Abstract
Information and material biological legacies that persist after catastrophic forest disturbance collectively constitute the ecological memory of the system and may strongly influence future stand development. Catastrophic disturbances often result in an influx of coarse woody debris (CWD), and this material legacy may provide beneficial microsites that affect successional and structural developmental pathways. We examined how microenvironmental characteristics influence the regeneration of woody plants in a subtropical woodland that experienced a large influx of CWD from a catastrophic wind disturbance. Specifically, we asked (1) what microenvironmental factors best explain woody plant density, richness, and height in the regeneration layer and (2) does woody plant density, richness, and height benefit from the large influx of CWD to a degree that competition dynamics and succession may be modified? Data were collected in a Pinus palustris woodland that had experienced an EF3 tornado and was subjected to a four-year prescribed fire rotation. We documented live woody plants <5 cm diameter at breast height, soil, and site characteristics and tested for differences in seedling and sapling density, species richness, and height in relation to CWD proximity. We used a random forest machine learning algorithm to examine the influence of microenvironmental conditions on the characteristics of woody plants in the regeneration layer. Woody plant density and species richness were not significantly different by proximity to CWD, but plants near CWD were slightly taller than plants away from CWD. The best predictors of woody plant density, richness, and height were abiotic site characteristics including slope gradient and azimuth, organic matter depth and weight, and soil water content. Results indicated that the regeneration of woody plants in this P. palustris woodland was not strongly influenced by the influx of CWD, but by other biological legacies such as existing root networks and soil characteristics. Our study highlights the need to consider ecological memory in forest management decision-making after catastrophic disturbance. Information and material legacies shape recovery patterns, but, depending on the system, some legacies will be more influential on successional and developmental pathways than others.
1. Introduction
Disturbances are important drivers of forest developmental and successional patterns. Forest canopy disturbances are regularly classified along a gradient based on their spatial extent, magnitude, and frequency. Along this gradient, catastrophic disturbances are among the most infrequent but the most severe, often removing most or all overstory vegetation [1,2,3]. In early forest disturbance ecology theory, catastrophic disturbances were considered to homogenize ecosystems and eliminate nearly all pre-disturbance biotic and abiotic stand conditions [4,5,6,7]. As our understanding of forest disturbance ecology has evolved, it is now understood that even after catastrophic disturbance, information and material biological legacies, components of ecological memory, persist [8]. These biological legacies of species adaptation and materials support recovery toward pre-disturbance conditions and enhance ecological resilience [9]. Rather than great homogenizing events that result in the elimination of pre-disturbance biophysical stand elements, catastrophic disturbances are now thought of as editors that retain, eliminate, and alter stand conditions [10,11]. Catastrophic disturbances may result in relatively complex, heterogeneous patterns of surviving organisms and residual structures (biological legacies), and these legacies may influence the succession and development of the regenerating stand [12,13]. For example, seed banks, spores, and fungal hyphae that persist post-disturbance can influence initial stand successional and developmental patterns [10]. Additionally, structural remnants such as snags or logs (i.e., coarse woody debris (CWD)) are also biological legacies that can strongly impact forest ecosystem structure, function, and composition [11,14]. The intrastand spatial patterns of biological legacies, such as undisturbed neighborhoods within a stand or patches of high CWD loading, may also modify ecosystem resilience [2,10]. Therefore, the presence and spatial distribution of biological legacies are important to consider in post-disturbance management and recovery.
Catastrophic wind disturbances largely eliminate the forest canopy, which reduces overhead competition and increases resource availability in the regeneration layer [15]. The post-disturbance regeneration of woody plants is influenced by microsite characteristics of the forest floor, the persistence of resting buds and seedbanks, the survival of seedlings, saplings, and small trees, and the arrival of new propagules [1]. Regeneration for many woody plant species requires an adequate seed supply and suitable substrate and microenvironmental conditions (e.g., bare mineral soil and abundant moisture) [16]. Thus, catastrophic disturbances will often lead to the recruitment of new individuals [17,18]. Additionally, catastrophic disturbances often result in an influx of CWD to the system. In general, CWD can aid in the establishment of some woody plant species by providing a substrate with high moisture and nutrient content [19,20], less interspecific competition [21], and a reduced risk of pathogenic soil fungi [22]. CWD can also provide shelter to seedlings and saplings from wind, animal browsing, and harsh environmental conditions [15,23]. The volume of CWD input following a wind disturbance is positively related to windthrow severity [24]. Characteristics of catastrophic wind disturbances result in the addition of CWD in a range of decay classes and amounts, and these attributes influence the ecosystem functions of the CWD [25]. An influx of CWD after catastrophic disturbance may differentially impact woody plant species, thereby altering successional and developmental pathways [26,27,28].
The Pinus palustris ecosystem is highly diverse, and among the most endangered forest types in the United States because of land-use changes and fire suppression [29,30,31]. Studies that have focused on the possible influence of biological legacies, such as CWD, on succession and stand development have predominately been conducted in boreal forests. The influence of CWD in biodiverse subtropical ecosystems that have relatively low woody debris volume, are frequently burned, and where moisture is not as limited, such as the Pinus palustris ecosystem, have received relatively little attention [32,33,34]. Furthermore, a notable lack of research exists on the influence of microenvironmental variability on woody plants following catastrophic tornado disturbance. Tornado frequency is anticipated to increase in the southeastern United States in the future [35]. Therefore, more work is needed to understand the drivers of intrastand patterns of forest recovery. This research has implications for the management and recovery of catastrophically disturbed forest ecosystems in the context of altered disturbance regimes.
Variability in the presence, abundance, size, or spatial arrangement of biological legacies following catastrophic disturbances can make developmental and successional trajectories difficult to predict [13]. The overarching goal of this study was to examine how variability in microenvironmental conditions influenced woody plant regeneration patterns following a catastrophic wind disturbance in a subtropical woodland. Specifically, this study sought to model the microenvironmental drivers of woody plant communities to help inform post-disturbance management. Additionally, the results supplement our understanding of the influence of biological legacies on the recovery of Pinus woodlands following natural catastrophic disturbance. The goals of this study were met by answering the following questions: (1) Which microenvironmental factors best explain woody plant density, richness, and height in the regeneration layer? and (2) Does woody plant density, richness, and height benefit from the large influx of CWD to a degree that competition dynamics and succession may be modified? Localized variability of site properties can influence woody plant regeneration [15,36,37]. Therefore, it was anticipated that (1) some soil characteristics, such as bulk density, soil water content, or organic matter depth, would influence woody plant regeneration patterns. CWD may alter microenvironmental conditions and, as a result, facilitate the establishment and growth of a variety of plant species in a range of forest types by providing shade, reducing temperature, and increasing humidity through decreased evapotranspiration [15,38]. Therefore, it was hypothesized that (2) altered microenvironmental conditions as a function of CWD input across the study site would influence patterns in density, richness, and height of the regeneration layer [25,39,40].
2. Materials and Methods
2.1. Study Site
This study was conducted in the Oakmulgee Ranger District of the Talladega National Forest in central Alabama, USA. The Oakmulgee Ranger District is in the Fall Line Hills [41]. The Fall Line Hills comprise sedimentary rock belts that serve as a transition zone for the Coastal Plain and Appalachian Highlands [42]. Soils are mainly derived from the Maubila series, which is described as deep and moderately well drained with a subangular blocky structure [43]. Specifically, data were collected over an area with two variations of the Maubila series, the Maubila flaggy loam and the Maubila–Smithdale complex [43] (Figure 1). The two soil delineations both have ca. 14 cm of available water storage, and the minimum water table depth for both is 23 cm [43].
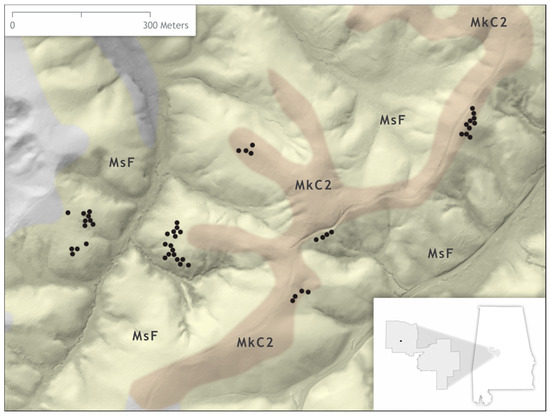
Figure 1.
Map depicting the coarse woody debris (CWD) sampled on the Oakmulgee Ranger District of the Talladega National Forest, Alabama, USA expressed as black dots, and the soil map units of the study site. The MsF soil map unit represents the Maubila–Smithdale complex and MkC2 indicates areas of Maubila flaggy loam [43].
The regional climate is humid mesothermal, characterized by year-round rainfall; long, hot summers; and brief, mild winters [44]. The 30 year normal annual precipitation and temperature are 1376 mm and 17 °C, respectively [45]. February has the highest precipitation at 139 mm, and October has the lowest at 87 mm [45]. The coldest month is January, with a mean temperature of 7 °C, and the warmest month is July, with a mean temperature of 27 °C [45]. Since 2000, severe droughts have occurred in the study area every 5–7 years, and the last severe drought was in 2017 [46].
The study was conducted in the central Pinus palustris belt of Alabama [47], and the plant communities liken to the Quercus–Pinus forest region [48]. Pinus palustris historically dominated the forest canopy of the study area; however, Pinus taeda, Pinus echinata, and various hardwood species can reach subcanopy and canopy positions [49,50]. The ecosystem supports a diverse herbaceous layer and an open mid-story [51,52]. Fire is an important disturbance in the advancement of Pinus palustris systems, maintaining the characteristically diverse understory and preventing succession to hardwood dominance [53]. Additionally, Pinus palustris woodlands require frequent low-intensity surface fires to decrease interspecific competition and expose bare mineral soil for seed germination [54].
Tornadoes occur frequently in this region [47,55]. The National Climatic Data Center has recorded 27 tornado events in the county since 1950, and three of these events were of EF3 or greater magnitude [56]. On 27 April 2011 an EF3 long-tracked wedge tornado, one of the 362 confirmed tornadoes of the 2011 super outbreak, moved through the Oakmulgee Ranger District. In fact, this was one of three tornadoes to impact the forest during the 2011 super outbreak. The EF3 tornado had maximum wind speeds of 233 kph and a maximum path width of 1609 m [57]. The tornado created a distinct edge between the undisturbed forest and the swath of catastrophically disturbed forest, and reduced basal area from 22 m2 ha−1 in the undisturbed forest to 1 m2 ha−1 in the tornado swath [58].
To promote Pinus palustris recovery, low-intensity prescribed fires are set every 2–4 years in P. palustris-dominated stands across the Oakmulgee Ranger District. Since the tornado in 2011, two operational, prescribed fires have been conducted, in April 2014 and April 2018. We sampled in the second growing season after the 2018 prescribed fire.
2.2. Field Methods
In the summer of 2019, 350 quadrats (300 quadrats contiguous to CWD and 50 quadrats located away from CWD) were established in stands that had experienced catastrophic wind disturbance and were not salvage-harvested on the Oakmulgee Ranger District. CWD sampling only occurred in a single compartment on the district to ensure consistency in prescribed fire, land-use, and management history. Transects, beginning from random starting points, were used to locate and sample CWD that met the following criteria: >3 m in length and ≥10 cm in diameter.
Our sampling scheme, modified from Chećko et al. [59], consisted of a group of three 1 m2 quadrats placed equidistant on the ground on both sides of the CWD to analyze microsite characteristics. Thus, six quadrats were established around one CWD fragment with three quadrats on one side of the log and the other three quadrats on the other side of the CWD (Figure 2). The quadrats were uniformly established by placing Quadrat 1 on the downslope side of the CWD, and then moving clockwise to assign the five remaining quadrats. A seventh quadrat was established at least 2 m away from the focal log and any other CWD.
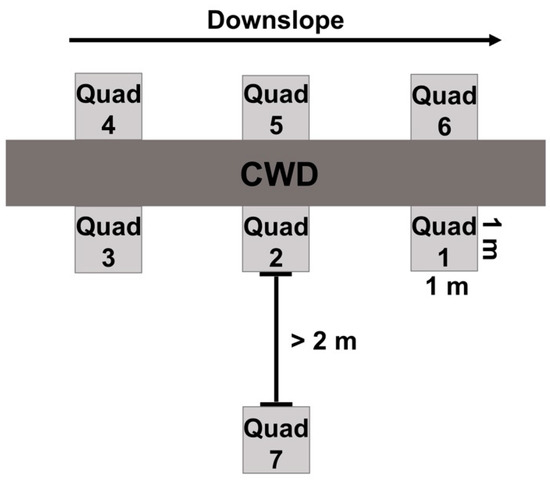
Figure 2.
Representation of 1 m2 quadrat establishment around each piece of coarse woody debris (CWD). Quadrat 7 was established colinear to Quadrat 2, and at least 2 m away from the focal piece of CWD and any other CWD.
For every quadrat contiguous to CWD, multiple attributes of the focal CWD were recorded: tree species to the lowest taxon possible, diameter, length, presence of rot, decay class, and azimuth. Azimuth of CWD was always recorded from upslope to downslope. The area where data were collected was under a uniform prescribed fire plan; therefore, all of the CWD analyzed was subjected to prescribed fire. However, if the CWD had visual signs of combustion where a quadrat was established, it was scored as charred [60,61]. CWD was also categorized as being either on the ground (i.e., resting on the ground) or elevated (i.e., the quadrat was placed adjacent to a section of the log that was suspended). Decay class was determined using a seven decay class system adopted from Ulyshen et al. [62] that accounted for the relatively rapid decomposition of sapwood, but common decomposition of heartwood that occurs in pines of the southeastern USA [63].
For every 1 m2 quadrat, including the seventh quadrat, the presence, species, and height of all live woody plants <5 cm diameter at breast height (dbh) were noted. Individual plants were classified as either seedlings (<1 m in height) or saplings (≥1 m in height, <5 cm dbh). Organic matter depth was documented to 0.25 cm using a ruler. We took four organic matter depth measurements equidistant from a transect perpendicular to the log through each quadrat (i.e., four readings quadrat−1). This method allowed for the calculation of an organic matter depth average for each quadrat and the understanding of how organic matter depth changed with distance from CWD. Organic matter measurements were not taken in portions of a quadrat where pieces of bark had recently sloughed off during CWD decay. The slope aspect and topographic position (linear, concave, or convex) for each quadrat were recorded. To examine how the amount of CWD influenced plant and soil traits, quadrats located alongside CWD were classified as ungrouped, grouped, or extremely grouped by adapting the methods of Pinno & Das Gupta [37] and Chećko et al. [59]. A quadrat was considered grouped if CWD or a stump was less than 1 m away from the quadrat edge, and classified as extremely grouped if the additional CWD was within the quadrat boundaries.
Soil samples were taken from the top of the soil profile using a 4 cm by 7.5 cm steel pipe with a volume of 94.25 cm3, following the methods of LaFevor [64]. Samples were collected at the center of each of the seven quadrats. Rocks occasionally impeded soil sample collection and forced the collection to occur closer to a quadrat edge than the true center. Samples were collected by removing all organic matter from the top of the soil, hammering down the steel pipe, removing the sample with a trowel, and then placing the sample in a labeled, double-sealed plastic bag. After collection, the samples were transferred to a separate bag for protection and moved to the laboratory to be weighed, dried at 105 °C for 24 h, and then reweighed [65,66]. The samples were then dry-sieved (2 mm) to remove all organic matter and rocks, which were subsequently weighed.
2.3. Laboratory and Analytical Methods
We studied a natural wind disturbance to better understand how catastrophic disturbance and a large influx of CWD might influence woody plant regeneration patterns; therefore, experimental replicability was not possible. The ecological knowledge to be gained from studying natural disturbance events often outweighs potential issues of pseudoreplication; however, we sought to mitigate possible issues of pseudoreplication through the statistical treatment of our data [67]. Seedling and sapling density and richness were determined for each quadrat. Density was calculated as the sum of woody seedlings and saplings in each quadrat, and richness was calculated as the number of woody seedling and sapling species. Organic matter depth was averaged by quadrat. Relative density was calculated for woody plant seedlings and saplings. Soil water content was calculated by subtracting the soil wet weight by the dry weight and dividing the difference by the dry weight.
For this study, significance was determined at p < 0.05. To assess significant differences in microsite characteristics, the six quadrats adjacent to a piece of CWD were classified as “near” and the seventh quadrat at least 2 m away from CWD was classified as “away.” We calculated the average density, richness, and height of the six near quadrats to compare to the paired away quadrat of each log (n = 50). Analysis was further stratified by seedlings and saplings. Shapiro–Wilks tests for normality and Levene’s tests for equality of variance were used to ensure assumptions of parametric tests were met. Data were not normally distributed and variances were unequal. Therefore, a paired-samples Wilcoxon signed-rank test was used to statistically analyze the differences in woody plant density, richness, and height near CWD versus away [68].
To determine the influence of microsite characteristics on woody plants, we applied a random forest (RF) machine learning algorithm. RF is a complex, non-parametric tool derived from classification and regression trees and is capable of modeling complex interactions between variables with high accuracy [69,70]. The algorithm combines many trees, each generated by bootstrap samples, and retains samples for internal cross-validation (out of bag estimates, OOB). The final model output is the mean result of all trees. Model outputs include a variable importance measure, which ranks each predictor variable by mean increase in error (% IncMSE) when observed values of predictor variables are permuted through the model [69]. These values can be used to infer which predictor variable has the greatest influence on the independent variable within the model. RF was selected because of the large number of recorded CWD and soil characteristics (n = 18), and the hypothesized complex interactions between microsite characteristics and woody plant communities. We fit three independent RF models to predict woody plant density, richness, and height, incorporating 500 trees into each model. Density and richness were modeled for each individual quadrat (n = 300) and height was modeled for each individual woody plant (n = 2609). We did not include the 50 quadrats that were located away from CWD in the RF models because they were not associated with any CWD characteristics. For each model, we removed predictor variables that had negative % IncMSE values, as these variables had no influence on the woody plant characteristics, and thus reduced the predictive ability and increased the error of the models. To account for potential multicollinearity of predictor variables, we tested for correlation among all predictor variables and removed variables that were strongly correlated to one or more predictor variables. All models were cross-validated with a randomly selected training and testing subset of the data. RF was conducted using the package “randomForest” in R version 1.2.5001 [71].
3. Results
3.1. Relative Density of Woody Plant Species
Vaccinium arboreum and Rhus copallinum were the most common woody plant seedling species (Table 1). Rhus copallinum, Vaccinium arboreum, and Pinus palustris seedlings exhibited the greatest relative frequencies, with Rhus copallinum seedlings appearing in 54% of quadrats, Vaccinium arboreum seedlings in 38%, and Pinus palustris seedlings in 33% of quadrats. A total of 15 woody plant species were reported only as seedlings, including Pinus palustris and five Quercus species. The three woody plant species with the highest densities in the sapling stage were Rhus copallinum, Oxydendrum arboreum, and Vaccinium arboreum (Table 2). Rhus copallinum saplings appeared in 25% of quadrats and Vaccinium arboreum saplings were found in 14% of quadrats. About 36% of woody plant species in the sapling stage were found in only one quadrat, including Asimina parviflora, Hamamelis virginiana, Liquidambar styraciflua, Pinus taeda, Quercus marilandica, Quercus stellata, Quercus velutina, and Rhus glabra.

Table 1.
Relative density of seedlings (live woody stems <1 m height) in a Pinus palustris woodland following catastrophic wind disturbance, ordered by relative density.

Table 2.
Relative density of saplings (live woody stems ≥1 m in height, <5 cm dbh) in a Pinus palustris woodland following catastrophic wind disturbance, ordered by relative density.
3.2. CWD Proximity and Woody Plant Density
Seedling density was not significantly different by proximity to CWD (p = 0.23, Figure 3a). The mean density of seedlings near CWD was 7.98 stems m−2 ± 0.59 (SE), and the mean density of woody plant seedlings away from CWD was 10.10 stems m−2 ± 1.38. The greatest seedling density was found in a plot located away from CWD, where there were 54 Vaccinium arboreum seedlings. There were 21 quadrats that had a seedling density ≥ 20 stems m−2, and there were 317 quadrats with a seedling density <20 stems m−2. There were 29 quadrats that were reported to have only one seedling, which included quadrats that were both near CWD and away from CWD. Sapling density was not significantly different by proximity to CWD (p = 0.17, Figure 3b). The mean density of saplings near CWD was 0.74 stems m−2 ± 0.14, and the mean density of saplings away from CWD was 0.5 stems m−2 ± 0.13. The greatest sapling density recorded in a quadrat was attributed to 19 Oxydendrum arboreum stems m−2, and the quadrat with the second highest density had 10 woody plant stems m−2. All other quadrats had woody plant sapling densities that were less than 10 stems m−2. Almost 45% of quadrats had a sapling density of 1 stem m−2.
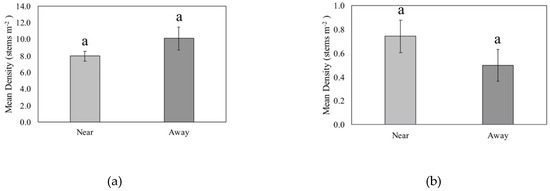
Figure 3.
Mean density of (a) seedlings (live woody stems <1 m height) and (b) saplings (live woody stems ≥1 m height, <5 cm dbh) of quadrats located near and away from coarse woody debris (CWD) in a Pinus palustris woodland following catastrophic disturbance. No significant differences (p < 0.05) were found.
3.3. Proximity to CWD and Woody Plant Richness
Seedling richness did not differ between quadrats near CWD and quadrats located away from CWD (p = 0.78, Figure 4a). The mean richness near CWD was 0.30 species m−2 ± 0.05 (SE), and the mean richness away from CWD was 0.34 species m−2 ± 0.07. One quadrat had a richness of 7 species m−2, and it was near CWD. Every other quadrat had a richness value of <7. Quadrats with only one species present comprised 25% of all quadrats with woody plant seedlings. Woody plant sapling richness was also not significantly different between quadrats near CWD and quadrats away (p = 0.80, Figure 4b). Sapling richness near CWD was 0.31 species m−2 ± 0.04 and sapling richness away from CWD was 0.32 species m−2 ± 0.08. Sapling richness was documented to be only either 2 species m−2 or 1 species m−2. Quadrats with a sapling richness of 2 species m−2 comprised 13% of quadrats, and quadrats with a richness of 1 species m−2 comprised 87% of quadrats with saplings present.
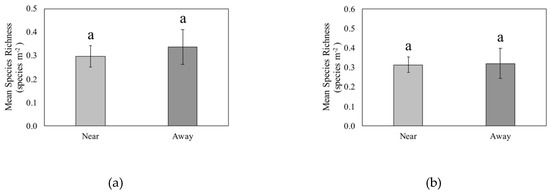
Figure 4.
Mean species richness of (a) seedlings (live woody stems <1 m height) and (b) saplings (live woody stems ≥1 m height, <5 cm dbh) by quadrats located near and away from coarse woody debris (CWD) in a Pinus palustris woodland following catastrophic disturbance. No significant differences (p < 0.05) were found.
3.4. Proximity to CWD and Woody Plant Height
Seedling height was significantly greater in quadrats that were near CWD than in quadrats that were away from CWD (p < 0.001, Figure 5a). The mean height for seedlings near CWD was 33.57 cm ± 0.51 (SE), and seedlings away from CWD had a mean height of 25.20 cm ± 0.93. Seedling heights ranged from 1 cm to 99 cm. Similarly, sapling height was significantly greater near CWD than away (p < 0.001, Figure 5b). Saplings near CWD had a mean height of 131.77 cm ± 2.13 and saplings away from CWD had a mean height of 120.92 cm ± 4.65.
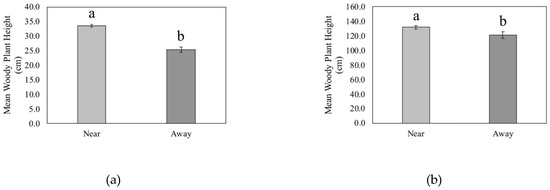
Figure 5.
Mean height of (a) seedlings (live woody stems <1 m height) and (b) saplings (live woody stems ≥1 m height, <5 cm dbh) of quadrats located near and away from coarse woody debris (CWD) in a Pinus palustris woodland following catastrophic disturbance. Different letters indicate significant differences at p < 0.05.
3.5. Influence of Microenvironmental Conditions on Woody Plant Characteristics
The RF models explained 17.2% of the variance in woody plant density, 18.4% of the variance in woody plant richness, and 21.7% of the variance in woody plant height. The woody plant density model included 16 of the 18 predictor variables. Topographic position and CWD species had negative %IncMSE values, and were thus removed from the final density model. Slope, decay class, and slope azimuth had the greatest impact on woody plant density (Figure 6). Of the soil characteristics, wet bulk density and soil water content were the most important predictors of density (Figure 6). The woody plant richness model included 12 of the 18 predictor variables. Topographic position, CWD species, presence of char, presence of rot, the quadrat slope position relative to CWD, and proximity to additional CWD were excluded from the final model. Wet bulk density, organic matter weight, and CWD azimuth were the most important predictors of woody plant richness (Figure 7). The woody plant height model had the highest predictive capability and explained the most variance of the three models (21.7%). Each of the 17 predictor variables were included in the height model. The three most influential variables on woody plant height were soil-related. These included organic matter depth, soil water content, and organic matter weight (Figure 8). Of the three models, the microsite characteristics that consistently had the greatest impact on woody plants (based on %IncMSE) were slope, organic matter depth and weight, and wet bulk density.
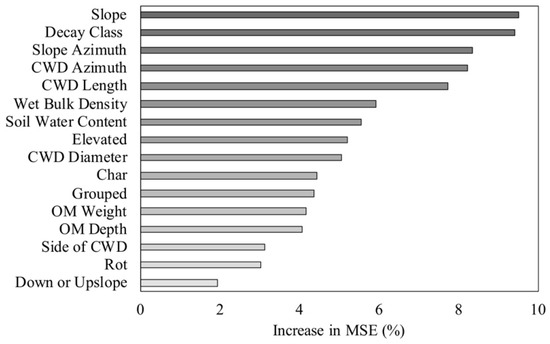
Figure 6.
Importance ranking for the influence of coarse woody debris (CWD) attributes and site characteristics on woody plant density for stems <5 cm dbh in a Pinus palustris woodland after catastrophic disturbance. The increase in mean square error (MSE) indicates the increase in model error when the given variable was randomly permuted.
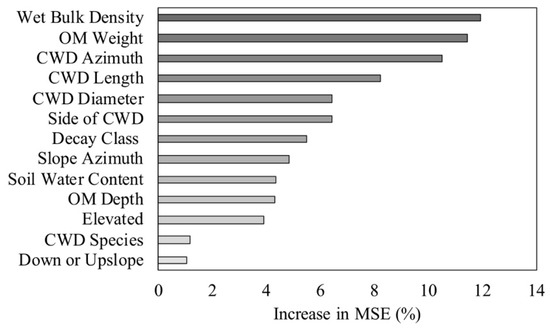
Figure 7.
Importance ranking for the influence of coarse woody debris (CWD) attributes and site characteristics on woody plant richness for stems <5 cm dbh in a Pinus palustris woodland after catastrophic disturbance. The increase in mean square error (MSE) indicates the increase in model error when the given variable was randomly permuted.
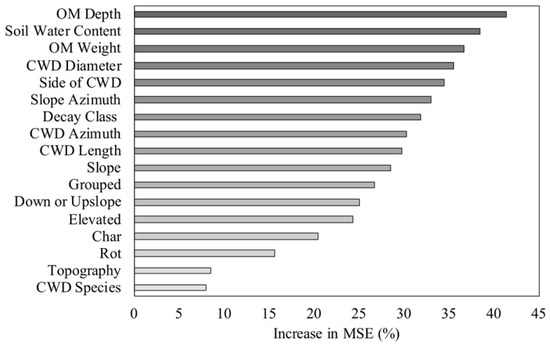
Figure 8.
Importance ranking for the influence of coarse woody debris (CWD) attributes and site characteristics on woody plant height for stems <5 cm dbh in a Pinus palustris woodland after catastrophic disturbance. The increase in mean square error (MSE) indicates the increase in model error when the given variable was randomly permuted.
4. Discussion
4.1. Relative Density of Woody Plant Species
Our findings indicate that Pinus palustris was primarily in the grass stage during the second growing season after the prescribed fire (2350 P. palustris seedlings ha−1 [72]), and the Pinus taeda individuals were mostly new seeds that had germinated since the April 2018 burn (the second prescribed fire since the catastrophic wind disturbance). Although not recorded in our study, the lack of Pinus palustris saplings was also attributed in part to the recruitment of individuals to the tree size class (>5 cm dbh) eight years post-disturbance. Working in the same stands, Kleinman et al. [72] noted increased post-fire Pinus palustris sapling density in catastrophically disturbed sites, but found that Pinus palustris individuals remained outnumbered by hardwood species (56 P. palustris saplings ha−1 vs. 776 hardwood saplings with canopy potential ha−1). However, these sites did exhibit fire-mediated sapling mortality, which indicated that hardwood and herbaceous species produced surface fuels that aided in the spread of fire in addition to the limited Pinus litter [72]. We suggest that continued use of frequent prescribed fire will allow Pinus palustris individuals to recruit into larger size classes while hardwood individuals will continue to be top-killed and thus restricted to smaller size classes. Therefore, we suggest that fire is more important than microenvironmental conditions for the recovery of these Pinus palustris stands. Many individuals on this study site were hardwood sprouts (Table 1 and Table 2). Hardwood regeneration can be prolific after prescribed fire because of sprouting [73,74,75]. The high frequency of hardwood sprouts after fire is typically attributed to hardwood rootstocks that can survive fire and facilitate hardwood sprouting [76,77]. High densities of hardwoods have been documented in the second growing season after fire in mixed Quercus–Pinus systems [73,78] and indeed have been recorded in these Pinus palustris woodlands [79].
For both seedlings and saplings, Vaccinium arboreum and Rhus copallinum were among the woody plant species with the greatest density. Vaccinium arboreum seedlings can form large colonies from root sprouts [80]. Similarly, the underground rhizomes of Rhus copallinum allow it to resprout readily after fire, and past research has illustrated that the species can dominate extremely disturbed sites [81]. The prevalence of Vaccinium arboreum and Rhus copallinum on this site was also shown in past studies [82], and is likely a result of the recent catastrophic disturbance that eliminated most all overstory vegetation and subsequent prescribed fire. The high abundance of sprouters recorded in this study may also indicate that microenvironmental variability influences non-sprouters more drastically than sprouters, although this was not directly measured.
4.2. Proximity to CWD and Woody Plant Density, Richness, and Height
Seedling and sapling density were not significantly different near CWD and away. The difference in seedling density was about two quadrat−1, and the difference in sapling density was less than one sapling quadrat−1. Only one Pinus individual was classified as a sapling; therefore, the majority of saplings were hardwoods and likely originated from rootstocks that produced shoots that had grown to the sapling layer since the prescribed fire of the prior growing season. We hypothesized that sapling density would be greater near CWD because CWD might provide some protection to root and bud systems found belowground from prescribed fire char (60% CWD was uncharred and 40% was charred). CWD has been found to protect mycorrhizal fungi and fungal hyphae from fire, and to aid in the re-establishment of these species after fire [83]. The protection of root and bud systems could have allowed individuals to resprout more readily after fire, and protect them from future prescribed fire by helping individuals to reach greater height classes more quickly.
No significant difference was found in seedling and sapling richness by proximity to CWD. This pattern may have been because the majority of individuals in our study site originated from root stocks and not from seed. CWD can be beneficial for many small-seeded species, because the seeds can be trapped in the ridges of CWD [59,84]. However, the majority of woody plants in this study originated from an underground root system; therefore, the CWD had little influence on hardwood species density. The catastrophic wind disturbance removed nearly all mature overstory trees, and thus reduced seed availability for natural regeneration of the overstory. The residual trees that survived the tornado were primarily Pinus palustris, but residual canopy basal area was low (1 m2 ha−1) [72]. Therefore, it was likely that most of the hardwood individuals recorded were biological legacies from the pre-disturbed stand, and not a function of intrastand microenvironmental variability. Species richness can potentially be increased by CWD providing more microtopographic diversity [85,86,87]. However, past research has found that CWD may not create sufficient microtopographic diversity to influence species richness patterns [88]. Other studies found that CWD was not a significant influence on vascular plant richness [89,90], and our results corroborated these findings.
Seedling and sapling height were found to be significantly greater in quadrats near CWD than in quadrats located away from CWD. However, the pattern may not be ecologically meaningful because the observed difference in seedling height was only 8 cm, and for sapling height the difference was 11 cm (i.e., seedlings near CWD were ca. 32% taller than seedlings away from CWD and saplings near CWD were ca. 9% taller than saplings away from CWD). Past CWD studies conducted in various forest types have found that CWD presence can improve individual growth rates [84,91,92]. CWD has been shown to provide a windbreak for seedlings located one to seven barrier heights on the leeward side of the log and one barrier height on the windward side, and this protection from wind can contribute to greater plant growth [93]. Additionally, a sheltering object can alter temperature and humidity values over a relatively short distance when compared to wind velocity [94]. The presence of CWD can result in a more favorable microclimate for seedling growth by reducing temperature changes and increasing humidity through decreased evapotranspiration [15]. Regardless of the height differences by CWD proximity, we posit that taller, fire-sensitive woody plants near CWD are as likely to be top-killed by a subsequent prescribed fire as woody plants away from CWD.
4.3. Influence of Microenvironmental Conditions on Woody Plant Characteristics
The RF results indicated that seedling and sapling patterns were influenced by CWD, along with other site variables. Slope, slope azimuth, and decay class were the best predictors of woody plant density. Wet bulk density, organic matter weight, and CWD azimuth were the best predictors of woody plant richness. Organic matter depth, soil water content, and organic matter weight were the best predictors of woody plant height. Previous CWD studies have found that site properties can be more dominant factors in determining plant community characteristics, and that CWD may be a minor contributing factor [36,37]. However, in each of the three models, at least two of the top five most influential predictor variables were CWD attributes. Therefore, it could be argued that CWD is nearly as influential as other microsite properties on woody plant density, richness, and height in the regeneration layer.
The Pinus palustris ecosystem in the southeastern USA typically supports some of the lowest volumes of CWD when compared to other forest types and regions [62,95]. Little has been reported on the role of CWD in Pinus palustris woodlands specifically; however, one herpetofaunal study was completed on upland Pinus taeda stands in the southeastern US coastal plain. Davis et al. [96] found that the amphibians and reptiles of the study site were not reliant on CWD for habitat. They concluded that the herpetofauna must have adapted to the characteristically low CWD volumes that are typical of the Pinus palustris ecosystem that historically dominated the study area [96,97]. The results from our study may indicate a similar relationship between CWD and woody plants. It could be that ecosystems in the southeastern US, which do not have historically high volumes of CWD, support woody plant species and animal populations that have largely adapted to survive without CWD. Additionally, our results indicate that CWD inputs may not differentially benefit woody plant species that occur in this system.
The variance explained by the RF models for woody plant density, richness, and height were relatively low—not higher than 22%. This low value may indicate that we did not measure particular variables that have a more significant influence on woody plant regeneration. Undocumented soil characteristics may have a greater influence on woody plant assemblages than soil water content or wet bulk density. For example, soil hardness and water repellency can play important roles in woody plant regeneration [15]. Additionally, CWD may have modified soil nutrients that influence woody plant density, richness, or height. Carbon and nitrogen soil content, soil base saturation, and pH may increase with an influx of CWD [25,98,99].
5. Conclusions
We analyzed woody plant regeneration patterns in a Pinus palustris woodland that experienced a catastrophic wind disturbance. The stands were under a four-year prescribed fire rotation and had been burned twice since the catastrophic event. In this analysis, the best predictors of woody plant density, richness, and height were site characteristics—slope, wet bulk density, and soil water content—and CWD attributes—decay class, CWD azimuth and CWD diameter. Results also indicated that proximity to CWD alone may not drive woody plant regeneration patterns. We suggest that woodland ecosystems located in the humid subtropics, that experience frequent prescribed fire, and that have historically low volumes of CWD support woody plant species that may not be dependent on and do not differentially benefit from CWD. A large influx of CWD has been found to alter carbon and nitrogen storage [100], fungi [101], animals [25], soil fertility [36], and soil erosion [102], and these variables can all influence competitive dynamics and favor some woody plant species over others. However, in the Pinus palustris woodland studied here, pre-disturbance biotic and abiotic conditions seem to be more impactful on woody plant communities than the CWD pulse, such as existing root networks in hardwood species and soil characteristics.
Salvage logging is a common forest operation that occurs after catastrophic disturbances to mitigate the loss of profits from downed or damaged trees and to reduce the risk of insect outbreak and fire [103]. Salvage logging is a contested practice because the ecological consequences are not yet fully understood [104,105,106]. Past research at this study site found that salvage logging did not negatively influence woody plant diversity; however, it did negatively impact ground flora diversity and macrofungal species richness and fruiting abundance [79,101]. Additionally, research conducted in other Pinus spp. stands found that salvage logging can benefit open-habitat avian species [107,108]. The results from our study indicate that retention of CWD after catastrophic disturbance may not alter the regeneration potential of woody plants in Pinus palustris woodlands. The decision to salvage harvest should be based on possible impacts on organisms of management concern. Here, we only documented seedlings and saplings, and we acknowledge that findings are contingent upon the response variables studied [109] and that salvage harvesting influences biophysical conditions other than just CWD loading.
Climate change and other stressors are modifying forest disturbance regimes, and the need to understand disturbance processes and patterns and their influence on forest composition, structure, and function is essential for forest management [110,111]. Information and material legacies from pre-disturbance conditions are critical in driving successional and developmental pathways in developing stands [9,112]. CWD is considered a material biological legacy that can strongly impact forest ecosystem functioning [11,14]. However, in our study we did not find that an influx of CWD significantly influenced woody plant seedling and sapling density or richness in a Pinus palustris woodland. This finding indicates that CWD as a component of ecological memory did not strengthen or weaken resilience in our study system. With a predicted increase in tornado frequency in the southeastern US [35], it is essential to understand how catastrophic disturbances and their legacies will influence resilience of these systems.
Author Contributions
A.T.L. and J.L.H. conceived and designed the study; A.T.L. and J.D.G. completed field data collection and data analysis; D.J.K. assisted random forest analysis and provided manuscript revisions; A.T.L., J.D.G., and J.L.H. wrote the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
We thank the USDA Forest Service, Talladega National Forest, Oakmulgee Ranger District for logistical support, the University of Alabama Cartographic Research Lab for assistance with map preparation, and the anonymous reviewers for helpful comments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Oliver, C.D.; Larson, B.C. Forest Stand Dynamics; John Wiley and Sons: Hoboken, NJ, USA, 1996. [Google Scholar]
- Foster, D.R.; Knight, D.H.; Franklin, J.F. Landscape patterns and legacies resulting from large, infrequent forest disturbances. Ecosystems 1998, 1, 497–510. [Google Scholar] [CrossRef]
- Hart, J.L.; Kleinman, J.S. What are intermediate-severity forest disturbances and why are they important? Forests 2018, 9, 579. [Google Scholar] [CrossRef]
- Clements, F.E. Plant Succession: An Analysis of the Development of Vegetation; No. 242; Carnegie Institution of Washington Publication: Washington, DC, USA, 1916; pp. 1–512. [Google Scholar] [CrossRef]
- Connell, J.H.; Slatyer, R.O. Mechanisms of succession in natural communities and their role in community stability and organization. Am. Nat. 1977, 111, 1119–1144. [Google Scholar] [CrossRef]
- Loehle, C. Strategy space and the disturbance spectrum: A life-history model for tree species coexistence. Am. Nat. 2000, 156, 14–33. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, R.J.; Bush, M.B.; Richards, K.J.E.M. Plant recolonization and vegetation succession on the Krakatau Islands, Indonesia. Ecol. Monogr. 1989, 59, 59–123. [Google Scholar] [CrossRef]
- Webster, C.R.; Dickinson, Y.L.; Burton, J.I.; Frelich, L.E.; Jenkins, M.A.; Kern, C.C.; Raymond, P.; Saunders, M.R.; Walters, M.B.; Willis, J.L. Promoting and maintaining diversity in contemporary hardwood forests: Confronting contemporary drivers of change and the loss of ecological memory. For. Ecol. Manag. 2018, 421, 98–108. [Google Scholar] [CrossRef]
- Johnstone, J.F.; Allen, C.D.; Franklin, J.F.; Frelich, L.E.; Harvey, B.J.; Higuera, P.E.; Mack, M.C.; Meentemeyer, R.K.; Metz, M.R.; Perry, G.L.; et al. Changing disturbance regimes, ecological memory, and forest resilience. Front. Ecol. Environ. 2016, 14, 369–378. [Google Scholar] [CrossRef]
- Franklin, J.F.; Lindenmayer, D.; MacMahon, J.A.; McKee, A.; Magnuson, J.; Perry, D.A.; Waide, R.; Foster, D. Threads of continuity. Conserv. Biol. 2000, 1, 8–16. [Google Scholar] [CrossRef]
- Franklin, J.F.; Spies, T.A.; Van Pelt, R.; Carey, A.B.; Thornburgh, D.A.; Berg, D.R.; Lindenmayer, D.B.; Harmon, M.E.; Keeton, W.S.; Shaw, D.C.; et al. Disturbances and structural development of natural forest ecosystems with silvicultural implications, using Douglas-fir forests as an example. For. Ecol. Manag. 2002, 155, 399–423. [Google Scholar] [CrossRef]
- Swanson, M.E.; Franklin, J.F.; Beschta, R.L.; Crisafulli, C.M.; DellaSala, D.A.; Hutto, R.L.; Lindenmayer, D.B.; Swanson, F.J. The forgotten stage of forest succession: Early-successional ecosystems on forest sites. Front. Ecol. Environ. 2011, 9, 117–125. [Google Scholar] [CrossRef]
- Turner, M.G.; Baker, W.L.; Peterson, C.J.; Peet, R.K. Factors influencing succession: Lessons from large, infrequent natural disturbances. Ecosystems 1998, 1, 511–523. [Google Scholar] [CrossRef]
- Franklin, J.F.; MacMahon, J.A. Messages from a mountain. Science 2000, 288, 1183–1184. [Google Scholar] [CrossRef]
- Bailey, T.G.; Davidson, N.J.; Close, D.C. Understanding the regeneration niche: Microsite attributes and recruitment of eucalypts in dry forests. For. Ecol. Manag. 2012, 269, 229–238. [Google Scholar] [CrossRef]
- Baskin, C.; Baskin, J.M. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Henry, J.D.; Swan, J.M.A. Reconstructing Forest History from Live and Dead Plant Material—An Approach to the Study of Forest Succession in Southwest New Hampshire. Ecology 1974, 55, 772–783. [Google Scholar] [CrossRef]
- Lorimer, C.G. Age structure and disturbance history of a southern Appalachian virgin forest. Ecology 1980, 61, 1169–1184. [Google Scholar] [CrossRef]
- Zielonka, T. When does dead wood turn into a substrate for spruce replacement? J. Veg. Sci. 2006, 17, 739–746. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Pulliam, W.M.; Lodge, D.J.; Quinones-Orfila, V.; Fetcher, N.; Guzman-Grajales, S.; Parrotta, J.A.; Asbury, C.E.; Walker, L.R.; Waide, R.B. Nitrogen immobilization by decomposing woody debris and the recovery of tropical wet forest from hurricane damage. Oikos 1995, 72, 314–322. [Google Scholar] [CrossRef]
- Harmon, M.E.; Franklin, J.F. Tree seedlings on logs in Picea-Tsuga forests of Oregon and Washington. Ecology 1989, 70, 48–59. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Miyamaoto, T. Racodium snow blight in Japan. For. Pathol. 2005, 35, 1–7. [Google Scholar] [CrossRef]
- Whyte, H.D.; Lusk, C.H. Woody debris in treefall gaps shelters palatable plant species from deer browsing, in an old-growth temperate forest. For. Ecol. Manag. 2019, 448, 198–207. [Google Scholar] [CrossRef]
- Waldron, K.; Ruel, J.C.; Gauthier, S. Forest structural attributes after windthrow and consequences of salvage logging. For. Ecol. Manag. 2013, 289, 28–37. [Google Scholar] [CrossRef]
- Harmon, M.E.; Franklin, J.F.; Swanson, F.J.; Sollins, P.; Gregory, S.V.; Lattin, J.D.; Anderson, N.H.; Cline, S.P.; Aumen, N.G.; Sedell, J.R.; et al. Ecology of coarse woody debris in temperate ecosystems. In Advances in Ecological Research; MacFadyn, A., Ford, E.D., Eds.; Academic Press: Orlando, FL, USA, 1986; Volume 15, pp. 133–302. [Google Scholar]
- Cornett, M.W.; Reich, P.B.; Puettmann, K.J.; Frelich, L.E. Seedbed and moisture availability determine safe sites for early Thuja occidentalis (Cupressaceae) regeneration. Am. J. Bot. 2000, 87, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Haskell, D.E.; Flaspohler, D.J.; Webster, C.R.; Meyer, M.W. Variation in soil temperature, moisture, and plant growth with the addition of downed woody material on lakeshore restoration sites. Restor. Ecol. 2012, 20, 113–121. [Google Scholar] [CrossRef]
- Stroheker, S.; Weiss, M.; Sieber, T.N.; Bugmann, H. Ecological Factors Influencing Norway Spruce Regeneration on Nurse Logs in a Subalpine Virgin Forest. Forests 2018, 9, 120. [Google Scholar] [CrossRef]
- Frost, C. History and future of the longleaf pine ecosystem. In The Longleaf Pine Ecosystem; Jose, S., Jokela, E.J., Miller, D.L., Eds.; Springer: New York, NY, USA, 2007; pp. 9–48. [Google Scholar] [CrossRef]
- Kirkman, L.K.; Coffey, K.L.; Mitchell, R.J.; Moser, E.B. Ground cover recovery patterns and life-history traits: Implications for restoration obstacles and opportunities in a species-rich savanna. J. Ecol. 2004, 92, 409–421. [Google Scholar] [CrossRef]
- Noss, R.F.; LaRoe, E.T.; Scott, J.M. Endangered Ecosystems of the United States: A Preliminary Assessment of Loss and Degradation; U.S. Department of the Interior, National Biological Service: Washington, DC, USA, 1995.
- Dirzo, R.; Raven, P.H. Global state of biodiversity and loss. Annu. Rev. Env. Resour. 2003, 28, 137–167. [Google Scholar] [CrossRef]
- Hansen, M.C.; Stehman, S.V.; Potapov, P.V.; Loveland, T.R.; Townshend, J.R.; De Fries, R.S.; Pittman, K.W.; Arunarwati, B.; Stolle, F.; Steininger, M.K.; et al. Humid tropical forest clearing from 2000 to 2005 quantified by using multitemporal and multiresolution remotely sensed data. Proc. Natl. Acad. Sci. USA 2008, 105, 9439–9444. [Google Scholar] [CrossRef]
- Seibold, S.; Bässler, C.; Brandl, R.; Gossner, M.M.; Thorn, S.; Ulyshen, M.D.; Müller, J. Experimental studies of dead-wood biodiversity—A review identifying global gaps in knowledge. Biol. Conserv. 2015, 191, 139–149. [Google Scholar] [CrossRef]
- Gensini, V.A.; Brooks, H.E. Spatial trends in United States tornado frequency. NPJ Clim. Atmos. Sci. 2018, 1, 1–5. [Google Scholar] [CrossRef]
- Brais, S.; Paré, D.; Lierman, C. Tree bole mineralization rates of four species of the Canadian eastern boreal forest: Implications for nutrient dynamics following stand- replacing disturbances. Can. J. For. Res. 2006, 36, 2331–2340. [Google Scholar] [CrossRef]
- Pinno, B.; Das Gupta, S. Coarse Woody Debris as a Land Reclamation Amendment at an Oil Sands Mining Operation in Boreal Alberta, Canada. Sustainability 2018, 10, 1640. [Google Scholar] [CrossRef]
- White, P.J.; McGill, B.J.; Lechowicz, M.J. Detecting changes in forest floor habitat after canopy disturbance. Ecol. Res. 2012, 27, 397–406. [Google Scholar] [CrossRef]
- Han, A.R.; Kim, H.J.; Jung, J.B.; Park, P.S. Seed germination and initial seedling survival of the subalpine tree species, Picea jezoensis, on different forest floor substrates under elevated temperature. For. Ecol. Manag. 2018, 429, 579–588. [Google Scholar] [CrossRef]
- Vítková, L.; Bače, R.; Kjučukov, P.; Svoboda, M. Deadwood management in Central European forests: Key considerations for practical implementation. For. Ecol. Manag. 2018, 429, 394–405. [Google Scholar] [CrossRef]
- Fenneman, N.M. Physiography of Eastern United States; McGraw-Hill: New York, NY, USA, 1938. [Google Scholar]
- Shankman, D.; Hart, J.L. The fall line: A physiographic forest-vegetation boundary. Geogr. Rev. 2007, 97, 502–519. [Google Scholar] [CrossRef]
- USDA NRCS (United States Department of Agriculture, Natural Resources Conservation Service). Web Soil Survey. Available online: https://websoilsurvey.nrcs.usda.gov/ (accessed on 23 April 2020).
- Thornthwaite, C.W. An approach toward a rational classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- PRISM Climate Group. Northwest Alliance for Computational Science and Engineering. Available online: http://www.prism.oregonstate.edu/ (accessed on 23 April 2020).
- USDA NOAA (United States Department of Agriculture, National Oceanic and Atmospheric Administration). United States Drought Monitor. Available online: https://data.nal.usda.gov/dataset/united-states-drought-monitor (accessed on 15 May 2020).
- Harper, R.M. Forests of Alabama Geological Survey of Alabama; Monograph 10; Wetumpka Printing Company: Wetumpka, AL, USA, 1943. [Google Scholar]
- Braun, E.L. Deciduous Forests of Eastern North America; The Blackburn Press: Caldwell, NJ, USA, 1950. [Google Scholar]
- Beckett, S.; Golden, M.S. Forest vegetation and vascular flora of Reed Brake Research Natural Area, Alabama. Castanea 1982, 47, 368–392. [Google Scholar]
- Cox, L.E.; Hart, J.L. Two centuries of forest compositional and structural changes in the Alabama Fall Line Hills. Am. Midl. Nat. 2015, 174, 218–238. [Google Scholar] [CrossRef]
- Kleinman, J.S.; Hart, J.L. Vascular flora of longleaf pine woodlands after wind disturbance and salvage harvesting in the Alabama Fall Line Hills. Castanea 2018, 83, 183–195. [Google Scholar] [CrossRef]
- Walker, J.L.; Silletti, A.M. Restoring the ground layer of longleaf pine ecosystems. In The Longleaf Pine Ecosystem; Jose, S., Jokela, E.J., Miller, D.L., Eds.; Springer: New York, NY, USA, 2007; pp. 297–333. [Google Scholar] [CrossRef]
- Van Lear, D.H.; Carroll, W.D.; Kapeluck, P.R.; Johnson, R. History and restoration of the longleaf pine-grassland ecosystem: Implications for species at risk. For. Ecol. Manag. 2005, 211, 150–165. [Google Scholar] [CrossRef]
- Platt, W.J.; Evans, G.W.; Rathbun, S.L. The population dynamics of a long-lived conifer (Pinus palustris). Am. Nat. 1988, 131, 491–525. [Google Scholar] [CrossRef]
- Reed, F.W. A Working Plan for Forest Lands in Central Alabama; US Department of Agriculture, Forest Service: Washington, DC, USA, 1905.
- NCDC (National Climatic Data Center). Storm Events Database. Available online: https://www.ncdc.noaa.gov/stormevents/ (accessed on 25 April 2020).
- NWS (National Weather Service). Sawyerville-Eoline (Greene, Hale and Bibb Counties) EF-3 Tornado 27 April 2011. Available online: https://www.weather.gov/bmx/event_04272011sawyerville (accessed on 25 April 2020).
- Goode, J.D.; Kleinman, J.S.; Hart, J.L.; Bhuta, A.A. Edge influence on composition and structure of a Pinus palustris woodland following catastrophic wind disturbance. Can. J. For. Res. 2020, 50, 332–341. [Google Scholar] [CrossRef]
- Chećko, E.; Jaroszewicz, B.; Olejniczak, K.; Kwiatkowska-Falińska, A.J. The importance of coarse woody debris for vascular plants in temperate mixed deciduous forests. Can. J. For. Res. 2015, 45, 1154–1163. [Google Scholar] [CrossRef]
- Donato, D.C.; Campbell, J.L.; Fontaine, J.B.; Law, B.E. Quantifying char in postfire woody detritus inventories. Fire Ecol. 2009, 5, 104–115. [Google Scholar] [CrossRef]
- Knapp, E.E.; Keeley, J.E.; Ballenger, E.A.; Brennan, T.J. Fuel reduction and coarse woody debris dynamics with early season and late season prescribed fire in a Sierra Nevada mixed conifer forest. For. Ecol. Manag. 2005, 208, 383–397. [Google Scholar] [CrossRef]
- Ulyshen, M.D.; Horn, S.; Pokswinski, S.; McHugh, J.V.; Hiers, J.K. A comparison of coarse woody debris volume and variety between old-growth and secondary longleaf pine forests in the southeastern United States. For. Ecol. Manag. 2018, 429, 124–132. [Google Scholar] [CrossRef]
- Schowalter, T.D.; Zhang, Y.L.; Sabin, T.E. Decomposition and nutrient dynamics of oak Quercus spp. logs after five years of decomposition. Ecography 1998, 21, 3–10. [Google Scholar] [CrossRef]
- LaFevor, M.C. Restoration of degraded agricultural terraces: Rebuilding landscape structure and process. J. Environ. Manag. 2014, 138, 32–42. [Google Scholar] [CrossRef]
- Dorau, K.; Mansfeldt, T. Comparison of redox potential dynamics in a diked marsh soil: 1990 to 1993 versus 2011 to 2014. J. Plant Nutr. Soil Sci. 2016, 179, 641–651. [Google Scholar] [CrossRef]
- Tome, J.B., Jr.; Dechen, A.R.; Atkinson, R.J. Effects of moist storage and different drying temperatures on the extractability of iron, copper, manganese, and zinc in soil samples. Commun. Soil Sci. Plan. 1996, 27, 2591–2611. [Google Scholar] [CrossRef]
- Davies, G.M.; Gray, A. Don’t let spurious accusations of pseudoreplication limit our ability to learn from natural experiments (and other messy kinds of ecological monitoring). Ecol. Evol. 2015, 5, 5295–5304. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: http://www.R-project.org/ (accessed on 25 April 2020).
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Cutler, D.R.; Edwards, T.C., Jr.; Beard, K.H.; Cutler, A.; Hess, K.T.; Gibson, J.; Lawler, J.J. Random forests for classification in ecology. Ecology 2007, 88, 2783–2792. [Google Scholar] [CrossRef] [PubMed]
- Liaw, A.; Wiener, M. Classification and Regression by random Forest. R News 2002, 2, 18–22. [Google Scholar]
- Kleinman, J.S.; Goode, J.D.; Hart, J.L.; Dey, D.C. Prescribed fire effects on Pinus palustris woodland development after catastrophic wind disturbance and salvage logging. For. Ecol. Manag. 2020, in press. [Google Scholar] [CrossRef]
- Clendenin, M.; Ross, W.G. Effects of cool season prescribed fire on understory vegetation in a mixed pine hardwood forest of east Texas. Tex. J. Sci. 2001, 53, 65–78. [Google Scholar]
- Dey, D.C.; Hartman, G. Returning fire to Ozark Highland forest ecosystems: Effects on advance regeneration. For. Ecol. Manag. 2005, 217, 37–53. [Google Scholar] [CrossRef]
- Fan, Z.; Ma, Z.; Dey, D.C.; Roberts, S.D. Response of advance reproduction of oaks and associated species to repeated prescribed fires in upland oak-hickory forests, Missouri. For. Ecol. Manag. 2012, 266, 160–169. [Google Scholar] [CrossRef]
- Olson, D.F.; Boyce, S.G. Factors affecting acorn production and germination and early growth of seedlings and seedling sprouts. In Oak Symposium Proceedings; US Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: Upper Darby, PA, USA, 1971; pp. 44–48. [Google Scholar]
- Whelan, A.W.; Bigelow, S.W.; Nieminen, M.F.; Jack, S.B. Fire season, overstory density and groundcover composition affect understory hardwood sprout demography in longleaf pine woodlands. Forests 2018, 9, 423. [Google Scholar] [CrossRef]
- Kuddes-Fischer, L.M.; Arthur, M.A. Response of understory vegetation and tree regeneration to a single prescribed fire in oak-pine forests. Nat. Area J. 2002, 22, 43–52. [Google Scholar]
- Kleinman, J.S.; Ford, S.A.; Hart, J.L. Catastrophic wind and salvage harvesting effects on woodland plants. For. Ecol. Manag. 2017, 403, 112–125. [Google Scholar] [CrossRef]
- Lyrene, P.M. First report of Vaccinium arboreum hybrids with cultivated highbush blueberry. HortScience 2011, 46, 563–566. [Google Scholar] [CrossRef]
- Freeman, D.C.; Brown, M.L.; Duda, J.J.; Graham, J.H.; Emlen, J.M.; Krzysik, A.J.; Balbach, H.E.; Kovacic, D.A.; Zak, J.C. Photosynthesis and fluctuating asymmetry as indicators of plant response to soil disturbance in the fall-line sandhills of Georgia: A case study using Rhus copallinum and Ipomoea pandurata. Int. J. Plant Sci. 2004, 165, 805–816. [Google Scholar] [CrossRef]
- Kleinman, J.S.; Hart, J.L. Response by vertical strata to catastrophic wind in restored Pinus palustris stands. J. Torrey Bot. Soc. 2017, 144, 423–438. [Google Scholar] [CrossRef]
- Perry, D.A.; Amaranthus, M.P.; Borchers, J.G.; Borchers, S.L.; Brainerd, R.E. Bootstrapping in ecosystems. Bioscience 1989, 39, 230–237. [Google Scholar] [CrossRef]
- Orman, O.; Adamus, M.; Szewczyk, J. Regeneration processes on coarse woody debris in mixed forests: Do tree germinants and seedlings have species-specific responses when grown on coarse woody debris? J. Ecol. 2016, 104, 1809–1818. [Google Scholar] [CrossRef]
- Kennedy, P.G.; Quinn, T. Understory plant establishment on old-growth stumps and the forest floor in western Washington. For. Ecol. Manag. 2001, 154, 193–200. [Google Scholar] [CrossRef]
- Sleeper, B.E.; Ficklin, R.L. Edaphic and vegetative responses to forested wetland restoration with created microtopography in Arkansas. Ecol. Restor. 2016, 34, 117–123. [Google Scholar] [CrossRef]
- Titus, J.H. Microtopography and woody plant regeneration in a hardwood floodplain swamp in Florida. Bull. Torrey Bot. Club 1990, 117, 429–437. [Google Scholar] [CrossRef]
- Russell, K.N.; Beauchamp, V.B. Plant species diversity in restored and created Delmarva Bay wetlands. Wetlands 2017, 37, 1119–1133. [Google Scholar] [CrossRef]
- Åström, M.; Dynesius, M.; Hylander, K.; Nilsson, C. Effects of slash harvest on bryophytes and vascular plants in southern boreal forest clear-cuts. J. Appl. Ecol. 2005, 42, 1194–1202. [Google Scholar] [CrossRef]
- Goldin, S.R.; Brookhouse, M.T. Effects of coarse woody debris on understorey plants in a temperate Australian woodland. Appl. Veg. Sci. 2015, 18, 134–142. [Google Scholar] [CrossRef]
- Bace, R.; Svoboda, M.; Janda, P. Density and height structure of seedlings in subalpine spruce forests of Central Europe: Logs vs. stumps as a favourable substrate. Silva Fenn. 2011, 45, 1065–1078. [Google Scholar] [CrossRef]
- Holeksa, J.; Saniga, M.; Szwagrzyk, J.; Dziedzic, T.; Ferenc, S.; Wodka, M. Altitudinal variability of stand structure and regeneration in the subalpine spruce forests of the Pol’ana biosphere reserve, Central Slovakia. Eur. J. For. Res. 2007, 126, 303–313. [Google Scholar] [CrossRef]
- Bird, P.R.; Jackson, T.T.; Kearney, G.A.; Roache, A. Effects of windbreak structure on shelter characteristics. Aust. J. Exp. Agric. 2007, 47, 727–737. [Google Scholar] [CrossRef]
- Cleugh, H.A.; Hughes, D.E. Impact of shelter on crop microclimates: A synthesis of results from wind tunnel and field experiments. Aust. J. Exp. Agric. 2002, 42, 679–701. [Google Scholar] [CrossRef]
- Ulyshen, M.D. Saproxylic Insects: Diversity, Ecology and Conservation; Springer International Publishing: New York, NY, USA, 2018. [Google Scholar]
- Davis, J.C.; Castleberry, S.B.; Kilgo, J.C. Influence of coarse woody debris on herpetofaunal communities in upland pine stands of the southeastern Coastal Plain. For. Ecol. Manag. 2010, 259, 1111–1117. [Google Scholar] [CrossRef]
- White, D.L. Land-use history. In Ecology and Management of a Forested Landscape: Fifty Years of Natural Resource Stewardship on the Savannah River Site; Kilgo, J.C., Blake, J.I., Eds.; Island Press: Washington, DC, USA, 2005; pp. 2–12. [Google Scholar]
- Błońska, E.; Kacprzyk, M.; Spolnik, A. Effect of deadwood of different tree species in various stages of decomposition on biochemical soil properties and carbon storage. Ecol. Res. 2017, 32, 193–203. [Google Scholar] [CrossRef]
- Stutz, K.P.; Lang, F. Potentials and unknowns in managing coarse woody debris for soil functioning. Forests 2017, 8, 37. [Google Scholar] [CrossRef]
- Laiho, R.; Prescott, C.E. Decay and nutrient dynamics of coarse woody debris in northern coniferous forests: A synthesis. Can. J. For. Res. 2004, 34, 763–777. [Google Scholar] [CrossRef]
- Ford, S.A.; Kleinman, J.S.; Hart, J.L. Effects of wind disturbance and salvage harvesting on macrofungal communities in a Pinus woodland. For. Ecol. Manag. 2018, 407, 31–46. [Google Scholar] [CrossRef]
- McIntyre, S.; Cunningham, R.B.; Donnelly, C.F.; Manning, A.D. Restoration of eucalypt grassy woodland: Effects of experimental interventions on ground-layer vegetation. Aust. J. Bot. 2015, 62, 570–579. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Gustafsson, L.; Benayas, J.M.R.; Castro, J. Does post-disturbance salvage logging affect the provision of ecosystem services? A systematic review protocol. Environ. Evid. 2015, 4, 16. [Google Scholar] [CrossRef]
- Beschta, R.L.; Rhodes, J.J.; Kauffman, J.B.; Gresswell, R.E.; Minshall, G.W.; Karr, J.R.; Perry, D.A.; Hauer, F.R.; Frissell, C.A. Postfire management on forested public lands of the western United States. Conserv. Biol. 2004, 18, 957–967. [Google Scholar] [CrossRef]
- DellaSala, D.A.; Karr, J.R.; Schoennagel, T.; Perry, D.; Noss, R.F.; Lindenmayer, D.; Beschta, R.; Hutto, R.L.; Swanson, M.E.; Evans, J. Post-fire logging debate ignores many issues. Science 2006, 314, 51–52. [Google Scholar] [CrossRef] [PubMed]
- Lindenmayer, D.B.; Noss, R.F. Salvage logging, ecosystem processes, and biodiversity conservation. Conserv. Biol. 2006, 20, 949–958. [Google Scholar] [CrossRef]
- Kroll, A.J.; Duke, S.D.; Hane, M.E.; Johnson, J.R.; Rochelle, M.; Betts, M.G.; Arnett, E.B. Landscape composition influences avian colonization of experimentally created snags. Biol. Conserv. 2012, 152, 145–151. [Google Scholar] [CrossRef]
- Rost, J.; Clavero, M.; Brotons, L.; Pons, P. The effect of postfire salvage logging on bird communities in Mediterranean pine forests: The benefits for declining species. J. Appl. Ecol. 2012, 49, 644–651. [Google Scholar] [CrossRef]
- Kleinman, J.S.; Goode, J.D.; Fries, A.C.; Hart, J.L. Ecological consequences of compound disturbances in forest ecosystems: A systematic review. Ecosphere 2019, 10, e02962. [Google Scholar] [CrossRef]
- Trumbore, S.; Brando, P.; Hartmann, H. Forest health and global change. Science 2015, 349, 814–818. [Google Scholar] [CrossRef]
- Turner, M.G. Disturbance and landscape dynamics in a changing world. Ecology 2010, 91, 2833–2849. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.W.; Jackson, S.T. Novel climates, no-analog communities, and ecological surprises. Front. Ecol. Environ. 2007, 5, 475–482. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).