Abstract
Litter decomposition plays a critical role in regulating biogeochemical cycles in terrestrial ecosystems and is profoundly impacted by increasing atmospheric nitrogen (N) deposition. Here, a N manipulation experiment was conducted to explore the effects of N additions (0 kg N ha−1 yr−1, 20 kg N ha−1 yr−1 and 40 kg N ha−1 yr−1) on decay rates and nutrients release of two contrasting species, the evergreen and nutrient-poor Michelia wilsonii and the deciduous and nutrient-rich Camptotheca acuminata, using a litterbag approach at the western edge of the Sichuan Basin of China. The decay rate and the mineralization of N and phosphorus (P) was faster in nutrient-rich C. acuminata litter than in nutrient-poor M. wilsonii litter, regardless of N regimes. N additions tended to decrease the decay constant (k value) in M. wilsonii litter, but had no effect on C. acuminata litter. N additions had no significant effects on carbon (C) release of both litter types. N additions showed negative effects on N and P release of M. wilsonii litter, particularly in the late decomposition stage. Moreover, for C. acuminata litter, N additions did not affect N release, but retarded P release in the late stage. N additions did not affect the C:N ratio in both litter types. However, N additions—especially high-N addition treatments—tended to reduce C:P and N:P ratios in both species. The effect of N addition on N and P remaining was stronger in M. wilsonii litter than in C. acuminata litter. The results of this study indicate that N additions retarded the nutrients release of two foliar litters. Thus, rising N deposition might favor the retention of N and P via litter decomposition in this specific area experiencing significant N deposition.
1. Introduction
The decomposition of plant litter is a key determinant of carbon (C) and nutrient cycling in terrestrial ecosystems. It is a primary source of energy and nutrients for complex detritus-based food webs and constitutes an important ecosystem service by recycling nutrients, renewing soil fertility, and carbon sequestration [1]. Studies have demonstrated that litter decomposition is controlled by both internal chemical components and external environmental conditions (e.g., climate and soil decomposers) [2]. Reactive nitrogen (N) deposition has increased dramatically over the last several decades in tropical areas [3,4,5]. Elevated N deposition consequently increases soil N availability [6], which could further have complex and profound influences on litter decomposition in subtropical forests.
Nitrogen deposition, as a global change driver, has great potential to affect plant litter decomposition in terrestrial ecosystems [7]. It has been reported that N additions cause positive, negative, or neutral effects on litter decomposition [8,9,10]. The effect of N input on decomposition is regulated by substrate quality, N addition rates, and ambient N deposition [4,6]. In N-deficient ecosystems, N addition generally stimulates the decomposition by increasing soil N availability or reducing the C:N litter ratios [11]. However, in N-sufficient sites, the added N often shows an inhibitory or neutral effect on litter decomposition and nutrients mineralization due to the saturation of soil N availability and the suppression of enzymic synthesis [12,13,14,15,16]. The response of litter decomposition to environmental changes may largely depend on the litter’s initial nutrient conditions [17,18]. As far as we know, nutrient-rich litter generally decays faster relative to nutrient-poor litter [19,20]. Regarding N additions, the effect of added N on litter decomposition and nutrient release generally varies among litter types and decomposition stages. A previous meta-analysis also indicated that N additions stimulated the decomposition rate of high-nutrient litter, but inhibited the decomposition of low-nutrient litter [4]. Additionally, the impact of N additions is positive in the initial phase, but negative in the late stage [7,21]. As a result, it is important to investigate N deposition responses of different-quality litters coexisting in an ecosystem using a N manipulation experiment, particularly in high-N-deposition subtropical forest ecosystems.
N deposition has risen significantly in China in the last several decades [22]. Increasing N deposition may be favorable to C sequestration in subtropical forests [23]. Atmospheric wet N deposition on the western edge of the Sichuan Basin (known as the “rainy zone of west China”) is much higher than the mean value across China, as a result of rich rainfall and reactive N [24]. Previous studies have indicated that extremely high N additions (100 and 150 kg N ha−1 yr−1) constrained the litter decomposition rate in subtropical forests [25,26,27]. However, the effect of N deposition on foliage decomposition and nutrients mineralization may vary between litter types (nutrient-rich vs. nutrient-poor) or decomposition stages (early vs. late). To examine the aforementioned scientific hypothesis, a field experiment was conducted to examine the effect of simulated N deposition on mass loss and C, N, and phosphorus (P) release, as well as the stoichiometric dynamics of two contrasting broad-leaved litter types. Michelia wilsonii is an evergreen tree species, but Camptotheca acuminata is a deciduous tree species. There were significant differences in litter matrix between the two tree species. Initial N and P concentrations were lower in the M. wilsonii litter relative to those of the C. acuminata litter. Conversely, lignin, cellulose, C:N, C:P, and lignin:N were significantly higher in the M. wilsonii litter versus the C. acuminata litter [28]. The main objectives of this study were to: (i) examine the effect of N additions on the decay rate as well as the C, N, and P release of two litters; and (ii) determine if the N-induced effect varied between two contrasting litter types.
2. Materials and Methods
2.1. Site Description and Experimental Design
The study was conducted in the Dujiangyan Experimental Forest of the Sichuan Agricultural University, Southwestern China (31°01’–31°04’ N, 103°37’–103°43’ E, 896−1320 m a.s.l.). This forest is characterized by a canopy composed of M. wilsonii and C. acuminata. Mean annual precipitation is 1243 mm. Mean annual temperature is 15.2 °C, with a minimum in January (−1.4 °C) and maximum in July (31.6 °C). The soil type is classified as Ferralsol with old alluvial yellow loam, according to the FAO (Food and Agriculture Organization of the United Nations) classification system. The basic topsoil properties (0−20 cm) are as follows: organic C 15.76 g kg−1, total N 1.92 g kg−1, total P 0.32 g kg−1, and pH 5.73.
Experimental plots were established in November 2015, according to the ambient atmospheric wet N deposition (36.2 kg N ha−1yr−1) in the study site [29]. Three N addition levels of 10 × 10 m in triplicates were established for this experiment: control (Control: 0 kg N ha−1 yr−1), low-N addition (LN: 20 kg N ha−1 yr−1) and high-N addition (HN: 40 kg N ha−1 yr−1). Both LN and HN treatments roughly simulate the scenarios that wet N deposition would be increased by about 50% and 100%, respectively. Ammonium nitrate (NH4NO3) solution was applied monthly in 12 equal applications from December 2015 to December 2016. There were no significant differences in soil temperature or moisture under different N addition treatments [28].
2.2. Litter Bag Experiment
Both M. wilsonii and C. acuminata were chosen in this study because they are the two dominant tree species in this site. In addition, they represent two contrasting litter types (evergreen and rich-nutrient vs. deciduous and low-nutrient) in subtropical areas. We collected fresh litter from the forest stand adjacent to the N manipulation site in late October 2015. Air-dried material of approximately 15.00 ± 0.01 g of two species was placed into 20 × 25 cm nylon mesh bags with a 1.0 mm mesh on the surface and 0.055 mm mesh on the bottom. Duplicate litter bags were placed on the surface in the respective treatment section in early January 2016. Meanwhile, two litterbags of each species were retrieved from each plot for determination of initial chemical properties, and were harvested after 46, 105, 164, 284 and 351 days from each plot. Soil particles and other extraneous materials were removed from the harvested litters, and the cleaned samples were then oven-dried (85 °C for 48 h) to constant mass and the mass remaining was weighed.
2.3. Chemical Analysis
The oven-dried litter samples were ground and sieved. The sieved samples were used for chemical analysis. The organic carbon (C), total nitrogen (N), and total phosphorus (P) concentrations were determined with the methods of dichromate oxidation, Kjeldahl determination (KDN, Top Ltd., Zhejiang, China) and phosphomolybdenum yellow spectrophotometry (TU-1901, Puxi Ltd., Beijing, China), respectively.
2.4. Calculations and Statistical Analysis
Decomposition rates were calculated from the remaining dry mass using a single negative exponential decay model y = e−kt, where y is the fraction of mass remaining at time t, t is the time elapsed in years and k is the annual decay constant [30]. The remaining amount of elements C, N and P on each sampling date was determined by multiplying the litter elements’ (C, N and P) concentration by the litter mass and the litter mass remaining, and comparing it to the initial litter elements’ (C, N and P) mass. The stoichiometric rates (C:N, C:P and N:P) were calculated on an element mass basis. Repeated-measure analysis of the variance was used to test the effects of tree species, N additions, sampling data and their interactions on elements (C, N and P) remaining and stoichiometric ratios. For a specific date, one-way analysis of variance was used to determine the chemistry addition effect on the measured parameters. Differences among means were considered significant at the p < 0.05 level. All statistical tests were performed using the Software Statistical Package for the Social Sciences (SPSS) version 22.0 (IBM SPSS Statistics Inc., Chicago, IL, USA).
3. Results
3.1. Decay Constant
Regardless of N additions, decay constants (k values) markedly differed between species (Figure 1). C. acuminata litter always decomposed faster than M. wilsonii litter. For M. wilsonii litter, the k values were significantly lower in the N addition treatments than in the control. However, N additions had no significant effect on k values of C. acuminata (p > 0.05).
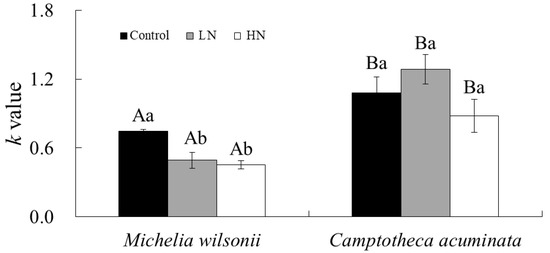
Figure 1.
The decomposition constant (k, year−1) of M. wilsonii and C. acuminata over 1-year decomposition under three N treatments. Values are expressed as mean ± SE. Different lowercase letters indicate significant differences between N additions for the same species; different capital letters denote significant differences between tree species for the same N addition. Control: no N addition (0 kg N ha−1 yr−1); HN: high-N addition (40 kg N ha−1 yr−1); LN: low-N addition (20 kg N ha−1 yr−1).
3.2. Litter C, N and P Remaining
The amount of C remaining in both species decreased significantly as decomposition proceeded (Table 1; Figure 2a,b). After 168-day fast loss, C loss in M. wilsonii gradually slowed down and retained 24–32% after one year of decomposition. Similarly, C loss of C. acuminata litter occurred rapidly and left 11–17% until the end of the experiment (Figure 2a,b). N treatments had no significant effect on litter C loss (p = 0.139, Table 1).

Table 1.
Results of repeated-measure ANOVA showing the effects of sampling dates (T), tree species (S), N treatments (N) and their interactions on element (C, N and P) remaining and their C:N:P stoichiometry.
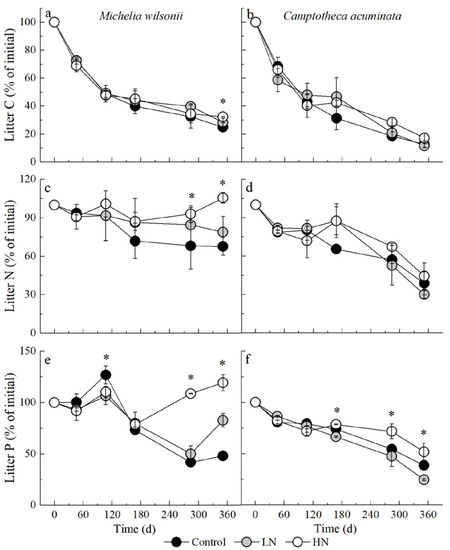
Figure 2.
C, N and P remaining of M. wilsonii and C. acuminata over 1-year decomposition under three N treatments. Values are expressed as mean ± SE. * Indicates significant differences among N additions. Control: no N addition (0 kg N ha−1 yr−1); HN: high-N addition (40 kg N ha−1 yr−1); LN: low-N addition (20 kg N ha−1 yr−1).
For M. wilsonii, the remaining N was higher in the N treatment plots than in the control plots during the late stage, especially after 284 and 351 days (Table 1; Figure 2c,d). By the end of the experiment, the remaining N for M. wilsonii was 68%, 79% and 106% for the control, LN and HN plots, respectively (Figure 2c,d). However, N addition did not significantly affect C. acuminata N release. The remaining N for C. acuminata was 61%, 56% and 70% for the control, LN and HN plots, respectively (Figure 2c,d). Regardless of N treatments, M. wilsonii N loss rates were significantly slower than those of C. acuminata after one year of incubation (Table 1; Figure 2c,d). Statistical analysis indicated that the effect of N addition on the remaining N in litter was dependent on litter type and decomposition period (Table 1).
Both litter types showed different patterns in P release throughout the experimental period (Figure 2e,f; Table 1). The P content of M. wilsonii litter clearly accumulated in the late decomposition stage (Figure 2e). However, the C. acuminata P content was always released during the decomposition process (Figure 2f). After one year of decomposition, the P remaining in M. wilsonii litter was 48.2%, 82.7% and 119.4%, in the control, LN and HN plots, respectively. However, the P remaining in C. acuminata, was only 38.8%, 24.6% and 51.9% in the control, LN and HN plots, respectively. The ANOVA analysis showed that N treatments, species, decomposition stages and their interactions all had significant influences on the P remaining (Table 1).
3.3. Stoichiometric Dynamic
Regardless of tree species and N treatment, the C:N ratio generally declined as litter decomposition progressed (Table 1; Figure 3a,b). N additions did not affect litter C:N ratio, but the M. wilsonii C:N ratio was often higher than the C. acuminata C:N ratio, regardless of the N treatment (Table 1; Figure 3a,b). The ANOVA analysis showed that the interactions between N additions and tree species or decomposition stages had no significant impact on the litter C:N ratio (Table 1; Figure 3a,b).
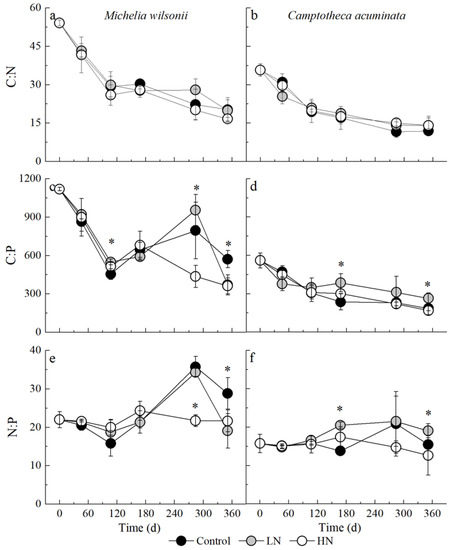
Figure 3.
C, N and P stoichiometric ratios of M. wilsonii and C. acuminata over 1-year decomposition under three N treatments. Values are expressed as means ± SE. * Indicates significant differences among N additions. Control: no N addition (0 kg N ha−1 yr−1); HN: high-N addition (40 kg N ha−1 yr−1); LN: low-N addition (20 kg N ha−1 yr−1).
Irrespective of N treatment, the C:P ratio of M. wilsonii litter remarkably declined during the first 105 days of decomposition but tended to increase after 286 days. N treatments significantly decreased C:P ratios in 351 days, compared to control plots (Figure 3c). However, the C. acuminata C:P ratio generally decreased over the one-year decomposition (Figure 3d). The ANOVA analysis showed that tree species, N additions, sampling dates and their interactions all had a significant impact on C:P ratio (Table 1).
4. Discussion
The results of this study showed significant differences in litter decomposition and nutrient release between tree species. Generally, litter with high initial nutrient content and low C:N ratio were suggested to have higher decomposition rates [31]. In our study, nutrient content was higher, but stoichiometric ratios were lower in C. acuminata litter than in M. wilsonii litter. Consequently, the decomposition rate of C. acuminata litter was often faster than that of M. wilsonii litter, irrespective of the N regime.
However, N additions had different effects on the decay rate of the two types of litter. N additions decreased the decomposition rate of M. wilsonii litter, but did not have an effect on C. acuminata litter. A previous study also reported that extremely high N additions inhibited litter decomposition rate in subtropical forests [14]. Knorr et al. [4] concluded that ambient N deposition and N addition levels are two important factors regulating litter decomposition. Litter decomposition may be inhibited when N addition rates are 2–20 times higher than the ambient N deposition amount. In the present case, N addition levels were roughly between 0.5 and 1 times higher than the background N deposition. Our fertilization rate was relatively low compared to other simulated N deposition experiments. On the other hand, elevated N may increase soil P limitation in a site, and the P effect may override the N effect [32]. Therefore, litter P content might regulate the decomposition rate in high ambient N deposition and P-poor ecosystems. Thus, N additions exhibited a greater suppression on P-low M. wilsonii litter compared to high-P C. acuminata litter.
Nutrient release in decomposing litter is positively regulated by total N content or stoichiometric ratios (e.g., C:N and C:P) [33]. Previous studies reported that N in decomposing litter starts to release when the C:N ratio of the litter is <40 [21]. The results in our study noted the initial C:N ratio of M. wilsonii and C. acuminata was 54 and 36, respectively. Net N release of both tree species occurred during the entire experiment. The rate of N release was greater in N-rich C. acuminata than in N-poor M. wilsonii. This may be because the M. wilsonii C:N ratio is much higher than that of C. acuminata. N additions slowed down the net N release in decaying litter. In addition, low-N litter M. wilsonii could be more easily affected by external N addition relative to high-N litter C. acuminata. Litter N mineralization may also be mediated by initial litter N content [34]. Simulated N deposition suppressed the net N release of foliar litter in a natural evergreen broad-leaved forest and in a bamboo forest with a high background N deposition area [35]. Moreover, the suppression of N addition on litter N mineralization was stronger in N-poor Liquidambar formosana litter than in N-rich Alnus cremastogyne [36]. This study also found that the effect of N addition on litter N release differed between contrasting litters, with M. wilsonii being stronger than C. acuminata. It is important to note that N was even immobilized in the M. wilsonii litter in the HN treatment by the end of the experiment. Net N immobilizations generally occur as decomposers access N exogenous to the litter and convert it to microbial biomass or exo-enzymes [37]. In this region, microbes may not meet their basic N requirements. N addition treatments could, to some extent, provide more external N for biological enrichment, implying that the N release in decomposing litters may be partly offset by microbial immobilization.
Similar to N release, the P pattern in decomposing litter is primarily regulated by litter C:P ratios [14,37]. The immobilization of litter P has been seen to occur when the C:P ratio is greater than 700 [38]. The initial C:P ratio for C. acuminata litter was 560. Net P release occurred in C. acuminata litter under all the N additions throughout the experimental period. However, the initial C:P ratio for M. wilsonii litter was 1192. As a result, microbes often immobilized more P in the litter. In addition, HN addition resulted in the net P immobilization or inhibitory effect of litter P in both litter types by the end of the experimental period. P input is usually minimal in natural ecosystems, therefore, N additions may, to some extent, strengthen the P limitation of the ecosystems. Litter P could be rapidly recycled internally during the decomposition processes, which could in turn hinder P release to the soil [39]. Our study found that N additions tended to favor the accumulation of P. Some studies have also found an accumulation of P with N deposition [40]. The litter P accumulation induced by N deposition might attribute to a relative shortage of P associated with the stoichiometric constraint theory [41], but further supporting evidence is necessary.
Irrespectively of tree species and N additions, the C:N ratio generally decreased as litter decomposition progressed. This result is in line with the observations obtained in many other tree species [42,43,44]. The C:P and N:P ratios pattern was different between two contrasting tree species during the decomposition process. This is because the nutrients release pattern differed between C. acuminata and M. wilsonii during decomposition. In addition, previous studies have also found that simulated N deposition altered the C, N and P stoichiometry [16,20]. As mentioned above, litter C release was similar among N addition treatments, but litter P release declined with increasing N additions. To alleviate the N-induced negative effect on litter P release, the litter P demand will be increased under N additions [39]. Therefore, N additions, especially under the HN treatment, tended to decrease C:P ratios in the late period of decomposition. On the other hand, N-inhibited magnitude in litter P was greater compared to that in litter N during the late period. N additions also tended to decrease litter N:P stoichiometry in the late period of decomposition.
5. Conclusions
This study investigated the litter decomposition and nutrients release of two contrasting litter types following gradient N additions in a subtropical forest with high N background. The decay rate and the mineralization of N and P was faster in nutrient-rich C. acuminata litter than in nutrient-poor M. wilsonii litter, regardless of N regime. The effect of N addition had negative effects on the decomposition rate as well as N and P release in M. wilsonii litter during the late decay phase. For C. acuminata, N additions did not affect N release, but retarded P release in the late stage. Overall, the effect of N addition on the remaining N and P was stronger in M. wilsonii litter than in C. acuminata litter, probably due to the elevated C:N and C:P ratios during decomposition. Increasing N deposition may slow down the nutrient release from decomposing litter in the subtropical forests where significant N deposition occurs. Additionally, N-induced effects may, to some extent, be dependent on the initial quality and the decay stage.
Author Contributions
Z.X. conceived and designed study; L.Z. (Liyan Zhuang), Q.L. and K.Y. performed the experiments; Z.L., C.Y. analyzed data; B.T., L.Z. (Li Zhang), R.Y. and R.B. contributed reagents/methods/analysis tools; L.Z. (Liyan Zhuang), Q.L. and Z.X. wrote the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by Program of Sichuan Excellent Youth Sci-Tech Foundation (20JCQN0145), the National Natural Science Foundation of China (31500509 and 31700542) and National Key Research and Development Program of China (2016YFC0502505 and 2017YFC0505003) and the Key Program of Sichuan Education Department (17ZA0321).
Acknowledgments
The authors thank the following people for their help in this research. Tong Wei and Ji Yuan provided field assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Abelho, M. Litter traits and decomposer complexity set the stage for a global decomposition model. Funct. Ecol. 2016, 30, 674–675. [Google Scholar] [CrossRef]
- Zhang, D.Q.; Hui, D.F.; Luo, Y.Q.; Zhou, G.Y. Rates of litter decomposition in terrestrial ecosystems: Global patterns and controlling factors. J. Plant Ecol. 2008, 1, 85–93. [Google Scholar] [CrossRef]
- Matson, P.; Lohse, K.A.; Hall, S.J. The globalization of nitrogen deposition: Consequences for terrestrial ecosystems. Ambio A J. Hum. Environ. 2002, 31, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Knorr, M.; Frey, S.D.; Curtis, P.S. Nitrogen addition and litter decomposition: A meta-analysis. Ecology 2005, 86, 3252–3257. [Google Scholar] [CrossRef]
- Liu, X.J.; Duan, L.; Mo, J.M.; Du, E.Z.; Shen, J.L.; Lu, X.K.; Zhang, Y.; Zhou, X.B.; He, C.N.; Zhang, F.S. Nitrogen deposition and its ecological impact in China: An overview. Environ. Pollut. 2011, 159, 2251–2264. [Google Scholar] [CrossRef] [PubMed]
- Manning, P.; Saunders, M.; Bardgett, R.D.; Bonkowski, M.; Bradford, M.A.; Ellis, R.J.; Kandeler, E.; Marhan, S.; Tscherko, D. Direct and indirect effects of nitrogen deposition on litter decomposition. Soil Biol. Biochem. 2008, 40, 688–698. [Google Scholar] [CrossRef]
- Berg, B.; Matzner, E. Effect of N deposition on decomposition of plant litter and soil organic matter in forest systems. Environ. Rev. 1997, 5, 1–25. [Google Scholar] [CrossRef]
- Liu, P.; Sun, O.J.; Huang, J.; Li, L.; Han, X. Nonadditive effects of litter mixtures on decomposition and correlation with initial litter N and P concentrations in grassland plant species of northern China. Biol. Fertil. Soils. 2007, 44, 211–216. [Google Scholar] [CrossRef]
- Berg, B. Decomposition patterns for foliar litter—A theory for influencing factors. Soil Biol. Biochem. 2014, 78, 222–232. [Google Scholar] [CrossRef]
- Zhang, T.A.; Luo, Y.; Chen, H.Y.; Ruan, H. Responses of litter decomposition and nutrient release to N addition: A meta-analysis of terrestrial ecosystems. Appl. Soil Ecol. 2018, 128, 35–42. [Google Scholar] [CrossRef]
- Norris, M.D.; Avis, P.G.; Reich, P.B.; Hobbie, S.E. Positive feedbacks between decomposition and soil nitrogen availability along fertility gradients. Plant Soil 2013, 367, 347–361. [Google Scholar] [CrossRef]
- Downs, M.R.; Nadelhoffer, K.J.; Melillo, J.M.; Aber, J.D. Immobilization of a N-15-labeled nitrate addition by decomposing forest litter. Oecologia 1996, 105, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Mo, J.; Brown, S.; Xue, J.; Fang, Y.; Li, Z. Response of litter decomposition to simulated N deposition in disturbed, rehabilitated and mature forests in subtropical China. Plant Soil 2006, 282, 135–151. [Google Scholar] [CrossRef]
- Zhou, S.X.; Huang, C.D.; Han, B.H.; Xiao, Y.X.; Tang, J.D.; Xiang, Y.B.; Luo, C. Simulated nitrogen deposition significantly suppresses the decomposition of forest litter in a natural evergreen broad-leaved forest in the rainy area of Western China. Plant Soil 2017, 420, 135–145. [Google Scholar] [CrossRef]
- Carreiro, M.; Sinsabaugh, R.; Repert, D.; Parkhurst, D. Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 2000, 81, 2359–2365. [Google Scholar] [CrossRef]
- Hobbie, S.E. Nitrogen effects on decomposition: A five-year experiment in eight temperate sites. Ecology 2008, 89, 2633–2644. [Google Scholar] [CrossRef]
- Reich, P.B.; Oleksyn, J.; Modrzynski, J.; Mrozinski, P.; Hobbie, S.E.; Eissenstat, D.M.; Chorover, J.; Chadwick, O.A.; Hale, C.M.; Tjoelker, M.G. Linking litter calcium, earthworms and soil properties: A common garden test with 14 tree species. Ecol. Lett. 2005, 8, 811–818. [Google Scholar] [CrossRef]
- Zhang, W.D.; Chao, L.; Yang, Q.P.; Wang, Q.K.; Fang, Y.T.; Wang, S.L. Litter quality mediated nitrogen effect on plant litter decomposition regardless of soil fauna presence. Ecology 2016, 97, 2834–2843. [Google Scholar] [CrossRef]
- Micks, P.; Downs, M.R.; Magill, A.H.; Nadelhoffer, K.J.; Aber, J.D. Decomposing litter as a sink for 15N-enriched additions to an oak forest and a red pine plantation. For. Ecol. Manag. 2004, 196, 71–87. [Google Scholar] [CrossRef]
- Zhu, X.M.; Chen, H.; Zhang, W.; Huang, J.; Fu, S.L.; Liu, Z.F.; Mo, J.M. Effects of nitrogen addition on litter decomposition and nutrient release in two tropical plantations with N2-fixing vs. non-N2-fixing tree species. Plant Soil 2016, 399, 61–74. [Google Scholar] [CrossRef]
- Parton, W.; Silver, W.L.; Burke, I.C.; Grassens, L.; Harmon, M.E.; Currie, W.S.; King, J.Y.; Adair, E.C.; Brandt, L.A.; Hart, S.C. Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 2007, 315, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.R.; Jia, Y.L.; He, N.P.; Zhu, J.X.; Chen, Z.; Wang, Q.F.; Piao, S.L.; Liu, X.J.; He, H.L.; Guo, X.B.; et al. Stabilization of atmospheric nitrogen deposition in China over the past decade. Nat. Geosci. 2019, 12, 424–429. [Google Scholar] [CrossRef]
- Yu, G.R.; Chen, Z.; Piao, S.L.; Peng, C.H.; Ciais, P.; Wang, Q.F.; Li, X.R.; Zhu, X.J. High carbon dioxide uptake by subtropical forest ecosystems in the East Asian monsoon region. Proc. Natl. Acad. Sci. USA 2014, 111, 4910–4915. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhao, Y.H.; Liu, X.J.; Dore, A.J.; Zhang, L.; Liu, L.; Cheng, M.M. Atmospheric nitrogen deposition in the Yangtze River basin: Spatial pattern and source attribution. Environ. Pollut. 2018, 232, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Tu, L.H.; Hu, H.L.; Chen, G.; Peng, Y.; Xiao, Y.L.; Hu, T.X.; Zhang, L.; Li, X.W.; Liu, L.; Tang, Y. Nitrogen addition significantly affects forest litter decomposition under high levels of ambient nitrogen deposition. PLoS ONE 2014, 9, e88752. [Google Scholar] [CrossRef]
- Chen, G.T.; Tu, L.H.; Peng, Y.; Hu, H.L.; Hu, T.X.; Xu, Z.F.; Liu, L.; Tang, Y. Effect of nitrogen additions on root morphology and chemistry in a subtropical bamboo forest. Plant Soil 2017, 412, 441–451. [Google Scholar] [CrossRef]
- Tu, L.H.; Hu, T.X.; Zhang, J.; Li, X.W.; Hu, H.L.; Liu, L.; Xiao, Y.L. Nitrogen addition stimulates different components of soil respiration in a subtropical bamboo ecosystem. Soil Biol. Biochem. 2013, 58, 255–264. [Google Scholar] [CrossRef]
- Liu, Q.; Zhuang, L.Y.; Ni, X.Y.; You, C.M.; Yang, W.Q.; Wu, F.Z.; Tan, B.; Yue, K.; Liu, Y.; Zhang, L.; et al. Nitrogen additions stimulate litter humification in a subtropical forest, southwestern China. Sci. Rep. 2018, 8, 17525. [Google Scholar] [CrossRef]
- Yang, K.J.; Yang, Y.L.; Xu, Z.F.; Wu, Q.G. Soil respiration in a subtropical forest of southwestern China: Components, patterns and controls. PLoS ONE 2018, 13, e0204341. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.C.; Amatangelo, K.; Dorrepaal, E.; Eviner, V.T.; Godoy, O.; Hobbie, S.E.; Hoorens, B.; Kurokawa, H.; Pérez-Harguindeguy, N.; et al. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol. Lett. 2008, 11, 1065–1071. [Google Scholar] [CrossRef]
- Marklein, A.R.; Houlton, B.Z. Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol. 2012, 193, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Aerts, R. Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: A triangular relationship. Oikos 1997, 79, 439–449. [Google Scholar] [CrossRef]
- Moore, T.R.; Trofymow, J.A.; Prescott, C.E.; Fyles, J.; Titus, B.D. Patterns of carbon, nitrogen and phosphorus dynamics in decomposing foliar litter in Canadian forests. Ecosystems 2006, 9, 46–62. [Google Scholar] [CrossRef]
- Tu, L.H.; Hu, H.L.; Hu, T.X.; Zhang, J.; Liu, L.; Li, R.H.; Dai, H.Z.; Luo, S.H. Decomposition of different litter fractions in a subtropical bamboo ecosystem as affected by experimental nitrogen deposition. Pedosphere 2011, 21, 685–695. [Google Scholar] [CrossRef]
- Kuperman, R.G. Litter decomposition and nutrient dynamics in oak–hickory forests along a historic gradient of nitrogen and sulfur deposition. Soil Biol. Biochem. 1999, 31, 237–244. [Google Scholar] [CrossRef]
- Zhou, G.; Zhang, J.; Qiu, X.; Wei, F.; Xu, X. Decomposing litter and associated microbial activity responses to nitrogen deposition in two subtropical forests containing nitrogen-fixing or non-nitrogen-fixing tree species. Sci. Rep. 2018, 8, 129334. [Google Scholar] [CrossRef]
- Zheng, Z.M.; Mamuti, M.; Liu, H.M.; Shu, Y.Q.; Hu, S.J.; Wang, X.H.; Li, B.B.; Lin, L.; Li, X. Effects of nutrient additions on litter decomposition regulated by phosphorus-induced changes in litter chemistry in a subtropical forest, China. For. Ecol. Manag. 2017, 400, 123–128. [Google Scholar] [CrossRef]
- Vivanco, L.; Austin, A.T. Nitrogen addition stimulates forest litter decomposition and disrupts species interactions in Patagonia, Argentina. Glob. Chang. Biol. 2011, 17, 1963–1974. [Google Scholar] [CrossRef]
- Chen, H.; Dong, S.; Liu, L.; Ma, C.; Zhang, T.; Zhu, X.; Mo, J. Effects of experimental nitrogen and phosphorus addition on litter decomposition in an old-growth tropical forest. PLoS ONE 2013, 8, e84101. [Google Scholar] [CrossRef]
- Gong, J.; Zhu, C.; Yang, L.; Yang, B.; Wang, B.; Baoyin, T.T.; Liu, M.; Zhang, Z.; Shi, J. Effects of nitrogen addition on above-and belowground litter decomposition and nutrient dynamics in the litter-soil continuum in the temperate steppe of Inner Mongolia, China. J. Arid Environ. 2020, 172, 104036. [Google Scholar] [CrossRef]
- Yuan, Z.; Chen, H.Y. Negative effects of fertilization on plant nutrient resorption. Ecology 2015, 96, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Cotrufo, M.F.; Ineson, P.; Roberts, J.D. Decomposition of birch leaf litters with varying C-to-N ratios. Soil Biol. Biochem. 1995, 27, 1219–1221. [Google Scholar] [CrossRef]
- Finn, D.; Page, K.; Catton, K.; Strounina, E.; Kienzle, M.; Robertson, F.; Armstrong, R.; Dalal, R. Effect of added nitrogen on plant litter decomposition depends on initial soil carbon and nitrogen stoichiometry. Soil Biol. Biochem. 2015, 91, 160–168. [Google Scholar] [CrossRef]
- Manzoni, S.; Trofymow, J.A.; Jackson, R.B.; Porporato, A. Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol. Monogr. 2010, 80, 89–106. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).