The Assessment of Two Species of Soapberry as Resources for High-Quality Biodiesel Production with an Optimized Method of Ultrasound-Assisted Oil Extraction
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Ultrasound-Assisted Extraction (UAE)
2.3. Determination of Extraction Yield
2.4. Optimization of Ultrasonic-assisted Extraction
2.5. Fatty Acids Composition
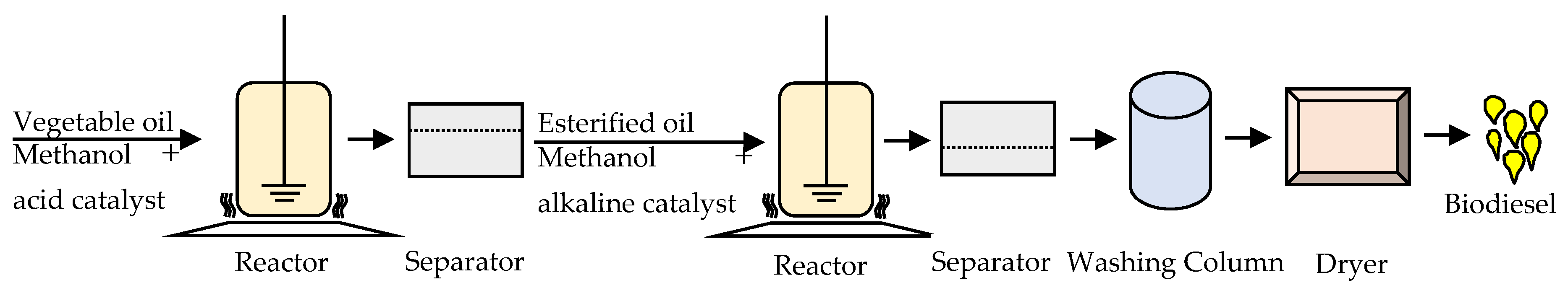
2.6. Preparation of Biodiesel
2.7. Properties of Biodiesel
3. Results
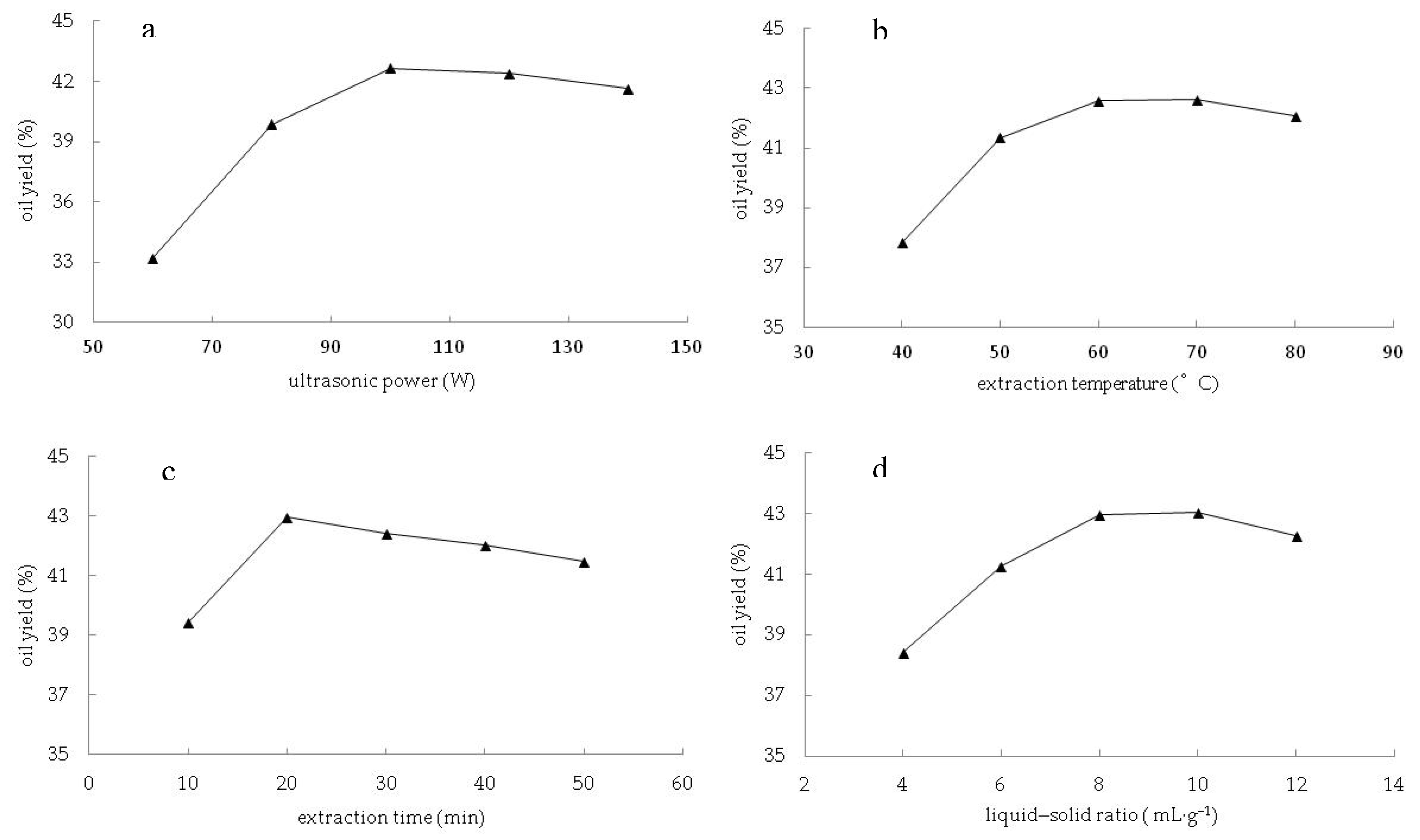
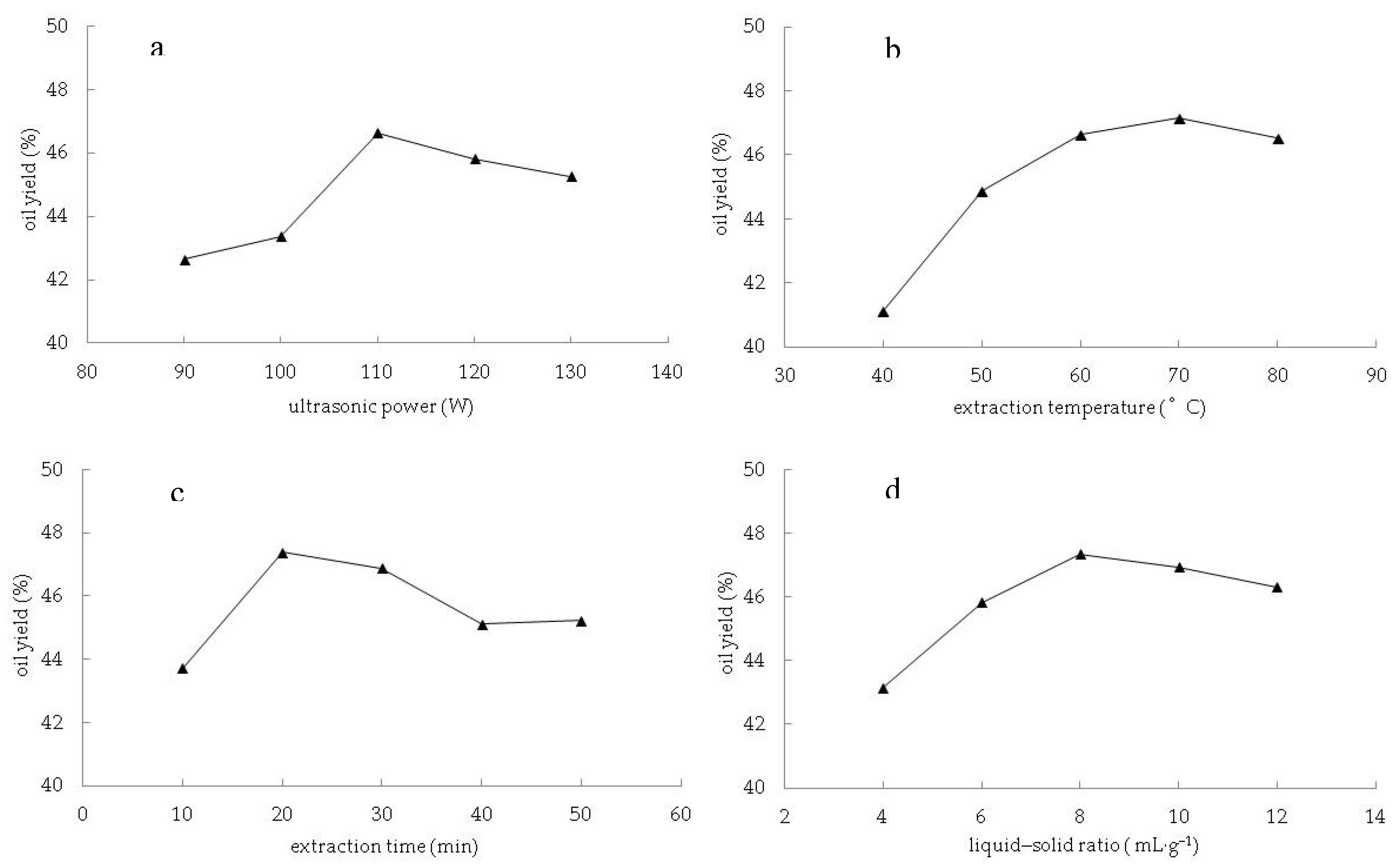
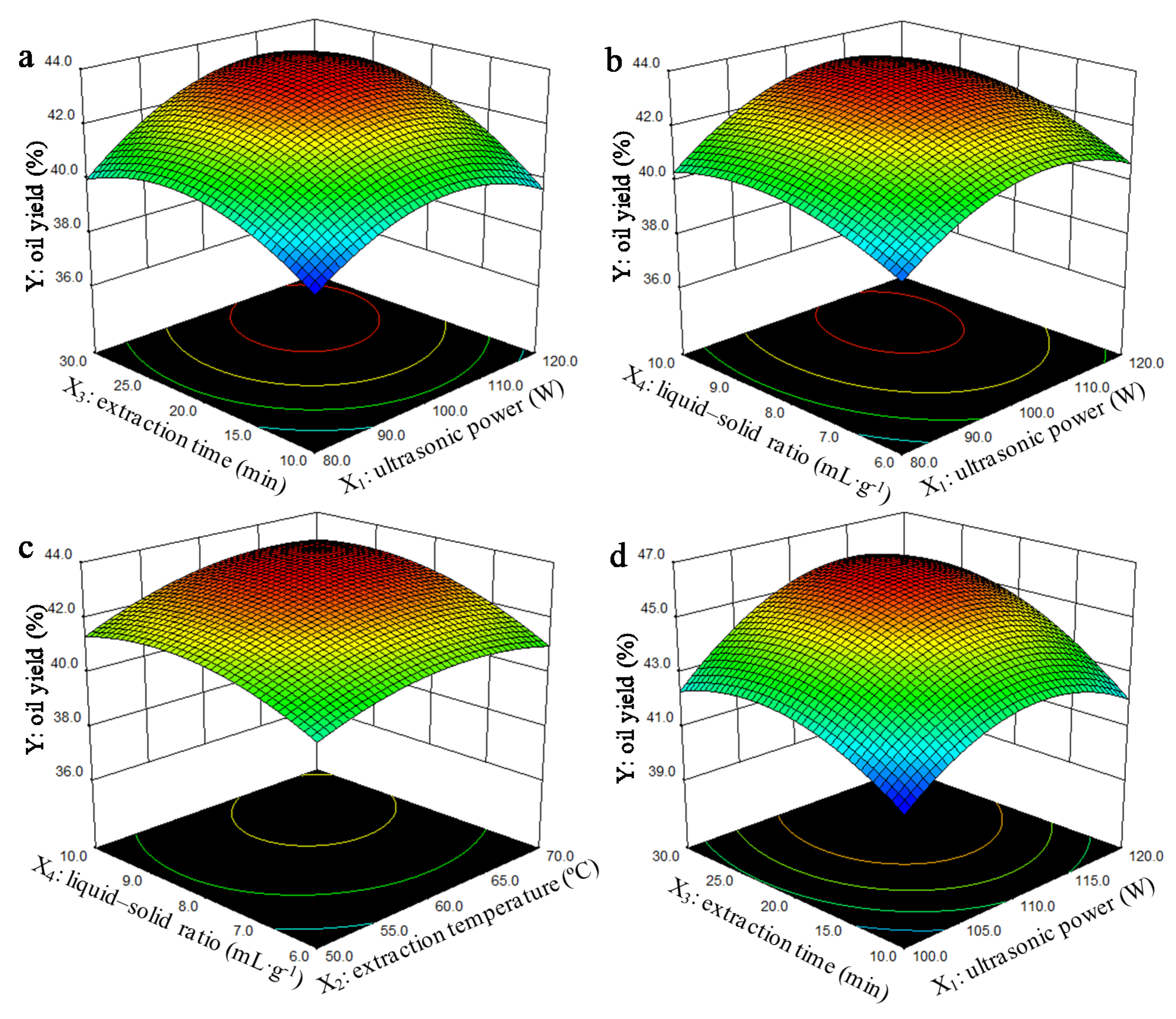
3.1. Parameters Optimization for Oil Extraction by Response Surface Methodology
3.1.1. Model Fitting and Analysis of Variance
3.1.2. Influence of the Parameters on the Oil Yield and Verification of Optimum Conditions
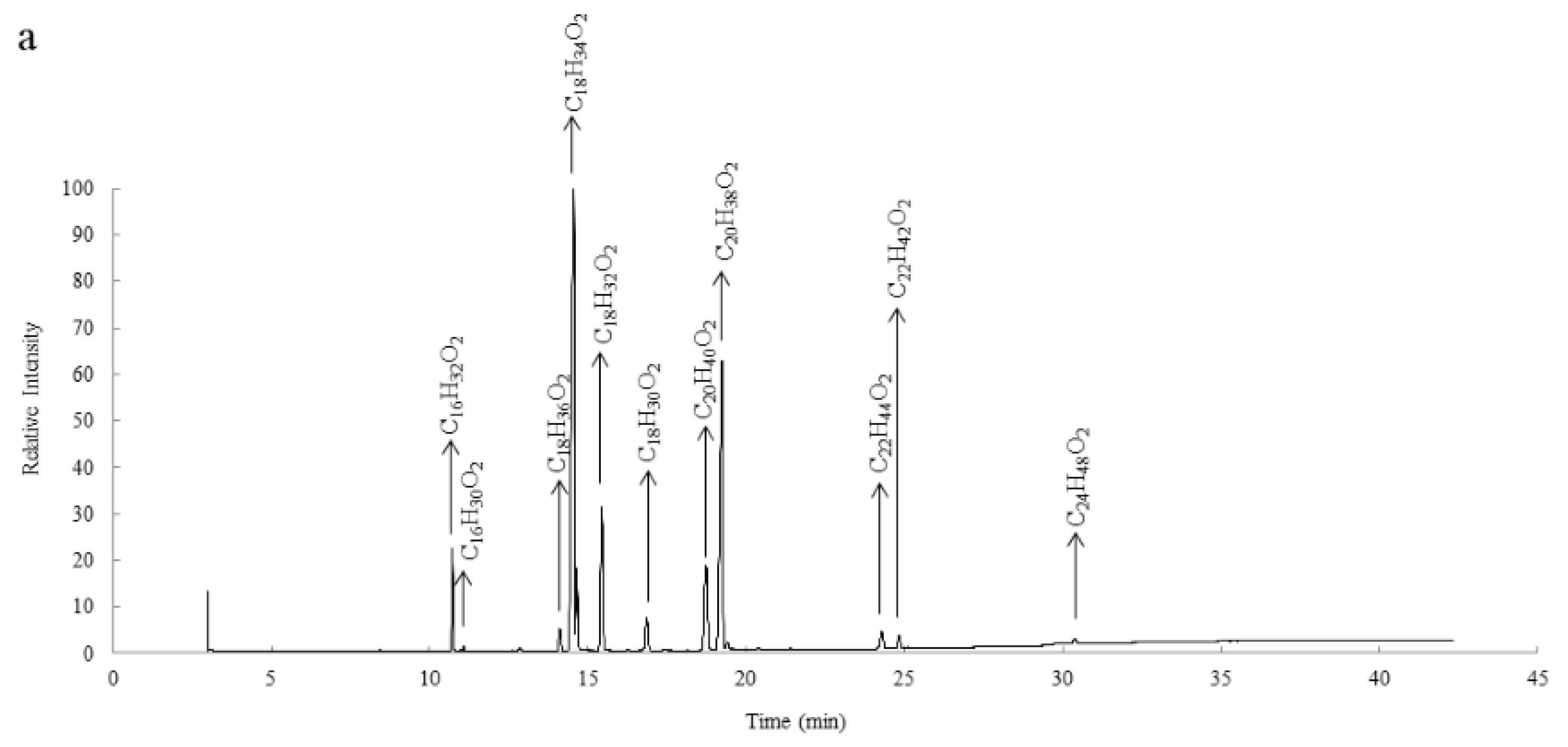
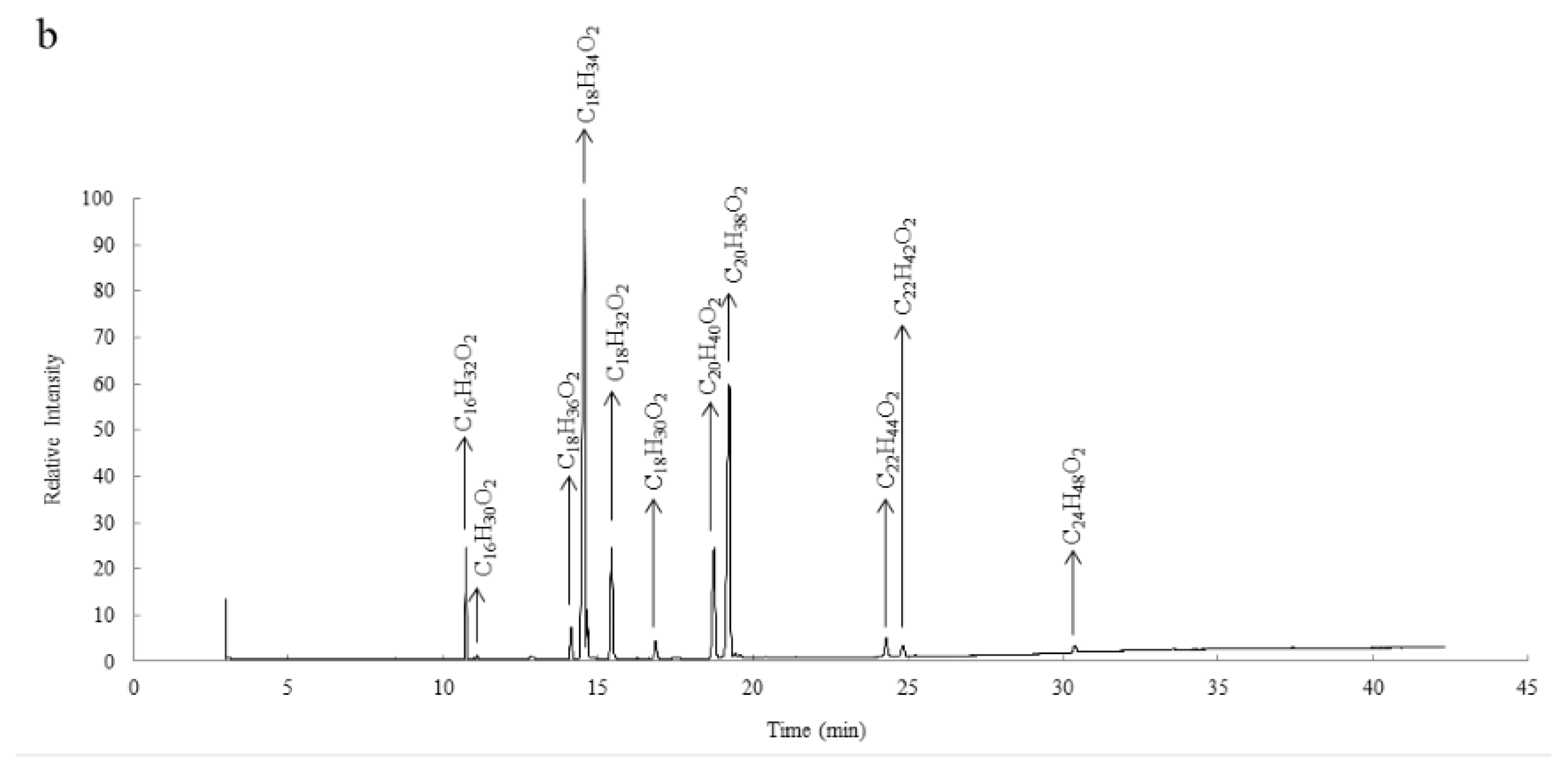
3.2. Fatty Acids Composition of S. delavayi and S. mukorossi Kernel Oils
3.3. Fuel Properties
4. Discussion
4.1. Effect of Process Parameters and Process Optimization
4.2. Effects of Seed oil Components on the Biodiesel Properties
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| B30 | 30% by volume of biodiesel in petrodiesel |
| SD | steam distillation |
| MP | mechanical press |
| SE | solvent extraction |
| EE | enzymatic extraction |
| SFE | supercritical fluid extraction |
| MAE | microwave-assisted extraction |
| UAE | ultrasound-assisted extraction |
| CPE | cold-pressing extraction |
| SXE | Soxhlet extraction |
| RSM | response surface methodology |
| BBD | Box-Behnken design |
| GC-MS | gas chromatography-mass spectrometer |
| EI | electron ionization |
| SD | S. delavayi |
| SM | S. mukorossi |
| SDMEs | S. delavayi methyl esters |
| SMMEs | S. mukorossi methyl esters |
| ASTM | American Society for Testing and Materials |
| EU | European Union |
| ISSR | inter-simple sequence repeat |
| KV | Kinematic viscosity |
| CV | calorific value |
| CFPP | cold filter plugging point |
| CN | cetane number |
| OS | oxidation stability |
| FP | Flash point |
Appendix A
References
- Xia, N.H.; Gadek, P.A. Sapindaceae. In Flora of China; Wu, Z.Y., Raven, P.H., Eds.; Science Press: Beijing, China, 2007; Missouri Botanical Garden Press: St. Louis, MO, USA, 2007; Volume 12, pp. 11–12. [Google Scholar]
- SFA. National Forestry Biomass Energy Development Plan for 2011–2020; The State Forestry Administration of the People’s Republic of China: Beijing, China, 2013.
- Maity, J.P.; Huang, Y.M.; Fan, C.W.; Chen, C.C.; Li, C.Y.; Hsu, C.M.; Chang, Y.F.; Wu, C.I.; Chen, C.Y.; Jean, J.S. Evaluation of remediation process with soapberry derived saponin for removal of heavy metals from contaminated soils in Hai-Pu, Taiwan. J. Environ. Sci. 2013, 25, 1180–1185. [Google Scholar] [CrossRef]
- Upadhyay, A.; Singh, D.K. Molluscicidal activity of Sapindus mukorossi, and Terminalia chebula, against the freshwater snail Lymnaea acuminata. Chemosphere 2011, 83, 468–474. [Google Scholar] [CrossRef]
- Singh, R.; Kumari, N. Comparative determination of phytochemicals and antioxidant activity from leaf and fruit of Sapindus mukorrossi Gaertn-Avaluable medicinal tree. Ind. Crop. Prod. 2015, 73, 1–8. [Google Scholar] [CrossRef]
- Ogunkunle, O.; Ahmed, N.A. A review of global current scenario of biodiesel adoption and combustion in vehicular diesel engines. Energy Rep. 2018, 5, 1560–1579. [Google Scholar] [CrossRef]
- Demirbas, A. Importance of biodiesel as transportation fuel. Energy Policy 2007, 35, 4661–4670. [Google Scholar] [CrossRef]
- Mahlia, T.M.I.; Syazmi, Z.A.H.S.; Mofjur, M.; Abas, A.E.P.; Bilad, M.R.; Ong, H.C.; Silitonga, A.S. Patent landscape review on biodiesel production: Technology updates. Renew. Sust. Energy Rev. 2020, 118, 1601–1620. [Google Scholar] [CrossRef]
- Yesilyurt, M.K.; Cesur, C.; Aslan, V.; Yilbasi, Z. The production of biodiesel from safflower (Carthamus tinctorius L.) oil as a potential feedstock and its usage in compression ignition engine: A comprehensive review. Renew. Sust. Energy Rev. 2020, in press. [Google Scholar] [CrossRef]
- Ambat, I.; Srivastava, V.; Sillanpää, M. Recent advancement in biodiesel production methodologies using various feedstock: A review. Renew. Sust. Energy Rev. 2018, 90, 356–369. [Google Scholar] [CrossRef]
- Karmakar, B.; Halder, G. Progress and future of biodiesel synthesis: Advancements in oil extraction and conversion technologies. Energy Conv. Manag. 2019, 182, 307–339. [Google Scholar] [CrossRef]
- Yusoff, M.M.; Gordon, M.H.; Niranjan, K. Aqueous enzyme assisted oil extraction from oilseeds and emulsion de-Emulsifying methods: A review. Trends Food Sci. Technol. 2015, 41, 60–82. [Google Scholar] [CrossRef]
- Tan, C.X.; Chong, G.H.; Hamzah, H.; Ghazali, H.M. Comparison of subcritical CO2 and ultrasound-Assisted aqueous methods with the conventional solvent method in the extraction of avocado oil. J. Supercrit. Fluids 2018, 135, 45–51. [Google Scholar] [CrossRef]
- Koubaa, M.; Mhemdi, H.; Barba, F.J.; Roohinejad, S.; Greiner, R.; Vorobiev, E. Oilseed treatment by ultrasounds and microwaves to improve oil yield and quality: An overview. Food Res. Int. 2016, 85, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Carabajal, M.; Teglia, C.M.; Cerutti, S.; Culzoni, M.J.; Goicoechea, H.C. Applications of liquid-phase microextraction procedures to complex samples assisted by response surface methodology for optimization. Microchem J. 2020, 152, 104436. [Google Scholar] [CrossRef]
- Tarley, C.R.T.; Silveira, G.; Santos, W.N.L.D.; Matos, G.D.; Da Silva, E.G.P.; Bezerra, M.A.; Miró, M.; Ferreira, S.L.C. Chemometric tools in electroanalytical chemistry: Methods for optimization based on factorial design and response surface methodology. Microchem. J. 2009, 92, 58–67. [Google Scholar] [CrossRef]
- Yusri, I.M.; Majeed, A.P.P.A.; Mamat, R.; Ghazali, M.F.; Awad, O.I.; Azmi, W.H. A review on the application of response surface method and artificial neural network in engine performance and exhaust emissions characteristics in alternative fuel. Renew. Sust. Energy Rev. 2018, 90, 665–686. [Google Scholar] [CrossRef]
- Zhao, X. Comparison of Sapindus mukorossi Gaertn from different areas in Jiangxi province and study of preparation of biodiesel and other comprehensive utilizations. Master′s Thesis, Jiangxi Agricultural University, Nanchang, China, 2014. [Google Scholar]
- Sun, C.; Jia, L.; Xi, B.; Liu, J.; Wang, L.; Weng, X. Genetic diversity and association analyses of fruit traits with microsatellite ISSRs in Sapindus. J. For. Res. 2019, 30, 193–203. [Google Scholar] [CrossRef]
- Suh, H.K.; Lee, C.S. A review on atomization and exhaust emissions of a biodiesel-Fueled compression ignition engine. Renew. Sust. Energy Rev. 2016, 58, 1601–1620. [Google Scholar] [CrossRef]
- Bhuiya, M.M.K.; Rasul, M.G.; Khan, M.M.K.; Ashwath, N.; Azad, A.K.; Hazrat, M.A. Prospects of 2nd generation biodiesel as a sustainable fuel-part 2: Properties, performance and emission characteristics. Renew. Sust. Energ. Rev. 2016, 55, 1129–1146. [Google Scholar] [CrossRef]
- Anahas, A.M.P.; Muralitharan, G. Characterization of heterocystous cyanobacterial strains for biodiesel production based on fatty acid content analysis and hydrocarbon production. Energy Conv. Manag. 2018, 157, 423–437. [Google Scholar] [CrossRef]
- Tian, Y.; Xu, Z.; Zheng, B.; Lo, M.Y. Optimization of ultrasonic-Assisted extraction of pomegranate (Punica granatum L.) seed oil. Ultrason. Sonochem. 2013, 20, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Senrayan, J.; Venkatachalam, S. Optimization of ultrasound-assisted solvent extraction (UASE) based on oil yield, antioxidant activity and evaluation of fatty acid composition and thermal stability of Coriandrum sativum L. seed oil. Food Sci. Biotechnol. 2019, 28, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Xu, Y.; Gao, X.; Zhu, X.; Du, M.; Wang, Y.; Deng, R.; Gao, J. Optimization of ultrasonic-assisted extraction of oil from the seed kernels and isolation of monoterpene glycosides from the oil residue of Paeonia lactiflora Pall. Ind. Crop. Prod. 2017, 107, 260–270. [Google Scholar] [CrossRef]
- Hu, B.; Li, C.; Qin, W.; Zhang, Z.; Liu, Y.; Zhang, Q.; Liu, A.; Jia, R.; Yin, Z.; Han, X.; et al. A method for extracting oil from tea (Camelia sinensis) seed by microwave in combination with ultrasonic and evaluation of its quality. Ind. Crop. Prod. 2019, 131, 234–242. [Google Scholar] [CrossRef]
- Ara, K.M.; Karami, M.; Raofie, F. Application of response surface methodology for the optimization of supercritical carbon dioxide extraction and ultrasound-assisted extraction of Capparis spinosa seed oil. J. Supercrit. Fluids 2014, 85, 173–182. [Google Scholar] [CrossRef]
- Maran, J.P.; Manikandan, S.; Nivetha, C.V.; Dinesh, R. Ultrasound assisted extraction of bioactive compounds from Nephelium lappaceum L. fruit peel using central composite face centered response surface design. Arab. J. Chem. 2017, 10, S1145–S1157. [Google Scholar]
- Xu, G.; Liang, C.; Huang, P.; Liu, Q.; Xu, Y.; Ding, C.; Li, T. Optimization of rice lipid production from ultrasound-assisted extraction by response surface methodology. J. Cereal Sci. 2016, 70, 23–28. [Google Scholar] [CrossRef]
- Almeida, B.; Valli, E.; Bendini, A.; Toschi, T.G. Semi-industrial ultrasound-assisted virgin olive oil extraction: Impact on quality. Eur. J. Lipid Sci. Technol. 2017, 119, 1600230. [Google Scholar] [CrossRef]
- Mohammadpour, H.; Sadrameli, S.M.; Eslami, F.; Asoodeh, A. Optimization of ultrasound-Assisted extraction of Moringa peregrina oil with response surface methodology and comparison with Soxhlet method. Ind. Crop. Prod. 2019, 131, 106–116. [Google Scholar] [CrossRef]
- Górnaś, P.; Siger, A.; Rudzińska, M.; Grygier, A.; Marszałkiewicz, S.; Ying, Q.; Sobieszczańska, N.; Segliņa, D. Impact of the Extraction Technique and Genotype on the Oil Yield and Composition of Lipophilic Compounds in the Oil Recovered from Japanese Quince (Chaenomeles japonica) Seeds. Eur. J. Lipid Sci. Technol. 2019, 121, 1800262. [Google Scholar] [CrossRef]
- Chhetri, A.B.; Tango, M.S.; Budge, S.M.; Watts, K.C.; Islam, M.R. Non-Edible plant oils as new sources for biodiesel production. Int. J. Mol. Sci. 2008, 9, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Wu, H.; Weng, Z.; Wang, H.; Yao, W. Characteristics and extraction of Sapindus mukorossi Gaertn. kernel oil with microwave pretreatment. China Oils Fats 2013, 38, 1–3. [Google Scholar]
- Sun, C.; Wang, J.; Duan, J.; Zhao, G.; Weng, X.; Jia, L. Association of fruit and seed traits of Sapindus mukorossi Germplasm with environmental factors in southern china. Forests 2017, 8, 491. [Google Scholar] [CrossRef]
- Liu, Z.; Gui, M.; Xu, T.; Zhang, L.; Kong, L.; Qin, L.; Zou, Z. Efcient aqueous enzymatic-Ultrasonication extraction of oil from Sapindus mukorossi seed kernels. Ind. Crop. Prod. 2019, 134, 124–133. [Google Scholar] [CrossRef]
- Yaşar, F. Comparision of fuel properties of biodiesel fuels produced from different oils to determine the most suitable feedstock type. Fuel 2020, 264, 116817. [Google Scholar] [CrossRef]
- Refaat, A.A. Correlation between the chemical structure of biodiesel and its physical properties. Int. J. Environ. Sci. Technol. 2009, 6, 677–694. [Google Scholar] [CrossRef]
- Ramírez-Verduzco, L.F.; Rodríguez-Rodríguez, J.E.; Jaramillo-Jacob, A.D.R. Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 2012, 91, 102–111. [Google Scholar] [CrossRef]
- Sajjadi, B.; Raman, A.A.A.; Arandiyan, H. A comprehensive review on properties of edible and non-Edible vegetable oil-Based biodiesel: Composition, specifications and prediction models. Renew. Sust. Energ. Rev. 2016, 63, 62–92. [Google Scholar] [CrossRef]
- Singh, D.; Sharma, D.; Soni, S.L.; Sharma, S.; Kumari, D. Chemical compositions, properties, and standards for different generation biodiesels: A review. Fuel 2019, 253, 60–71. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Jia, L.M.; Chen, Z.G.; Zhao, X.M.; Su, S.C. Development of new non-Food woody oil-Plant resources and comparative analysis of biodiesel properties. China Oils Fats 2016, 41, 1–8. [Google Scholar]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sust. Energ. Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Knothe, G.; Steidley, K.R. Kinematic viscosity of biodiesel fuel components and related compounds. Influence of compound structure and comparison to petrodiesel fuel components. Fuel 2005, 84, 1059–1065. [Google Scholar]
- Kumar, N.; Chauhan, S.R. Performance and emission characteristics of biodiesel from different origins: A review. Renew. Sust. Energy Rev. 2013, 21, 633–658. [Google Scholar]
- Dunn, R.O. Cold flow properties of biodiesel: A guide to getting an accurate analysis. Biofuels 2015, 6, 115–128. [Google Scholar] [CrossRef]
- Kumar, N. Oxidative stability of biodiesel: Causes, effects and prevention. Fuel 2017, 190, 328–350. [Google Scholar] [CrossRef]
- Sierra-Cantor, J.F.; Guerrero-Fajardo, C.A. Methods for improving the cold flow properties of biodiesel with high saturated fatty acids content: A review. Renew. Sustain. Energy Rev. 2017, 72, 774–790. [Google Scholar] [CrossRef]
| Fruit Traits | S. delavayi | S. mukorossi |
|---|---|---|
| 100-fruit dry weight (g) | 431.878 ± 44.538 | 406.070 ± 28.940 |
| 100-pericarp dry weight (g) | 252.089 ± 39.684 | 245.275 ± 26.143 |
| 100-seed dry weight (g) | 179.789 ± 15.984 | 160.795 ± 16.004 |
| 100-seed shell dry weight (g) | 120.211 ± 12.688 | 116.683 ± 6.543 |
| 100-seed kernel dry weight (g) | 59.578 ± 6.595 | 44.112 ± 9.812 |
| Seed content of fruit (%) | 41.630 ± 5.100 | 39.598 ± 4.873 |
| Kernel content of seed (%) | 33.138 ± 3.504 | 24.626 ± 4.570 |
| Fruit horizontal diameter (mm) | 20.830 ± 0.891 | 21.898 ± 1.520 |
| Fruit vertical diameter (mm) | 19.537 ± 0.587 | 19.348 ± 1.274 |
| Fruit lateral diameter (mm) | 23.142 ± 4.037 | 18.435 ± 1.134 |
| Pericarp thickness (mm) | 1.757 ± 0.164 | 1.703 ± 0.303 |
| Runs | S. delavayi | S. mukorossi | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| X1 (W) | X2 (°C) | X3 (min) | X4 (mL·g−1) | Y1 (%) | Y2 (%) | X1 (W) | X2 (°C) | X3 (min) | X4 (mL·g−1) | Y1 (%) | Y2 (%) | |
| 1 | 80 (−1) | 50 (−1) | 20 (0) | 8 (0) | 39.37 | 39.46 | 100 (−1) | 60 (−1) | 20 (0) | 8 (0) | 43.51 | 44.08 |
| 2 | 120 (1) | 50 (−1) | 20 (0) | 8 (0) | 41.28 | 41.11 | 120 (1) | 60 (−1) | 20 (0) | 8 (0) | 46.56 | 46.17 |
| 3 | 80 (−1) | 70 (1) | 20 (0) | 8 (0) | 40.46 | 40.46 | 100 (−1) | 80 (1) | 20 (0) | 8 (0) | 43.31 | 43.67 |
| 4 | 120 (1) | 70 (1) | 20 (0) | 8 (0) | 42.60 | 42.33 | 120 (1) | 80 (1) | 20 (0) | 8 (0) | 46.34 | 45.74 |
| 5 | 100 (0) | 60 (0) | 10 (−1) | 6 (−1) | 39.30 | 39.13 | 110 (0) | 70 (0) | 10 (−1) | 6 (−1) | 43.23 | 43.13 |
| 6 | 100 (0) | 60 (0) | 30 (1) | 6 (−1) | 41.33 | 41.13 | 110 (0) | 70 (0) | 30 (1) | 6 (−1) | 45.46 | 45.28 |
| 7 | 100 (0) | 60 (0) | 10 (−1) | 10 (1) | 40.43 | 40.46 | 110 (0) | 70 (0) | 10 (−1) | 10 (1) | 44.37 | 44.52 |
| 8 | 100 (0) | 60 (0) | 30 (1) | 10 (1) | 42.71 | 42.71 | 110 (0) | 70 (0) | 30 (1) | 10 (1) | 47.10 | 47.16 |
| 9 | 80 (−1) | 60 (0) | 20 (0) | 6 (−1) | 39.26 | 39.31 | 100 (−1) | 70 (0) | 20 (0) | 6 (−1) | 43.19 | 42.95 |
| 10 | 120 (1) | 60 (0) | 20 (0) | 6 (−1) | 40.63 | 40.67 | 120 (1) | 70 (0) | 20 (0) | 6 (−1) | 44.42 | 44.63 |
| 11 | 80 (−1) | 60 (0) | 20 (0) | 10 (1) | 40.38 | 40.36 | 100 (−1) | 70 (0) | 20 (0) | 10 (1) | 44.69 | 44.19 |
| 12 | 120 (1) | 60 (0) | 20 (0) | 10 (1) | 42.55 | 42.52 | 120 (1) | 70 (0) | 20 (0) | 10 (1) | 46.71 | 46.66 |
| 13 | 100 (0) | 50 (−1) | 10 (−1) | 8 (0) | 39.34 | 39.37 | 110 (0) | 60 (−1) | 10 (−1) | 8 (0) | 44.32 | 44.28 |
| 14 | 100 (0) | 70 (1) | 10 (−1) | 8 (0) | 40.56 | 40.47 | 110 (0) | 80 (1) | 10 (−1) | 8 (0) | 44.27 | 43.97 |
| 15 | 100 (0) | 50 (−1) | 30 (1) | 8 (0) | 41.37 | 41.48 | 110 (0) | 60 (−1) | 30 (1) | 8 (0) | 46.78 | 46.79 |
| 16 | 100 (0) | 70 (1) | 30 (1) | 8 (0) | 42.62 | 42.61 | 110 (0) | 80 (1) | 30 (1) | 8 (0) | 46.51 | 46.26 |
| 17 | 80 (−1) | 60 (0) | 10 (−1) | 8 (0) | 38.84 | 38.81 | 100 (−1) | 70 (0) | 10 (−1) | 8 (0) | 42.72 | 42.61 |
| 18 | 120 (1) | 60 (0) | 10 (−1) | 8 (0) | 39.41 | 39.65 | 120 (1) | 70 (0) | 10 (−1) | 8 (0) | 43.35 | 43.74 |
| 19 | 80 (−1) | 60 (0) | 30 (1) | 8 (0) | 40.10 | 40.02 | 100 (−1) | 70 (0) | 30 (1) | 8 (0) | 44.11 | 44.05 |
| 20 | 120 (1) | 60 (0) | 30 (1) | 8 (0) | 42.50 | 42.69 | 120 (1) | 70 (0) | 30 (1) | 8 (0) | 46.65 | 47.09 |
| 21 | 100 (0) | 50 (−1) | 20 (0) | 6 (−1) | 40.32 | 40.35 | 110 (0) | 60 (−1) | 20 (0) | 6 (−1) | 44.83 | 44.75 |
| 22 | 100 (0) | 70 (1) | 20 (0) | 6 (−1) | 40.75 | 41.01 | 110 (0) | 80 (1) | 20 (0) | 6 (−1) | 44.35 | 44.74 |
| 23 | 100 (0) | 50 (−1) | 20 (0) | 10 (1) | 41.45 | 41.35 | 110 (0) | 60 (−1) | 20 (0) | 10 (1) | 46.86 | 46.80 |
| 24 | 100 (0) | 70 (1) | 20 (0) | 10 (1) | 42.79 | 42.91 | 110 (0) | 80 (1) | 20 (0) | 10 (1) | 45.56 | 45.97 |
| 25 | 100 (0) | 60 (0) | 20 (0) | 8 (0) | 42.88 | 42.97 | 110 (0) | 70 (0) | 20 (0) | 8 (0) | 47.00 | 47.29 |
| 26 | 100 (0) | 60 (0) | 20 (0) | 8 (0) | 42.96 | 42.97 | 110 (0) | 70 (0) | 20 (0) | 8 (0) | 47.26 | 47.29 |
| 27 | 100 (0) | 60 (0) | 20 (0) | 8 (0) | 43.11 | 42.97 | 110 (0) | 70 (0) | 20 (0) | 8 (0) | 47.42 | 47.29 |
| 28 | 100 (0) | 60 (0) | 20 (0) | 8 (0) | 42.85 | 42.97 | 110 (0) | 70 (0) | 20 (0) | 8 (0) | 47.13 | 47.29 |
| 29 | 100 (0) | 60 (0) | 20 (0) | 8 (0) | 43.03 | 42.97 | 110 (0) | 70 (0) | 20 (0) | 8 (0) | 47.66 | 47.29 |
| S. delavayi | S. mukorossi | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Source | SS | df | MS | F–value | p-Value | Source | SS | df | MS | F–value | p-Value |
| Model | 55.29 | 14 | 3.95 | 123.48 | <0.0001 | Model | 61.30 | 14 | 4.38 | 26.62 | <0.0001 |
| X1 | 9.29 | 1 | 9.29 | 290.55 | <0.0001 | X1 | 12.03 | 1 | 12.03 | 73.14 | <0.0001 |
| X2 | 3.69 | 1 | 3.69 | 115.22 | <0.0001 | X3 | 15.85 | 1 | 15.85 | 96.39 | <0.0001 |
| X3 | 13.55 | 1 | 13.55 | 423.57 | <0.0001 | X4 | 7.41 | 1 | 7.41 | 45.05 | <0.0001 |
| X4 | 6.34 | 1 | 6.34 | 198.12 | <0.0001 | X1X3 | 0.84 | 1 | 0.84 | 5.12 | 0.0400 |
| X1X3 | 0.84 | 1 | 0.84 | 26.18 | 0.0002 | X12 | 16.69 | 1 | 16.69 | 101.45 | <0.0001 |
| X1X4 | 0.16 | 1 | 0.16 | 5.00 | 0.0421 | X22 | 3.05 | 1 | 3.05 | 18.56 | 0.0007 |
| X2X4 | 0.21 | 1 | 0.21 | 6.47 | 0.0234 | X32 | 9.44 | 1 | 9.44 | 57.38 | <0.0001 |
| X12 | 12.87 | 1 | 12.87 | 402.30 | <0.0001 | X42 | 6.20 | 1 | 6.20 | 37.72 | <0.0001 |
| X22 | 3.34 | 1 | 3.34 | 104.31 | <0.0001 | ||||||
| X32 | 10.42 | 1 | 10.42 | 325.66 | <0.0001 | ||||||
| X42 | 4.61 | 1 | 4.61 | 144.27 | <0.0001 | ||||||
| ANOVA | |||||||||||
| Residual | 0.45 | 14 | 0.032 | Residual | 2.30 | 14 | 0.16 | ||||
| Lack of Fit | 0.40 | 10 | 0.040 | 3.52 | 0.1184 | Lack of Fit | 2.06 | 10 | 0.21 | 3.37 | 0.1264 |
| Pure Error | 0.046 | 4 | 0.011 | Pure Error | 0.24 | 4 | 0.061 | ||||
| Total SS | 55.74 | 28 | Total SS | 63.61 | 28 | ||||||
| R2 = 0.9920; Adj R2 = 0.9839; Pred R2 = 0.9572; Adeq Precision = 32.348 | R2 = 0.9638; Adj R2 = 0.9276; Pred R2 = 0.8076; Adeq Precision = 15.434 | ||||||||||
| Fatty Acid | Molecular Formula | Retention Time of SD | S. delavayi (%) | Retention Time of SM | S. mukorossi (%) |
|---|---|---|---|---|---|
| Palmitic acid | C16H32O2 | 10.729 | 4.83 ± 0.17 | 10.732 | 5.32 ± 0.31 |
| Palmitoleic acid | C16H30O2 | 11.086 | 0.28 ± 0.04 | 11.089 | 0.33 ± 0.02 |
| Stearic acid | C18H36O2 | 14.123 | 1.43 ± 0.06 | 14.124 | 1.81 ± 0.17 |
| Oleic acid | C18H34O2 | 14.564 | 52.32 ± 1.28 | 14.560 | 55.21 ± 1.83 |
| Linoleic acid | C18H32O2 | 15.442 | 8.45 ± 0.72 | 15.440 | 6.63 ± 0.54 |
| Linolenic acid | C18H30O2 | 16.861 | 2.66 ± 0.32 | 16.859 | 1.20 ± 0.16 |
| Arachidic acid | C20H40O2 | 18.731 | 6.28 ± 0.29 | 18.744 | 7.15 ± 0.37 |
| Eicosenoic acid | C20H38O2 | 19.237 | 21.45 ± 0.81 | 19.234 | 20.03 ± 1.12 |
| Behenic acid | C22H44O2 | 24.283 | 1.09 ± 0.08 | 24.287 | 1.18 ± 0.11 |
| Erucic acid | C22H42O2 | 24.825 | 0.86 ± 0.17 | 24.832 | 0.75 ± 0.20 |
| Lignoceric acid | C24H48O2 | 30.379 | 0.35 ± 0.07 | 30.380 | 0.39 ± 0.06 |
| saturated fatty acids | ΣC n:0 | — | 13.98 | — | 15.85 |
| monounsaturated fatty acids | ΣC n:1 | — | 74.91 | — | 76.32 |
| polyunsaturated fatty acids | ΣC n:m = 2,3 | — | 11.11 | — | 7.83 |
| S. delavayi | S. mukorossi | ASTMD6751 | EN14214 | GB25199 | |
|---|---|---|---|---|---|
| Density (15 °C; kg·m−3) | 882 | 879 | — 1 | 860~900 | 820~900 (20°C) |
| KV (40 °C; mm2·s−1) | 4.42 | 4.48 | 1.9~6.0 | 3.5~5.0 | 1.9~6.0 |
| CV (MJ·kg−1) | 39.63 | 39.96 | — 2 | — 2 | — 2 |
| CFPP (°C) | 4 | 5 | — 2 | — 1 | — 3 |
| CN | 53 | 54 | ≥47 | ≥51 | ≥49(S50); ≥51(S10) |
| OS (110 °C; h) | 3.6 | 3.9 | ≥3 | ≥8 | ≥6 |
| FP (°C) | 152 | 147 | ≥93 | ≥101 | ≥130 |
| Acid value (mg KOH·g−1) | 0.16 | 0.14 | ≤0.5 | ≤0.5 | ≤0.5 |
| Sulfur content (mg·kg−1) | ND | ND | ≤15(S15); ≤500(S500) | ≤10 | ≤50(S50); ≤10(S10) |
| Sulfated ash content (%mass) | ND | ND | ≤0.02 | ≤0.02 | ≤0.02 |
| Copper strip corrosion (50 °C; 3 h) | 1a | 1a | No. 3 max | No. 1 max | No. 1 max |
| Free glycerol (% mass) | 0.0 | 0.0 | ≤0.02 | ≤0.02 | ≤0.02 |
| Total glycerol (% mass) | 0.1 | 0.1 | ≤0.24 | ≤0.25 | ≤0.24 |
| Water Content (mg·kg−1) | 119 | 102 | ≤500 | ≤500 | ≤500 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.-Q.; Jin, L.; Duan, J.; Zhao, G.-C.; Xu, Y.-Y.; Liu, J.-M.; Jia, L.-M.; Hao, Y.-B.; Su, S.-C. The Assessment of Two Species of Soapberry as Resources for High-Quality Biodiesel Production with an Optimized Method of Ultrasound-Assisted Oil Extraction. Forests 2020, 11, 212. https://doi.org/10.3390/f11020212
Zhang Y-Q, Jin L, Duan J, Zhao G-C, Xu Y-Y, Liu J-M, Jia L-M, Hao Y-B, Su S-C. The Assessment of Two Species of Soapberry as Resources for High-Quality Biodiesel Production with an Optimized Method of Ultrasound-Assisted Oil Extraction. Forests. 2020; 11(2):212. https://doi.org/10.3390/f11020212
Chicago/Turabian StyleZhang, Yun-Qi, Lin Jin, Jie Duan, Guo-Chun Zhao, Yuan-Yuan Xu, Ji-Ming Liu, Li-Ming Jia, Yan-Bin Hao, and Shu-Chai Su. 2020. "The Assessment of Two Species of Soapberry as Resources for High-Quality Biodiesel Production with an Optimized Method of Ultrasound-Assisted Oil Extraction" Forests 11, no. 2: 212. https://doi.org/10.3390/f11020212
APA StyleZhang, Y.-Q., Jin, L., Duan, J., Zhao, G.-C., Xu, Y.-Y., Liu, J.-M., Jia, L.-M., Hao, Y.-B., & Su, S.-C. (2020). The Assessment of Two Species of Soapberry as Resources for High-Quality Biodiesel Production with an Optimized Method of Ultrasound-Assisted Oil Extraction. Forests, 11(2), 212. https://doi.org/10.3390/f11020212







