Effect of Different Soil Treatments with Hydrogel on the Performance of Drought-Sensitive and Tolerant Tree Species in a Semi-Arid Region
Abstract
1. Introduction
2. Materials and Methods
- (1)
- CO (control)
- (2)
- OM (hydrogel and sawdust)
- (3)
- HM (hydrogel and organic fertilizer)
- (4)
- HC (hydrogel and compost)
- (5)
- HK (hydrogel and wheat straw)
- (6)
- HS (hydrogel and subsoil)
- (7)
- HP (hydrogel and subsoil with a cobbles cover)
2.1. Photosynthetic Pigments
2.2. Rubisco
2.3. Nitrogen
2.4. Electrical Conductivity
2.5. Prolin
2.6. Abscisic Acid
2.7. Total Protein
2.8. Zinc
2.9. Relative Water Content
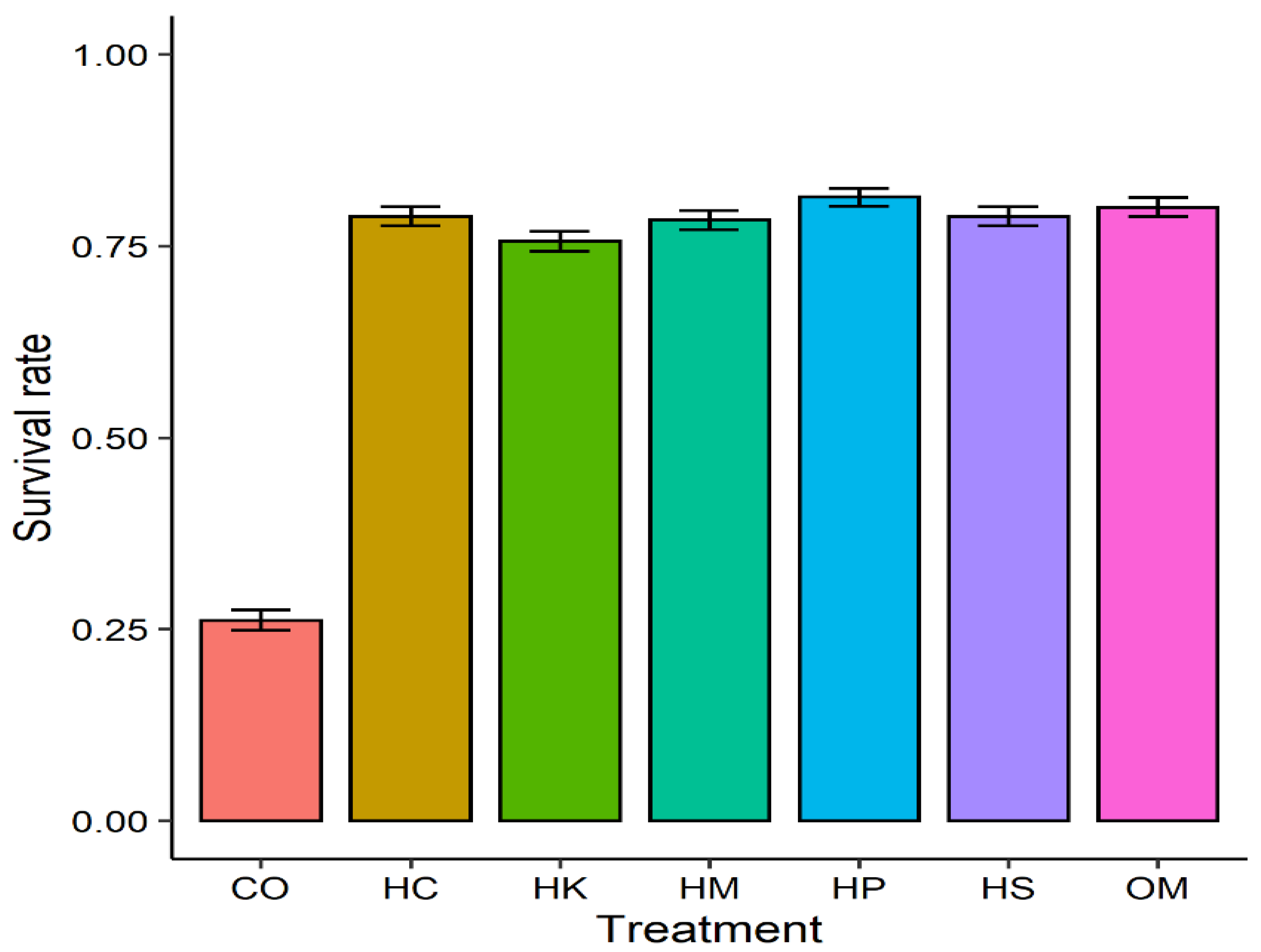
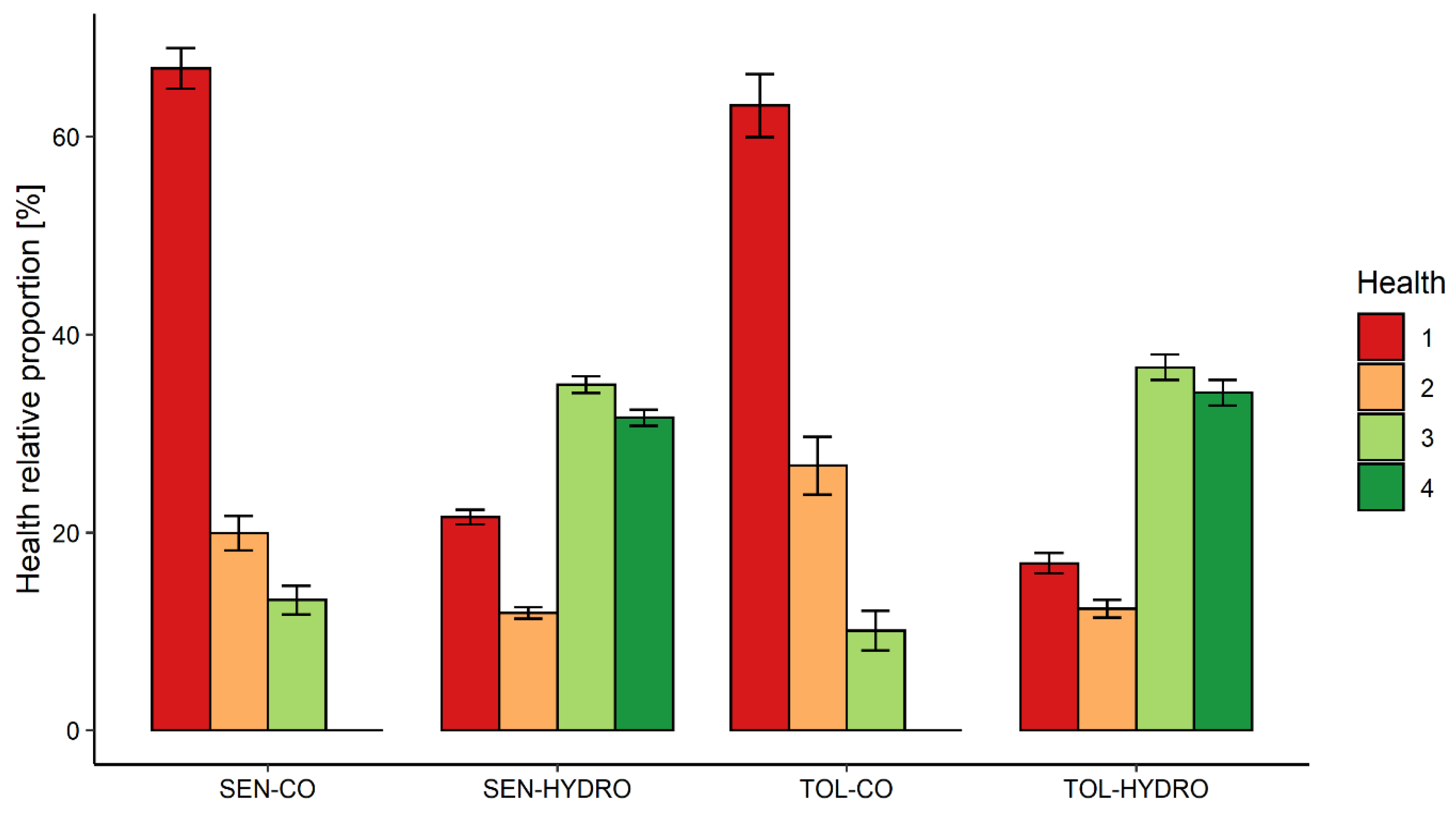
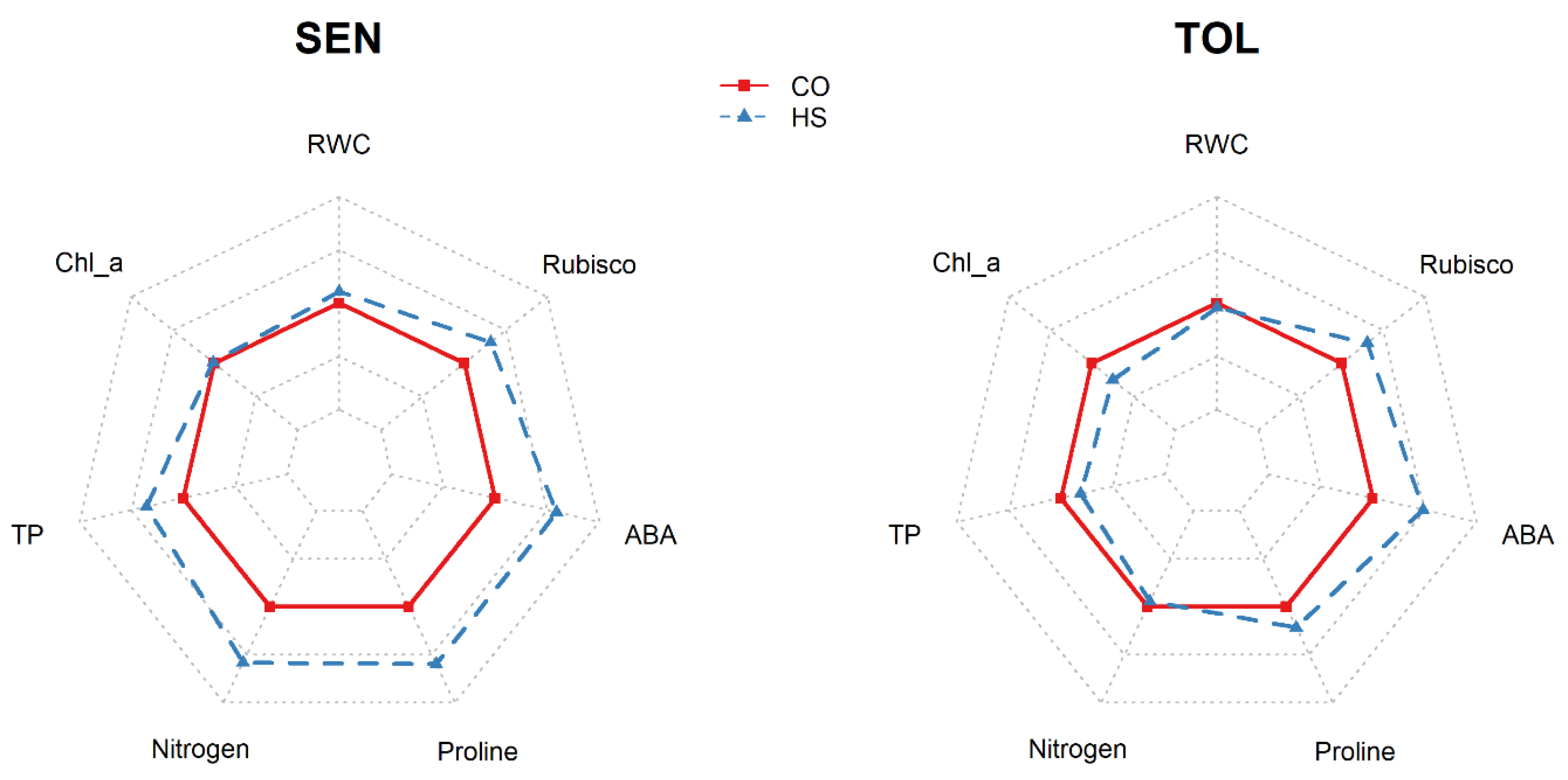
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Species | Treatment | Emmean | SE | df | Lower.CL | Upper.CL |
|---|---|---|---|---|---|---|
| SEN | CO | 2.23 | 0.04505 | 3619 | 2.14 | 2.32 |
| TOL | CO | 1.89 | 0.07821 | 2979 | 1.74 | 2.04 |
| SEN | HYDRO | 2.38 | 0.00983 | 4345 | 2.36 | 2.40 |
| TOL | HYDRO | 2.12 | 0.01857 | 3930 | 2.08 | 2.15 |
| Contrast | Estimate | SE | df | t-Ratio | p-Value |
|---|---|---|---|---|---|
| CO_SEN—CO_TOL | 0.339 | 0.0903 | 3170 | 3.759 | 0.0010 |
| CO_SEN—HYDRO_SEN | −0.150 | 0.0461 | 3735 | −3.254 | 0.0063 |
| CO_SEN—HYDRO_TOL | 0.111 | 0.0487 | 3858 | 2.278 | 0.1034 |
| CO_TOL—HYDRO_SEN | −0.489 | 0.0788 | 3020 | −6.208 | <0.0001 |
| CO_TOL—HYDRO_TOL | −0.228 | 0.0804 | 3081 | −2.840 | 0.0235 |
| HYDRO_SEN—HYDRO_TOL | 0.261 | 0.0210 | 4056 | 12.423 | <0.0001 |
| Treatment | CO | HC | HK | HM | HP | HS |
|---|---|---|---|---|---|---|
| HC | <2 × 10−16 | - | - | - | - | - |
| HK | <2 × 10−16 | 1.00 | - | - | - | - |
| HM | <2 × 10−16 | 1.00 | 1.00 | - | - | - |
| HP | <2 × 10−16 | 1.00 | 0.16 | 1.00 | - | - |
| HS | <2 × 10−16 | 1.00 | 1.00 | 1.00 | 1.00 | - |
| OM | <2 × 10−16 | 1.00 | 0.74 | 1.00 | 1.00 | 1.00 |
| Treatment | Species | Health | Height Increase (cm) | Survival (%) | Osmotic Potential (bar) | Species | Health | Height Increase (cm) | Survival (%) | Osmotic Potential (bar) |
|---|---|---|---|---|---|---|---|---|---|---|
| CO | Acer | 1.3 ± 0.5 | 14.9 ± 7.5 | 23.7 ± 43.1 | 15.3 ± 7.9 | Jugla | 1.3 ± 0.6 | 9.0 ± 3.6 | 21.1 ± 41.3 | 14.0 ± 7.3 |
| HC | Acer | 2.6 ± 1.1 | 26.1 ± 17.5 | 71.1 ± 46.0 | 7.4 ± 6.2 | Jugla | 2.4 ± 1.2 | 15.9 ± 9.5 | 65.8 ± 48.1 | 11.1 ± 2.5 |
| HK | Acer | 2.5 ± 1.0 | 22.4 ± 15.2 | 73.7 ± 44.6 | 13.6 ± 5.6 | Jugla | 2.4 ± 1.1 | 17.0 ± 9.2 | 68.4 ± 47.1 | 10.3 ± 5.3 |
| HM | Acer | 2.3 ± 1.3 | 26.0 ± 21.9 | 57.9 ± 50.0 | 12.8 ± 6.0 | Jugla | 2.3 ± 1.4 | 14.4 ± 11.1 | 50.0 ± 50.7 | 9.9 ± 5.4 |
| HP | Acer | 2.4 ± 1.3 | 18.9 ± 8.9 | 57.9 ± 50.0 | 10.7 ± 5.7 | Jugla | 2.0 ± 1.0 | 14.4 ± 10.0 | 57.9 ± 50.0 | 11.3 ± 6.0 |
| HS | Acer | 2.3 ± 1.2 | 23.0 ± 13.9 | 63.2 ± 48.9 | 14.8 ± 4.5 | Jugla | 2.4 ± 1.1 | 15.3 ± 10.0 | 63.2 ± 48.9 | 13.3 ± 3.7 |
| OM | Acer | 2.5 ± 1.3 | 26.0 ± 21.6 | 60.5 ± 49.5 | 13.7 ± 6.9 | Jugla | 2.4 ± 1.2 | 13.7 ± 9.3 | 68.4 ± 47.1 | 10.8 ± 6.3 |
| CO | Ailan | 1.6 ± 0.8 | 25.8 ± 20.8 | 42.1 ± 50.0 | 20.6 ± 1.7 | Junip | 1.5 ± 0.7 | 6.9 ± 2.8 | 36.8 ± 48.9 | 22.0 ± 0.0 |
| HC | Ailan | 2.6 ± 1.3 | 22.3 ± 9.3 | 78.9 ± 41.3 | 18.3 ± 2.6 | Junip | 3.1 ± 1.0 | 8.8 ± 3.7 | 86.8 ± 34.3 | 21.3 ± 1.0 |
| HK | Ailan | 2.9 ± 1.2 | 36.5 ± 28.2 | 73.7 ± 44.6 | 19.9 ± 1.9 | Junip | 2.7 ± 1.1 | 11.9 ± 6.6 | 73.7 ± 44.6 | 21.4 ± 0.9 |
| HM | Ailan | 2.6 ± 1.2 | 19.0 ± 6.6 | 71.1 ± 46.0 | 19.3 ± 3.4 | Junip | 2.9 ± 1.0 | 8.2 ± 3.8 | 89.5 ± 31.1 | 22.0 ± 0.0 |
| HP | Ailan | 2.8 ± 1.1 | 24.6 ± 12.4 | 81.6 ± 39.3 | 18.8 ± 2.4 | Junip | 2.9 ± 1.0 | 13.2 ± 8.2 | 84.2 ± 37.0 | 18.0 ± 8.9 |
| HS | Ailan | 2.7 ± 1.2 | 21.5 ± 9.6 | 76.3 ± 43.1 | 18.5 ± 2.3 | Junip | 2.6 ± 1.2 | 12.1 ± 6.0 | 71.1 ± 46.0 | 21.6 ± 1.0 |
| OM | Ailan | 2.8 ± 1.1 | 22.2 ± 11.3 | 81.6 ± 39.3 | 18.3 ± 2.6 | Junip | 3.1 ± 1.1 | 9.4 ± 5.5 | 89.5 ± 31.1 | 21.3 ± 1.6 |
| CO | Azadi | 1.6 ± 0.9 | 24.3 ± 15.4 | 35.3 ± 48.5 | 21.3 ± 1.0 | Morus | 1.4 ± 0.7 | 18.9 ± 8.6 | 31.6 ± 47.1 | 19.5 ± 2.4 |
| HC | Azadi | 3.1 ± 1.0 | 17.8 ± 8.6 | 88.2 ± 32.7 | 21.5 ± 1.2 | Morus | 2.9 ± 1.1 | 20.9 ± 10.6 | 81.6 ± 39.3 | 18.4 ± 1.4 |
| HK | Azadi | 2.6 ± 1.2 | 17.9 ± 8.9 | 76.5 ± 43.1 | 19.8 ± 1.8 | Morus | 3.1 ± 0.9 | 26.5 ± 16.7 | 89.5 ± 31.1 | 16.4 ± 3.1 |
| HM | Azadi | 3.2 ± 1.1 | 27.4 ± 24.3 | 85.3 ± 35.9 | 22.0 ± 0.0 | Morus | 3.2 ± 0.8 | 23.5 ± 11.7 | 94.7 ± 22.6 | 20.3 ± 3.6 |
| HP | Azadi | 3.1 ± 1.0 | 23.8 ± 11.1 | 85.3 ± 35.9 | 19.1 ± 2.8 | Morus | 3.1 ± 1.0 | 28.7 ± 22.5 | 89.5 ± 31.1 | 17.8 ± 3.3 |
| HS | Azadi | 2.9 ± 1.1 | 22.0 ± 12.0 | 85.3 ± 35.9 | 18.9 ± 3.4 | Morus | 3.1 ± 1.1 | 25.3 ± 14.1 | 86.8 ± 34.3 | 18.1 ± 3.6 |
| OM | Azadi | 3.0 ± 1.1 | 25.1 ± 17.4 | 82.4 ± 38.7 | 20.0 ± 1.8 | Morus | 3.1 ± 1.2 | 25.2 ± 11.8 | 81.6 ± 39.3 | 18.2 ± 3.0 |
| CO | Berbe | 1.4 ± 0.5 | 21.8 ± 22.7 | 36.8 ± 48.9 | 21.5 ± 1.2 | Olea | 1.8 ± 0.9 | 9.9 ± 9.4 | 52.6 ± 50.6 | 22.0 ± 0.0 |
| HC | Berbe | 2.4 ± 1.2 | 21.1 ± 16.5 | 68.4 ± 47.1 | 21.0 ± 1.5 | Olea | 3.4 ± 0.8 | 11.3 ± 10.7 | 97.4 ± 16.2 | 20.0 ± 2.4 |
| HK | Berbe | 2.4 ± 1.1 | 18.4 ± 14.2 | 71.1 ± 46.0 | 17.8 ± 8.8 | Olea | 3.1 ± 0.9 | 14.4 ± 10.3 | 94.7 ± 22.6 | 21.7 ± 0.5 |
| HM | Berbe | 2.4 ± 1.1 | 22.5 ± 17.9 | 76.3 ± 43.1 | 21.3 ± 1.2 | Olea | 3.4 ± 0.7 | 15.8 ± 13.7 | 100.0 ± 0.0 | 20.3 ± 1.5 |
| HP | Berbe | 2.4 ± 1.1 | 20.3 ± 16.5 | 68.4 ± 47.1 | 21.5 ± 0.8 | Olea | 3.1 ± 0.8 | 13.5 ± 8.1 | 94.7 ± 22.6 | 20.3 ± 2.3 |
| HS | Berbe | 2.3 ± 1.2 | 25.1 ± 21.9 | 71.1 ± 46.0 | 21.8 ± 0.4 | Olea | 3.1 ± 0.9 | 15.1 ± 13.3 | 92.1 ± 27.3 | 21.2 ± 1.0 |
| OM | Berbe | 2.9 ± 1.1 | 24.5 ± 21.0 | 78.9 ± 41.3 | 21.0 ± 1.5 | Olea | 3.3 ± 0.8 | 15.5 ± 12.8 | 97.4 ± 16.2 | 19.2 ± 1.3 |
| CO | Celti | 1.7 ± 0.7 | 15.0 ± 7.7 | 52.6 ± 50.6 | 21.3 ± 1.2 | Peld | 1.4 ± 0.7 | 9.2 ± 5.6 | 31.6 ± 47.1 | 18.0 ± 3.4 |
| HC | Celti | 3.1 ± 0.6 | 18.6 ± 8.8 | 100.0 ± 0.0 | 19.3 ± 2.4 | Peld | 2.7 ± 1.0 | 9.5 ± 4.2 | 81.6 ± 39.3 | 19.7 ± 2.3 |
| HK | Celti | 3.0 ± 0.8 | 16.5 ± 10.1 | 94.7 ± 22.6 | 17.9 ± 4.5 | Peld | 2.5 ± 1.2 | 1.5 ± 4.8 | 68.4 ± 47.1 | 17.0 ± 4.5 |
| HM | Celti | 3.1 ± 0.7 | 19.5 ± 10.7 | 94.7 ± 22.6 | 21.1 ± 1.5 | Peld | 2.6 ± 1.2 | 10.0 ± 4.4 | 65.8 ± 48.1 | 16.2 ± 8.6 |
| HP | Celti | 3.2 ± 0.8 | 16.2 ± 10.6 | 97.4 ± 16.2 | 19.4 ± 2.2 | Peld | 2.4 ± 1.2 | 8.9 ± 3.9 | 68.4 ± 47.1 | 17.5 ± 1.6 |
| HS | Celti | 3.1 ± 0.7 | 17.0 ± 9.7 | 97.4 ± 16.2 | 20.0 ± 1.8 | Peld | 2.5 ± 1.2 | 13.2 ± 7.9 | 63.2 ± 48.9 | 20.0 ± 3.1 |
| OM | Celti | 3.2 ± 0.7 | 16.4 ± 7.0 | 97.4 ± 16.2 | 17.8 ± 4.9 | Peld | 2.6 ± 1.1 | 11.5 ± 6.6 | 73.7 ± 44.6 | 15.8 ± 2.6 |
| CO | Cerci | 1.6 ± 0.7 | 7.3 ± 3.5 | 50.0 ± 50.7 | 22.0 ± 0.0 | Plata | 1.0 ± 0.2 | 5.0 ± 0.8 | 2.6 ± 16.2 | 12.8 ± 10.1 |
| HC | Cerci | 3.3 ± 0.7 | 9.8 ± 6.8 | 100.0 ± 0.0 | 21.3 ± 1.0 | Plata | 2.1 ± 1.3 | 18.4 ± 12.1 | 47.4 ± 50.6 | 14.5 ± 2.5 |
| HK | Cerci | 3.2 ± 0.7 | 11.8 ± 7.6 | 97.4 ± 16.2 | 20.3 ± 2.6 | Plata | 1.8 ± 1.1 | 16.3 ± 11.1 | 39.5 ± 49.5 | 15.3 ± 3.4 |
| HM | Cerci | 3.3 ± 0.8 | 8.8 ± 5.4 | 94.7 ± 22.6 | 21.8 ± 0.4 | Plata | 1.8 ± 1.1 | 20.2 ± 11.3 | 36.8 ± 48.9 | 13.2 ± 2.8 |
| HP | Cerci | 3.1 ± 0.6 | 7.2 ± 3.9 | 94.7 ± 22.6 | 20.5 ± 2.0 | Plata | 1.6 ± 1.1 | 13,9 ± 6.0 | 31.6 ± 47.1 | 13.7 ± 2.7 |
| HS | Cerci | 3.1 ± 0.7 | 10.4 ± 7.5 | 97.4 ± 16.2 | 22.0 ± 0.0 | Plata | 1.7 ± 1.1 | 17.3 ± 5.0 | 31.6 ± 47.1 | 14.8 ± 3.1 |
| OM | Cerci | 3.2 ± 0.8 | 11.8 ± 10.9 | 92.1 ± 27.3 | 21.5 ± 1.2 | Plata | 1.7 ± 1.0 | 16.0 ± 6,4 | 34.2 ± 48.1 | 10.5 ± 5.6 |
| CO | Cupre | 1.6 ± 0.8 | 10.4 ± 6.5 | 47.4 ± 50.6 | 22.0 ± 0.0 | Platy | 1.6 ± 0.9 | 10.1 ± 4.9 | 34.2 ± 48.1 | 21.5 ± 1.2 |
| HC | Cupre | 2.9 ± 1.0 | 12.8 ± 6.7 | 81.6 ± 39.3 | 18.5 ± 4.6 | Platy | 3.1 ± 1.0 | 9.7 ± 5.5 | 84.2 ± 37.0 | 21.0 ± 2.4 |
| HK | Cupre | 2.9 ± 1.0 | 13.2 ± 8.2 | 89.5 ± 31.1 | 21.8 ± 0.4 | Platy | 3.1 ± 1.0 | 9.0 ± 6.2 | 81.6 ± 39.3 | 22.0 ± 0.0 |
| HM | Cupre | 2.9 ± 1.0 | 11.5 ± 5.0 | 86.8 ± 34.3 | 22.0 ± 0.0 | Platy | 3.1 ± 1.1 | 11.3 ± 4.7 | 86.8 ± 34.3 | 21.3 ± 1.0 |
| HP | Cupre | 2.7 ± 1.0 | 9.9 ± 5.0 | 84.2 ± 37.0 | 20.0 ± 3.2 | Platy | 3.3 ± 0.9 | 9.7 ± 3.8 | 86.8 ± 34.3 | 20.7 ± 2.4 |
| HS | Cupre | 2.9 ± 0.9 | 11.2 ± 6.0 | 89.5 ± 31.1 | 20.9 ± 1.2 | Platy | 3.0 ± 1.1 | 9.4 ± 5.0 | 81.6 ± 39.3 | 20.8 ± 1.8 |
| OM | Cupre | 3.1 ± 1.0 | 15.5 ± 9.5 | 89.5 ± 31.1 | 21.3 ± 1.0 | Platy | 3.3 ± 0.9 | 12.8 ± 8.4 | 94.7 ± 22.6 | 20.0 ± 3-3 |
| CO | Elaea | 1.4 ± 0.6 | 12.4 ± 5.5 | 36.8 ± 48.9 | 21.7 ± 0.8 | Pnig | 1.4 ± 0.6 | 11.1 ± 6.0 | 34.2 ± 48.1 | 21.2 ± 1.3 |
| HC | Elaea | 2.9 ± 1.0 | 17.7 ± 14.2 | 86.8 ± 34.3 | 19.6 ± 1.4 | Pnig | 2.9 ± 1.1 | 13 ± 9.0 | 14.0 ± 10.3 | 21.6 ± 3.2 |
| HK | Elaea | 3.0 ± 0.9 | 21.6 ± 18.5 | 89.5 ± 31.1 | 21.7 ± 0.8 | Pnig | 2.9 ± 1.1 | 13 ± 9.0 | 81.6 ± 39.3 | 15.0 ± 2.1 |
| HM | Elaea | 3.1 ± 0.9 | 16.6 ± 10.0 | 92.1 ± 27.3 | 21.4 ± 0.9 | Pnig | 3.2 ± 1.0 | 14.5 ± 8.8 | 86.8 ± 34.3 | 16.4 ± 5.4 |
| HP | Elaea | 2.9 ± 0.9 | 23.4 ± 17.4 | 92.1 ± 27.3 | 20.5 ± 2.1 | Pnig | 3.0 ± 1.1 | 13.4 ± 10 | 81.6 ± 39.3 | 18.6 ± 3.4 |
| HS | Elaea | 3.0 ± 0.9 | 16.5 ± 12.0 | 94.7 ± 22.6 | 20.7 ± 2.8 | Pnig | 3.4 ± 0.9 | 14.7 ± 9.7 | 92.1 ± 27.3 | 16.1 ± 5.2 |
| OM | Elaea | 2.9 ± 1.0 | 16.8 ± 11.3 | 81.6 ± 39.3 | 21.1 ± 0.8 | Pnig | 3.5 ± 0.7 | 14.5 ± 9.2 | 97.4 ± 16.2 | 12.3 ± 6.4 |
| CO | Eryt | 1.9 ± 0.9 | 22.8 ± 17.7 | 55.3 ± 50.4 | 20.8 ± 2.9 | Popul | 1.2 ± 0.5 | 31.4 ± 49.7 | 13.2 ± 34.3 | 14.1 ± 7.2 |
| HC | Eryt | 3.3 ± 1.0 | 24.9 ± 12.6 | 97.4 ± 16.2 | 21.7 ± 0.8 | Popul | 2.0 ± 1.1 | 43.4 ± 44.6 | 50.0 ± 50.7 | 19.0 ± 1.9 |
| HK | Eryt | 3.1 ± 1.0 | 25.7 ± 16.2 | 94.7 ± 22.6 | 22.0 ± 0.0 | Popul | 2.2 ± 1.1 | 33.3 ± 24.2 | 63.2 ± 48.9 | 19.0 ± 1.3 |
| HM | Eryt | 3.2 ± 1.2 | 29.4 ± 22.3 | 94.7 ± 22.6 | 21.2 ± 1.0 | Popul | 2.2 ± 1.1 | 31.3 ± 31.2 | 65.8 ± 48.1 | 13.5 ± 7.2 |
| HP | Eryt | 3.2 ± 1.0 | 23.9 ± 13.7 | 97.4 ± 16.2 | 21.3 ± 1.6 | Popul | 2.4 ± 1.1 | 34.7 ± 31.9 | 73.7 ± 44.6 | 15.0 ± 7.8 |
| HS | Eryt | 2.9 ± 1.1 | 26.3 ± 16.5 | 97.4 ± 16.2 | 21.7 ± 0.8 | Popul | 2.1 ± 1.1 | 32.2 ± 22.0 | 55.3 ± 50.4 | 16.9 ± 1.1 |
| OM | Eryt | 3.4 ± 1.1 | 28.0 ± 16.0 | 94.7 ± 22.6 | 22.0 ± 0.0 | Popul | 2.1 ± 1.2 | 27.7 ± 23.8 | 47.4 ± 50.6 | 12.3 ± 6.4 |
| CO | Fraxi | 1.3 ± 0.5 | 20.4 ± 13.5 | 28.9 ± 46.0 | 21.5 ± 1.2 | Robin | 12.3 ± 6.4 | 21.4 ± 16.9 | 28.9 ± 46.0 | 21.7 ± 0.5 |
| HC | Fraxi | 2.7 ± 1.1 | 18.9 ± 12.9 | 81.6 ± 39.3 | 18.2 ± 8.9 | Robin | 3.0 ± 1.0 | 25.5 ± 17.7 | 84.2 ± 37.0 | 21.5 ± 1.2 |
| HK | Fraxi | 2.9 ± 1.0 | 17.1 ± 11.9 | 89.5 ± 31.1 | 19.9 ± 2.8 | Robin | 3.0 ± 1.1 | 23.1 ± 16.2 | 84.2 ± 37.0 | 20.8 ± 1.7 |
| HM | Fraxi | 2.8 ± 1.1 | 21.2 ± 17.4 | 81.6 ± 39.3 | 21.5 ± 1.2 | Robin | 2.6 ± 1.0 | 18.1 ± 10.7 | 76.3 ± 43.1 | 19.2 ± 3.0 |
| HP | Fraxi | 2.5 ± 0.9 | 28.3 ± 26.3 | 84.2 ± 37.0 | 21.3 ± 1.6 | Robin | 3.1 ± 0.9 | 34.6 ± 37.0 | 94.7 ± 22.6 | 20.0 ± 2.6 |
| HS | Fraxi | 2.7 ± 0.9 | 22.8 ± 26.4 | 84.2 ± 37.0 | 20.7 ± 3.3 | Robin | 3.2 ± 0.7 | 21.7 ± 18.5 | 94.7 ± 22.6 | 19.0 ± 3.9 |
| HS | Fraxi | 3.1 ± 1.0 | 28.2 ± 27.1 | 89.5 ± 31.1 | 20.8 ± 1.3 | Robin | 2.8 ± 1.0 | 23.4 ± 17.0 | 89.5 ± 31.1 | 19.9 ± 1.8 |
| Species | Treatment | RWC (%) | C (chl a) (mg/g FW) | C (chl b) (mg/g FW) | LDW (mg) | Rubisco (Umg/Protein) | Nitrogen (%) | EC (µs/cm) | Prolin (µmol/g FW) | ABA (ppm) | TP (mg/g DW) | Zn (mg/kg DW) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acer negundo | CO | 72.7 ± 3.5 | 3.97 ± 0.28 | 1.20 ± 0.08 | 0.152 ± 0.02 | 89.2 ± 4.3 | 0.184 ± 0.012 | 1712 ± 75 | 1.50 ± 0.11 | 7.83 ± 0.45 | 76.5 ± 5.2 | 25.0 ± 1.4 |
| HS | 76.2 ± 2.3 | 4.24 ± 0.23 | 1.16 ± 0.10 | 0.191 ± 0.01 | 104.3 ± 5.7 | 0.246 ± 0.015 | 1871 ± 77 | 2.05 ± 0.16 | 8.22 ± 0.41 | 90.4 ± 6.8 | 29.5 ± 2.7 | |
| Ailanthus altissima | CO | 41.3 ± 2.6 | 4.96 ± 0.38 | 2.80 ± 0.22 | 0.166 ± 0.01 | 195.1 ± 8.5 | 0.152 ± 0.013 | 1145 ± 68 | 2.60 ± 0.18 | 5.30 ± 0.35 | 71.5 ± 5.2 | 13.5 ± 0.9 |
| HS | 43.1 ± 1.8 | 4.25 ± 0.31 | 2.66 ± 0.21 | 0.128 ± 0.01 | 190.6 ± 8.5 | 0.136 ± 0.010 | 1138 ± 59 | 2.68 ± 0.19 | 5.72 ± 0.39 | 62.3 ± 4.3 | 16.0 ± 0.8 | |
| Azadirachta indica | CO | 45.3 ± 2.7 | 3.18 ± 0.23 | 1.71 ± 0.14 | 0.161 ± 0.02 | 134.7 ± 6.5 | 0.161 ± 0.008 | 1425 ± 66 | 2.68 ± 0.17 | 6.15 ± 0.49 | 145.7 ± 9.12 | 20.0 ± 1.5 |
| HS | 41.9 ± 1.3 | 2.52 ± 0.16 | 1.53 ± 0.08 | 0.120 ± 0.01 | 130.5 ± 6.1 | 0.142 ± 0.010 | 1516 ± 68 | 2.80 ± 0.18 | 7.61 ± 0.29 | 138.2 ± 10.4 | 48.0 ± 3.4 | |
| Berberis vulgaris | CO | 71.2 ± 3.1 | 2.91 ± 0.21 | 2.82 ± 0.13 | 0.188 ± 0.02 | 93.5 ± 4.4 | 0.120 ± 0.010 | 1760 ± 80 | 0.94 ± 0.09 | 4.95 ± 0.31 | 42.9 ± 4.2 | 13.0 ± 0.6 |
| HS | 80.4 ± 3.3 | 3.15 ± 0.17 | 2.98 ± 0.18 | 0.194 ± 0.02 | 98.2 ± 4.9 | 0.112 ± 0.080 | 1715 ± 71 | 1.21 ± 0.08 | 5.30 ± 0.33 | 50.2 ± 5.1 | 21.0 ± 0.7 | |
| Celtis caucasica | CO | 60.4 ± 3.1 | 3.10 ± 0.21 | 2.04 ± 0.18 | 0.227 ± 0.02 | 109.1 ± 7.2 | 0.182 ± 0.013 | 1245 ± 60 | 1.82 ± 0.06 | 5.61 ± 0.32 | 49.5 ± 2.8 | 12.3 ± 0.8 |
| HS | 63.2 ± 2.1 | 2.34 ± 0.14 | 1.61 ± 0.12 | 0.190 ± 0.02 | 103.7 ± 5.5 | 0.187 ± 0.012 | 1260 ± 58 | 2.02 ± 0.14 | 6.18 ± 0.41 | 47.4 ± 2.6 | 14.0 ± 0.5 | |
| Cercis siliquastrum | CO | 55.3 ± 2.7 | 2.62 ± 0.21 | 2.24 ± 0.21 | 0.172 ± 0.01 | 100.2 ± 6.2 | 0.256 ± 0.019 | 1315 ± 66 | 1.50 ± 0.10 | 2.95 ± 0.20 | 60.5 ± 5.1 | 62.0 ± 3.7 |
| HS | 57.4 ± 1.5 | 2.78 ± 0.15 | 2.74 ± 0.17 | 0.193 ± 0.01 | 117.6 ± 6.2 | 0.292 ± 0.017 | 1217 ± 52 | 1.77 ± 0.08 | 3.35 ± 0.24 | 69.3 ± 5.1 | 54.0 ± 4.9 | |
| Cupressus arizonica | CO | 33.7 ± 1.9 | 2.95 ± 0.20 | 2.35 ± 0.20 | 0.228 ± 0.02 | 81.5 ± 6.6 | 0.191 ± 0.016 | 1914 ± 82 | 3.60 ± 0.27 | 4.48 ± 0.39 | 80.4 ± 4.2 | 33.0 ± 1.8 |
| HS | 30.4 ± 1.3 | 2.61 ± 0.16 | 1.81 ± 0.13 | 0.207 ± 0.02 | 77.8 ± 3.5 | 0.162 ± 0.010 | 1812 ± 72 | 3.78 ± 0.27 | 5.12 ± 0.39 | 71.5 ± 5.3 | 14.5 ± 1.9 | |
| Eleagnus angustifolia | CO | 54.2 ± 3.6 | 3.81 ± 0.28 | 1.51 ± 0.14 | 0.241 ± 0.02 | 70.7 ± 3.6 | 0.168 ± 0.012 | 1570 ± 63 | 1.72 ± 0.11 | 3.30 ± 0.25 | 218.3 ± 19.5 | 52.0 ± 2.9 |
| HS | 52.4 ± 1.1 | 3.52 ± 0.28 | 1.15 ± 0.08 | 0.190 ± 0.02 | 66.5 ± 3.4 | 0.177 ± 0.010 | 1690 ± 67 | 1.80 ± 0.14 | 3.46 ± 0.25 | 205.0 ± 12.5 | 51.0 ± 3.2 | |
| Erythrostemon gilliesii | CO | 74.1 ± 3.8 | 3.62 ± 0.25 | 2.12 ± 0.16 | 0.161 ± 0.01 | 81.5 ± 6.2 | 0.164 ± 0.017 | 1325 ± 72 | 2.20 ± 0.12 | 2.96 ± 0.24 | 40.4 ± 3.1 | 81.0 ± 5.9 |
| HS | 74.7 ± 3.0 | 3.42 ± 0.21 | 1.75 ± 0.14 | 0.147 ± 0.01 | 79.1 ± 4.1 | 0.153 ± 0.010 | 1248 ± 53 | 2.63 ± 0.18 | 3.40 ± 0.26 | 36.2 ± 2.8 | 25.5 ± 1.8 | |
| Fraxinus excelsior | CO | 50.5 ± 2.9 | 3.81 ± 0.24 | 2.91 ± 0.22 | 0.155 ± 0.01 | 113.1 ± 5.2 | 0.202 ± 0.014 | 1305 ± 58 | 2.85 ± 0.12 | 1.50 ± 0.08 | 52.3 ± 4.6 | 17.5 ± 0.8 |
| HS | 52.7 ± 1.4 | 3.98 ± 0.27 | 3.27 ± 0.21 | 0.171 ± 0.01 | 118.6 ± 6.3 | 0.226 ± 0.014 | 1174 ± 53 | 3.10 ± 0.24 | 1.83 ± 0.11 | 58.6 ± 4.4 | 26.0 ± 1.7 | |
| Juglans nigra | CO | 59.2 ± 3.7 | 4.72 ± 0.23 | 2.58 ± 0.18 | 0.256 ± 0.02 | 98.6 ± 4.7 | 0.268 ± 0.008 | 1815 ± 81 | 4.12 ± 0.27 | 1.46 ± 0.09 | 139.5 ± 11.4 | 36.0 ± 2.0 |
| HS | 61.1 ± 2.8 | 5.11 ± 0.39 | 2.78 ± 0.20 | 0.296 ± 0.02 | 116.6 ± 6.9 | 0.308 ± 0.022 | 1850 ± 71 | 4.45 ± 0.37 | 1.95 ± 0.09 | 143.1 ± 10.2 | 44.0 ± 2.9 | |
| Juniperus sp. | CO | 28.8 ± 1.3 | 4.03 ± 0.38 | 1.91 ± 0.14 | 0.252 ± 0.01 | 68.8 ± 3.5 | 0.228 ± 0.019 | 1390 ± 53 | 2.95 ± 0.18 | 3.52 ± 0.29 | 100.4 ± 9.2 | 15.0 ± 1.4 |
| HS | 34.5 ± 1.2 | 4.55 ± 0.34 | 2.50 ± 0.16 | 0.285 ± 0.02 | 83.4 ± 4.2 | 0.255 ± 0.013 | 1288 ± 59 | 3.20 ± 0.18 | 3.80 ± 0.25 | 112.4 ± 8.2 | 15.5 ± 0.9 | |
| Morus alba | CO | 67.8 ± 2.5 | 2.92 ± 0.21 | 1.80 ± 0.11 | 0.128 ± 0.01 | 129.4 ± 6.1 | 0.208 ± 0.014 | 1834 ± 83 | 1.12 ± 0.08 | 9.83 ± 0.71 | 37.8 ± 2.9 | 52.0 ± 2.8 |
| HS | 74.8 ± 2.1 | 3.27 ± 0.22 | 2.11 ± 0.14 | 0.159 ± 0.01 | 144.5 ± 7.1 | 0.238 ± 0.016 | 2085 ± 97 | 1.63 ± 0.07 | 10.59 ± 0.46 | 46.8 ± 3.2 | 57.5 ± 2.6 | |
| Olea europea | CO | 41.9 ± 3.2 | 5.21 ± 0.31 | 2.10 ± 0.18 | 0.288 ± 0.02 | 97.6 ± 5.1 | 0.181 ± 0.010 | 1810 ± 77 | 1.35 ± 0.09 | 4.77 ± 0.31 | 128.4 ± 9.4 | 27.0 ± 1.3 |
| HS | 43.2 ± 1.3 | 4.47 ± 0.31 | 1.52 ± 0.10 | 0.240 ± 0.02 | 94.3 ± 5.1 | 0.198 ± 0.011 | 1895 ± 61 | 1.50 ± 0.09 | 5.20 ± 0.31 | 120.1 ± 8.2 | 8.0 ± 0.6 | |
| Pinus eldarica | CO | 31.5 ± 2.0 | 4.61 ± 0.31 | 1.62 ± 0.12 | 0.241 ± 0.02 | 60.3 ± 3.8 | 0.254 ± 0.019 | 1746 ± 63 | 4.80 ± 0.28 | 2.56 ± 0.16 | 96.3 ± 7.2 | 13.5 ± 1.1 |
| HS | 28.1 ± 0.7 | 4.41 ± 0.32 | 1.22 ± 0.10 | 0.181 ± 0.02 | 55.7 ± 2.9 | 0.263 ± 0.016 | 1672 ± 68 | 4.88 ± 0.38 | 2.70 ± 0.16 | 90.5 ± 8.1 | 30.5 ± 2.4 | |
| Pinus nigra | CO | 38.5 ± 1.7 | 4.02 ± 0.31 | 2.37 ± 0.18 | 0.261 ± 0.02 | 109.1 ± 8.2 | 0.261 ± 0.019 | 1845 ± 71 | 5.75 ± 0.32 | 1.98 ± 0.12 | 108.2 ± 9.4 | 10.5 ± 0.9 |
| HS | 34.2 ± 1.1 | 4.18 ± 0.31 | 2.41 ± 0.20 | 0.248 ± 0.02 | 151.5 ± 7.2 | 0.250 ± 0.017 | 1738 ± 60 | 5.78 ± 0.36 | 2.40 ± 0.12 | 111.5 ± 8.4 | 34.5 ± 2.9 | |
| Platanus orientalis | CO | 72.4 ± 3.7 | 3.72 ± 0.29 | 1.86 ± 0.14 | 0.160 ± 0.01 | 169.8 ± 7.5 | 0.216 ± 0.014 | 1220 ± 62 | 1.92 ± 0.15 | 5.42 ± 0.37 | 62.5 ± 4.3 | 22.0 ± 1.2 |
| HS | 78.8 ± 2.4 | 4.08 ± 0.21 | 2.26 ± 0.17 | 0.188 ± 0.01 | 182.0 ± 8.4 | 0.263 ± 0.018 | 1214 ± 52 | 1.88 ± 0.11 | 5.05 ± 0.38 | 77.3 ± 5.6 | 24.0 ± 1.6 | |
| Platycladus orientalis | CO | 36.4 ± 1.4 | 3.93 ± 0.28 | 1.81 ± 0.13 | 0.258 ± 0.02 | 41.4 ± 3.6 | 0.215 ± 0.014 | 1695 ± 75 | 3.86 ± 0.22 | 4.91 ± 0.38 | 120.3 ± 8.8 | 11.0 ± 0.7 |
| HS | 30.5 ± 1.4 | 3.72 ± 0.21 | 1.34 ± 0.12 | 0.208 ± 0.02 | 38.1 ± 4.0 | 0.203 ± 0.014 | 1569 ± 59 | 4.12 ± 0.35 | 5.21 ± 0.37 | 104.3 ± 7.3 | 20.0 ± 1.9 | |
| Populus nigra | CO | 71.5 ± 3.7 | 4.10 ± 0.37 | 1.93 ± 0.17 | 0.182 ± 0.02 | 98.5 ± 4.6 | 0.236 ± 0.019 | 1316 ± 75 | 2.26 ± 0.18 | 1.95 ± 0.16 | 41.2 ± 3.6 | 65.0 ± 6.9 |
| HS | 76.2 ± 2.2 | 3.77 ± 0.26 | 1.82 ± 0.13 | 0.146 ± 0.01 | 96.3 ± 4.4 | 0.221 ± 0.017 | 1171 ± 66 | 2.42 ± 0.19 | 2.30 ± 0.09 | 37.5 ± 2.8 | 182.5 ± 8.7 | |
| Robinia pseudoacacia | CO | 59.6 ± 2.7 | 3.91 ± 0.29 | 3.22 ± 0.19 | 0.195 ± 0.01 | 112.2 ± 5.4 | 0.242 ± 0.019 | 2190 ± 88 | 0.93 ± 0.07 | 2.65 ± 0.19 | 76.3 ± 5.3 | 32.0 ± 3.9 |
| HS | 55.4 ± 2.2 | 4.28 ± 0.26 | 3.42 ± 0.26 | 0.254 ± 0.01 | 120.3 ± 6.2 | 0.277 ± 0.018 | 2307 ± 84 | 1.15 ± 0.09 | 3.25 ± 0.19 | 92.8 ± 7.4 | 42.5 ± 3.6 |
References
- Zohrabi, N.; Bavani, A.M.; Goodarzi, E.; Eslamian, S. Attribution of temperature and precipitation changes to greenhouse gases in northwest Iran. Quat. Int. 2014, 345, 130–137. [Google Scholar] [CrossRef]
- Daneshvar, M.R.M.; Ebrahimi, M.; Nejadsoleymani, H. An overview of climate change in Iran: Facts and statistics. Environ. Syst. Res. 2019, 8, 7. [Google Scholar] [CrossRef]
- Some’e, B.S.; Ezani, A.; Tabari, H. Spatiotemporal trends and change point of precipitation in Iran. Atmos. Res. 2012, 113, 1–12. [Google Scholar] [CrossRef]
- Karimi, N.; Golian, S.; Karimi, D. Monitoring deforestation in Iran, Jangal-Abr Forest using multi-temporal satellite images and spectral mixture analysis method. Arab. J. Geosci. 2016, 9, 214. [Google Scholar] [CrossRef]
- Khalyani, A.; Mayer, A. Spatial and temporal deforestation dynamics of Zagros forests (Iran) from 1972 to 2009. Landsc. Urban Plan. 2013, 117, 1–12. [Google Scholar] [CrossRef]
- Sohrabi, H.; Bakhtiarvand-Bakhtiari, S.; Ahmadi, K. Above-and below-ground biomass and carbon stocks of different tree plantations in central Iran. J. Arid Land 2016, 8, 138–145. [Google Scholar] [CrossRef]
- Azevedo, G.; de Souza, A.M.; de Azevedo, G.B.; Cerqueira, P.H. Minicutting rooting of eucalyptus with different doses of the hydrophylic polymer incorporated into the substrate. Sci. For. 2015, 43, 773–780. [Google Scholar] [CrossRef][Green Version]
- Varela, S.A.; Weigandt, M.N.; Willems, P.; Bianchi, E.; Diez, J.P.; Gyenge, J.E. Physiological status of conifer seedlings treated with radiation, drought and frost stress mitigation techniques: A laboratory assessment. New For. 2016, 47, 87–103. [Google Scholar] [CrossRef]
- Li, J.; Ma, X.; Sa, G.; Zhou, D.; Zheng, X.; Zhou, X.; Lu, C.; Lin, S.; Zhao, R.; Chen, S. Natural and synthetic hydrophilic polymers enhance salt and drought tolerance of Metasequoia glyptostroboides Hu and WC Cheng seedlings. Forests 2018, 9, 643. [Google Scholar] [CrossRef]
- Azevedo, G.; de Azevedo, G.B.; de Souza, A.M.; Mews, C.L.; de Sousa, J.R.L. Effect of hydrogel doses in the quality of Corymbia citriodora Hill & Johnson seedlings. Nativa 2016, 4, 244–248. [Google Scholar] [CrossRef][Green Version]
- Coello, J.; Ameztegui, A.; Rovira, P.; Fuentes, C.; Piqué, M. Innovative soil conditioners and mulches for forest restoration in semiarid conditions in northeast Spain. Ecol. Eng. 2018, 118, 52–65. [Google Scholar] [CrossRef]
- Landis, T.D.; Haase, D.L. Applications of hydrogels in the nursery and during outplanting. In Tech. Coords. National Proceedings: Forest and Conservation Nursery Associations-2011. Proceeding RMRS-P-68; Haase, D.L., Pinto, J.R., Riley, L.E., Eds.; Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2012; Volume 68, pp. 53–58. [Google Scholar]
- Repáč, I.; Vencurik, J.; Balanda, M. Effects of commercial products application on survival, growth and physiological parameters of Norway spruce and European beech plantations. Zpravy Lesn. Vyzk. 2013, 58, 167–175. [Google Scholar]
- Navroski, M.C.; Araújo, M.M.; Reininger, L.R.S.; Muniz, M.F.B.; Pereira, M.O. Doses of hydrogel influencing growth and nutritional content in seedlings Eucalyptus dunnii. Floresta 2015, 45, 315–328. [Google Scholar] [CrossRef]
- Chen, J.H.; Jiang, H.W.; Hsieh, E.J.; Chen, H.Y.; Chien, C.T.; Hsieh, H.L.; Lin, T.P. Drought and salt stress tolerance of an Arabidopsis glutathione S-transferase U17 knockout mutant are attributed to the combined effect of glutathione and abscisic acid. Plant Physiol. 2012, 158, 340–351. [Google Scholar] [CrossRef]
- Konzen, E.R.; Navroski, M.C.; Friederichs, G.; Ferrari, L.H.; Pereira, M.O.; Felippe, D. The use of hydrogel combined with appropriate substrate and fertilizer improve quality and growth performance of Mimosa scabrella Benth. seedlings. Cerne 2017, 23, 473–482. [Google Scholar] [CrossRef]
- Agaba, H.; Baguma Orikiriza, L.J.; Osoto Esegu, J.F.; Obua, J.; Kabasa, J.D.; Hüttermann, A. Effects of hydrogel amendment to different soils on plant available water and survival of trees under drought conditions. Clean Soil Air Water 2010, 38, 328–335. [Google Scholar] [CrossRef]
- Javari, M. Spatial-temporal variability of seasonal precipitation in Iran. Open Atmos. Sci. J. 2016, 10. [Google Scholar] [CrossRef]
- Liu, H.; Gleason, S.M.; Hao, G.; Hua, L.; He, P.; Goldstein, G.; Ye, Q. Hydraulic traits are coordinated with maximum plant height at the global scale. Sci. Adv. 2019, 5, 1332. [Google Scholar] [CrossRef]
- Tilki, F.; Dirik, H. Seed germination of three provenances of Pinus brutia (Ten.) as influenced by stratification, temperature and water stress. J. Environ. Biol. 2007, 28, 133–136. [Google Scholar]
- Froux, F.; Huc, R.; Ducrey, M.; Dreyer, E. Xylem hydraulic efficiency versus vulnerability in seedlings of four contrasting Mediterranean tree species (Cedrus atlantica, Cupressus sempervirens, Pinus halepensis and Pinus nigra). Ann. For. Sci. 2002, 59, 409–418. [Google Scholar] [CrossRef][Green Version]
- Yangyang, L.; Hui, S.; Ming’an, S. Leaf water use efficiency and its relationship with hydraulic characteristics in eight dominant trees and shrubs in Loess Hilly area during vegetation succession. Sci. Silvae Sin. 2010, 46, 67–73. [Google Scholar] [CrossRef]
- Murakami, Y.; Miki, N.H.; Yang, L.; Zhang, G.; Wang, L.H.; Yoshikawa, K. Water transport properties of seven woody species from the semi-arid Mu Us Sandy Land, China. Landsc. Ecol. Eng. 2016, 12, 209–220. [Google Scholar] [CrossRef]
- Wellburn, A.R. The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Barta, C.; Carmo-Silva, A.E.; Salvucci, M.E. Rubisco activase activity assays. Methods Mol. Biol. 2011, 684, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Carillo, P.; Gibon, Y. Protocol: Extraction and determination of proline. PrometheusWiki 2011. Available online: http://prometheuswiki.publish.csiro.au/tiki-index.php?page=Extraction+and+determination+of+proline (accessed on 12 February 2020).
- Okamoto, M.; Hanada, A.; Kamiya, Y.; Yamaguchi, S.; Nambara, E. Measurement of abscisic acid and gibberellins by gas chromatography/mass spectrometry. Methods Mol. Biol. 2009, 495, 53–60. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Diatloff, E.; Rengel, Z. Compilation of simple spectrophotometric techniques for the determination of elements in nutrient solutions. J. Plant Nutr. 2001, 24, 75–86. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Nakazawa, M. Fmsb: Functions for Medical Statistics Book with Some Demographic Data. R Package Version 0.6.3. 2018. Available online: https://CRAN.R-project.org/package=fmsb (accessed on 29 January 2020).
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1-141. 2019. Available online: http://CRAN.R-Project.Org/Package=Nlme (accessed on 28 January 2020).
- M’barki, N.; Aissaoui, F.; Chehab, H.; Dabbaghi, O.; del Giudice, T.; Boujnah, D.; Mechri, B. Cultivar dependent impact of soil amendment with water retaining polymer on olive (Olea europaea L.) under two water regimes. Agric. Water Manag. 2019, 216, 70–75. [Google Scholar] [CrossRef]
- Tawfik, M.M.; Badr, E.; Talooth, A.T. Smart farming practices to promote growth and productivity of atriplex (nummularia) in saline habitats. Int. J. Agric. For. Life Sci. 2019, 3, 161–170. [Google Scholar]
- Canellas, L.P.; Olivares, F.L.; Aguiar, N.O.; Jones, D.L.; Nebbioso, A.; Mazzei, P.; Piccolo, A. Humic and fulvic acids as biostimulants in horticulture. Sci. Hortic. 2015, 196, 15–27. [Google Scholar] [CrossRef]
- Yang, W.; Li, P.; Guo, S.; Song, R.; Yu, J. Co-application of soil superabsorbent polymer and foliar fulvic acid to increase tolerance to water deficit maize: Photosynthesis, water parameters, and proline. Chil. J. Agric. Res. 2019, 79, 435–446. [Google Scholar] [CrossRef]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments: A review. Plant Signal. Behav. 2012, 7, 1456–1466. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, L.; Zhou, H.; Li, M. Improved Viability of Areca (Areca catechu L.) Seedlings under Drought Stress Using a Superabsorbent Polymer. HortScience 2018, 53, 1872–1876. [Google Scholar] [CrossRef]
- Periman, R.D. The influence of prehistoric Anasazi cobble-mulch agricultural features of northern Rio Grande landscapes. In Tech Coords. Desired Future Conditions for Southwestern Riparian Ecosystems: Bringing Interests and Concerns Together; Shaw, D.W., Finch, D.M., Eds.; General Technical Report RM-GTR-272; Department of Agriculture, Forest Service, Rocky Mountain Forest and Range Experiment Station: Fort Collins, CO, USA, 1996; Volume 272, pp. 181–188. [Google Scholar]
| Tree Species | Origin | Vulnerability to Drought Stress (P50, MPa) |
|---|---|---|
| Acer negundo L. | N America | −1.7 [19] |
| Ailanthus altissima (Mill.) Swingle | E Asia | −1.2 [19] |
| Azadirachta indica A. Juss. | SE Asia | unknown |
| Berberis vulgaris L. | Europe, W Asia | −5.7 [19] |
| Celtis caucasica Willd. | SE Europe, Himalayas | from −0.8 to −1.5 (Celtis spp.) [19] |
| Cercis siliquastrum L. | W Asia, Mediterranean | −1.8 [19] |
| Cupressus arizonica Greene | N America | −11.0 (Cupressus spp.) [19] |
| Elaeagnus angustifolia L. | E Europe, Asia | unknown |
| Erythrostemon gilliesii (Hook.) Klotzsch | S America | from −2.1 to −2.5 (Erythrostemon spp.) [19] |
| Fraxinus excelsior L. | Europe | −2.8 [19] |
| Juglans nigra L. | N America | −2.0 [19] |
| Juniperus sp. | Holarctic region | −9.8 [19] |
| Morus alba L. | China | −0.2 [19] |
| Olea europea L. | S Europe, N Africa, W Asia | −7.2 [19] |
| Pinus brutia subsp. eldarica Tenore | Middle East, Russia | −3.1 [19] |
| Pinus nigra J. F. Arnold | S Europe | from −2.8 to −3.8 [20] |
| Platanus orientalis L. | SE Europe, SW Asia | −1.8 (Platanus hybrids) [21] |
| Platycladus orientalis (L.) Franco | Asia, Russia | −3.6 (Platycladus spp.) [19] |
| Populus nigra L. | Europe, Asia | −2.9 [19] |
| Robinia pseudoacacia L. | N America | from −0.5 to −0.9 [22,23] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomášková, I.; Svatoš, M.; Macků, J.; Vanická, H.; Resnerová, K.; Čepl, J.; Holuša, J.; Hosseini, S.M.; Dohrenbusch, A. Effect of Different Soil Treatments with Hydrogel on the Performance of Drought-Sensitive and Tolerant Tree Species in a Semi-Arid Region. Forests 2020, 11, 211. https://doi.org/10.3390/f11020211
Tomášková I, Svatoš M, Macků J, Vanická H, Resnerová K, Čepl J, Holuša J, Hosseini SM, Dohrenbusch A. Effect of Different Soil Treatments with Hydrogel on the Performance of Drought-Sensitive and Tolerant Tree Species in a Semi-Arid Region. Forests. 2020; 11(2):211. https://doi.org/10.3390/f11020211
Chicago/Turabian StyleTomášková, Ivana, Michal Svatoš, Jan Macků, Hana Vanická, Karolina Resnerová, Jaroslav Čepl, Jaroslav Holuša, Seyed Mohammad Hosseini, and Achim Dohrenbusch. 2020. "Effect of Different Soil Treatments with Hydrogel on the Performance of Drought-Sensitive and Tolerant Tree Species in a Semi-Arid Region" Forests 11, no. 2: 211. https://doi.org/10.3390/f11020211
APA StyleTomášková, I., Svatoš, M., Macků, J., Vanická, H., Resnerová, K., Čepl, J., Holuša, J., Hosseini, S. M., & Dohrenbusch, A. (2020). Effect of Different Soil Treatments with Hydrogel on the Performance of Drought-Sensitive and Tolerant Tree Species in a Semi-Arid Region. Forests, 11(2), 211. https://doi.org/10.3390/f11020211





