Hydrophobization and Photo-Stabilization of Radiata Pinewood: The Effect of the Esterification on Thermal and Mechanical Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Wood Samples
2.2. Esterification Process
2.3. Gravimetric Changes
2.4. Chemical Changes
2.5. Wettability
2.6. Moisture Sorption Measurements
2.7. UV Accelerated Ageing Test
2.8. Mechanical Changes
2.9. Oxidative Response of Esterified Wood
3. Results and Discussion
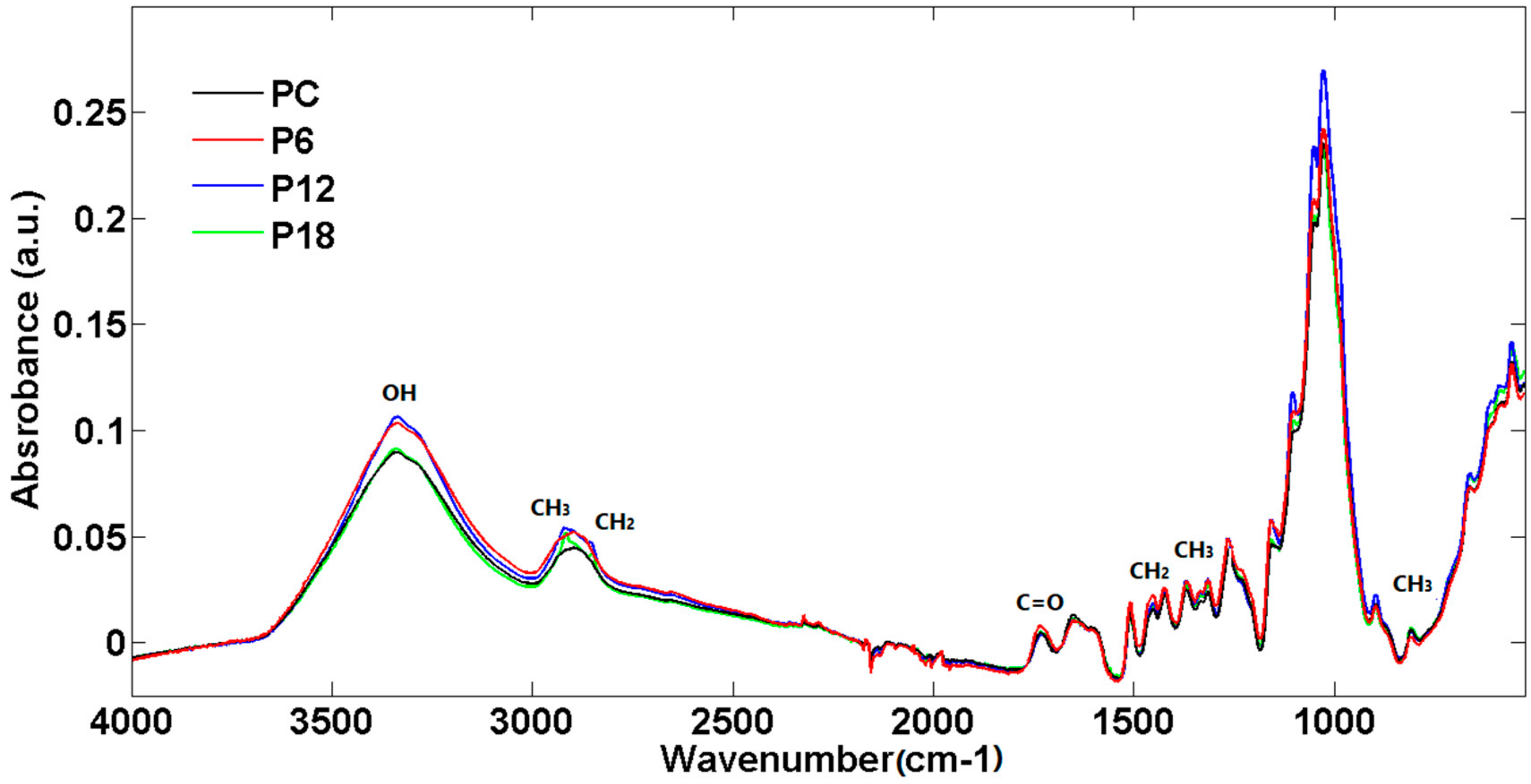
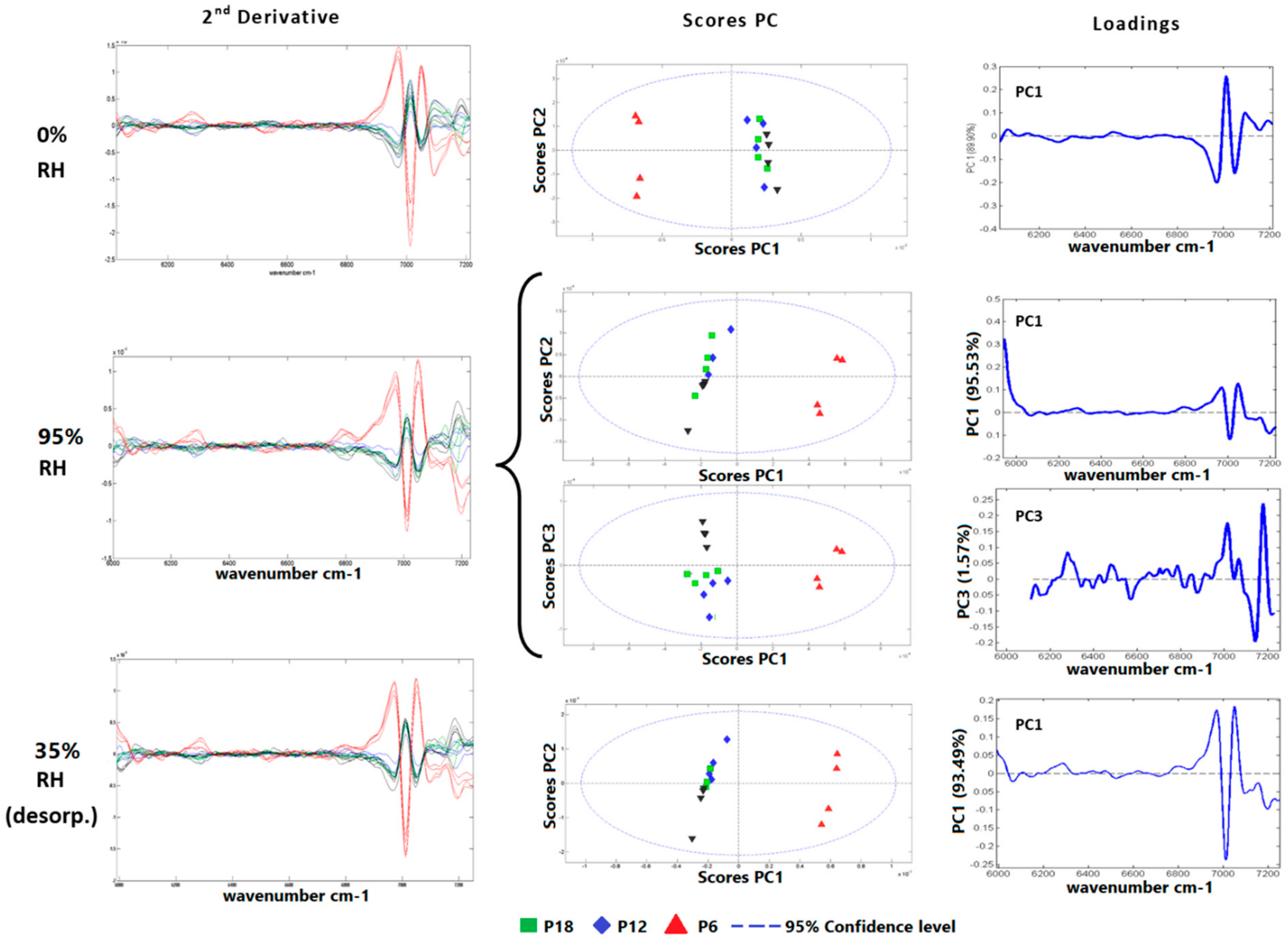
3.1. Spectroscopic Analysis of Esterified Wood
3.2. Physical Changes
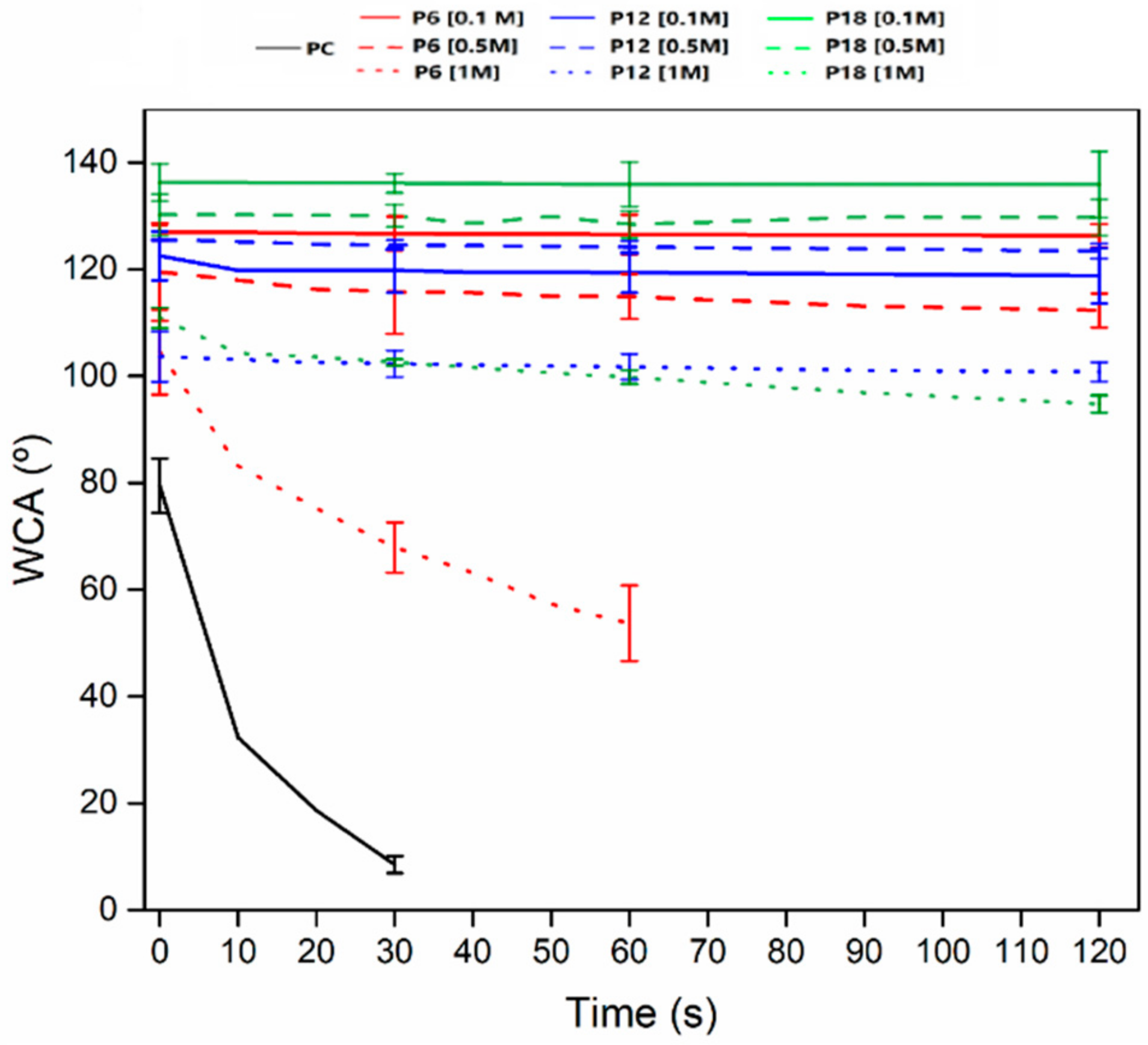
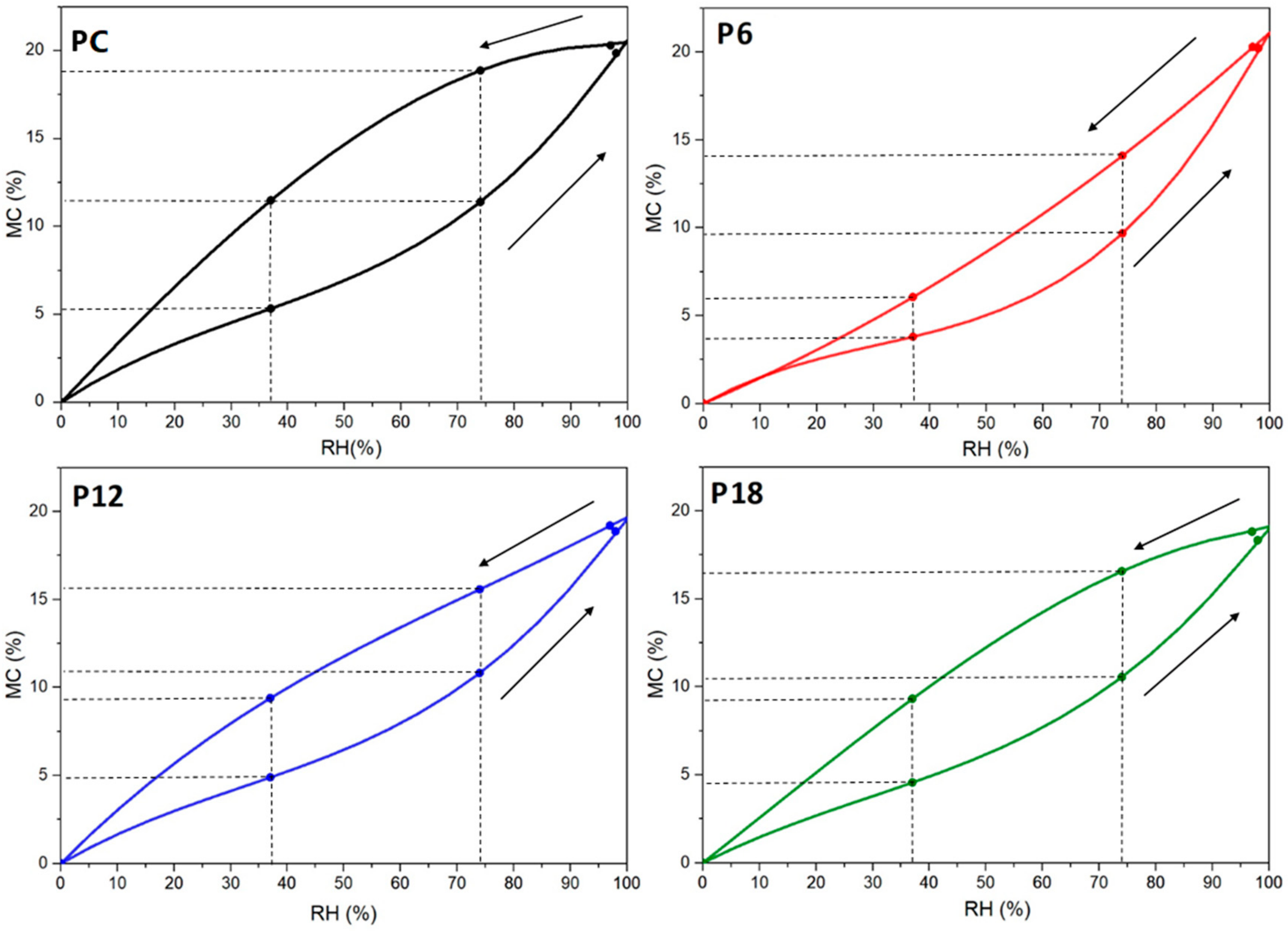
3.3. Wetting Behavior and Hygroscopic Properties
3.4. Photostability of Modified Wood
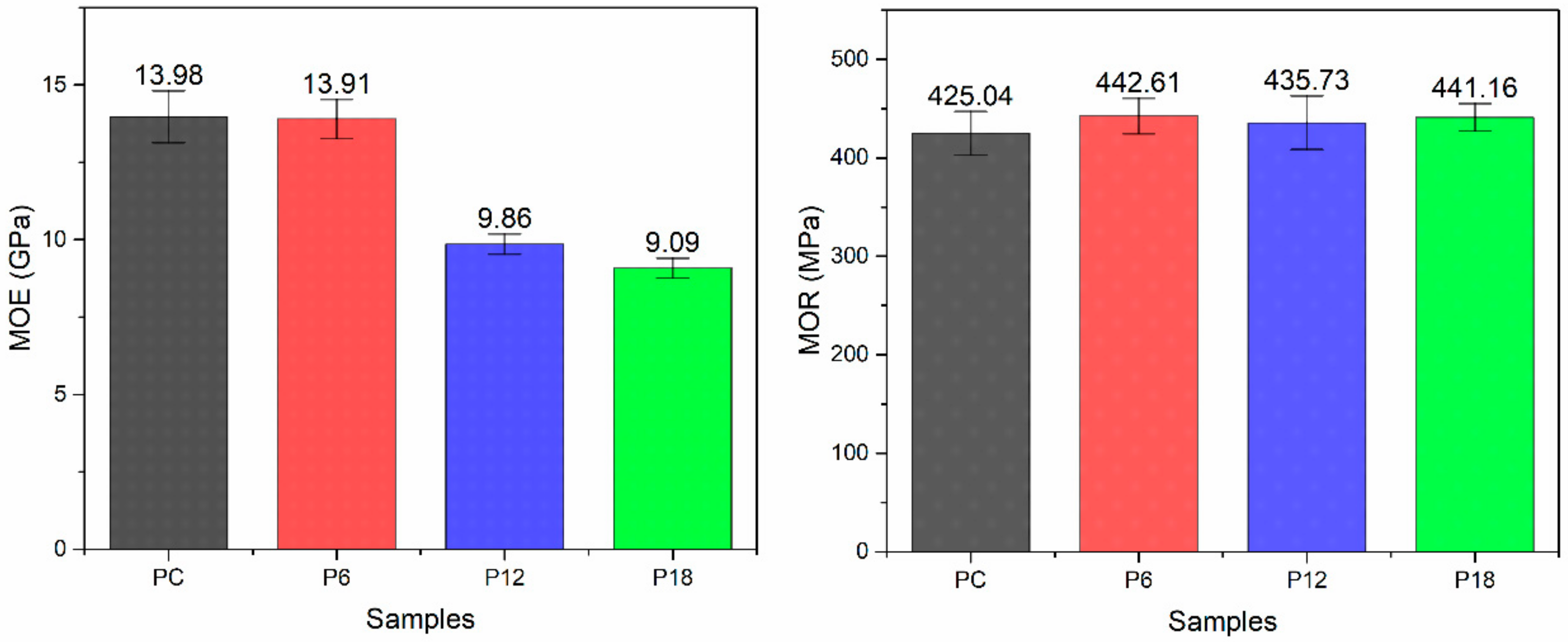
3.5. Mechanical Properties
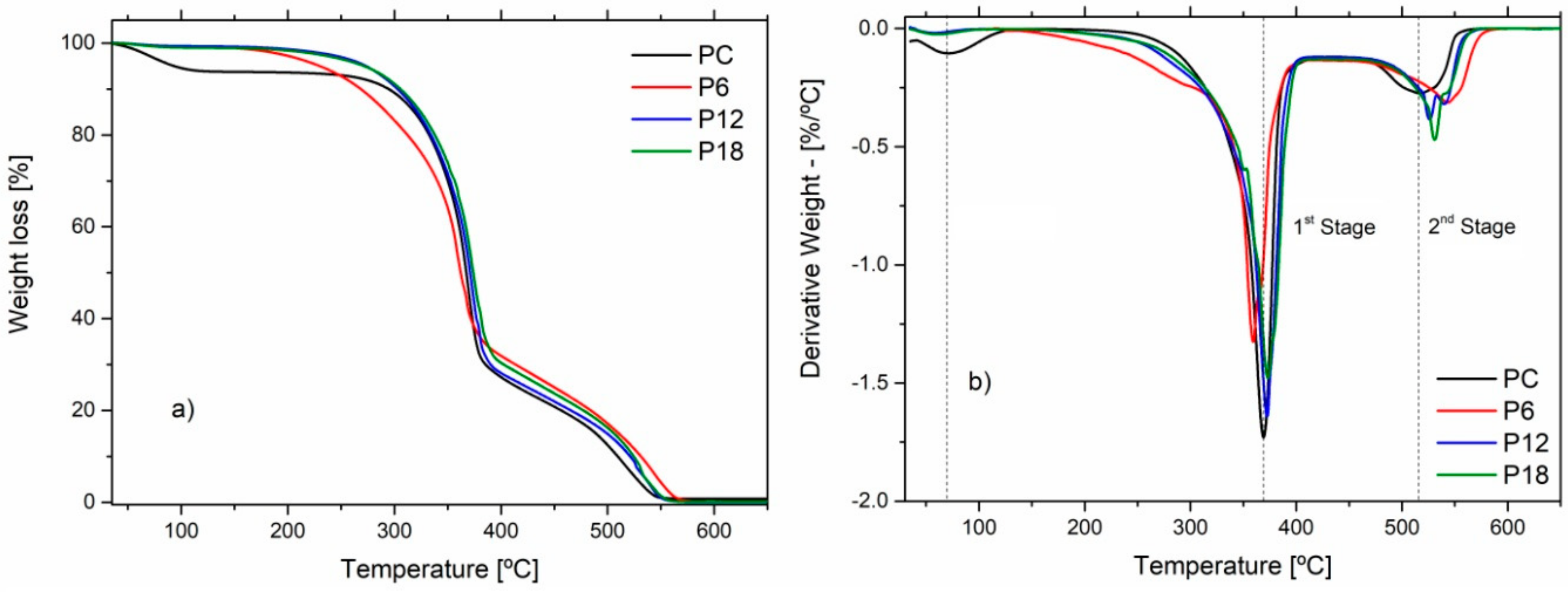
3.6. Thermal Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mantanis, G.I. Chemical modification of wood by acetylation or furfurylation: A review of the present scaled-up technologies. BioResources 2017, 12, 4478–4489. [Google Scholar] [CrossRef]
- Rowell, R.M.; Dickerson, J.P. Acetylation of wood. In Deterioration and Protection of Sustainable Biomaterials; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2014; Volume 1158, pp. 301–327. [Google Scholar]
- Jones, D. Wood Modification in Europe: A State-of-the-Art About Processes, Products and Applications; Firenze University Press: Firenze, Italy, 2019; ISBN 8864539700. [Google Scholar]
- Gérardin, P. New alternatives for wood preservation based on thermal and chemical modification of wood—A review. Ann. For. Sci. 2016, 73, 559–570. [Google Scholar] [CrossRef]
- Petrič, M. Surface modification of wood: A critical review. Rev. Adhes. Adhes. 2013, 1, 216–247. [Google Scholar] [CrossRef]
- Rowell, R.M. Chemical Modification of Wood. In Handbook of Wood Chemistry and Wood Composites, 2nd ed.; CRC Press/Taylor Francis Group: Boca Raton, FL, USA, 2013; Chapter 2; pp. 537–597. [Google Scholar]
- Teacă, C.A.; Tanasa, F. Wood surface modification-classic and modern approaches in wood chemical treatment by esterification reactions. Coatings 2020, 10, 629. [Google Scholar] [CrossRef]
- He, M.; Xu, D.; Li, C.; Ma, Y.; Dai, X.; Pan, X.; Fan, J.; He, Z.; Gui, S.; Dong, X.; et al. Cell wall bulking by maleic anhydride for wood durability improvement. Forests 2020, 11, 367. [Google Scholar] [CrossRef]
- Mubarok, M.; Militz, H.; Dumarçay, S.; Gérardin, P. Beech wood modification based on in situ esterification with sorbitol and citric acid. Wood Sci. Technol. 2020, 54, 479–502. [Google Scholar] [CrossRef]
- George, B.; Suttie, E.; Merlin, A.; Deglise, X. Photodegradation and photostabilisation of wood—The state of the art. Polym. Degrad. Stab. 2005, 88, 268–274. [Google Scholar] [CrossRef]
- Thiebaud, S.; Borredon, M.E. Solvent-Free Wood Esterification with Fatty-Acid Chlorides. Bioresour. Technol. 1995, 52, 169–173. [Google Scholar] [CrossRef]
- Rowell, R.M. Handbook of Wood Chemistry and Wood Composites, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9781439853818. [Google Scholar]
- Teacă, C.A.; Bodîrlău, R.; Spiridon, I. Maleic anhydride treatment of softwood—Effect on wood structure and properties. Cellul. Chem. Technol. 2014, 48, 863–868. [Google Scholar]
- Larnøy, E.; Karaca, A.; Gobakken, L.R.; Hill, C.A.S. Polyesterification of wood using sorbitol and citric acid under aqueous conditions. Int. Wood Prod. J. 2018, 9, 66–73. [Google Scholar] [CrossRef]
- Noël, M.; Grigsby, W.; Vitkeviciute, I.; Volkmer, T. Modifying wood with bio-polyesters: Analysis and performance. Int. Wood Prod. J. 2015, 6, 14–20. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Xie, Y.; Zhang, K. Functional nanomaterials through esterification of cellulose: A review of chemistry and application. Cellulose 2018, 25, 3703–3731. [Google Scholar] [CrossRef]
- Rahn, K.; Diamantoglou, M.; Klemm, D.; Berghmans, H.; Heinze, T. Homogeneous synthesis of cellulose p-toluenesulfonates in N,N-dimethylacetamide/LiCl solvent system. Angew. Makromol. Chem. 1996, 238, 143–163. [Google Scholar] [CrossRef]
- Doczekalska, B.; Bartkowiak, M.; Zakrzewski, R. Esterification of Willow Wood With Cyclic Acid Anhydrides. Wood Res. 2014, 59, 85–96. [Google Scholar]
- Zhang, Y.; Xue, Y.; Toghiani, H.; Zhang, J.; Pittman, C.U. Modification of Wood Flour Surfaces by Esterification with Acid Chlorides: Use in HDPE/Wood Flour Composites. Compos. Interfaces 2009, 16, 671–686. [Google Scholar] [CrossRef]
- Yusof, N.A.; Mukhair, H.; Malek, E.A.; Mohammad, F. Esterified Coconut Coir by Fatty Acid Chloride as Biosorbent in Oil Spill Removal. BioResources 2015, 10, 8025–8038. [Google Scholar] [CrossRef]
- Prakash, G.K.; Pandey, K.K.; Ram, R.K.D.; Mahadevan, K.M. Dimensional stability and photostability of octanoylated wood. Holzforschung 2006, 60, 539–542. [Google Scholar] [CrossRef]
- Prakash, G.K.; Mahadevan, K.M. Enhancing the properties of wood through chemical modification with palmitoyl chloride. Appl. Surf. Sci. 2008, 254, 1751–1756. [Google Scholar] [CrossRef]
- Salla, J.; Pandey, K.K.; Prakash, G.K.; Mahadevan, K.M. Photobleaching and dimensional stability of rubber wood esterified by fatty acid chlorides. J. Wood Chem. Technol. 2012, 32, 121–136. [Google Scholar] [CrossRef]
- Jebrane, M.; Sèbe, G. A new process for the esterification of wood by reaction with vinyl esters. Carbohydr. Polym. 2008, 72, 657–663. [Google Scholar] [CrossRef]
- Gordobil, O.; Robles, E.; Egüés, I.; Labidi, J. Lignin-ester derivatives as novel thermoplastic materials. RSC Adv. 2016, 6, 86909–86917. [Google Scholar] [CrossRef]
- Bodîrlǎu, R.; Teacǎ, C.A.; Roşu, D.; Roşu, L.; Varganici, C.D.; Coroabǎ, A. Physico-chemical properties investigation of softwood surface after treatment with organic anhydride. Cent. Eur. J. Chem. 2013, 11, 2098–2106. [Google Scholar] [CrossRef]
- Zhang, A.P.; Mei, L.; Zhao, Z.Z.; Xie, J.; Liu, C.F.; Sun, R.C. Homogeneous Esterification of Eucalyptus with Palmitoyl Chloride at Room Temperature. Bioresources 2013, 8, 4288–4297. [Google Scholar] [CrossRef]
- Fodor, F.; Németh, R.; Lankveld, C.; Hofmann, T. Effect of acetylation on the chemical composition of hornbeam (Carpinus betulus L.) in relation with the physical and mechanical properties. Wood Mater. Sci. Eng. 2018, 13, 271–278. [Google Scholar] [CrossRef]
- Thiebaud, S.; Borredon, M.E.; Baziard, G.; Senocq, F. Properties of wood esterified by fatty-acid chlorides. Bioresour. Technol. 1997, 59, 103–107. [Google Scholar] [CrossRef]
- Rowell, R.M. Chemical modification of wood: A short review. Wood Mater. Sci. Eng. 2006, 1, 29–33. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Yang, T.; Wang, J.; Xu, J.; Ma, E.; Cao, J. Hygroscopicity and dimensional stability of Populus euramericana Cv. modified by furfurylation combined with low hemicellulose pretreatment. J. Mater. Sci. 2019, 54, 13445–13456. [Google Scholar] [CrossRef]
- Hill, C.A.S.; Norton, A.J.; Newman, G. The water vapour sorption properties of Sitka spruce determined using a dynamic vapour sorption apparatus. Wood Sci. Technol. 2010, 44, 497–514. [Google Scholar] [CrossRef]
- Shi, J.; Avramidis, S. Water sorption hysteresis in wood: I review and experimental patterns—Geometric characteristics of scanning curves. Holzforschung 2017, 71, 307–316. [Google Scholar] [CrossRef]
- Tsuchikawa, S.; Yonenobu, H.; Siesler, H.W. Near-infrared spectroscopic observation of the ageing process in archaeological wood using a deuterium exchange method. Analyst 2005, 130, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Brereton, R.G.; Jansen, J.; Lopes, J.; Marini, F.; Pomerantsev, A.; Rodionova, O.; Roger, J.M.; Walczak, B.; Tauler, R. Chemometrics in analytical chemistry—Part II: Modeling, validation, and applications. Anal. Bioanal. Chem. 2018, 410, 6691–6704. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, T.; Kobori, H.; Tsuchikawa, S. Prediction of wood density independently of moisture conditions using near infrared spectroscopy. J. Near Infrared Spectrosc. 2012, 20, 353–359. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.C.; Fackler, K. A review of band assignments in near infrared spectra of wood and wood components. J. Near Infrared Spectrosc. 2011, 19, 287–308. [Google Scholar] [CrossRef]
- Chai, Y.; Liu, J.; Wang, Z.; Zhao, Y. Dimensional Stability and Mechanical Properties of Plantation Poplar Wood Esterified Using Acetic Anhydride. BioResources 2016, 12, 912–922. [Google Scholar] [CrossRef]
- Green, D.; Winandy, J.; Kretschmann, D. Mechanical Properties of Wood. In Wood Handbook: Wood as an Engineering Material; USDA: Madison, WI, USA, 1999. [Google Scholar]
- Belgacem, M.N.; Pizzi, A. Lignocellulosic Fibers and Wood Handbook: Renewable Materials for Today’s Environment; John Wiley & Sons: Hoboken, NJ, USA, 2016; ISBN 9781118773727. [Google Scholar]
- Brebu, M.; Vasile, C. Thermal degradation of lignin—A review. Cellul. Chem. Technol. 2010, 44, 353–363. [Google Scholar]
- Hagen, M.; Hereid, J.; Delichatsios, M.A.; Zhang, J.; Bakirtzis, D. Flammability assessment of fire-retarded Nordic Spruce wood using thermogravimetric analyses and cone calorimetry. Fire Saf. J. 2009, 44, 1053–1066. [Google Scholar] [CrossRef]
- Rowell, R.M.; Dietenberger, M.A. Thermal Properties, Combustion, and Fire Retardancy of Wood. In Handbook of Wood Chemistry and Wood Composites, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9781439853818. [Google Scholar]
- Wu, J.H.; Hsieh, T.Y.; Lin, H.Y.; Shiau, I.L.; Chang, S.T. Properties of wood plasticization with octanoyl chloride in a solvent-free system. Wood Sci. Technol. 2004, 37, 363–372. [Google Scholar] [CrossRef]
- Bodîrlău, R.; Teacă, C.A.; Spiridon, I. Chemical modification of beech wood: Effect on thermal stability. BioResources 2008, 3, 789–800. [Google Scholar]
| Sample Code | Pretreatment | Fatty Acid Chloride | Molar Concentration | Reaction Conditions (°C/h) | ||
|---|---|---|---|---|---|---|
| PC (Pinus radiata) | Reference | |||||
| P6 | Solvent extraction | C:6 | [0.1] | [0.5] | [1] | 100/3 |
| P12 | C:12 | [0.1] | [0.5] | [1] | 100/3 | |
| P18 | C:18 | [0.1] | [0.5] | [1] | 80/3 | |
| Samples | Classif. Analysis | Signal Processing | N | Err | P | Class Err. | N Pred. |
|---|---|---|---|---|---|---|---|
| PC | SVMDA | EMSC-SNV-MC | 15 | 0.100 | 0.800 | 0.261 | 13 |
| P6 | 15 | 0.000 | 1.000 | 0.000 | 15 | ||
| P12 | 15 | 0.015 | 0.900 | 0.158 | 14 | ||
| P18 | 15 | 0.105 | 0.750 | 0.363 | 10 |
| Chemical Modification | Concentration [M] | Anhydrous Density ρA [kg/m3] | Density (65% RH 25°C) ρR [kg/m3] | Moisture Content at 65% RH 25°C | WPG [%] | Product Retention after Leaching [%] |
|---|---|---|---|---|---|---|
| Control | - | 432.20 (25.26) | 471.08 (30.45) | 10.50 (1.5) | - | - |
| P6 | [0.1] | 439.76 (29.11) | 471.95 (22.86) | 8.7 (1.4) | 1.06 (0.45) | 37.45 |
| [0.5] | 481.15 (17.37) | 512.81 (16.91) | 6.2 (0.5) | 5.64 (1.26) | 60.57 | |
| [1] | 468.32 23.97) | 501.05 (28.95) | 6.1 (0.5) | 4.44 (0.66) | 30.88 | |
| P12 | [0.1] | 427.78 (27.68) | 458.23 (29.64) | 9.8 (1.6) | 1.39 (0.27) | 58.51 |
| [0.5] | 444.53 (13.70) | 474.52 (28.87) | 8.1 (0.8) | 3.26 (0.49) | 57.75 | |
| [1] | 441.94 (18.06) | 470.72 (20.14) | 7.8 (0.7) | 6.19 (1.99) | 59.09 | |
| P18 | [0.1] | 441.16 (33.60) | 474.46 (34.99) | 9.8 (1.4) | 1.93 (0.30) | 48.73 |
| [0.5] | 460.28 (15.65) | 494.23 (30.90) | 8.3 (0.9) | 2.67 (0.45) | 50.75 | |
| [1] | 436.94 (0.34) | 470.32 (41.76) | 7.6 (0.9) | 2.38 (0.34) | 63.49 |
| Time | 0 h | 240 h | |||||
|---|---|---|---|---|---|---|---|
| Sample | L | a | b | ΔL | Δa | Δb | ΔE |
| PC | 84.95 (1.05) | 3.98 (0.40) | 9.51 (0.53) | 12.11 | 3.03 | 13.69 | 8.37 |
| P6 | 85.80 (0.96) | 4.36 (0.45) | 12.96 (0.89) | 7.39 | 0.03 | 0.27 | 2.78 |
| P12 | 84.98 (0.60) | 4.02 (0.36) | 12.04 (0.55) | 8.24 | 1.02 | 2.19 | 4.41 |
| P18 | 84.64 (0.58) | 3.55 (0.22) | 10.75 (0.47) | 9.61 | 1.64 | 6.35 | 4.20 |
| Sample | T10% (°C) | T50% (°C) | Temperatures of Degradation (°C) | |
|---|---|---|---|---|
| 1st Stage | 2nd Stage | |||
| PC | 279.9 | 367.0 | 369.1 | 517.6 |
| P6 | 263.3 | 363.1 | 361.8 | 538.1 |
| P12 | 295.6 | 370.6 | 372.7 | 536.7 |
| P18 | 297.6 | 373.4 | 373.3 | 530.9 |
| Properties | Treatment | ||
|---|---|---|---|
| P6 | P12 | P18 | |
| WPG | ++ | ++ | + |
| Product retention | + | ++ | ++ |
| Hydrophobicity | + | ++ | ++ |
| Dimensional stability | ++ | + | + |
| Photostability | ++ | + | + |
| Modulus of elasticity/Modulus of rupture | =/+ | -/+ | -/+ |
| Thermal performance | = | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera Díaz, R.; Gordobil, O.; de Hoyos-Martinez, P.L.; Sandak, A.; Labidi, J. Hydrophobization and Photo-Stabilization of Radiata Pinewood: The Effect of the Esterification on Thermal and Mechanical Properties. Forests 2020, 11, 1243. https://doi.org/10.3390/f11121243
Herrera Díaz R, Gordobil O, de Hoyos-Martinez PL, Sandak A, Labidi J. Hydrophobization and Photo-Stabilization of Radiata Pinewood: The Effect of the Esterification on Thermal and Mechanical Properties. Forests. 2020; 11(12):1243. https://doi.org/10.3390/f11121243
Chicago/Turabian StyleHerrera Díaz, René, Oihana Gordobil, Pedro L. de Hoyos-Martinez, Anna Sandak, and Jalel Labidi. 2020. "Hydrophobization and Photo-Stabilization of Radiata Pinewood: The Effect of the Esterification on Thermal and Mechanical Properties" Forests 11, no. 12: 1243. https://doi.org/10.3390/f11121243
APA StyleHerrera Díaz, R., Gordobil, O., de Hoyos-Martinez, P. L., Sandak, A., & Labidi, J. (2020). Hydrophobization and Photo-Stabilization of Radiata Pinewood: The Effect of the Esterification on Thermal and Mechanical Properties. Forests, 11(12), 1243. https://doi.org/10.3390/f11121243









