Living in Drylands: Functional Adaptations of Trees and Shrubs to Cope with High Temperatures and Water Scarcity
Abstract
1. Introduction
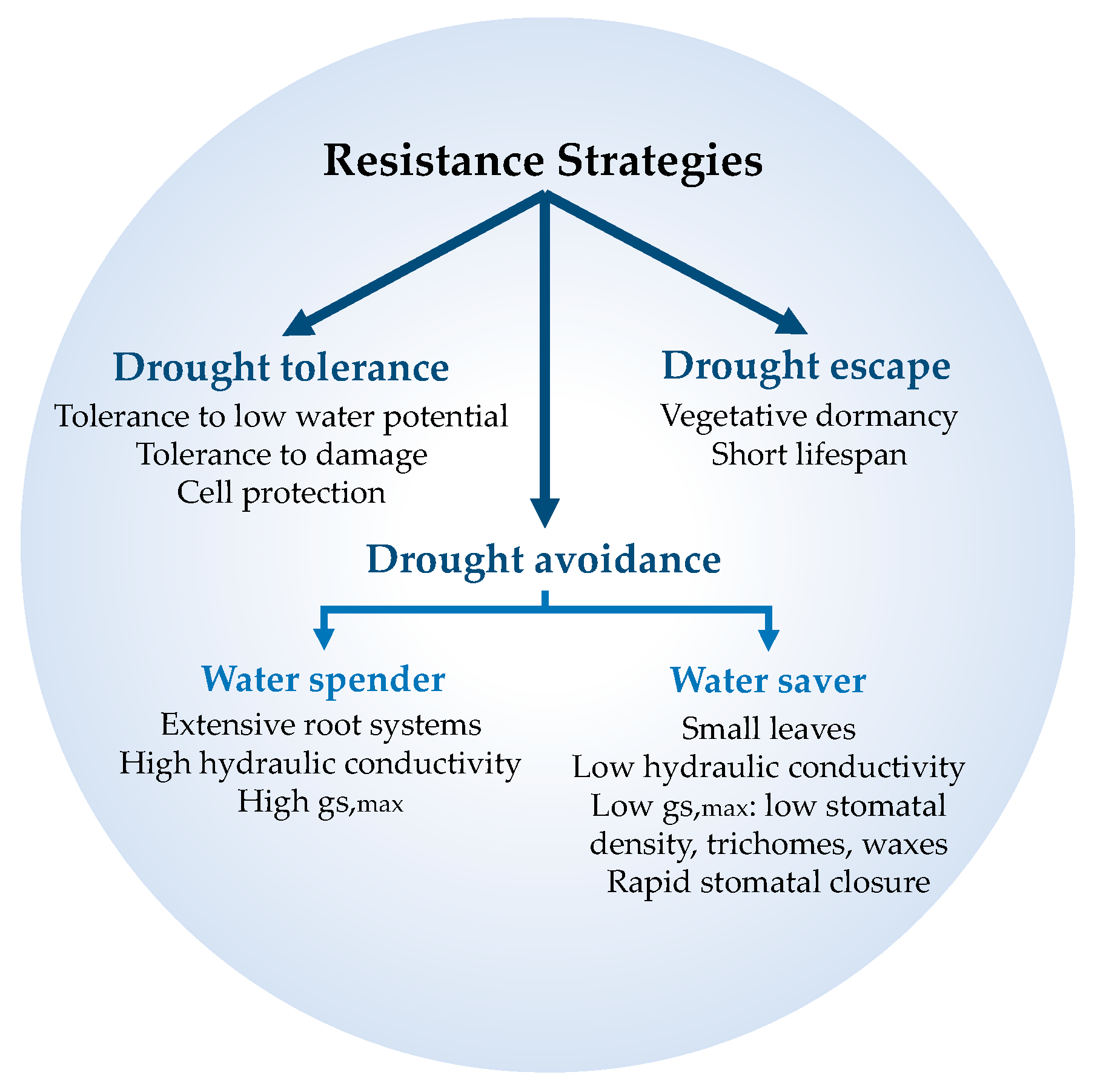
2. Plant Strategies to Cope with Drought
3. Plant Adaptations to Cope with High Temperatures under Drought Conditions
3.1. Reduction of Absorbed Light Energy
3.1.1. Changes in Leaf Angle
3.1.2. Increasing Leaf Reflectance
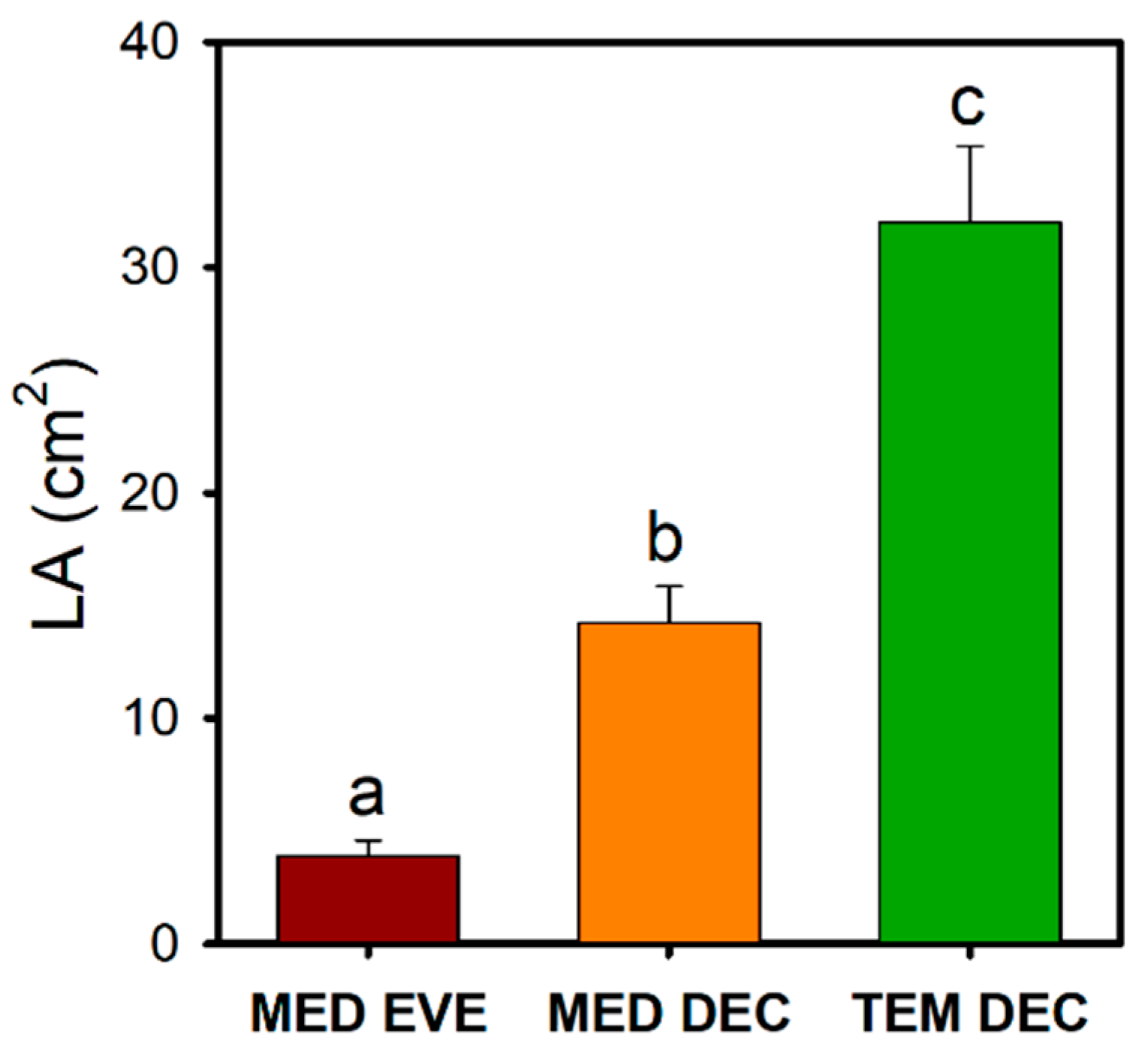
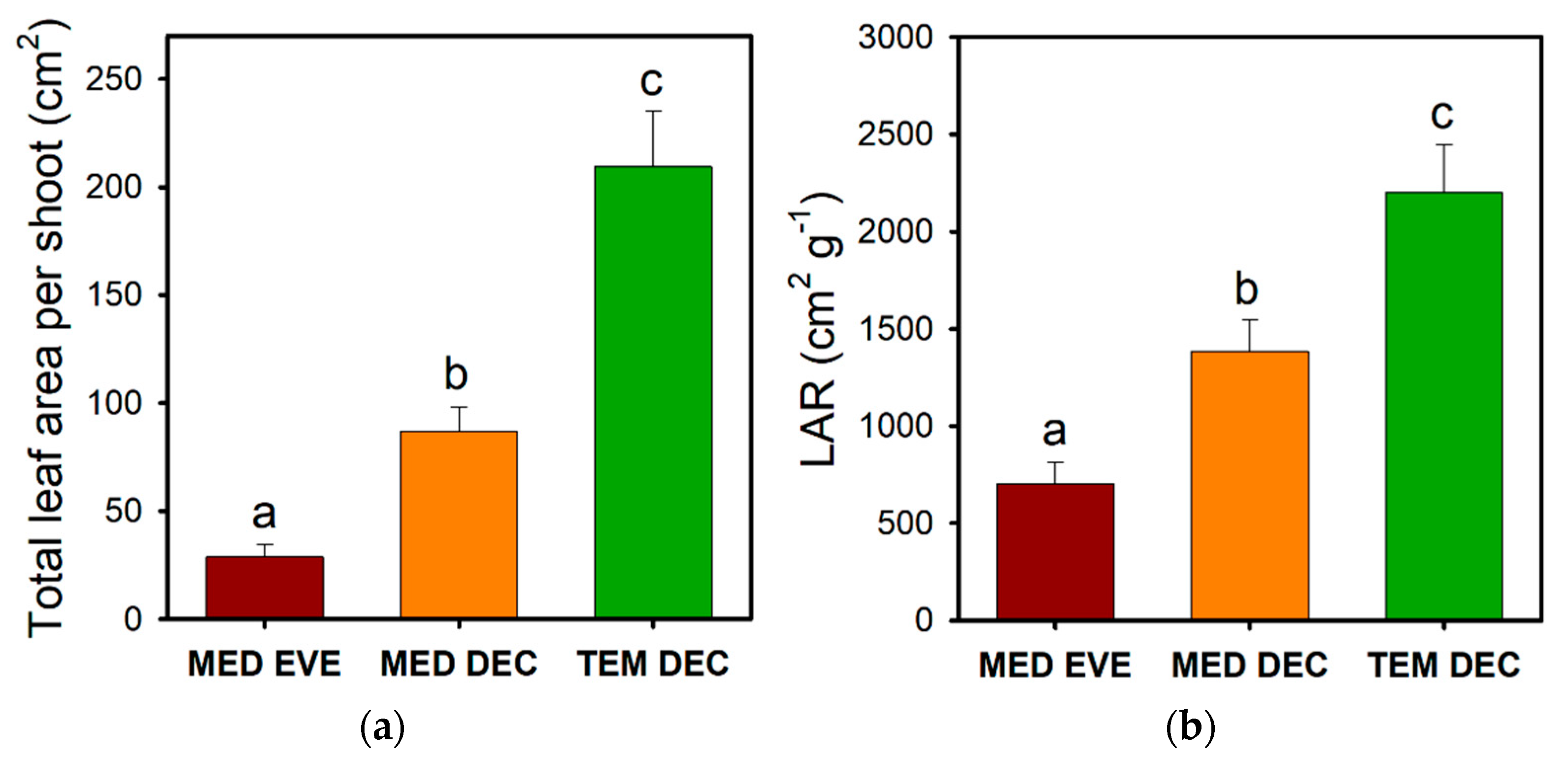
3.2. Leaf Size Reduction
3.3. Changes in Leaf Shape
3.4. Water Spender Strategy Enhances Leaf Cooling
3.5. Water Saver Strategy Regulates Water Loss at the Expense of a Reduced Ability for Leaf Cooling
4. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Schimper, A.F.W. Plant-Geography on a Physiological Basis; Clarendon Press: Oxford, UK, 1903. [Google Scholar]
- Althawadi, A.M.; Grace, J. Water use by the desert cucurbit Citrullus colocynthis (L.) Schrad. Oecologia 1986, 70, 475–480. [Google Scholar] [CrossRef]
- Bueno, A.; Alfarhan, A.; Arand, K.; Burghardt, M.; Deininger, A.-C.; Hedrich, R.; Leide, J.; Seufert, P.; Staiger, S.; Riederer, M. Effects of temperature on the cuticular transpiration barrier of two desert plants with water-spender and water-saver strategies. J. Exp. Bot. 2019, 70, 1613–1625. [Google Scholar] [CrossRef]
- Safriel, U.; Adeel, Z.; Niemeijer, D.; Puigdefabregas, J.; White, R.; Lal, R.; Winslow, M.; Ziedler, J.; Prince, S.; Archer, E.; et al. Dryland systems. In Ecosystems and Human Well-Being: Current State and Trends; Hassan, R., Scholes, R., Ash, N., Eds.; Findings of the Condition and Trends Working Group, Island Press: Washington, DC, USA, 2005; Volume 1, pp. 623–662. [Google Scholar]
- Quan, C.; Han, S.; Utescher, T.; Zhang, C.; Liu, Y.S.C. Validation of temperature-precipitation based aridity index: Paleoclimatic implications. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 386, 86–95. [Google Scholar] [CrossRef]
- Thornthwaite, C.W. An approach toward a rational classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- Mayr, S.; Schmid, P.; Laur, J.; Rosner, S.; Katline Charra-Vaskou, K.; Dämon, B.; Hacke, U.G. Uptake of water via branches helps timberline conifers refill embolized xylem in late winter. Plant Physiol. 2014, 164, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Mayr, S.; Hacke, U.; Schmid, P.; Schwienbacher, F.; Gruber, A. Frost drought in conifers at the alpine timberline: Xylem dysfunction and adaptations. Ecology 2006, 87, 3175–3185. [Google Scholar] [CrossRef]
- Santos Pereira, L.; Oweis, T.; Zairi, A. Irrigation management under water scarcity. Agric. Water Manag. 2002, 57, 175–206. [Google Scholar] [CrossRef]
- Cherlet, M.; Hutchinson, C.; Reynolds, J.; Hill, J.; Sommer, S.; von Maltitz, G. (Eds.) World Atlas of Desertification; Publication Office of the European Union: Luxembourg, 2018. [Google Scholar] [CrossRef]
- D’Odorico, P.; Porporato, A.; Runyian, C.W. (Eds.) Ecohydrology of arid and semiarid ecosystems: An introduction. In Dryland Ecohydrology; Springer Nature: Berlin/Heidelberg, Germany, 2019; pp. 1–30. [Google Scholar]
- Kirkham, M.B. (Ed.) Potential Evapotranspiration. In Principles of Soil and Plant Water Relations, 2nd ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 501–504. [Google Scholar]
- Cai, J.; Liu, Y.; Lei, T.; Santos Pereira, L. Estimating reference evapotranspiration with the FAO Penman–Monteith equation using daily weather forecast messages. Agric. For. Meteorol. 2017, 145, 22–35. [Google Scholar] [CrossRef]
- Sentelhas, P.C.; Gillespie, T.J.; Santos, E.A. Evaluation of FAO Penman–Monteith and alternative methods for estimating reference evapotranspiration with missing data in Southern Ontario, Canada. Agric. Water Manag. 2010, 97, 635–644. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A multiscalar drought index sensitive to global warming: The standardized precipitation evapotranspiration index. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef]
- Whitford, W.G.; Duval, B.D. (Eds.) Conceptual Framework, Paradigms, and Models. In Ecology of Desert Systems, 2nd ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 1–20. [Google Scholar]
- Gao, X.; Giorgi, F. Increased aridity in the Mediterranean region under greenhouse gas forcing estimated from high resolution simulations with a regional climate model. Glob. Planet. Chang. 2008, 62, 195–209. [Google Scholar] [CrossRef]
- Fu, Q.; Feng, S. Responses of terrestrial aridity to global warming. J. Geophys. Res. Atmos. 2014, 119. [Google Scholar] [CrossRef]
- Asadi Zarch, M.A.; Sivakumar, B.; Malekinezhad, H.; Sharma, A. Future aridity under conditions of global climate change. J. Hydrol. 2017, 554, 451–469. [Google Scholar] [CrossRef]
- Oberlander, T.M. Characterization of arid elements according to combined water balance parameters. J. Arid Environ. 1979, 2, 219–241. [Google Scholar] [CrossRef]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar] [CrossRef]
- Besnard, G.; Anthelm, F.; Baali-Cherif, D. The Laperrine’s olive tree (Oleaceae): A wild genetic resource of the cultivated olive and a model-species for studying the biogeography of the Saharan Mountains. Acta Bot. Gall. 2012, 159, 319–328. [Google Scholar] [CrossRef]
- Lionello, P.; Malanotte-Rizzoli, P.; Boscolo, R.; Alpert, P.; Artale, V.; Li, L.; Luterbacher, J.; May, W.; Trigo, R.; Tsimplis, M.; et al. The Mediterranean climate: An overview of the main characteristics and issues. In Mediterranean Climate Variability; Lionello, P., Malanotte-Rizzoli, P., Boscolo, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 1–26. ISBN 978-0-444-52170-5. [Google Scholar]
- Deitch, M.J.; Sapundjieff, M.J.; Feirer, S.T. Characterizing precipitation variability and trends in the world’s Mediterranean-Climate areas. Water 2017, 9, 259. [Google Scholar] [CrossRef]
- Walter, H. Vegetation of the Earth and Ecological Systems of the Geo-Biosphere, 3rd ed.; Springer: Berlin, Germany, 1985. [Google Scholar]
- Rivas-Martínez, S.; Rivas-Sáenz, S.; Penas-Merino, A. Worldwide Bioclimatic Classification System. Glob. Geobot. 2011, 1, 1–634. [Google Scholar]
- Gil-Pelegrín, E.; Saz, M.A.; Cuadrat, J.M.; Peguero-Pina, J.J.; Sancho-Knapik, D. Oaks under Mediterranean-type climates: Functional response to summer aridity. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus Quercus L.; Gil-Pelegrín, E., Peguero-Pina, J.J., Sancho-Knapik, D., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 137–193. ISBN 978-3-319-69099-5. [Google Scholar]
- Vilagrosa, A.; Bellot, J.; Vallejo, V.R.; Gil-Pelegrín, E. Cavitation, stomatal conductance, and leaf dieback in seedlings of two co-occurring Mediterranean shrubs during an intense drought. J. Exp. Bot. 2003, 54, 2015–2024. [Google Scholar] [CrossRef] [PubMed]
- Vilagrosa, A.; Morales, F.; Abadía, A.; Bellot, J.; Cochard, H.; Gil-Pelegrín, E. Are symplast tolerance to intense drought conditions and xylem vulnerability to cavitation coordinated? An integrated analysis of photosynthetic, hydraulic and leaf level processes in two Mediterranean drought-resistant species. Environ. Exp. Bot. 2010, 69, 233–242. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Morales, F.; Flexas, J.; Gil-Pelegrín, E.; Moya, I. Photochemistry, remotely sensed physiological reflectance index and de-epoxidation state of the xanthophyll cycle in Quercus coccifera under intense drought. Oecologia 2008, 156, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Peguero-Pina, J.J.; Gil-Pelegrín, E.; Morales, F. Three pools of zeaxanthin in Quercus coccifera leaves during light transitions with different roles in rapidly reversible photoprotective energy dissipation and photoprotection. J. Exp. Bot. 2013, 64, 1649–1661. [Google Scholar] [CrossRef] [PubMed]
- Abadía, A.; Gil, E.; Morales, F.; Montañés, L.; Montserrat, G.; Abadía, J. Marcescence and senescence in a submediterranean oak (Quercus subpyrenaica E.H. del Villar): Photosynthetic characteristics and nutrient composition. Plant Cell Environ. 1996, 19, 685–694. [Google Scholar] [CrossRef]
- Himrane, H.; Camarero, J.J.; Gil-Pelegrín, E. Morphological and ecophysiological variation of the hybrid oak Quercus subpyrenaica (Q. faginea × Q. pubescens). Trees 2004, 18, 566–575. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Martín, P.; Saz, M.A.; Gea-Izquierdo, G.; Cañellas, I.; Gil-Pelegrín, E. Evidence of vulnerability segmentation in a deciduous Mediterranean oak (Quercus subpyrenaica E. H. del Villar). Trees 2015, 29, 1917–1927. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sisó, S.; Sancho-Knapik, D.; Díaz-Espejo, A.; Flexas, J.; Galmés, J.; Gil-Pelegrín, E. Leaf morphological and physiological adaptations of a deciduous oak (Quercus faginea Lam.) to the Mediterranean climate: A comparison with a closely related temperate species (Quercus robur L.). Tree Physiol. 2016, 36, 287–299. [Google Scholar] [CrossRef]
- Levitt, J. Responses of Plants to Environmental Stresses, Vol II; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Lo Gullo, M.A.; Salleo, S. Different strategies of drought resistance in three Mediterranean sclerophyllous trees growing in the same environmental conditions. New Phytol. 1988, 108, 267–276. [Google Scholar] [CrossRef]
- Sakcali, M.S.; Ozturk, M. Eco-physiological behaviour of some mediterranean plants as suitable candidates for reclamation of degraded areas. J. Arid Environ. 2004, 57, 141–153. [Google Scholar] [CrossRef]
- Manes, F.; Vitale, M.; Donato, E.; Giannini, M.; Puppi, G. Different ability of three Mediterranean oak species to tolerate progressive water stress. Photosynthetica 2006, 44, 387–393. [Google Scholar] [CrossRef]
- Turner, N.C. Adaptation to water deficits: A changing perspective. Funct. Plant Biol. 1986, 13, 175–190. [Google Scholar] [CrossRef]
- Kozlowski, T.T.; Kramer, P.J.; Pallardy, S.G. The Physiological Ecology of Woody Plants; Academic Press: San Diego, CA, USA, 1991. [Google Scholar]
- Jones, H.G. Plants and Microclimate: A Quantitative Approach to Environmental Plant Physiology, 2nd ed.; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
- Larcher, W. Physiological Plant Ecology: Ecophysiology and Stress Physiology of Functional Groups; Springer: Berlin/Heidelberg, Germany, 1995. [Google Scholar]
- Volaire, F. A unified framework of plant adaptive strategies to drought: Crossing scales and disciplines. Glob. Chang. Biol. 2018, 24, 2929–2938. [Google Scholar] [CrossRef] [PubMed]
- Valladares, F.; Vilagrosa, A.; Peñuelas, J.; Ogaya, R.; Camarero, J.J.; Corcuera, L.; Sisó, S.; Gil-Pelegrín, E. Estrés hídrico: Ecofisiología y escalas de la sequía. In Ecología del bosque mediterráneo en un mundo cambiante; Valladares, F., Ed.; Ministerio de Medio Ambiente: Madrid, Spain, 2004; pp. 163–190. [Google Scholar]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Vilagrosa, A.; Hernández, E.I.; Luis, V.C.; Cochard, H.; Pausas, J.G. Physiological differences explain the co-existence of different regeneration strategies in Mediterranean ecosystems. New Phytol. 2014, 201, 1277–1288. [Google Scholar] [CrossRef]
- Grace, J. Environmental controls of gas exchange in tropical rain forests. In Physiological Plant Ecology; Press, M.C., Scholes, J.D., Barker, M.G., Eds.; British Ecological Society: London, UK, 1999. [Google Scholar]
- Law, B.E.; Williams, M.; Anthoni, P.M.; Baldocchi, D.D.; Unsworth, M.H. Measuring and modelling seasonal variation of carbon dioxide and water vapour exchange of a Pinus ponderosa forest subject to soil water deficit. Glob. Change Biol. 2000, 6, 613–630. [Google Scholar] [CrossRef]
- Wilson, K.B.; Baldocchi, D.D.; Hanson, P.J. Leaf age affects the seasonal pattern of photosynthetic capacity and net ecosystem exchange of carbon in a deciduous forest. Plant Cell Environ. 2001, 24, 571–583. [Google Scholar] [CrossRef]
- Knight, C.A.; Ackerly, D.D. Evolution and plasticity of photosynthetic thermal tolerance, specific leaf area and leaf size: Congeneric species from desert and coastal environments. New Phytol. 2003, 160, 337–347. [Google Scholar] [CrossRef]
- Braun, V.; Buchner, O.; Neuner, G. Thermotolerance of photosystem 2 of three alpine plant species under field conditions. Photosynthetica 2002, 40, 587–595. [Google Scholar] [CrossRef]
- Krause, G.H.; Winter, K.; Krause, B.; Jahns, P.; García, M.; Aranda, J.; Virgo, A. High-temperature tolerance of a tropical tree, Ficus insipida: Methodological reassessment and climate change considerations. Funct. Plant Biol. 2010, 37, 890–900. [Google Scholar] [CrossRef]
- Dreyer, E.; Le Roux, X.; Montpied, P.; Duadet, F.A.; Masson, F. Temperature response of leaf photosynthetic capacity in seedlings from seven temperate tree species. Tree Physiol. 2001, 21, 223–232. [Google Scholar] [CrossRef]
- Leon-Garcia, I.V.; Lasso, E. High heat tolerance in plants from the Andean highlands: Implications for paramos in a warmer world. PLoS ONE 2019, 14, e0224218. [Google Scholar] [CrossRef]
- Haldimann, P.; Gallé, A.; Feller, U. Impact of an exceptionally hot dry summer on photosynthetic traits in oak (Quercus pubescens) leaves. Tree Physiol. 2008, 28, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Gounaris, K.; Brain, A.P.; Quinn, P.J.; Williams, W.P. Structural reorganization of chloroplast thylakoid membranes in response to heat stress. Biochim. Biophys. Acta 1984, 766, 198–208. [Google Scholar] [CrossRef]
- Yordanov, I.; Dilova, S.; Petkova, R.; Pangelova, T.; Goltsev, V.; Süss, K.H. Mechanisms of the temperature damage and acclimation of the photosynthetic apparatus. Photobiochem. Photobiophys. 1986, 12, 147–155. [Google Scholar]
- Perdomo, J.A.; Capó-Bauçà, S.; Carmo-Silva, E.; Galmés, J. Rubisco and Rubisco Activase Play an Important Role in the Biochemical Limitations of Photosynthesis in Rice, Wheat, and Maize under High Temperature and Water Deficit. Front. Plant Sci. 2017, 8, 490. [Google Scholar] [CrossRef]
- Burghardt, M.; Riederer, M. Ecophysiological relevance of cuticular transpiration of deciduous and evergreen plants in relation to stomatal closure and leaf water potential. J. Exp. Bot. 2003, 54, 1941–1949. [Google Scholar] [CrossRef]
- Riederer, M. Thermodynamics of the water permeability of plant cuticles: Characterization of the polar pathway. J. Exp. Bot. 2003, 57, 2937–2942. [Google Scholar] [CrossRef] [PubMed]
- Fernández, V.; Bahamonde, H.A.; Peguero-Pina, J.J.; Gil-Pelegrín, E.; Sancho-Knapik, D.; Gil, L.; Goldbach, H.E.; Eichert, T. Physico-chemical properties of plant cuticles and their functional and ecological significance. J. Exp. Bot. 2017, 68, 5293–5306. [Google Scholar] [CrossRef]
- Yeats, T.H.; Rose, J.K.C. The formation and function of plant cuticles. Plant Physiol. 2013, 163, 5–20. [Google Scholar] [CrossRef]
- Bueno, A.; Sancho-Knapik, D.; Gil-Pelegrín, E.; Leide, J.; Peguero-Pina, J.J.; Burghardt, M.; Riederer, M. Cuticular wax coverage and its transpiration barrier properties in Quercus coccifera L. leaves: Does the environment matter? Tree Physiol. 2020, 40, 827–840. [Google Scholar] [CrossRef]
- Schuster, A.-C.; Burghardt, M.; Alfarhan, A.; Bueno, A.; Hedrich, R.; Leide, J.; Thomas, J.; Riederer, M. Effectiveness of cuticular transpiration barriers in a desert plant at controlling water loss at high temperatures. AoB Plants 2016, 8, plw027. [Google Scholar] [CrossRef]
- Vogel, S. Leaves in the lowest and highest winds: Temperature, force and shape. New Phytol. 2009, 183, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Ehleringer, J.R.; Comstock, J. Leaf absorptance and leaf angle: Mechanisms for stress avoidance. In Plant Response to Stress; Tenhunen, J.D., Catarino, F.M., Lange, O.L., Oechel, W.C., Eds.; NATO ASI Series (Series G: Ecological Sciences); Springer: Berlin, Heidelberg, Germany, 1987; Volume 15, pp. 55–76. [Google Scholar]
- Leigh, A.; Sevanto, S.; Close, J.D.; Nicotra, A.B. The influence of leaf size and shape on leaf thermal dynamics: Does theory hold up under natural conditions? Plant Cell Environ. 2017, 40, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Gibson, A.C. Structure-Function Relations of Warm Desert Plants; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Vogel, S. Convective cooling at low airspeeds and the shapes of broad leaves. J. Exp. Bot. 1970, 21, 91–101. [Google Scholar] [CrossRef]
- Jubany-Marí, T.; Munné -Bosch, S.; López-Carbonell, M.; Alegre, L. Hydrogen peroxide is involved in the acclimation of the Mediterranean shrub, Cistus albidus L., to summer drought. J. Exp. Bot. 2009, 60, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Feistler, A.M.; Habermann, G. Assessing the role of vertical leaves within the photosynthetic function of Styrax camporum under drought conditions. Photosynthetica 2012, 50, 613–622. [Google Scholar] [CrossRef]
- Medina, E. Adaptations of tropical trees to moisture stress. In Tropical Rain Forest Ecosystems: A. Structure and Function; Golley, F.G., Ed.; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1983; pp. 225–237. [Google Scholar]
- Ball, M.C.; Cowan, I.R.; Farquhar, G.D. Maintenance of leaf temperature and the optimisation of carbon gain in relation to water loss in a tropical mangrove forest. Funct. Plant Biol. 1988, 15, 263–276. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Clough, B.F. Influence of solar radiation and leaf angle on leaf xanthophyll concentrations in mangroves. Oecologia 1992, 91, 518–525. [Google Scholar] [CrossRef]
- Ehleringer, J.R.; Forseth, I.N. Diurnal leaf movements and productivity in canopies. In Plant Canopies: Their Growth, Form and Function; Russel, G., Marshall, B., Jarvis, P.G., Eds.; Cambridge University Press: Cambridge, UK, 1989; pp. 129–142. [Google Scholar]
- Ortiz, C.A.; Bravo, L.A.; Pinto, M.; Cardemil, L. Physiological and molecular responses of Prosopis chilensis under field and simulation conditions. Phytochemistry 1995, 40, 1375–1382. [Google Scholar] [CrossRef]
- Arena, C.; Vitale, L.; De Santo, A.V. Paraheliotropism in Robinia pseudoacacia L.: An efficient strategy to optimise photosynthetic performance under natural environmental conditions. Plant Biol. 2008, 10, 194–201. [Google Scholar] [CrossRef]
- Wan, C.; Sosebee, R.E. Water Relations and Transpiration of Honey Mesquite on 2 Sites in West Texas. J. Range Manag. 1991, 44, 156. [Google Scholar] [CrossRef]
- Villagra, P.E.; Vilela, A.; Giordano, C.; Alvarez, J.A. Ecophysiology of Prosopis Species From the Arid Lands of Argentina: What Do We Know About Adaptation to Stressful Environments? In Desert Plants: Biology and Biotechnology; Ramawat, K.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 321–340. [Google Scholar] [CrossRef]
- Chávez, R.O.; Clevers, J.G.P.W.; Herold, M.; Acevedo, E.; Ortiz, M. Assessing water stress of desert tamarugo trees using in situ data and very high spatial resolution remote sensing. Remote Sens. 2013, 5, 5064–5088. [Google Scholar] [CrossRef]
- Habermann, G.; Ellsworth, P.F.V.; Cazoto, J.L.; Feistler, A.M.; da Silva, L.; Donatti, D.A.; Machado, S.R. Leaf paraheliotropism in Styrax camporum confers increased light use efficiency and advantageous photosynthetic responses rather than photoprotection. Environ. Exp. Bot. 2011, 71, 10–17. [Google Scholar] [CrossRef]
- Heim, R.H.-J.; Jürgens, N.; Große-Stoltenberg, A.; Oldeland, J. The Effect of Epidermal Structures on Leaf Spectral Signatures of Ice Plants (Aizoaceae). Remote Sens. 2015, 7, 16901–16914. [Google Scholar] [CrossRef]
- Huggins, T.D.; Mohammed, S.; Sengodon, P.; Ibrahim, A.M.H.; Tilley, M.; Hays, D.B. Changes in leaf epicuticular wax load and its effect on leaf temperature and physiological traits in wheat cultivars (Triticum aestivum L.) exposed to high temperatures during anthesis. J. Agro. Crop. Sci. 2017, 204, 49–61. [Google Scholar] [CrossRef]
- Fernández-Marín, B.; Hernández, A.; Garcia-Plazaola, J.I.; Esteban, R.; Míguez, F.; Artetxe, U.; Gómez-Sagasti, M.T. Photoprotective Strategies of Mediterranean Plants in Relation to Morphological Traits and Natural Environmental Pressure: A Meta Analytical Approach. Front. Plant Sci. 2017, 8, 1051. [Google Scholar] [CrossRef]
- Werker, E. Trichome diversity and development. Ad. Bot. Res. 2000, 31, 1–35. [Google Scholar] [CrossRef]
- Bickford, C.P. Ecophysiology of leaf trichomes. Funct. Plant Biol. 2016, 43, 807–814. [Google Scholar] [CrossRef]
- Agrawal, A.A.; Fishbein, M.; Jetter, R.; Salminen, J.P.; Goldstein, J.B.; Freitag, A.E.; Sparks, J.P. Phylogenetic ecology of leaf surface traits in the milkweeds (Asclepias spp.): Chemistry, ecophysiology, and insect behavior. New Phytol. 2009, 183, 848–867. [Google Scholar] [CrossRef]
- Sakai, W.S.; Sanford, W.G. Ultrastructure of the water-absorbing trichomes of Pineapple (Ananas comosus, Bromeliaceae). Ann. Bot. 1980, 46, 7–11. [Google Scholar] [CrossRef]
- Fernández, V.; Sancho-Knapik, D.; Guzmán, P.; Peguero-Pina, J.J.; Gil, L.; Karabourniotis, G.; Khayet, M.; Fasseas, C.; Heredia-Guerrero, J.A.; Heredia, A.; et al. Wettability, polarity and water absorption of Quercus ilex leaves: Effect of leaf side and age. Plant Physiol. 2014, 166, 168–180. [Google Scholar] [CrossRef]
- Karabourniotis, G.; Bornman, J.F. Penetration of UV-A, UV-B and blue light through the leaf trichome layers of two xeromorphic plants, olive and oak, measured by optical fibre microprobes. Physiol. Plant. 1999, 105, 655–661. [Google Scholar] [CrossRef]
- Morales, F.; Abadía, A.; Abadía, J.; Montserrat, G.; Gil-Pelegrín, E. Trichomes and photosynthetic pigment composition changes: Responses of Quercus ilex subsp. ballota (Desf.) Samp. and Quercus coccifera L. to Mediterranean stress conditions. Trees 2002, 16, 504–510. [Google Scholar] [CrossRef][Green Version]
- Manetas, Y. The importance of being hairy: The adverse effects of hair removal on stem photosynthesis of Verbascum speciosum are due to solar UV-B radiation. New Phytol. 2003, 158, 503–508. [Google Scholar] [CrossRef]
- Ehleringer, J.R.; Mooney, H.A. Leaf hairs: Effects on physiological activity and adaptive value to a desert shrub. Oecologia 1978, 37, 183–200. [Google Scholar] [CrossRef] [PubMed]
- Ehleringer, J.R.; Björkman, O. A comparison of photosynthetic characteristics of Encelia species possessing glabrous and pubescent leaves. Plant Physiol. 1978, 62, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.K.; Nobel, P.S. Influences of seasonal changes in leaf morphology on water-use efficiency for three desert broadleaf shrubs. Ecology 1977, 58, 1033–1043. [Google Scholar] [CrossRef]
- Gates, D.M.; Keegan, H.J.; Schleter, J.C.; Weidner, V.R. Spectral properties of plants. Appl. Opt. 1965, 4, 11–20. [Google Scholar] [CrossRef]
- Ehleringer, J.R. Changes in leaf characteristics of species along elevational gradients in the Wasatch Front, Utah. Am. J. Bot. 1988, 75, 680–689. [Google Scholar] [CrossRef] [PubMed]
- Clark, C. Phylogeny and Adaptation in the Encelia Alliance (Asteraceae: Helliantheae). Aliso 1998, 17, 89–98. [Google Scholar] [CrossRef][Green Version]
- Ehleringer, J.R. Comparative ecophysiology of Encelia farinosa and Encelia frutescens. Energy-balance considerations. Oecologia 1988, 76, 553–561. [Google Scholar] [CrossRef]
- Ehleringer, J.R. The Influence of Water Stress and Temperature on Leaf Pubescence Development in Encelia farinosa. Am. J. Bot. 1982, 69, 670–675. [Google Scholar] [CrossRef]
- Ehleringer, J.R.; Björkman, O.; Mooney, H.A. Leaf pubescence: Effects on absorptance and photosynthesis in a desert shrub. Science 1976, 23, 376–377. [Google Scholar] [CrossRef] [PubMed]
- Ehleringer, J.R. Characterization of a glabrate Encelia farinosa mutant: Morphology, ecophysiology, and field observations. Oecologia 1983, 57, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Givnish, T.J. Comparative studies of leaf form: Assessing the relative roles of selective pressures and phylogenetic constraints. New Phytol. 1987, 106, 131–160. [Google Scholar] [CrossRef]
- Ackerly, D.D.; Knight, C.A.; Weiss, S.B.; Barton, K.; Starmer, K.P. Leaf size, specific leaf area and microhabitat distribution of woody plants in a California chaparral: Contrasting patterns in species level and community level analyses. Oecologia 2002, 130, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Peppe, D.J.; Royer, D.L.; Cariglino, B.; Oliver, S.Y.; Newman, S.; Leight, E.; Enikolopov, G.; Fernandez-Burgos, M.; Herrera, F.; Adams, J.M.; et al. Sensitivity of leaf size and shape to climate: Global patterns and paleoclimatic applications. New Phytol. 2011, 190, 724–739. [Google Scholar] [CrossRef] [PubMed]
- Baldocchi, D.D.; Xu, L. What limits evaporation from Mediterranean oak woodlands—the supply of moisture in the soil, physiological control by plants or the demand by the atmosphere? Adv. Water Resour. 2007, 30, 2113–2122. [Google Scholar] [CrossRef]
- Yates, M.J.; Verboom, G.A.; Rebelo, A.G.; Cramer, M.D. Ecophysiological significance of leaf size variation in Proteaceae from the Cape Floristic Region. Funct. Ecol. 2010, 24, 485–492. [Google Scholar] [CrossRef]
- Roth-Nebelsick, A. Computer-based analysis of steady-state and transient heat transfer of small-sized leaves by free and mixed convection. Plant Cell Environ. 2001, 24, 631–640. [Google Scholar] [CrossRef]
- Givnish, T.J.; Vermeij, G.J. Sizes and Shapes of Liane Leaves. Am. Nat. 1976, 110, 743–778. [Google Scholar] [CrossRef]
- Parkhurst, D.F.; Loucks, O.L. Optimal Leaf Size in Relation to Environment. J. Ecol. 1972, 60, 505–537. [Google Scholar] [CrossRef]
- Schuepp, P.H. Leaf boundary layers. New Phytol. 1993, 125, 477–507. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Cochard, H.; Barredo, G.; Villarroya, D.; Gil-Pelegrín, E. Hydraulic traits are associated with the distribution range of two closely related Mediterranean firs, Abies alba Mill. and Abies pinsapo Boiss. Tree Physiol. 2011, 31, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Barrón, E.; Camarero, J.J.; Vilagrosa, A.; Gil-Pelegrín, E. Morphological and physiological divergences within Quercus ilex support the existence of different ecotypes depending on climatic dryness. Ann. Bot. 2014, 114, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Esteso-Martínez, J.; Valladares, F.; Camarero, J.J.; Gil-Pelegrín, E. Crown architecture and leaf habit are associated with intrinsically different light-harvesting efficiencies in Quercus seedlings from contrasting environments. Ann. For. Sci. 2006, 63, 511–518. [Google Scholar] [CrossRef]
- Poorter, H.; Remkes, C. Leaf area ratio and net assimilation rate of 24 wild species differing in relative growth rate. Oecologia 1990, 83, 553–559. [Google Scholar] [CrossRef]
- Sisó, S.; Camarero, J.J.; Gil-Pelegrín, E. Relationship between hydraulic resistance and leaf morphology in broadleaf Quercus species: A new interpretation of leaf lobation. Trees 2001, 15, 341–345. [Google Scholar] [CrossRef]
- Balding, F.R.; Cunningham, L.G. A comparison of heat transfer characteristics of simple and pinnate leaf models. Bot. Gaz. 1976, 137, 65–74. [Google Scholar] [CrossRef]
- Vogel, S. “Sun Leaves” and “Shade Leaves”: Differences in Convective Heat Dissipation. Ecology 1968, 49, 1203–1204. [Google Scholar] [CrossRef]
- Givnish, T.J. Adaptive significance of compound leaves with particular reference to tropical trees. In Tropical Trees as Living Systems; Tomlinson, P.B., Zimmerman, M.H., Eds.; Cambridge University Press: Cambridge, UK, 1978; pp. 351–380. [Google Scholar]
- Stowe, L.G.; Brown, J.L. A geographic perspective on the ecology of compound leaves. Evolution 1981, 35, 818–821. [Google Scholar] [CrossRef]
- Talbert, C.M.; Holch, A.E. A study of the lobbing of sun and shade leaves. Ecology 1957, 38, 655–658. [Google Scholar] [CrossRef]
- Parkhurst, D.F.; Duncan, P.R.; Gates, D.M.; Kreith, F. Wind tunnel modeling of convection of heat between air and broad leaves of plants. Agric. Meteorol. 1968, 5, 33–47. [Google Scholar] [CrossRef]
- Baldocchi, D.D.; Ma, S.; Rambal, S.; Misson, L.; Ourcival, J.M.; Limousin, J.M.; Papale, D. On the differential advantages of evergreenness and deciduousness in Mediterranean oak woodlands: A flux perspective. Ecol. Appl. 2010, 20, 1583–1597. [Google Scholar] [CrossRef] [PubMed]
- Givnish, T.J. On the adaptive significance of leaf form. In Topics in Plant Population Biology; Solbrig, O.T., Jain, S., Johnson, G.B., Raven, P.H., Eds.; Columbia University Press: New York, NY, USA, 1979; pp. 375–407. [Google Scholar]
- Aparecido, L.M.T.; Woo, S.; Suazo, C.; Hultine, K.R.; Blonder, B. High water use in desert plants exposed to extreme heat. Ecol. Lett. 2020, 23, 1189–1200. [Google Scholar] [CrossRef]
- Smith, W.K. Temperatures of Desert Plants: Another Perspective on the Adaptability of Leaf Size. Science 1978, 201, 614–616. [Google Scholar] [CrossRef] [PubMed]
- Lawson, T.; Davey, P.A.; Yates, S.A.; Bechtold, U.; Baeshen, M.; Baeshen, N.; Mutwakil, M.Z.; Sabir, J.; Baker, N.R.; Mullineaux, P.M. C3 photosynthesis in the desert plant Rhazya stricta is fully functional at high temperatures and light intensities. New Phytol. 2014, 201, 862–873. [Google Scholar] [CrossRef]
- Mediavilla, S.; Escudero, A. Stomatal responses to drought at a Mediterranean site: A comparative study of co-occurring woody species differing in leaf longevity. Tree Physiol. 2003, 23, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.; Ingwers, M.W.; McGuire, M.A.; Teskey, R. Increase in leaf temperature opens stomata and decouples net photosynthesis from stomatal conductance in Pinus taeda and Populus deltoides x nigra. J. Exp. Bot. 2017, 68, 1757–1767. [Google Scholar] [CrossRef] [PubMed]
- Franks, P.J.; Beerling, D.J. Maximum leaf conductance driven by CO2 effects on stomatal size and density over geologic time. Proc. Natl. Acad. Sci. USA 2009, 106, 10343–10347. [Google Scholar] [CrossRef] [PubMed]
- Peguero-Pina, J.J.; Sisó, S.; Fernández-Marín, B.; Flexas, J.; Galmés, J.; García-Plazaola, J.I.; Niinemets, Ü.; Sancho-Knapik, D.; Gil-Pelegrín, E. Leaf functional plasticity decreases the water consumption without further consequences for carbon uptake in Quercus coccifera L. under Mediterranean conditions. Tree Physiol. 2016, 36, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Scareli-Santos, C.; Sánchez-Mondragón, M.L.; González-Rodríguez, A.; Oyama, K. Foliar micromorphology of Mexican oaks (Quercus: Fagaceae). Acta Bot. Mex. 2013, 104, 31–52. [Google Scholar] [CrossRef][Green Version]
- Panahi, P.; Jamzad, Z.; Pourmajidian, M.R.; Fallah, A.; Pourhashemi, M. Foliar epidermis morphology in Quercus (subgenus Quercus, section Quercus) in Iran. Acta Bot. Croat. 2012, 71, 95–113. [Google Scholar] [CrossRef]
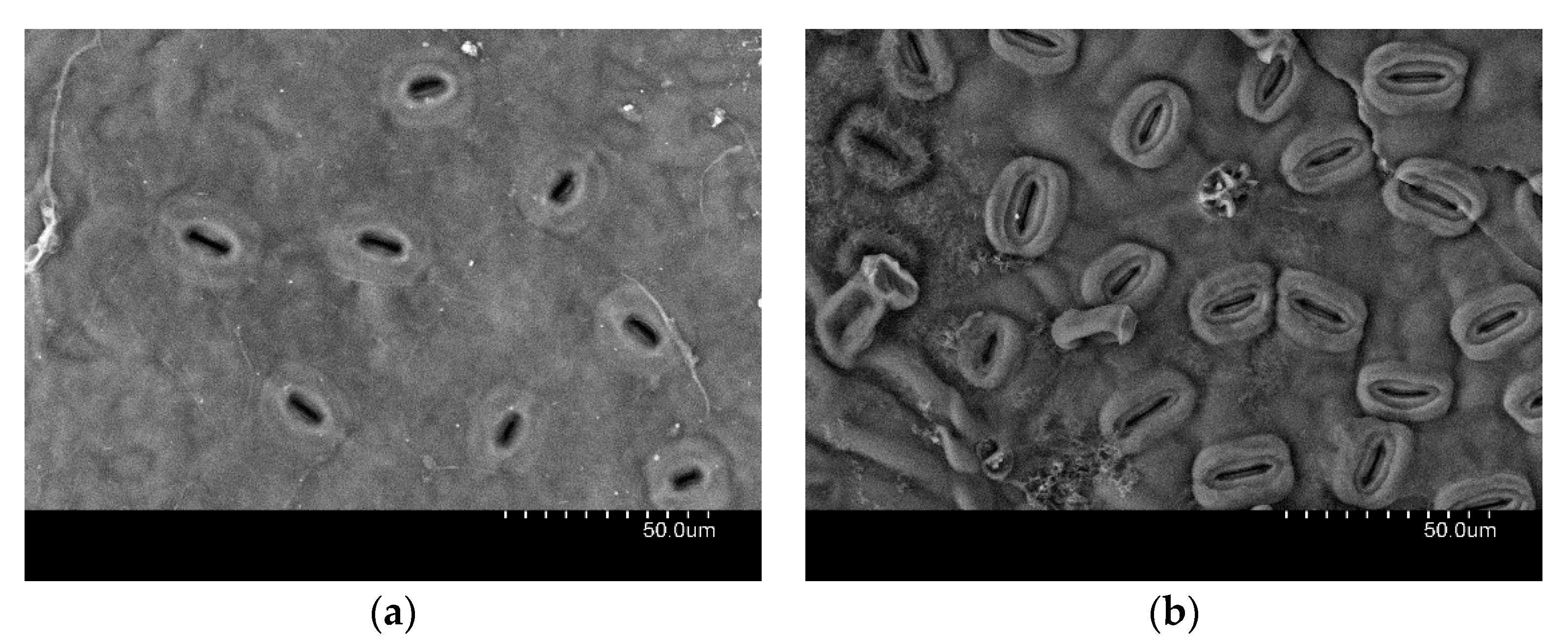
- Roth-Nebelsick, A.; Fernández, V.; Peguero-Pina, J.J.; Sancho-Knapik, D.; Gil-Pelegrín, E. Stomatal encryption by epicuticular waxes as a plastic trait modifying gas exchange in a Mediterranean evergreen species (Quercus coccifera L.). Plant Cell Environ. 2013, 36, 579–589. [Google Scholar] [CrossRef]
- Ripley, B.S.; Pammenter, N.W.; Smith, V.R. Function of leaf hairs revisited: The hair layer on leaves of Arctotheca populifolia reduces photoinhibition, but leads to higher leaf temperatures caused by lower transpiration rates. J. Plant Physiol. 1999, 155, 78–85. [Google Scholar] [CrossRef]
- Benz, B.W.; Martin, C.E. Foliar trichomes, boundary layers, and gas exchange in 12 species of epiphytic Tillandsia (Bromeliaceae). J. Plant Physiol. 2006, 163, 648–656. [Google Scholar] [CrossRef]
- Jordan, G.J.; Weston, P.H.; Carpenter, R.J.; Dillon, R.A.; Brodribb, T.J. The evolutionary relations of sunken, covered and encrypted stomata to dry habitats in Proteaceae. Am. J. Bot. 2008, 95, 521–530. [Google Scholar] [CrossRef]
- Roth-Nebelsick, A.; Hassiotou, F.; Veneklaas, E.J. Stomatal crypts have small effects on transpiration: A numerical model analysis. Plant Physiol. 2009, 151, 2018–2027. [Google Scholar] [CrossRef]
- García-Plazaola, J.I.; Esteban, R.; Hormaetxe, K.; Fernández-Marín, B.; Becerril, J.M. Photoprotective responses of Mediterranean and Atlantic trees to the extreme heat-wave of summer 2003 in Southwestern Europe. Trees 2008, 22, 385–392. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Morales, F.; Flexas, J.; Gil-Pelegrín, E. Differential photosynthetic performance and photoprotection mechanisms of three Mediterranean evergreen oaks under severe drought stress. Funct. Plant Biol. 2009, 36, 453–462. [Google Scholar] [CrossRef]

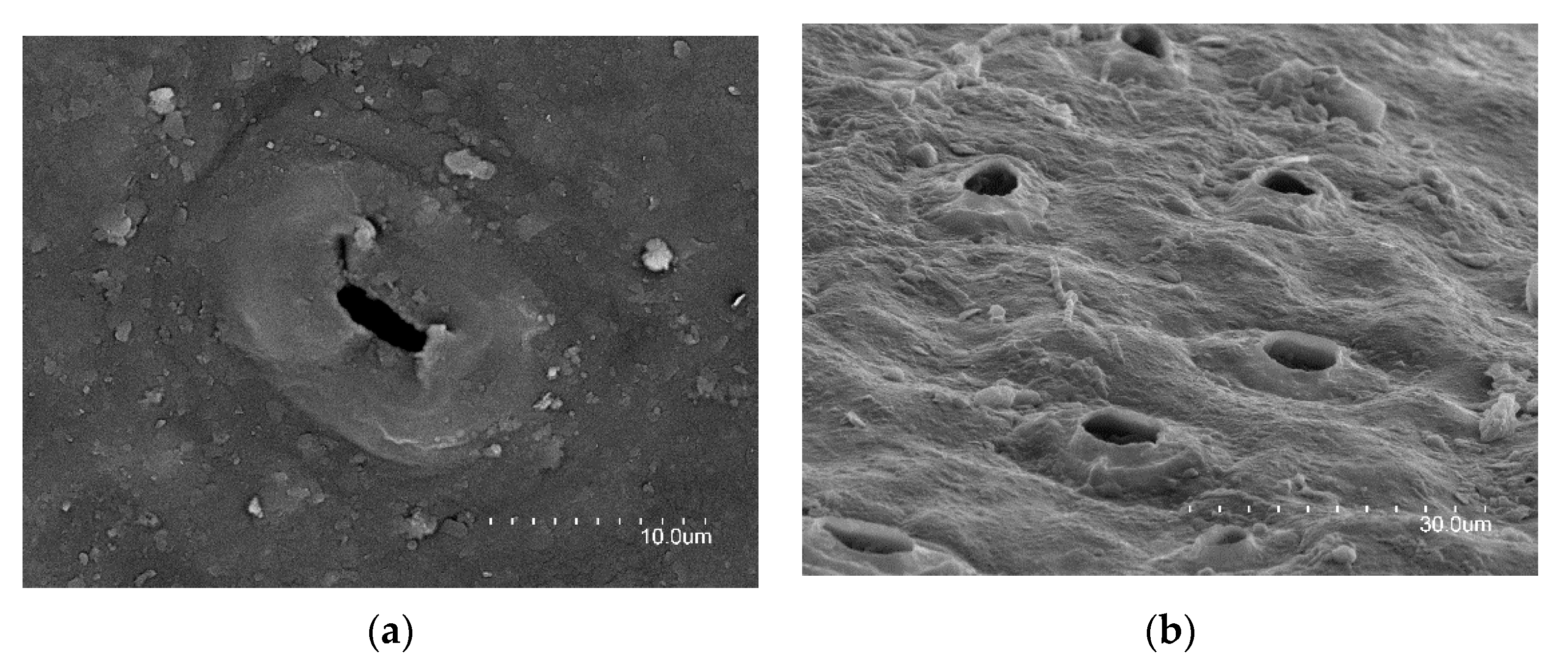
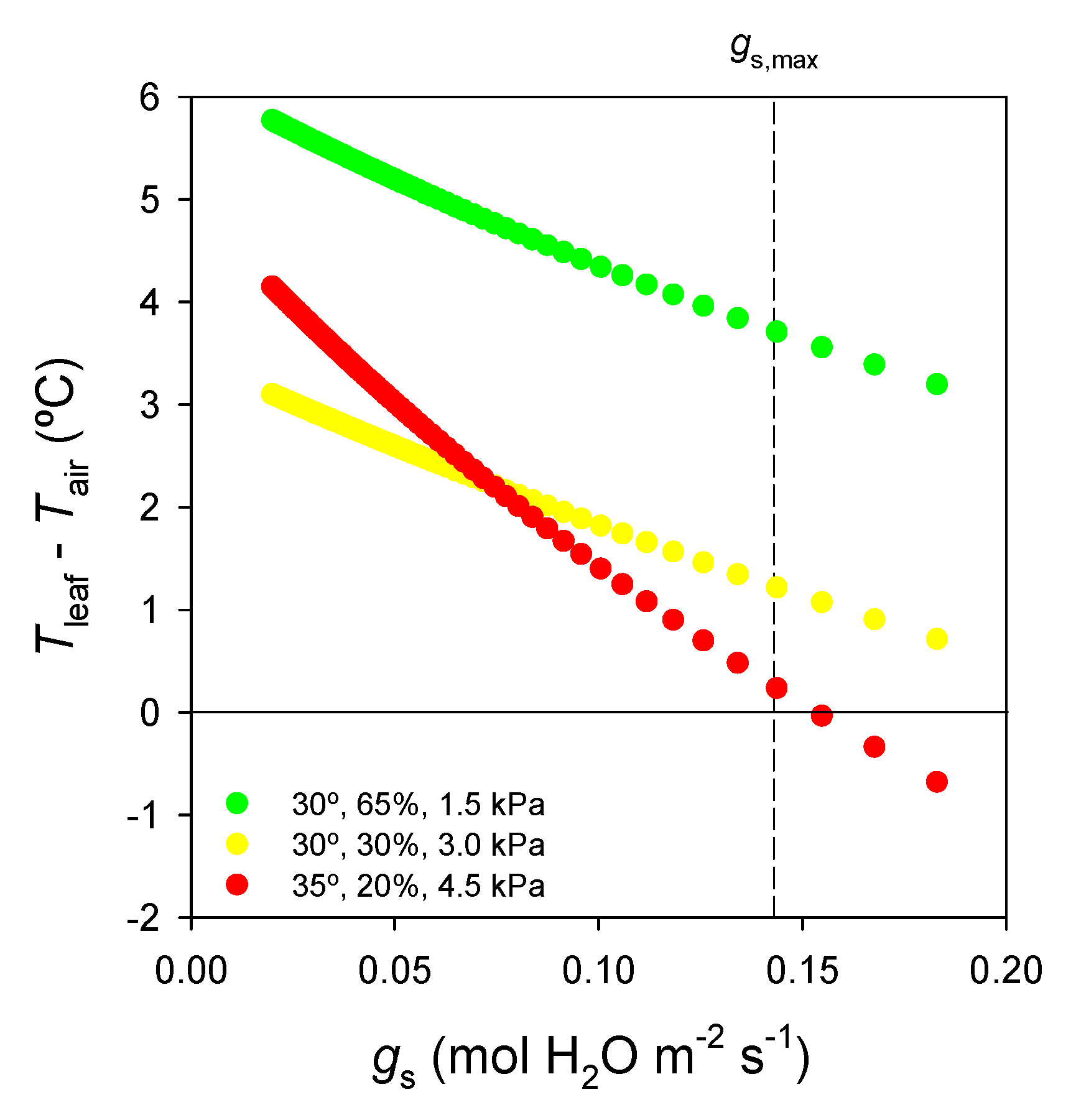
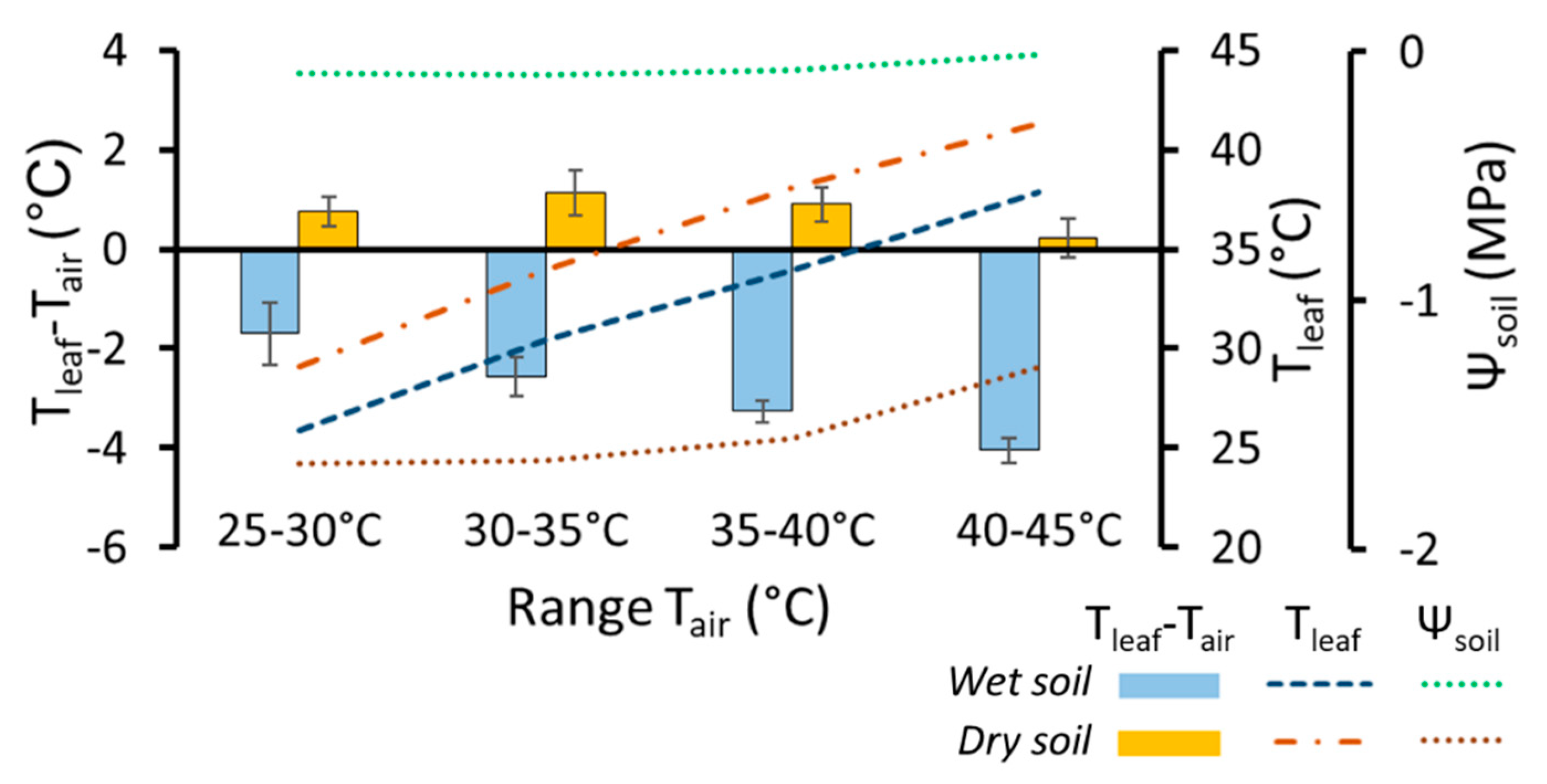
| Name of the Strategy according to Different Authors | ||||||
|---|---|---|---|---|---|---|
| Description | Levitt (1980) | Turner (1986) | Kozlowski et al. (1991) | Jones (1992) | Larcher (1995) | Volaire (2018) |
| Species that escape stress conditions (or complete their vegetative cycle before drought stress) | Escape | Escape | Avoidance | Avoidance | Drought evading | Dehydration escape |
| Species that avoid plant/cellular dehydration | Avoidance | Tolerance with high water potential | Dehydration postponement (avoidance) | Avoidance | Desiccation avoidant | Dehydration avoidance |
| Species that tolerate plant/cellular dehydration | Tolerance | Tolerance with low water potential | Dehydration tolerance | Tolerance | Desiccation tolerant | Dehydration tolerance |
| VPD (kPa) | Tleaf − Tair (°C) | Tleaf (°C) | E (mmol H2O m−2·s−1) |
|---|---|---|---|
| 1.5 | 3.6 | 33.6 | 33.3 |
| 3.0 | 1.1 | 31.1 | 28.9 |
| 4.5 | 0.0 | 35.0 | 79.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peguero-Pina, J.J.; Vilagrosa, A.; Alonso-Forn, D.; Ferrio, J.P.; Sancho-Knapik, D.; Gil-Pelegrín, E. Living in Drylands: Functional Adaptations of Trees and Shrubs to Cope with High Temperatures and Water Scarcity. Forests 2020, 11, 1028. https://doi.org/10.3390/f11101028
Peguero-Pina JJ, Vilagrosa A, Alonso-Forn D, Ferrio JP, Sancho-Knapik D, Gil-Pelegrín E. Living in Drylands: Functional Adaptations of Trees and Shrubs to Cope with High Temperatures and Water Scarcity. Forests. 2020; 11(10):1028. https://doi.org/10.3390/f11101028
Chicago/Turabian StylePeguero-Pina, José Javier, Alberto Vilagrosa, David Alonso-Forn, Juan Pedro Ferrio, Domingo Sancho-Knapik, and Eustaquio Gil-Pelegrín. 2020. "Living in Drylands: Functional Adaptations of Trees and Shrubs to Cope with High Temperatures and Water Scarcity" Forests 11, no. 10: 1028. https://doi.org/10.3390/f11101028
APA StylePeguero-Pina, J. J., Vilagrosa, A., Alonso-Forn, D., Ferrio, J. P., Sancho-Knapik, D., & Gil-Pelegrín, E. (2020). Living in Drylands: Functional Adaptations of Trees and Shrubs to Cope with High Temperatures and Water Scarcity. Forests, 11(10), 1028. https://doi.org/10.3390/f11101028









