The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes
Abstract
1. Introduction
2. Materials and Methods
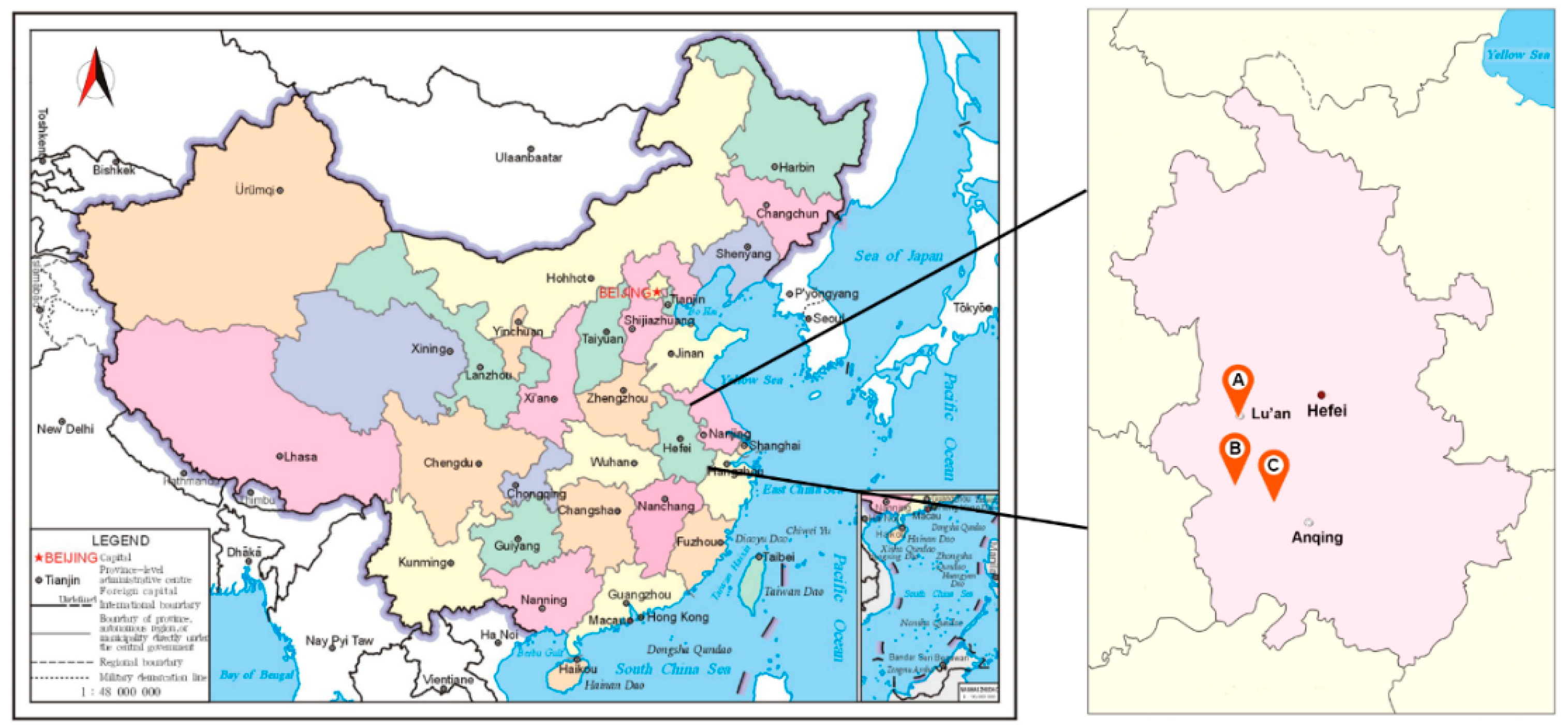
2.1. Plant Material
2.2. Experimental Design
2.3. Ecological Factors
2.3.1. Climate Factors
2.3.2. Soil Factors
2.4. Determination of Main Medicinal Components
2.4.1. Determination of Polysaccharide Content
2.4.2. Determination of Total Alkaloid Content
2.4.3. Determination of Total Flavonoid Content
2.5. Statistical Analysis
3. Results
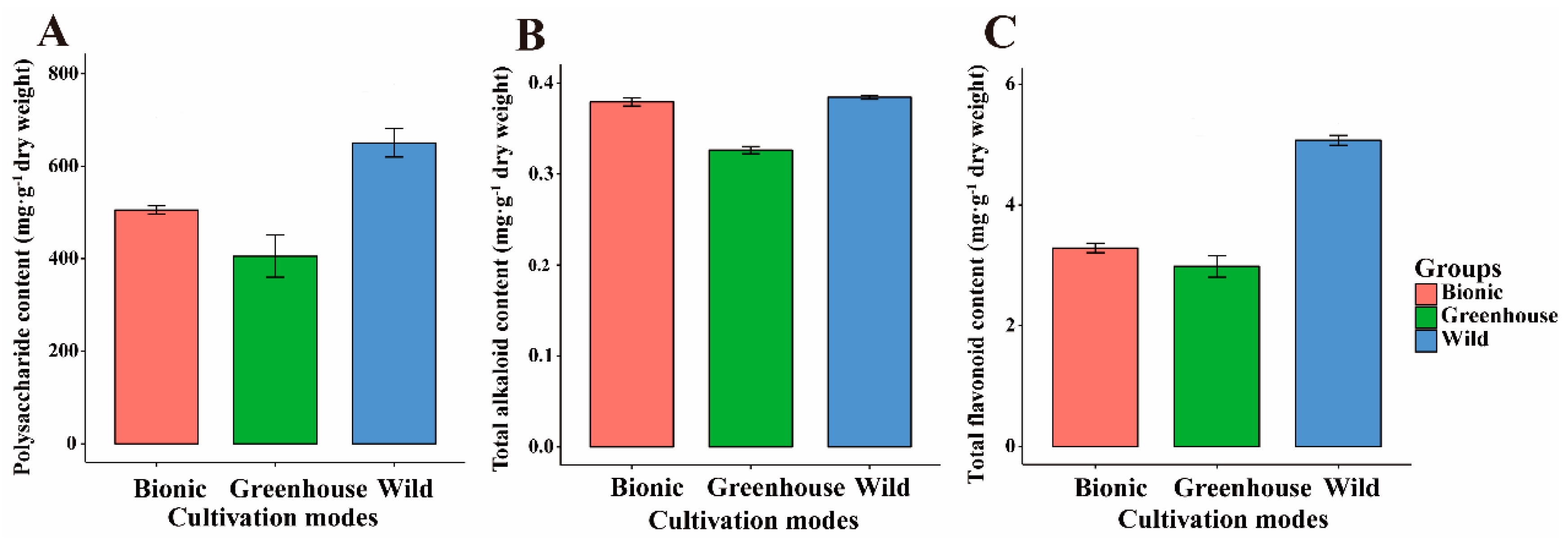
3.1. Main Medicinal Components Variation in Different Cultivation Modes
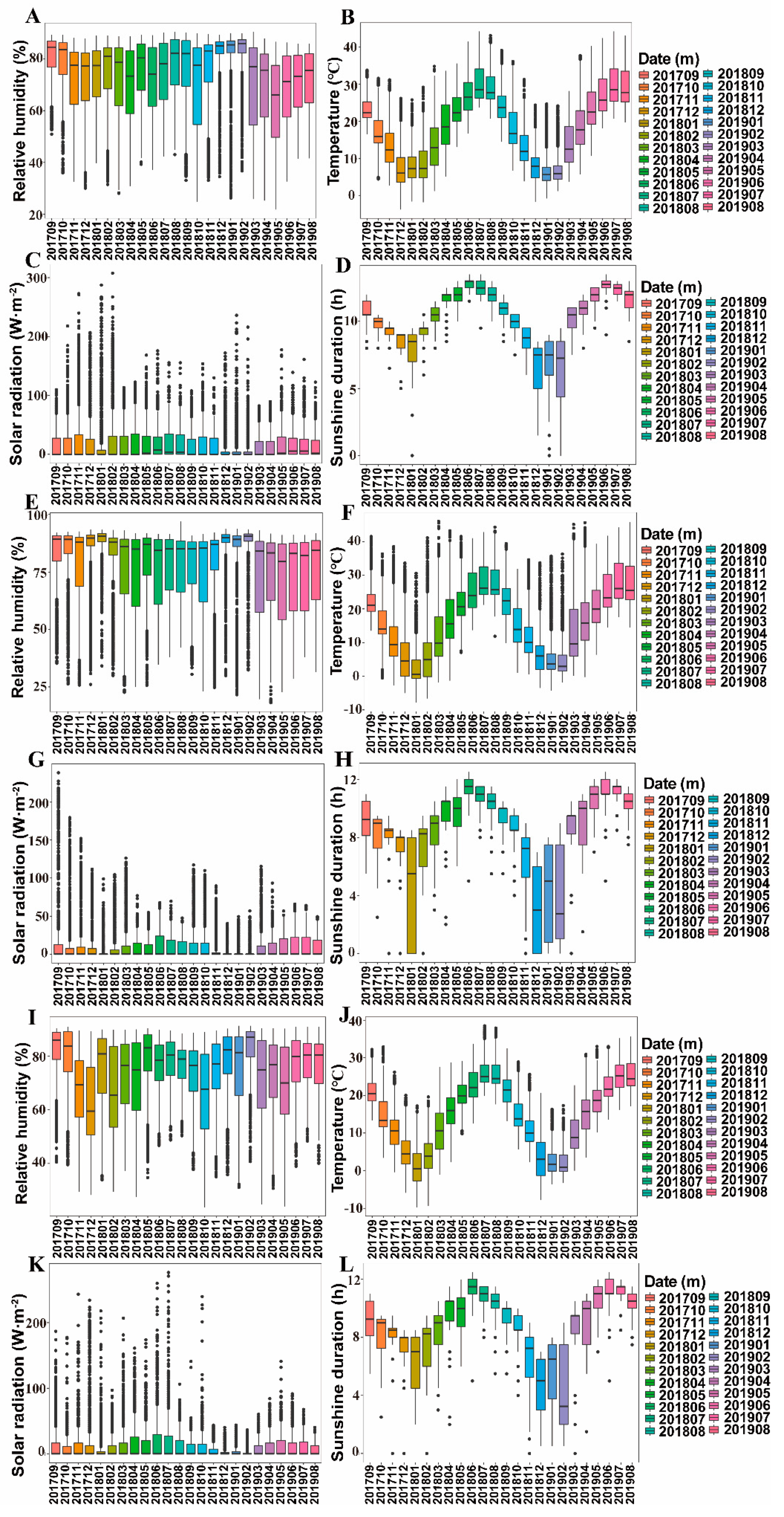
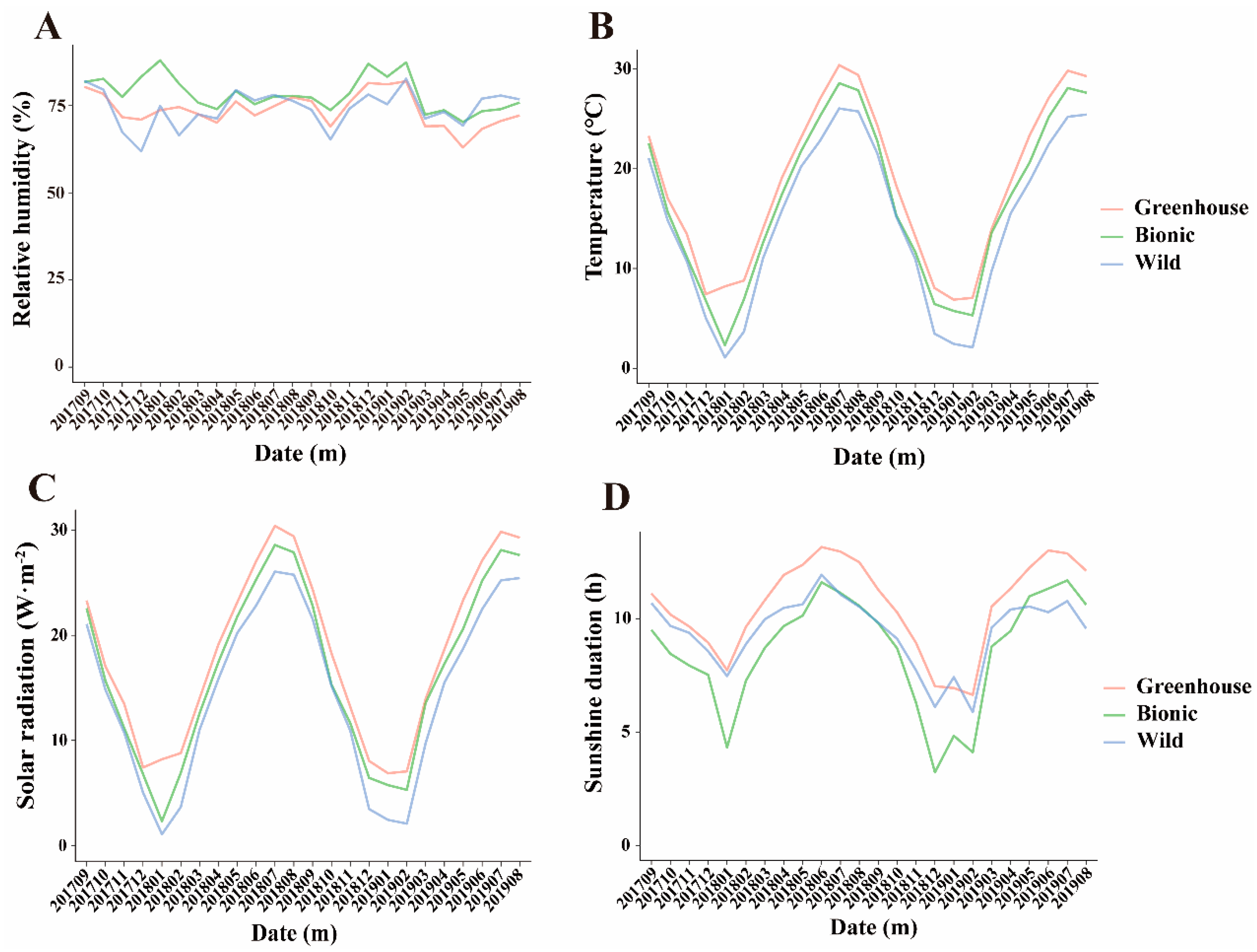
3.2. Ecological Factors Variation in Different Cultivation Modes
3.3. Ecological Factors Influencing the Main Medicinal Components
3.3.1. LDA
3.3.2. CA (Correlation Analysis)
3.3.3. PCA of Ecological Factors
3.3.4. Stepwise Multiple Linear Regression (SMLR) Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hou, B.; Tian, M.; Luo, J.; Ji, Y.; Xue, Q. Genetic diversity assessment and ex situ conservation strategy of the endangered Dendrobium officinale (orchidaceae) using new trinucleotide microsatellite markers. Plant Syst. Evolut. 2012, 298, 1483–1491. [Google Scholar] [CrossRef]
- Hou, B.; Luo, J.; Zhang, Y.; Niu, Z.; Xue, Q.; Ding, X. Iteration expansion and regional evolution: Phylogeography of Dendrobium officinale and four related taxa in southern china. Sci. Rep. 2017, 7, 43525. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, Z.; Ding, X.; Zhou, K.; Xu, L. Differentiation of dendrobium species used as ”huangcao shihu” by rdna its sequence analysis. Planta Med. 2006, 72, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ding, X.; Chu, B.; Zhou, Q.; Ding, G.; Gu, S. Genetic diversity analysis and conservation of the endangered chinese endemic herb Dendrobium officinale kimura et migo (orchidaceae) based on aflp. Genetica 2008, 133, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.Y.; Feng, Z.Y.; Zhang, X.X.; Xu, W.; Hou, B.W.; Ding, X.Y. Genetic diversity and population structure of an endangered orchid (Dendrobium loddigesii rolfe) from china revealed by srap markers. Sci. Hortic. 2011, 129, 877–881. [Google Scholar] [CrossRef]
- Chang, C.-C.; Ku, A.F.; Tseng, Y.-Y.; Yang, W.-B.; Fang, J.-M.; Wong, C.-H. 6,8-di-c-glycosyl flavonoids from Dendrobium huoshanense. J. Nat. Prod. 2010, 73, 229–232. [Google Scholar] [CrossRef]
- Xu, J.; Han, Q.-B.; Li, S.-L.; Chen, X.-J.; Wang, X.-N. Chemistry, bioactivity and quality control of Dendrobium, a commonly used tonic herb in traditional chinese medicine. Phytochem. Rev. 2013, 12, 341–367. [Google Scholar] [CrossRef]
- National Pharmacopoeia Committee. Pharmacopoeia of the People’s Republic of China. 1 (2010); China Medical Science Press: Beijing, China, 2010. [Google Scholar]
- Cao, H.; Ji, Y.; Li, S.; Lu, L.; Tian, M.; Yang, W.; Li, H. Extensive metabolic profiles of leaves and stems from the medicinal plant Dendrobium officinale kimura et migo. Metabolites 2019, 9, 215. [Google Scholar] [CrossRef]
- Jin-Ping, S.I.; Qiao-Xian, Y.U.; Song, X.S.; Shao, W.J. Artificial cultivation modes for Dendrobium officinale. China J. Chin. Mater. Med. 2013, 38, 481–484. [Google Scholar] [CrossRef]
- Wang, H.; You, D.; Cui, X.; Guo, Y. Effect of plastic greenhouse cultivation on inoculation and growth of Cistanche tubulosa. Chin. Agric. Sci. Bull. 2012, 28, 279–283. [Google Scholar]
- Shao, Q.S.; Ye, S.Y.; Zhou, A.C.; Wang, H.Z.; Xu, J.W. Current researches and prospects of seedling propagation and cultivation modes of jinxianlian. China J. Chin. Mater. Med. 2016, 41, 160–166. [Google Scholar]
- Kebaili, Z.; Hameurlaine, S.; Fellah, O.; Djermane, M.; Gherraf, N.; Zellagui, A.; Abidi, A.; Derouiche, K. Assessment of alkaloid content and antibacterial activity of hyoscyamus albus and hyoscyamus muticus collected in two different climatic regions in Algeria. J. Biochem. Technol. 2019, 10, 1–6. [Google Scholar]
- Hanif, A.; Juahir, H.; Lananan, F.; Kamarudin, M.; Adiana, G.; Azemin, A.; Yusra, A.I. Spatial variation of melaleuca cajuputi powell essential oils. J. Fundam. Appl. Sci. 2018, 10, 139–155. [Google Scholar]
- Fellah, O.; Hameurlaine, S.; Gherraf, N.; Zellagui, A.; Ali, T.; Abidi, A.; Altun, M.; Demirtas, I.; SahinYaglioglu, A. Anti-proliferative activity of ethyl acetate extracts of tamarix gallica l. Grown at different climatic conditions in Algeria. Acta Sci. Nat. 2018, 5, 23–31. [Google Scholar] [CrossRef]
- Pu, Y.; Wang, D.; Yan, Y.; Tian, H.; Peng, B.; Qin, X.; Ma, C.; Du, C.; Hu, B.; Zhang, F. Analysis of influencing factors of secondary metabolites contents in cultivated Polygala tenuifolia. China J. Chin. Mater. Med. 2017, 42, 3167–3177. [Google Scholar]
- Guo, L.P.; Wang, S.; Zhang, J.; Yang, G.; Zhao, M.X. Effects of ecological factors on secondary metabolites and inorganic elements ofscutellaria baicalensisand analysis of geoherblism. Sci. China Life Sci. 2013, 56, 1047–1056. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Thomas, R.; Sheard, R.; Moyer, J. Comparison of conventional and automated procedures for nitrogen, phosphorus, and potassium analysis of plant material using a single digestion 1. Agron. J. 1967, 59, 240–243. [Google Scholar] [CrossRef]
- Lu, R. Analysis methods of soil agricultural chemistry. China Agric. Sci. Technol. 2000, 107, 147–150. [Google Scholar]
- Huang, Y.; Ye, Y.; Yang, S. Feasibility of no3-n determination by dual wavelength spectrophotometric method. Chin. Agric. Sci. Bull. 2009, 25, 43–45. [Google Scholar]
- Yan, J.; Li, K.; Peng, X.; Huang, Z.; Liu, S.; Zhang, Q. The mechanism for exclusion of pinus massoniana during the succession in subtropical forest ecosystems: Light competition or stoichiometric homoeostasis? Sci. Rep. 2015, 5, 10994. [Google Scholar] [CrossRef] [PubMed]
- Abliz, A.; Halik, Ü.; Welp, M.; Zhang, L.-X. Effects of shelterbelt afforestation on soil properties in kökyar, nw china. Int. J. Appl. Environ. Sci. 2015, 10, 2017–2036. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Yang, F.; Wei, N.-N.; Gao, R.; Piao, X.-C.; Lian, M.-L. Effect of several medium factors on polysaccharide and alkaloid accumulation in protocorm-like bodies ofdendrobium candidumduring bioreactor culture. Acta Physiol. Plant. 2015, 37, 94. [Google Scholar] [CrossRef]
- Yuan, Y.; Yu, M.; Zhang, B.; Liu, X.; Zhang, J. Comparative nutritional characteristics of the three major chinese dendrobium species with different growth years. PLoS ONE 2019, 14, e0222666. [Google Scholar] [CrossRef]
- Bush, L.P.; Wilkinson, H.H.; Schardl, C.L. Bioprotective alkaloids of grass-fungal endophyte symbioses. Plant Physiol. 1997, 114, 1–7. [Google Scholar] [CrossRef]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Hossain, M.A.; Rahman, S.M. Total phenolics, flavonoids and antioxidant activity of tropical fruit pineapple. Food Res. Int. 2011, 44, 672–676. [Google Scholar] [CrossRef]
- Latt, Z.Z.; Wittenberg, H. Improving flood forecasting in a developing country: A comparative study of stepwise multiple linear regression and artificial neural network. Water Resour. Manag. 2014, 28, 2109–2128. [Google Scholar] [CrossRef]
- Rahimmalek, M.; Heidari, E.F.; Ehtemam, M.H.; Mohammadi, S. Essential oil variation in iranian ajowan (trachyspermum ammi (l.) sprague) populations collected from different geographical regions in relation to climatic factors. Ind. Crops Prod. 2017, 95, 591–598. [Google Scholar] [CrossRef]
- Valls, J.; Richard, T.; Trotin, F.; Monti, J.-P.; Mérillon, J.-M.; Vitrac, X. Carbon-14 biolabeling of flavanols and chlorogenic acids in crataegus monogyna cell suspension cultures. Food Chem. 2007, 105, 879–882. [Google Scholar] [CrossRef]
- Gairola, S.; Shariff, N.M.; Bhatt, A. Influence of climate change on production of secondary chemicals in high altitude medicinal plants: Issues needs immediate attention. J. Med. Plants Res. 2010, 4, 1825–1829. [Google Scholar]
- Yuan, Y.; Zhang, J.; Liu, X.; Meng, M.; Wang, J.; Lin, J. Tissue-specific transcriptome for Dendrobium officinale reveals genes involved in flavonoid biosynthesis. Genomics 2019, 19, 30700–30701. [Google Scholar] [CrossRef]
- Shen, C.; Guo, H.; Chen, H.; Shi, Y.; Meng, Y.; Lu, J.; Feng, S.; Wang, H. Identification and analysis of genes associated with the synthesis of bioactive constituents in Dendrobium officinale using rna-seq. Sci. Rep. 2017, 7, 187. [Google Scholar] [CrossRef] [PubMed]
- Peñuelas, J.; Llusià, J. Effects of carbon dioxide, water supply, and seasonality on terpene content and emission by rosmarinus officinalis. J. Chem. Ecol. 1997, 23, 979–993. [Google Scholar] [CrossRef]
- Zhong, G.; Zhang, L.; Zhang, L.; Yang, R.; Ding, C. A study on photosynthetic characteristics of different Salvia miltiorrhiza varieties. Acta Pratac. Sin. 2011, 20, 116–122. [Google Scholar]
- Peng, X.; Zhang, S. Competition for light and crop productivity in an agro-forestry system in the hilly region, Shangluo, China. Acta Ecol. Sin. 2012, 32, 2692–2698. [Google Scholar] [CrossRef]
- Liu, J.; Shu, Z.; Liang, Z.; Shi, X.; Zhang, Y. Uv-b radiation effects on phenolic changes and antioxidant activity in Salvia miltiorrhiza bunge leaf. J. Food Agric. Environ. 2013, 11, 2788–2791. [Google Scholar]
- Dong, J.E.; Ma, X.H.; Wei, Q.; Peng, S.B.; Zhang, S.C. Effects of growing location on the contents of secondary metabolites in the leaves of four selected superior clones of eucommia ulmoides. Ind. Crops Prod. 2011, 34, 1607–1614. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, D.; Liang, Z.; Liu, J.; Yan, K.; Zhu, Y.; Yang, S. Climatic factors control the geospatial distribution of active ingredients in Salvia miltiorrhiza bunge in china. Sci. Rep. 2019, 9, 904. [Google Scholar] [CrossRef]
- Huang, L.; Wentao, L.I.; Wang, Z.; Juan, F.U.; Chen, S. Correlative study between chemical constituents and ecological factors of notopterygii rhizoma et radix of endangered plateau plant. Acta Ecol. Sin. 2013, 33, 7667–7678. [Google Scholar]
- Pan, H.; Fang, C.; Zhou, T.; Wang, Q.; Chen, J. Accumulation of calycosin and its 7-o-β-d-glucoside and related gene expression in seedlings of astragalus membranaceus bge. Var. Mongholicus (bge.) hsiao induced by low temperature stress. Plant Cell Rep. 2007, 26, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Nam, M.H.; Heo, E.J.; Kim, J.Y.; Kim, S.I.; Kwon, K.H.; Seo, J.B.; Kwon, O.; Yoo, J.S.; Park, Y.M. Proteome analysis of the responses of panax ginseng ca meyer leaves to high light: Use of electrospray ionization quadrupole-time of flight mass spectrometry and expressed sequence tag data. Proteomics 2003, 3, 2351–2367. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.p.; Sui, X.x.; Sun, Q. Biological functions of secondary metabolism of medicinal plants and influences of ecological environment. Nat. Prod. Res. Dev. 2006, 18, 1027. [Google Scholar]
- Liu, W.; Liu, J.; Yin, D.; Zhao, X. Influence of ecological factors on the production of active substances in the anti-cancer plant sinopodophyllum hexandrum (royle) t.S. Ying. PLoS ONE 2015, 10, e0122981. [Google Scholar] [CrossRef]
- Shah, F.A.; Ren, Y.; Yuan, Y.J.; Fu, S.; Wang, Y.; Chen, H. Effect of plant age and geographical location on active paeonol and paeoniflorin accumulation in the roots of paeonia ostii. Pak. J. Bot. 2018, 50, 1785–1790. [Google Scholar]
- Antoniadis, V.; Levizou, E.; Shaheen, S.M.; Ok, Y.S.; Sebastian, A.; Baum, C.; Prasad, M.N.; Wenzel, W.W.; Rinklebe, J. Trace elements in the soil-plant interface: Phytoavailability, translocation, and phytoremediation–A review. Earth Sci. Rev. 2017, 171, 621–645. [Google Scholar] [CrossRef]
- Yan, L.I.; Zhou, X.; Lou, Z.; Xiao, X. Review of plant secondary metabolites and the factors that influence its accumulation. Jiangxi For. Sci. Technol. 2012, 54–60. [Google Scholar]
- Du, W.; Huang, H.; Garden, W.B. Correlation analysis of secondary metabolites and environmental factors in tripterygium wilfordii. Chin. Bull. Bot. 2008, 25, 707–713. [Google Scholar]
- Zhang, L.; Ye, Z.; Guo, Q. Effects of soil factor on active components of radix ophiopogonis. China J. Chin. Mater. Med. 2010, 35, 1372–1377. [Google Scholar]

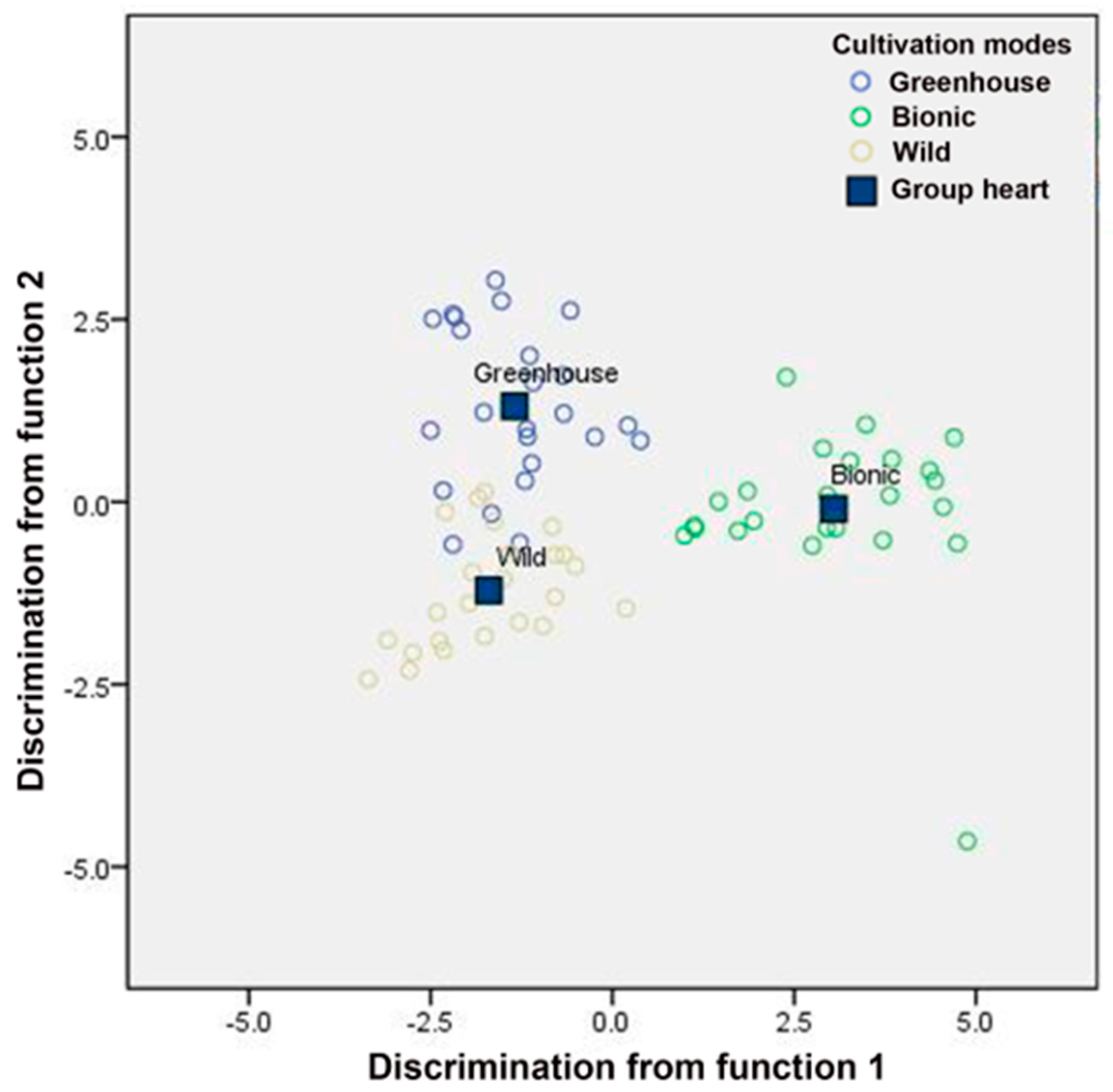
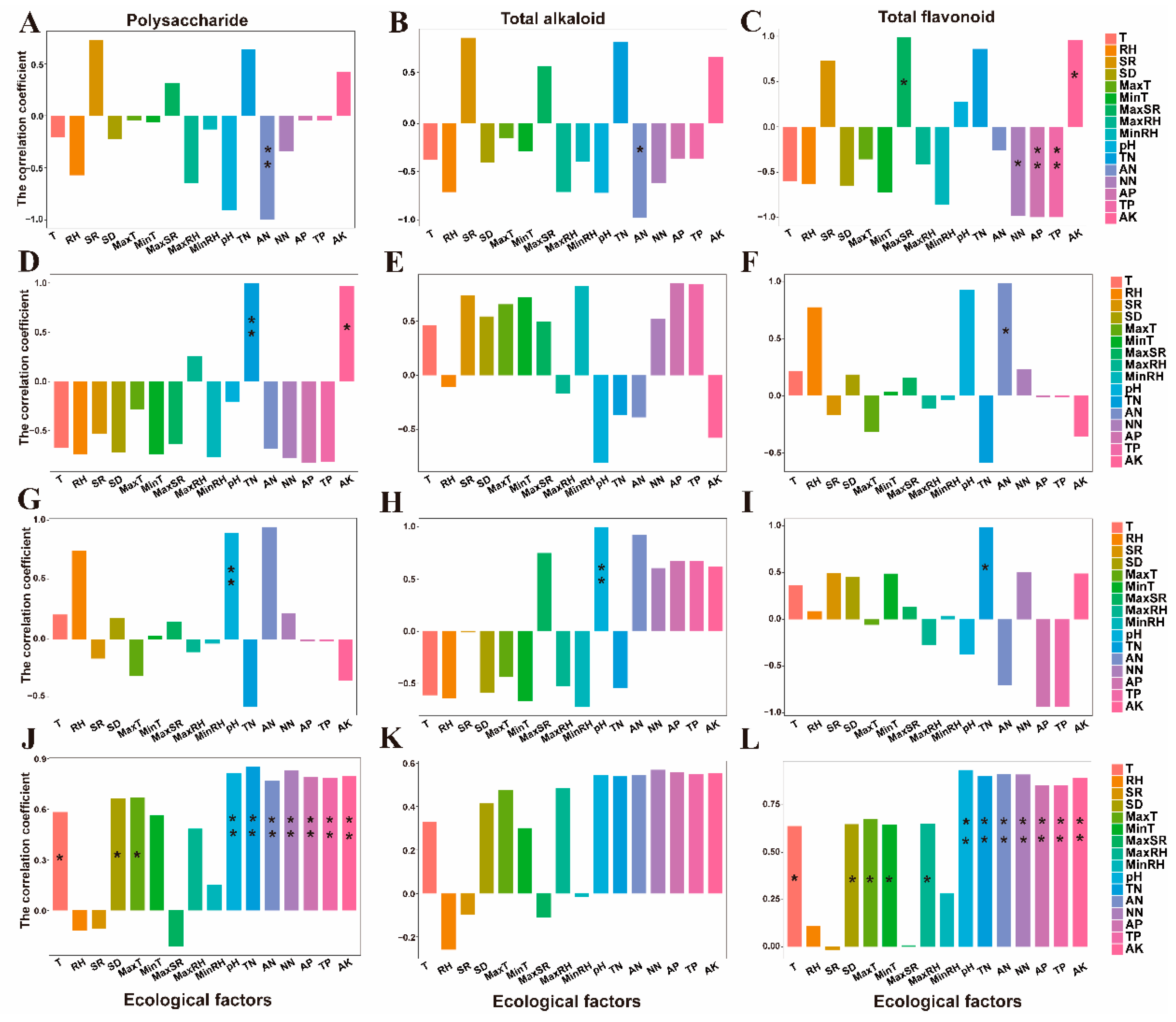
| Cultivation Modes | pH | TN | AN | NN | TP | AP | AK |
|---|---|---|---|---|---|---|---|
| Greenhouse and Bionic | 5.383 ± 0.068 | 0.307 ± 0.025 | 0.013 ± 0.001 | 0.0006 ± 0.0002 | 0.007 ± 0.006 | 0.001 ± 0.0009 | 0.011 ± 0.0009 |
| Wild | 6.287 ± 0.041 | 3.925 ± 0.775 | 0.029 ± 0.003 | 0.010 ± 0.0008 | 0.071 ± 0.134 | 0.014 ± 0.003 | 0.051 ± 0.009 |
| Principal Components | Eigenvalues | Contribution Rates (%) | Cumulative Contribution Rates (%) |
|---|---|---|---|
| 1 | 7.562 | 47.261 | 47.261 |
| 2 | 3.804 | 23.775 | 71.036 |
| 3 | 2.131 | 13.318 | 84.354 |
| 4 | 1.269 | 7.933 | 92.287 |
| Ecological Factors | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| Temperature | −0.125 | 0.978 | 0.05 | 0.035 |
| RH | −0.234 | −0.24 | −0.411 | 0.807 |
| SR | −0.218 | 0.249 | 0.85 | −0.074 |
| SD | 0.003 | 0.85 | 0.383 | −0.239 |
| MaxT | −0.531 | 0.692 | −0.236 | −0.049 |
| MinT | −0.029 | 0.946 | 0.104 | 0.191 |
| MaxSR | −0.002 | −0.167 | 0.885 | 0.138 |
| MaxRH | −0.161 | −0.254 | −0.733 | 0.228 |
| MinRH | 0.288 | 0.436 | 0.221 | 0.754 |
| pH | 0.995 | −0.066 | −0.004 | −0.001 |
| TN | 0.989 | −0.056 | −0.012 | 0.019 |
| AN | 0.994 | −0.067 | 0.002 | −0.009 |
| NN | 0.996 | −0.062 | −0.004 | 0.005 |
| AP | 0.993 | −0.058 | 0.004 | −0.004 |
| TP | 0.993 | −0.057 | 0.004 | −0.004 |
| AK | 0.992 | −0.069 | 0.001 | −0.006 |
| No. | Equation | R2 | F | P |
|---|---|---|---|---|
| Model I | Polysaccharide = 1.023 × TN + 0.287 × Max RH + 0.130 × Max T − 0.88 × Min RH | 0.918 | 199.036 | 0.019 |
| Model II | Alkaloid = 0.498 × Max RH + 0.858 × TP + 0.394 × Max T − 0.149 × Min RH − 0.181 × SD | 0.788 | 53.888 | 0.05 |
| Model III | Flavonoid = 1.464 × pH + 0.104 Max RH − 0.476 × AP | 0.973 | 863.144 | 0.004 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, Y.; Tang, X.; Jia, Z.; Li, C.; Ma, J.; Zhang, J. The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes. Forests 2020, 11, 94. https://doi.org/10.3390/f11010094
Yuan Y, Tang X, Jia Z, Li C, Ma J, Zhang J. The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes. Forests. 2020; 11(1):94. https://doi.org/10.3390/f11010094
Chicago/Turabian StyleYuan, Yingdan, Xinggang Tang, Zhaohui Jia, Chong Li, Jieyi Ma, and Jinchi Zhang. 2020. "The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes" Forests 11, no. 1: 94. https://doi.org/10.3390/f11010094
APA StyleYuan, Y., Tang, X., Jia, Z., Li, C., Ma, J., & Zhang, J. (2020). The Effects of Ecological Factors on the Main Medicinal Components of Dendrobium officinale under Different Cultivation Modes. Forests, 11(1), 94. https://doi.org/10.3390/f11010094





