The Persistence of Container Nursery Treatments on the Field Performance and Root System Morphology of Longleaf Pine Seedlings
Abstract
1. Introduction
2. Materials and Methods
2.1. Seedling Quality
2.2. Outplanting
2.3. Field and Laboratory Measurements
2.3.1. Growth and Survival
2.3.2. Root System Analysis
2.3.3. Biomass Allocation and Nutrients
2.4. Statistical Analysis
3. Results
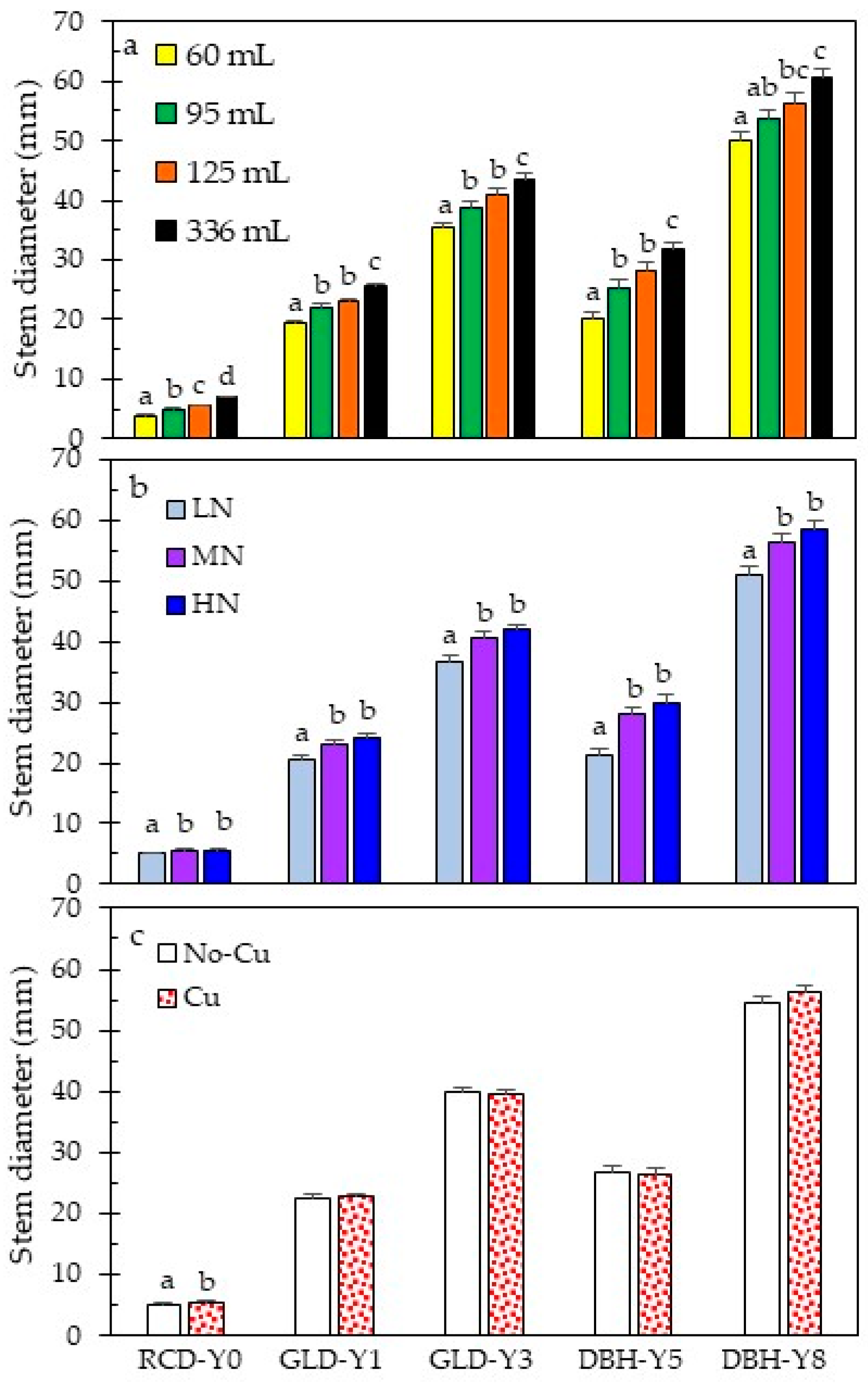
3.1. Stock Quality at Outplanting
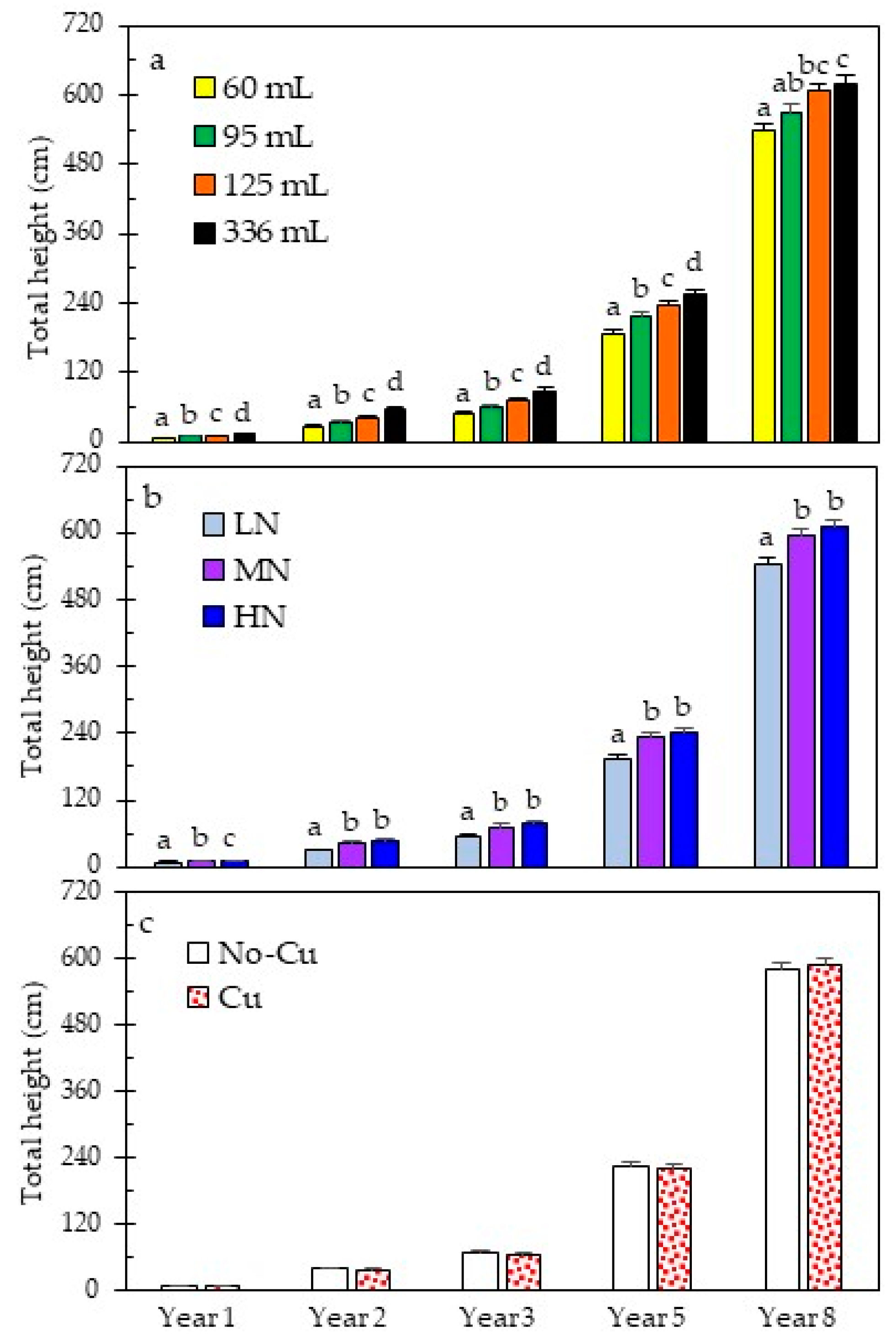
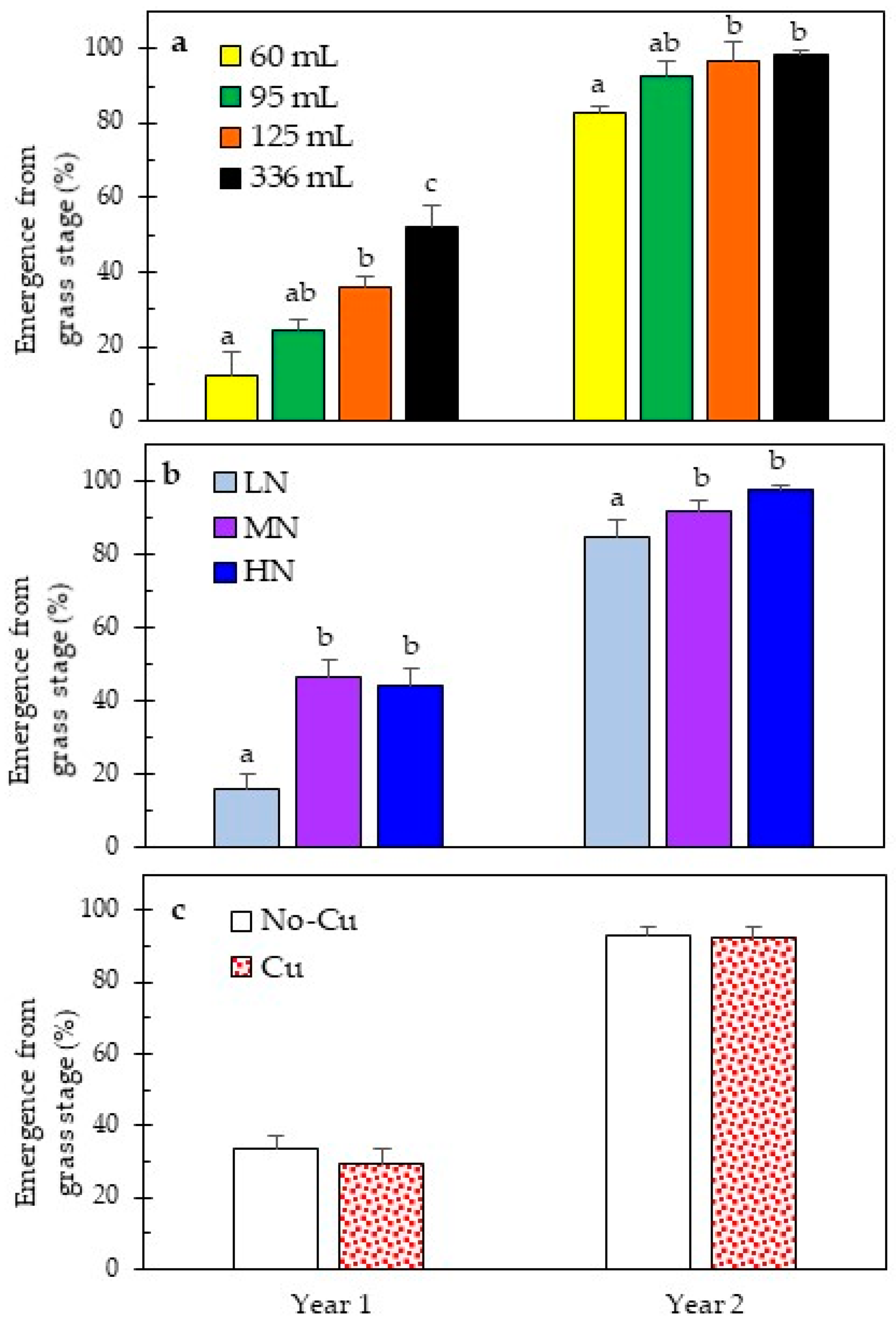
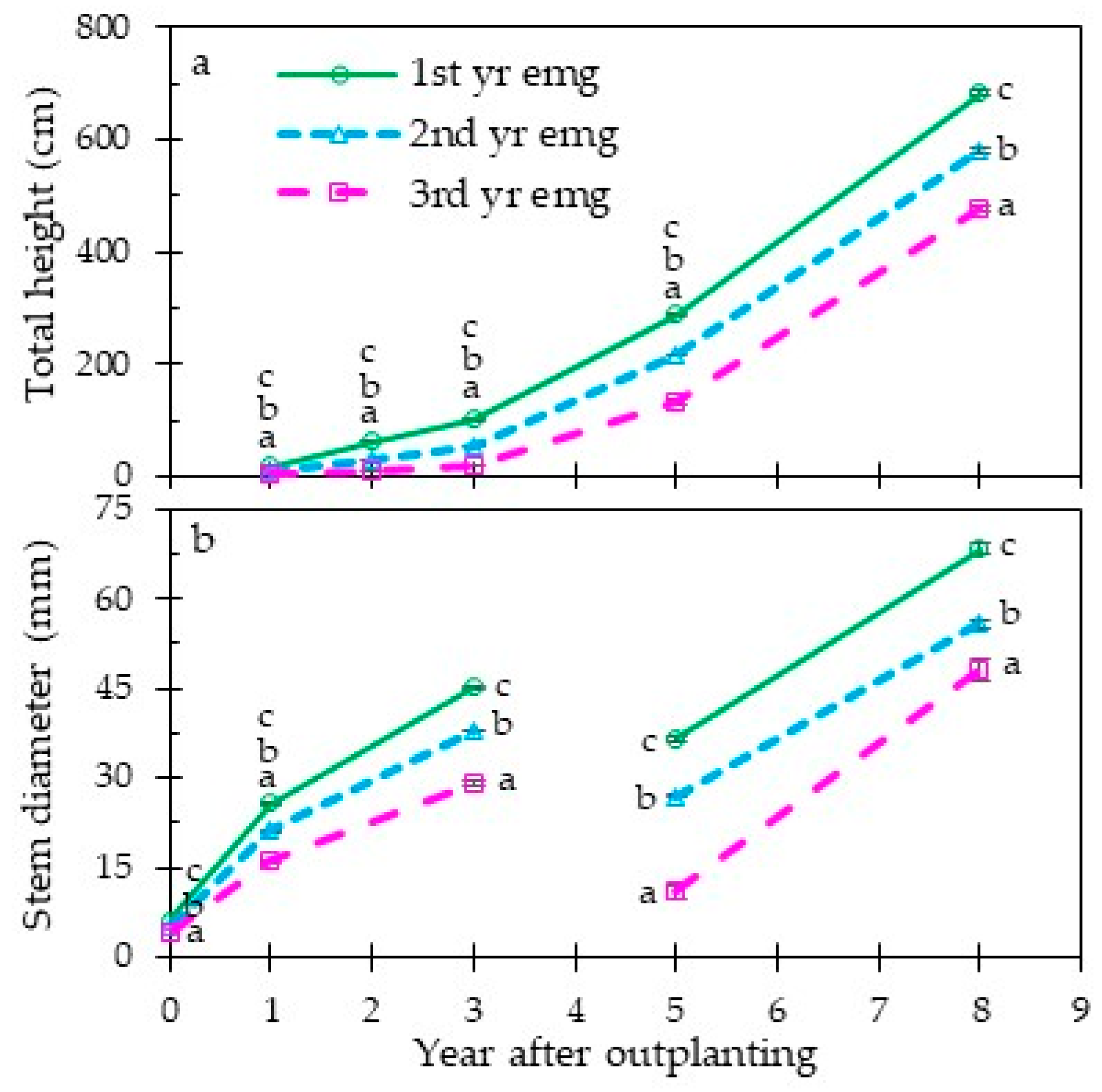
3.2. Early and Long-Term Field Performance
3.3. Seedling Physiology
3.3.1. Seedling Biomass and Allocation
3.3.2. Mineral Nutrient Concentrations and Contents
3.4. Seedling Root System
3.4.1. Vertical Anchorage
3.4.2. Plug Root Biomass and Allocation
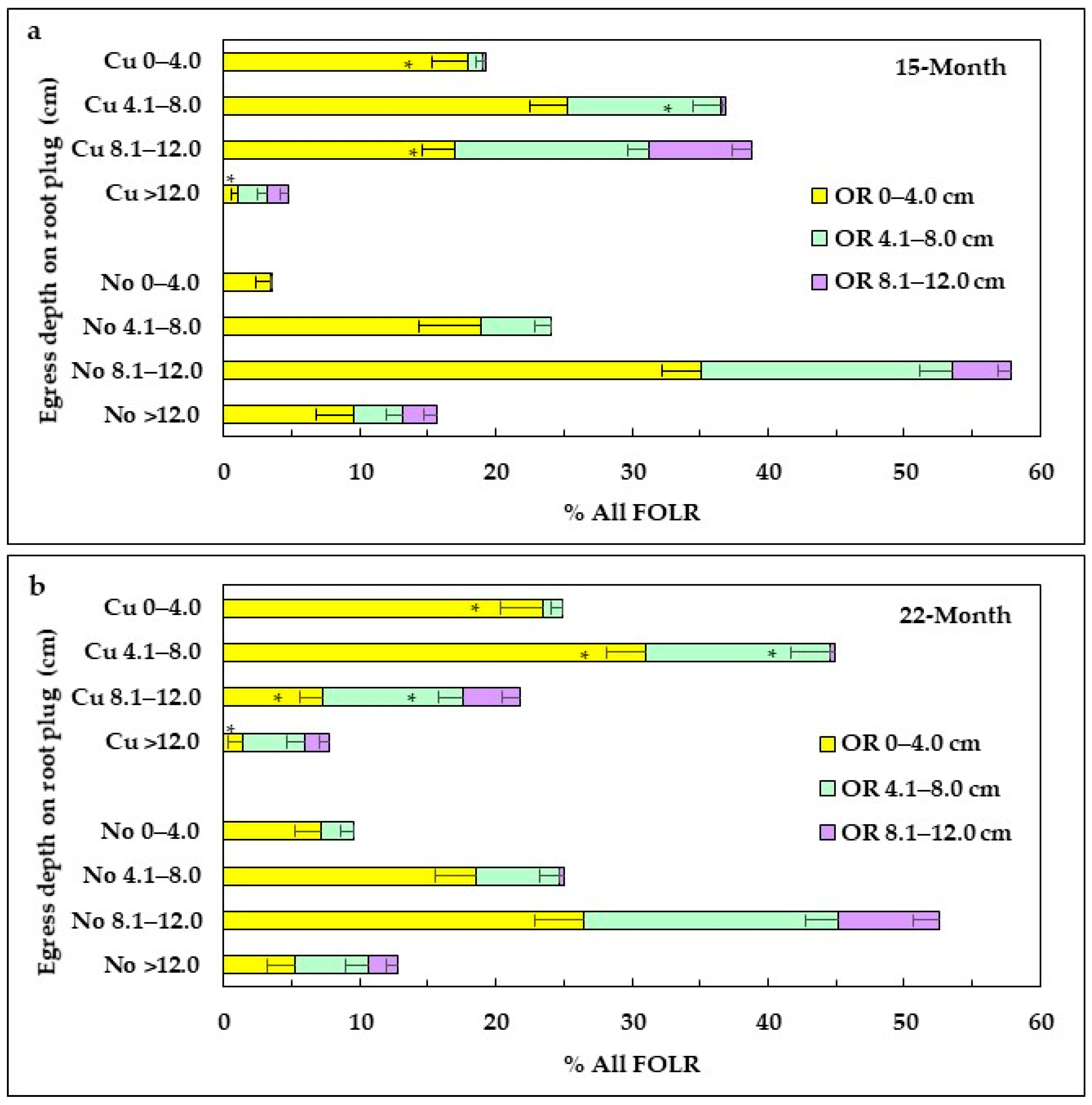
3.4.3. Morphology of FOLRs
FOLRs within the Root Plug
FOLR Outside the Root Plug
4. Discussion
4.1. Seedling Quality Attributes
4.2. Field Performance Attributes
4.3. Grass Stage Duration
4.4. Mineral Nutrition
4.5. Biomass Allocation
4.6. Root System Morphology
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Frost, C.C. Four centuries of changing landscape patterns in the longleaf pine ecosystem. In The Longleaf Pine Ecosystem: Ecology, Restoration and Management, Proceedings of the 18th Tall Timbers Fire Ecology Conference, Tallahassee, FL, USA, 30 May–2 June 1991; Hermann, S.M., Ed.; Tall Timbers Research Inc.: Tallahassee, FL, USA, 1993; pp. 17–43. [Google Scholar]
- Landers, J.L.; Van Lear, D.H.; Boyer, W.D. The longleaf pine forests of the Southeast: Requiem or renaissance? J. Forest. 1995, 93, 39–44. [Google Scholar]
- Outcalt, K.W. The longleaf pine ecosystem of the South. Native Plants J. 2000, 1, 42–53. [Google Scholar] [CrossRef]
- Frost, C. History and future of the longleaf pine ecosystem. In The Longleaf Pine Ecosystem-Ecology, Silviculture, and Restoration; Jose, S., Jokela, E.J., Miller, D.L., Eds.; Springer: New York, NY, USA, 2006; pp. 9–42. [Google Scholar]
- Barnett, J.P. Naval Stores: A History of an Early Industry Created from the South’s Forests. U.S. Department of Agriculture Forest Service; Southern Research Station: Asheville, NC, USA, 2019; p. 45. [Google Scholar]
- Jose, S.; Jokela, E.J.; Miller, D.L. The Longleaf Pine Ecosystem-Ecology, Silviculture, and Restoration; Springer: New York, NY, USA, 2006; p. 438. [Google Scholar]
- Van Lear, D.H.; Carroll, W.D.; Kapeluck, P.R.; Johnson, R. History and restoration of the longleaf pine-grassland ecosystem: Implications for species at risk. For. Ecol. Manag. 2005, 211, 150–165. [Google Scholar] [CrossRef]
- America’s Longleaf Restoration Initiative. Range-Wide Conservation Plan for Longleaf Pine. 2009. Available online: www.americanslongleaf.org (accessed on 31 July 2019).
- Gresham, C.A.; Williams, T.M.; Lipscomb, D.J. Hurricane Hugo wind damage to southeastern U.S. coastal forest tree species. Biotropica 1991, 23, 420–426. [Google Scholar] [CrossRef]
- Johnsen, K.H.; Butnor, J.R.; Kush, J.S.; Schmidtling, R.C.; Nelson, C.D. Hurricane Katrina winds damaged longleaf pine less than loblolly pine. South. J. Appl. For. 2009, 33, 178–181. [Google Scholar] [CrossRef]
- South, D.B.; Harris, S.W.; Barnett, J.P.; Hainds, M.J.; Gjerstad, D.H. Effect of container type and seedling size on survival and early height growth of Pinus palustris seedlings in Alabama, USA. For. Ecol. Manag. 2005, 204, 385–398. [Google Scholar] [CrossRef]
- Barnard, E.L.; Mayfield III, A.E. Insects and diseases of longleaf pine in the context of longleaf ecosystem restoration. In Proceedings of the 2009 Society of American Foresters Convention, Orlando, FL, USA, 30 September–4 October 2009; Society of American Foresters: Bethesda, MD, USA, 2009; p. 10. [Google Scholar]
- Lindström, A.; Rune, G. Root deformation in plantations of container-grown Scots pine trees: Effects on root growth, tree stability and stem straightness. Plant Soil 1999, 217, 29–37. [Google Scholar] [CrossRef]
- Burdett, A.N.; Coates, H.; Eremko, R.; Martin, P.A.F. Toppling in British Columbia’s lodgepole pine plantations: Significance, cause and prevention. Forest. Chron. 1986, 62, 433–439. [Google Scholar] [CrossRef]
- Balisky, A.C.; Salonius, P.; Walli, C.; Brinkman, D. Seedling roots and forest floor: Misplaced and neglected aspects of British Columbia’s reforestation effort? Forest. Chron. 1995, 71, 59–65. [Google Scholar] [CrossRef]
- Halter, M.R.; Chanway, C.P.; Harper, G.J. Growth reduction and root deformation of containerized lodgepole pine saplings 11 years after planting. For. Ecol. Manag. 1993, 56, 131–146. [Google Scholar] [CrossRef]
- Barnett, J.P.; Brissette, J.C. Producing Southern Pine Seedlings in Containers; U.S. Department of Agriculture Forest Service; Southern Forest Experiment Station: New Orleans, LA, USA, 1986; p. 71. [Google Scholar]
- Sung, S.S.; Haywood, J.D.; Zarnoch, S.J.; Sword Sayer, M.A. Long-term container effects on root system architecture of longleaf pine. In Proceedings of the 2009 Society of American Foresters Convention, Orlando, FL, USA, 30 September–4 October 2009; Society of American Foresters: Bethesda, MD, USA, 2009; p. 9. [Google Scholar]
- Burdett, A.N. Control of root morphogenesis for improved mechanical stability in container-grown lodgepole pine. Can. J. For. Res. 1978, 8, 483–486. [Google Scholar] [CrossRef]
- Sayer, M.A.S.; Haywood, J.D.; Sung, S.S. Cavity size and copper root pruning affect production and establishment of container-grown longleaf pine seedlings. For. Sci. 2009, 55, 377–389. [Google Scholar]
- Ruehle, J.L. The effect of cupric carbonate on root morphology of containerized mycorrhizal pine seedlings. Can. J. For. Res. 1985, 15, 586–592. [Google Scholar] [CrossRef]
- Krasowski, M.J. Root system modifications by nursery culture reflect on post-planting growth and development of coniferous seedlings. Forest. Chron. 2003, 79, 882–891. [Google Scholar] [CrossRef]
- Sayer, M.A.S.; Sung, S.S.; Haywood, J.D. Longleaf pine root system development and seedling quality in response to copper root pruning and cavity size. South. J. Appl. For. 2011, 35, 5–11. [Google Scholar] [CrossRef][Green Version]
- Dumroese, R.K.; Sung, S.-S.; Pinto, J.R.; Ross-Davis, A.; Scott, D.A. Morphology, gas exchange, and chlorophyll content of longleaf pine seedlings in response to rooting volume, copper root pruning, and nitrogen supply in a container nursery. New For. 2013, 44, 881–898. [Google Scholar] [CrossRef]
- Jones, M.D.; Kiiskila, S.; Flanagan, A. Field performance of pine stock types: Two-year results of a trial on interior lodgepole pine seedlings grown in StyroblocksTM, CopperblocksTM, or AirBlocksTM. BCJ Ecosyst. Manag. 2002, 2, 59–70. [Google Scholar]
- Rune, G. Slits in container wall improve root structure and stem straightness of outplanted Scots pine seedlings. Silva Fenn. 2003, 37, 333–342. [Google Scholar] [CrossRef][Green Version]
- Sung, S.S.; Haywood, J.D. Air lateral root pruning affects longleaf pine seedling root system morphology. In Proceedings of the 18th Biennial Southern Silvicultural Research Conference, Knoxville, TN, USA, 2–5 March 2015; Schweitzer, C., Clatterbuck, W.K., Oswalt, C., Eds.; U.S. Department of Agriculture Forest Service, Southern Research Station: Asheville, NC, USA, 2016; pp. 241–245. [Google Scholar]
- Grelen, H.E. May burning favors survival and early height growth of longleaf pine seedlings. South. J. Appl. For. 1983, 7, 16–20. [Google Scholar] [CrossRef]
- Nelson, L.R.; Zutter, B.R.; Gjerstad, D.H. Planted longleaf pine seedlings respond to herbaceous weed control using herbicides. South. J. Appl. For. 1985, 9, 236–240. [Google Scholar] [CrossRef]
- Haywood, J.D. Mulch and hexazinone herbicide shorten the time longleaf pine seedlings are in the grass stage and increase height growth. New For. 2000, 19, 279–290. [Google Scholar] [CrossRef]
- Ramsey, C.L.; Jose, S.; Brecke, B.J.; Merritt, S. Growth response of longleaf pine (Pinus palustris Mill.) seedlings to fertilization and herbaceous weed control in an old field in southern USA. For. Ecol. Manag. 2003, 172, 281–289. [Google Scholar] [CrossRef]
- Haywood, J.D. Influence of herbicides and felling, fertilization, and prescribed fire on longleaf pine establishment and growth through six growing seasons. New For. 2007, 33, 257–279. [Google Scholar] [CrossRef]
- Knapp, B.O.; Wang, G.G.; Walker, J.L.; Cohen, S. Effects of site preparation treatments on early growth and survival of planted longleaf pine (Pinus palustris Mill.) seedlings in North Carolina. For. Ecol. Manag. 2006, 226, 122–128. [Google Scholar] [CrossRef]
- Hu, H.; Wang, G.G.; Walker, J.L.; Knapp, B.O. Silvicultural treatments for converting loblolly pine to longleaf pine dominance: Effects on resource availability and their relationships with planted longleaf pine seedlings. For. Ecol. Manag. 2012, 282, 115–123. [Google Scholar] [CrossRef]
- Barnett, J.P.; Hainds, M.J.; Hernandez, G.A. Interim guidelines for growing longleaf seedlings in containers. In Proceedings of Workshops on Growing Longleaf Pine in Containers-1999 and 2001, Jesup, GA, USA, 21–23 September 1999 and Tifton, GA, USA 16–18 January 2001; Barnett, J.P., Dumroese, R.K., Moorhead, D.J., Eds.; U.S. Department of Agriculture Forest Service; Southern Research Station: Asheville, NC, USA, 2002; pp. 27–29. [Google Scholar]
- Dumroese, R.K.; Barnett, J.P.; Jackson, D.P.; Hainds, M.J. 2008 Interim guidelines for growing longleaf pine seedlings in container nurseries. In National Proceedings of Forest and Conservation Nursey Associations-2008, Missoula, MT, USA, 23–25 June 2008 and Asheville, NC, USA, 21–24 July 2008; Dumroese, R.K., Riley, L.E., Eds.; Tech. Coords.; U.S. Department of Agriculture Forest Service; Rocky Mountain Research Station: Fort Collins, CO, USA, 2009; pp. 101–107. [Google Scholar]
- Haywood, J.D.; Sung, S.S.; Sayer, M.A.S. Copper root pruning and container cavity size influence longleaf pine growth through five growing seasons. South. J. Appl. For. 2012, 36, 146–151. [Google Scholar] [CrossRef]
- Jackson, D.P.; Dumroese, R.K.; Barnett, J.P. Nursery response of container Pinus palustris seedlings to nitrogen supply and subsequent effects on outplanting performance. For. Ecol. Manag. 2012, 265, 1–12. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Montville, M.E.; Pinto, J.R. Using container weights to determine irrigation needs: A simple method. Nativ. Plants J. 2015, 16, 67–71. [Google Scholar] [CrossRef]
- Kormanik, P.P.; Ruehle, J.L.; Muse, H.D. Frequency distribution and heritability of first-order lateral roots in loblolly pine seedlings. For. Sci. 1990, 36, 802–814. [Google Scholar]
- Kerr, A., Jr.; Griffis, B.J.; Powell, J.W.; Edwards, J.P.; Venson, R.L.; Long, J.K.; Kilpatrick, W.W. Soil Survey of Rapides Parish Louisiana; U.S. Department of Agriculture Soil Conservation Service and Forest Service in cooperation with Louisiana State University, Louisiana Agricultural Experiment Station: Baton Rouge, LA, USA, 1980; p. 87. [Google Scholar]
- Esau, K. Anatomy of Seed Plants, 2nd ed.; John Wiley Songs: New York, NY, USA, 1977; p. 550. [Google Scholar]
- South, D.B.; Shelton, J.; Enebak, S.A. Geotropic lateral roots of container-grown longleaf pine seedlings. Nativ. Plants J. 2001, 2, 126–130. [Google Scholar] [CrossRef][Green Version]
- SAS Institute, Inc. SAS/STAT 9.1 User’s Guide; SAS Institute, Inc.: Cary, NC, USA, 2004; p. 5121. [Google Scholar]
- Dumroese, R.K.; Landis, T.D.; Pinto, J.R.; Haase, D.L.; Wilkinson, K.M.; Davis, A.S. Meeting forest restoration challenges: Using the Target Plant Concept. Reforesta 2016, 1, 37–52. [Google Scholar] [CrossRef]
- Ritchie, G.A.; Landis, T.D.; Dumroese, R.K.; Haase, D.L. Chapter 2: Assessing plant quality. In The Container Tree Nursery Manual. Volume 7: Seedling Processing, Storage, and Outplanting; Landis, T.D., Dumroese, R.K., Haase, D.L., Eds.; Agriculture Handbook 674; U.S. Department of Agriculture Forest Service: Washington, DC, USA, 2010; pp. 17–81. [Google Scholar]
- Davis, A.S.; Jacobs, D.F. Quantifying root system quality of nursery seedlings and relationship to outplanting performance. New For. 2005, 30, 295–311. [Google Scholar] [CrossRef]
- Grossnickle, S.C.; MacDonald, J.E. Why seedlings grow: Influence of plant attributes. New For. 2018, 49, 1–34. [Google Scholar] [CrossRef]
- Davis, A.S.; Ross-Davis, A.L.; Dumroese, R.K. Nursery culture impacts cold hardiness in longleaf pine (Pinus palustris) seedlings. Restor. Ecol. 2011, 19, 717–719. [Google Scholar] [CrossRef]
- Burdett, A.N.; Martin, P.A.F. Chemical root pruning of coniferous seedlings. HortScience 1982, 17, 622–624. [Google Scholar]
- Dumroese, R.K.; Wenny, D.L. An assessment of ponderosa pine seedlings grown in copper-coated polybags. Tree Plant. Notes 1997, 48, 60–64. [Google Scholar]
- Kormanik, P.P.; Sung, S.S.; Zarnoch, S.J. Immature loblolly pine growth and biomass accumulation: Correlations with seedlings initial first-order lateral roots. South. J. Appl. For. 1998, 22, 117–123. [Google Scholar] [CrossRef]
- Koskela, J. A process-based model for the grass stage pine seedlings. Silva Fenn. 2000, 34, 3–20. [Google Scholar] [CrossRef]
- Keeley, J.E. Ecology and evolution of pine life histories. Ann. For. Sci. 2012, 69, 445–453. [Google Scholar] [CrossRef]
- Straka, T.J. Financial breakeven point for competition control in longleaf pine (Pinus palustris Mill.) reestablishment. New For. 2010, 40, 165–173. [Google Scholar] [CrossRef]
- Grossnickle, S.C.; El-Kassaby, Y. Bareroot versus container stocktypes: A performance comparison. New For. 2016, 47, 1–51. [Google Scholar] [CrossRef]
- Rodríguez-Trejo, D.A.; Duryea, M.L.; White, T.L.; English, J.R.; McGuire, J. Artificially regenerating longleaf pine in canopy gaps: Initial survival and growth during a year of drought. For. Ecol. Manag. 2003, 180, 25–36. [Google Scholar] [CrossRef]
- Samuelson, L.J.; Stokes, T.A. Leaf physiological and morphological responses to shade in grass-stage seedlings and young trees of longleaf pine. Forests 2012, 3, 684–699. [Google Scholar] [CrossRef]
- Belvins, D.; Allen, H.L.; Colbert, S.; Gardnew, W. Nutrition Management for Longleaf Pinestraw; Woodland Owners Notes; North Carolina Cooperative Extension Service: Raleigh, NC, USA, 1996; p. 8. [Google Scholar]
- Burdett, A.N.; Simpson, D.G.; Thompson, C.F. Root development and plantation establishment success. Plant Soil 1983, 71, 103–110. [Google Scholar] [CrossRef]
- Sung, S.S.; Dumroese, R.K. Root system architecture: The invisible trait in container longleaf pine seedlings. In National Proceedings of Forest and Conservation Nursery Associations-2012, Chattanooga, TN, USA, 16–19 July 2012 and Bend, OR, USA, 11–13 August 2012; Haase, D.L., Pinto, J.R., Wilkinson, K.M., Eds.; U.S. Department of Agriculture Forest Service; Rocky Mountain Research Station: Fort Collins, CO, USA, 2014; pp. 26–31. [Google Scholar]
| Treatment | RCD (mm) | FOLR (#) | LFOLR (cm) | Fascicle (#) |
|---|---|---|---|---|
| Cavity volume (mL) | ||||
| 60 | 3.65 (0.18) a | 2.8 (0.4) a | 10.1 (1.0) a | 6.5 (0.4) a |
| 95 | 5.01 (0.17) b | 3.1 (0.3) a,b | 12.3 (0.6) a,b | 9.9 (1.1) a |
| 125 | 5.84 (0.33) c | 4.8 (0.4) b,c | 10.3 (0.5) a | 10.3 (1.9) a |
| 336 | 7.13 (0.24) d | 5.7 (0.7) c | 13.8 (1.2) b | 16.9 (2.1) b |
| p-value | <0.0001 | <0.0001 | 0.0018 | <0.0001 |
| Nitrogen rate | ||||
| Low | 4.86 (0.28) a | 3.9 (0.3) | 10.8 (0.7) | 6.9 (0.4) a |
| Medium | 5.55 (0.34) b | 4.3 (0.5) | 11.8 (0.8) | 10.9 (1.4) b |
| High | 5.82 (0.35) b | 4.1 (0.5) | 12.2 (0.9) | 15.0 (1.9) c |
| p-value | 0.0013 | 0.7258 | 0.2101 | <0.0001 |
| Root pruning | ||||
| No-Cu | 5.14 (0.27) a | 3.9 (0.4) | 13.4 (0.5) b | 11.3 (1.5) |
| Cu | 5.67 (0.26) b | 4.3 (0.4) | 10.0 (0.7) a | 10.6 (0.9) |
| p-value | 0.0127 | 0.4086 | <0.0001 | 0.5891 |
| Variable | Cavity Volume (C) | Nitrogen Rate (N) | Copper Root Pruning (Cu) | C × N | C × Cu | N × Cu | C × N × Cu |
|---|---|---|---|---|---|---|---|
| RCD-Year 0 | <0.0001 | <0.0001 | 0.0003 | 0.0020 | 0.8631 | 0.7274 | 0.0412 |
| GLD-Year 1 | <0.0001 | <0.0001 | 0.5702 | 0.5174 | 0.3485 | 0.7078 | 0.0835 |
| HT-Year 1 | <0.0001 | <0.0001 | 0.0843 | 0.3809 | 0.2679 | 0.2534 | 0.5140 |
| EMGS-Year 1 | <0.0001 | <0.0001 | 0.1034 | 0.3494 | 0.5656 | 0.4868 | 0.6788 |
| HT-Year 2 | <0.0001 | <0.0001 | 0.1484 | 0.0458 | 0.1653 | 0.7920 | 0.2804 |
| EMGS-Year 2 | 0.0003 | 0.0002 | 0.6159 | 0.1688 | 0.6792 | 0.5754 | 0.3431 |
| GLD-Year 3 | <0.0001 | <0.0001 | 0.3764 | 0.8142 | 0.1071 | 0.4136 | 0.5348 |
| HT-Year 3 | <0.0001 | <0.0001 | 0.2205 | 0.1711 | 0.2517 | 0.9654 | 0.3698 |
| DBH-Year 5 | <0.0001 | <0.0001 | 0.6495 | 0.3578 | 0.0871 | 0.5675 | 0.1485 |
| HT-Year 5 | <0.0001 | <0.0001 | 0.4894 | 0.3255 | 0.1149 | 0.5319 | 0.1114 |
| DBH-Year 8 | <0.0001 | 0.0003 | 0.2853 | 0.1955 | 0.2073 | 0.3605 | 0.4752 |
| HT-Year 8 | <0.0001 | <0.0001 | 0.6922 | 0.0639 | 0.0613 | 0.4151 | 0.1327 |
| Treatment | Seedling Biomass (g) | Foliage (%) | Stem (%) | Taproot+ (%) | FOLR (%) | Fine Root (%) |
|---|---|---|---|---|---|---|
| 15-Month Seedlings (March 2009) | ||||||
| Cavity volume (mL) | ||||||
| 60 | 45 (4) a | 31.1 (0.7) | 12.8 (0.8) | 31.9 (1.4) | 21.2 (1.4) | 3.0 (0.2) |
| 95 | 64 (5) a,b | 32.4 (0.8) | 11.3 (0.6) | 32.6 (2.1) | 21.4 (1.5) | 2.3 (0.2) |
| 125 | 68 (6) b | 30.7 (1.0) | 11.7 (0.9) | 29.7 (1.7) | 25.3 (1.8) | 2.5 (0.2) |
| 336 | 108 (9) c | 32.8 (0.8) | 12.2 (0.5) | 26.8 (1.5) | 25.9 (1.0) | 2.3 (0.6) |
| p-value | <0.0001 | 0.1632 | 0.4325 | 0.0483 | 0.0545 | 0.5555 |
| Nitrogen rate | ||||||
| Low | 53 (7) a | 30.1 (0.7) a | 11.5 (0.4) | 33.4 (1.8) b | 21.7 (1.6) | 2.7 (0.2) |
| Medium | 72 (6) b | 31.9 (0.6) a,b | 11.7 (0.7) | 30.5 (1.4) a,b | 23.1 (1.2) | 2.8 (0.4) |
| High | 88 (7) b | 33.2 (0.7) b | 12.8 (0.6) | 26.4 (0.9) a | 25.5 (0.9) | 2.1 (0.2) |
| p-value | <0.0001 | 0.0071 | 0.2905 | 0.0013 | 0.1326 | 0.2356 |
| Root pruning | ||||||
| No-Cu | 74 (7) | 31.2 (0.6) | 11.6 (0.3) | 31.3 (1.2) | 23.3 (1.0) | 2.7 (0.3) |
| Cu | 69 (5) | 32.3 (0.6) | 12.4 (0.6) | 29.3 (1.2) | 23.7 (1.1) | 2.4 (0.2) |
| p-value | 0.3918 | 0.1579 | 0.2830 | 0.2165 | 0.7918 | 0.4260 |
| 22-Month Seedlings (October 2009) | ||||||
| Cavity volume (mL) | ||||||
| 60 | 133 (22) a | 60.0 (1.9) b | 16.4 (1.8) a | 14.7 (0.9) | 7.5 (0.8) | 1.5 (0.3) b |
| 95 | 210 (20) a,b | 57.5 (1.2) b | 18.6 (1.3) a,b | 13.5 (0.8) | 9.6 (0.8) | 0.9 (0.2) a |
| 125 | 228 (26) b | 56.5 (1.4) a,b | 20.3 (1.4) a,b | 13.2 (0.8) | 9.4 (0.7) | 0.7 (0.1) a |
| 336 | 296 (33) b | 52.0 (1.5) a | 23.6 (1.7) b | 13.7 (1.0) | 10.1 (0.8) | 0.5 (0.1) a |
| p-value | 0.0001 | 0.0015 | 0.0040 | 0.5560 | 0.1334 | 0.0003 |
| Nitrogen rate | ||||||
| Low | 149 (16) a | 59.4 (1.0) b | 15.4 (0.9) a | 15.5 (0.6) b | 8.6 (0.7) | 1.2 (0.2) |
| Medium | 242 (28) b | 57.1 (1.4) a,b | 20.5 (1.5) b | 12.5 (0.9) a | 9.1 (0.7) | 0.7 (0.2) |
| High | 259 (22) b | 53.0 (1.6) a | 23.3 (1.4) b | 13.3 (0.7) a,b | 9.6 (0.7) | 0.8 (0.1) |
| p-value | 0.0005 | 0.0014 | <0.0001 | 0.0101 | 0.5928 | 0.0260 |
| Root pruning | ||||||
| No-Cu | 234 (22) | 55.4 (1.1) | 20.1 (1.2) | 13.5 (0.7) | 10.3 (0.6) b | 0.8 (0.1) |
| Cu | 200 (18) | 57.6 (1.2) | 19.4 (1.2) | 14.1 (0.6) | 8.0 (0.5) a | 1.0 (0.1) |
| p-value | 0.1416 | 0.1099 | 0.6246 | 0.4740 | 0.0071 | 0.2345 |
| Nutrient | 15-Month Foliage Concentration | 15-Month Seedling Content (mg) | 22-Month Foliage Concentration | 22-Month Seedling Content (mg) |
|---|---|---|---|---|
| (%) | (mg) | (%) | (mg) | |
| N | 1.27 (0.01) | 680 (46) | 0.89 (0.01) | 1884 (121) |
| P | 0.08 (0.002) | 54 (4) | 0.08 (0.002) ⁼ | 163 (10) |
| K | 0.52 (0.01) | 269 (18) | 0.56 (0.02) | 962 (61) |
| Mg | 0.11 (0.002) | 73 (5) | 0.11 (0.002) ⁼ | 219 (12) |
| Ca | 0.44 (0.01) | 135 (9) | 0.20 (0.01) | 340 (22) |
| (ppm) | (mg) | (ppm) | (mg) | |
| Cu | 5.6 (0.1) | 0.3 (0.0) | 4.5 (0.1) | 1.0 (0.1) |
| B | 15 (1) | 2 (0.1) | 20 (1) | 4 (0.3) |
| Zn | 63 (1) | 3 (0.2) | 40 (1) | 8 (0.4) |
| Fe | 52 (2) | 8 (1) | 114 (9) | 19 (1.7) |
| Mn | 665 (16) | 19 (1) | 241(8) | 35 (2.5) |
| Treatment | 15-Month Vertical Anchorage Depth (cm) | 15-Month Root Anchorage Biomass (g) | 22-Month Vertical Anchorage Depth (cm) | 22-Month Root Anchorage Biomass (g) |
|---|---|---|---|---|
| Cavity volume (mL) | ||||
| 60 | 18.7 (1.8) a | 13.7 (1.3) a | 24.9 (3.3) | 18.3 (3.0) a |
| 95 | 19.3 (2.0) a,b | 19.1 (1.8) a | 21.5 (3.1) | 26.6 (2.9) a,b |
| 125 | 26.5 (2.1) b | 18.0 (1.2) a | 25.3 (3.7) | 26.2 (2.5) a,b |
| 336 | 20.2 (2.7) a,b | 27.9 (3.0) b | 19.3 (3.2) | 35.7 (3.7) b |
| p-value | 0.0229 | <0.0001 | 0.4557 | 0.0008 |
| Nitrogen rate | ||||
| Low | 18.2 (1.7) a | 15.7 (2.0) a | 19.8 (2.1) | 21.0 (2.6) a |
| Medium | 19.0 (1.8) a | 20.6 (1.7) a,b | 19.9 (2.6) | 27.5 (3.0) a,b |
| High | 26.3 (2.0) b | 22.7 (2.0) b | 28.5 (3.4) | 31.6 (2.7) b |
| p-value | 0.0019 | 0.0151 | 0.0345 | 0.0110 |
| Root pruning | ||||
| No-Cu | 22.9 (1.7) | 21.2 (1.9) | 23.9 (2.5) | 29.5 (2.8) |
| Cu | 19.4 (1.4) | 18.2 (1.3) | 21.5 (2.2) | 23.9 (1.8) |
| p-value | 0.0731 | 0.1288 | 0.4248 | 0.0510 |
| Treatment | Plug Root Biomass (g) | Taproot (%) | FOLR (%) | Fine Root (%) |
|---|---|---|---|---|
| 15-Month Seedlings (March 2009) | ||||
| Cavity volume (mL) | ||||
| 60 | 14.1 (1.1) a | 66.5 (1.9) a | 24.3 (2.3) a | 9.2 (0.7) |
| 95 | 20.4 (1.4) b | 64.1 (2.3) a | 29.2 (2.3) a | 6.7 (0.5) |
| 125 | 20.6 (1.5) b | 59.6 (2.3) a,b | 32.8 (2.4) a,b | 7.6 (0.6) |
| 336 | 33.8 (2.7) c | 53.2 (2.1) b | 40.0 (2.1) b | 6.8 (1.4) |
| p-value | <0.0001 | 0.0004 | <0.0001 | 0.2207 |
| Nitrogen rate | ||||
| Low | 17.9 (2.2) a | 63.9 (2.5) a | 28.1 (2.5) a | 8.0 (0.7) |
| Medium | 22.9 (1.8) b | 61.2 (1.9) a,b | 31.0 (1.9) a,b | 7.7 (0.9) |
| High | 25.9 (2.1) b | 57.3 (1.7) b | 35.7 (2.1) b | 6.9 (0.7) |
| p-value | 0.0005 | 0.0517 | 0.0220 | 0.5985 |
| Root pruning | ||||
| No-Cu | 24.2 (1.9) a | 58.1 (1.6) a | 34.5 (1.7) a | 7.4 (0.7) |
| Cu | 20.5 (1.5) b | 63.2 (1.7) b | 29.0 (1.8) b | 7.8 (0.6) |
| p-value | 0.0188 | 0.0139 | 0.0110 | 0.7219 |
| 22-Month Seedlings (October 2009) | ||||
| Cavity volume (mL) | ||||
| 60 | 17.3 (2.3) a | 66.8 (2.5) | 23.1 (2.6) a | 10.1 (1.7) a |
| 95 | 28.3 (2.9) b | 64.8 (2.5) | 29.2 (2.9) a,b | 6.0 (1.0) b |
| 125 | 29.2 (2.7) b | 62.3 (2.7) | 32.6 (2.8) b | 5.2 (0.6) b |
| 336 | 41.7 (3.8) c | 59.1 (2.6) | 37.3 (2.8) b | 3.5 (0.5) b |
| p-value | <0.0001 | 0.1089 | 0.0005 | <0.0001 |
| Nitrogen rate | ||||
| Low | 22.4 (2.3) a | 67.4 (2.1) | 25.1 (2.5) a | 7.4 (1.1) |
| Medium | 29.9 (3.4) b | 61.6 (2.3) | 33.0 (2.3) b | 5.4 (1.0) |
| High | 35.1 (231) b | 60.7 (2.2) | 33.6 (2.7) b | 5.8 (0.9) |
| p-value | 0.0006 | 0.0382 | 0.0057 | 0.1919 |
| Root pruning | ||||
| No-Cu | 33.6 (2.8) a | 58.5 (1.8) a | 36.2 (2.0) a | 5.3 (0.9) |
| Cu | 24.6 (1.9) b | 68.0 (1.5) b | 24.9 (1.8) b | 7.1 (0.7) |
| p-value | 0.0004 | 0.0001 | <0.0001 | 0.0591 |
| Treatment | FOLR Number | Mean FOLR Length (cm) | Total FOLR Length (cm) | DI |
|---|---|---|---|---|
| 15-Month Seedlings (March 2009) | ||||
| Cavity volume (mL) | ||||
| 60 | 5.7 (0.4) a | 6.3 (0.7) a | 36.2 (5.0) a | 3.0 (0.6) a |
| 95 | 6.6 (0.5) a | 8.0 (0.6) b | 54.5 (5.9) a | 4.8 (0.7) a |
| 125 | 7.5 (0.6) a | 7.6 (0.4) a,b | 59.0 (5.6) a | 5.3 (0.6) a |
| 336 | 9.6 (0.6) b | 10.4 (0.7) c | 101.7 (10.3) b | 9.2 (1.1) b |
| p-value | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Nitrogen rate | ||||
| Low | 5.8 (0.5) a | 7.9 (0.6) | 49.4 (7.3) a | 4.4 (0.7) a |
| Medium | 7.6 (0.5) b | 8.2 (0.5) | 64.9 (7.0) a,b | 5.8 (0.7) a,b |
| High | 8.7 (0.5) b | 8.2 (0.6) | 74.2 (8.3) b | 6.6 (0.9) b |
| p-value | <0.0001 | 0.6912 | 0.0047 | 0.0165 |
| Root pruning | ||||
| No-Cu | 7.3 (0.5) | 9.9 (0.4)b | 76.9 (7.2) b | 7.4 (0.7) b |
| Cu | 7.4 (0.4) | 6.2 (0.3) a | 48.1 (4.2) a | 3.8 (0.4) a |
| p-value | 0.7158 | <0.0001 | <0.0001 | <0.0001 |
| 22-Month Seedlings (October 2009) | ||||
| Cavity volume (mL) | ||||
| 60 | 6.3 (0.6) a | 7.0 (0.7) a | 46.2 (6.9) a | 4.4 (0.8) a |
| 95 | 8.2 (0.8) a,b | 7.3 (0.7) a | 64.1 (10.9) a | 6.6 (1.5) a |
| 125 | 7.9 (0.7) a | 7.8 (0.6) a | 64.7 (8.3) a | 7.1 (1.1) a |
| 336 | 10.9 (0.9) b | 10.4 (0.9) b | 117.7 (15.9) b | 12.7 (2.0) b |
| p-value | 0.0006 | <0.0001 | <0.0001 | 0.0002 |
| Nitrogen rate | ||||
| Low | 6.8 (0.6) a | 7.3 (0.6) | 51.8 (7.2) a | 5.4 (1.1) a |
| Medium | 8.5 (0.8) a,b | 8.5 (0.6) | 76.6 (10.9) a,b | 8.4 (1.5) a,b |
| High | 9.8 (0.7) b | 8.6 (0.7) | 91.2 (12.6) b | 9.3 (1.4) b |
| p-value | 0.0076 | 0.0665 | 0.0033 | 0.0378 |
| Root pruning | ||||
| No-Cu | 8.8 (0.6) | 10.2 (0.5) b | 94.9 (9.8) b | 10.6 (1.2) b |
| Cu | 7.8 (0.6) | 6.1 (0.3) a | 51.5 (6.1) a | 4.8 (0.8) a |
| p-value | 0.1806 | <0.0001 | <0.0001 | <0.0001 |
| Egress from Root Plug (cm) | Cavity Volume (C) | Nitrogen Rate (N) | Copper Root Pruning (Cu) | C × N | C × Cu | N × Cu | C × N × Cu |
|---|---|---|---|---|---|---|---|
| 15-Month Seedlings (March 2009) | |||||||
| 0–4.0 | 0.5016 | 0.9933 | <0.0001 | 0.5024 | 0.1036 | 0.8204 | 0.4237 |
| 4.1–8.0 | 0.0627 | 0.1681 | 0.0219 | 0.7383 | 0.8408 | 0.7116 | 0.5560 |
| 8.1–12.0 | 0.0005 | 0.3796 | 0.0002 | 0.8830 | 0.0056 | 0.3341 | 0.7559 |
| >12.0 | <0.0001 | 0.2303 | <0.0001 | 0.4101 | <0.0001 | 0.8293 | 0.9987 |
| 22–Month Seedlings (October 2009) | |||||||
| 0–4.0 | 0.0311 | 0.7943 | <0.0001 | 0.8622 | 0.0077 | 0.1007 | 0.8669 |
| 4.1–8.0 | 0.8383 | 0.0230 | <0.0001 | 0.7750 | 0.3028 | 0.1679 | 0.7650 |
| 8.1–12.0 | 0.1274 | 0.2828 | <0.0001 | 0.7692 | 0.3711 | 0.6554 | 0.3101 |
| >12.0 | <0.0001 | <0.0001 | 0.1314 | 0.0001 | 0.3872 | 0.1218 | 0.6118 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sung, S.-J.S.; Dumroese, R.K.; Pinto, J.R.; Sayer, M.A.S. The Persistence of Container Nursery Treatments on the Field Performance and Root System Morphology of Longleaf Pine Seedlings. Forests 2019, 10, 807. https://doi.org/10.3390/f10090807
Sung S-JS, Dumroese RK, Pinto JR, Sayer MAS. The Persistence of Container Nursery Treatments on the Field Performance and Root System Morphology of Longleaf Pine Seedlings. Forests. 2019; 10(9):807. https://doi.org/10.3390/f10090807
Chicago/Turabian StyleSung, Shi-Jean S., R. Kasten Dumroese, Jeremiah R. Pinto, and Mary Anne S. Sayer. 2019. "The Persistence of Container Nursery Treatments on the Field Performance and Root System Morphology of Longleaf Pine Seedlings" Forests 10, no. 9: 807. https://doi.org/10.3390/f10090807
APA StyleSung, S.-J. S., Dumroese, R. K., Pinto, J. R., & Sayer, M. A. S. (2019). The Persistence of Container Nursery Treatments on the Field Performance and Root System Morphology of Longleaf Pine Seedlings. Forests, 10(9), 807. https://doi.org/10.3390/f10090807





