Effects of Vegetation Restoration on the Distribution of Nutrients, Glomalin-Related Soil Protein, and Enzyme Activity in Soil Aggregates on the Loess Plateau, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Site Selection
2.2. Soil Sampling
2.3. Soil Physicochemical Properties and Enzyme Analysis
2.4. Data Analysis
3. Results
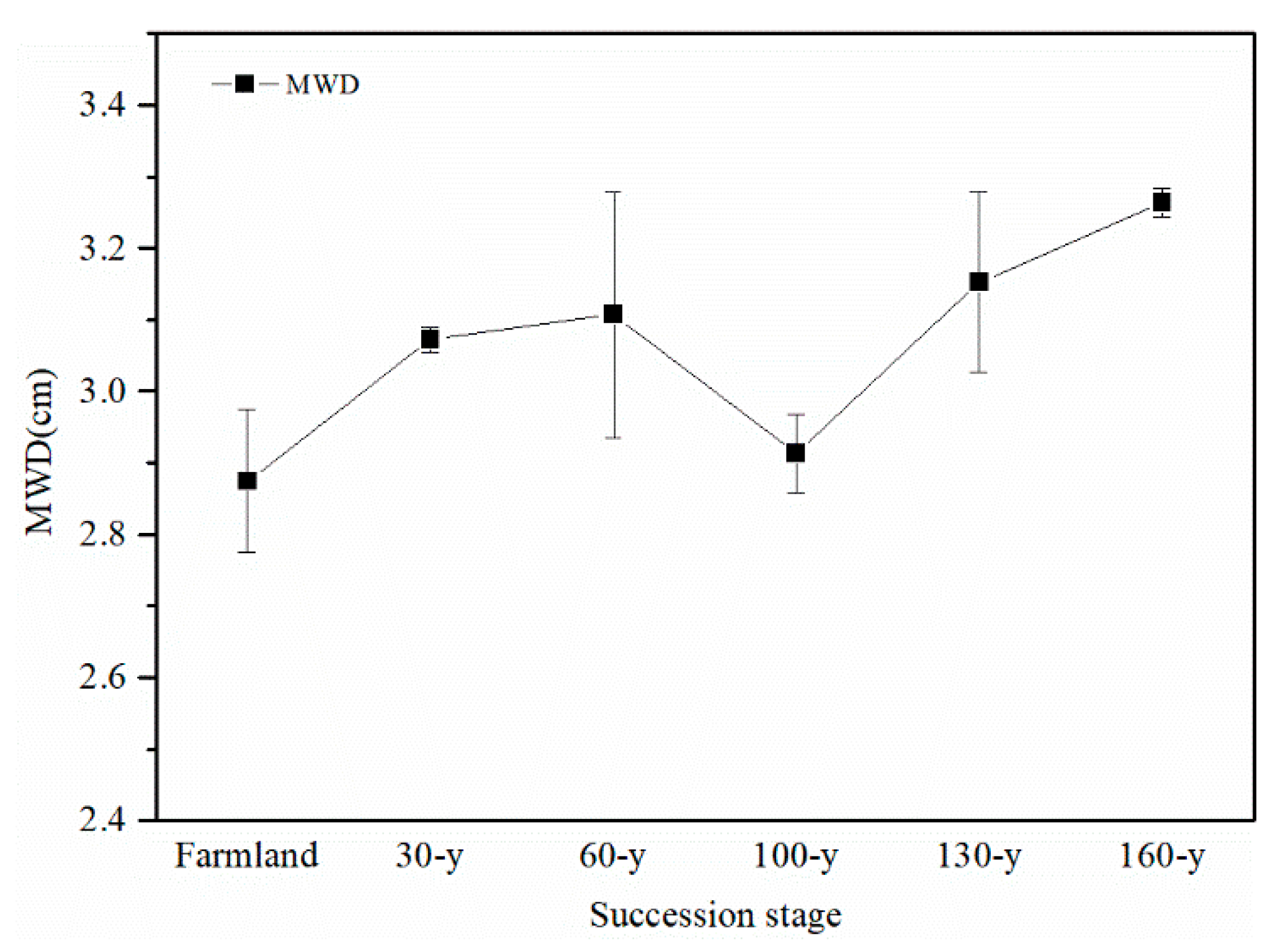
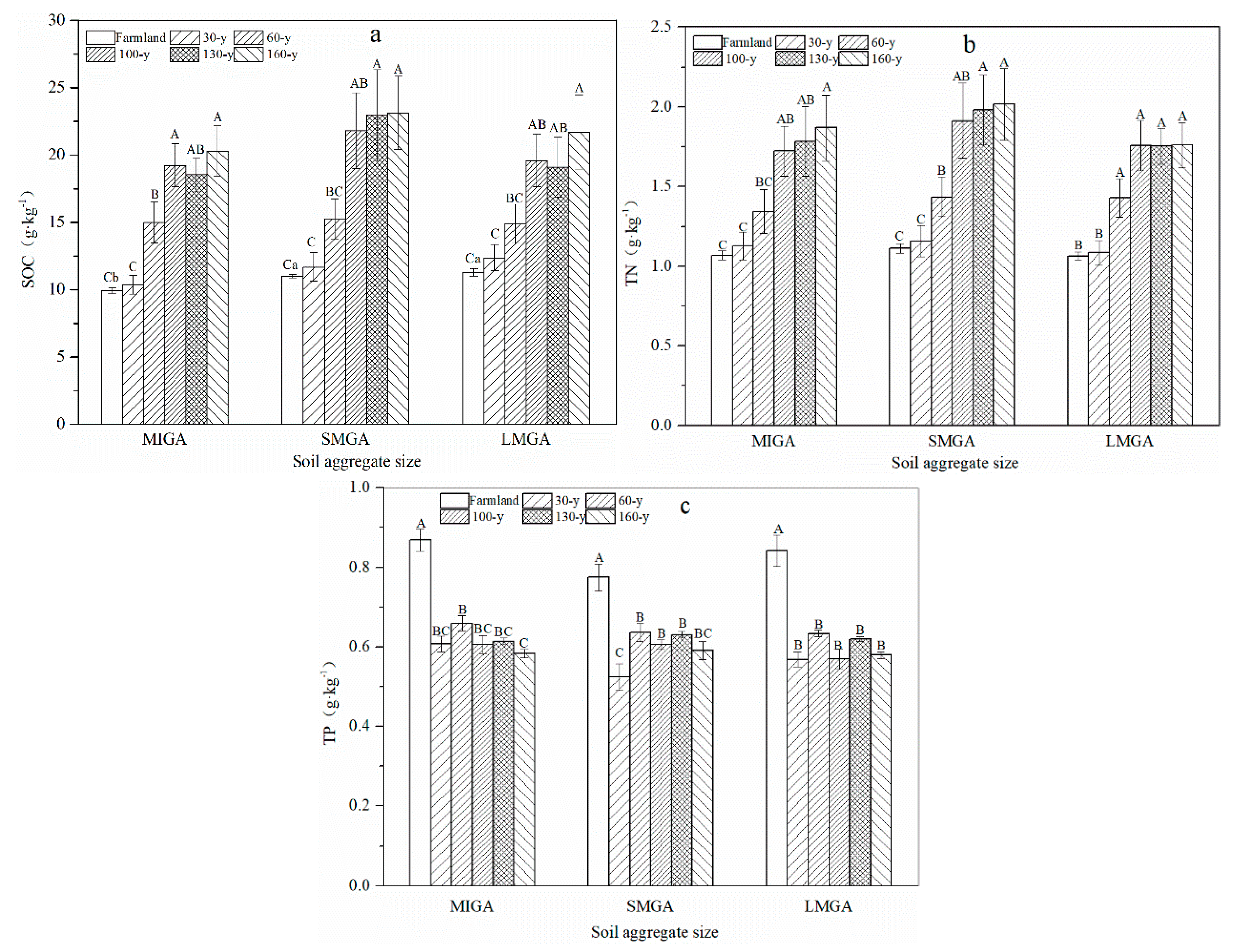
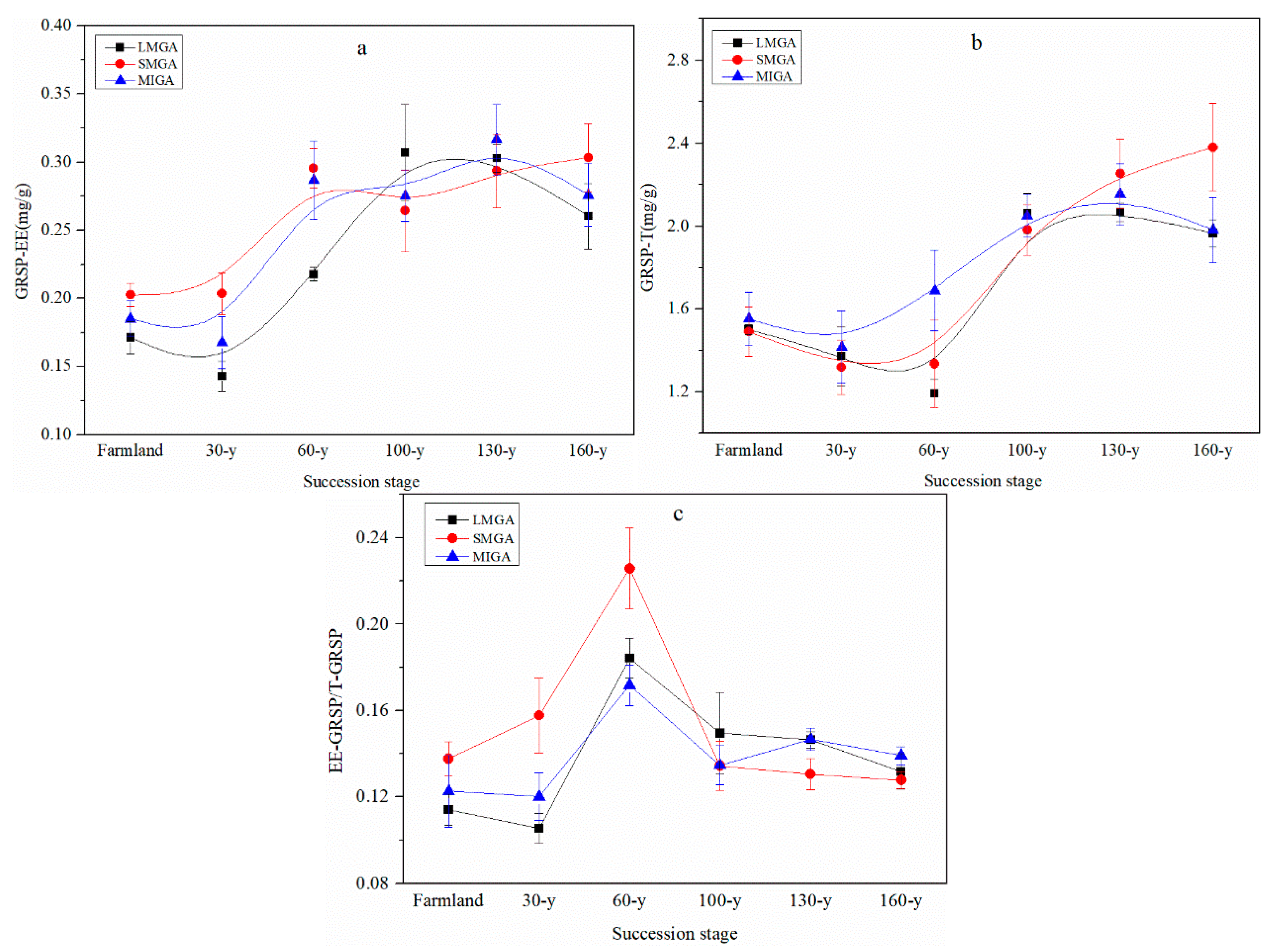
3.1. Aggregate Stability and Content of Nutrients and GRSP
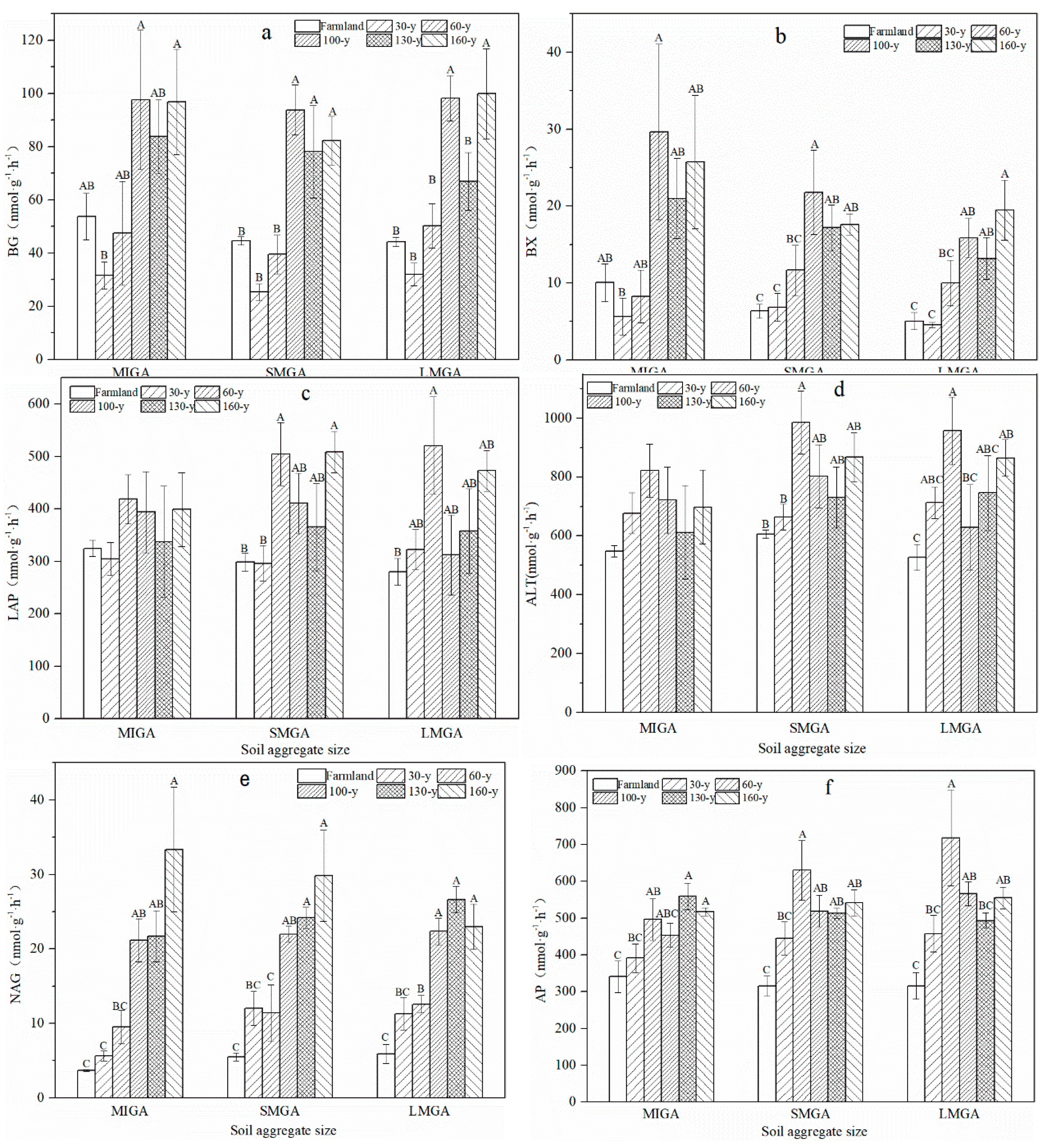
3.2. Enzyme Activity and Nutrient Turnover
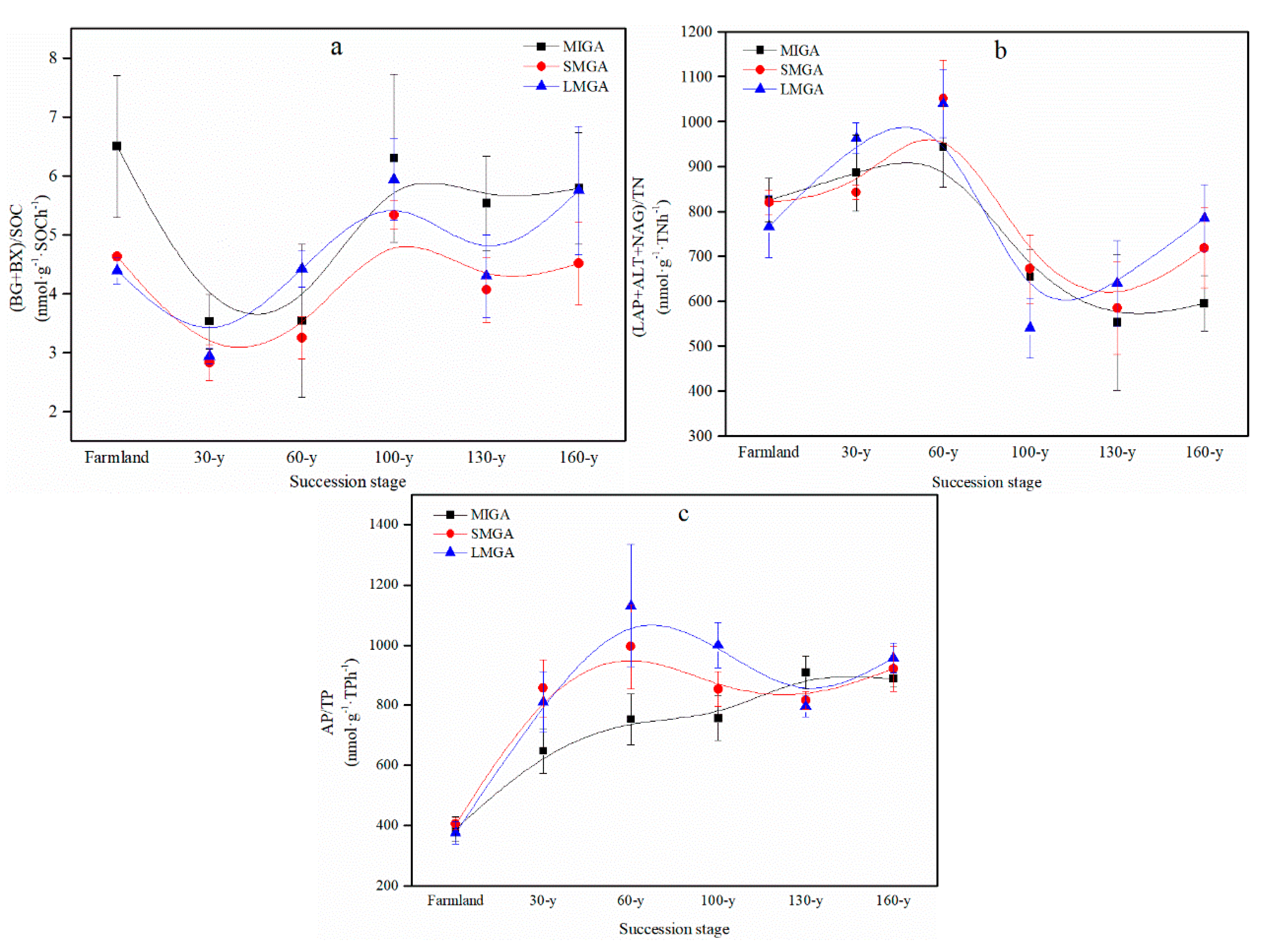
3.3. Ecological Stoichiometry
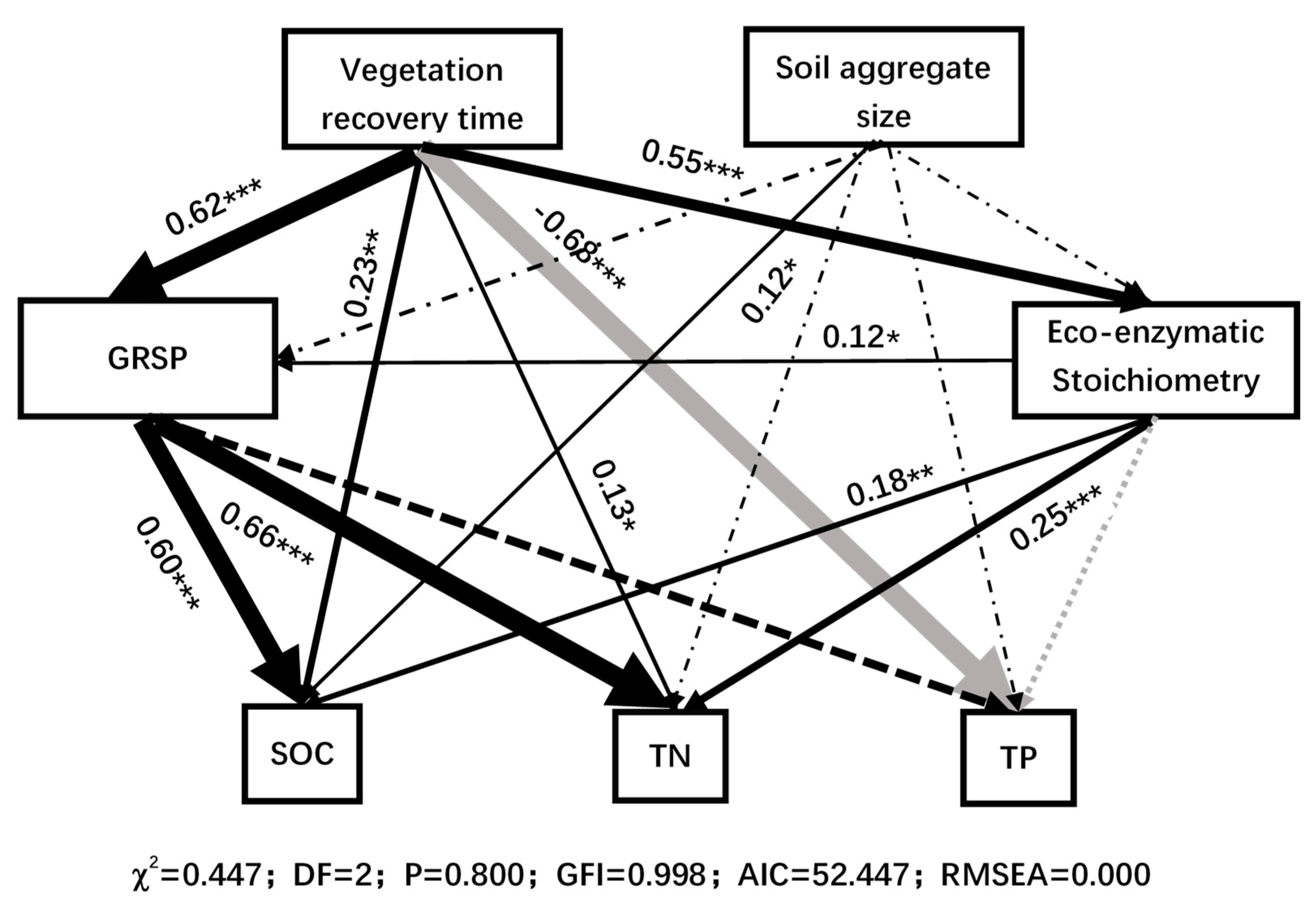
3.4. Comprehensive Response of Soil Aggregate Nutrients and Enzymes to Vegetation Restoration
4. Discussion
4.1. Effects of Vegetation Restoration on Soil Aggregate Nutrients and Glomalin
4.2. Effects of Vegetation Restoration on Soil Aggregate Enzyme Activity and Nutrient Turnover
4.3. Response of Ecological Stoichiometry to Vegetation Restoration
4.4. The Effect of GRSP on Nutrient Balance
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tian, J.; Mc Cormack, L.; Wang, J.; Guo, D.; Wang, Q.; Zhang, X.; Yu, G.; Blagodatskaya, E.; Kuzyakov, Y. Linkages between the soil organic matter fractions and the microbial metabolic functional diversity within a broad-leaved Korean pine forest. Eur. J. Soil Biol. 2015, 66, 57–64. [Google Scholar] [CrossRef]
- Deng, L.; Liu, S.; Kim, D.G.; Peng, C.; Sweeney, S.; Shangguan, Z. Past and future carbon sequestration benefits of China’s grain for green program. Glob. Environ. Chang. Hum. Policy Dimens. 2017, 47, 13–20. [Google Scholar] [CrossRef]
- Marin-Spiotta, E.; Sharma, S. Carbon storage in successional and plantation forest soils: A tropical analysis. Global Ecol. Biogeogr. 2013, 22, 105–117. [Google Scholar] [CrossRef]
- Qiu, L.P.; Wei, X.R.; Gao, J.L.; Zhang, X.C. Dynamics of soil aggregate-associated organic carbon along an afforestation chronosequence. Plant Soil 2015, 391, 237–251. [Google Scholar] [CrossRef]
- Zhang, C.; Xue, S.; Liu, G.B.; Song, Z.L. A comparison of soil qualities of different revegetation types in the Loess Plateau, China. Plant Soil 2011, 347, 163–178. [Google Scholar] [CrossRef]
- Deng, L.; Wang, K.B.; Zhu, G.Y.; Liu, Y.L.; Chen, L.; Shangguan, Z.P. Changes of soil carbon in five land use stages following 10 years of vegetation succession on the Loess Plateau, China. Catena 2018, 171, 185–192. [Google Scholar] [CrossRef]
- Barthes, B.; Roose, E. Aggregate stability as an indicator of soil susceptibility to runoff and erosion; validation at several levels. Catena 2002, 47, 133–149. [Google Scholar] [CrossRef]
- Cheng, M.; Xiang, Y.; Xue, Z.; An, S.; Darboux, F. Soil aggregation and intra-aggregate carbon fractions in relation to vegetation succession on the Loess Plateau, China. Catena 2015, 124, 77–84. [Google Scholar] [CrossRef]
- Zhu, G.; Lei, D.; Shangguan, Z. Effects of soil aggregate stability on soil N following land use changes under erodible environment. Agric. Ecosyst. Environ. 2018, 262, 18–28. [Google Scholar] [CrossRef]
- Deng, L.; Kim, D.-G.; Peng, C.; Shangguan, Z. Controls of soil and aggregate-associated organic carbon variations following natural vegetation restoration on the Loess Plateau in China. Land Degrad. Dev. 2018, 29, 3974–3984. [Google Scholar] [CrossRef]
- Zhu, G.-Y.; Shangguan, Z.-P.; Deng, L. Soil aggregate stability and aggregate-associated carbon and nitrogen in natural restoration grassland and Chinese red pine plantation on the Loess Plateau. Catena 2017, 149, 253–260. [Google Scholar] [CrossRef]
- Bach, E.M.; Hofmockel, K.S. Soil aggregate isolation method affects measures of intra-aggregate extracellular enzyme activity. Soil Biol. Biochem. 2014, 69, 54–62. [Google Scholar] [CrossRef]
- Wright, S.F.; Upadhyaya, A. A survey of soils for aggregate stability and glomalin, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant Soil 1998, 198, 97–107. [Google Scholar] [CrossRef]
- Nichols, K.A.; Wright, S.F. Carbon and nitrogen in operationally defined soil organic matter pools. Biol. Fertil. Soils 2006, 43, 215–220. [Google Scholar] [CrossRef]
- Rillig, M.C. Arbuscular mycorrhizae, glomalin, and soil aggregation. Can. J. Soil Sci. 2004, 84, 355–363. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Wright, S.F.; Ruess, C.R.W. Soil Stocks of Glomalin Produced by Arbuscular Mycorrhizal Fungi across a Tropical Rain Forest Landscape. J. Ecol. 2004, 92, 278–287. [Google Scholar] [CrossRef]
- Rillig, M.C.; Wright, S.F.; Nichols, K.A.; Schmidt, W.F.; Torn, M.S. Large contribution of arbuscular mycorrhizal fungi to soil carbon pools in tropical forest soils. Plant Soil 2001, 233, 167–177. [Google Scholar] [CrossRef]
- Fokom, R.; Adamou, S.; Teugwa, M.C.; Boyogueno, A.B.; Nana, W.L.; Ngonkeu, M.E.L.; Tchameni, N.S.; Nwaga, D.; Ndzomo, G.T.; Zollo, P.A. Glomalin related soil protein, carbon, nitrogen and soil aggregate stability as affected by land use variation in the humid forest zone of south Cameroon. Soil Tillage Res. 2012, 120, 69–75. [Google Scholar] [CrossRef]
- Vasconcellos, R.L.; Bonfim, J.A.; Baretta, D.; Cardoso, E.J. Arbuscular Mycorrhizal Fungi and Glomalin related Soil Protein as Potential Indicators of Soil Quality in a Recuperation Gradient of the Atlantic Forest in Brazil. Land Degrad. Dev. 2016, 27, 325–334. [Google Scholar] [CrossRef]
- Sinsabaugh, R.L.; Hill, B.H.; Follstad Shah, J.J. Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 2009, 462, 795–798. [Google Scholar] [CrossRef]
- Chen, H.; Li, D.; Xiao, K.; Wang, K. Soil microbial processes and resource limitation in karst and non-karst forests. Funct. Ecol. 2018, 32, 1400–1409. [Google Scholar] [CrossRef]
- Zhao, F.Z.; Ren, C.J.; Han, X.H.; Yang, G.H.; Wang, J.; Doughty, R. Changes of soil microbial and enzyme activities are linked to soil C, N and P stoichiometry in afforested ecosystems. For. Ecol. Manag. 2018, 427, 289–295. [Google Scholar] [CrossRef]
- Sinsabaugh, R.L.; Lauber, C.L.; Weintraub, M.N.; Ahmed, B.; Allison, S.D.; Crenshaw, C.; Contosta, A.R.; Cusack, D.; Frey, S.; Gallo, M.E.; et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 2008, 11, 1252–1264. [Google Scholar] [CrossRef]
- Waring, B.G.; Weintraub, S.R.; Sinsabaugh, R.L. Ecoenzymatic stoichiometry of microbial nutrient acquisition in tropical soils. Biogeochemistry 2014, 117, 101–113. [Google Scholar] [CrossRef]
- Reich, P.B.; Oleksyn, J. Global patterns of plant leaf N and P in relation to temperature and latitude. Proc. Natl. Acad. Sci. USA 2004, 101, 11001–11006. [Google Scholar] [CrossRef]
- Walker, T.W.; Syers, J.K. The fate of phosphorus during pedogenesis. Geoderma 1976, 15, 1–19. [Google Scholar] [CrossRef]
- Howarth, V.R.W. Nitrogen Limitation on Land and in the Sea: How Can It Occur? Biogeochemistry 1991, 13, 87–115. [Google Scholar]
- Peng, X.; Wang, W. Stoichiometry of soil extracellular enzyme activity along a climatic transect in temperate grasslands of northern China. Soil Biol. Biochem. 2016, 98, 74–84. [Google Scholar] [CrossRef]
- Fu, X.L.; Shao, M.A.; Wei, X.R.; Horton, R. Soil organic carbon and total nitrogen as affected by vegetation types in Northern Loess Plateau of China. Geoderma 2010, 155, 31–35. [Google Scholar] [CrossRef]
- Wang, B.; Xue, S.; Liu, G.B.; Zhang, G.H.; Li, G.; Ren, Z.P. Changes in soil nutrient and enzyme activities under different vegetations in the Loess Plateau area, Northwest China. Catena 2012, 92, 186–195. [Google Scholar] [CrossRef]
- An, S.-S.; Cheng, Y.; Huang, Y.-M.; Liu, D. Effects of Revegetation on Soil Microbial Biomass, Enzyme Activities, and Nutrient Cycling on the Loess Plateau in China. Restor. Ecol. 2013, 21, 600–607. [Google Scholar] [CrossRef]
- Deng, L.; Wang, K.-B.; Shangguan, Z.-P. Long-term natural succession improves nitrogen storage capacity of soil on the Loess Plateau, China. Soil Res. 2014, 52, 262–270. [Google Scholar] [CrossRef]
- Jia, G.M.; Cao, J.; Wang, C.Y.; Wang, G. Microbial biomass and nutrients in soil at the different stages of secondary forest succession in Ziwulin, northwest China. For. Ecol. Manag. 2005, 217, 117–125. [Google Scholar] [CrossRef]
- Wang, G.; Wang, C.Y.; Wang, W.Y.; Wang, Q.J. Capacity of soil to protect organic carbon and biochemical characteristics of density fractions in Ziwulin Haplic Greyxems soil. Chin. Sci. Bull. 2005, 50, 27–32. [Google Scholar] [CrossRef]
- Zou, H.-Y.; Liu, G.-B.; Wang, H.-S. The vegetation development in North Ziwulin forest region in last fifty years. Xibei Zhiwu Xuebao 2002, 22, 1–8. [Google Scholar]
- Chen, C.D. The vegetation and its roles in soil and water conservation in the secondary forest area in the boundary of Shaanxi and Gansu provinces. Acta Phytoecol. Geobot. Sin. 1954, 2, 152–2231. [Google Scholar]
- Wang, K.; Shao, R.; Shangguan, Z. Changes in species richness and community productivity during succession on the Loess Plateau (China). Pol. J. Ecol. 2010, 58, 501–510. [Google Scholar]
- Nie, M.; Pendall, E.; Bell, C.; Wallenstein, M.D. Soil aggregate size distribution mediates microbial climate change feedbacks. Soil Biol. Biochem. 2014, 68, 357–365. [Google Scholar] [CrossRef]
- Kalembasa, S.J.; Jenkinson, D.S. A comparative study of titrimetric and gravimetric methods for the determination of organic carbon in soil. J. Sci. Food Agric. 1973, 24, 1085–1090. [Google Scholar] [CrossRef]
- Jackson, M.L. Soil Chemical Analysis—Advanced Course; Scientific Publishers: Jodhpur, India, 1979. [Google Scholar]
- Schade, J.D.; Kyle, M.; Hobbie, S.E.; Fagan, W.F.; Elser, J.J. Stoichiometric tracking of soil nutrients by a desert insect herbivore. Ecol. Lett. 2003, 6, 96–101. [Google Scholar] [CrossRef]
- Wright, S.F.; Upadhyaya, A. Extraction of an abundant and unusual protein from soil and comparison with hyphal protein of arbuscular mycorrhizal fungi. Soil Sci. 1996, 161, 575–586. [Google Scholar] [CrossRef]
- Saiya-Cork, K.R.; Sinsabaugh, R.L.; Zak, D.R. The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol. Biochem. 2002, 34, 1309–1315. [Google Scholar] [CrossRef]
- Steinweg, J.M.; Dukes, J.S.; Wallenstein, M.D. Modeling the effects of temperature and moisture on soil enzyme activity: Linking laboratory assays to continuous field data. Soil Biol. Biochem. 2012, 55, 85–92. [Google Scholar] [CrossRef]
- Veen, G.F.; Olff, H.; Duyts, H.; van der Putten, W.H. Vertebrate herbivores influence soil nematodes by modifying plant communities. Ecology 2010, 91, 828–835. [Google Scholar] [CrossRef]
- Zhang, J.; Ai, Z.; Liang, C.; Wang, G.; Liu, G.; Xue, S. How microbes cope with short-term N addition in a Pinus tabuliformis forest-ecological stoichiometry. Geoderma 2019, 337, 630–640. [Google Scholar] [CrossRef]
- Wang, Y.X.; Ran, L.S.; Fang, N.F.; Shi, Z.H. Aggregate stability and associated organic carbon and nitrogen as affected by soil erosion and vegetation rehabilitation on the Loess Plateau. Catena 2018, 167, 257–265. [Google Scholar] [CrossRef]
- Shi, J.; Cui, L.L. Soil carbon change and its affecting factors following afforestation in China. Landsc. Urban Plan. 2010, 98, 75–85. [Google Scholar] [CrossRef]
- Yang, Y.; Luo, Y. Carbon: Nitrogen stoichiometry in forest ecosystems during stand development. Glob. Ecol. Biogeogr. 2011, 20, 354–361. [Google Scholar] [CrossRef]
- Cheng, Y.; Li, P.; Xu, G.; Li, Z.; Cheng, S.; Gao, H. Spatial distribution of soil total phosphorus in Yingwugou watershed of the Dan River, China. Catena 2016, 136, 175–181. [Google Scholar] [CrossRef]
- Kooijman, A.M.; Jongejans, J.; Sevink, J. Parent material effects on Mediterranean woodland ecosystems in NE Spain. Catena 2005, 59, 55–68. [Google Scholar] [CrossRef]
- Treseder, K.K.; Cross, A. Global distributions of arbuscular mycorrhizal fungi. Ecosystems 2006, 9, 305–316. [Google Scholar] [CrossRef]
- Treseder, K.K.; Turner, K.M. Glomalin in Ecosystems. Soil Sci. Soc. Am. J. 2007, 71, 1257–1266. [Google Scholar] [CrossRef]
- Treseder, K.K. A meta-analysis of mycorrhizal responses to nitrogen, phosphorus, and atmospheric CO2 in field studies. New Phytol. 2004, 164, 347–355. [Google Scholar] [CrossRef]
- Jorge-Araújo, P.; Quiquampoix, H.; Matumoto-Pintro, P.T.; Staunton, S. Glomalin-related soil protein in French temperate forest soils: Interference in the Bradford assay caused by co-extracted humic substances. Eur. J. Soil Sci. 2015, 66, 311–319. [Google Scholar] [CrossRef]
- Jiang, J.P.; Xiong, Y.C.; Jiang, H.M.; Ye, D.Y.; Song, Y.J.; Li, F.M. Soil Microbial Activity During Secondary Vegetation Succession in Semiarid Abandoned Lands of Loess Plateau. Pedosphere 2009, 19, 735–747. [Google Scholar] [CrossRef]
- Ren, C.; Zhao, F.; Kang, D.; Yang, G.; Han, X.; Tong, X.; Feng, Y.; Ren, G. Linkages of C:N:P stoichiometry and bacterial community in soil following afforestation of former farmland. For. Ecol. Manag. 2016, 376, 59–66. [Google Scholar] [CrossRef]
- Miller, M.; Palojarvi, A.; Rangger, A.; Reeslev, M.; Kjoller, A. The use of fluorogenic substrates to measure fungal presence and activity in soil. Appl. Environ. Microbiol. 1998, 64, 613–617. [Google Scholar]
- Muruganandam, S.; Israel, D.W.; Robarge, W.P. Activities of Nitrogen-Mineralization Enzymes Associated with Soil Aggregate Size Fractions of Three Tillage Systems. Soil Sci. Soc. Am. J. 2009, 73, 751–759. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, J.; Zhao, S. Response of soil microorganisms to vegetational succession in Ziwuling Forest. Front. Environ. Sci. Eng. China 2009, 3, 421–427. [Google Scholar] [CrossRef]
- Sinsabaugh, M.R.L. A Theoretical Model of Litter Decay and Microbial Interaction. Ecol. Monogr. 2006, 76, 151–174. [Google Scholar]
- Sinsabaugh, R.L.; Shah, J.J.F. Ecoenzymatic Stoichiometry and Ecological Theory. Annu. Rev. Ecol. Evol. Syst. 2012, 43, 313–343. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Farrington, H. Nutrient limitation and soil development: Experimental test of a biogeochemical theory. Biogeochemistry 1997, 37, 63–75. [Google Scholar] [CrossRef]
- Feng, X.; Fu, B.; Lu, N.; Zeng, Y.; Wu, B. How ecological restoration alters ecosystem services: An analysis of carbon sequestration in China’s Loess Plateau. Sci. Rep. 2013, 3, 3. [Google Scholar] [CrossRef]
- Zhang, J.; Tang, X.; Zhong, S.; Yin, G.; Gao, Y.; He, X. Recalcitrant carbon components in glomalin-related soil protein facilitate soil organic carbon preservation in tropical forests. Sci. Rep. 2017, 7, 2391. [Google Scholar] [CrossRef]
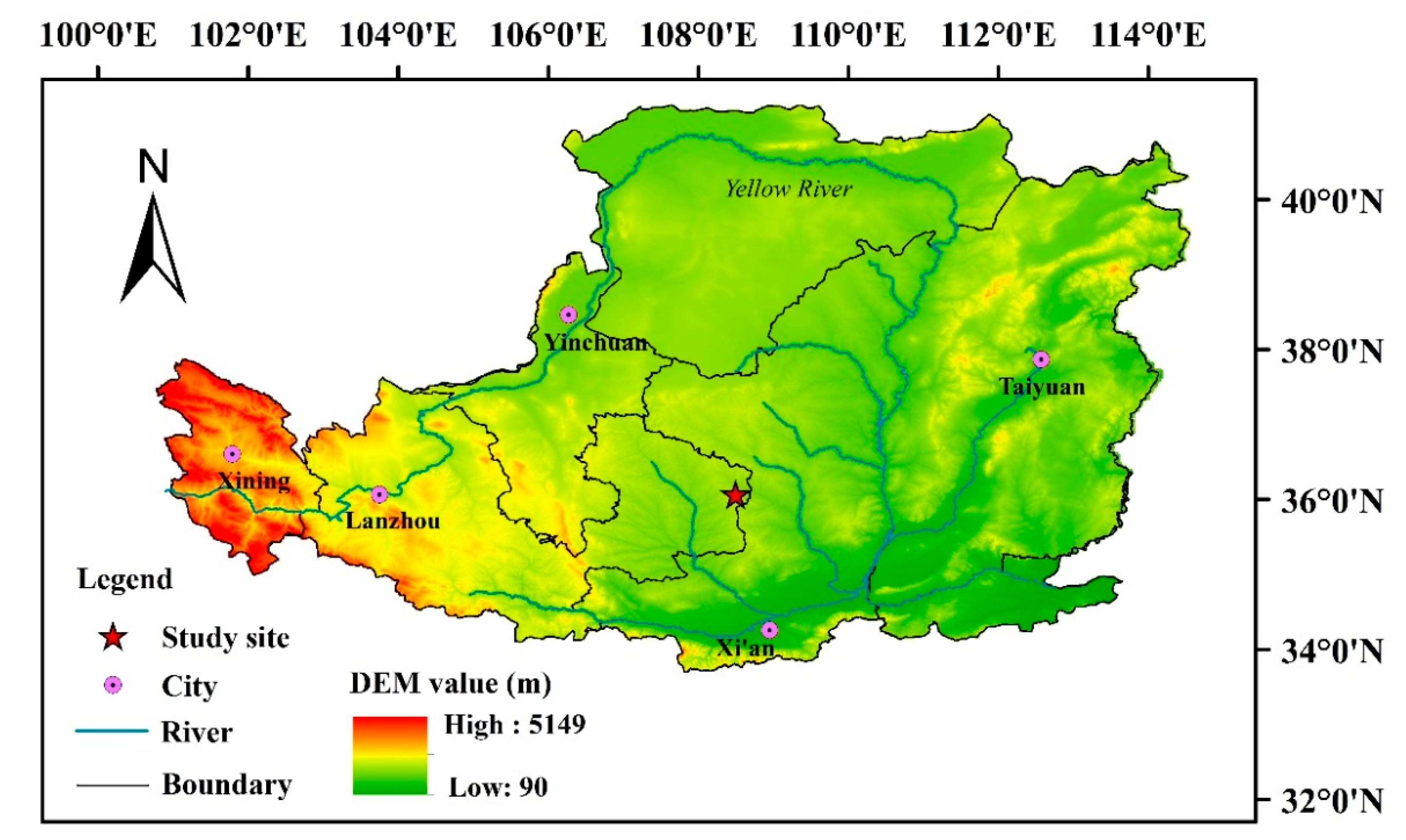
| Site | Succession Stage (in Years) | Biome | Altitude (m) | Slope (°) | Main Plant Species |
|---|---|---|---|---|---|
| S1 | 0-y | Farmland | 1280 | 0 | Corn |
| S2 | 30-y | Herbaceous | 1416 | 15 | Bothriochloa ischaemum, Lespedeza dahurica |
| S3 | 60-y | Shrub | 1346 | 21 | Sophora davidii, Carex lanceolata, Hippophae rhamnoides |
| S4 | 100-y | The pioneer forest | 1445 | 14 | Populus davidiana, Spiraea schneideriana, Carex lanceolata |
| S5 | 130-y | The mingled forest | 1440 | 18 | Populus davidiana, Quercus liaotungensis, Betula platyphylla |
| S6 | 160-y | The climax forest | 1427 | 22 | Quercus liaotungensis, Rosa hugonis, Carex lanceolata |
| Succession Stage | Soil C: N Ratio | Soil C: P Ratio | Soil N: P Ratio | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MIGA | SMGA | LMGA | MIGA | SMGA | LMGA | MIGA | SMGA | LMGA | |
| Farmland | 9.299 Bb | 9.894 Bab | 10.604 a | 11.471 C | 14.285 B | 13.518 C | 1.230 B | 1.437 C | 1.264 D |
| 30-y | 9.221 Bc | 10.086 Bb | 11.379 a | 17.016 BC | 22.498 B | 21.662 BC | 1.855 B | 2.205 BC | 1.908 CD |
| 60-y | 11.206 A | 10.597 B | 10.428 | 22.683 B | 24.002 B | 23.425 BC | 2.037 B | 2.258 BC | 2.257 BC |
| 100-y | 11.185 A | 11.374 A | 11.124 | 31.971 A | 35.748 A | 34.762 A | 2.844 A | 3.155 AB | 3.081 A |
| 130-y | 10.661 AB | 11.449 A | 10.854 | 30.181 A | 36.438 A | 30.810 AB | 2.905 A | 3.144 AB | 2.829 AB |
| 160-y | 10.955 A | 11.439 A | 12.214 | 34.834 A | 39.810 A | 37.676 A | 3.206 A | 3.415 A | 3.036 A |
| Succession Stage | Enzymatic C: N Ratio | Enzymatic C: P Ratio | Enzymatic N: P Ratio | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MIGA | SMGA | LMGA | MIGA | SMGA | LMGA | MIGA | SMGA | LMGA | |
| Farmland | 0.077 AB | 0.055 BC | 0.062 BCD | 0.191 AB | 0.165 A | 0.161 A | 2.811 | 3.089 A | 2.740 A |
| 30-y | 0.037 B | 0.034 C | 0.035 D | 0.098 B | 0.074 B | 0.083 B | 2.624 | 2.220 B | 2.367 AB |
| 60-y | 0.044 B | 0.034 C | 0.045 CD | 0.110 B | 0.082 B | 0.099 B | 2.539 | 2.413 AB | 2.169 AB |
| 100-y | 0.105 A | 0.094 A | 0.126 A | 0.272 A | 0.222 A | 0.200 A | 2.489 | 2.439 AB | 1.673 B |
| 130-y | 0.119 A | 0.088 AB | 0.072 BC | 0.185 AB | 0.188 A | 0.163 A | 1.727 | 2.179 B | 2.303 AB |
| 160-y | 0.107 A | 0.071 AB | 0.086 B | 0.237 AB | 0.190 A | 0.215 A | 2.170 | 2.632 AB | 2.453 AB |
| C | N | P | C:N | C:P | N:P | BG | BX | ALT | LAP | NAG | AP | Enzymatic C:N | Enzymatic C:P | Enzymatic N:P | GRSP-EE | GRSP-T | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | 1.00 | ||||||||||||||||
| N | 0.947 ** | 1.00 | |||||||||||||||
| P | −0.387 ** | −0.363 ** | 1.00 | ||||||||||||||
| C:N | 0.620 ** | 0.350 ** | −0.302 ** | 1.00 | |||||||||||||
| C:P | 0.968 ** | 0.915 ** | −0.582 ** | 0.613 ** | 1.00 | ||||||||||||
| N:P | 0.917 ** | 0.953 ** | −0.603 ** | 0.387 ** | 0.962 ** | 1.00 | |||||||||||
| BG | 0.739 ** | 0.783 ** | −0.17 | 0.258 * | 0.693 ** | 0.719 ** | 1.00 | ||||||||||
| BX | 0.679 ** | 0.772 ** | −0.21 | 0.13 | 0.650 ** | 0.720 ** | 0.820 ** | 1.00 | |||||||||
| ALT | 0.311 ** | 0.340 ** | −0.23 | 0.11 | 0.307 ** | 0.334 ** | 0.299 * | 0.346 ** | 1.00 | ||||||||
| LAP | 0.417 ** | 0.473 ** | −0.18 | 0.11 | 0.415 ** | 0.460 ** | 0.397 ** | 0.520 ** | 0.713 ** | 1.00 | |||||||
| NAG | 0.751 ** | 0.747 ** | −0.468 ** | 0.418 ** | 0.770 ** | 0.773 ** | 0.628 ** | 0.624 ** | 0.263 * | 0.331 ** | 1.00 | ||||||
| AP | 0.425 ** | 0.428 ** | −0.385 ** | 0.261 * | 0.448 ** | 0.459 ** | 0.289 * | 0.327 ** | 0.523 ** | 0.613 ** | 0.320 ** | 1.00 | |||||
| enzymatic C:N | 0.588 ** | 0.624 ** | −0.06 | 0.20 | 0.534 ** | 0.552 ** | 0.815 ** | 0.653 ** | -0.21 | -0.04 | 0.480 ** | 0.11 | 1.00 | ||||
| enzymatic C:P | 0.569 ** | 0.636 ** | 0.04 | 0.09 | 0.500 ** | 0.537 ** | 0.898 ** | 0.783 ** | 0.13 | 0.23 | 0.503 ** | −0.09 | 0.780 ** | 1.00 | |||
| enzymatic N:P | −0.100 | −0.04 | 0.20 | −0.23 | −0.13 | −0.10 | 0.04 | 0.09 | 0.441 ** | 0.300 * | −0.06 | −0.444 ** | −0.305 ** | 0.286 * | 1.00 | ||
| GRSP-EE | 0.780 ** | 0.819 ** | −0.273 * | 0.322 ** | 0.735 ** | 0.761 ** | 0.606 ** | 0.595 ** | 0.269 * | 0.381 ** | 0.637 ** | 0.407 ** | 0.533 ** | 0.457 ** | −0.12 | 1.00 | |
| GRSP-T | 0.794 ** | 0.819 ** | −0.23 | 0.336 ** | 0.759 ** | 0.771 ** | 0.720 ** | 0.602 ** | 0.11 | 0.277 * | 0.698 ** | 0.13 | 0.642 ** | 0.669 ** | 0.06 | 0.716 ** | 1.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiao, L.; Li, Y.; Song, Y.; Zhai, J.; Wu, Y.; Chen, W.; Liu, G.; Xue, S. Effects of Vegetation Restoration on the Distribution of Nutrients, Glomalin-Related Soil Protein, and Enzyme Activity in Soil Aggregates on the Loess Plateau, China. Forests 2019, 10, 796. https://doi.org/10.3390/f10090796
Qiao L, Li Y, Song Y, Zhai J, Wu Y, Chen W, Liu G, Xue S. Effects of Vegetation Restoration on the Distribution of Nutrients, Glomalin-Related Soil Protein, and Enzyme Activity in Soil Aggregates on the Loess Plateau, China. Forests. 2019; 10(9):796. https://doi.org/10.3390/f10090796
Chicago/Turabian StyleQiao, Leilei, Yuanze Li, Yahui Song, Jiaying Zhai, Yang Wu, Wenjing Chen, Guobin Liu, and Sha Xue. 2019. "Effects of Vegetation Restoration on the Distribution of Nutrients, Glomalin-Related Soil Protein, and Enzyme Activity in Soil Aggregates on the Loess Plateau, China" Forests 10, no. 9: 796. https://doi.org/10.3390/f10090796
APA StyleQiao, L., Li, Y., Song, Y., Zhai, J., Wu, Y., Chen, W., Liu, G., & Xue, S. (2019). Effects of Vegetation Restoration on the Distribution of Nutrients, Glomalin-Related Soil Protein, and Enzyme Activity in Soil Aggregates on the Loess Plateau, China. Forests, 10(9), 796. https://doi.org/10.3390/f10090796





