Effects of Tree Shape on the Microclimate and Fruit Quality Parameters of Camellia oleifera Abel
Abstract
1. Introduction
2. Materials and Methods
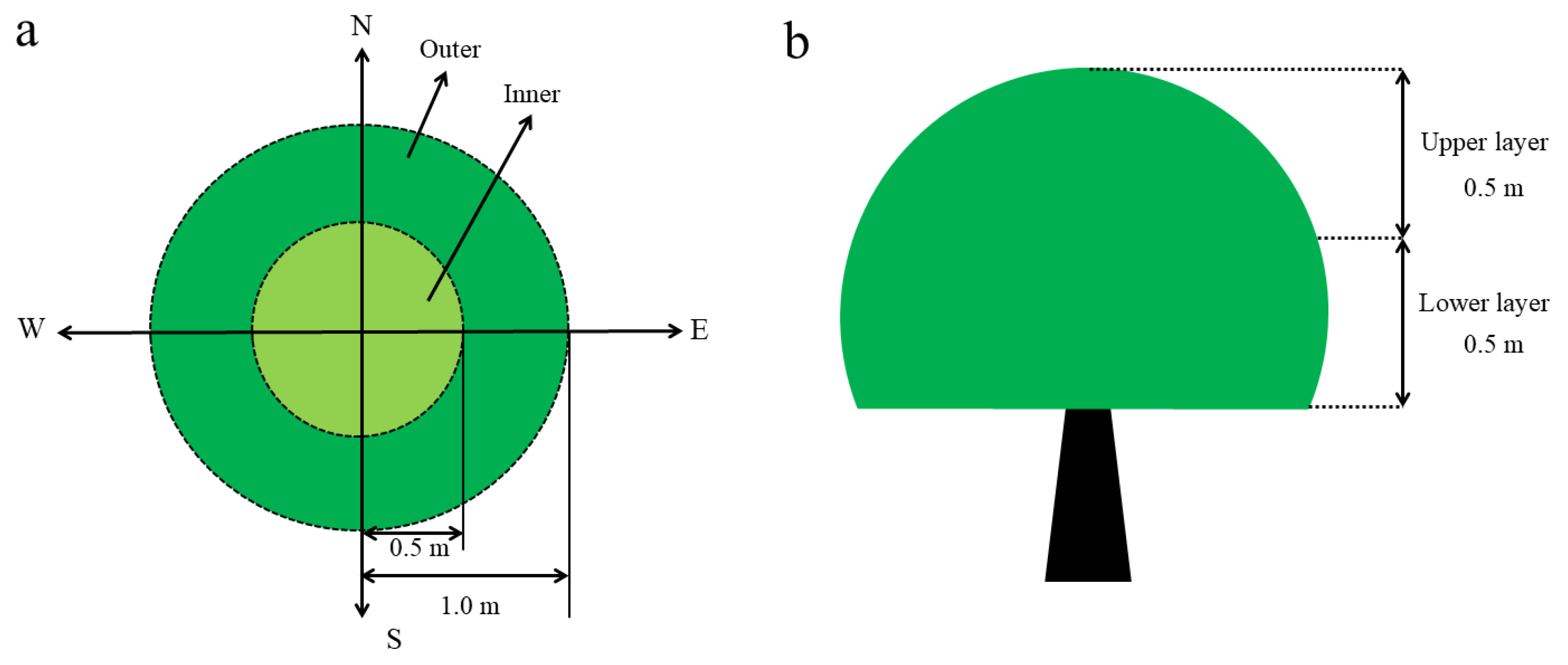
2.1. Experimental Site Description and Plant Materials
2.2. Canopy Microclimate.
2.3. Fruit Quality Parameters
2.4. Statistical Analysis
3. Results
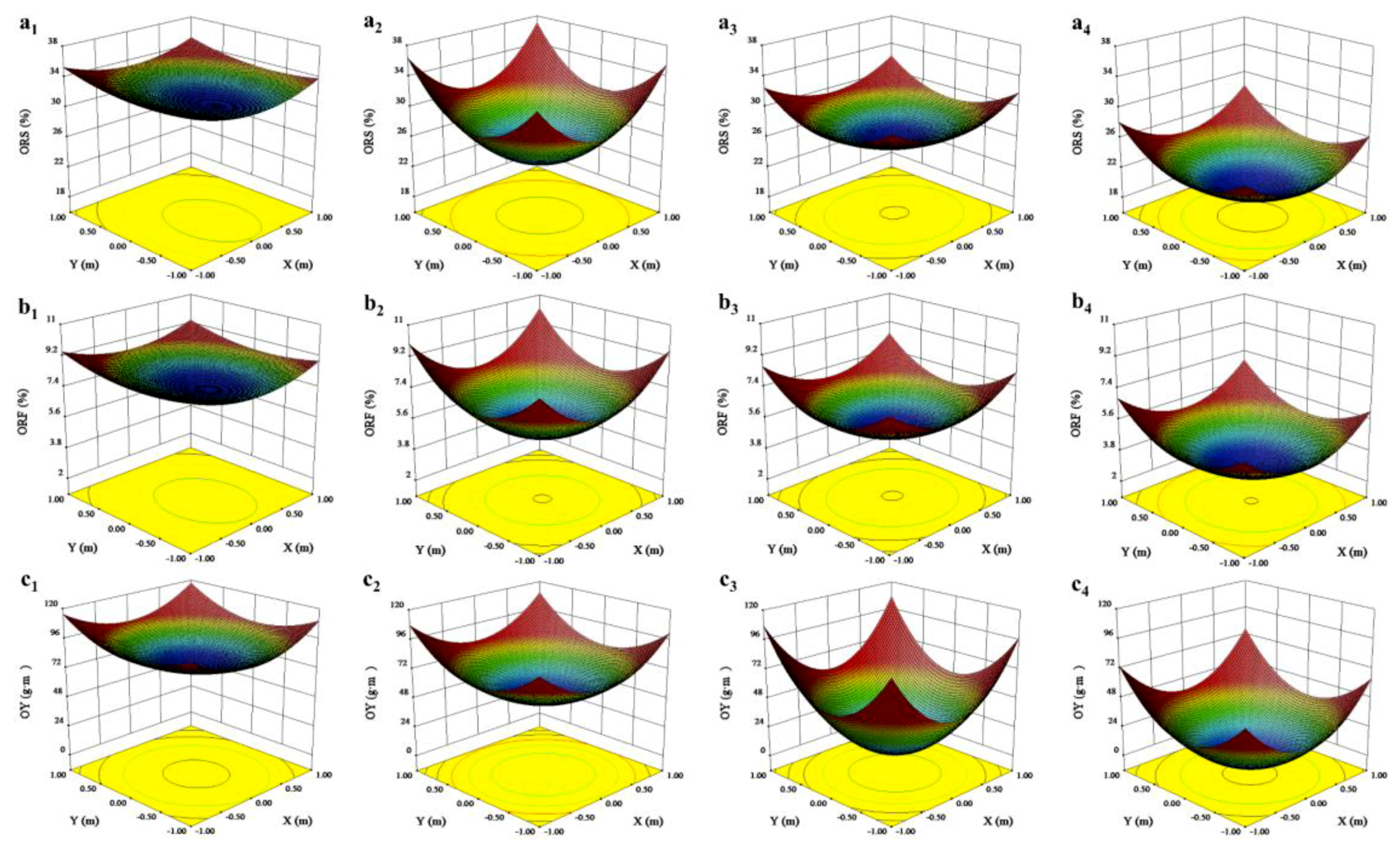
3.1. Effects of Canopy Shape on Fruit Quality Parameters
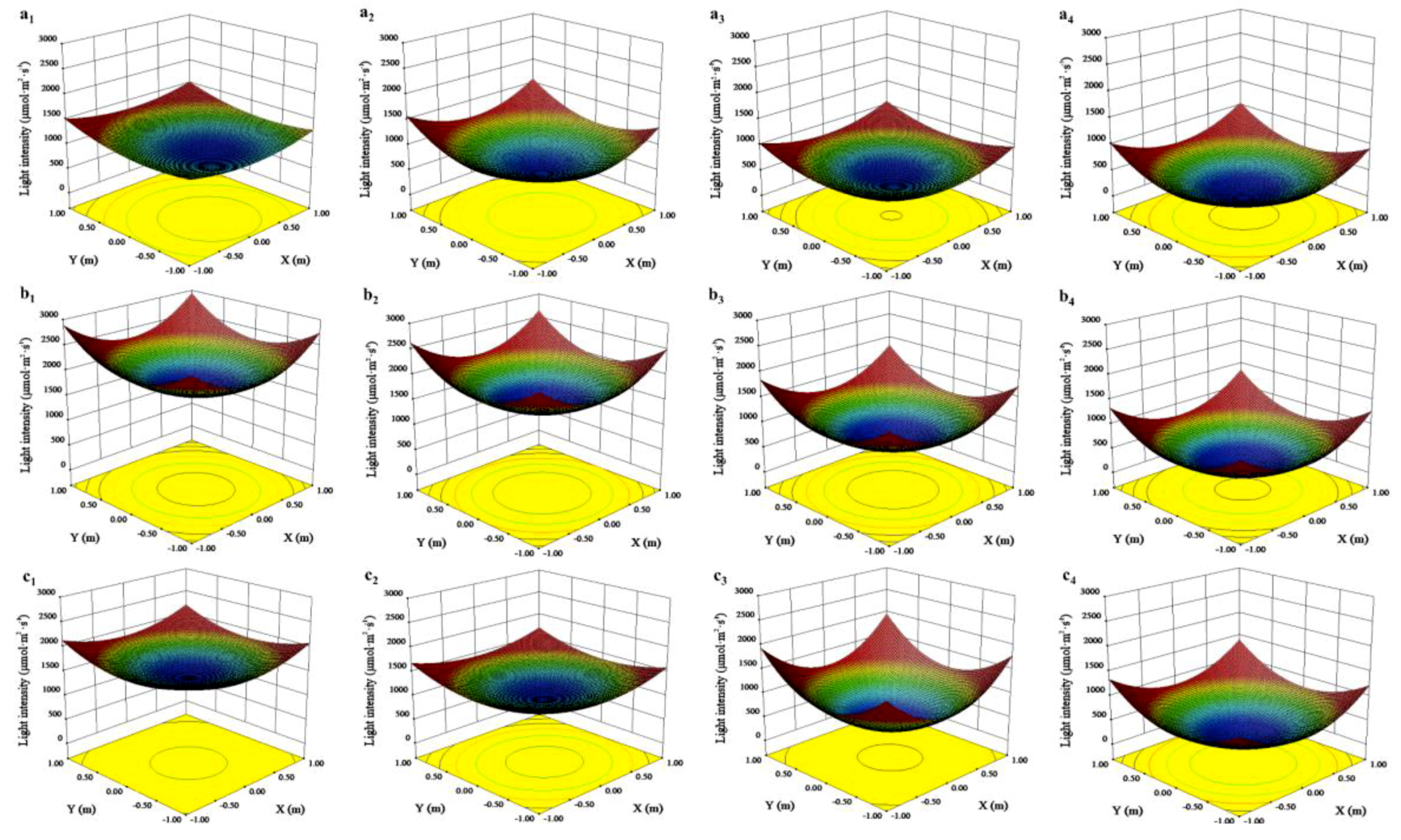
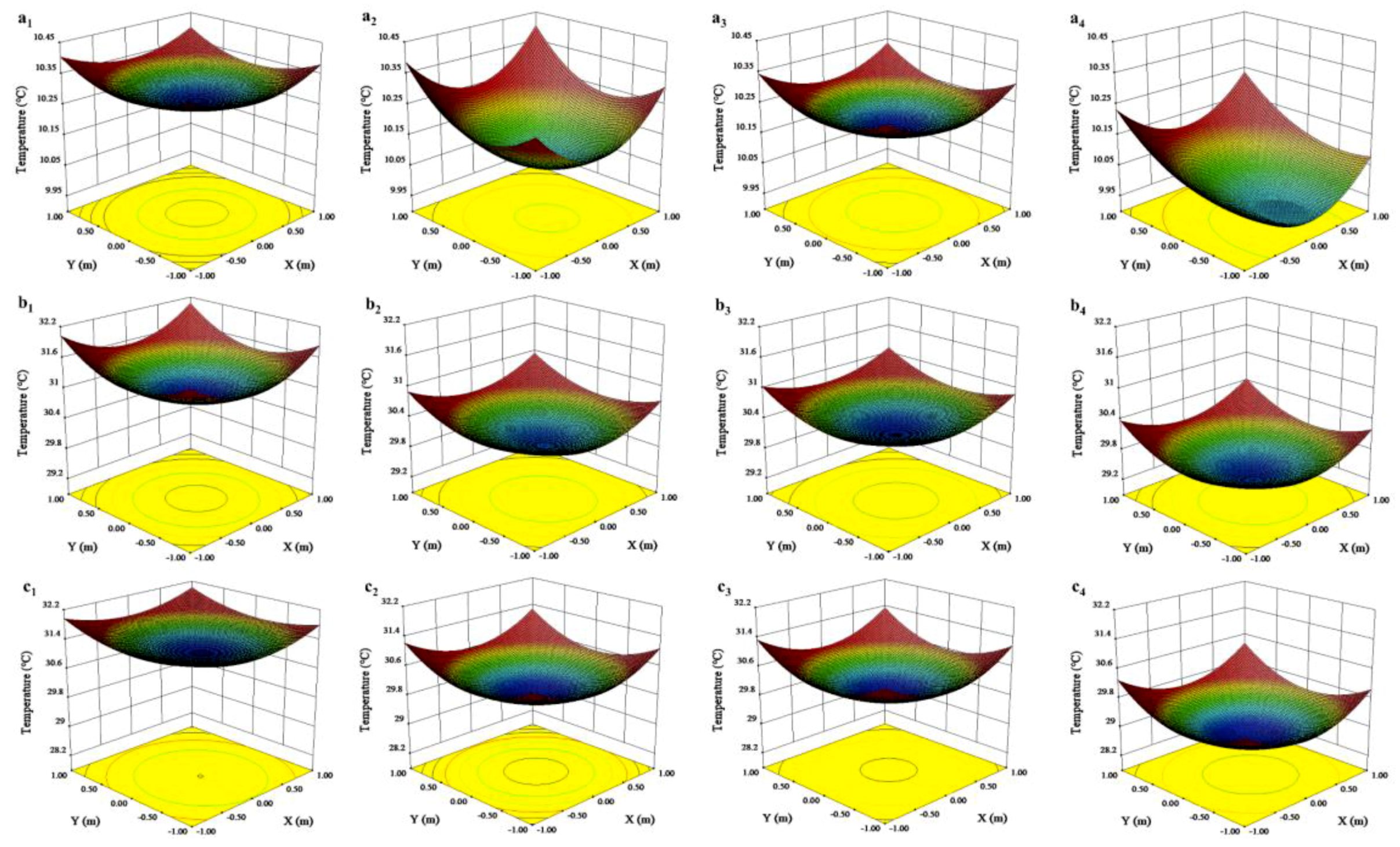
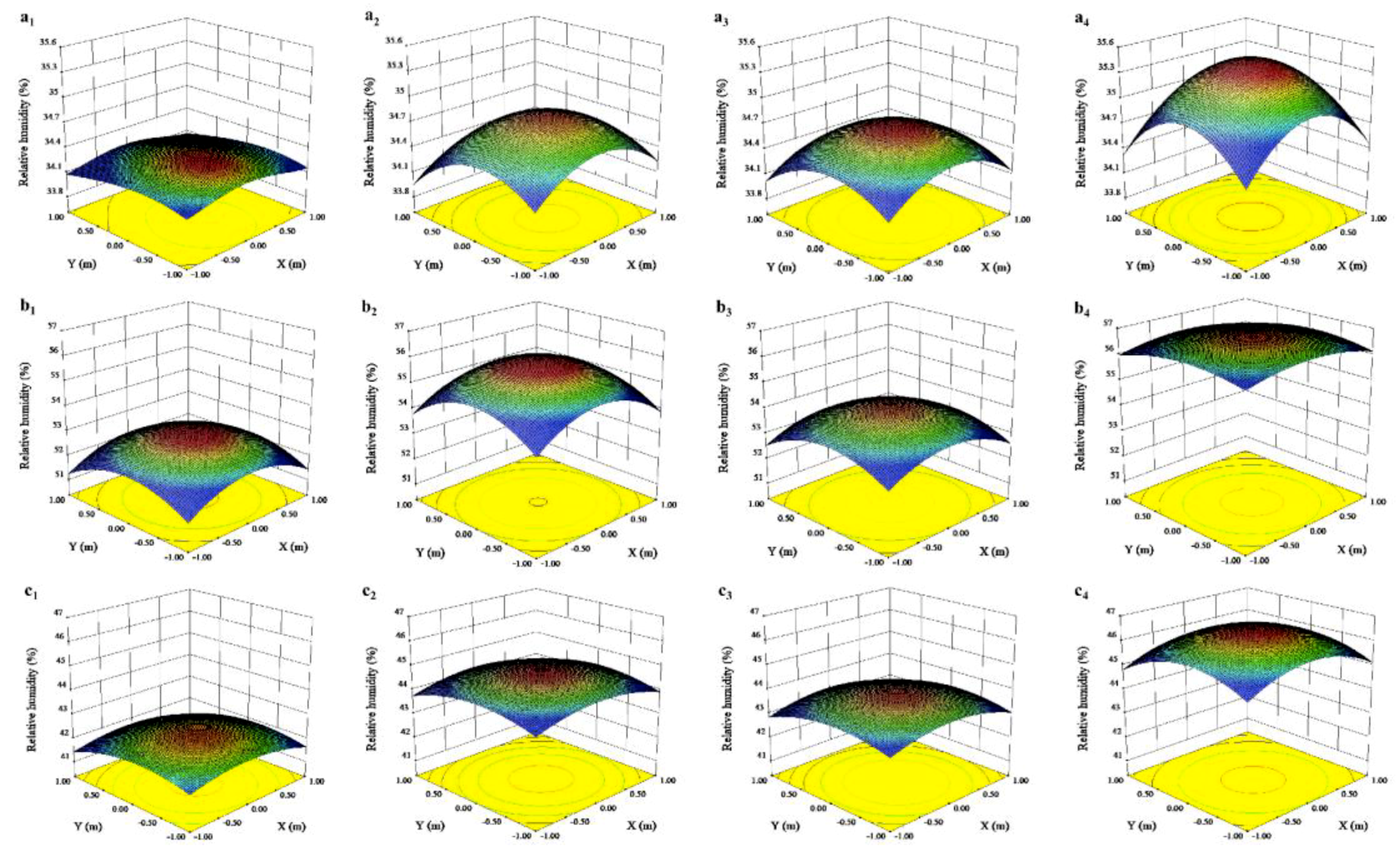
3.2. Effects of Canopy Shape on Microclimate
3.3. Correlations between Canopy Microclimates and the Oil Yield Parameters of Different Canopy Shapes
4. Discussion
4.1. Effects of Canopy Shape on Microclimate
4.2. Effects of Canopy Shape on Fruit Quality Parameters
4.3. Correlations between Canopy Microclimates and the Oil Yield Parameters of Different Canopy Shapes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Xia, E.; Jiang, J.; Huang, H.; Zhang, L.; Zhang, H.; Gao, L. Transcriptome analysis of the oil-rich tea plant, Camellia oleifera, reveals candidate genes related to lipid metabolism. PLoS ONE 2014, 9, e104150. [Google Scholar] [CrossRef] [PubMed]
- Long, Z.; Wang, D. Chemical constituents of olive oil and from Camellia oleifera seed oil. J. Chin. Cereals Oils Assoc. 2008, 23, 121–123. [Google Scholar]
- Ma, J.; Ye, H.; Rui, Y.; Chen, G.; Zhang, N. Fatty acid composition of Camellia oleifera oil. J. Verbrauch Lebensm 2011, 6, 9–12. [Google Scholar] [CrossRef]
- Singh, S.; Singh, S.; Sharma, R. Pruning alters fruit quality of mango cultivars (Mangifera indica L.) under high density planting. J. Trop. Agric. 2010, 48, 55–57. [Google Scholar]
- Singh, S.; Singh, S.; Sharma, R.; Patel, V. Influence of pruning intensity on flowering, fruit yields and floral malformation in three mango cultivars planted under high density. Indian J. Hortic. 2010, 67, 84–89. [Google Scholar]
- Li, K.; Lakso, A.; Piccioni, R.; Robinson, T. Summer pruning effects on fruit size, fruit quality, return bloom and fine root survival in apple trees. J. Hortic. Sci. Biotechnol. 2003, 78, 7. [Google Scholar] [CrossRef]
- Li, K.; Lasko, A. Photosynthesis characteristics of apple spur leaves after summer pruning to improve exposure to light. Hortscience 2004, 39, 969–972. [Google Scholar] [CrossRef]
- Calatayud, A.; Roca, D.; Gorbe, E.; Martínez, P.F. Light acclimation in rose (Rosa hybrida cv. Grand Gala) leaves after pruning: Effects on chlorophyll a fluorescence, nitrate reductase, ammonium and carbohydrate. Sci. Hortic. 2007, 111, 152–159. [Google Scholar] [CrossRef]
- Hanson, E.; Hancock, J.; Ramsdell, D.; Schilder, A.; Vanee, G.; Ledebuhr, R. Sprayer type and pruning affect the incidence of blueberry fruit rots. Hortscience 2000, 35, 235–238. [Google Scholar] [CrossRef]
- Cooley, D.; Autio, W. Summer pruning of apple: impacts on disease management. Adv. Hortic Sci 2011, 25, 199–204. [Google Scholar]
- Yi, K. Scientific pruning of Camellia oleifera. Hunan For. Sci. Technol. 2003, 30, 1–64. [Google Scholar]
- Robinson, T.; Lakso, A.; Ren, Z. Modifying apple tree canopies for improved production efficiency. HortScience 1991, 26, 1005–1012. [Google Scholar] [CrossRef]
- Cheng, X.; Liao, K.; Li, N.; Zhao, S.; Sun, H.; Mansur, N.; Liu, J. Analysis on the composition and structure of branches of two kinds of tree shapes in Korla fragrant pear. Agric. Sci. Tech. 2013, 14, 954–958. [Google Scholar]
- Tukiran, J.; Ariffin, J.; Ghani, A. Cooling effects of two types of tree canopy shape in Penang, Malaysia. Int. J. Geomate 2016, 11, 2275–2283. [Google Scholar] [CrossRef]
- Kappel, K.; Brownlee, R. Early performance of ‘Conference’ pear on four training systems. Hortscience 2001, 36, 69–71. [Google Scholar] [CrossRef]
- Jung, S.; Choi, H. Light penetration, growth, and fruit productivity in ‘Fuji’ apple trees trained to four growing systems. Sci. Hortic. 2010, 125, 672–678. [Google Scholar] [CrossRef]
- Lordan, J.; Francescatto, P.; Dominguez, L.; Robinson, T. Long-term effects of tree density and tree shape on apple orchard performance, a 20 year study-part 1, agronomic analysis. Sci. Hortic. 2018, 238, 303–317. [Google Scholar] [CrossRef]
- Sobierajski, G.; Silva, T.; Hernandes, J.; Pedro Júnior, M. Y-shaped and fruiting wall peach orchard training system in subtropical Brazil. Available online: http://www.scielo.br/scielo.php?pid=S0006-87052019005007102&script=sci_arttext (accessed on 12 November 2018).
- Duan, W.; Yuan, D.; Gao, C.; Gong, C.; Lei, X.; Liao, T. Relationship between tree composition and light utilization of Camellia oleifera. For. Res. 2013, 26, 118–122. [Google Scholar]
- Siles, P.; Vaast, P.; Dreyer, E.; Harmand, J. Rainfall partitioning into throughfall, stemflow and interception loss in a coffee (Coffea arabica L.) monoculture compared to an agroforestry system with Inga densiflora. J. Hydrol. 2010, 395, 39–48. [Google Scholar] [CrossRef]
- Marini, R.; Marini, M. Seasonal changes in specific leaf weight, net photosynthesis, and chlorophyll content of peach leaves as affected by light penetration and canopy position. J. Am. Soc. Hortic. Sci. 1983, 108, 600–605. [Google Scholar]
- Zhang, S.; Li, B.; Liu, Y.; Zhang, L.; Wang, Z.; Han, M. Fractal characteristics of two-dimensional images of ‘Fuji’ apple trees trained to two tree configurations after their winter pruning. Sci. Hortic. 2011, 130, 102–108. [Google Scholar] [CrossRef]
- Gao, Z.; Zhao, C.; Cheng, J.; Zhang, X. Tree structure and 3-D distribution of radiation in canopy of apple trees with different canopy structures in china. Chin. J. Eco-Agric. 2012, 20, 63–68. [Google Scholar] [CrossRef]
- Gao, Z.; Li, Z. Light use efficiency distribution as a function of different tree shapes in apple. J. Anim. Plant. Sci. 2015, 25, 247–253. [Google Scholar]
- Middleton, S.; McWaters, A. Increasing the yield and fruit quality of Australian apple orchards; Horticulture Australia Ltd.: Sydney, Australia, 2001. [Google Scholar]
- Hao, J. The Effect of Topiary Work on the Growth Characteristics and Photosynthetic Physiology of Camellia Sapling. Master’s Thesis, Jiangxi Agricultural University, Nanchang, China, 2011. [Google Scholar]
- Jiang, Z. The Research on Classification Transformation Technology of Low Yield Stands of Camellia oleifera. Master’s Thesis, Central South University of Forestry and Technology, Changsha, China, 2012. [Google Scholar]
- Franck, N.; Vaast, P.; Ge´nard, M.; Dauzat, J. Soluble sugars mediate sink feedback down–regulation of leaf photosynthesis of Coffea arabica in the field. Tree Physiol 2006, 26, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Siles, P.; Harmand, J.; Vaast, P. Effects of Inga densiflora, on the microclimate of coffee (Coffea arabica L.) and overall biomass under optimal growing conditions in Costa Rica. Agrofor. Syst. 2010, 78, 269–286. [Google Scholar] [CrossRef]
- Proietti, P.; Nasini, L.; Famiani, F.; Guelfi, P.; Standardi, A. Influence of light availability on fruit and oil characteristics in Olea europaea L. Acta Hortic. 2012, 949, 243–250. [Google Scholar] [CrossRef]
- Schaffer, B.; Gaye, G. Effect of pruning on light interception, specific leaf density, leaf chlorophyll content of mango. Sci. Hortic. 1989, 41, 55–61. [Google Scholar] [CrossRef]
- Cain, J. Effects of mechanical pruning of apple hedgerows with a slotting saw on light penetration and fruiting. J. Am. Soc. Hortic. Sci. 1971, 96, 664–667. [Google Scholar]
- Seeley, E.; Micke, W.; Kammereck, R. ‘Delicious’ apple fruit size and quality as influenced by radiant flux density in the immediate growing environment. J. Am. Soc. Hortic. Sci. 1980, 109, 19–24. [Google Scholar]
- Hunter, J.; Volschenk, C.; Zorer, R. Vineyard row orientation of Vitis vinifera, L. cv. Shiraz/101-14 Mgt: Climatic profiles and vine physiological status. Agric. For. Meteorol. 2016, 228–229, 104–119. [Google Scholar] [CrossRef]
- Lombard, P.; Westwood, N. Effect of hedgerow orientation on pear fruiting. Acta Hort. 1977, 69, 175–182. [Google Scholar] [CrossRef]
- Khemira, H.; Lombard, P.; Sugar, D.; Azarenko, A. Hedgerow orientation affects canopy exposure, flowering and fruiting of ‘Anjou’ pear trees. HortScience 1993, 28, 984–987. [Google Scholar] [CrossRef]
- Hatfield, J.; Prueger, J. Temperature extremes: effect on plant growth and development. Weather Clim. Extrem. 2015, 10, 4–10. [Google Scholar] [CrossRef]
- Feng, H.; Yuan, F.; Skinkis, P.; Qian, M. Influence of cluster zone leaf removal on Pinot noir grape chemical and volatile composition. Food Chem. 2015, 173, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Trad, M.; Gaaliche, B.; Renard, C.; Mars, M. Inter-and intra-tree variability in quality of figs. Influence of altitude, leaf area and fruit position in the canopy. Sci. Hortic. 2013, 162, 49–54. [Google Scholar] [CrossRef]
- Zhang, J.; Serra, S.; Leisso, R.; Musacchi, S. Effect of light microclimate on the quality of ‘d’Anjou’ pears in mature open-centre tree architecture. Biosyst. Eng. 2016, 141, 1–11. [Google Scholar] [CrossRef]
- Sharma, R.; Singh, R. Pruning intensity modifies canopy microclimate, and influences sex ratio, malformation incidence and development of fruited panicles in ‘Amrapali’ mango (Mangifera indica L.). Sci. Hortic. 2006, 109, 118–122. [Google Scholar] [CrossRef]
- Amarante, C.; Steffens, C.; Argenta, L. Yield and fruit quality of ‘Gala’ and ‘Fuji’ apple trees protected by white anti-hail net. Sci. Hortic. 2011, 129, 79–85. [Google Scholar] [CrossRef]
- Tan, H.; Liu, S.; Yang, X. Shaping and pruning of Camellia oleifera. For. Chin. 2009, 1, 57. [Google Scholar]
- Liu, J. Shaping and pruning techniques for high yield Camellia oleifera. Mod. Hortic. 2017, 21, 73. [Google Scholar]
- Pan, H.; Liu, J.; Zhou, G. Study on relationship between the tree composition and yield of Camellia oleifera. J. Jiangxi Agric. Univ. 2011, 33, 58–62. [Google Scholar]
- Wen, Y.; Su, S.; Ma, L.; Yang, S.; Wang, Y.; Wang, X. Effects of canopy microclimate on fruit yield and quality of Camellia oleifera. Sci. Hortic. 2018, 235, 132–141. [Google Scholar]
- Yuan, D.; Zhou, F.; Tan, X.; He, C.; Yuan, J.; Fan, X. Flower bud differentiation and development of male and female gametophytes in Camellia oleifera. J. Cent. South. Univ. For. Technol. 2011, 31, 65–70. [Google Scholar]
- Wang, X. Research on Phenology and Blossom Biology Of Oil-Tea Camellia. Master’s Thesis, Central South University of Forestry and Technology, Changsha, China, 2011. [Google Scholar]
- Tang, L.; Hou, C.; Huang, H.; Chen, C.; Zou, J. Light interception efficiency analysis based on three-dimensional peach canopy models. Ecol. Inform. 2015, 30, 60–67. [Google Scholar] [CrossRef]
- Marini, R.P. Pruning Peach Trees. Available online: https://www.pubs.ext.vt.edu/422/422-020/422-020.html (accessed on 12 October 2018).
- Foley, J.; Costa, M.; Delire, C.; Ramankutty, N.; Snyder, P. Green surprise? How terrestrial ecosystems could affect earth’s climate. Front. Ecol. Environ. 2003, 1, 38–44. [Google Scholar]
- Fouché, J.; Roberts, S.; Midgley, S.; Steyn, W. Peel color and blemishes in ‘Granny Smith’ apples in relation to canopy light environment. Hortscience 2010, 45, 899–905. [Google Scholar]
- Sinoquet, H.; Roux, X.; Adam, B.; Ameglio, T.; Daudet, F. RATP: A model for simulating the spatial distribution of radiation absorption, transpiration and photosynthesis within canopies: application to an isolated tree crown. Plant. Cell Environ. 2010, 24, 395–406. [Google Scholar] [CrossRef]
- Xue, H.; Han, Y.; Li, Y.; Wang, G. Spatial distribution of light interception by different plant population densities and its relationship with yield. Field Crops Res. 2015, 184, 17–27. [Google Scholar] [CrossRef]
- Connor, D.; Gómez-del-Campo, M.; Trentacoste, E. Relationships between olive yield components and simulated irradiance within hedgerows of various row orientations and spacings. Sci. Hortic. 2016, 198, 12–20. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Ahn, T.; Shin, J.; Park, K.; Son, J. Sweet pepper (Capsicum annuum L.) canopy photosynthesis modeling using 3D plant architecture and light ray-tracing. Front. Plant. Sci. 2016, 7, 1321. [Google Scholar] [CrossRef]
- He, F.; Wang, F.; Wei, Q.; Wang, X.; Zhang, Q. Relationships between the distribution of relative canopy light intensity and the peach yield and quality. Agric. Sci. Chin. 2008, 7, 297–302. [Google Scholar] [CrossRef]
- Zhang, Y.; Wen, Y.; Bai, Q.; Ma, Z.; Ye, H.; Su, S. Spatio-temporal effects of canopy microclimate on fruit yield and quality of Sapindus mukorossi Gaertn. Sci. Hortic. 2019, 251, 136–149. [Google Scholar] [CrossRef]
- Gao, Y.; Gao, S.; Jia, L.; Dai, T.; Wei, X.; Duan, J.; Liu, S.; Weng, X. Canopy characteristics and light distribution in Sapindus mukorossi, Gaertn. Are influenced by crown architecture manipulation in the hilly terrain of southeast china. Sci. Hortic. 2018, 240, 11–22. [Google Scholar] [CrossRef]
- Yuri, J.; Ibarra-Romero, M.; Vásquez, J.; Lepe, V.; González-Talice, J.; del Pozo, A. Reduction of apple tree height (Malus domestica borkh) cv. Ultra-Red Gala/MM111 does not decrease fruit yield and quality. Sci. Hortic. 2011, 130, 191–196. [Google Scholar] [CrossRef]
- Lauri, P.; Costes, E.; Regnard, J.; Brun, L.; Simon, S.; Monney, P.; Sinoquet, H. Does knowledge on fruit tree architecture and its implications for orchard management improve horticultural sustainability? An overview of recent advances in the apple. Acta Hortic. 2009, 817, 243–250. [Google Scholar] [CrossRef]
- Walcroft, A.; Roux, L.; Diaz-Espejo, A.; Dones, N.; Sinoquet, H. Effects of crown development on leaf irradiance, leaf morphology and photosynthetic capacity in a peach tree. Tree Physiol. 2002, 22, 929–938. [Google Scholar] [CrossRef]
- Agele, S.; Maraiyesa, I.; Adeniji, I. Effects of variety and row spacing on radiation interception, partitioning of dry matter and seed set efficiency in late season sunflower (Helianthus annuus L.) in a humid zone of Nigeria. Afr. J. Agric. Res. 2007, 2, 80–88. [Google Scholar]
- Yuan, J.; Shi, B.; Wu, Z.; Tang, X. Response of fruit quality and leaf photosynthesis to different sink-source relationships in Camellia oleifera. Plant. Physiol. J. 2015, 51, 1287–1292. [Google Scholar]
- Acebedo, M.; Cuevas, J.; Cañete, M. Processes affecting fruit distribution and its quality in the canopy of olive trees. Adv. Hortic. Sci. 2000, 14, 169–175. [Google Scholar]
- Cherbiy-Hoffmann, S.; Searles, P.; Hall, A.; Rousseaux, M. Influence of light environment on yield determinants and components in large olive hedgerows following mechanical pruning in the subtropics of the Southern Hemisphere. Sci. Hortic. 2012, 137, 36–42. [Google Scholar] [CrossRef]
- Tombesi, A.; Cartechini, A. L’effetto dell’ombreggiamento della chioma sulla differenziazione delle gemme a fiore dell’olivo. Riv. Di Ortoflorofrutti. It. 1986, 70, 277–285. [Google Scholar]
- Zhou, C.; Yao, X.; Lin, P.; Lu, J. Growth characteristics and dynamic analysis of water and oil content on oil-tea Camellia fruit. J. Yangzhou Univ. 2013, 34, 49–53. [Google Scholar]
- He, Y. Studies on Photosynthetic Characteristics of Oil-Tea Camellia Clones at the Fat Conversation Period of Fruit. Master’s Thesis, Central South University of Forestry and Technology, Changsha, China, 2010. [Google Scholar]
- Lestari, R.; Ebert, G.; Huyskenskeil, S. The effect of light and water supply on growth, net CO2 assimilation rate and mineral content of Salak (Salacca zalacca (Gaertn.) Voss) Seedlings. Int. J. Biol. 2011, 3, 94–104. [Google Scholar] [CrossRef]


| Position | Fruit Quality Parameter | Open Center | Round Head | Significance |
|---|---|---|---|---|
| Lower layer | FY (g·m−3) | 646.17 ± 39.81 | 575.08 ± 36.71 | NS |
| Upper layer | 1048.83 ± 15.40 | 1072.19 ± 10.45 | NS | |
| Significance | * | * | ||
| Lower layer | FSR (%) | 50.23 ± 1.81 | 47.83 ± 1.76 | NS |
| Upper layer | 55.76 ± 0.88 | 53.82 ± 0.80 | * | |
| Significance | * | * | ||
| Lower layer | DSR (%) | 45.59 ± 0.32 | 41.20 ± 0.29 | * |
| Upper layer | 47.41 ± 0.26 | 47.50 ± 0.25 | NS | |
| Significance | * | * | ||
| Lower layer | KPS (%) | 48.92 ± 0.46 | 42.85 ± 0.32 | * |
| Upper layer | 51.70 ± 0.71 | 50.52 ± 0.62 | NS | |
| Significance | * | * | ||
| Lower layer | ORS (%) | 28.91 ± 0.72 | 20.14 ± 0.71 | * |
| Upper layer | 31.34 ± 0.81 | 29.42 ± 0.51 | * | |
| Significance | * | * | ||
| Lower layer | ORF (%) | 5.48 ± 0.15 | 3.44 ± 0.10 | * |
| Upper layer | 7.70 ± 0.33 | 7.07 ± 0.31 | NS | |
| Significance | * | * | ||
| Lower layer | OY (g·m−3) | 35.41 ± 5.97 | 19.78 ± 3.67 | * |
| Upper layer | 80.76 ± 4.03 | 75.80 ± 2.65 | * | |
| Significance | * | * |
| Position | Fruit Quality Parameter | Open Center | Round Head | Significance |
|---|---|---|---|---|
| Inner canopy | FY (g·m−3) | 771.40 ± 27.66 | 703.48 ± 15.20 | * |
| Outer canopy | 923.60 ± 33.12 | 943.79 ± 31.20 | NS | |
| Significance | * | * | ||
| Inner canopy | FSR (%) | 52.43 ± 0.90 | 49.09 ± 0.89 | * |
| Outer canopy | 55.43 ± 0.79 | 52.56 ± 0.71 | * | |
| Significance | * | * | ||
| Inner canopy | DSR (%) | 45.49 ± 0.31 | 41.73 ± 0.30 | * |
| Outer canopy | 47.53 ± 0.34 | 46.98 ± 0.37 | NS | |
| Significance | * | * | ||
| Inner canopy | KPS (%) | 49.14 ± 0.44 | 45.15 ± 0.31 | * |
| Outer canopy | 51.59 ± 0.46 | 48.22 ± 0.39 | * | |
| Significance | * | * | ||
| Inner canopy | ORS (%) | 30.24 ± 0.78 | 23.75 ± 0.43 | * |
| Outer canopy | 30.98 ± 0.31 | 24.81 ± 0.29 | * | |
| Significance | NS | * | ||
| Inner canopy | ORF (%) | 6.79 ± 0.11 | 4.79 ± 0.09 | * |
| Outer canopy | 7.60 ± 0.16 | 5.72 ± 0.14 | * | |
| Significance | * | * | ||
| Inner canopy | OY (g·m−3) | 52.38 ± 4.14 | 33.70 ± 3.17 | * |
| Outer canopy | 70.19 ± 5.30 | 53.98 ± 4.37 | * | |
| Significance | * | * |
| Fruit Quality Parameter | Open Center | Round Head | ||||||
|---|---|---|---|---|---|---|---|---|
| LI | LO | UI | UO | LI | LO | UI | UO | |
| FY (g·m−3) | 545.70 ± 21.22 d | 746.64 ± 13.16 c | 997.1 ± 17.21 b | 1100.56 ± 13.22 a | 441.62 ± 12.10 d | 708.54 ± 10.12 c | 965.34 ± 21.14 b | 1061.04 ± 13.22 a |
| FSR (%) | 49.91 ± 0.40 d | 50.55 ± 0.21 c | 54.95 ± 0.54 b | 55.91 ± 0.41 a | 45.62 ± 0.61 d | 50.04 ± 0.60 c | 52.56 ± 0.71 b | 55.08 ± 0.27 a |
| DSR (%) | 45.01 ± 0.31 c | 46.17 ± 0.50 b | 45.97 ± 0.45 b | 48.89 ± 0.70 a | 36.39 ± 0.28 d | 46.01 ± 0.20 c | 46.53 ± 0.31 b | 48.47 ± 0.20 a |
| KPS (%) | 47.30 ± 0.31 c | 50.54 ± 0.23 b | 50.76 ± 0.22 b | 52.64 ± 0.60 a | 40.37 ± 0.41 d | 45.33 ± 0.77 c | 49.93 ± 0.37 b | 51.11 ± 0.31 a |
| ORS (%) | 28.54 ± 0.40 d | 29.28 ± 0.30 c | 31.94 ± 0.31 b | 32.68 ± 0.40 a | 19.66 ± 0.62 d | 22.09 ± 0.38 c | 27.55 ± 0.54 b | 30.29 ± 0.38 a |
| ORF (%) | 5.32 ± 0.12 d | 6.23 ± 0.20 c | 7.31 ± 0.33 b | 7.92 ± 0.20 a | 3.41 ± 0.14 d | 5.12 ± 0.18 c | 6.69 ± 0.23 b | 7.29 ± 0.21 a |
| OY (g·m−3) | 21.94 ± 2.11 d | 48.88 ± 4.32 c | 76.12 ± 3.20 b | 84.21 ± 3.77 a | 10.66 ± 2.14 d | 33.32 ± 3.01 c | 60.49 ± 4.14 b | 78.21 ± 1.02 a |
| Month | Position | Light Intensity (μmol·m2·s−1) | Temperature (°C) | Relative Humidity (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Open Center | Round Head | Significance | Open Center | Round Head | Significance | Open Center | Round Head | Significance | ||
| 1–2 | Lower layer | 661.53 ± 24.47 | 442.96 ± 12.24 | * | 10.23 ± 0.02 | 10.03 ± 0.01 | * | 34.53 ± 0.02 | 35.54 ± 0.02 | * |
| Upper layer | 1168.29 ± 29.86 | 908.29 ± 13.63 | * | 10.31 ± 0.01 | 10.17 ± 0.01 | * | 34.28 ± 0.01 | 34.61 ± 0.02 | * | |
| Significance | * | * | * | * | * | * | ||||
| Inner canopy | 832.75 ± 21.87 | 498.43 ± 15.01 | * | 10.24 ± 0.01 | 10.04 ± 0.01 | * | 34.48 ± 0.02 | 35.04 ± 0.01 | * | |
| Outer canopy | 997.06 ± 24.89 | 602.83 ± 14.84 | * | 10.29 ± 0.01 | 10.14 ± 0.01 | * | 34.32 ± 0.02 | 34.70 ± 0.01 | * | |
| Significance | * | * | * | * | * | * | ||||
| 7–9 | Lower layer | 1295.19 ± 35.84 | 733.10 ± 24.85 | * | 30.54 ± 0.11 | 29.44 ± 0.06 | * | 53.71 ± 0.17 | 57.44 ± 0.14 | * |
| Upper layer | 1889.25 ± 37.04 | 1476.20 ± 21.47 | * | 31.25 ± 0.08 | 30.31 ± 0.07 | * | 52.54 ± 0.09 | 55.34 ± 0.14 | * | |
| Significance | * | * | * | * | * | * | ||||
| Inner canopy | 1529.20 ± 26.67 | 925.16 ± 33.72 | * | 30.74 ± 0.06 | 29.86 ± 0.06 | * | 53.44 ± 0.07 | 56.08 ± 0.14 | * | |
| Outer canopy | 1638.24 ± 28.53 | 1261.15 ± 32.29 | * | 31.05 ± 0.07 | 30.16 ± 0.05 | * | 52.81 ± 0.01 | 55.59 ± 0.01 | * | |
| Significance | * | * | * | * | * | * | ||||
| 10 | Lower layer | 858.54 ± 22.62 | 529.78 ± 10.97 | * | 30.35 ± 0.14 | 29.00 ± 0.09 | * | 43.67 ± 0.06 | 46.10 ± 0.09 | * |
| Upper layer | 1423.85 ± 21.65 | 1209.05 ± 14.11 | * | 31.32 ± 0.11 | 30.31 ± 0.12 | * | 42.14 ± 0.06 | 44.56 ± 0.08 | * | |
| Significance | * | * | * | * | * | * | ||||
| Inner canopy | 1098.15 ± 21.67 | 781.94 ± 10.49 | * | 30.70 ± 0.06 | 29.52 ± 0.06 | * | 43.08 ± 0.07 | 45.57 ± 0.12 | * | |
| Outer canopy | 1325.86 ± 22.16 | 1078.89 ± 12.86 | * | 31.13 ± 0.07 | 30.02 ± 0.05 | * | 42.65 ± 0.06 | 45.08 ± 0.10 | * | |
| Significance | * | * | * | * | * | * | ||||
| Month | Position | Light Intensity (μmol·m2·s−1) | Temperature (°C) | Relative Humidity (%) | |||
|---|---|---|---|---|---|---|---|
| Open Center | Round Head | Open Center | Round Head | Open Center | Round Head | ||
| 1–2 | LI | 564.34 ± 13.05 d | 244.50 ± 12.22 d | 10.19 ± 0.01 d | 9.99 ± 0.03 d | 34.65 ± 0.01 a | 35.34 ± 0.02 a |
| LO | 758.68 ± 15.12 c | 561.42 ± 16.00 c | 10.25 ± 0.01 c | 10.06 ± 0.02 c | 34.41 ± 0.02 b | 34.94 ± 0.01 b | |
| UI | 1101.14 ± 29.91 b | 752.35 ± 16.18 b | 10.29 ± 0.01 b | 10.12 ± 0.01 b | 34.31 ± 0.01 c | 34.75 ± 0.02 c | |
| UO | 1235.44 ± 22.24 a | 1064.24 ± 21.00 a | 10.33 ± 0.01 a | 10.22 ± 0.01 a | 34.23 ± 0.01 d | 34.47 ± 0.02 d | |
| 7–9 | LI | 788.16 ± 6.98 d | 290.53 ± 8.24 d | 30.42 ± 0.04 d | 29.55 ± 0.02 d | 54.01 ± 0.09 a | 56.42 ± 0.09 a |
| LO | 1222.23 ± 15.79 c | 729.66 ± 14.76 c | 30.66 ± 0.05 c | 29.87 ± 0.02 c | 53.40 ± 0.01 b | 56.45 ± 0.06 b | |
| UI | 1830.24 ± 17.12 b | 1559.78 ± 14.41 b | 31.07 ± 0.04 b | 30.16 ± 0.02 b | 52.87 ± 0.01 c | 55.74 ± 0.06 c | |
| UO | 2254.26 ± 26.32 a | 1992.63 ± 25.94 a | 31.44 ± 0.04 a | 30.45 ± 0.03 a | 52.21 ± 0.01 d | 54.94 ± 0.06 d | |
| 10 | LI | 580.71 ± 4.63 d | 348.03 ± 13.61 d | 30.10 ± 0.04 d | 28.97 ± 0.05 d | 43.86 ± 0.05 a | 46.40 ± 0.07 a |
| LO | 1136.36 ± 20.26 c | 755.53 ± 11.00 c | 30.60 ± 0.06 c | 29.50 ± 0.06 c | 43.48 ± 0.04 b | 45.79 ± 0.05 b | |
| UI | 1615.60 ± 29.05 b | 1215.86 ± 26.21 b | 31.29 ± 0.06 b | 30.07 ± 0.06 b | 42.31 ± 0.04 c | 44.74 ± 0.05 c | |
| UO | 1832.11 ± 33.35 a | 1402.24 ± 26.83 a | 31.55 ± 0.07 a | 30.54 ± 0.06 a | 41.98 ± 0.03 d | 44.37 ± 0.04 d | |
| Month | Canopy Shape | Correlation Coefficient | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Light Intensity | Temperature | Relative Humidity | ||||||||
| ORS | ORF | OY | ORS | ORF | OY | ORS | ORF | OY | ||
| 1–2 | Open center | 0.839 ** | 0.834 * | 0.882 ** | 0.743 * | 0.754 * | 0.794 * | −0.852 ** | −0.854 ** | −0.886 ** |
| Round head | 0.899 ** | 0.875 ** | 0.913 ** | 0.831 * | 0.877 ** | 0.895 ** | −0.830 * | −0.818 * | −0.819 * | |
| 7–9 | Open center | 0.870 ** | 0.855 ** | 0.919 ** | 0.840 ** | 0.839 ** | 0.881 ** | −0.856 ** | −0.858 ** | −0.885 ** |
| Round head | 0.906 ** | 0.887 ** | 0.926 ** | 0.846 ** | 0.884 ** | 0.890 ** | −0.849 ** | −0.844 ** | −0.837 ** | |
| 10 | Open center | 0.859 ** | 0.865 ** | 0.903 ** | 0.846 ** | 0.850 ** | 0.893 ** | −0.841 ** | −0.838 ** | −0.885 ** |
| Round head | 0.870 ** | 0.903 ** | 0.922 ** | 0.853 ** | 0.869 ** | 0.911 ** | −0.842 ** | −0.893 ** | −0.895 ** | |
| Month | Canopy Shape | Light Intensity (103 μmol·m2·s−1) | Temperature (°C) | Relative Humidity (%) | Suitable Range of Values | |
|---|---|---|---|---|---|---|
| ORS | 1–2 | Open center | Y = −1.10X2 + 7.55X + 24.24 | NS | Y = 4.70X2 − 333.42X + 5933.30 | <3432 μmol·m2·s−1 |
| (p = 0.0481, R2 = 0.704) | (p = 0.0421, R2 = 0.726) | >35.47% | ||||
| Round head | Y = −17.10X2 + 43.05X + 4.82 | Y = 209.01X2 − 4170.20X + 20822 | Y = 8.66X2 − 616.74X + 11005 | <1259 μmol·m2·s−1 | ||
| (p = 0.0129, R2 = 0.824) | (p = 0.0388, R2 = 0.727) | (p = 0.0327, R2 = 0.745) | >9.98°C | |||
| >35.61% | ||||||
| 7–9 | Open center | Y = −5.99X2 + 23.94X + 7.60 | Y = −0.21X2 + 16.98X − 290.70 | Y = 0.16X2 − 19.53X + 605.47 | <1998 μmol·m2·s−1 | |
| (p = 0.0022, R2 = 0.818) | (p = 0.0132, R2 = 0.697) | (p = 0.0311, R2 = 0.630) | <40.43 °C | |||
| <53.70% | ||||||
| Round head | Y = 1.09X2 + 9.45X + 12.92 | Y = 7.15X2 − 416.58X + 6085.70 | Y = 2.63X2 − 301.59X + 8649.60 | >733 μmol·m2·s−1 | ||
| (p = 0.0213, R2 = 0.768) | (p = 0.0233, R2 = 0.765) | (p = 0.0123, R2 = 0.825) | >29.13 °C | |||
| <57.45% | ||||||
| 10 | Open center | Y = 8.99X2 − 16.47X + 36.58 (p = 0.0089, R2 = 0.603) | Y = 0.64X2 − 36.97X + 558.30 | Y = −0.11X2 + 7.26X − 85.75 | >916 μmol·m2·s−1 | |
| (p = 0.0135, R2 = 0.652) | (p = 0.0145, R2 = 0.686) | >28.88 °C | ||||
| <43.70 | ||||||
| Round head | Y = 3.33X2 − 7.38X + 15.28 | Y = 1.11X2 − 58.84X + 792.98 | Y = −0.45X2 + 34.33X − 611.64 | >1108 μmol·m2·s−1 | ||
| (p = 0.0310, R2 = 0.700) | (p = 0.0073, R2 = 0.696) | (p = 0.0035, R2 = 0.732) | >26.50 °C | |||
| <38.14% | ||||||
| ORF | 1–2 | Open center | Y = −2.61X2 + 9.88X − 0.0662 | NS | Y = 1.71X2 − 126.50X + 2337 | <1893 μmol·m2·s−1 |
| (p = 0.0477, R2 = 0.703) | (p = 0.0373, R2 = 0.729) | > 36.99% | ||||
| Round head | Y = 2.00X2 + 4.41X + 1.49 | Y = 223.83X2 − 4501.40X + 22636 | Y = 3.69X2 − 262.82X + 4674 | >443 μmol·m2·s−1 | ||
| (p = 0.0016, R2 = 0.766) | (p = 0.0232, R2 = 0.778) | (p = 0.0025, R2 = 0.909) | >10.06 °C | |||
| >35.61% | ||||||
| 7–9 | Open center | Y = −6.37X2 + 24.78X − 16.22 | Y = −0.96X2 + 62.91X − 1019.30 | Y = −0.034X2 + 1.64X + 15.61 | <1945 μmol·m2·s−1 | |
| (p = 0.0011, R2 = 0.731) | (p = 0.0037, R2 = 0.699) | (p = 0.0056, R2 = 0.646) | <32.77 °C | |||
| <53.70% | ||||||
| Round head | Y = 1.69X2 + 0.87X + 2.05 | Y = 3.21X2 − 187.66X + 2748.70 | Y = 0.99X2 − 112.90X + 3233.50 | >733 μmol·m2·s−1 | ||
| (p = 0.0016, R2 = 0.863) | (p = 0.0032, R2 = 0.899) | (p = 0.0096, R2 = 0.939) | >29.23 °C | |||
| >57.02% | ||||||
| 10 | Open center | Y = 6.81X2 − 11.76X + 10.70 | Y = 0.26X2 − 13.48X + 175.39 | Y = −0.24X2 + 18.83X − 360.66 | >863 μmol·m2·s−1 | |
| (p = 0.0410, R2 = 0.579) | (p = 0.0316, R2 = 0.662) | (p = 0.0033, R2 = 0.700) | >25.92 °C | |||
| <39.23 % | ||||||
| Round head | Y = 2.99X2 − 0.14X + 2.74 | Y = 0.85X2 − 47.55X + 670.02 | Y = 0.11X2 − 12.28X + 337.56 | >23.41 μmol·m2·s−1 | ||
| (p = 0.0013, R2 = 0.893) | (p = 0.0014, R2= 0.889) | (p = 0.0006, R2 = 0.915) | >27.97 °C | |||
| <46.10% | ||||||
| OY | 1–2 | Open center | Y = −27.97X2 + 148.73X − 52.20 | Y = −1717.30X2 + 35808X − 186554 | Y = 239.04X2 − 16614X − 288696 | <2652 μmol·m2·s−1 |
| (p = 0.0016, R2 = 0.780) | (p = 0.0432, R2 = 0.718) | (p = 0.0021, R2 = 0.817) | <10.43 °C | |||
| >34.75% | ||||||
| Round head | Y = 12.81X2 + 97.26X − 23.07 | Y = 2973.80X2 − 59717X + 299830 | Y = 91.66X2 − 6486.70X + 114738 | >443 μmol·m2·s−1 | ||
| (p = 0.0111, R2 = 0.834) | (p = 0.0013, R2 = 0.901) | (p = 0.0010, R2 = 0.866) | >10.04 °C | |||
| >35.38% | ||||||
| 7–9 | Open center | Y = −102.75X2 + 413.38X − 331.92 | Y = −25.06X2 + 1619X − 26035 | Y = 9.35X2 − 1034.00X + 28611 | <2012 μmol·m2·s−1 | |
| (p = 0.0050, R2 = 0.789) | (p = 0.0056, R2 = 0.836) | (p = 0.0010, R2 = 0.654) | <32.30 °C | |||
| <53.70 % | ||||||
| Round head | Y = 62.33X2 − 62.07X + 32.62 | Y = 87.37X2 − 5155.50X + 76070 | Y = 21.65X2 − 2466.90X + 70300 | >498 μmol·m2·s−1 | ||
| (p = 0.0022, R2 = 0.869) | (p = 0.0024, R2 = 0.950) | (p = 0.0011, R2= 0.943) | >29.50 °C | |||
| >56.97% | ||||||
| 10 | Open center | Y = 159.25X2 − 282.44X + 161.94 | Y = −0.019X2 + 51.29X + 1505.70 | Y = 3.70X2 − 352.15X + 8347.90 | >887 μmol·m2·s−1 | |
| (p = 0.0039, R2 = 0.803) | (p = 0.0011, R2 = 0.771) | (p = 0.0342, R2 = 0.661) | >30.40 °C | |||
| <43.70% | ||||||
| Round head | Y = 97.80X2 − 87.51X + 38.67 | Y = 26.78X2 − 1544.70X + 22290 | Y = 9.43X2 − 893.43X + 21173 | >447 μmol·m2·s−1 | ||
| (p = 0.0014, R2 = 0.912) | (p = 0.0010, R2 = 0.904) | (p = 0.0012, R2 = 0.908) | >28.84 °C | |||
| <46.10% |
| Canopy Shape | Month | Light Intensity (μmol·m2·s−1) | Temperature (°C) | Relative Humidity (%) |
|---|---|---|---|---|
| Open center | 1–2 | <1893 | <10.43 | >36.99 |
| 7–9 | <1954 | <32.30 | <53.70 | |
| 10 | >916 | >30.40 | <39.23 | |
| Round head | 1–2 | 443–1259 | >10.06 | >35.61 |
| 7–9 | >733 | >29.50 | 57.02–57.45 | |
| 10 | >1108 | >28.84 | <38.14 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, Y.; Zhang, Y.; Su, S.; Yang, S.; Ma, L.; Zhang, L.; Wang, X. Effects of Tree Shape on the Microclimate and Fruit Quality Parameters of Camellia oleifera Abel. Forests 2019, 10, 563. https://doi.org/10.3390/f10070563
Wen Y, Zhang Y, Su S, Yang S, Ma L, Zhang L, Wang X. Effects of Tree Shape on the Microclimate and Fruit Quality Parameters of Camellia oleifera Abel. Forests. 2019; 10(7):563. https://doi.org/10.3390/f10070563
Chicago/Turabian StyleWen, Yue, Yunqi Zhang, Shuchai Su, Shaoyan Yang, Lvyi Ma, Lingyun Zhang, and Xiangnan Wang. 2019. "Effects of Tree Shape on the Microclimate and Fruit Quality Parameters of Camellia oleifera Abel" Forests 10, no. 7: 563. https://doi.org/10.3390/f10070563
APA StyleWen, Y., Zhang, Y., Su, S., Yang, S., Ma, L., Zhang, L., & Wang, X. (2019). Effects of Tree Shape on the Microclimate and Fruit Quality Parameters of Camellia oleifera Abel. Forests, 10(7), 563. https://doi.org/10.3390/f10070563





