Not Seeing the Forest for the Trees: The Oversight of Defaunation in REDD+ and Global Forest Governance
Abstract
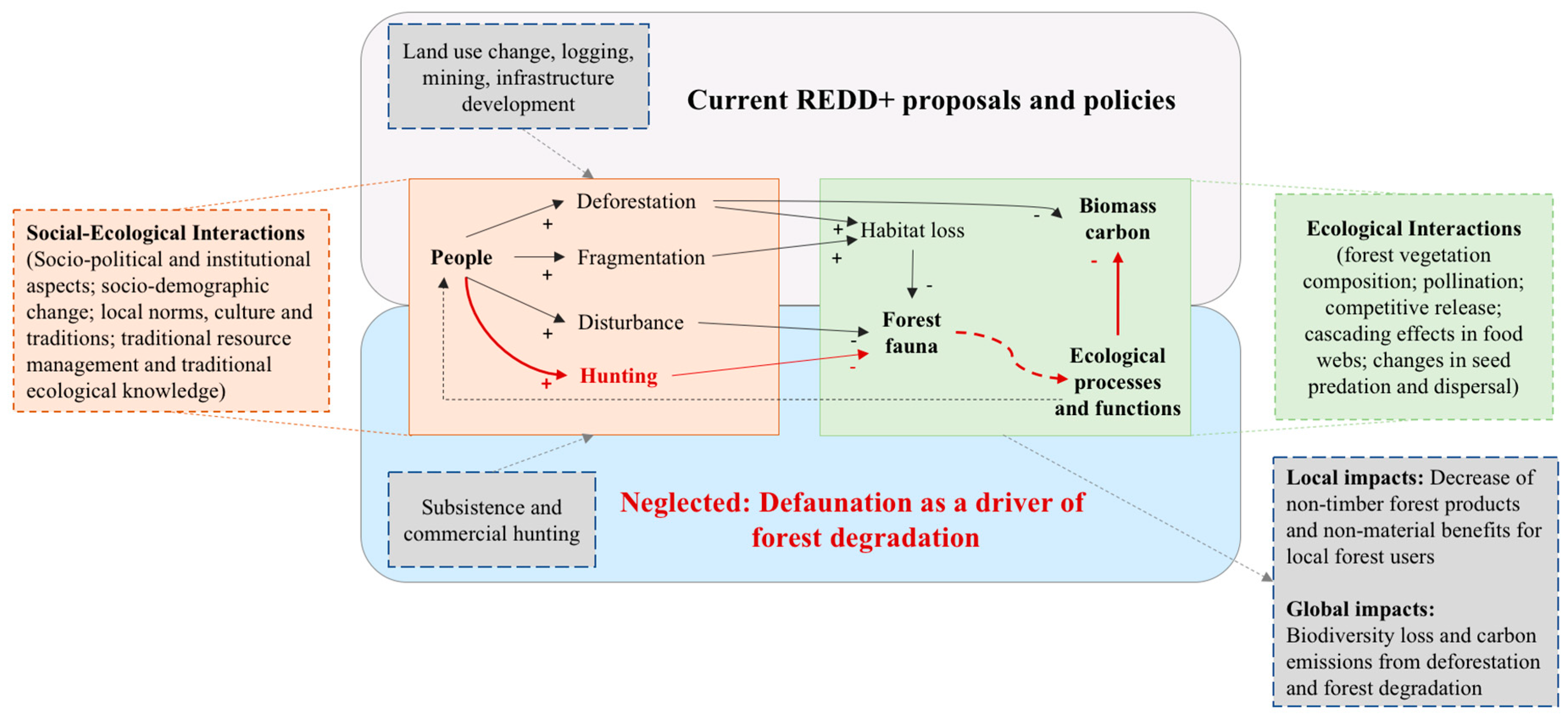
1. Introduction
1.1. The Role of Forest and Fauna in Livelihoods
1.2. Consequences of Defaunation
1.3. Objectives
2. Materials and Methods
3. Results
3.1. Forest Biodiversity and the UNFCCC
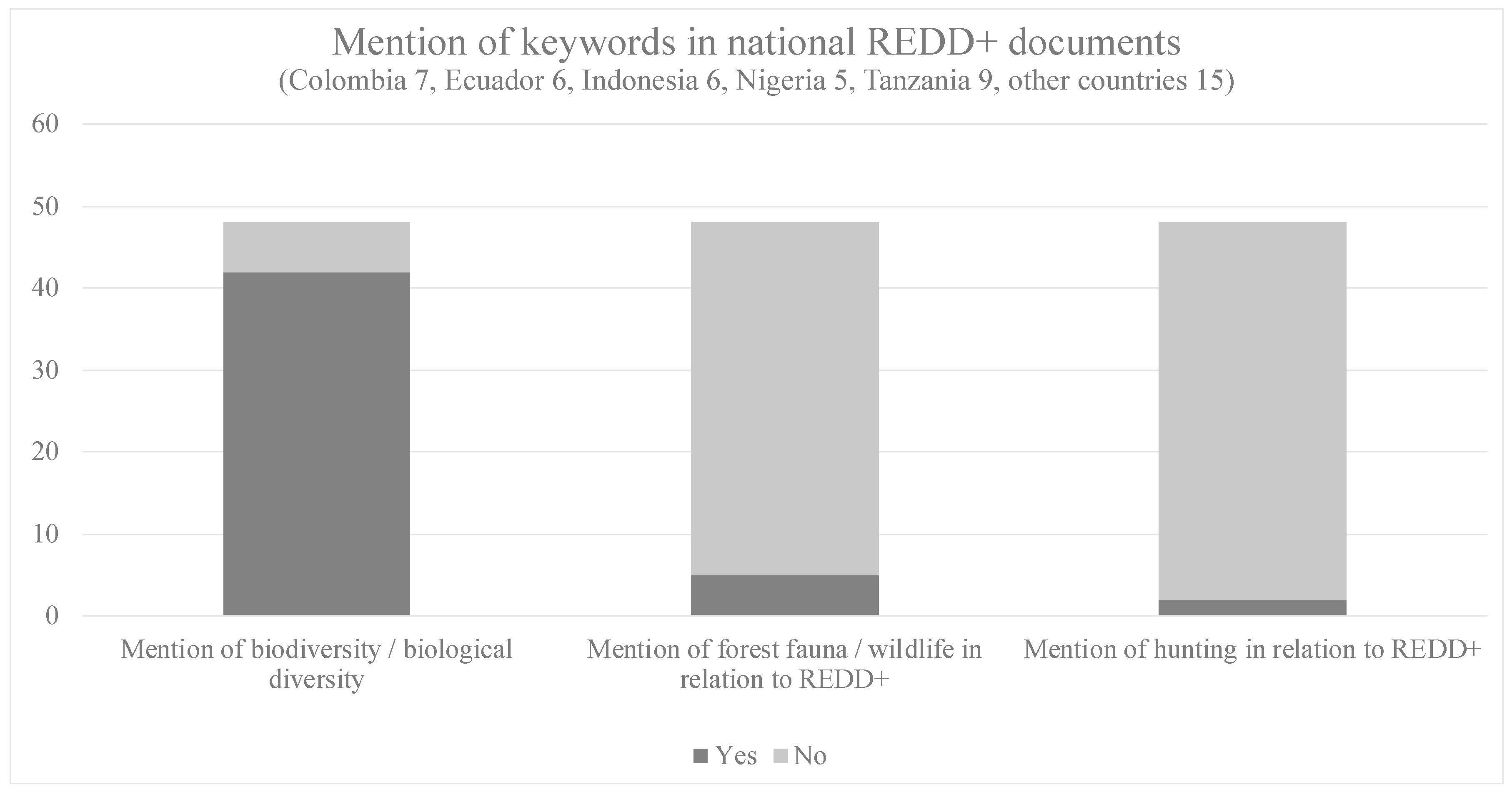
3.2. National REDD+ Programs
3.3. Sub-National REDD+ Projects
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Malmer, A.; Murdiyarso, D.; Bruijnzeel, L.A.; Ilstedt, U. Carbon sequestration in tropical forests and water: A critical look at the basis for commonly used generalizations. Chang. Biol. 2010, 16, 599–604. [Google Scholar] [CrossRef]
- Canadell, J.G.; Raupach, M.R.; Canadell, J. Managing Forests for Climate Change Mitigation. Science 2008, 320, 1456–1457. [Google Scholar] [CrossRef]
- Van der Werf, G.R.; Morton, D.C.; DeFries, R.S.; Olivier, J.G.J.; Kasibhatla, P.S.; Jackson, R.B.; Collatz, G.J.; Randerson, J.T. CO2 emissions from forest loss. Nat. Geosci 2009, 2, 737–738. [Google Scholar] [CrossRef]
- Jackson, R.B.; Randerson, J.T.; Canadell, J.G.; Anderson, R.G.; Avissar, R.; Baldocchi, D.D.; Bonan, G.B.; Caldeira, K.; Diffenbaugh, N.S.; Field, C.B.; et al. Protecting climate with forests. Environ. Res. Lett. 2008, 3, 044006. [Google Scholar] [CrossRef]
- Butchart, S.H.M.; Walpole, M.; Collen, B.; Van Strien, A.; Scharlemann, J.P.W.; Almond, R.E.A.; Baillie, J.E.M.; Bomhard, B.; Brown, C.; Bruno, J.; et al. Global Biodiversity: Indicators of Recent Declines. Science 2010, 328, 1164–1168. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nat. Cell Boil. 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Sunderland, T.C.H.; Powell, B.; Ickowitz, A.; Foli, S.; Pinedo-Vasquez, M.; Nasi, R.; Padoch, C. Food Security and Nutrition: The Role of Forests; Center for International Forestry Research (CIFOR): Bogor, Indonesia, 2013. [Google Scholar]
- Wunder, S.; Angelsen, A.; Belcher, B. Forests, Livelihoods, and Conservation: Broadening the Empirical Base. World Dev. 2014, 64, S1–S11. [Google Scholar] [CrossRef]
- Dirzo, R.; Raven, P.H. Global State of Biodiversity and Loss. Annu. Resour. 2003, 28, 137–167. [Google Scholar] [CrossRef]
- Turner, I.M. Species Loss in Fragments of Tropical Rain Forest: A Review of the Evidence. J. Appl. Ecol. 1996, 33, 200. [Google Scholar] [CrossRef]
- Estrada, A.; Garber, P.A.; Rylands, A.B.; Roos, C.; Fernandez-Duque, E.; Di Fiore, A.; Nekaris, K.A.-I.; Nijman, V.; Heymann, E.W.; Lambert, J.E.; et al. Impending extinction crisis of the world’s primates: Why primates matter. Sci. Adv. 2017, 3, e1600946. [Google Scholar] [CrossRef]
- Barlow, J.; Lennox, G.D.; Ferreira, J.; Berenguer, E.; Lees, A.C.; Mac Nally, R.; Thomson, J.R.; Ferraz, S.F.D.B.; Louzada, J.; Oliveira, V.H.F.; et al. Anthropogenic disturbance in tropical forests can double biodiversity loss from deforestation. Nat. Cell Boil. 2016, 535, 144–147. [Google Scholar] [CrossRef]
- Pearson, T.R.H.; Brown, S.; Murray, L.; Sidman, G. Greenhouse gas emissions from tropical forest degradation: An underestimated source. Carbon Balance Manag. 2017, 12, 3. [Google Scholar] [CrossRef]
- Pimentel, D. Soil Erosion: A Food and Environmental Threat. Environ. Dev. Sustain. 2006, 8, 119–137. [Google Scholar] [CrossRef]
- Southgate, D.; Whitaker, M. Promoting Resource Degradation in Latin America: Tropical Deforestation, Soil Erosion, and Coastal Ecosystem Disturbance in Ecuador. Econ. Dev. Cult. Chang. 1992, 40, 787–807. [Google Scholar] [CrossRef]
- Makarieva, A.M.; Gorshkov, V.G. Biotic pump of atmospheric moisture as driver of the hydrological cycle on land. Hydrol. Earth Sci. 2007, 11, 1013–1033. [Google Scholar] [CrossRef]
- Angelsen, A.; Jagger, P.; Babigumira, R.; Belcher, B.; Hogarth, N.J.; Bauch, S.; Börner, J.; Smith-Hall, C.; Wunder, S. Environmental Income and Rural Livelihoods: A Global-Comparative Analysis. World Dev. 2014, 64, S12–S28. [Google Scholar] [CrossRef]
- Pistorius, T. From RED to REDD+: The evolution of a forest-based mitigation approach for developing countries. Curr. Opin. Environ. Sustain. 2012, 4, 638–645. [Google Scholar] [CrossRef]
- UNFCCC. Decisions adopted by the Conference of the Parties—Decision 1/CP.16; United Nations Framework Convention on Climate Change: Cancun, Mexico, 2011. [Google Scholar]
- Kanowski, P.J.; McDermott, C.L.; Cashore, B.W. Implementing REDD+: Lessons from analysis of forest governance. Environ. Sci. Policy 2011, 14, 111–117. [Google Scholar] [CrossRef]
- Stiem, L.; Krause, T. Exploring the Impact of Social Norms and Perceptions on Women’s Participation in Customary Forest and Land Governance in the Democratic Republic of Congo—Implications for REDD+. Int. For. Rev. 2016, 18, 110–122. [Google Scholar] [CrossRef]
- Nuesiri, E. Feigning Democracy: Performing Representation in the UN-REDD Funded Nigeria-REDD Programme. Conserv. Soc. 2017, 15, 384–399. [Google Scholar] [CrossRef]
- Aguilar-Støen, M. Global forest conservation initiatives as spaces for participation in Colombia and Costa Rica. Geoforum 2015, 61, 36–44. [Google Scholar] [CrossRef]
- Chomba, S.; Kariuki, J.; Lund, J.F.; Sinclair, F. Roots of inequity: How the implementation of REDD+ reinforces past injustices. Land Use Policy 2016, 50, 202–213. [Google Scholar] [CrossRef]
- Karsenty, A.; Vogel, A.; Castell, F. “Carbon rights”, REDD+ and payments for environmental services. Environ. Sci. Policy 2014, 35, 20–29. [Google Scholar] [CrossRef]
- Fletcher, R.; Dressler, W.; Büscher, B.; Anderson, Z.R. Questioning REDD+ and the future of market-based conservation. Conserv. Biol. 2016, 30, 673–675. [Google Scholar] [CrossRef]
- Corbera, E. Problematizing REDD+ as an experiment in payments for ecosystem services. Curr. Opin. Environ. Sustain. 2012, 4, 612–619. [Google Scholar] [CrossRef]
- Cabello, J.; Gilbertson, T. A colonial mechanism to enclose lands: A critical review of two REDD+-focused special issues. Ephemera 2012, 12, 162–180. [Google Scholar]
- Angelsen, A.; Brockhaus, M.; Duchelle, A.E.; Larson, A.; Martius, C.; Sunderlin, W.D.; Verchot, L.; Wong, G.; Wunder, S. Learning from REDD+: A response to Fletcher et al. Conserv. Biol. 2017, 31, 718–720. [Google Scholar] [CrossRef]
- UNFCCC. Adoption of the Paris Agreement—Draft decision -/CP.21; United Nations Framework Convention on Climate Change: Paris, France, 2015; p. 32. [Google Scholar]
- Hein, J.; Guarin, A.; Frommé, E.; Pauw, P. Deforestation and the Paris climate agreement: An assessment of REDD + in the national climate action plans. For. Policy Econ. 2018, 90, 7–11. [Google Scholar] [CrossRef]
- Hinsley, A.; Entwistle, A.; Pio, D.V. Does the long-term success of REDD+ also depend on biodiversity? Oryx 2015, 49, 216–221. [Google Scholar] [CrossRef]
- Krause, T.; Zambonino, H. More than just trees—Animal species diversity and participatory forest monitoring in the Ecuadorian Amazon. Int. J. Biodivers. Sci. Serv. Manag. 2013, 9, 225–238. [Google Scholar] [CrossRef]
- Brodie, J.F. Carbon Costs and Bushmeat Benefits of Hunting in Tropical Forests. Ecol. Econ. 2018, 152, 22–26. [Google Scholar] [CrossRef]
- Weatherley-Singh, J.; Gupta, A. Drivers of deforestation and REDD+ benefit-sharing: A meta-analysis of the (missing) link. Environ. Sci. Policy 2015, 54, 97–105. [Google Scholar] [CrossRef]
- FAO. The state of the world’s forests 2018—Forest pathways to sustainable development. In The State of the World; Food and Agricultural Organization of the United Nations: Rome, Italy, 2018; p. 139. [Google Scholar]
- Gardner, T.A.; Burgess, N.D.; Aguilar-Amuchastegui, N.; Barlow, J.; Berenguer, E.; Clements, T.; Danielsen, F.; Ferreira, J.; Foden, W.; Kapos, V.; et al. A framework for integrating biodiversity concerns into national REDD+ programmes. Biol. Conserv. 2012, 154 (Suppl. C), 61–71. [Google Scholar] [CrossRef]
- Angelsen, A.; Dokken, T. Environmental Reliance, Climate Exposure, and Vulnerability—A Cross-Section Analysis of Structural and Stochastic Poverty. In Policy Research Working Paper 7474—Shock Waves: Managing the Impact of Climate Change on Poverty; The World Bank: Washington, DC, USA, 2015; p. 47. [Google Scholar]
- Angelsen, A.; Dokken, T. Climate exposure, vulnerability and environmental reliance: A cross-section analysis of structural and stochastic poverty. Dev. Econ. 2018, 23, 257–278. [Google Scholar] [CrossRef]
- De Merode, E.; Homewood, K.; Cowlishaw, G. The value of bushmeat and other wild foods to rural households living in extreme poverty in Democratic Republic of Congo. Boil. Conserv. 2004, 118, 573–581. [Google Scholar] [CrossRef]
- Schulte-Herbruggen, B.; Cowlishaw, G.; Homewood, K.; Rowcliffe, J.M. The Importance of Bushmeat in the Livelihoods of West African Cash-Crop Farmers Living in a Faunally-Depleted Landscape. PLoS ONE 2013, 8, e72807. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.R.; Meilby, H.; Smith-Hall, C.; Pouliot, M.; Treue, T. The Importance of Wild Meat in the Global South. Ecol. Econ. 2018, 146, 696–705. [Google Scholar] [CrossRef]
- Angelsen, A.; Wunder, S. Exploring the Forest-Poverty Link: Key Concepts, Issues and Research Implications; CIFOR Occasional Paper; Center for International Forestry Research: Bogot, Indonesia, 2003. [Google Scholar]
- Babulo, B.; Muys, B.; Nega, F.; Tollens, E.; Nyssen, J.; Deckers, J.; Mathijs, E. The economic contribution of forest resource use to rural livelihoods in Tigray, Northern Ethiopia. For. Policy Econ. 2009, 11, 109–117. [Google Scholar] [CrossRef]
- Nielsen, M.R.; Pouliot, M.; Meilby, H.; Smith-Hall, C.; Angelsen, A. Global patterns and determinants of the economic importance of bushmeat. Boil. Conserv. 2017, 215, 277–287. [Google Scholar] [CrossRef]
- Tieguhong, J. Supplies of bushmeat for livelihoods in logging towns in the Congo Basin. J. Hortic. For. 2009, 1, 65–80. [Google Scholar]
- Kümpel, N.F.; Milner-Gulland, E.J.; Cowlishaw, G.; Rowcliffe, J.M. Incentives for Hunting: The Role of Bushmeat in the Household Economy in Rural Equatorial Guinea. Hum. Ecol. 2010, 38, 251–264. [Google Scholar] [CrossRef]
- Sarti, F.M.; Adams, C.; Morsello, C.; Van Vliet, N.; Schor, T.; Yagüe, B.; Tellez, L.; Quiceno-Mesa, M.P.; Cruz, D. Beyond protein intake: Bushmeat as source of micronutrients in the Amazon. Ecol. Soc. 2015, 20. [Google Scholar] [CrossRef]
- Rowland, D.; Ickowitz, A.M.Y.; Powell, B.; Nasi, R.; Sunderland, T. Forest foods and healthy diets: Quantifying the contributions. Environ. Conserv. 2017, 44, 102–114. [Google Scholar] [CrossRef]
- Fa, J.E.; Currie, D.; Meeuwig, J. Bushmeat and food security in the Congo Basin: Linkages between wildlife and people’s future. Environ. Conserv. 2003, 30, 71–78. [Google Scholar] [CrossRef]
- Golden, C.D.; Fernald, L.C.H.; Brashares, J.S.; Rasolofoniaina, B.J.R.; Kremen, C.; Rasolofoniaina, B.J.R. Benefits of wildlife consumption to child nutrition in a biodiversity hotspot. Proc. Natl. Acad. Sci. USA 2011, 108, 19653–19656. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.G.; Bennett, E.L. (Eds.) Hunting for Sustainability in Tropical Forests; Columbia University Press: New York, NY, USA, 2000. [Google Scholar]
- Milner-Gulland, E.J.; Bennett, E.L. Wild Meat: The Bigger Picture. Trends Ecol. Evol. 2003, 18, 351–357. [Google Scholar] [CrossRef]
- Gray, T.N.E.; Hughes, A.C.; Laurance, W.F.; Long, B.; Lynam, A.J.; O’Kelly, H.; Ripple, W.J.; Seng, T.; Scotson, L.; Wilkinson, N.M. The wildlife snaring crisis: An insidious and pervasive threat to biodiversity in Southeast Asia. Biodivers. Conserv. 2018, 27, 1031–1037. [Google Scholar] [CrossRef]
- Ripple, W.J.; Abernethy, K.; Betts, M.G.; Chapron, G.; Dirzo, R.; Galetti, M.; Levi, T.; Lindsey, P.A.; Macdonald, D.W.; Machovina, B.; et al. Bushmeat hunting and extinction risk to the world’s mammals. Soc. Open Sci. 2016, 3, 160498. [Google Scholar] [CrossRef]
- Redford, K.H. The Empty Forest. BioScience 1992, 42, 412–422. [Google Scholar] [CrossRef]
- Wilkie, D.S.; Bennett, E.L.; Peres, C.A.; Cunningham, A.A. The empty forest revisited. Ann. N. Y. Acad. Sci. 2011, 1223, 120–128. [Google Scholar] [CrossRef]
- Malhi, Y.; Gardner, T.A.; Goldsmith, G.R.; Silman, M.R.; Zelazowski, P. Tropical Forests in the Anthropocene. Annu. Resour. 2014, 39, 125–159. [Google Scholar] [CrossRef]
- Kurten, E.L. Cascading effects of contemporaneous defaunation on tropical forest communities. Boil. Conserv. 2013, 163, 22–32. [Google Scholar] [CrossRef]
- Dirzo, R.; Young, H.S.; Galetti, M.; Ceballos, G.; Isaac, N.J.B.; Collen, B. Defaunation in the Anthropocene. Science 2014, 345, 401–406. [Google Scholar] [CrossRef]
- Young, H.S.; McCauley, D.J.; Galetti, M.; Dirzo, R. Patterns, Causes, and Consequences of Anthropocene Defaunation. Annu. Ecol. Evol. Syst. 2016, 47, 333–358. [Google Scholar] [CrossRef]
- Dirzo, R.; Miranda, A. Altered Patterns of Herbivory and Diversity in the Forest Understory: A Case Study of the Possible Consequences of Contemporary Defaunation; Wiley: Hoboken, NJ, USA, 1991; pp. 273–287. [Google Scholar]
- Galetti, M.; Dirzo, R. Ecological and evolutionary consequences of living in a defaunated world. Boil. Conserv. 2013, 163, 1–6. [Google Scholar] [CrossRef]
- Beaune, D.; Fruth, B.; Bollache, L.; Hohmann, G.; Bretagnolle, F. Doom of the elephant-dependent trees in a Congo tropical forest. Ecol. Manag. 2013, 295, 109–117. [Google Scholar] [CrossRef]
- Caughlin, T.T.; Ferguson, J.M.; Lichstein, J.W.; Zuidema, P.A.; Bunyavejchewin, S.; Levey, D.J. Loss of animal seed dispersal increases extinction risk in a tropical tree species due to pervasive negative density dependence across life stages. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142095. [Google Scholar] [CrossRef]
- Terborgh, J.; Nuñez-Iturri, G.; Pitman, N.C.A.; Valverde, F.H.C.; Álvarez, P.; Swamy, V.; Pringle, E.G.; Paine, C.E.T. Tree recruitment in an empty forest. Ecology 2008, 89, 1757–1768. [Google Scholar] [CrossRef]
- Berzaghi, F.; Verbeeck, H.; Nielsen, M.R.; Doughty, C.E.; Bretagnolle, F.; Marchetti, M.; Scarascia-Mugnozza, G.; Scarascia-Mugnozza, G. Assessing the role of megafauna in tropical forest ecosystems and biogeochemical cycles - the potential of vegetation models. Ecography 2018, 41, 1934–1954. [Google Scholar] [CrossRef]
- Harrison, R.D.; Tan, S.; Plotkin, J.B.; Slik, F.; Detto, M.; Brenes, T.; Itoh, A.; Davies, S.J. Consequences of defaunation for a tropical tree community. Ecol. Lett. 2013, 16, 687–694. [Google Scholar] [CrossRef]
- Bunker, D.E.; Declerck, F.; Bradford, J.C.; Colwell, R.K.; Perfecto, I.; Phillips, O.L.; Sankaran, M.; Naeem, S. Species Loss and Aboveground Carbon Storage in a Tropical Forest. Science 2005, 310, 1029–1031. [Google Scholar] [CrossRef] [PubMed]
- Brodie, J.F.; Gibbs, H.K. Bushmeat Hunting As Climate Threat. Science 2009, 326, 364–365. [Google Scholar] [CrossRef] [PubMed]
- Wright, I.J.; Ackerly, D.D.; Bongers, F.; Harms, K.E.; Ibarra-Manriquez, G.; Martinez-Ramos, M.; Mazer, S.J.; Muller-Landau, H.C.; Paz, H.; Pitman, N.C.A.; et al. Relationships among ecologically important dimensions of plant trait variation in seven neotropical forests. Ann. Bot. 2007, 99, 1003–1015. [Google Scholar] [CrossRef]
- Queenborough, S.A.; Mazer, S.J.; Vamosi, S.M.; Garwood, N.C.; Valencia, R.; Freckleton, R.P. Seed mass, abundance and breeding system among tropical forest species: Do dioecious species exhibit compensatory reproduction or abundances? J. Ecol. 2009, 97, 555–566. [Google Scholar] [CrossRef]
- Bello, C.; Galetti, M.; Pizo, M.A.; Magnago, L.F.S.; Rocha, M.F.; Lima, R.A.F.; Peres, C.A.; Ovaskainen, O.; Jordano, P. Defaunation affects carbon storage in tropical forests. Sci. Adv. 2015, 1, e1501105. [Google Scholar] [CrossRef] [PubMed]
- Peres, C.A.; Emilio, T.; Schietti, J.; Desmouliere, S.J.M.; Levi, T. Dispersal limitation induces long-term biomass collapse in overhunted Amazonian forests. Proc. Natl. Acad. Sci. USA 2016, 113, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Osuri, A.M.; Ratnam, J.; Varma, V.; Alvarez-Loayza, P.; Astaiza, J.H.; Bradford, M.; Fletcher, C.; Ndoundou-Hockemba, M.; Jansen, P.A.; Kenfack, D.; et al. Contrasting effects of defaunation on aboveground carbon storage across the global tropics. Nat. Commun. 2016, 7, 11351. [Google Scholar] [CrossRef]
- Harris, N.L.; Brown, S.; Hagen, S.C.; Saatchi, S.S.; Petrova, S.; Salas, W.; Hansen, M.C.; Potapov, P.V.; Lotsch, A. Baseline Map of Carbon Emissions from Deforestation in Tropical Regions. Science 2012, 336, 1573–1576. [Google Scholar] [CrossRef]
- UNFCCC. Decision 11/CP.19 Modalities for National Forest Monitoring Systems; United Framework Convention on Climate Change: Warsaw, Poland, 2013; p. 43. [Google Scholar]
- Neha, J.; Edward, T.A.M.; Natalia, W.; Jorge, T.; Julian, M.R.; Andrea, E.; Murray, C.; Martin, R.J.; Rasmus, F. Mapping dynamics of deforestation and forest degradation in tropical forests using radar satellite data. Environ. Res. Lett. 2015, 10, 034014. [Google Scholar]
- Guariguata, M.R.; Heymell, V.; Sabogal, C.; Thompson, I.D.; Okabe, K.; Bahamondez, C.; Nasi, R. An Operational Framework for Defining and Monitoring Forest Degradation. Ecol. Soc. 2013, 18, 20. [Google Scholar]
- Panfil, S.N.; Harvey, C.A. REDD+ and Biodiversity Conservation: A Review of the Biodiversity Goals, Monitoring Methods, and Impacts of 80 REDD+ Projects. Conserv. Lett. 2015, 9, 143–150. [Google Scholar] [CrossRef]
- Blomley, T.; Lukumbuzya, K.; Brodnig, G. Participatory Forest Management and REDD+ in Tanzania; The World Bank: Washington, DC, USA, 2011; p. 32. [Google Scholar]
- UN-REDD. National Programme Final Report—Ecuador; Ministry of Environment of Ecuador: Quito, Ecuador, 2015; p. 95. [Google Scholar]
- MAE. Bosques para el Buen Vivir—Plan de Acción REDD+ Ecuador (2016–2025); Ministry of Environment of Ecuador: Quito, Ecuador, 2016; p. 223. [Google Scholar]
- UN-REDD. UN-REDD Colombia National Programme Document; Ministry of Environment and Sustainable Development: Bogotá, Colombia, 2015; p. 70.
- UN-REDD. National Programme Annual Report—Federal Republic of Nigeria; UN-REDD: Abuja, Nigeria, 2015; p. 39. [Google Scholar]
- Federal Republic of Nigeria. REDD+ Readiness Preparation Proposal (R-PP); Federal Ministry of Environment: Abuja, Nigeria, 2014; p. 170.
- UN-REDD. Community Based REDD+ Country Plan for Nigeria; UNREDD and The GEF Small Grants Programme: Abuja, Nigeria, 2015; p. 28. [Google Scholar]
- PlanVivo. Reducing Emissions from Deforestation and Forest Degradation in the Yaeda Valley, Northern Tanzania; Plan Vivo: Edinburgh, UK, 2015; p. 49. [Google Scholar]
- Joshi L, J.; van Noordwijk, M.; Pradhan, U. Investment in Carbon Stocks in the Eastern Buffer Zone of Lamandau River Wildlife Reserve, Central Kalimantan Province, Indonesia: A REDD+ Feasibility Study; World Agroforestry Center (ICRAF) Southeast Asia Regional Office: Bogor, Indonesia, 2010; p. 105. [Google Scholar]
- van Vliet, N.; Quiceno Mesa, M.P.; Cruz-Antia, D.; Neves De Aquino, L.J.; Moreno, J.; Nasi, R. The uncovered volumes of bushmeat commercialized in the Amazonian trifrontier between Colombia, Peru & Brazil. Ethnobiol. Conserv. 2014, 2014, 3. [Google Scholar]
- Nasi, R.; Taber, A.; Van Vliet, N. Empty forests, empty stomachs? Bushmeat and livelihoods in the Congo and Amazon Basins. Int. For. 2011, 13, 355–368. [Google Scholar] [CrossRef]
- Culot, L.; Bello, C.; Batista, J.L.F.; Couto, H.T.Z.D.; Galetti, M.; Couto, H.T.Z. Synergistic effects of seed disperser and predator loss on recruitment success and long-term consequences for carbon stocks in tropical rainforests. Sci. Rep. 2017, 7, 7662. [Google Scholar] [CrossRef]
- Effiom, E.O.; Nuñez-Iturri, G.; Smith, H.G.; Ottosson, U.; Olsson, O. Bushmeat hunting changes regeneration of African rainforests. Proc. Soc. B Boil. Sci. 2013, 280, 20130246. [Google Scholar] [CrossRef]
- Phelps, J.; Friess, D.A.; Webb, E.L. Win-win REDD+ approaches belie carbon-biodiversity trade-offs. Biol. Conserv. 2012, 154, 53–60. [Google Scholar] [CrossRef]
- den Besten, J.W.; Arts, B.; Verkooijen, P. The evolution of REDD+: An analysis of discursive-institutional dynamics. Environ. Sci. Policy 2014, 35, 40–48. [Google Scholar] [CrossRef]
- Gallemore, C. Transaction costs in the evolution of transnational polycentric governance. Int. Environ. Agreem. Politics Law Econ. 2017, 17, 639–654. [Google Scholar] [CrossRef]
- Marrocoli, S.; Nielsen, M.R.; Morgan, D.; Van Loon, T.; Kulik, L.; Kühl, H. Using wildlife indicators to facilitate wildlife monitoring in hunter-self monitoring schemes. Ecol. Indic. in press.
- Precious Woods. Résumé Public du Plan d’Aménagement; Precious Woods: Libreville, Gabon, 2018; p. 31. [Google Scholar]
- Cawthorn, D.-M.; Hoffman, L.C. The bushmeat and food security nexus: A global account of the contributions, conundrums and ethical collisions. Food Res. Int. 2015, 76, 906–925. [Google Scholar] [CrossRef]
- Vieira, M.A.R.D.M.; Von Muhlen, E.M.; Shepard, G.H. Participatory Monitoring and Management of Subsistence Hunting in the Piagaçu-Purus Reserve, Brazil. Conserv. Soc. 2015, 13, 254. [Google Scholar] [CrossRef]
- Rist, J.; Cowlishaw, G.; Rowcliffe, M.; Milner-Gulland, E. Hunter Reporting of Catch per Unit Effort as a Monitoring Tool in a Bushmeat-Harvesting System. Conserv. Boil. 2010, 24, 489–499. [Google Scholar] [CrossRef]
- Meijaard, E.; Mengersen, K.; Buchori, D.; Nurcahyo, A.; Ancrenaz, M.; Wich, S.; Atmoko, S.S.U.; Tjiu, A.; Prasetyo, D.; Nardiyono; et al. Why Don’t We Ask? A Complementary Method for Assessing the Status of Great Apes. PLoS ONE 2011, 6, e18008. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, K.B.; Sténs, A.; Sandström, C.; Johansson, J.; Lidskog, R.; Ranius, T.; Roberge, J.M. The Swedish forestry model: More of everything? For. Policy Econ. 2017, 77, 44–55. [Google Scholar] [CrossRef]
- Chazdon, R.L.; Brancalion, P.H.S.; Laestadius, L.; Bennett-Curry, A.; Buckingham, K.; Kumar, C.; Moll-Rocek, J.; Vieira, I.C.G.; Wilson, S.J. When is a forest a forest? Forest concepts and definitions in the era of forest and landscape restoration. AMBIO 2016, 45, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Antunes, A.P.; Fewster, R.M.; Venticinque, E.M.; Peres, C.A.; Levi, T.; Rohe, F.; Shepard, G.H. Empty forest or empty rivers? A century of commercial hunting in Amazonia. Sci. Adv. 2016, 2, e1600936. [Google Scholar] [CrossRef] [PubMed]
- Stokstad, E. The empty forest. Science 2014, 345, 396–399. [Google Scholar] [CrossRef]
- Harrison, R.D. Emptying the Forest: Hunting and the Extirpation of Wildlife from Tropical Nature Reserves. BioScience 2011, 61, 919–924. [Google Scholar] [CrossRef]
- Huntingford, C.; Zelazowski, P.; Galbraith, D.; Mercado, L.M.; Sitch, S.; Fisher, R.; Lomas, M.; Walker, A.P.; Jones, C.D.; Booth, B.B.B.; et al. Simulated resilience of tropical rainforests to CO2-induced climate change. Nat. Geosci. 2013, 6, 268–273. [Google Scholar] [CrossRef]
- International Labour Organization (ILO). C 169 Indigenous and Tribal Peoples Convention, 1989; ILO: Geneva, Switzerland, 1989. [Google Scholar]
- UNDRIP. United Nations Declaration on the Rights of Indigenous Peoples; United Nations: New York, NY, USA, 2008. [Google Scholar]
- Rakotomamonjy, S.N.; Jones, J.P.G.; Razafimanahaka, J.H.; Ramamonjisoa, B.; Williams, S.J. The effects of environmental education on children’s and parents’ knowledge and attitudes towards lemurs in rural Madagascar. Anim. Conserv. 2015, 18, 157–166. [Google Scholar] [CrossRef]
- Duporge, I.; Hodgetts, T.; Brett, M. What spatially explicit quantitative evidence exists that shows the effect of land tenure on illegal hunting of endangered terrestrial mammals in sub-Saharan Africa? A systematic map protocol. Environ. Évid. 2018, 7, 27. [Google Scholar] [CrossRef]
- van Vliet, N. “Bushmeat Crisis” and “Cultural Imperialism” in Wildlife Management? Taking Value Orientations Into Account for a More Sustainable and Culturally Acceptable Wildmeat Sector. Front. Ecol. Evol. 2018, 6, 112. [Google Scholar] [CrossRef]
- Mockrin, M.H.; Redford, K.H. Potential for spatial management of hunted mammal populations in tropical forests. Conserv. Lett. 2011, 4, 255–263. [Google Scholar] [CrossRef]
- Wright, J.H.; Hill, N.A.; Roe, D.; Rowcliffe, J.M.; Kumpel, N.F.; Day, M.; Booker, F.; Milner-Gulland, E.J. Reframing the concept of alternative livelihoods. Conserv. Biol. 2016, 30, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Wicander, S.; Coad, L. Can the Provision of Alternative Livelihoods Reduce the Impact of Wild Meat Hunting in West and Central Africa? Conserv. Soc. 2018, 16, 441. [Google Scholar] [CrossRef]
| Document Reference | Text Excerpts Referring to Forests and/or Biodiversity | Keywords Mentioned | ||
|---|---|---|---|---|
| Biodiversity | Forest Fauna | Hunting | ||
| United Nations Framework Convention on Climate Change (UNFCCC; 1992), Article 4, 1d | Promote sustainable management, and promote and cooperate in the conservation and enhancement, as appropriate, of sinks and reservoirs of all greenhouse gases not controlled by the Montreal Protocol, including biomass, forests and oceans as well as other terrestrial, coastal and marine ecosystems; | No | No | No |
| UNFCCC/CP/2007/6/Add.1-COP 13 Bali | Decision 2/CP.13 “Recognizing that reducing emissions from deforestation and forest degradation in developing countries can promote co-benefits and may complement the aims and objectives of other relevant international conventions and agreements,” | No 1 | No | No |
| UNFCCC/CP/2009/11/Add.1-COP 15 Copenhagen | Decision 4/CP.15 “Recognizing the importance of promoting sustainable management of forests and co-benefits, including biodiversity, that may complement the aims and objectives of national forest programmes and relevant international conventions and agreements,” | Yes | No | No |
| UNFCCC/CP/2010/7/Add.1 (Cancun Safeguards); Decision 1/CP.16, Appendix 1, paragraph 2 | Appendix 1, 2(e) “Following safeguards should be promoted and supported ‘That actions are consistent with the conservation of natural forests and biological diversity, ensuring that the actions referred to in paragraph 70 of this decision are not used for the conversion of natural forests, but are instead used to incentivize the protection and conservation of natural forests and their ecosystem services, and to enhance other social and environmental benefits (Taking into account the need for sustainable livelihoods of indigenous peoples and local communities and their interdependence on forests in most countries)’” | Yes | No | No |
| UNFCCC/CP/2013/10/Add.1-COP 19 Warsaw | Decision 9/CP.19 – “22. Recognizes the importance of incentivizing non-carbon benefits for the long-term sustainability of the implementation of the activities referred to in decision 1/CP.16, paragraph 70, and noting the work on methodological issues referred to in decision 1/CP.18, paragraph 40;” | No | No | No |
| UNFCCC/CP/2015/10/Add.3-COP 21 Paris | Decision 18/CP. 21 – “Decides that methodological issues related to non-carbon benefits resulting from the implementation of the activities referred to in decision 1/CP.16, paragraph 70, do not constitute a requirement for developing country Parties seeking to receive support for the implementation of the actions and activities referred to in decision 1/CP.16 or results-based payments pursuant to decision 9/CP.19” | No | No | No |
| FCP 2 Carbon Fund Methodological Framework 2016 | “Non-Carbon Benefits: any benefits produced by or in relation to the implementation and operation of the ER Program, other than ERs and Monetary and Non-Monetary Benefits, as specified in the ER Program Document, and, as relevant, any Safeguards Plans. Such Non-Carbon Benefits may include, but not be limited to, the improvement of local livelihoods, building of transparent and effective forest governance structures, making progress on securing land tenure, and enhancing or maintaining biodiversity and/or other ecosystem services.” | Yes | No | No |
| Climate, Community & Biodiversity Standards: v3.1 3 (2017) | Well-designed projects also contribute to biodiversity conservation by restoring and protecting the world’s natural ecosystems, saving threatened animal and plant species from extinction and maintaining resilient and productive natural life-support for humankind. Through effective planning and implementation, all of these positive outcomes can be achieved cost-effectively. | Yes | No | No |
| Plan Vivo Standard (2013) 4 | Project interventions may include any improved land management activities that can generate demonstrable ecosystem service benefits, improve the livelihoods of participants and maintain or enhance biodiversity. The protection of, or provision of, biodiversity is often characterized as one of a variety of ecosystem services. Maintenance of biodiversity is arguably better understood as an overarching requirement for all ecosystem services in that biodiversity is a prerequisite for healthy ecosystems which provide a range of ecosystem services. Because of this, regardless of the focus of the PES scheme, Plan Vivo project interventions must be designed in a way that maintains or enhances biodiversity. | Yes | No | No |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krause, T.; Nielsen, M.R. Not Seeing the Forest for the Trees: The Oversight of Defaunation in REDD+ and Global Forest Governance. Forests 2019, 10, 344. https://doi.org/10.3390/f10040344
Krause T, Nielsen MR. Not Seeing the Forest for the Trees: The Oversight of Defaunation in REDD+ and Global Forest Governance. Forests. 2019; 10(4):344. https://doi.org/10.3390/f10040344
Chicago/Turabian StyleKrause, Torsten, and Martin Reinhardt Nielsen. 2019. "Not Seeing the Forest for the Trees: The Oversight of Defaunation in REDD+ and Global Forest Governance" Forests 10, no. 4: 344. https://doi.org/10.3390/f10040344
APA StyleKrause, T., & Nielsen, M. R. (2019). Not Seeing the Forest for the Trees: The Oversight of Defaunation in REDD+ and Global Forest Governance. Forests, 10(4), 344. https://doi.org/10.3390/f10040344





