Effects of Bark Stripping on Timber Production and Structure of Norway Spruce Forests in Relation to Climatic Factors
Abstract
1. Introduction
2. Materials and Methods
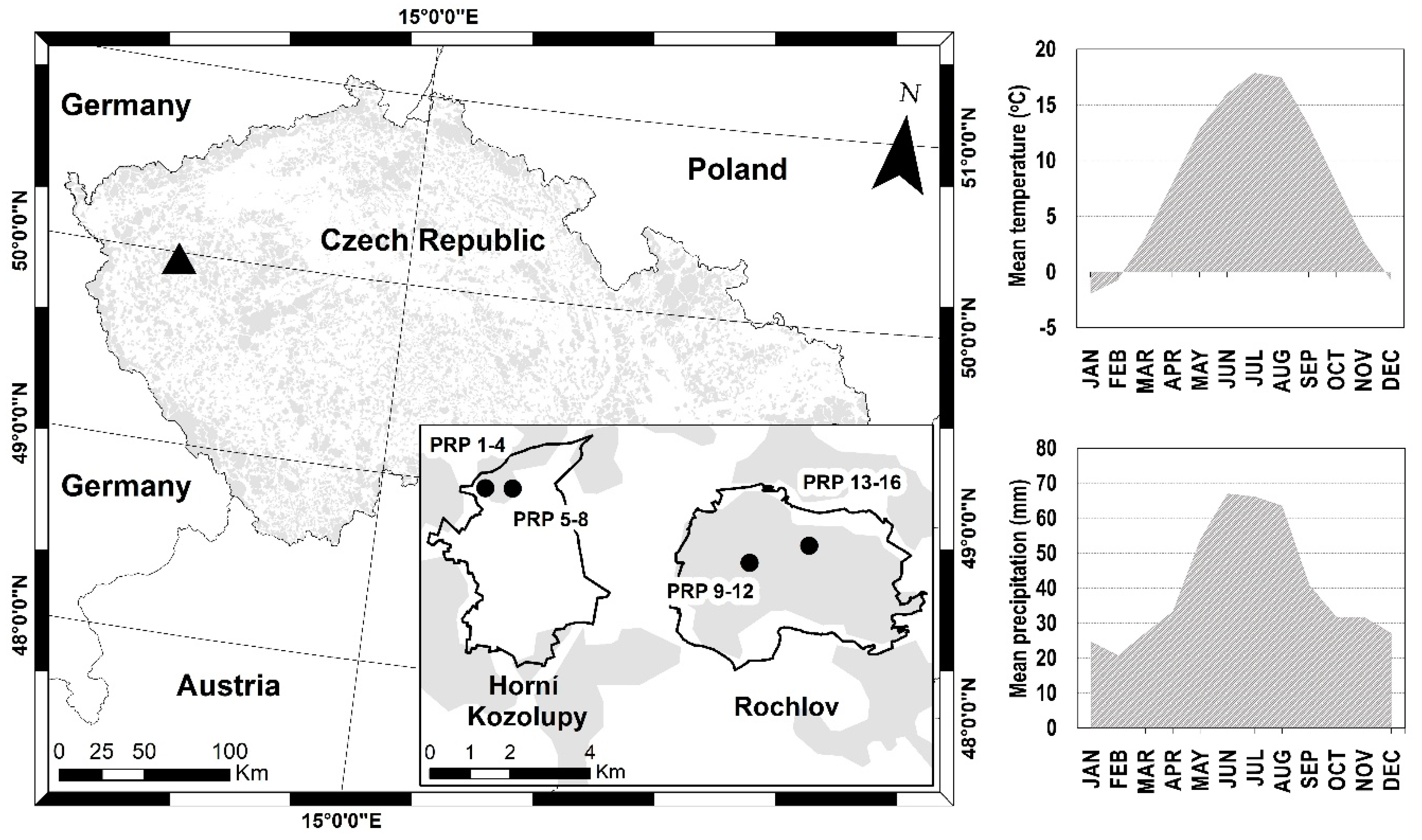
2.1. Study Area
2.2. Data Collection
2.3. Data Analyses
3. Results
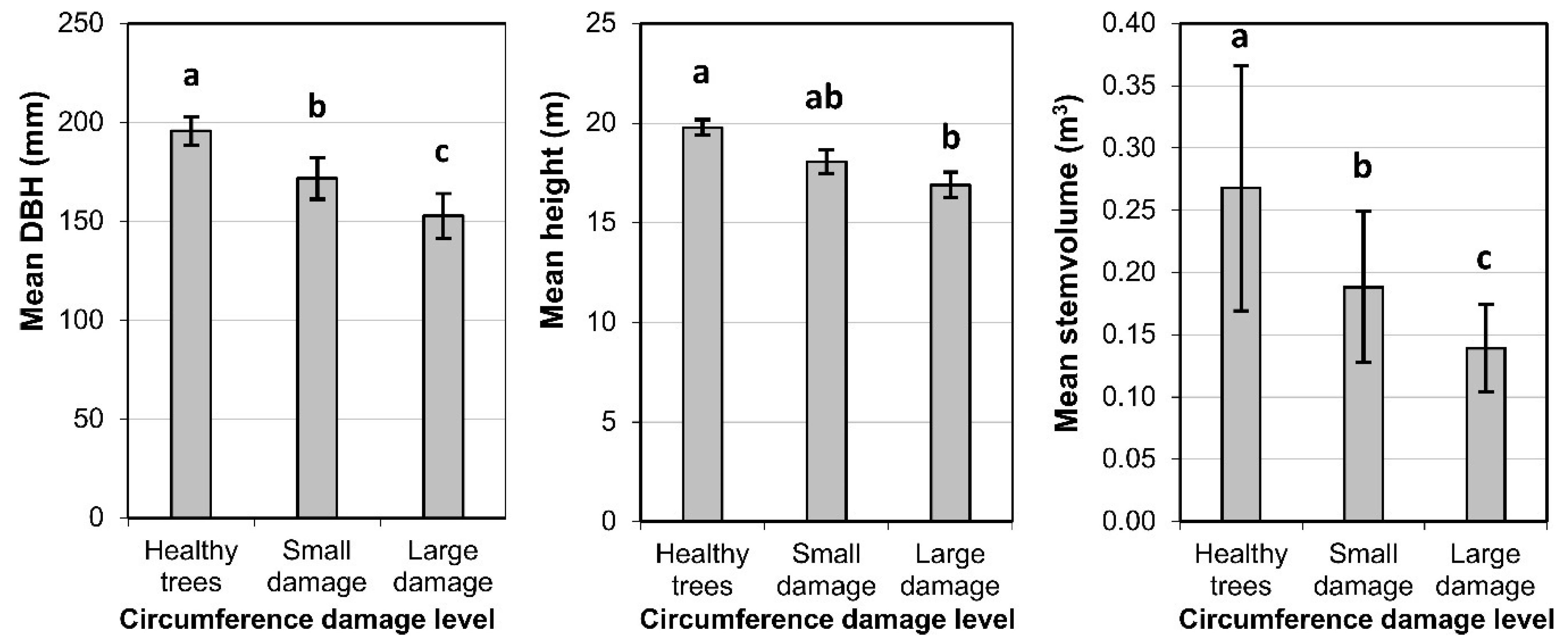
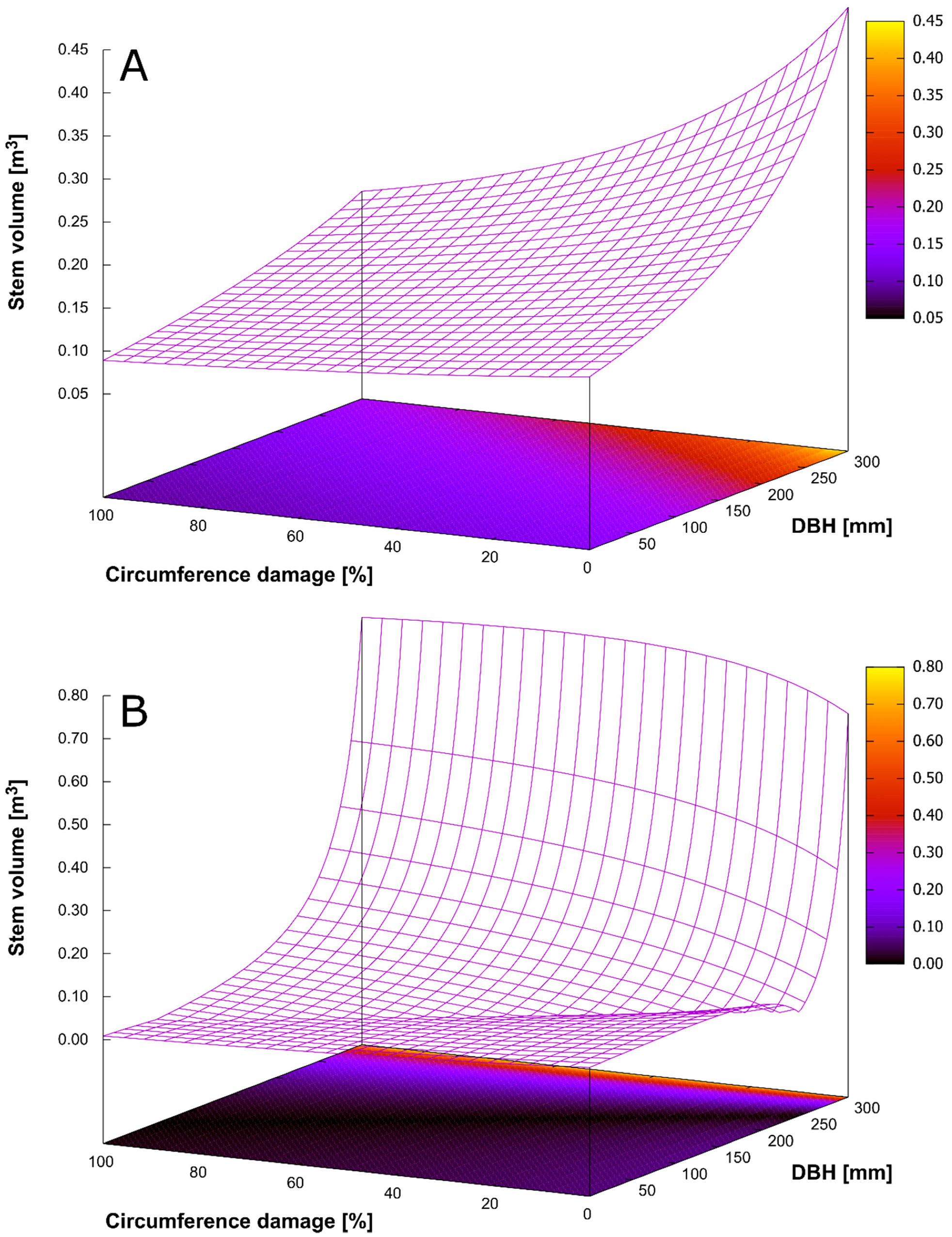
3.1. Production of Spruce Stands
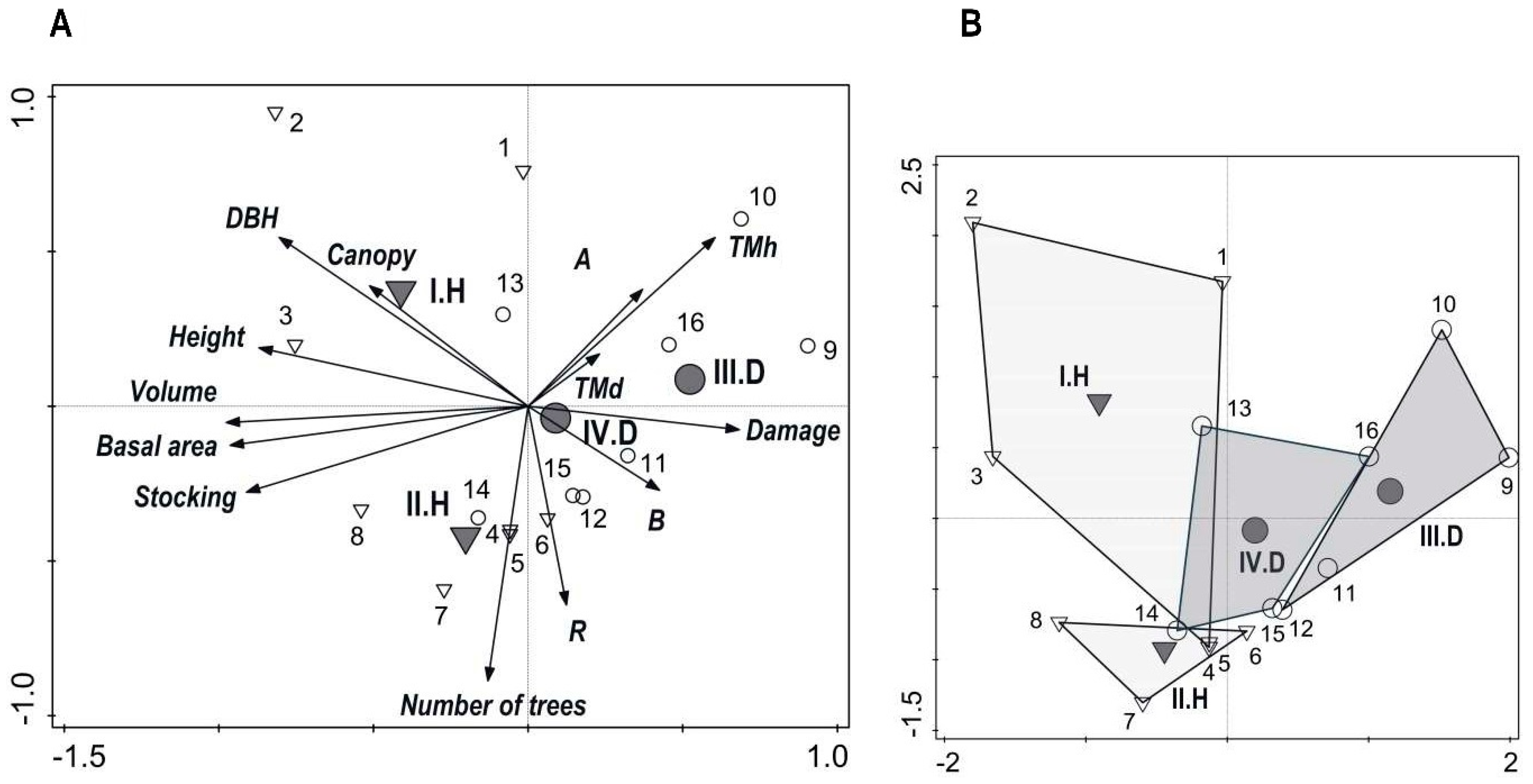
3.2. Stand Diversity and Structure
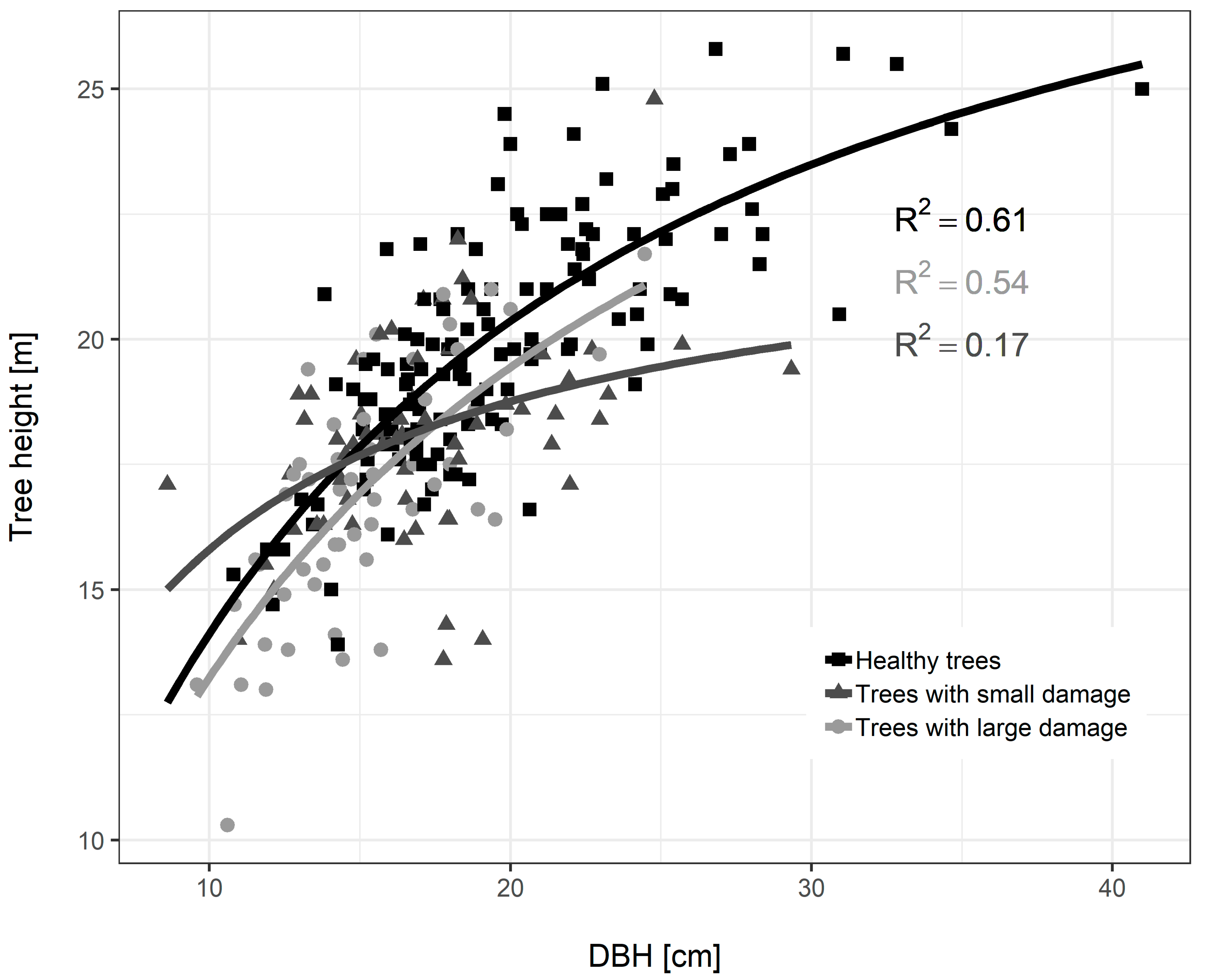
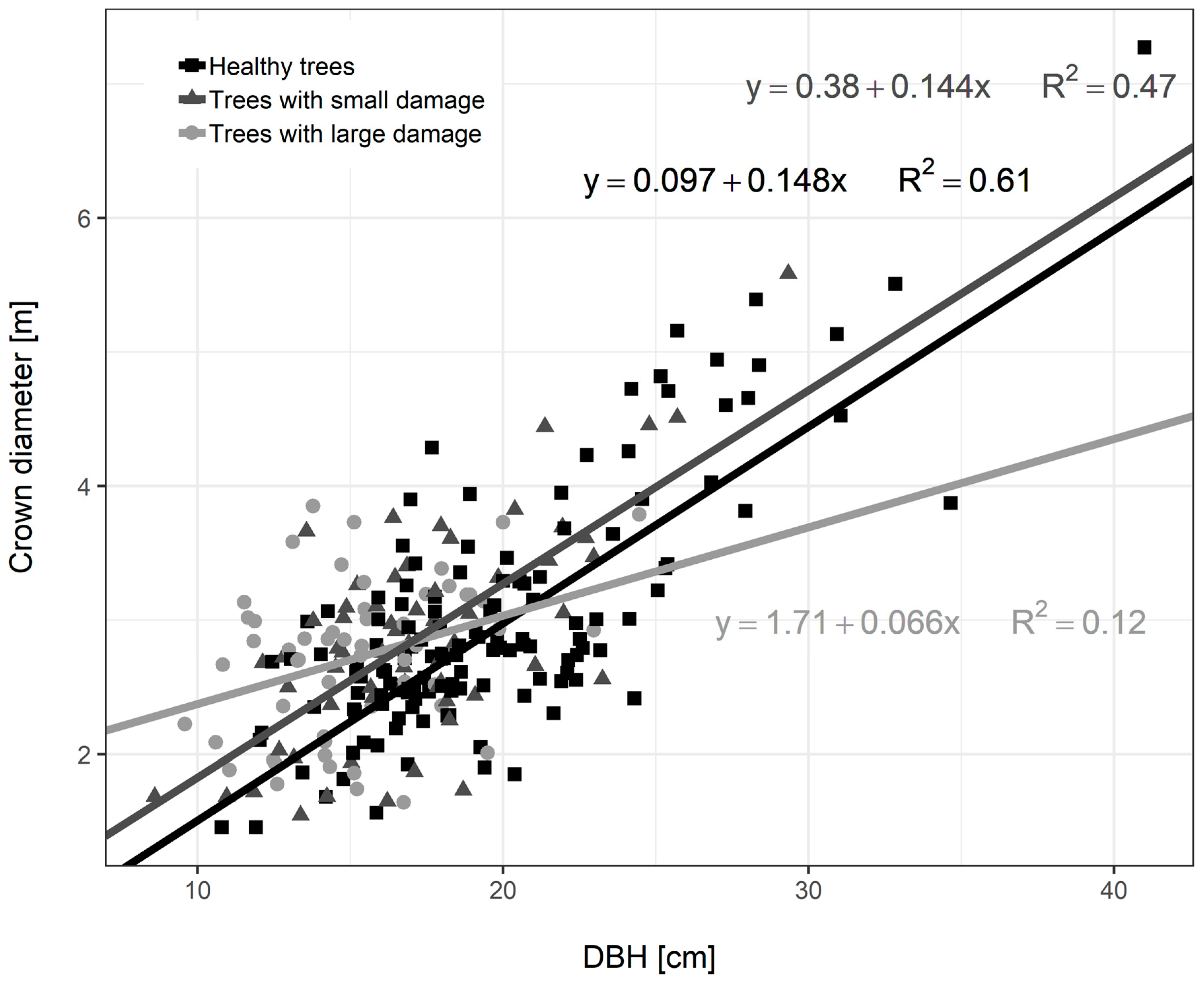
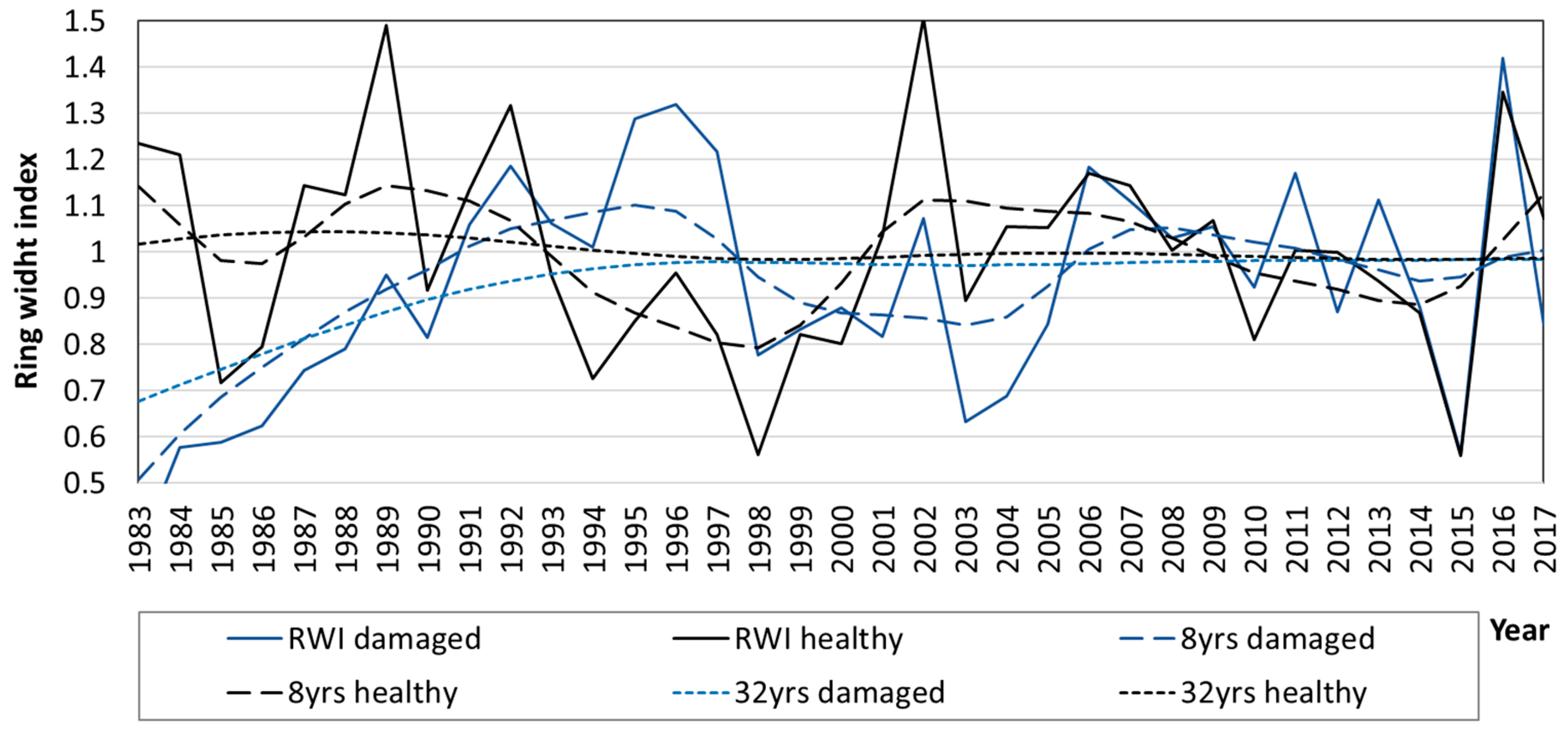
3.3. Diameter Growth of Norway Spruce
3.4. Bark Stripping Damage and Stem Rot
3.5. Relationships among Production, Diversity, and Game Damage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ellis, E.C.; Goldewijk, K.K.; Siebert, S.; Lightman, D.; Ramankutty, N. Anthropogenic transformation of the biomes, 1700 to 2000. Glob. Ecol. Biogeogr. 2010, 19, 589–606. [Google Scholar] [CrossRef]
- Kreidenweis, U.; Humpenöder, F.; Kehoe, L.; Kuemmerle, T.; Bodirsky, B.L.; Lotze-Campen, H.; Popp, A. Pasture intensification is insufficient to relieve pressure on conservation priority areas in open agricultural markets. Glob. Chang. Biol. 2018, 24, 3199–3213. [Google Scholar] [CrossRef]
- Malek, Ž.; Verburg, P.H.; R Geijzendorffer, I.; Bondeau, A.; Cramer, W. Global change effects on land management in the Mediterranean region. Glob. Environ. Chang. 2018, 50, 238–254. [Google Scholar] [CrossRef]
- Baltzinger, M.; Mårell, A.; Archaux, F.; Pérot, T.; Leterme, F.; Deconchat, M. Overabundant ungulates in French Sologne? Increasing red deer and wild boar pressure may not threaten woodland birds in mature forest stands. Basic Appl. Ecol. 2016, 17, 552–563. [Google Scholar] [CrossRef]
- Bleier, N.; Lehoczki, R.; Újváry, D.; Szemethy, L.; Csányi, S. Relationships between wild ungulates density and crop damage in Hungary. Acta Theriol. (Warsz.) 2012, 57, 351–359. [Google Scholar] [CrossRef]
- Fattebert, J.; Baubet, E.; Slotow, R.; Fischer, C. Landscape effects on wild boar home range size under contrasting harvest regimes in a human-dominated agro-ecosystem. Eur. J. Wildl. Res. 2017, 63, 32. [Google Scholar] [CrossRef]
- Hagen, R.; Heurich, M.; Kröschel, M.; Herdtfelder, M. Synchrony in hunting bags: Reaction on climatic and human induced changes? Sci. Total Environ. 2014, 468–469, 140–146. [Google Scholar] [CrossRef]
- Heurich, M.; Brand, T.T.G.; Kaandorp, M.Y.; Šustr, P.; Müller, J.; Reineking, B. Country, cover or protection: What shapes the distribution of red deer and roe deer in the Bohemian Forest Ecosystem? PLoS ONE 2015, 10, e0120960. [Google Scholar] [CrossRef] [PubMed]
- Thulin, C.G.; Malmsten, J.; Ericsson, G. Opportunities and challenges with growing wildlife populations and zoonotic diseases in Sweden. Eur. J. Wildl. Res. 2015, 61, 649–656. [Google Scholar] [CrossRef]
- Kuijper, D.P.J.; de Kleine, C.; Churski, M.; van Hooft, P.; Bubnicki, J.; Jedrzejewska, B. Landscape of fear in Europe: Wolves affect spatial patterns of ungulate browsing in Bialowieża Primeval Forest, Poland. Ecography 2013, 36, 1263–1275. [Google Scholar] [CrossRef]
- Biedrzycka, A.; Solarz, W.; Okarma, H. Hybridization between native and introduced species of deer in Eastern Europe. J. Mammal. 2012, 93, 1331–1341. [Google Scholar] [CrossRef]
- Côté, S.; Rooney, T.; Tremblay, J.-P.; Dussault, C.; Waller, D. Ecological impacts of deer overabundance. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 113–147. [Google Scholar] [CrossRef]
- Barančeková, M.; Krojerová-Prokešová, J.; Voloshina, I.V.; Myslenkov, A.I.; Kawata, Y.; Oshida, T.; Lamka, J.; Koubek, P. The origin and genetic variability of the Czech sika deer population. Ecol. Res. 2012, 27, 991–1003. [Google Scholar] [CrossRef]
- Bartoš, L. Sika Deer in Continental Europe. In Sika Deer; McCullough, D.R., Takatsuki, S., Kaji, K., Eds.; Springer: Tokyo, Japan, 2009; pp. 573–594. [Google Scholar]
- Pitra, C.; Rehbein, S.; Lutz, W. Tracing the genetic roots of the sika deer Cervus nippon naturalized in Germany and Austria. Eur. J. Wildl. Res. 2005, 51, 237–241. [Google Scholar] [CrossRef]
- Hannaford, J.; Pinn, E.H.; Diaz, A. The impact of sika deer grazing on the vegetation and infauna of Arne saltmarsh. Mar. Pollut. Bull. 2006, 53, 56–62. [Google Scholar] [CrossRef]
- Pérez-Espona, S.; Pemberton, J.M.; Putman, R. Red and sika deer in the British Isles, current management issues and management policy. Mamm. Biol. 2009, 74, 247–262. [Google Scholar] [CrossRef]
- Putman, R.J.; Moore, N.P. Impact of deer in lowland Britain on agriculture, forestry and conservation habitats. Mamm. Rev. 1998, 28, 141–164. [Google Scholar] [CrossRef]
- Bergvall, U.A.; Leimar, O. Directional associational plant defense from Red deer (Cervus elaphus) foraging decisions. Ecosphere 2017, 8, e01714. [Google Scholar] [CrossRef]
- Reimoser, F.; Putman, R.J. Impact of large ungulates on agriculture, forestry and conservation in Europe. In Ungulate Management in Europe: Problems and Practices; Putman, R.J., Apollonio, M., Andersen, R., Eds.; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Vospernik, S. Probability of bark stripping damage by red deer (Cervus elaphus) in Austria. Silva Fenn. 2006, 40, 589–601. [Google Scholar] [CrossRef][Green Version]
- Miller, G.R.; Kinnaird, J.W.; Cummins, R.P. Liability of Saplings to Browsing on a Red Deer Range in the Scottish Highlands. J. Appl. Ecol. 1982, 19, 941–951. [Google Scholar] [CrossRef]
- Kupferschmid, A.D.; Zimmermann, S.; Bugmann, H. Browsing regime and growth response of naturally regenerated Abies alba saplings along light gradients. For. Ecol. Manag. 2013, 310, 393–404. [Google Scholar] [CrossRef]
- Bobrowski, M.; Gillich, B.; Stolter, C. Modelling browsing of deer on beech and birch in northern Germany. For. Ecol. Manag. 2015, 358, 212–221. [Google Scholar] [CrossRef]
- Moser, B.; Schütz, M.; Hindenlang, K.E. Importance of alternative food resources for browsing by roe deer on deciduous trees: The role of food availability and species quality. For. Ecol. Manag. 2006, 226, 248–255. [Google Scholar] [CrossRef]
- Vandenberghe, C.; Freléchoux, F.; Buttler, A. The influence of competition from herbaceous vegetation and shade on simulated browsing tolerance of coniferous and deciduous saplings. Oikos 2008, 117, 415–423. [Google Scholar] [CrossRef]
- Gill, R.M.A. The influence of large herbivores on tree recruitment and forest dynamics. In Large Herbivore Ecology and Ecosystem Dynamics; Kjell, D., Bergström, D.P., Pastor, J., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 170–193. [Google Scholar]
- Faber, W.E. Bark stripping by moose on young pinus sylvestris in south–central Sweden. Scand. J. For. Res. 1996, 11, 300–306. [Google Scholar] [CrossRef]
- Gossow, H. Fütterungsstandort und Rotwild schäle. Österreichische Forstzeitung 1988, 6, 53–54. [Google Scholar]
- Borkowski, J.; Ukalski, K. Bark stripping by red deer in a post-disturbance area: The importance of security cover. For. Ecol. Manag. 2012, 263, 17–23. [Google Scholar] [CrossRef]
- Jerina, K.; Dajčman, M.; Adamič, M. Red deer (Cervus elaphus) bark stripping on spruce with regard to spatial distribution of supplemental feeding places. Zb. gozdarstva Lesar. 2008, 86, 33–43. [Google Scholar]
- Welch, D.; Staines, B.W.; Scott, D.; Catt, D.C. Bark-stripping damage by red deer in Sitka spruce in Western Scotland. II. Qound size and position. Forestry 1988, 61, 245–254. [Google Scholar] [CrossRef]
- Gill, R.M.A. A Review of Damage by Mammals in North Temperate Forests: 1. Deer. Forestry 1992, 65, 145–169. [Google Scholar] [CrossRef]
- Welch, D.; Scott, D. Observations on bark-stripping by red deer in a Picea sitchensis forest in Western Scotland over a 35-year period. Scand. J. For. Res. 2017, 32, 473–480. [Google Scholar] [CrossRef][Green Version]
- El Atta, H.A.; Hayes, A.J. Decay in Norway spruce caused by Stereum sanguinolentum Alb. & Schw. ex Fr. developing from extraction wounds. Forestry 1987, 60, 1–111. [Google Scholar]
- Månsson, J.; Jarnemo, A. Bark-stripping on Norway spruce by red deer in Sweden: Level of damage and relation to tree characteristics. Scand. J. For. Res. 2013, 28, 117–125. [Google Scholar] [CrossRef]
- Verheyden, H.; Ballon, P.; Bernard, V.; Saint-andrieux, C. Variations in bark-stripping by red deer Cervus elaphus across Europe. Mamm. Rev. 2006, 36, 217–234. [Google Scholar] [CrossRef]
- Vasiliauskas, R.; Stenlid, J.; Johansson, M. Fungi in bark peeling wounds of Picea abies in central Sweden. Eur. J. For. Pathol. 1996, 26, 285–296. [Google Scholar] [CrossRef]
- Akashi, N.; Terazawa, K. Bark stripping damage to conifer plantations in relation to the abundance of sika deer in Hokkaido, Japan. For. Ecol. Manag. 2005, 208, 77–83. [Google Scholar] [CrossRef]
- Hanzal, V.; Košinová, K.; Pokorný, R.; Janiszewski, P.; Hart, V. Weight parameters of body parts in sika deer (Cervus nippon nippon) from the Konstantinolázeňsko microregion, the Czech Republic. Cent. Eur. For. J. 2018, 64, 16–23. [Google Scholar] [CrossRef]
- Welch, D.; Scott, D. An estimate of timber degrade in Sitka spruce due to bark stripping by deer in a Scottish plantation. Forestry 2008, 81, 489–497. [Google Scholar] [CrossRef][Green Version]
- CZSO. Výsledek mysliveckého hospodaření [The summary of game management in the Czech Republic]; CZSO: Prague, Czech Republic, 2018.
- Loison, A.; Toïgo, C.; Gaillard, J.M. Large herbivore biodiversity in European mountain ecosystems: Current status and challenges for the future. In Alpine Biodiversity—Pattern, Process and Change in Europe; Nagy, L., Ed.; Springer: Berlin, Germany, 2003; pp. 351–366. [Google Scholar]
- Ward, A.I. Expanding ranges of wild and feral deer in Great Britain. Mamm. Rev. 2005, 35, 165–173. [Google Scholar] [CrossRef]
- Allombert, S.; Stockton, S.; Martin, J.L. A natural experiment on the impact of overabundant deer on forest invertebrates. Conserv. Biol. 2005, 19, 1917–1929. [Google Scholar] [CrossRef]
- Ammer, C. Impact of ungulates on structure and dynamics of natural regeneration of mixed mountain forests in the Bavarian Alps. For. Ecol. Manag. 1996, 88, 43–53. [Google Scholar] [CrossRef]
- Gill, R.M.A.; Beardall, V. The impact of deer on woodlands: The effects of browsing and seed dispersal on vegetation structure and composition. Forestry 2001, 74, 209–218. [Google Scholar] [CrossRef]
- Fuller, R.J.; Gill, R.M.A. Ecological impacts of increasing numbers of deer in British woodland. Forestry 2001, 74, 193–199. [Google Scholar] [CrossRef]
- CHMI Czech Hydrometeorological Institute. Available online: Portal.chmi.cz (accessed on 10 February 2019).
- Köppen, W. Das {Geographische} {System} der {Klimate}; Gebrüder Borntraeger: Berlin, Germany, 1936; Volume 1. [Google Scholar]
- Quitt, E. Klimatické oblasti Československa [Climatic Regions of Czechoslovakia].; Academia: Prague, Czech Republic, 1971. [Google Scholar]
- Institute of Forest Ecosystem Research—Monitoring and Mapping Solutions; Institute of Forest Ecosystem Research: Jílové u Prahy, Czech Republic, 2017.
- Černý, M.; Cienciala, E.; Russ, R. Metodika terénního šetření v systému inventarizace krajiny CzechTerra [Survey Methodology in the CzechTerra Landscape Inventory System]; IFER—Ústav pro výzkum lesních ekosystémů, s.r.o.: Prague, Czech Republic, 2009. [Google Scholar]
- Forest Management Institute. National Forest Inventory in the Czech Republic 2001–2004; FMI: Brandýs nad Labem, Czech Republic, 2007. [Google Scholar]
- Kosmala, M.; Suchocka, M. Influence of Mechanical Damage on the Condition of Trees; Annals of Warsaw University of Life Sciences—SGGW: Warsaw, Poland, 2008. [Google Scholar]
- Petráš, R.; Pajtík, J. Sústava česko-slovenských objemových tabuliek drevín. For. J. Lesn. časopis 1991, 37, 49–56. [Google Scholar]
- Reineke, L.H. Prefecting a stand-density index for evenaged forests. J. Agric. Res. 1933, 46, 627–638. [Google Scholar]
- Crookston, N.L.; Stage, A.R. Percent Canopy Cover and Stand Structure Statistics from the Forest Vegetation Simulator; US Department of Agriculture, Forest Service, Rocky Mountain Research Station: Ogden, UT, USA, 1999.
- Naslund, M. Skogsförsöksanstaltens gallringsförsök i tallskog; Swedish Institute of Experimental Forestry: Stockholm, Sweden, 1937; Volume 29.
- Fabrika, M. Growth simulator SIBYLA and the possibilities of its applications in forest management. Lesn. časopis For. J. 2003, 49. [Google Scholar]
- Pretzsch, H. Wissen nutzbar machen für das Management von Waldökosystemen. Allg. Forstzeitschrift/Der Wald 2006, 1158–1159. [Google Scholar]
- Füldner, K. Strukturbeschreibung in Mischbeständen [Structure description of mixed stands]. Forstarchiv 1995, 66, 235–606. [Google Scholar]
- Jaehne, S.; Dohrenbusch, A. Ein Verfahren zur beurteilung der bestandesdiversitiit. Eur. J. For. Res. 1997, 116, 333–345. [Google Scholar] [CrossRef]
- Clark, P.J.; Evans, F.C. Distance to Nearest Neighbor as a Measure of Spatial Relationships in Populations. Ecology 1954, 35, 445–453. [Google Scholar] [CrossRef]
- Pretzsch, H. Forest Dynamics, Growth and Yield; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Yamaguchi, D.K. A simple method for cross-dating increment cores from living trees. Can. J. For. Res. 1991, 21, 414–416. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Bunn, A.G. A dendrochronology program library in R (dplR). Dendrochronologia 2008, 26, 115–124. [Google Scholar] [CrossRef]
- Bunn, A.G. Statistical and visual crossdating in R using the dplR library. Dendrochronologia 2010, 28, 251–258. [Google Scholar] [CrossRef]
- Schweingruber, F.H.; Eckstein, D.; Serre-Bachet, F.; Bräker, O.U. Identification, presentation and interpretation of event years and pointer years in dendrochronology. Dendrochronologia 1990, 8, 9–38. [Google Scholar]
- Biondi, F.; Waikul, K. DENDROCLIM2002: A C++ program for statistical calibration of climate signals in tree-ring chronologies. Comput. Geosci. 2004, 30, 303–311. [Google Scholar] [CrossRef]
- Siegel, S.; Castellan, N.J., Jr. Nonparametric Statistics for the Behavioral Sciences, 2nd ed.; Mcgraw-Hill Book Company: New York, NY, USA, 1988; ISBN 0-07-057357-3. [Google Scholar]
- Lepš, J.; Šmilauer, P. Multivariate Analysis of Ecological Data Using CANOCO; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Völk, F.H. Bedeutung von Waldstruktur und Rotwildhege für die Schälhäufigkeit in den alpinen Bundesländern Österreichs. Z. Jagdwiss. 1999, 45, 1–16. [Google Scholar] [CrossRef]
- Girompaire, L.; Ballon, P. Conséquences de l’écorcage du cerf élaphe dans le massif des Vosges Alsaciennes. Rev. For. Française 1992, 6, 501–511. [Google Scholar] [CrossRef]
- Simon, J.; Kolář, C. Economic evaluation of bark stripping by red deer on the basis of analysis on a time growth series of spruce stands in the Hrubý Jeseník Mts. J. For. Sci. 2001, 47, 402–409. [Google Scholar]
- Eidmann, F.E. Wertminderung von Fichtenbestanden durch den schälschäden des Rotwildes. Forstarchiv 1952, 23, 65–69. [Google Scholar]
- Ueckermann, E. The causes of bark-stripping by cervids (Untersuchungen über die ursache des schälens des rotwildes). Z. Jagdwiss. 1956, 2. [Google Scholar]
- Pheiffer, J.; Hartfiel, B. Relation between winter feeding and the bark-stripping behaviour of red deer in the Eifel (Beziehungen zwischen der Winterfütterung und dem Schälverhalten des Rotwildes in der Eifel). Z. Jagdwiss. 1984, 30, 243–355. [Google Scholar]
- Shibata, E.; Torazawa, Y. Effects of bark stripping by sika deer, Cervus nippon, on wind damage to coniferous trees in subalpine forest of central Japan. J. For. Res. 2008, 13, 296–301. [Google Scholar] [CrossRef]
- Wolf, R. Historie chovu jelena siky na území České republiky. Introd. Spárkatá Zvěř 1999, 99, 52–56. [Google Scholar]
- Yokoyama, S.; Maeji, I.; Ueda, T.; Ando, M.; Shibata, E. Impact of bark stripping by sika deer, Cervus nippon, on subalpine coniferous forests in central Japan. For. Ecol. Manag. 2001, 140, 93–99. [Google Scholar] [CrossRef]
- Akashi, N.; Nakashizuka, T. Effects of bark-stripping by Sika deer (Cervus nippon) on population dynamics of a mixed forest in Japan. For. Ecol. Manag. 1999, 113, 75–82. [Google Scholar] [CrossRef]
- Vacek, Z.; Vacek, S.; Bílek, L.; Král, J.; Remeš, J.; Bulušek, D.; Králícek, I. Ungulate impact on natural regeneration in spruce-beech-fir stands in Černý důl nature reserve in the Orlické hory Mountains, case study from central sudetes. Forests 2014, 5, 2929–2946. [Google Scholar] [CrossRef]
- Gill, R.M.A. A review of damage by mammals in north temperate forest 3. Forestry 1992, 65, 363–388. [Google Scholar] [CrossRef]
- Vacek, Z. Structure and dynamics of spruce-beech-fir forests in Nature Reserves of the Orlické hory Mts. in relation to ungulate game. Cent. Eur. For. J. 2017, 63, 23–34. [Google Scholar] [CrossRef]
- Slanař, J.; Vacek, Z.; Vacek, S.; Bulušek, D.; Cukor, J.; Štefančík, I.; Bílek, L.; Krl, J. Long-term transformation of submontane spruce-beech forests in the Jizerské hory Mts.: Dynamics of natural regeneration. Cent. Eur. For. J. 2017, 63, 212–224. [Google Scholar] [CrossRef]
- Pulkkinen, P.; Pijykkij, T. Inherited narrow crown form, harvest index and stem biomass production in Norway spruce, Picea abies. Tree Physiol. 1990, 6, 381–391. [Google Scholar] [CrossRef]
- Lenz, O. Damage caused to trees by red deer. Schweiz. Z. Forstwes. 1964, 115, 13–14. [Google Scholar]
- Koltzenburg, C. Schutzwirkung durch mechanischbiologischen Schalschutz in Fichtenbestanden. Forst-Holzwirt 1985, 40, 471–475. [Google Scholar]
- Čermák, P.; Jankovský, L.; Lička, D.; Beránek, J.; Glogar, J. Damage to spruce stands by deer barking and subsequent rots in Forest Range Proklest, the Křtiny Training Forest Enterprise “Masaryk Forest” (the Drahany Upland). Acta Univ. Agric. Silvic. Mendel. Brun. 2004, 52, 165–174. [Google Scholar] [CrossRef]
- Büchsenmeister, R. The current situation as regards peeling damage in the forest: Austrian Forest Inventory 1986/90. Förderungsdienst 1995, 43, 312–320. [Google Scholar]
- Vasiliauskas, R.; Stenlid, J. Spread of Stereum sanguinolentum Vegetative Compatibility Groups within a Stand and within Stems of Picea abies. Silva Fenn. 1998, 32, 301–309. [Google Scholar] [CrossRef]
- Vasiliauskas, R. Patterns of wounding and decay in stems of Quercus robur due to bark peeling. Scand. J. For. Res. 1998, 13, 437–441. [Google Scholar] [CrossRef]
- Čermák, P.; Jankovský, L. Škody ohryzem, loupáním a následnými hnilobami; Lesnická práce: Kostelec nad Černými lesy, Czech Republic, 2006. [Google Scholar]
- Roll-Hansen, F.; Roll-Hansen, H. Microorganisms which invade Picea abies in seasonal stem wounds. For. Pathol. 1980, 10, 396–410. [Google Scholar] [CrossRef]
- Kallio, T.; Hallaksela, A.M. Biological-Control of Heterobasidion-Annosum (Fr) Bref (Fomes-annosus) in Finland. Eur. J. For. Pathol. 1979, 9, 298–308. [Google Scholar] [CrossRef]
- Welch, D.; Scott, D.; Staines, B. Bark stripping damage by red deer in Sitka spruce in Western Scotland. III. Trends in wound condition. Forestry 1997, 70, 113–120. [Google Scholar] [CrossRef]
- Jarnemo, A.; Minderman, J.; Bunnefeld, N.; Zidar, J.; Mansson, J. Managing landscapes for multiple objectives: Alternative forage can reduce the conflict between deer and forestry. Ecosphere 2014, 5. [Google Scholar] [CrossRef]
- Král, J.; Vacek, S.; Vacek, Z.; Putalová, T.; Bulušek, D.; Štefančík, I. Structure, development and health status of spruce forests affected by air pollution in the western Krkonoše Mts. in 1979–2014. For. J. 2015, 61, 175–187. [Google Scholar] [CrossRef]
- Králíček, I.; Vacek, Z.; Vacek, S.; Remeš, J.; Bulušek, D.; Král, J.; Štefančík, I.; Putalová, T. Dynamics and structure of mountain autochthonous spruce-beech forests: Impact of hilltop phenomenon, air pollutants and climate. Dendrobiology 2017, 77, 119–137. [Google Scholar] [CrossRef]
- Wilson, R.J.S.; Hopfmueller, M. Dendrochronological investigations of Norway spruce along an elevational transect in the Bavarian Forest, Germany. Dendrochronologia 2001, 19, 67–79. [Google Scholar]
- Savva, Y.; Oleksyn, J.; Reich, P.B.; Tjoelker, M.G.; Vaganov, E.A.; Modrzynski, J. Interannual growth response of Norway spruce to climate along an altitudinal gradient in the Tatra Mountains, Poland. Trees Struct. Funct. 2006, 20, 735–746. [Google Scholar] [CrossRef]
- Büntgen, U.; Frank, D.C.; Kaczka, R.J.; Verstege, A.; Zwijacz-Kozica, T.; Esper, J. Growth responses to climate in a multi-species tree-ring network in the {Western} {Carpathian} {Tatra} {Mountains}, {Poland} and {Slovakia}. Tree Physiol. 2007, 27, 689–702. [Google Scholar] [CrossRef]
- Meyer, F.D.; Bräker, O.U. Climate response in dominant and suppressed spruce trees, Picea abies (L.) karst., on a subalpine and lower montane site in Switzerland. Ecoscience 2001, 8, 105–114. [Google Scholar] [CrossRef]
- Vacek, S.; Hůnová, I.; Vacek, Z.; Hejcmanová, P.; Podrázský, V.; Král, J.; Putalová, T.; Moser, W.K. Effects of air pollution and climatic factors on Norway spruce forests in the Orlické hory Mts. (Czech Republic), 1979–2014. Eur. J. For. Res. 2015, 134, 1127–1142. [Google Scholar] [CrossRef]
- Andreassen, K.; Solberg, S.; Tveito, O.E.; Lystad, S.L. Regional differences in climatic responses of Norway spruce (Picea abies L. Karst) growth in Norway. For. Ecol. Manag. 2006, 222, 211–221. [Google Scholar] [CrossRef]
- Hauck, M.; Zimmermann, J.; Jacob, M.; Dulamsuren, C.; Bade, C.; Ahrends, B.; Leuschner, C. Rapid recovery of stem increment in Norway spruce at reduced SO2 levels in the Harz Mountains, Germany. Environ. Pollut. 2012, 164, 132–141. [Google Scholar] [CrossRef]
- Mäkinen, H.; Nöjd, P.; Kahle, H.P.; Neumann, U.; Tveite, B.; Mielikäinen, K.; Röhle, H.; Spiecker, H. Radial growth variation of Norway spruce (Picea abies (L.) Karst.) across latitudinal and altitudinal gradients in central and northern Europe. For. Ecol. Manag. 2002, 171, 243–259. [Google Scholar] [CrossRef]
- Putalová, T.; Vacek, Z.; Vacek, S.; Štefančík, I.; Bulušek, D.; Král, J. Tree-ring widths as an indicator of air pollution stress and climate conditions in different Norway spruce forest stands in the Krkonoše Mts. Cent. Eur. For. J. 2019, 65, 21–33. [Google Scholar]
- Kolář, T.; Čermák, P.; Oulehle, F.; Trnka, M.; Štěpánek, P.; Cudlín, P.; Hruška, J.; Büntgen, U.; Rybníček, M. Pollution control enhanced spruce growth in the “Black Triangle” near the Czech-Polish border. Sci. Total Environ. 2015, 538, 703–711. [Google Scholar] [CrossRef]
- Vacek, Z.; Vacek, S.; Bulušek, D.; Podrázský, V.; Remeš, J.; Král, J.; Putalová, T. Effect of fungal pathogens and climatic factors on production, biodiversity and health status of ash mountain forests. Dendrobiology 2017, 77, 161–175. [Google Scholar] [CrossRef]
- Mäkinen, H.; Nöjd, P.; Mielikäinen, K. Climatic signal in annual growth variation in damaged and healthy stands of Norway spruce [Picea abies (L.) Karst.] in southern Finland. Trees Struct. Funct. 2001, 15, 177–185. [Google Scholar] [CrossRef]
- Bradshaw, R.H.W.; Cowling, S.A.; Sykes, M.T. The effects of climate change on the distribution and management of Picea abies in Southern Scandinavia. Can. J. For. Res. 2000, 30, 1992–1998. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Gigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Eastaugh, C.S.; Pötzelsberger, E.; Hasenauer, H. Assessing the impacts of climate change and nitrogen deposition on Norway spruce (Picea abies L. Karst) growth in Austria with BIOME-BGC. Tree Physiol. 2011, 31, 262–274. [Google Scholar] [CrossRef]
- Lindner, M.; Fitzgerald, J.B.; Zimmermann, N.E.; Reyer, C.; Delzon, S.; van der Maaten, E.; Schelhaas, M.J.; Lasch, P.; Eggers, J.; van der Maaten-Theunissen, M.; et al. Climate change and European forests: What do we know, what are the uncertainties, and what are the implications for forest management? J. Environ. Manag. 2014, 146, 69–83. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Schlyter, P.; Stjernquist, I.; Bärring, L.; Jönsson, A.M.; Nilsson, C. Assessment of the impacts of climate change and weather extremes on boreal forests in northern Europe, focusing on Norway spruce. Clim. Res. 2006, 31, 75–84. [Google Scholar] [CrossRef]
- Hlásny, T.; Barka, I.; Roessiger, J.; Kulla, L.; Trombik, J.; Sarvašová, Z.; Bucha, T.; Kovalčík, M.; Čihák, T. Conversion of Norway spruce forests in the face of climate change: A case study in Central Europe. Eur. J. For. Res. 2017, 136, 1013–1028. [Google Scholar] [CrossRef]
- Brázdil, R.; Stucki, P.; Szabó, P.; Řezníčková, L.; Dolák, L.; Dobrovolný, P.; Tolasz, R.; Kotyza, O.; Chromá, K.; Suchánková, S. Windstorms and forest disturbances in the Czech Lands: 1801–2015. Agric. For. Meteorol. 2018, 250–251, 47–63. [Google Scholar] [CrossRef]
- Trnka, M.; Brázdil, R.; Možný, M.; Štěpánek, P.; Dobrovolný, P.; Zahradníček, P.; Balek, J.; Semerádová, D.; Dumbrovský, M.; Hlavinka, P.; et al. Soil moisture trends in the Czech Republic between 1961 and 2012. Int. J. Climatol. 2015, 35, 3733–3747. [Google Scholar] [CrossRef]
- Hanewinkel, M.; Cullmann, D.A.; Schelhass, M.-J.; Nabuurs, G.-J.; Zimmermann, N.E. Climate change may cause severe loss in the economic value of European forest land. Nat. Clim. Chang. 2012, 3, 203–207. [Google Scholar] [CrossRef]
- Neuner, S.; Albrecht, A.; Cullmann, D.; Engels, F.; Griess, V.C.; Hahn, W.A.; Hanewinkel, M.; Härtl, F.; Kölling, C.; Staupendahl, K.; et al. Survival of Norway spruce remains higher in mixed stands under a dryer and warmer climate. Glob. Chang. Biol. 2015, 21, 935–946. [Google Scholar] [CrossRef]
| Criterion | Quantifiers | Label | Reference | Evaluation |
|---|---|---|---|---|
| Horizontal structure | Aggregation index | R | [64] | mean value R = 1; aggregation R < 1; regularity R > 1 |
| Vertical diversity | Arten-Profile index | A | [61] | range 0–1; balanced vertical structure A < 0.3; selection forest A > 0.9 |
| Structure differentiation | Diameter dif. | TM | [62] | range 0–1; low TM < 0.3; very high differentiation TM > 0.7 |
| Height dif. | TMh | |||
| Crown dif. | K | [63] | low K < 1; very high differentiation K > 3 | |
| Complex diversity | Stand diversity | B | [63] | monotonous structure B < 4; uneven structure B = 6–8; very diverse structure B > 9 |
| PRP | Age | DBH ± SD | Height ± SD | Stem Volume ± SD | Number of Trees | Basal Area | Stand Volume | HDR | MAI | CC | CPA | SDI |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| y | cm | m | m3 | Trees ha−1 | m2 ha–1 | m3 ha–1 | m3 ha–1 y−1 | % | ha | |||
| 1LD | 43 | 20.9 ± 3.9 | 20.30 ± 2.43 | 0.304 ± 0.154 | 1200 | 41.0 | 365 | 97.1 | 8.5 | 81.1 | 1.67 | 0.74 |
| 2LD | 43 | 26.3 ± 7.6. | 21.47 ± 2.02 | 0.492 ± 0.378 | 1100 | 59.2 | 541 | 81.6 | 12.6 | 93.9 | 2.80 | 0.98 |
| 3LD | 43 | 23.8 ± 4.0 | 23.56 ± 1.46 | 0.446 ± 0.184 | 1300 | 57.5 | 580 | 99.0 | 13.5 | 85.4 | 1.92 | 0.98 |
| 4LD | 43 | 16.8 ± 3.2 | 18.33 ± 1.35 | 0.179 ± 0.077 | 1900 | 42.1 | 341 | 109.1 | 7.9 | 84.6 | 1.87 | 0.84 |
| 5LD | 43 | 17.3 ± 2.7 | 19.22 ± 1.54 | 0.201 ± 0.085 | 1800 | 42.5 | 363 | 111.1 | 8.4 | 78.6 | 1.54 | 0.82 |
| 6LD | 43 | 17.6 ± 3.7 | 17.24 ± 2.15 | 0.186 ± 0.088 | 1700 | 41.1 | 316 | 98.0 | 7.3 | 78.9 | 1.56 | 0.81 |
| 7LD | 43 | 18.1 ± 3.2 | 19.97 ± 1.60 | 0.227 ± 0.095 | 1900 | 48.6 | 430 | 110.3 | 10.0 | 77.3 | 1.48 | 0.93 |
| 8LD | 43 | 19.4 ± 4.0 | 20.49 ± 2.03 | 0.267 ± 0.153 | 2200 | 64.7 | 588 | 105.6 | 13.7 | 82.3 | 1.73 | 0.99 |
| 9HD | 41 | 14.7 ± 2.7 | 15.76 ± 1.88 | 0.117 ± 0.048 | 1400 | 23.7 | 164 | 107.2 | 4.0 | 77.6 | 1.50 | 0.49 |
| 10HD | 41 | 17.1 ± 4.9 | 17.25 ± 3.16 | 0.185 ± 0.159 | 1300 | 29.8 | 240 | 100.9 | 5.9 | 80.0 | 1.62 | 0.58 |
| 11HD | 41 | 17.8 ± 3.1 | 17.40 ± 2.23 | 0.187 ± 0.080 | 1500 | 37.1 | 280 | 97.8 | 6.8 | 81.3 | 1.68 | 0.73 |
| 12HD | 41 | 17.2 ± 3.6 | 17.44 ± 2.28 | 0.180 ± 0.093 | 1800 | 41.9 | 323 | 101.4 | 7.9 | 80.6 | 1.64 | 0.81 |
| 13HD | 43 | 19.5 ± 5.0 | 17.92 ± 1.79 | 0.232 ± 0.146 | 1500 | 44.6 | 348 | 91.9 | 8.1 | 92.6 | 2.61 | 0.83 |
| 14HD | 43 | 18.0 ± 4.7 | 17.80 ± 1.67 | 0.200 ± 0.143 | 2000 | 51.0 | 400 | 98.9 | 9.3 | 91.5 | 2.46 | 0.97 |
| 15HD | 43 | 17.3 ± 3.7 | 19.09 ± 1.91 | 0.199 ± 0.110 | 1600 | 37.4 | 318 | 110.3 | 7.4 | 83.6 | 1.81 | 0.73 |
| 16HD | 43 | 18.3 ± 3.5 | 17.60 ± 2.61 | 0.205 ± 0.117 | 1200 | 31.4 | 246 | 96.2 | 5.7 | 78.4 | 1.53 | 0.60 |
| PRP | R | A | TMd | TMh | K | B |
|---|---|---|---|---|---|---|
| 1LD | 1.153 | 0.494 | 0.206 | 0.167 | 0.639 | 2.213 |
| 2LD | 1.023 | 0.450 | 0.171 | 0.063 | 0.632 | 2.065 |
| 3LD | 1.219 | 0.103 | 0.159 | 0.084 | 0.754 | 1.952 |
| 4LD | 1.173 | 0.213 | 0.202 | 0.071 | 0.622 | 2.811 |
| 5LD | 1.173 | 0.213 | 0.202 | 0.071 | 0.622 | 2.811 |
| 6LD | 1.173 | 0.213 | 0.202 | 0.071 | 0.622 | 2.811 |
| 7LD | 1.362 * | 0.311 | 0.160 | 0.075 | 0.543 | 2.019 |
| 8LD | 1.170 | 0.448 | 0.166 | 0.089 | 0.607 | 2.680 |
| 9HD | 1.143 | 0.554 | 0.167 | 0.127 | 0.791 | 2.413 |
| 10HD | 1.138 | 0.628 | 0.264 | 0.170 | 0.728 | 2.813 |
| 11HD | 1.233 | 0.627 | 0.108 | 0.107 | 0.697 | 3.041 |
| 12HD | 1.246 | 0.501 | 0.218 | 0.079 | 0.726 | 2.334 |
| 13HD | 1.187 | 0.369 | 0.222 | 0.098 | 0.812 | 1.976 |
| 14HD | 1.328 * | 0.374 | 0.225 | 0.085 | 1.002 | 2.074 |
| 15HD | 1.340 * | 0.218 | 0.186 | 0.100 | 0.776 | 2.201 |
| 16HD | 1.290 | 0.391 | 0.193 | 0.120 | 0.558 | 2.314 |
| Generalized Linear Model (Gamma Distribution) | Link Function: Inverse Value | |
|---|---|---|
| Explanatory Variable | Value | p-Value |
| Constant | 7.643 | <0.001 |
| DBH | −0.018 | <0.001 |
| Circumference damage | 0.036 | <0.001 |
| Null deviance | 92.0 | |
| Residual deviance | 32.6 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cukor, J.; Vacek, Z.; Linda, R.; Vacek, S.; Marada, P.; Šimůnek, V.; Havránek, F. Effects of Bark Stripping on Timber Production and Structure of Norway Spruce Forests in Relation to Climatic Factors. Forests 2019, 10, 320. https://doi.org/10.3390/f10040320
Cukor J, Vacek Z, Linda R, Vacek S, Marada P, Šimůnek V, Havránek F. Effects of Bark Stripping on Timber Production and Structure of Norway Spruce Forests in Relation to Climatic Factors. Forests. 2019; 10(4):320. https://doi.org/10.3390/f10040320
Chicago/Turabian StyleCukor, Jan, Zdeněk Vacek, Rostislav Linda, Stanislav Vacek, Petr Marada, Václav Šimůnek, and František Havránek. 2019. "Effects of Bark Stripping on Timber Production and Structure of Norway Spruce Forests in Relation to Climatic Factors" Forests 10, no. 4: 320. https://doi.org/10.3390/f10040320
APA StyleCukor, J., Vacek, Z., Linda, R., Vacek, S., Marada, P., Šimůnek, V., & Havránek, F. (2019). Effects of Bark Stripping on Timber Production and Structure of Norway Spruce Forests in Relation to Climatic Factors. Forests, 10(4), 320. https://doi.org/10.3390/f10040320






