Leaf Physiological Responses to Drought Stress and Community Assembly in an Asian Savanna
Abstract
1. Introduction
- How do leaf water relations differ among the four dominant deciduous dipterocarp species;
- How do the four species differ in their physiological responses to drought stress in response to a progressive dry-down; and
- Are there other adaptive traits such as leaf shedding or resprouting among the four species that mitigate the impacts of seasonal drought?
2. Materials and Methods
2.1. Study Site and Species
2.2. Experimental Design
2.2.1. Material Preparations
2.2.2. Water Relations
2.2.3. Water Potential and Drought Stress Performances in a Dry-Down Experiment
2.3. Data Analyses
3. Results
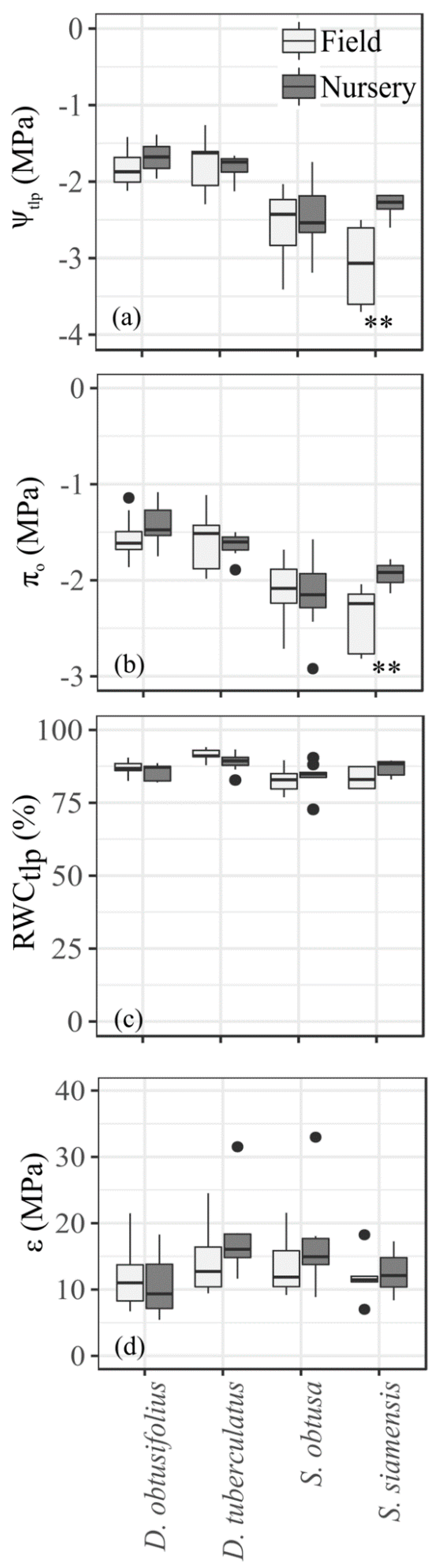
3.1. Leaf Water Relations
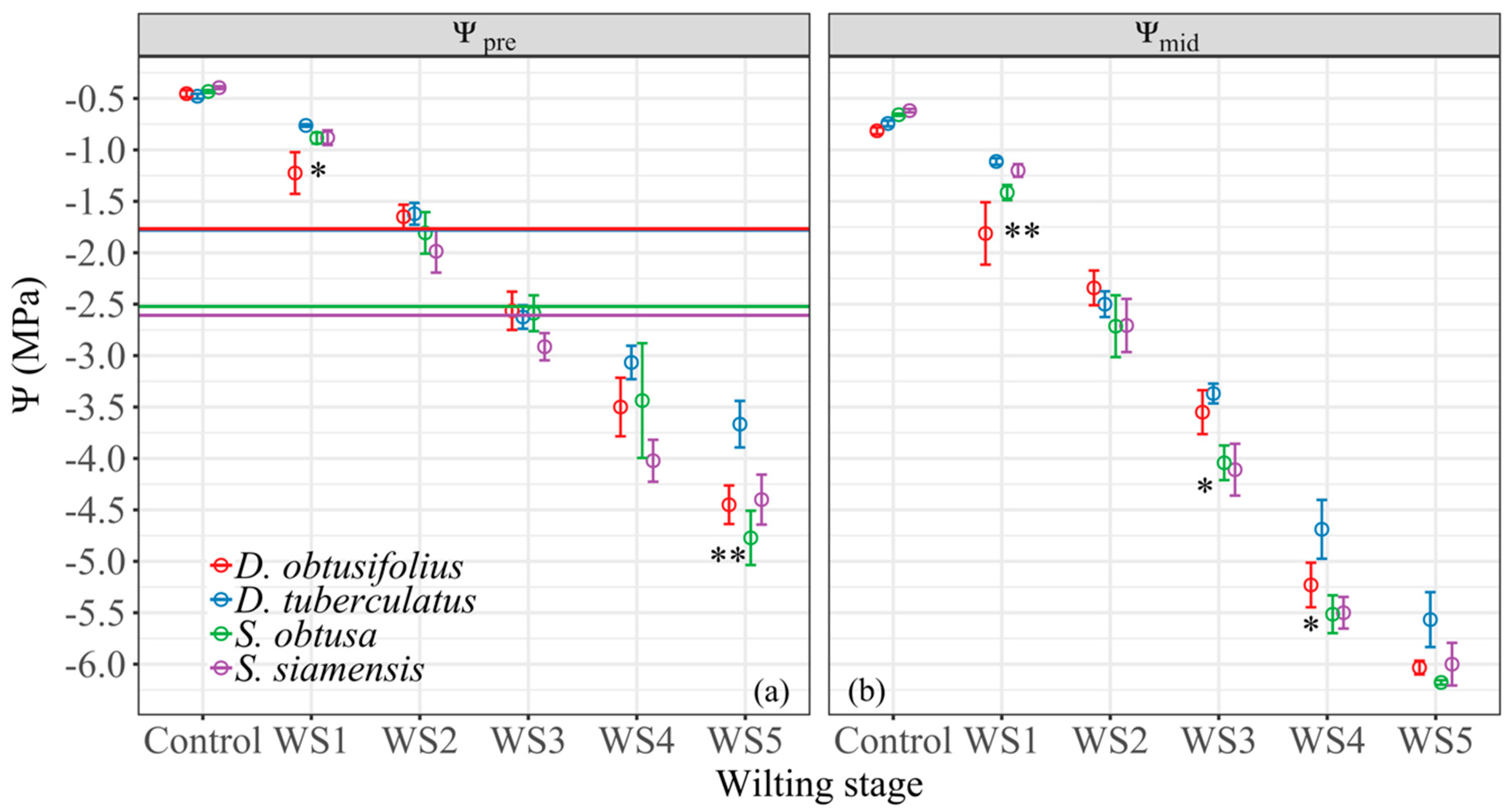
3.2. Water Potential at Wilting Stages
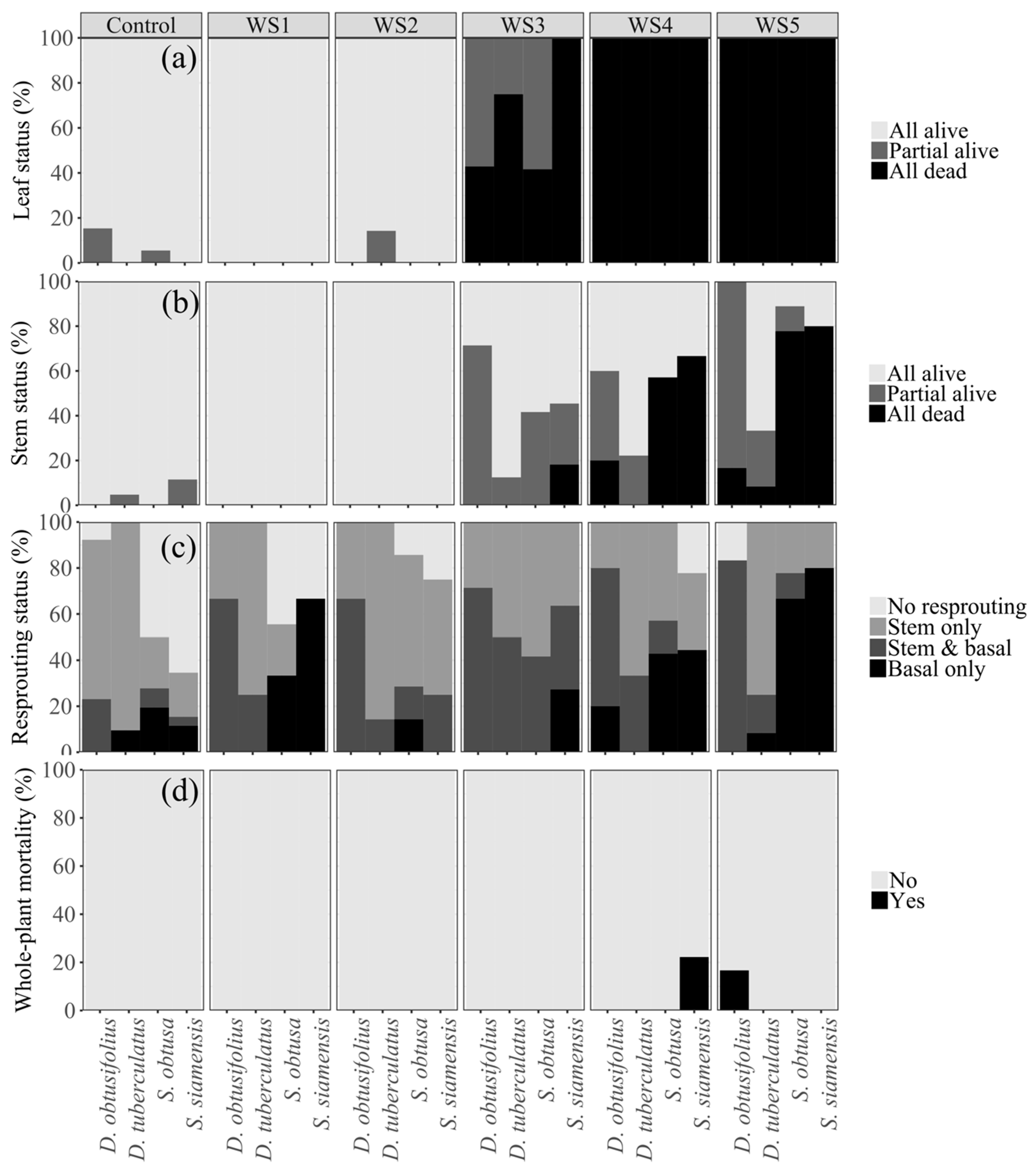
3.3. Leaf and Stem Responses following Drought Stress
3.4. Seedling Resprouting and Whole-Plant Mortality after Reirrigation
4. Discussion
4.1. Physiological Drought Tolerance Traits and Drought Stress Performances
4.2. Drought Adaptive Traits and Seedling Survival
4.3. Drought Stress and Community Assembly Patterns
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Condit, R.; Hubbell, S.P.; Foster, R.B. Mortality Rates of 205 Neotropical Tree and Shrub Species and the Impact of a Severe Drought. Ecol. Monogr. 1995, 65, 419–439. [Google Scholar] [CrossRef]
- Condit, R. Ecological Implications of Changes in Drought Patterns: Shifts in Forest Composition in Panama. Clim. Chang. 1998, 39, 413–427. [Google Scholar] [CrossRef]
- Delissio, L.J.; Primack, R.B. The impact of drought on the population dynamics of canopy-tree seedlings in an aseasonal Malaysian rain forest. J. Trop. Ecol. 2003, 19, 489–500. [Google Scholar] [CrossRef]
- Engelbrecht, B.M.J.; Kursar, T.A.; Tyree, M.T. Drought effects on seedling survival in a tropical moist forest. Trees 2005, 19, 312–321. [Google Scholar] [CrossRef]
- Phillips, O.L.; van der Heijden, G.; Lewis, S.L.; Lopez-Gonzalez, G.; Aragão, L.E.O.C.; Lloyd, J.; Malhi, Y.; Monteagudo, A.; Almeida, S.; Dávila, E.Á.; et al. Drought-mortality relationships for tropical forests. New Phytol. 2010, 187, 631–646. [Google Scholar] [CrossRef]
- Li, X.; Liu, F. Drought Stress Memory and Drought Stress Tolerance in Plants: Biochemical and Molecular Basis. In Drought Stress Tolerance in Plants; Hossain, M.A., Wani, S.H., Bhattacharjee, S., Burritt, D.J., Tran, L.-S.P., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 1, pp. 17–44. [Google Scholar]
- Baltzer, J.L.; Davies, S.J.; Bunyavejchewin, S.; Noor, N.S.M. The role of desiccation tolerance in determining tree species distributions along the Malay–Thai Peninsula. Funct. Ecol. 2008, 22, 221–231. [Google Scholar] [CrossRef]
- Jones, H.G.; Sutherland, R.A. Stomatal control of xylem embolism. Plant Cell Environ. 1991, 14, 607–612. [Google Scholar] [CrossRef]
- Garkoti, S.C.; Zobel, D.B.; Singh, S.P. Variation in drought response of sal (Shorea robusta) seedlings. Tree Physiol. 2003, 23, 1021–1030. [Google Scholar] [CrossRef]
- Cao, K.-F. Water relations and gas exchange of tropical saplings during a prolonged drought in a Bornean heath forest, with reference to root architecture. J. Trop. Ecol. 2000, 16, 101–116. [Google Scholar] [CrossRef]
- Reich, P.B.; Borchert, R. Water Stress and Tree Phenology in a Tropical Dry Forest in the Lowlands of Costa Rica. J. Ecol. 1984, 72, 61–74. [Google Scholar] [CrossRef]
- Engelbrecht, B.M.J.; Kursar, T.A. Comparative drought-resistance of seedlings of 28 species of co-occurring tropical woody plants. Oecologia 2003, 136, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Poorter, L.; Markesteijn, L. Seedling Traits Determine Drought Tolerance of Tropical Tree Species. Biotropica 2008, 40, 321–331. [Google Scholar] [CrossRef]
- Pausas, J.G.; Pratt, R.B.; Keeley, J.E.; Jacobsen, A.L.; Ramirez, A.R.; Vilagrosa, A.; Paula, S.; Kaneakua-Pia, I.N.; Davis, S.D. Towards understanding resprouting at the global scale. New Phytol. 2015, 209, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Blackman, C.J.; Creek, D.; Maier, C.; Aspinwall, M.J.; Drake, J.E.; Pfautsch, S.; O’Grady, A.; Delzon, S.; Medlyn, B.E.; Tissue, D.T.; et al. Drought response strategies and hydraulic traits contribute to mechanistic understanding of plant dry-down to hydraulic failure. Tree Physiol. 2019, 197, 1–15. [Google Scholar] [CrossRef]
- Brodribb, T.J.; Holbrook, N.M.; Edwards, E.J.; Gutiérrez, M.V. Relations between stomatal closure, leaf turgor and xylem vulnerability in eight tropical dry forest trees. Plant Cell Environ. 2003, 26, 443–450. [Google Scholar] [CrossRef]
- Hesla, B.I.; Tieszen, H.L.; Boutton, T.W. Seasonal water relations of savanna shrubs and grasses in Kenya, East Africa. J. Arid Environ. 1985, 8, 15–31. [Google Scholar] [CrossRef]
- Rundel, P.W. Dry forest ecosystems of Thailand. In Seasonally Dry Tropical Forests; Bullock, S.H., Mooney, H.A., Medina, E., Eds.; Island Press/Center for Resource Economics, Cambridge University Press: Cambridge, UK, 1995; pp. 93–123. [Google Scholar]
- Stott, P. The savanna forests of mainland southeast Asia: An ecological survey. Prog. Phys. Geog. 1984, 8, 315–335. [Google Scholar] [CrossRef]
- Champion, H.; Seth, S. A revised survey of the forest types of India; Gov. India Press: Delhi, India, 1968.
- Ashton, P.S. Toward a regional classification of the humid tropics of Asia. Tropics 1991, 1, 1–12. [Google Scholar] [CrossRef][Green Version]
- Ratnam, J.; Bond, W.J.; Fensham, R.J.; Hoffmann, W.A.; Archibald, S.; Lehmann, C.E.R.; Anderson, M.T.; Higgins, S.I.; Sankaran, M. When is a forest a savanna, and why does it matter? Global. Ecol. Biogeogr. 2011, 20, 653–660. [Google Scholar] [CrossRef]
- Ratnam, J.; Tomlinson, K.W.; Rasquinha, D.N.; Sankaran, M. Savannahs of Asia: Antiquity, biogeography, and an uncertain future. Phil. Trans. 2016, 371, 20150305. [Google Scholar] [CrossRef]
- Bunyavejchewin, S. Canopy structure of the dry dipterocarp forest of Thailand. Thai For. Bull. 1983, 14, 1–132. [Google Scholar]
- Dinh, D.Q. Contribution to Study on Natural Regeneration of the Deciduous Dipterocarp Forest in Easup, Dak Lak Province, Vietnam. Ph.D. Thesis, Forest Science Institute of Vietnam, Hanoi, Vietnam, 1993. [Google Scholar]
- Nguyen, T.T.; Murphy, B.P.; Baker, P.J. The existence of a fire-mediated tree-recruitment bottleneck in an Asian savanna. J. Biogeogr. 2019, 46, 745–756. [Google Scholar] [CrossRef]
- Ogawa, H.; Yoda, K.; Kira, T.; Ogino, K.; Shidel, T.; Ratanawongse, D.; Apasutaya, C. Comparative Ecological Studies on Three Main Types of Forest Vegetation in Thailand. I. Structure and Floristic Composition. Nat. Life Southeast Asia 1965, 4, 13–48. [Google Scholar]
- Kutintara, U. Structure of the dry Dipterocarp Forest. Ph.D. Thesis, Colorado State University, Fort Collins, CO, USA, 1975. [Google Scholar]
- Nguyen, T.T.; Baker, P.J. Structure and composition of deciduous dipterocarp forest in Central Vietnam: Patterns of species dominance and regeneration failure. Plant Ecol. Divers. 2016, 9, 589–601. [Google Scholar] [CrossRef]
- Tyree, M.T.; Engelbrecht, B.M.J.; Vargas, G.; Kursar, T.A. Desiccation Tolerance of Five Tropical Seedlings in Panama. Relationship to a Field Assessment of Drought Performance. Plant Physiol. 2003, 132, 1439–1447. [Google Scholar] [CrossRef]
- Bartlett, M.K.; Zhang, Y.; Yang, J.; Kreidler, N.; Sun, S.W.; Lin, L.; Hu, Y.H.; Cao, K.F.; Sack, L. Drought tolerance as a driver of tropical forest assembly: Resolving spatial signatures for multiple processes. Ecology 2016, 97, 503–514. [Google Scholar] [CrossRef]
- Ho, V.C. Study on Solutions for Biodiversity Conservation of YokDon National Park. Ph.D. Thesis, Vietnam Forestry University, Hanoi, Vietnam, 2008; pp. 1–216. [Google Scholar]
- Van Tan, P.; Thanh, N.D.; Van Hiep, N. A review of evidence of recent climate change in the Central Highlands of Vietnam. Coffee Clim. Enabling Eff. Response 2013, 1–49. [Google Scholar]
- Sankaran, M.; Ratnam, J. African and Asian Savannas. In Encyclopedia of Biodiversity; Elsevier: Amsterdam, The Netherlands, 2013; pp. 58–74. [Google Scholar]
- Parr, C.L.; Lehmann, C.E.R.; Bond, W.J.; Hoffmann, W.A.; Andersen, A.N. Tropical grassy biomes: Misunderstood, neglected, and under threat. Trends Ecol. Evol. 2014, 29, 205–213. [Google Scholar] [CrossRef]
- Tran, C.V. Study on Applicability of Mathematical Simulation for Structural and Dynamic Properties of Deciduous Dipterocarp Forest in the Central Highlands of Vietnam. Ph.D. Thesis, Forest Science Institute of Vietnam, Hanoi, Vietnam, 1991. [Google Scholar]
- Tyree, M.T.; Hammel, H.T. The Measurement of the Turgor Pressure and the Water Relations of Plants by the Pressure-bomb Technique. J. Exp. Bot. 1972, 23, 267–282. [Google Scholar] [CrossRef]
- Bartlett, M.K.; Scoffoni, C.; Sack, L. The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: A global meta-analysis. Ecol. Lett. 2012, 15, 393–405. [Google Scholar] [CrossRef]
- Turner, N.C. Measurement of plant water status by the pressure chamber technique. Irrig. Sci. 1998, 9, 289–308. [Google Scholar] [CrossRef]
- Schulte, P.J.; Hinckley, T.M. A comparison of pressure-volume curve data analysis techniques. J. Exp. Bot. 1985, 36, 1590–1602. [Google Scholar] [CrossRef]
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. 2018. Available online: https://www.R-project.org (accessed on 24 May 2018).
- Levitt, J.V. Responses of Plants to Environmental Stresses; Academic Press: New York, NY, USA, 1972. [Google Scholar]
- Fu, P.-L.; Jiang, Y.-J.; Wang, A.-Y.; Brodribb, T.J.; Zhang, J.-L.; Zhu, S.-D.; Cao, K.-F. Stem hydraulic traits and leaf water-stress tolerance are co-ordinated with the leaf phenology of angiosperm trees in an Asian tropical dry karst forest. Ann. Bot. 2012, 110, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Sobrado, M.A. Aspects of tissue water relations and seasonal changes of leaf water potential components of evergreen and deciduous species coexisting in tropical dry forests. Oecologia 1986, 68, 413–416. [Google Scholar] [CrossRef]
- Stott, P. Stability and stress in the savanna forests of mainland South-East Asia. J. Trop. For. Sci. 1990, 17, 373–383. [Google Scholar] [CrossRef]
- Shukla, A.; Mehrotra, R.C.; Guleria, J.S. Emergence and extinction of Dipterocarpaceae in western India with reference to climate change: Fossil wood evidences. J. Earth Syst. Sci. 2013, 122, 1373–1386. [Google Scholar] [CrossRef]
- Lenz, T.I.; Wright, I.J.; Westoby, M. Interrelations among pressure-volume curve traits across species and water availability gradients. Physiol. Plant. 2006, 127, 423–433. [Google Scholar] [CrossRef]
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant Physiological Ecology; Springer: Berlin/Heidelberg, Germany, 2008; pp. 1–624. [Google Scholar]
- Scoffoni, C.; Vuong, C.; Diep, S.; Cochard, H.; Sack, L. Leaf shrinkage with dehydration: Coordination with hydraulic vulnerability and drought tolerance. Plant Physiol. 2014, 164, 1772–1788. [Google Scholar] [CrossRef]
- Hoffmann, W.A.; Orthen, B.; Franco, A.C. Constraints to seedling success of savanna and forest trees across the savanna-forest boundary. Oecologia 2004, 140, 252–260. [Google Scholar] [CrossRef]
- Bunyavejchewin, S.; Baker, P.J.; Davies, S.J. Seasonally Dry Tropical Forests in Continental Southeast Asia: Structure, Composition, and Dynamics. In The Ecology and Conservation of Seasonally Dry Forests in Asia; McShea, W.J., Davies, S.J., Bhumpakphan, N., Eds.; Smithsonian Institution Scholarly Press: Washington, DC, USA, 2011; pp. 9–35. [Google Scholar]
- Schutz, A.E.N.; Bond, W.J.; Cramer, M.D. Juggling carbon: Allocation patterns of a dominant tree in a fire-prone savanna. Oecologia 2009, 160, 235–246. [Google Scholar] [CrossRef]
- Clarke, P.J.; Lawes, M.J.; Midgley, J.J.; Lamont, B.B.; Ojeda, F.; Burrows, G.E.; Enright, N.J.; Knox, K.J.E. Resprouting as a key functional trait: How buds, protection and resources drive persistence after fire. New Phytol. 2012, 197, 19–35. [Google Scholar] [CrossRef]
- Zeppel, M.J.B.; Harrison, S.P.; Adams, H.D.; Kelley, D.I.; Li, G.; Tissue, D.T.; Dawson, T.E.; Fensham, R.; Medlyn, B.E.; Palmer, A.; et al. Drought and resprouting plants. New Phytol. 2014, 206, 583–589. [Google Scholar] [CrossRef]
- Smith, M.G.; Arndt, S.K.; Miller, R.E.; Kasel, S.; Bennett, L.T. Trees use more non-structural carbohydrate reserves during epicormic than basal resprouting. Tree Physiol. 2018, 38, 1779–1791. [Google Scholar] [CrossRef]
- Smith, M.G.; Miller, R.E.; Arndt, S.K.; Kasel, S.; Bennett, L.T. Whole-tree distribution and temporal variation of non-structural carbohydrates in broadleaf evergreen trees. Tree Physiol. 2017, 31, 1–12. [Google Scholar] [CrossRef]
| Wilting Stage | Leaf Visual Symptoms |
|---|---|
| WS1 | Leaves look healthy with no signs of wilting |
| WS2 | Leaves become softer, slight changes to leaf angle |
| WS3 | Leaves soft, nearly parallel with stem, brighter color, slightly curved |
| WS4 | Slightly crunchy, curved, dried, necrotic tissue, brighter or brown |
| WS5 | Very dried, curved, brighter or brown |
| Status | Rating | Criteria |
|---|---|---|
| Leaf | All dead | All remaining leaves or the only remaining leaf from the measurements were dead |
| Partially alive | At least one of the remaining leaves from the measurements was alive | |
| All alive | All remaining leaves or the only remaining leaf from the measurements were alive | |
| Stem | All dead | All multiple or single stems were dead |
| Partially alive | At least one of the multiple stems was alive | |
| All alive | All the multiple stems or single stem were alive | |
| Resprouting | No resprouting | No new shoot/leaves emerged |
| Stem only | New shoots/leaves only emerged from the stems/seedling tops | |
| Stem and basal | New shoots/leaves emerged from both the base and stems/ seedling tops | |
| Basal only | New shoots only emerged from the base |
| Models Explaining the Above-Ground Mortality | df | AIC |
|---|---|---|
| Model1: D + H + D*H + Species + Wilting stage + No. of stem | 134 | 80 |
| Model2: D + H + D*H + Species + Wilting stage | 135 | 78 |
| Model3: D + H + D*H + Species | 139 | 126 |
| Model4: D + H + D*H + Wilting stage | 138 | 93 |
| Model5: D + Species + Wilting stage | 137 | 75 |
| Model6: H + Species + Wilting stage | 137 | 84 |
| Model7: Species + Wilting stage | 138 | 83 |
| Model of Resprouting after Treatment | ||
| Model1: D + H + D*H + Species + Wilting stage + No. of stem + Stem status | 132 | 180 |
| Model2: D + H + D*H + Species + Writing stage + No. of stem | 134 | 193 |
| Model3: D + H + D*H + Species + Wilting stage + Stem status | 133 | 185 |
| Model4: D + H + D*H + Species + No. of stem + Stem status | 136 | 177 |
| Model5: D + H + D*H + No. of stem + Stem status | 139 | 178 |
| Model6: D + Species + No. of stem + Stem status | 138 | 176 |
| Model7: H + Species + No. of stem + Stem status | 138 | 177 |
| Model8: H*Species + No. of stem + Stem status | 135 | 181 |
| Model9: D*Species + No. of stem + Stem status | 135 | 179 |
| Model10: Species + No. of stem + Stem status | 139 | 175 |
| Model11: No. of stem + Stem status | 140 | 178 |
| Model12: Species + Stem status | 140 | 178 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.T.; Arndt, S.K.; Baker, P.J. Leaf Physiological Responses to Drought Stress and Community Assembly in an Asian Savanna. Forests 2019, 10, 1119. https://doi.org/10.3390/f10121119
Nguyen TT, Arndt SK, Baker PJ. Leaf Physiological Responses to Drought Stress and Community Assembly in an Asian Savanna. Forests. 2019; 10(12):1119. https://doi.org/10.3390/f10121119
Chicago/Turabian StyleNguyen, Thuy T., Stefan K. Arndt, and Patrick J. Baker. 2019. "Leaf Physiological Responses to Drought Stress and Community Assembly in an Asian Savanna" Forests 10, no. 12: 1119. https://doi.org/10.3390/f10121119
APA StyleNguyen, T. T., Arndt, S. K., & Baker, P. J. (2019). Leaf Physiological Responses to Drought Stress and Community Assembly in an Asian Savanna. Forests, 10(12), 1119. https://doi.org/10.3390/f10121119






