Tree Water Use, Water Use Efficiency, and Carbon Isotope Discrimination in Relation to Growth Potential in Populus deltoides and Hybrids under Field Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. Experimental Design
2.3. Clone Selection for Water Use Study
2.4. Tree Sap Flow and Transpiration
2.5. Canopy Stomatal Conductance and Specific Hydraulic Conductance
2.6. Biometrics
2.7. Isotope-Wood and Foliar δ13C
2.8. Statistical Analysis
3. Results
3.1. Environmental Conditions
3.2. Growth
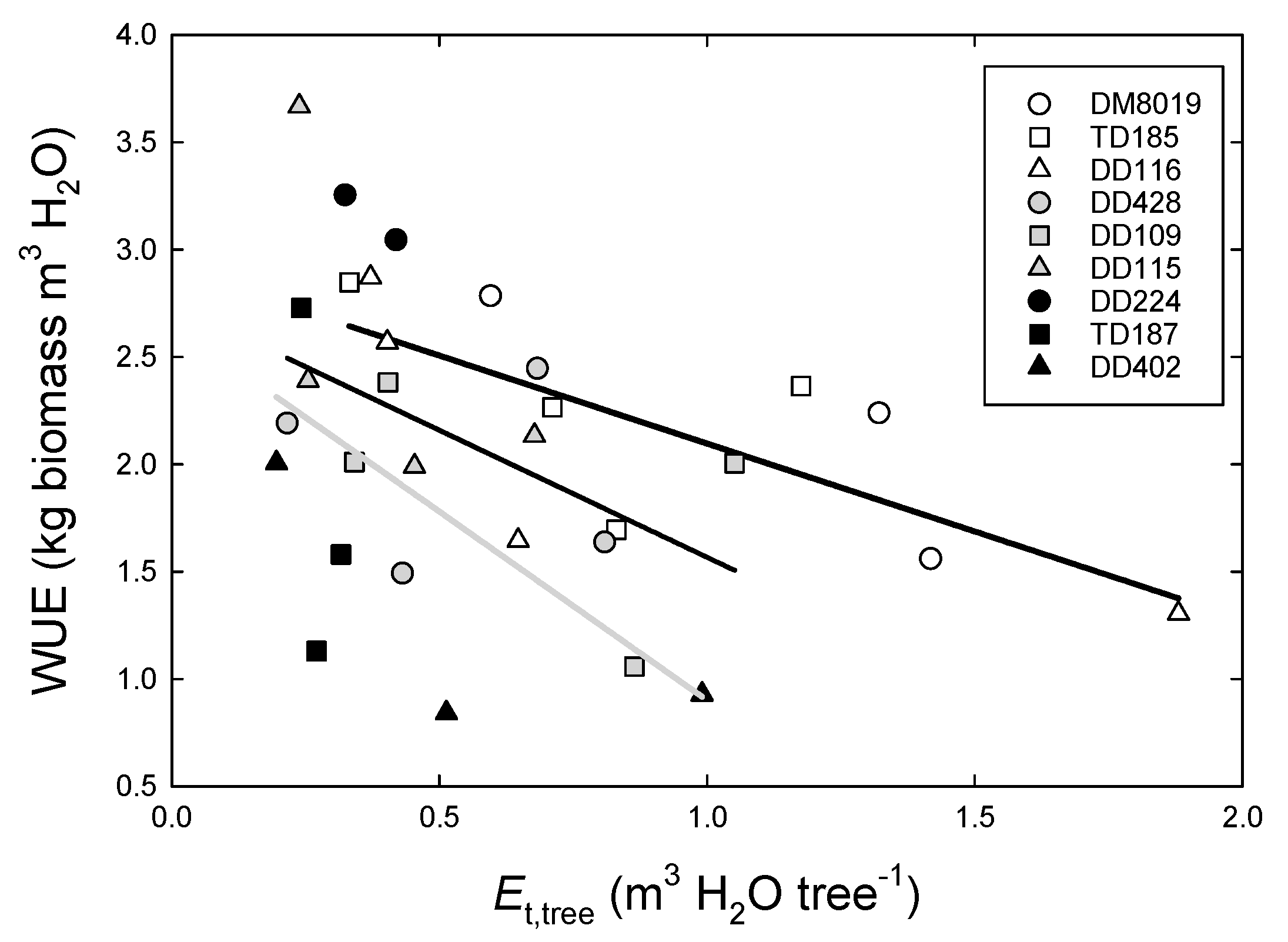
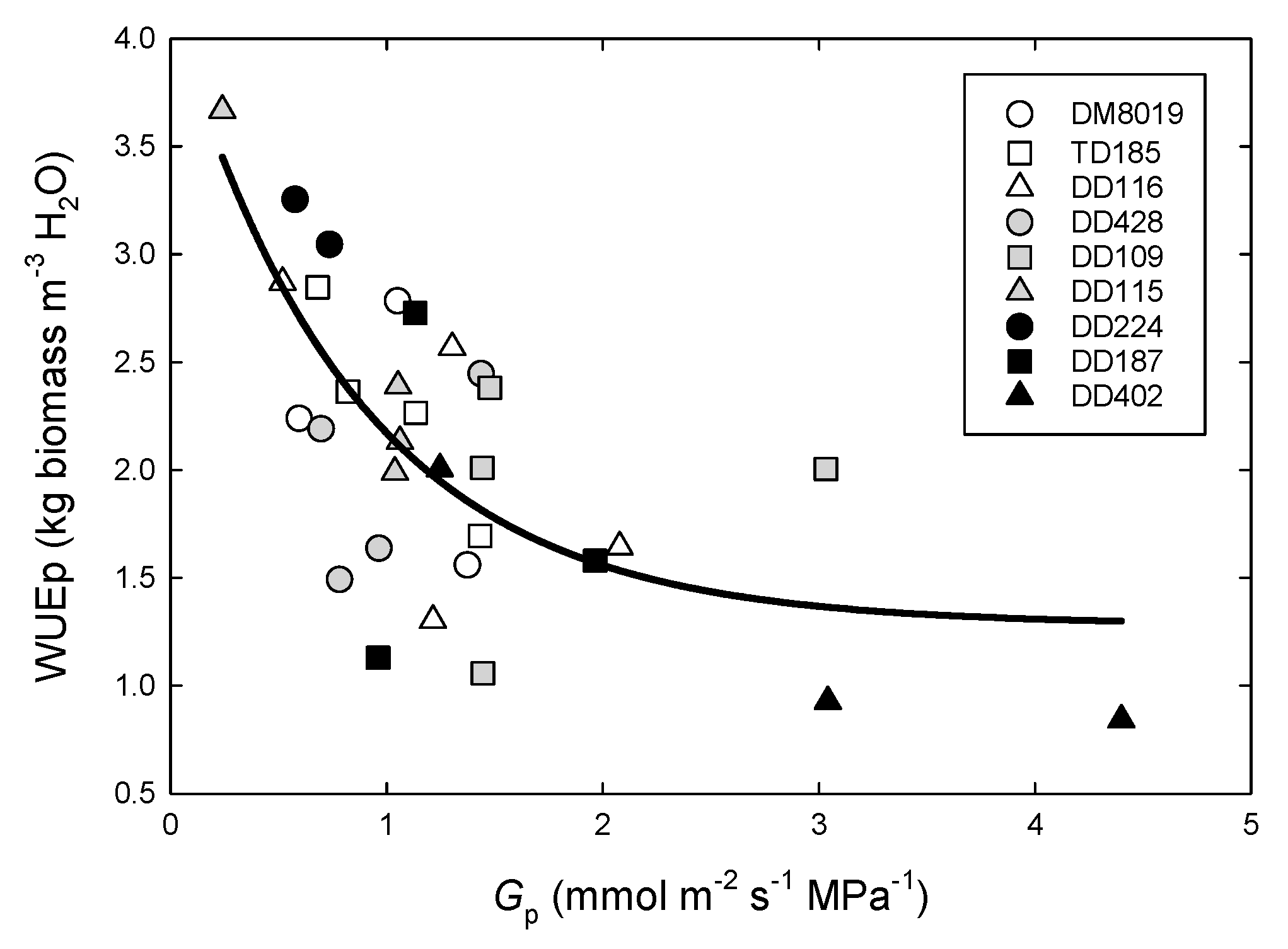
3.3. Tree Water Use and WUEp
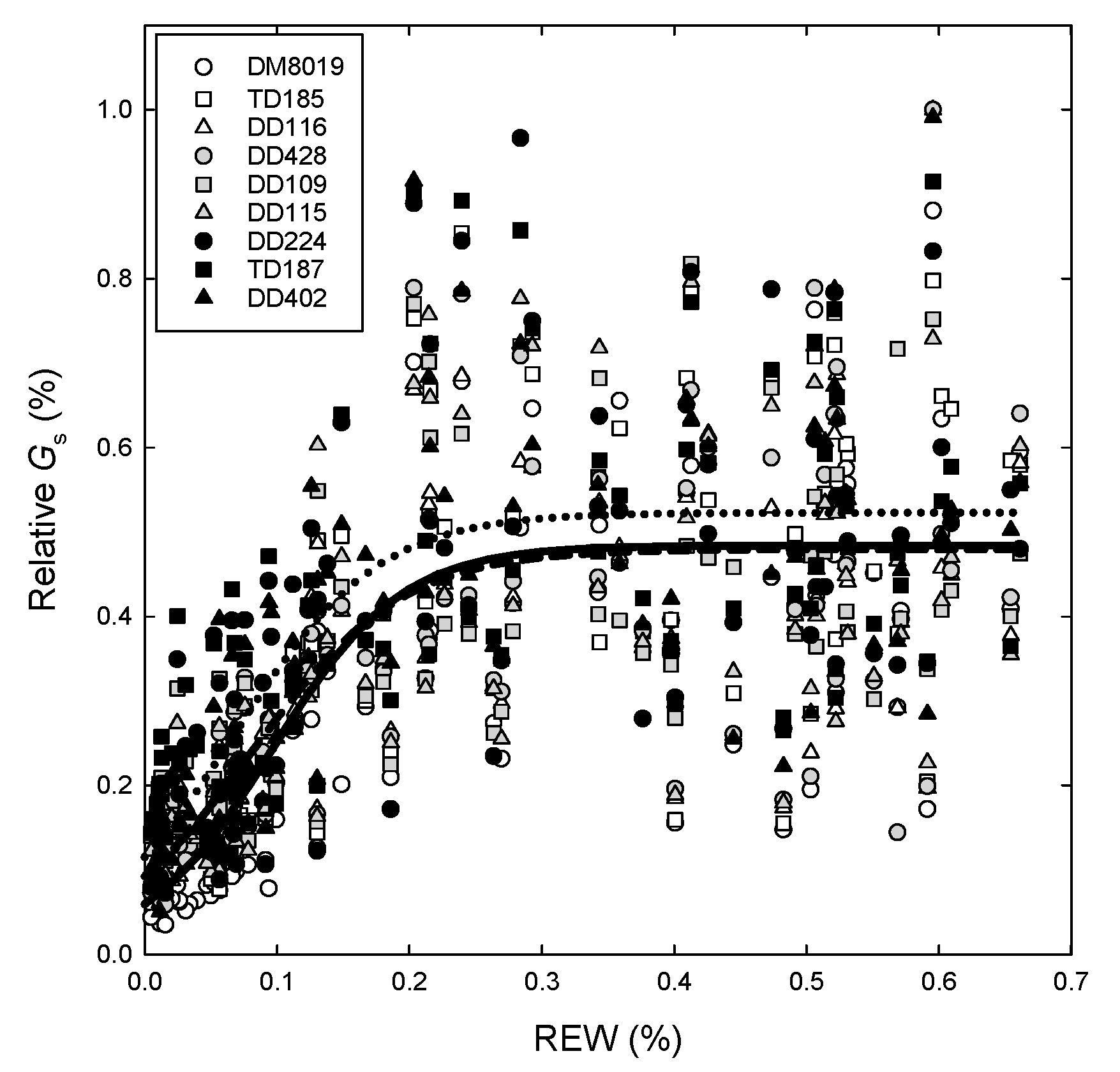
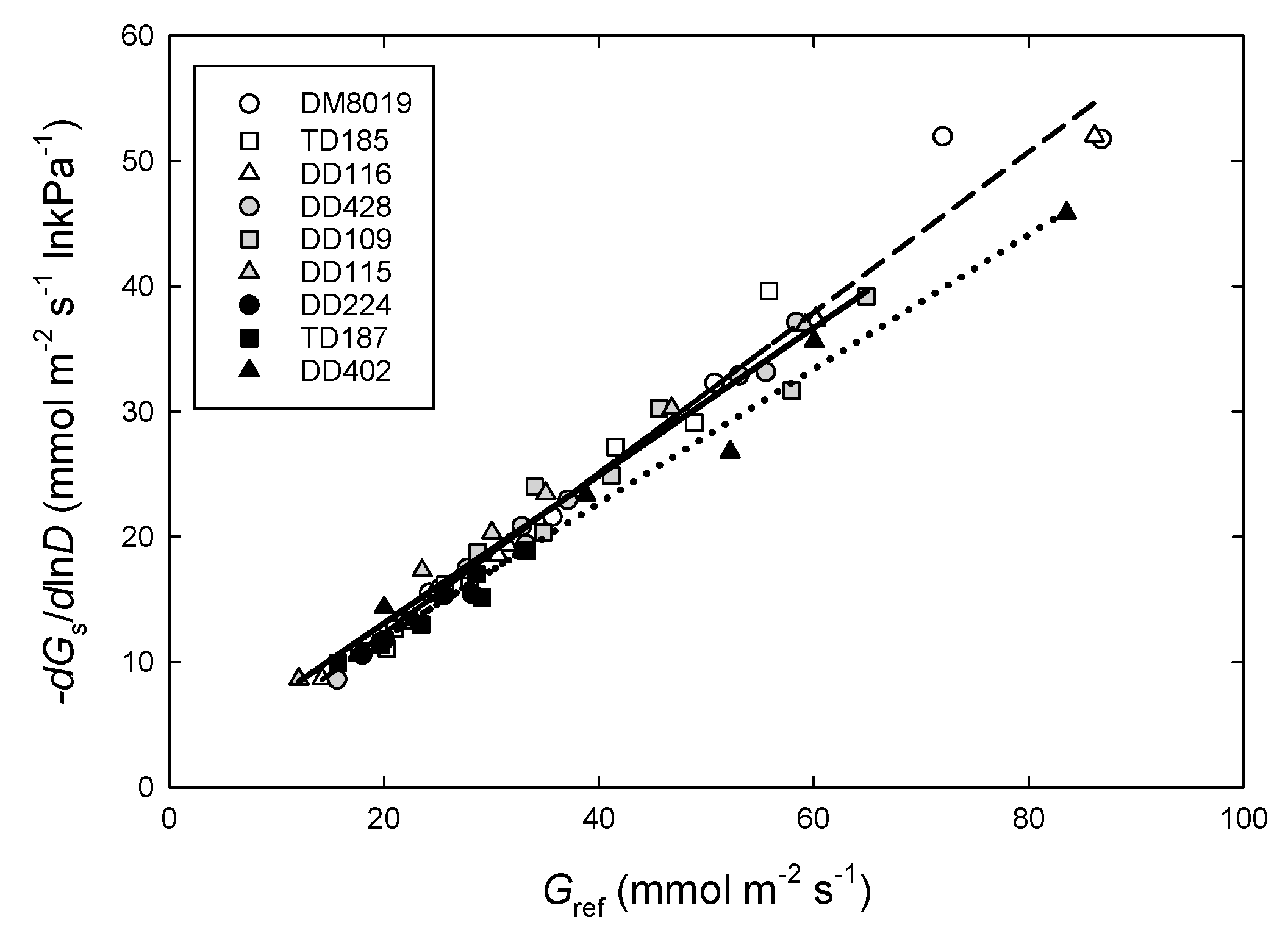
3.4. Canopy Stomatal Conductance and Hydraulic Conductance
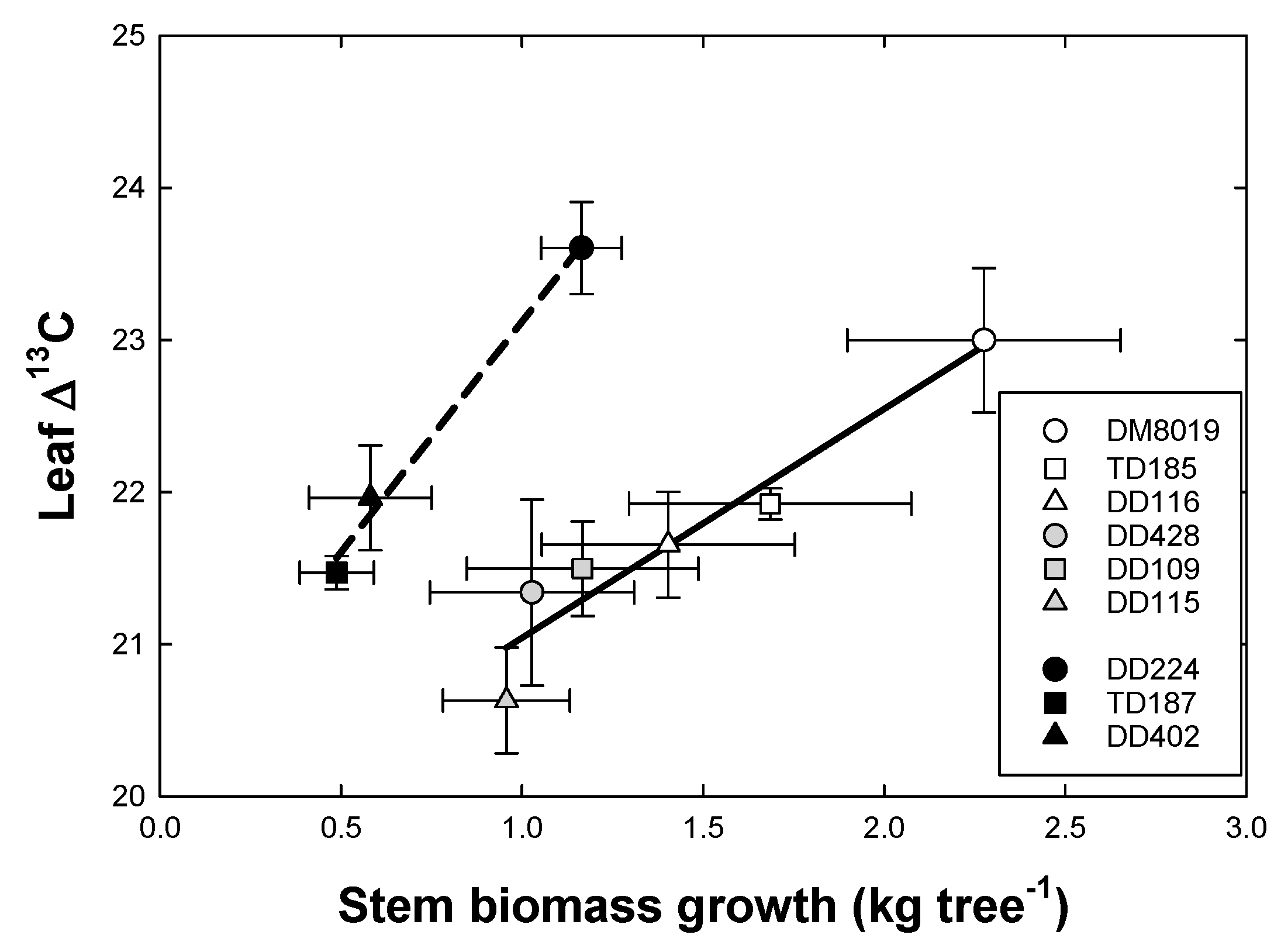
3.5. Stable Isotopes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perlack, R.D.E.; Laurence, M.; Anthony, F.T., Jr.; Matt, H.L.; Craig, C.B.; Mark, E.D.; Robin, L.G.; Lynn, L.W.; Jacob, M.W.; Anna, M.S.; et al. U.S. Billion-Ton Update: Biomass Supply for a Bioenergy and Bioproducts Industry; U.S. Department of Energy, Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2011; p. 227. [Google Scholar]
- Vance, E.; Loehle, C.; Wigley, T.; Weatherford, P. Scientific Basis for Sustainable Management of Eucalyptus and Populus as Short-Rotation Woody Crops in the U.S. Forests 2014, 5, 901–918. [Google Scholar] [CrossRef]
- King, J.S.; Ceulemans, R.; Albaugh, J.M.; Dillen, S.Y.; Domec, J.-C.; Fichot, R.; Fischer, M.; Leggett, Z.; Sucre, E.; Trnka, M.; et al. The Challenge of Lignocellulosic Bioenergy in a Water-Limited World. BioScience 2013, 63, 102–117. [Google Scholar] [CrossRef]
- Ghezehei, S.B.; Nichols, E.G.; Maier, C.A.; Hazel, D.W. Adaptability of Populus to Physiography and Growing Conditions in the Southeastern USA. Forests 2019, 10, 118. [Google Scholar] [CrossRef]
- Braatne, J.H.; Hinckley, T.M.; Stettler, R.F. Influence of soil water on the physiological and morphological components of plant water balance in Populus trichocarpa, Populus deltoides and their F1 hybrids. Tree Physiol. 1992, 11, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Oren, R.; Hinckley, T.M. Actual and potential transpiration and carbon assimilation in an irrigated poplar plantation. Tree Physiol. 2008, 28, 559–577. [Google Scholar] [CrossRef] [PubMed]
- Jassal, R.S.; Black, T.A.; Arevalo, C.; Jones, H.; Bhatti, J.S.; Sidders, D. Carbon sequestration and water use of a young hybrid poplar plantation in north-central Alberta. Biomass Bioenergy 2013, 56, 323–333. [Google Scholar] [CrossRef]
- Navarro, A.; Facciotto, G.; Campi, P.; Mastrorilli, M. Physiological adaptations of five poplar genotypes grown under SRC in the semi-arid Mediterranean environment. Trees 2014, 28, 983–994. [Google Scholar] [CrossRef]
- Hinckley, T.M.; Brooks, J.R.; Čermák, J.; Ceulemans, R.; Kučera, J.; Meinzer, F.C.; Roberts, D.A. Water flux in a hybrid poplar stand. Tree Physiol. 1994, 14, 1005–1018. [Google Scholar] [CrossRef]
- Tschaplinski, T.J.; Tuskan, G.A.; Gunderson, C.A. Water-stress tolerance of black and eastern cottonwood clones and four hybrid progeny. I. Growth, water relations, and gas exchange. Can. J. For. Res. 1994, 24, 364–371. [Google Scholar] [CrossRef]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processs and long-term consequences. Ann. Forst Sci. 2006, 63, 625–644. [Google Scholar] [CrossRef]
- Ceulemans, R.; Deraedt, W. Production physiology and growth potential of poplars under short-rotation forestry culture. For. Ecol. Manag. 1999, 121, 9–23. [Google Scholar] [CrossRef]
- Monclus, R.; Dreyer, E.; Villar, M.; Delmotte, F.M.; Delay, D.; Petit, J.-M.; Barbaroux, C.; Le Thiec, D.; Bréchet, C.; Brignolas, F. Impact of drought on productivity and water use efficiency in 29 genotypes of Populus deltoides × Populus nigra. New Phytol. 2006, 169, 765–777. [Google Scholar] [CrossRef] [PubMed]
- Ghezehei, S.B.; Nichols, E.G.; Hazel, D.W. Early Clonal Survival and Growth of Poplars Grown on North Carolina Piedmont and Mountain Marginal Lands. BioEnergy Res. 2016, 9, 548–558. [Google Scholar] [CrossRef]
- Isebrands, J.G.; Richardson, J. Poplars and Willows: Trees for Society and the Environment; CABI: Oxford, UK, 2014. [Google Scholar]
- Zalesny, R.S.; Headlee, W.L.; Gopalakrishnan, G.; Bauer, E.O.; Hall, R.B.; Hazel, D.W.; Isebrands, J.G.; Licht, L.A.; Negri, M.C.; Nichols, E.G.; et al. Ecosystem services of poplar at long-term phytoremediation sites in the Midwest and Southeast, United States. Wiley Interdiscip. Rev. Energy Environ. 2019, 8, e349. [Google Scholar] [CrossRef]
- Hubbard, R.M.; Ryan, M.G.; Stiller, V. Stomatal conductance and photosynthesis vary linearly with plant hydraulic conductance in ponderosa pine. Plant Cell Environ. 2001, 24, 113–121. [Google Scholar] [CrossRef]
- Brodribb, T.J.; McAdam, S.A.M.; Jordan, G.J.; Feild, T.S. Evolution of stomatal responsiveness to CO2 and optimization of water-use efficiency among land plants. New Phytol. 2009, 183, 839–847. [Google Scholar] [CrossRef]
- Tyree, M.T. Hydraulic limits on tree performance: Transpiration, carbon gain and growth of trees. Trees 2003, 17, 95–100. [Google Scholar] [CrossRef]
- Whitehead, D.; Beadle, C.L. Physiological regulation of productivity and water use in Eucalptus: A review. For. Ecol. Manag. 2004, 193, 113–140. [Google Scholar] [CrossRef]
- McCarthy, H.R.; Pataki, D.E.; Jenerette, G.D. Plant water-use efficiency as a metric of urban ecosystem services. Ecol. Appl. 2011, 21, 3115–3127. [Google Scholar] [CrossRef]
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant Physiological Ecology; Springer: New York, NY, USA, 2008. [Google Scholar]
- Farquhar, G.D.; Hubick, K.T.; Condon, A.G.; Richards, R.A. Carbon Isotope Fractionation and Plant Water-Use Efficiency. In Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Springer: New York, NY, USA, 1989; pp. 21–40. [Google Scholar] [CrossRef]
- Rae, A.M.; Robinson, K.M.; Street, N.R.; Taylor, G. Morphological and physiological traits influencing biomass productivity in short-rotation coppice poplar. Can. J. For. Res. 2004, 34, 1488–1498. [Google Scholar] [CrossRef]
- Zhang, X.; Zang, R.; Li, C. Population differences in physiological and morphological adaptations of Populus davidiana seedlings in response to progressive drought stress. Plant Sci. 2004, 166, 791–797. [Google Scholar] [CrossRef]
- Marron, N.; Villar, M.; Dreyer, E.; Delay, D.; Boudouresque, E.; Petit, J.-M.; Delmotte, F.M.; Guehl, J.-M.; Brignolas, F. Diversity of leaf traits related to productivity in 31 Populus deltoides × Populus nigra clones. Tree Physiol. 2005, 25, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Dillen, S.Y.; Marron, N.; Koch, B.; Ceulemans, R. Genetic Variation of Stomatal Traits and Carbon Isotope Discrimination in Two Hybrid Poplar Families (Populus deltoides ‘S9-2’ × P. nigra ‘Ghoy’ and P. deltoides ‘S9-2’ × P. trichocarpa ‘V24’). Ann. Bot. 2008, 102, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Ripullone, F.; Lauteri, M.; Grassi, G.; Amato, M.; Borghetti, M. Variation in nitrogen supply changes water-use efficiency of Pseudotsuga menziesii and Populus x euroamericana; a comparison of three approaches to determine water-use efficiency. Tree Physiol. 2004, 24, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Bonhomme, L.; Barbaroux, C.; Monclus, R.; Morabito, D.; Berthelot, A.; Villar, M.; Dreyer, E.; Brignolas, F. Genetic variation in productivity, leaf traits and carbon isotope discrimination in hybrid poplars cultivated on contrasting sites. Ann. For. Sci. 2008, 65, 503. [Google Scholar] [CrossRef]
- Monclus, R.; Villar, M.; Barbaroux, C.; Bastien, C.; Fichot, R.; Delmotte, F.M.; Delay, D.; Petit, J.M.; Bréchet, C.; Dreyer, E.; et al. Productivity, water-use efficiency and tolerance to moderate water deficit correlate in 33 poplar genotypes from a Populus deltoides × Populus trichocarpa F1 progeny. Tree Physiol. 2009, 29, 1329–1339. [Google Scholar] [CrossRef]
- Fischer, M.; Fichot, R.; Albaugh, J.M.; Ceulemans, R.; Domec, J.C.; Trnka, M.; King, J.S. Populus and Salix grown in a short-rotation coppice for bioenergy: Ecophysiology, aboveground productivity, and stand-level water use efficiency. In Sustainable Biofuels, an Ecological Assessment of the Future Energy; Ajay, K.B., Zenone, T., Chen, J., Eds.; HEP deGruyter: Berlin, Germany, 2015; pp. 155–194. [Google Scholar]
- Silim, S.; Nash, R.; Reynard, D.; White, B.; Schroeder, W. Leaf gas exchange and water potential responses to drought in nine poplar (Populus spp.) clones with contrasting drought tolerance. Trees 2009, 23, 959–969. [Google Scholar] [CrossRef]
- Arango-Velez, A.; Zwiazek, J.J.; Thomas, B.R.; Tyree, M.T. Stomatal factors and vulnerability of stem xylem to cavitation in poplars. Physiol. Plant. 2011, 143, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Attia, Z.; Domec, J.-C.; Oren, R.; Way, D.A.; Moshelion, M. Growth and physiological responses of isohydric and anisohydric poplars to drought. J. Exp. Bot. 2015. [Google Scholar] [CrossRef]
- Schulte, P.J.; Hinckley, T.M.; Stettler, R.F. Stomatal responses of Populus to leaf water potential. Can. J. Bot. 1987, 65, 255–260. [Google Scholar] [CrossRef]
- Yin, C.; Wang, X.; Duan, B.; Luo, J.; Li, C. Early growth, dry matter allocation and water use efficiency of two sympatric Populus species as affected by water stress. Environ. Exp. Bot. 2005, 53, 315–322. [Google Scholar] [CrossRef]
- Pointeau, V.M.; Guy, R.D. Comparative resource-use efficiencies and growth of Populus trichocarpa and Populus balsamifera under glasshouse conditions. Botany 2014, 92, 443–451. [Google Scholar] [CrossRef]
- Schimel, D.S.; Braswell, B.H.; McKeown, R.; Ojima, D.S.; Parton, W.J.; Pulliam, W. Climate and nitrogen controls on the geography and timescales of terrestrial biogeochemical cycling. Glob. Biogeochem. Cycles 1996, 10, 677–692. [Google Scholar] [CrossRef]
- Chaves, M.M.; Osorio, M.J.; Pereira, J.S. Water use efficiency and photosynthesis. In Water Use Efficiency in Plant Biology; Bacon, M.A., Ed.; Blackwell Publishing: Oxford, UK, 2004; pp. 42–74. [Google Scholar]
- Fardusi, M.J.; Ferrio, J.P.; Comas, C.; Voltas, J.; de Dios, V.R.; Serrano, L. Intra-specific association between carbon isotope composition and productivity in woody plants: A meta-analysis. Plant Sci. 2016, 251, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Condon, A.G.; Richards, R.A.; Rebetzke, G.J.; Farquhar, G.D. Breeding for high water-use efficiency. J. Exp. Bot. 2004, 55, 2447–2460. [Google Scholar] [CrossRef]
- Sinclair, T.R. Is transpiration efficiency a viable plant trait in breeding for crop improvement? Funct. Plant Biol. 2012, 39, 359–365. [Google Scholar] [CrossRef]
- Binkley, D.; Stape, J.L.; Ryan, M.G. Thinking about efficiency of resource use in forests. For. Ecol. Manag. 2004, 193, 5–16. [Google Scholar] [CrossRef]
- Binkley, D. Understanding the Role of Resource Use Efficiency in Determining the Growth of Trees and Forests. In Forests in Development: A Vital Balance; Schlichter, T., Montes, L., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 13–26. [Google Scholar] [CrossRef]
- Monclus, R.; Dreyer, E.; Delmotte, F.M.; Villar, M.; Delay, D.; Boudouresque, E.; Petit, J.-M.; Marron, N.; Bréchet, C.; Brignolas, F. Productivity, leaf traits and carbon isotope discrimination in 29 Populus deltoides × P. nigra clones. New Phytol. 2005, 167, 53–62. [Google Scholar] [CrossRef]
- Shifflett, S.D.; Hazel, D.W.; Frederick, D.J.; Nichols, E.G. Species Trials of Short Rotation Woody Crops on Two Wastewater Application Sites in North Carolina, USA. BioEnergy Res. 2014, 7, 157–173. [Google Scholar] [CrossRef]
- Coyle, D.R.; Coleman, M.D.; Durant, J.A.; Newman, L.A. Survival and growth of 31 Populus clones in South Carolina. Biomass Bioenergy 2006, 30, 750–758. [Google Scholar] [CrossRef]
- Zalesny, R.S.; Hall, R.B.; Zalesny, J.A.; McMahon, B.G.; Berguson, W.E.; Stanosz, G.R. Biomass and Genotype × Environment Interactions of Populus Energy Crops in the Midwestern United States. BioEnergy Res. 2009, 2, 106–122. [Google Scholar] [CrossRef]
- Ghezehei, S.B.; Shifflett, S.D.; Hazel, D.W.; Nichols, E.G. SRWC bioenergy productivity and economic feasibility on marginal lands. J. Environ. Manag. 2015, 160, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.G. Growth and Leaf Area Dynamics of Short-Rotation Populus Genotypes; North Carolina State University: Raleigh, NC, USA, 2016. [Google Scholar]
- Albaugh, T.J.; Albaugh, J.M.; Fox, T.R.; Allen, H.L.; Rubilar, R.A.; Trichet, P.; Loustau, D.; Linder, S. Tamm Review: Light use efficiency and carbon storage in nutrient and water experiments on major forest plantation species. For. Ecol. Manag. 2016, 376, 333–342. [Google Scholar] [CrossRef]
- Granier, A. Une nouvelle methode pour la measure du flux de seve brute dans le tronc des arbres. Ann. For. Sci. 1985, 42, 193–200. [Google Scholar] [CrossRef]
- Jones, H.G. Plants and Microclimate; Cambridge University Press: Oxford, UK, 1986. [Google Scholar]
- Samuelson, L.J.; Stokes, T.A.; Coleman, M.D. Influence of irrigation and fertilization on transpiration and hydraulic properties of Populus deltoides. Tree Physiol. 2007, 27, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Edwards, W.R.N.; Booker, R.E. Radial Variation in the Axial Conductivity of Populus and its Significance in Heat Pulse Velocity Measurement. J. Exp. Bot. 1984, 35, 551–561. [Google Scholar] [CrossRef]
- Lambs, L.; Muller, É. Sap flow and water transfer in the Garonne River riparian woodland, France: First results on poplar and willow. Ann. For. Sci. 2002, 59, 301–315. [Google Scholar] [CrossRef]
- Nobel, P.S. Physicochemical & Environmental Plant Physiology; Academic Press: San Diego, CA, USA, 1999. [Google Scholar]
- Oren, R.; Sperry, J.S.; Katul, G.G.; Pataki, D.E.; Ewers, B.E.; Phillips, N.; Schafer, K.V.R. Survey and synthesis of intra- and interspecific variation in stomatal sensitivity to vapour pressure deficit. Plant Cell Environ. 1999, 22, 1515–1526. [Google Scholar] [CrossRef]
- Ewers, B.E.; Oren, R. Analyses of assumptions and errors in the calculation of stomatal conductance from sap flux measurments. Tree Physiol. 2000, 20, 579–589. [Google Scholar] [CrossRef]
- Wullschleger, S.D.; Meinzer, F.C.; Vertessy, R.A. A review of whole-plant water use studies in tree. Tree Physiol. 1998, 18, 499–512. [Google Scholar] [CrossRef]
- Meinzer, F.C.; Goldstein, G.; Jackson, P.; Holbrook, N.M.; Gutiérrez, M.V.; Cavelier, J. Environmental and physiological regulation of transpiration in tropical forest gap species: The influence of boundary layer and hydraulic properties. Oecologia 1995, 101, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Franks, P.J. Stomatal control and hydraulic conductance, with special reference to tall trees. Tree Physiol. 2004, 24, 865–878. [Google Scholar] [CrossRef] [PubMed]
- Zarnoch, S.J. Testing Hypotheses for Differences Between Linear Regression Lines; SRS–17; USDA Forest Service, Southern Research Station: Asheville, NC, USA, 2009. [Google Scholar]
- Cannell, M.G.R.; Sheppard, L.J.; Milne, R. Light Use Efficiency and Woody Biomass Production of Poplar and Willow. Forestry 1988, 61, 125–136. [Google Scholar] [CrossRef]
- Curtis, P.S.; Vogel, C.S.; Wang, X.; Pregitzer, K.S.; Zak, D.R.; Lussenhop, J.; Kubiske, M.; Teeri, J.A. Gas exchange, leaf nitrogen, and growth efficiency of Populus tremuloides in a CO2-enriched atmosphere. Ecol. Appl. 2000, 10, 3–17. [Google Scholar]
- Pellis, A.; Laureysens, I.; Ceulemans, R. Growth and production of a short rotation coppice culture of poplar I. Clonal differences in leaf characteristics in relation to biomass production. Biomass Bioenergy 2004, 27, 9–19. [Google Scholar] [CrossRef]
- Broeckx, L.; Vanbeveren, S.; Verlinden, M.; Ceulemans, R. First vs. second rotation of a poplar short rotation coppice: Leaf area development, light interception and radiation use efficiency. iForest—Biogeosci. For. 2015, 8, 565–573. [Google Scholar] [CrossRef]
- Dowell, R.C.; Gibbins, D.; Rhoads, J.L.; Pallardy, S.G. Biomass production physiology and soil carbon dynamics in short-rotation-grown Populus deltoides and P. deltoides × P. nigra hybrids. For. Ecol. Manag. 2009, 257, 134–142. [Google Scholar] [CrossRef]
- Albaugh, T.J.; Allen, H.L.; Dougherty, P.M.; Johnsen, K.H. Long term growth responses of loblolly pine to optimal nutrient and water resource availability. For. Ecol. Manag. 2004, 192, 3–19. [Google Scholar] [CrossRef]
- Stape, J.L.; Binkley, D.; Ryan, M.G. Production and carbon allocation in clonal Eucalyptus plantation with water and nutrient manipulations. For. Ecol. Manag. 2008, 255, 920–930. [Google Scholar] [CrossRef]
- Ryan, M.G.; Stape, J.L.; Binkley, D.; Fonseca, S.; Loos, R.A.; Takahashi, E.N.; Silva, C.R.; Hakamada, R.E.; Ferreira, J.M.; Lima, A.M.N.; et al. Factors controlling Eucalyptus productivity: How water availability and stand structure alter production and carbon allocation. For. Ecol. Manag. 2010, 259, 1695–1703. [Google Scholar] [CrossRef]
- Rasheed, F.; Dreyer, E.; Richard, B.; Brignolis, F.; Montpied, P.; Thiec, D.L. Genotype differences in 13C discrimination between atmospheric and leaf matter match differences in transpiration efficiency at leaf and whole-plant levels in hybrid Populus deltoides x nigra. Plant Cell Environ. 2013, 36, 87–102. [Google Scholar] [CrossRef] [PubMed]
- Le Roux, D.; Stock, W.D.; Bond, W.J.; Maphanga, D. Dry mass allocation, water use efficiency and δ13C in clones of Eucalyptus grandis, E. grandis × camaldulensis and E. grandis × nitens grown under two irrigation regimes. Tree Physiol. 1996, 16, 497–502. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Forrester, D.I.; Collopy, J.J.; Beadle, C.L.; Warren, C.R.; Baker, T.G. Effect of thinning, pruning and nitrogen fertilizer application on transpiration, photosynthesis and water-use effliciency in a young Eucalyptus nitens plantation. For. Ecol. Manag. 2012, 266, 286–300. [Google Scholar] [CrossRef]
- Stape, J.L.; Binkley, D.; Ryan, M.G. Eucalyptus production and the supply, use and efficiency of use of water, light and nitrogen across a geographic gradient in Brazil. For. Ecol. Manag. 2004, 193, 17–31. [Google Scholar] [CrossRef]
- Binkley, D.; Stape, J.L.; Ryan, M.G.; Barnard, H.R.; Fownes, J. Age-related decline in forest ecosystem growth: An individual-tree, stand structure hypothesis. Ecosystems 2002, 5, 58–67. [Google Scholar] [CrossRef]
- Otto, M.S.G.; Hubbard, R.M.; Binkley, D.; Stape, J.L. Dominant clonal Eucalyptus grandis x urophylla trees use water more efficiently. For. Ecol. Manag. 2014, 328, 117–121. [Google Scholar] [CrossRef]
- Donovan, L.A.; Ehleringer, J.R. Carbon isotope discrimination, water-use efficiency, growth, and mortality in a natural shrub population. Oecologia 1994, 100, 347–354. [Google Scholar] [CrossRef]
- Flanagan, L.B.; Johnsen, K.H. Genetic variation in carbon isotope discrimination and its relationship to growth under field conditions in full-sib families of Picea mariana. Can. J. For. Res. 1995, 25, 39–47. [Google Scholar] [CrossRef]
- Coleman, M.D.; Dickson, R.E.; Isebrands, J.G. Growth and physiology of aspen supplied with different fertilizer addition rates. Physiol. Plant. 1998, 103, 513–526. [Google Scholar] [CrossRef]
- Casella, E.; Ceulemans, R. Spatial distribution of leaf morphological and physiological characteristics in relation to local radiation regime within the canopies of 3-year-old Populus clones in coppice culture. Tree Physiol. 2002, 22, 1277–1288. [Google Scholar] [CrossRef][Green Version]
- Sparks, J.P.; Ehleringer, J.R. Leaf carbon isotope discrimination and nitrogen content for riparian trees along elevational transects. Oecologia 1997, 109, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Voltas, J.; Serrano, L.; Hernández, M.; Pemán, J. Carbon Isotope Discrimination, Gas Exchange and Stem Growth of Four Euramerican Hybrid Poplars under Different Watering Regimes. New For. 2006, 31, 435–451. [Google Scholar] [CrossRef]
- Schifman, L.A.; Stella, J.C.; Volk, T.A.; Teece, M.A. Carbon isotope variation in shrub willow (Salix spp.) ring-wood as an indicator of long-term water status, growth and survival. Biomass Bioenergy 2012, 36, 316–326. [Google Scholar] [CrossRef]
- Toillon, J.; Rollin, B.; Dallé, E.; Feinard-Duranceau, M.; Bastien, J.-C.; Brignolas, F.; Marron, N. Variability and plasticity of productivity, water-use efficiency, and nitrogen exportation rate in Salix short rotation coppice. Biomass Bioenergy 2013, 56, 392–404. [Google Scholar] [CrossRef]
- Sun, Z.J.; Livingston, N.J.; Guy, R.D.; Ethier, G.J. Stable carbon isotopes as indicators of increased water use efficiecny and productivity in white spruce (Picea glauca (Moench) Voss) seedlings. Plant Cell Environ. 1996, 19, 887–894. [Google Scholar] [CrossRef]
- Bond, W.J.; Stock, W.D. Preliminary Assessment of the Grading of Eucalyptus Clones Using Carbon Isotope Discrimination. S. Afr. For. J. 1990, 154, 51–55. [Google Scholar] [CrossRef]
- Pita, P.; Soria, F.; Cañas, I.; Toval, G.; Pardos, J.A. Carbon isotope discrimination and its relationship to drought resistance under field conditions in genotypes of Eucalyptus globulus Labill. For. Ecol. Manag. 2001, 141, 211–221. [Google Scholar] [CrossRef]
- Chamaillard, S.; Fichot, R.; Vincent-Barbaroux, C.; Bastien, C.; Depierreux, C.; Dreyer, E.; Villar, M.; Brignolas, F. Variations in bulk leaf carbon isotope discrimination, growth and related leaf traits among three Populus nigra L. populations. Tree Physiol. 2011, 31, 1076–1087. [Google Scholar] [CrossRef]
- Verlinden, M.S.; Fichot, R.; Broeckx, L.S.; Vanholme, B.; Boerjan, W.; Ceulemans, R. Carbon isotope compositions (δ13C) of leaf, wood and holocellulose differ among genotypes of poplar and between previous land uses in a short-rotation biomass plantation. Plant Cell Environ. 2015, 38, 144–156. [Google Scholar] [CrossRef]
- Cernusak, L.A.; Aranda, J.; Marshall, J.D.; Winter, K. Large variation in whole-plant water-use efficiency among tropical tree species. New Phytol. 2007, 173, 294–305. [Google Scholar] [CrossRef]
- Zhang, J.; Marshall, J.D. Population differences in water-use efficiency of well-watered and water-stressed western larch seedlings. Can. J. For. Res. 1994, 24, 92–99. [Google Scholar] [CrossRef]
- Seibt, U.; Rajabi, A.; Griffiths, H.; Berry, J.A. Carbon isotopes and water use efficiency: Sense and sensitivity. Oecologia 2008, 155, 441. [Google Scholar] [CrossRef] [PubMed]
- Fichot, R.; Barigah, T.S.; Chamaillard, S.; Le Thiec, D.; Laurans, F.; Cochard, H.; Brignolas, F. Common trade-offs between xylem resistance to cavitation and other physiological traits do not hold among unrelated Populus deltoides × Populus nigra hybrids. Plant Cell Environ. 2010, 33, 1553–1568. [Google Scholar] [CrossRef] [PubMed]
- Fichot, R.; Laurans, F.; Monclus, R.; Moreau, A.; Pilate, G.; Brignolas, F. Xylem anatomy correlates with gas exchange, water-use efficiency and growth performance under contrasting water regimes: Evidence from Populus deltoides × Populus nigra hybrids. Tree Physiol. 2009, 29, 1537–1549. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, D. Regulation of stomatal conductance and transpiration in forest canopies. Tree Physiol. 1998, 18, 633–644. [Google Scholar] [CrossRef]
- O’Grady, A.P.; Worledge, D.; Wilkinson, A.; Battaglia, M. Photosynthesis and respiration decline with light intensity in dominant and suppressed Eucalyptus globulus canopies. Funct. Plant Biol. 2008, 35, 439–447. [Google Scholar] [CrossRef]
- Wullschleger, S.D.; Wilson, K.B.; Hanson, P.J. Environmental control of whole-plant transpiration, canopy conductance and estimates of the decoupling coefficient for large red maple trees. Agric. For. Meteorol. 2000, 104, 157–168. [Google Scholar] [CrossRef]
- Broadmeadow, M.S.J.; Griffiths, H. 8—Carbon Isotope Discrimination and the Coupling of CO2 Fluxes within Forest Canopies. In Stable Isotopes and Plant Carbon-Water Relations; Ehleringer, J.R., Hall, A.E., Farquhar, G.D., Eds.; Academic Press: San Diego, CA, USA, 1993; pp. 109–129. [Google Scholar] [CrossRef]
- Hubbard, R.M.; Bond, B.J.; Ryan, M.G. Evidence that hydraulic conductance limits photosynthesis in old Pinus ponderosa trees. Tree Physiol. 1999, 19, 165–172. [Google Scholar] [CrossRef]
- Williams, M.; Bond, B.J.; Ryan, M.G. Evaluating different soil and plant hydraulic constraints on tree function using a model and sap flow data from ponderosa pine. Plant Cell Environ. 2001, 24, 679–690. [Google Scholar] [CrossRef]
- Samuelson, L.J.; McLemore, P.C.; Somers, G.L. Relationship between foliar d13C and hydraulic pathway length in Pinus palustris. For. Sci. 2003, 49, 790–798. [Google Scholar]
- Forrester, D.I. Transpiration and water-use efficiency in mixed-species forests versus monocultures: Effects of tree size, stand density and season. Tree Physiol. 2015, 35, 289–304. [Google Scholar] [CrossRef] [PubMed]
- Bloemen, J.; Fichot, R.; Horemans, J.A.; Broeckx, L.S.; Verlinden, M.S.; Zenone, T.; Ceulemans, R. Water use of a multigenotype poplar short-rotation coppice from tree to stand scale. GCB Bioenergy 2017, 9, 370–384. [Google Scholar] [CrossRef] [PubMed]
- Mencuccini, M. The ecological significance of long-distance water transport: Short-term regulation, long-term acclimation and the hydraulic costs of stature across plant life forms. Plant Cell Environ. 2003, 26, 163–182. [Google Scholar] [CrossRef]
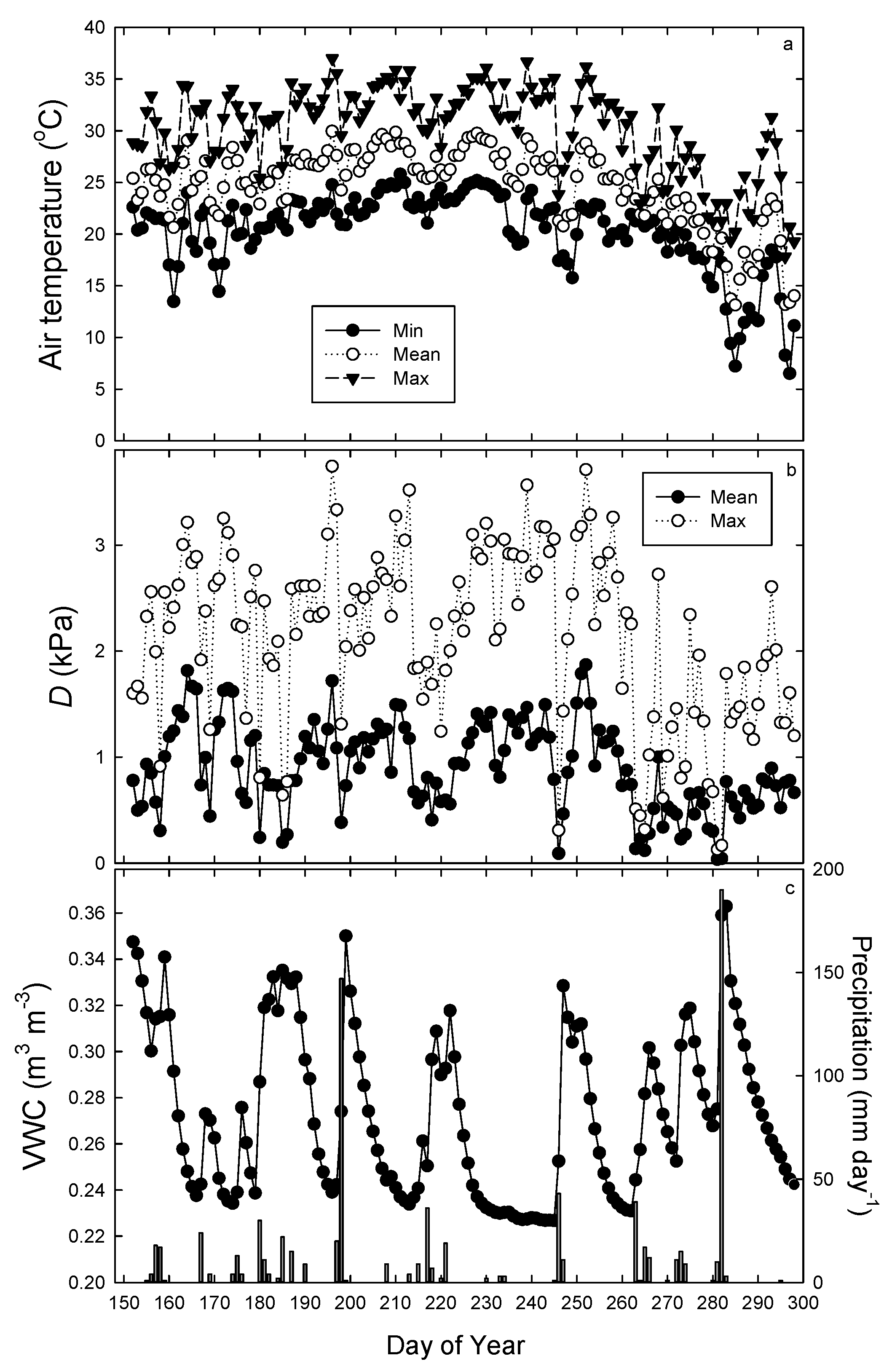
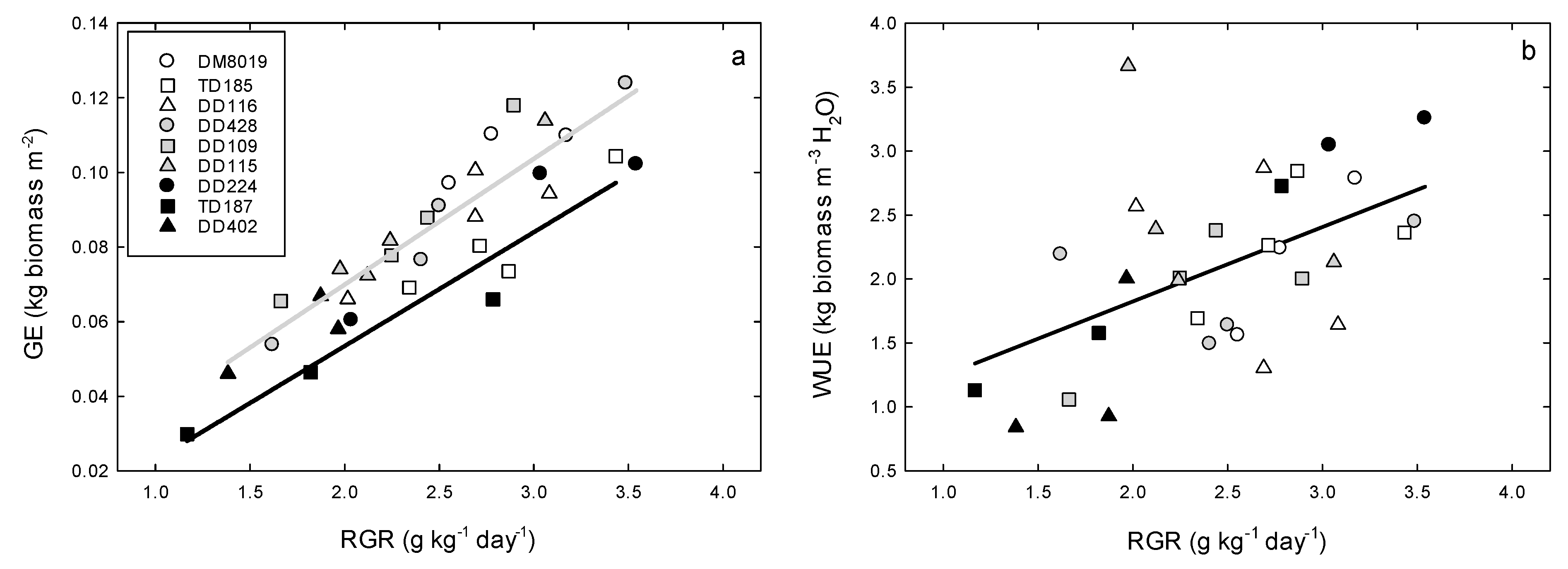
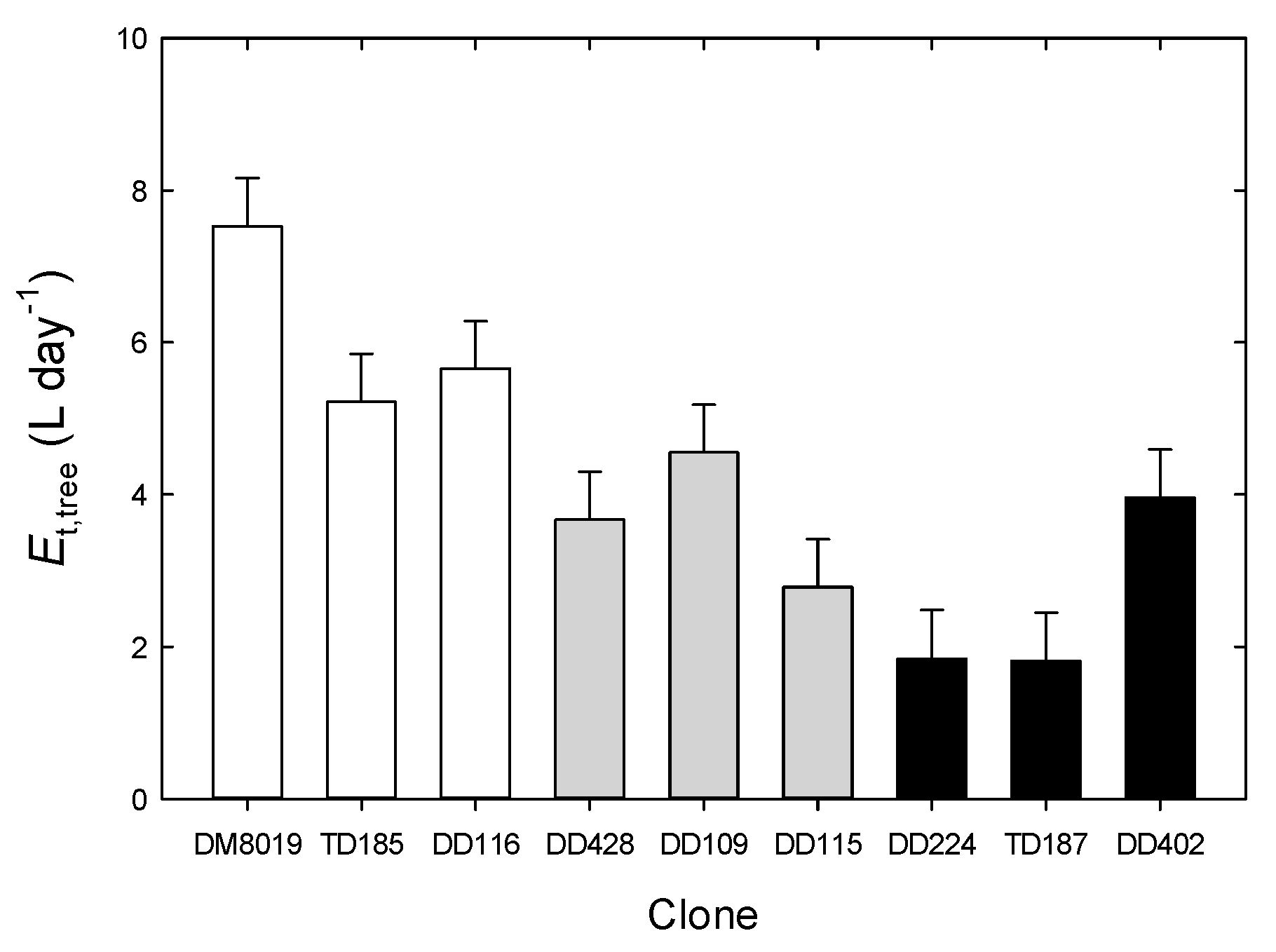
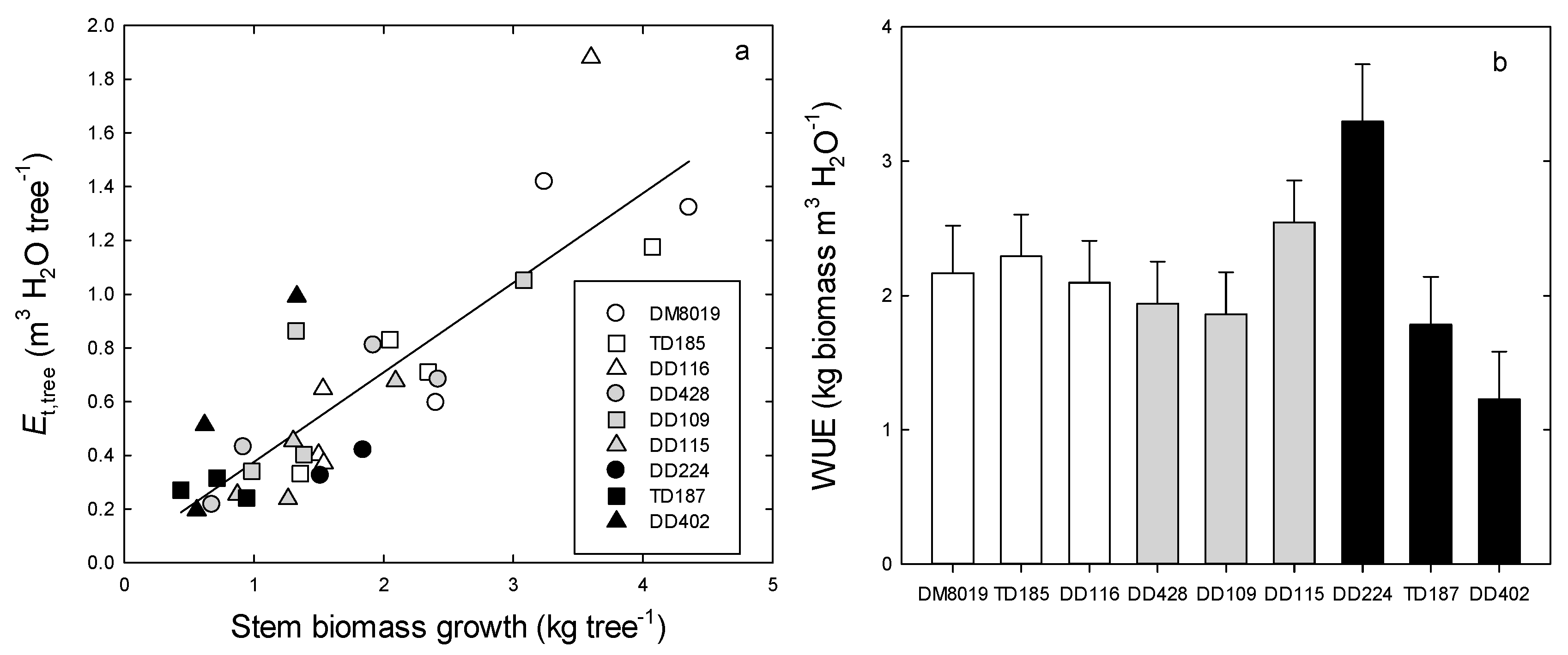
| Clone | Group | Height (m) | Initial DBH (cm) | Initial Stem Biomass (kg tree−1) | Stem Growth (kg tree−1) | RGR (g kg−1 day−1) | Foliage Biomass (kg tree−1) | Root Biomass (kg tree−1) | SLA (cm2 g−1) | Leaf Area (Al, m2) | Sapwood Area (As, cm2) | Al:As (m2 cm−2) | Al:Root (cm2 g−1) | AH (cm2 m−3) | GE (kg tree−1 m−2) | n |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DM8019 | H | 9.70 (0.62) | 7.76 (0.59) | 5.59 (0.70) | 2.28 (0.32) | 2.84 (0.33) | 1.59 (0.18) | 1.46 (0.17) | 135.2 (7.0) | 21.5 (2.4) | 48.6 (5.46) | 0.445 (0.003) | 148.0 (1.2) | 0.240 (0.018) | 0.106 (0.011) | 3 |
| TD185 | H | 8.70 (0.54) | 6.60 (0.50) | 4.00 (0.60) | 1.69 (0.28) | 2.84 (0.28) | 1.24 (0.15) | 1.12 (0.14) | 161.2 (6.1) | 20.0 (2.1) | 37.1 (4.65) | 0.539 (0.003) | 178.6 (1.0) | 0.215 (0.017) | 0.082 (0.010) | 4 |
| DD116 | H | 8.85 (0.54) | 6.32 (0.50) | 3.71 (0.60) | 1.40 (0.28) | 2.62 (0.28) | 1.13 (0.15) | 1.02 (0.14) | 140.0 (6.1) | 15.8 (2.1) | 33.9 (4.65) | 0.469 (0.003) | 155.9 (1.0) | 0.243 (0.017) | 0.087 (0.010) | 4 |
| DD428 | I | 8.48 (0.54) | 5.45 (0.50) | 2.68 (0.60) | 1.03 (0.28) | 2.50 (0.28) | 0.89 (0.15) | 0.79 (0.17) | 127.2 (6.1) | 11.3 (2.1) | 26.4 (4.65) | 0.428 (0.003) | 143.2 (1.01) | 0.276 (0.017) | 0.086 (0.010) | 4 |
| DD109 | I | 8.70 (0.54) | 6.08 (0.50) | 3.39 (0.60) | 1.17 (0.28) | 2.31 (0.28) | 1.06 (0.15) | 0.95 (0.14) | 121.7 (6.2) | 12.9 (2.1) | 31.5 (4.65) | 0.409 (0.003) | 135.9 (1.0) | 0.282 (0.017) | 0.087 (0.010) | 4 |
| DD115 | I | 8.45 (0.54) | 5.55 (0.50) | 2.75 (0.60) | 0.96 (0.28) | 2.35 (0.28) | 0.89 (0.15) | 0.80 (0.14) | 122.8 (6.1) | 11.0 (2.1) | 26.4 (4.65) | 0.415 (0.003) | 138.1 (1.0) | 0.286 (0.017) | 0.086 (0.010) | 4 |
| DD224 | L | 7.43 (0.62) | 4.99 (0.59) | 2.20 (0.70) | 0.95 (0.32) | 2.88 (0.33) | 0.74 (0.18) | 0.65 (0.17) | 142.6 (6.1) | 10.5 (2.4) | 22.0 (5.47) | 0.479 (0.003) | 161.8 (1.2) | 0.290 (0.018) | 0.087 (0.011) | 3 |
| TD187 | L | 8.10 (0.62) | 4.50 (0.59) | 1.76 (0.70) | 0.49 (0.32) | 1.93 (0.33) | 0.63 (0.18) | 0.55 (0.17) | 164.5 (6.2) | 10.3 (2.4) | 18.8 (5.47) | 0.548 (0.003) | 188.0 (1.2) | 0.229 (0.018) | 0.047 (0.010) | 3 |
| DD402 | L | 7.70 (0.62) | 5.03 (0.59) | 2.30 (0.70) | 0.58 (0.32) | 1.71 (0.28) | 0.77 (0.18) | 0.68 (0.17) | 129.6 (6.1) | 10.0 (2.4) | 23.0 (5.47) | 0.432 (0.003) | 147.0 (1.2) | 0.309 (0.018) | 0.057 (0.011) | 3 |
| p> F | ||||||||||||||||
| Clone | 0.345 | 0.017 | 0.020 | 0.016 | 0.157 | 0.017 | 0.017 | <0.001 | 0.006 | <0.001 | <0.001 | <0.001 | 0.001 | 0.041 | ||
| Group | 0.027 | <0.001 | <0.001 | <0.001 | 0.091 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.002 | 0.018 |
| Group | REW | Ψpd | Ψmd | Ψdiff | Gp |
|---|---|---|---|---|---|
| H | Dry | −0.51 (0.07) | −1.52 (0.17) | 1.01 (0.19) | 0.699 (0.269) |
| Wet | −0.28 (0.07) | −1.96 (0.17) | 1.69 (0.19) | 1.040 (0.269) | |
| I | Dry | −0.57 (0.07) | −1.71 (0.17) | 1.13 (0.17) | 0.788 (0.258) |
| Wet | −0.29 (0.07) | −1.90 (0.17) | 1.61 (0.17) | 1.147 (0.258) | |
| L | Dry | −0.650 (0.08) | −1.43 (0.18) | 0.77 (0.19) | 1.695 (0.316) |
| Wet | −0.283 (0.08) | −1.65 (0.18) | 1.37 (0.19) | 1.620 (0.316) | |
| p> F | |||||
| Group (Grp) | 0.615 | 0.116 | 0.083 | 0.022 | |
| Day | <0.001 | 0.006 | <0.001 | 0.370 | |
| Grp×Day | 0.626 | 0.537 | 0.753 | 0.717 |
| Clone | Group | Δ13C Foliage | Δ13C Wood |
|---|---|---|---|
| DM8019 | H | 23.07 (0.41) | 21.63 (0.22) |
| TD185 | H | 22.04 (0.37) | 21.34 (0.19) |
| DD116 | H | 21.65 (0.37) | 21.44 (0.19) |
| DD428 | I | 21.39 (0.37) | 21.54 (0.19) |
| DD109 | I | 21.50 (0.37) | 22.55 (0.19) |
| DD115 | I | 20.63 (0.37) | 21.60 (0.19) |
| DD224 | L | 23.60 (0.37) | 23.26 (0.19) |
| TD187 | L | 21.47 (0.37) | 21.65 (0.19) |
| DD402 | L | 21.86 (0.37) | 22.07 (0.19) |
| p> F | |||
| Clone | <0.0001 | <0.0001 | |
| Group | 0.009 | 0.004 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maier, C.A.; Burley, J.; Cook, R.; Ghezehei, S.B.; Hazel, D.W.; Nichols, E.G. Tree Water Use, Water Use Efficiency, and Carbon Isotope Discrimination in Relation to Growth Potential in Populus deltoides and Hybrids under Field Conditions. Forests 2019, 10, 993. https://doi.org/10.3390/f10110993
Maier CA, Burley J, Cook R, Ghezehei SB, Hazel DW, Nichols EG. Tree Water Use, Water Use Efficiency, and Carbon Isotope Discrimination in Relation to Growth Potential in Populus deltoides and Hybrids under Field Conditions. Forests. 2019; 10(11):993. https://doi.org/10.3390/f10110993
Chicago/Turabian StyleMaier, Christopher A., Joel Burley, Rachel Cook, Solomon B. Ghezehei, Dennis W. Hazel, and Elizabeth G. Nichols. 2019. "Tree Water Use, Water Use Efficiency, and Carbon Isotope Discrimination in Relation to Growth Potential in Populus deltoides and Hybrids under Field Conditions" Forests 10, no. 11: 993. https://doi.org/10.3390/f10110993
APA StyleMaier, C. A., Burley, J., Cook, R., Ghezehei, S. B., Hazel, D. W., & Nichols, E. G. (2019). Tree Water Use, Water Use Efficiency, and Carbon Isotope Discrimination in Relation to Growth Potential in Populus deltoides and Hybrids under Field Conditions. Forests, 10(11), 993. https://doi.org/10.3390/f10110993




