1. Introduction
The boreal ecotype of woodland caribou (
Rangifer tarandus caribou) have evolved to occupy a niche unexploited by other northern ungulates [
1]. Caribou tend to select low-productivity forests where ground lichens are a dominant understory component and have evolved physiological adaptations to consume these lichens as a major component of their diet, particularly in winter [
2,
3,
4]. By frequenting lichen-rich landscapes, caribou can acquire forage and distance themselves from more productive forests which support higher densities of ungulates (e.g., moose (
Alces alces)) and thus predators (e.g., wolves (
Canis lupus) [
1]. However, stand-replacing forest fires are a common occurrence in woodland caribou habitat and because ground lichens are highly flammable, large quantities of lichen are lost in these disturbances [
5]. Since lichens are slow-growing, they take several decades to recover following fire [
6,
7]. Fires therefore create a constantly shifting mosaic of lichen availability across the landscape, which can influence the distribution and habitat selection of woodland caribou [
5,
8].
Given the importance of ground lichens in caribou ecology, it may be useful to map the abundance of ground lichens across caribou ranges for research and/or management purposes. Ground lichens are typically found in mature conifer stands with sparse canopy closure and nitrogen-poor, acidic substrate conditions [
9,
10,
11]. Proxies for the growing conditions preferred by ground lichens can be found in forest inventory layers, which often contain attributes for stand age, soil type, tree species composition and forest structure (e.g., canopy closure). Most forest inventories utilize ecological classification systems to divide the landscape into discrete vegetation communities called ‘ecosites’. Each ecosite is characterized by consistent physical features (soil type, soil depth, nutrient availability) and the resulting vegetation community (trees, shrubs and herbaceous plants) [
12]. Several researchers have used forest inventory layers to predict the occurrence and abundance of ground lichens [
13,
14,
15]. A disadvantage of forest inventory layers is they are generally unavailable for boreal caribou ranges beyond the range of active forest management. In addition, forest inventory layers are typically updated on long time horizons (e.g., 10–20 years) as part of a forest management planning process, which can make it difficult to update lichen abundance maps to reflect changes to ecosite conditions [
13].
Remote sensing has become an essential tool in landscape ecology [
16], particularly due to the availability of Landsat satellite imagery [
17]. Landsat satellites capture images of the Earth’s surface approximately bi-weekly, allowing researchers to update spatial layers as conditions change [
17]. Landsat imagery is composed of several spectral bands that capture different portions of the electromagnetic spectrum. The ground lichens caribou eat contain usnic acid, which produces a unique spectral signature in the blue and short-wave infrared wavelengths [
18]. Being pale in colour, lichens can also be distinguished from green vegetation using the normalized difference vegetation index [
9], which uses the red and near-infrared wavelengths to quantify vegetation greenness (
Appendix C) [
19]. The unique spectral properties of usnic lichens in the near- and short-wave infrared wavelengths led to the incorporation of the normalized difference moisture index (NDMI;
Appendix C) [
20] in several lichen remote sensing studies [
21,
22]. Studies have proven that Landsat spectral properties can be used to obtain reasonable estimates of lichen abundance in northern boreal and tundra systems [
18,
21,
22]. The unique spectral signature of ground lichens can be captured by the moderate spatial resolution of Landsat imagery (30 m pixels) in northern boreal and tundra ecosystems because tree cover is sparse or non-existent [
14]. In the continuous boreal forest, which is characterized by relatively dense tree cover, the unique spectral signature of ground lichens may be masked by the tree canopy [
14]. Higher resolution satellite imagery such as SPOT 6 (6 m pixels) and QuickBird (2.5 m pixels) may be able to capture the unique spectral signature of ground lichens in densely treed areas [
9], but these platforms do not capture the short-wave infrared portion of the electromagnetic spectrum, which has proven useful in modelling lichens in previous studies [
18,
21].
Landscape nutrition models often integrate remote sensing and Geographic Information System (GIS) data (e.g., topography, disturbances, forest structure) to generate spatial predictions of forage abundance from field observations. Such models have been generated for multiple, wide-ranging mammal species, including grizzly bears [
23,
24], elk [
25] and woodland caribou [
26]. Landscape nutrition models can include multiple food types, including seasonally available plant species and prey biomass [
24]. Quantifying forage abundance across the landscape can allow researchers to study the influence of nutrition on survival and fecundity [
25]. Forage layers can also be used to identify potential high-quality habitats to target for protection or areas of overlap between humans and wildlife that present a high risk of conflict [
23]. In conjunction with spatial predictions of predation risk, forage layers can be used to study the tradeoffs between nutrition and predator avoidance experienced by prey species [
26,
27].
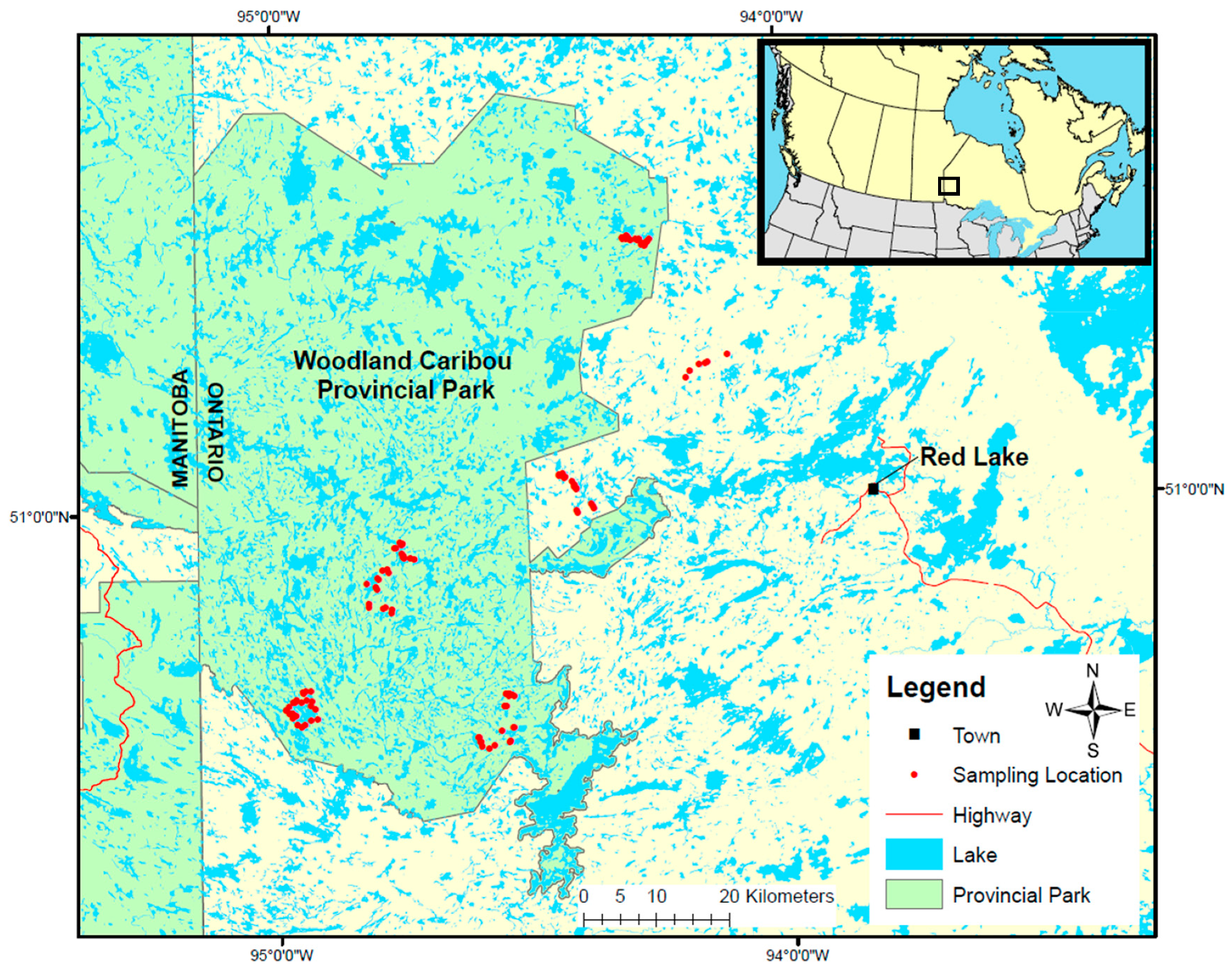
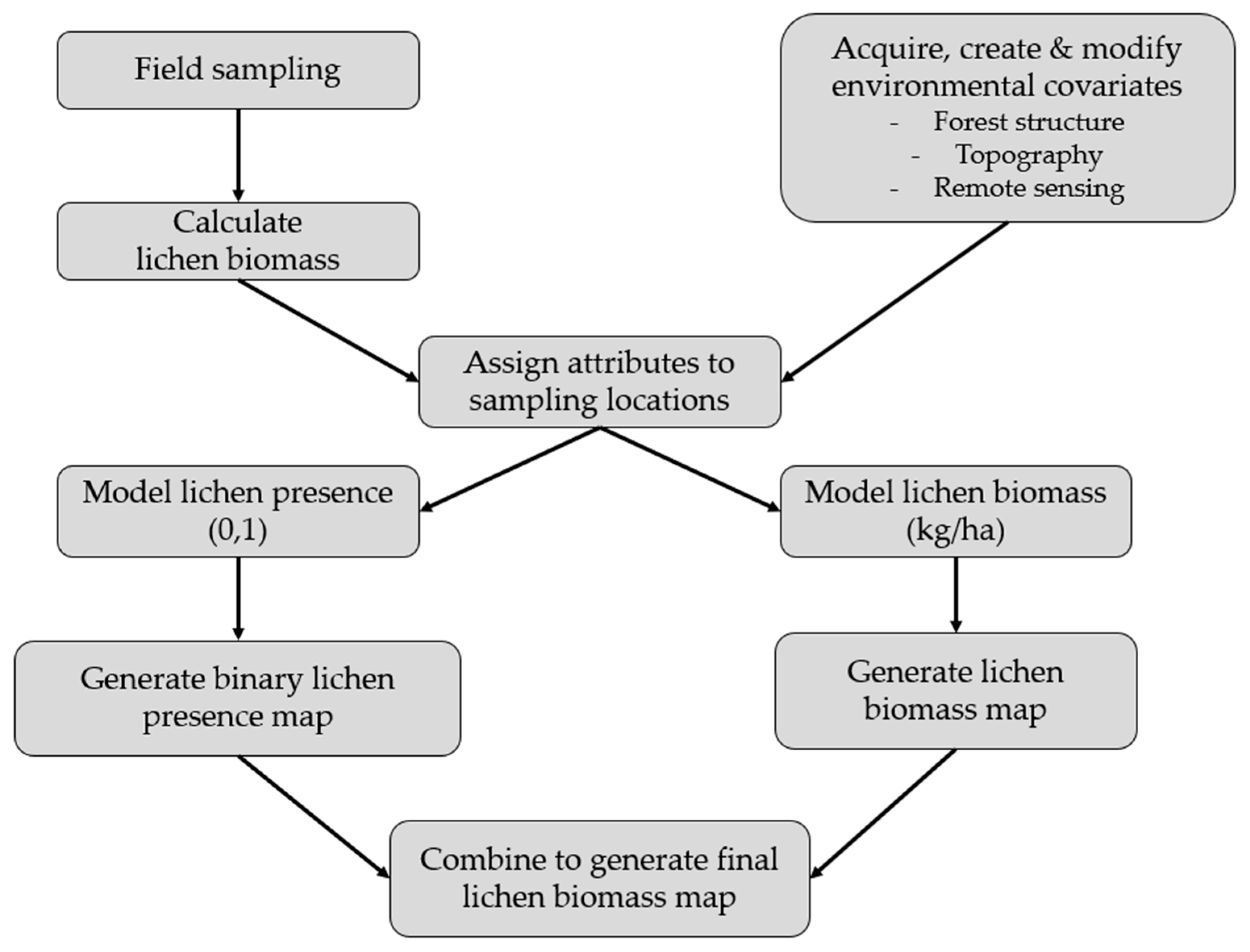
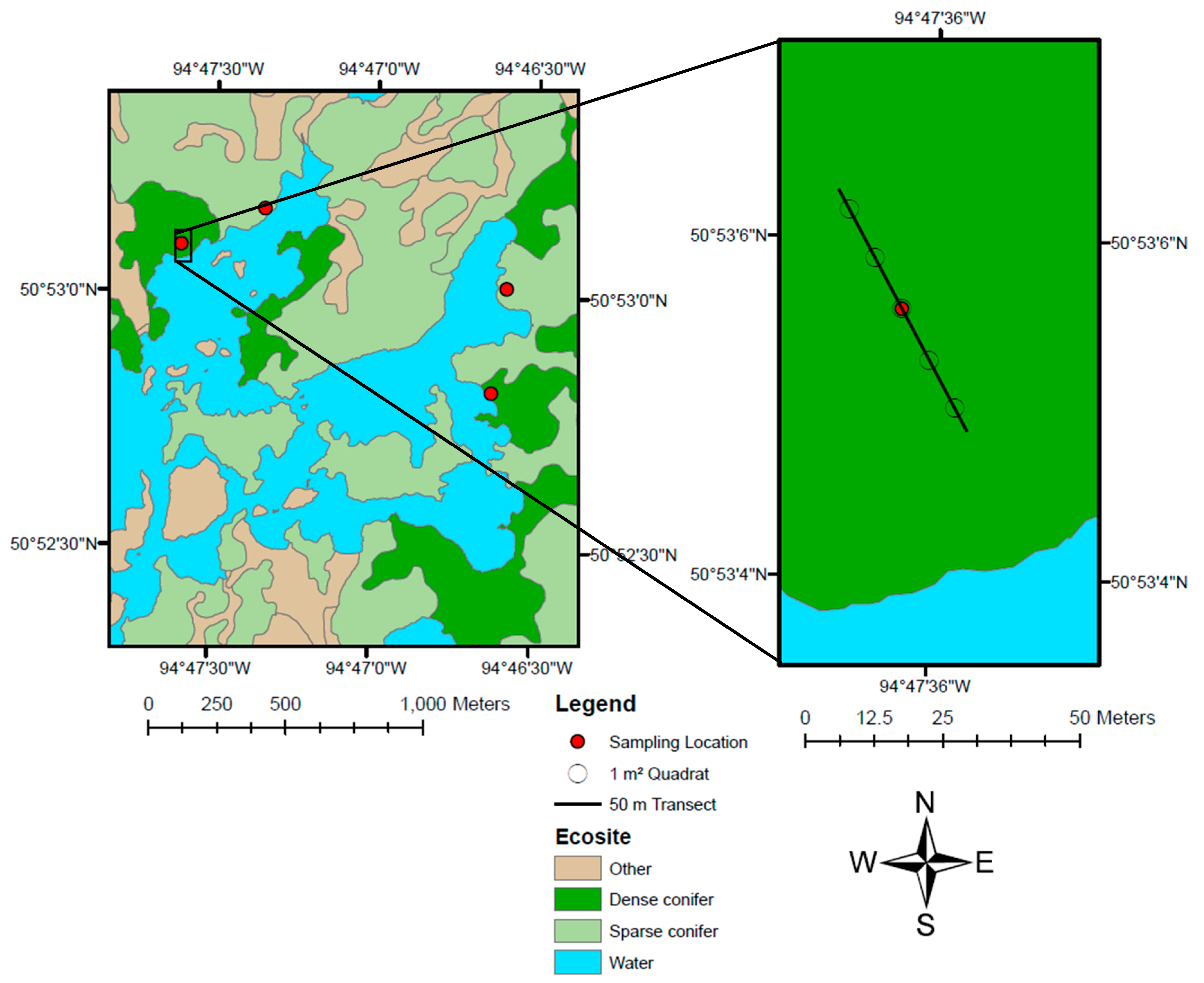
In this study, we create a predictive model of lichen abundance in the boreal forest of Woodland Caribou Provincial Park, in Ontario, Canada and interpolate this model to create a spatial prediction (map) of lichen biomass. We use a regression modelling approach, first conducting field sampling within the study area to parameterize relationships between lichen abundance and environmental conditions (forest type, time-since-fire, canopy closure). We then relate lichen presence and lichen abundance to remote sensing and GIS data and use an a priori model selection procedure to identify the best explanatory variables. We interpolate the top lichen presence and abundance models across the study area and combine them to generate a map predicting lichen biomass (kg/ha). We show that our approach is straightforward and could be applied in other boreal caribou ranges with site-specific field data. Lichen maps could help managers develop more effective conservation strategies for woodland caribou. Managers could use lichen maps to track the availability of this important food resource over time and ensure a constant supply of lichen-rich habitat through resource or fire management planning. Paired with GPS collar locations, lichen maps could be used to identify the quantity of lichen in stands selected by caribou, aiding in the delineation of suitable habitat patches based on available forage resources.
4. Discussion
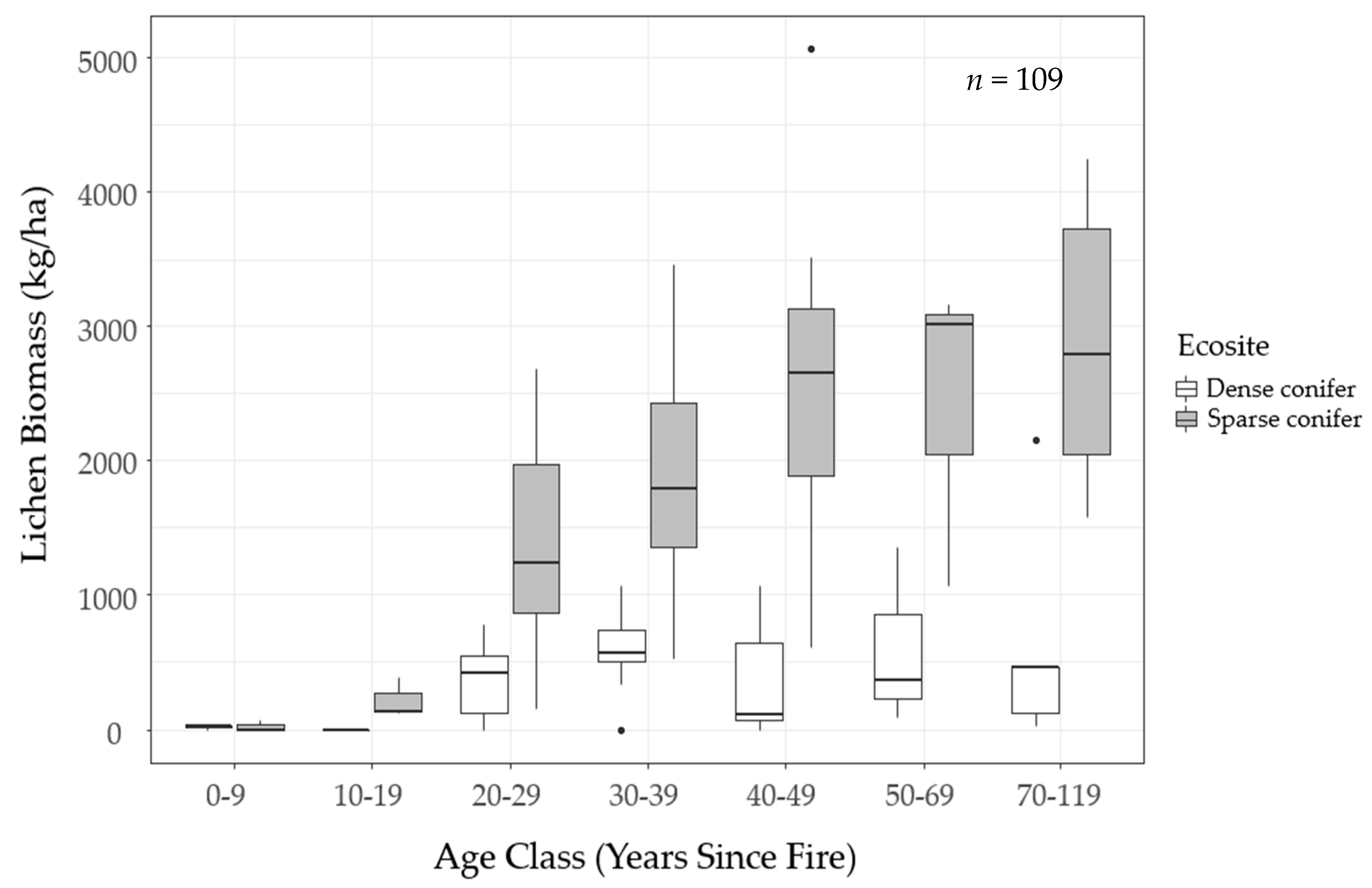
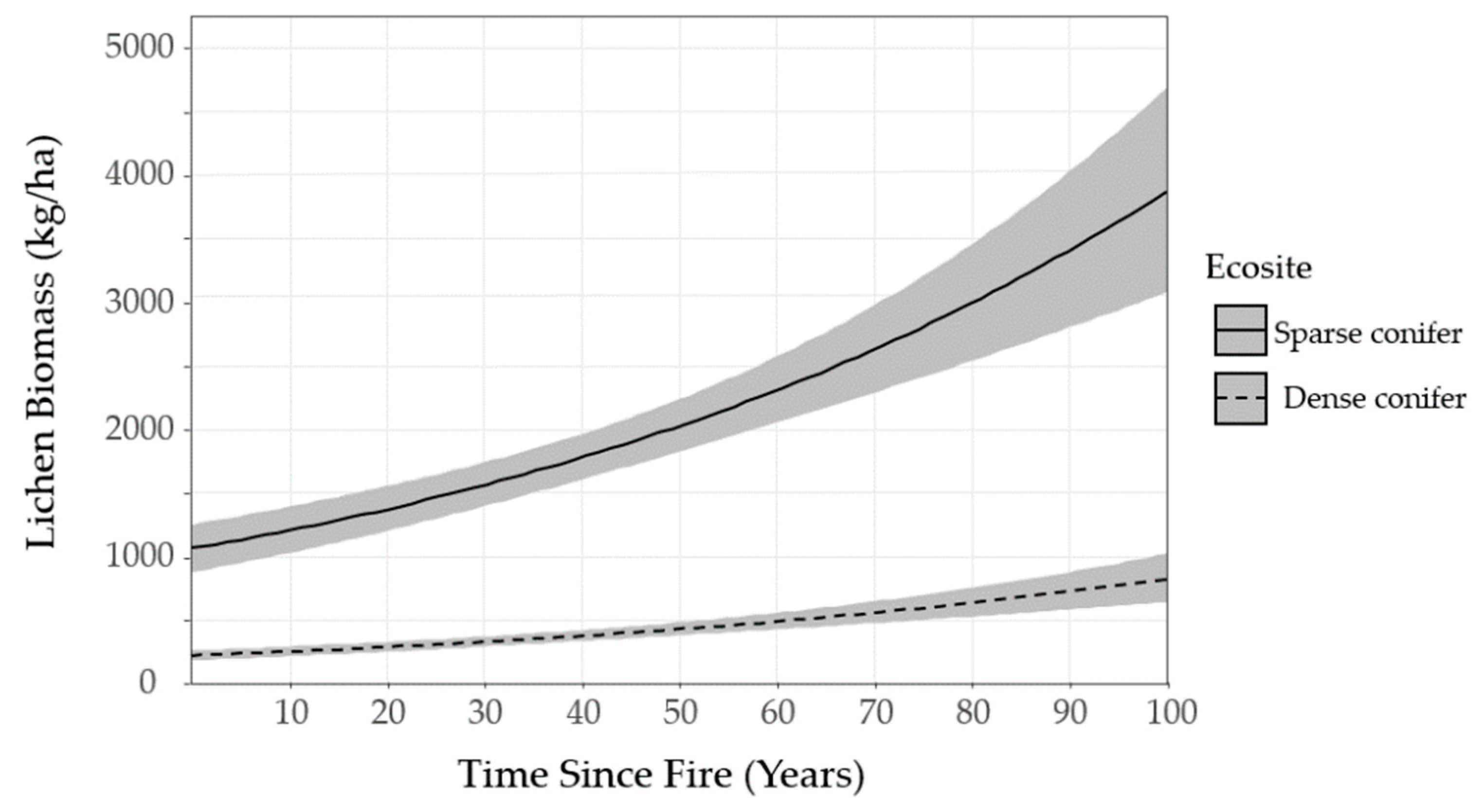
We mapped lichen biomass across Woodland Caribou Provincial Park using a spatial modelling approach that can provide a framework to generate lichen biomass maps for resource management and ecological research in Canada’s boreal forest. By relating our field observations of lichen abundance to forest structure, topographic and remote sensing attributes, we were able to identify environmental features useful in predicting ground lichens. We found that time-since-fire and ecosite were important predictors of ground lichens. Probability of occurrence and biomass of ground lichens was negatively associated with dense conifer ecosites (
Table 5,
Table 7) and such stands demonstrated low lichen abundance (<1000 kg/ha) across the local time-since-fire continuum (
Figure 6). Conversely, sparse conifer ecosites supported very low lichen abundance in the first 20 years after fire, but lichen biomass increased steadily from 20–50 years post-fire (
Figure 5). Mature sparse conifer (≥ 70 years old) supported approximately three times more lichen biomass than dense conifer of the same age (
Figure 6).
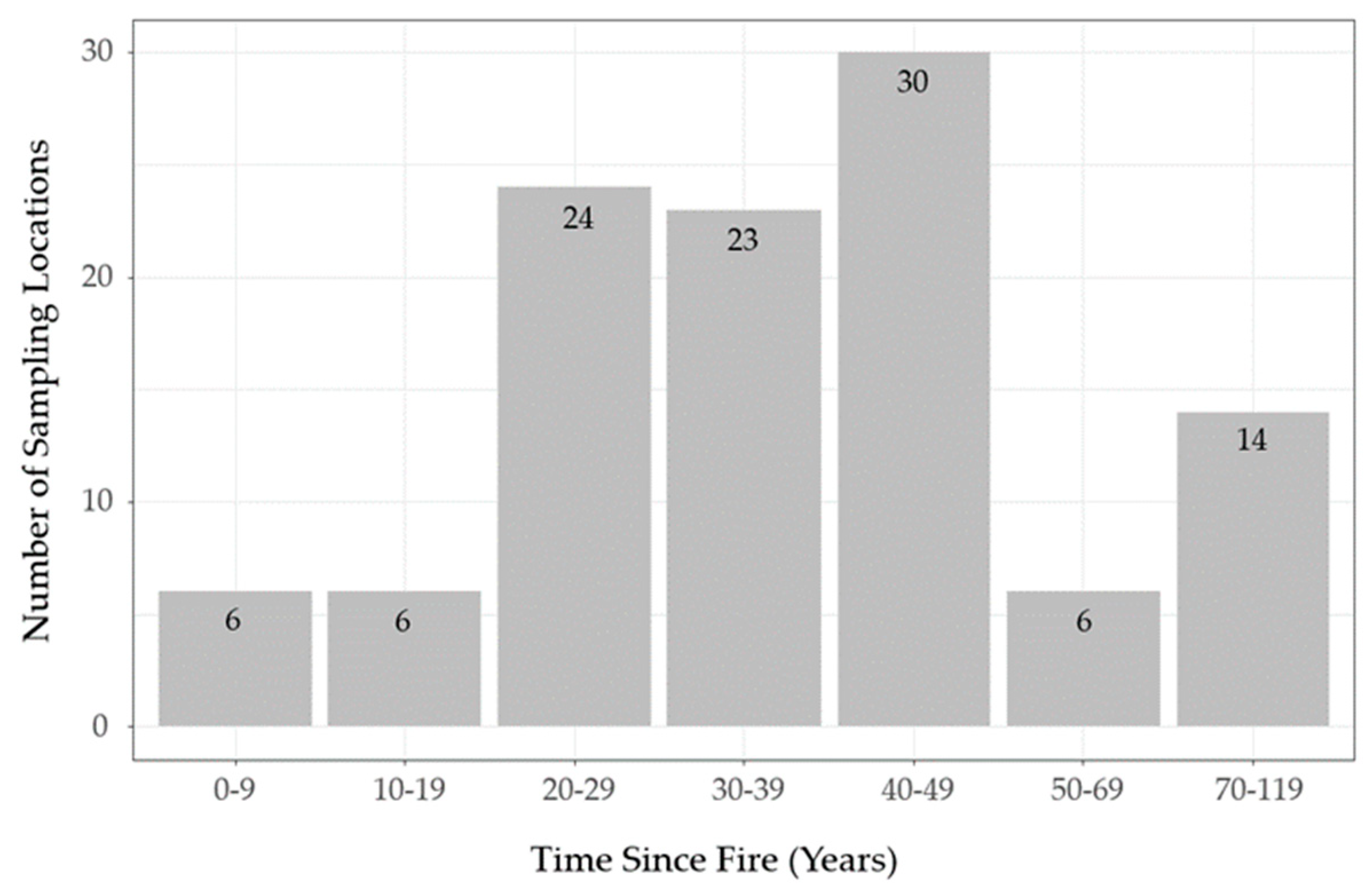
The lichen abundance model appears to overestimate lichen biomass in young sparse conifer stands (0−19 years post-fire;
Figure 6) relative to what was observed in the field (
Figure 5). Similarly, the model appears to exaggerate the accumulation of lichen biomass in older stands (≥ 50 years old). These discrepancies could be due to the unbalanced sampling design we employed, as we focused most of our sampling effort on middle-aged stands due to a secondary objective to test post-fire lichen recovery. This resulted in few observations at the young (0−19 years post-fire,
n = 12) and older (50−119 years post-fire,
n = 20) portions of the local post-fire continuum (
Figure 3). In addition, our field observations suggest lichen biomass may follow a non-linear pattern with time-since-fire in sparse conifer ecosites (
Figure 5). We were unable to fully capture this trend in our analysis because generalized linear models assume a linear relationship between the response variable and the predictor variables. Other model types such as generalized additive models can improve predictions of non-linear trends [
46]. Species distribution models [
46] such as those developed through MaxEnt [
49], provide a highly flexible workflow for mapping the distribution of plants, and have been used to map the presence of lichens [
50]. Future research could incorporate these approaches to generate lichen maps for caribou conservation.
In our study, lichen presence was positively associated with canopy closure (
Table 5). Conversely, lichen biomass was negatively associated with canopy closure (
Table 7). Lichen growth is typically maximized at intermediate levels of canopy closure (~40%) [
51], beyond which the growth of mosses is promoted at the expense of lichens [
6]. Thus, lichens may require a minimum amount canopy closure to be present at a site but experience reduced growth at high levels of canopy closure, perhaps explaining the opposing responses of lichen presence and biomass observed here. In the oldest stands we sampled (70−119 years old), high mortality of mature trees created large gaps in the canopy and increased sun exposure at ground level. This promoted the growth of juniper shrubs (
Juniperus communis L.), which often covered the ground lichens, possibly reducing access to foraging caribou. We had limited observations in overmature conifer stands (
n = 14) and suggest future work should measure lichen biomass and caribou habitat selection in mature (50−70 years old) and overmature stands (≥70 years old) to estimate the optimal renewal period for caribou habitat. This information is essential to develop effective fire response and resource management plans that consider caribou conservation.
Most previous studies quantifying lichen over large areas only used remote sensing [
9,
18,
21,
22] or environmental [
13,
14] data. We anticipated that combining forest structure and topographic attributes with remote sensing attributes would provide the best results. Contrary to our expectations, models with only forest structure and/or topographic attributes were just as predictive as models including remote sensing attributes. The candidate models for both lichen presence and lichen abundance had small differences between AICc scores (∆AIC
C < 2) (
Table 4;
Table 6), which would typically indicate support for multiple candidate models [
52]. However, the best candidate model for lichen presence and lichen abundance was the base model, the most parsimonious of the candidate set, only containing ecosite (i.e., dense conifer vs. sparse conifer), time-since-fire and canopy closure as covariates. The penalty weight assigned by AIC
c to more complex models [
52] indicates that the additional topographic and remote sensing covariates did not improve explanatory power over the base model.
The lack of support for candidate models with remote sensing covariates could arise from multiple sources. First, environmental and remote sensing covariates are often correlated. We controlled for collinearity within models but, because we were interested in predicting lichen abundance rather than inferring ecological relationships, we did not account for correlation amongst models. Second, the coarse spatial resolution of Landsat imagery can cause trees to mask the spectral signature of ground lichens [
14]. Keim et al. (2017) [
9] report an
R2 = 0.74 for a lichen map generated using QuickBird satellite imagery (2.5 m pixels) and LiDAR data (1 m pixels). They found that QuickBird imagery predicted lichens better than SPOT (6 m pixels) and Landsat (30 m pixels) imagery in their study area in the continuous boreal forest of northeastern Alberta [
9]. We suggest that the continued incorporation of finer resolution satellite [
9] or UAV imagery [
53] may help improve the accuracy of lichen mapping in years to come.
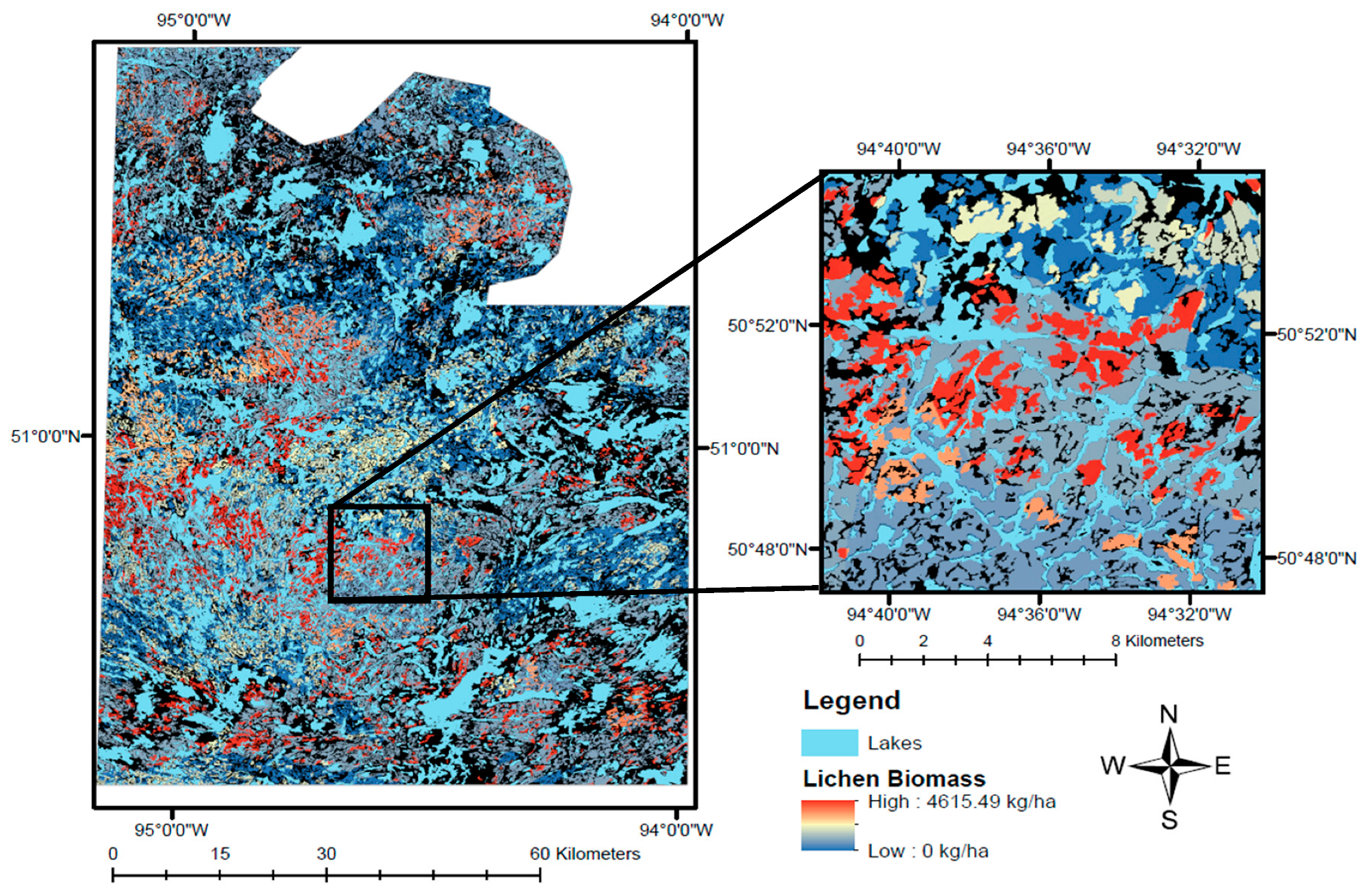
The lichen abundance map we generated in this study (
Figure 7) highlights the patchy distribution of lichens on the landscape, which is driven primarily by the prevalence of fire in the study area. Lichen-rich forest is relatively restricted on the landscape, only occurring in mature, sparse conifer ecosites (≥50 years post-fire), where biomass often exceeds 3000 kg/ha (
Figure 5). Historically, many researchers used habitat type as a proxy for lichen abundance [
54,
55,
56]. However, some studies have explicitly measured lichen availability and suggest caribou preferentially select stands with ≥3000 kg/ha of ground lichens as winter habitat [
8,
57,
58]. Given that animal nutrition is necessarily related to the amount of available food, ecologists and land managers should strive to understand caribou’s use of lichen biomass across time and space. In particular, identifying the use of lichen biomass during different seasons could be used to delineate nutritionally important habitat patches.
The relatively low accuracy of our map (
R2 = 0.39) is unsurprising given we used relatively coarse spatial covariates (30 m pixels) to model the presence and abundance of lichens, which are responding to environmental conditions at a very small scale (i.e., microsite). However, we feel that our lichen map provides a reasonable estimation of lichen biomass across our study area and suggest our modelling approach provides a useful framework for researchers to apply and improve in future lichen mapping projects. Most previous research mapped lichen cover [
9,
13,
18], but we suggest lichen biomass is more biologically relevant than lichen cover as biomass is more closely related to animal energetics and fitness [
26]. We stress the importance of validating biomass conversion factors and landscape covariates for new study areas, as growing conditions for lichen may vary. For example, in northern Alberta, peatlands are a dominant landscape feature. Previous studies indicate peatlands support much lower lichen abundance than upland sites [
35], however raised ‘islands’ of drier peat within bogs can provide better conditions for lichen growth and support locally abundant ground lichens [
9,
11]. In other parts of the boreal forest, sandy areas dominated by jack pine support thick mats of ground lichens [
33]. Integrating abiotic information, such as substrate type, groundwater depth and terrain ruggedness into spatial models may improve lichen predictive mapping, especially when the study area spans multiple biophysical regions. We incorporated data from numerous sources, data types and spatial resolutions to map the abundance of lichens in our study (
Table 2). Researchers must be cognizant of the vintage of the source data in each layer they incorporate in their modelling framework to ensure temporal consistency. We suggest future research should focus on incorporating multiple sources of information, including time-since-fire and attributes derived from high resolution satellite imagery (e.g., spectral values, landcover type, forest structure [
59]). This will improve spatio-temporal consistency and repeatability. We also encourage researchers to ensure they are selecting the most appropriate model for predicting the distribution of lichens and suggest utilizing generalized additive models [
48] may be of particular utility to address some of the deficiencies of this study. Once a lichen abundance map has been generated, we encourage researchers to conduct independent validation using additional field sampling to improve certainty in their spatial predictions.