Carbon Pools in Old-Growth Scots Pine Stands in Hemiboreal Latvia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fieldwork
2.3. Data Analysis
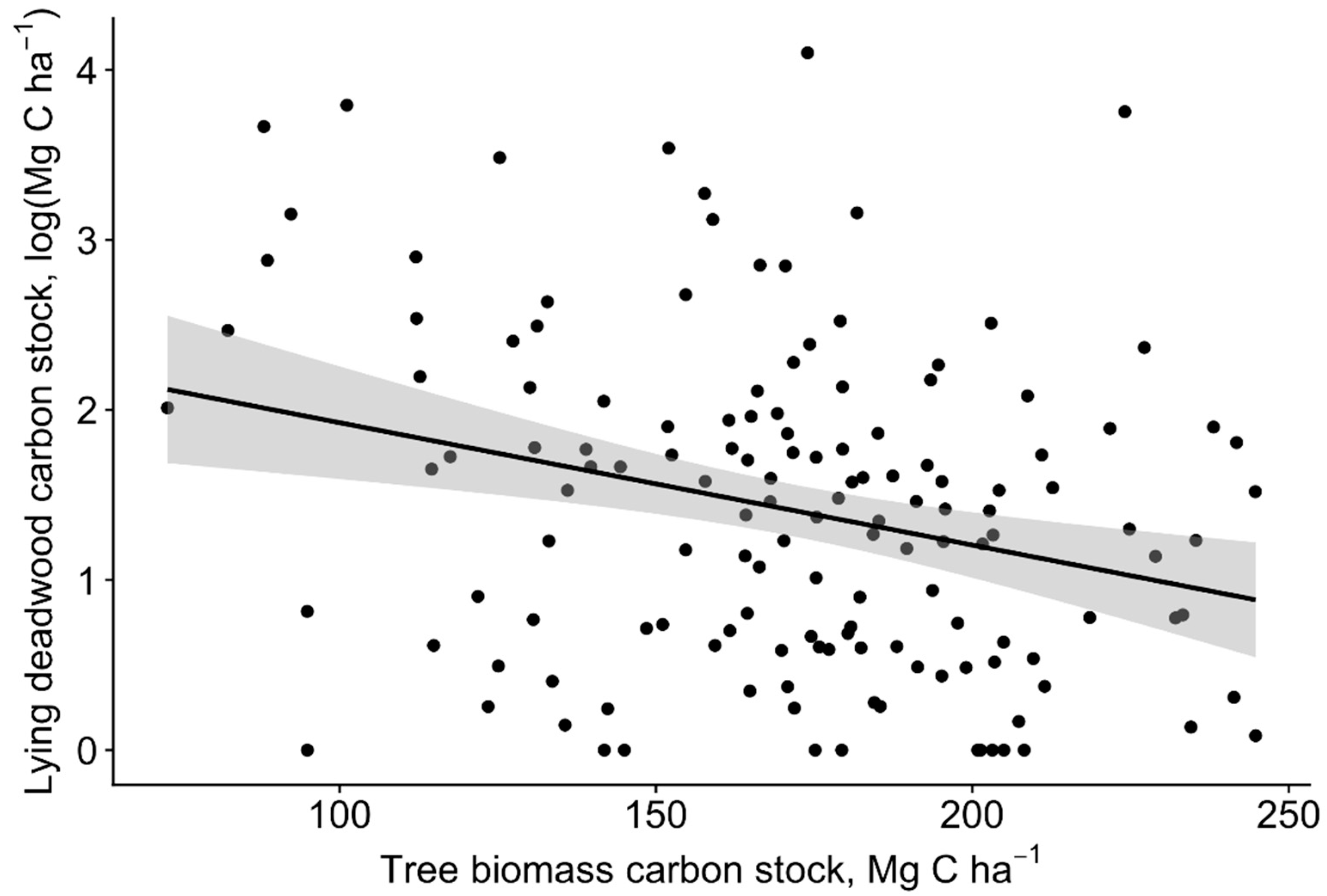
3. Results
3.1. Volume
3.2. Carbon Pools
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Park, A.; Puettmann, K.; Wilson, E.; Messier, C.; Kames, S.; Dhar, A. Can boreal and temperate forest management be adapted to the uncertainties of 21st century climate change? Crit. Rev. Plant Sci. 2014, 33, 251–285. [Google Scholar] [CrossRef]
- Birdsey, R.; Pan, Y. Trends in management of the world’s forests and impacts on carbon stocks. For. Ecol. Manag. 2015, 355, 83–90. [Google Scholar] [CrossRef]
- Bradshaw, C.J.A.; Warkentin, I.G. Global estimates of boreal forest carbon stocks and flux. Glob. Planet. Chang. 2015, 128, 24–30. [Google Scholar] [CrossRef]
- Luyssaert, S.; Schulze, E.D.; Börner, A.; Knohl, A.; Hessenmöller, D.; Law, B.E.; Ciais, P.; Grace, J. Old-growth forests as global carbon sinks. Nature 2008, 455, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Angst, G.; Messinger, J.; Greiner, M.; Häusler, W.; Hertel, D.; Kirfel, K.; Kögel-Knabner, I.; Leuschner, C.; Rethemeyer, J.; Mueller, C.W. Soil organic carbon stocks in topsoil and subsoil controlled by parent material, carbon input in the rhizosphere, and microbial-derived compounds. Soil Biol. Biochem. 2018, 122, 19–30. [Google Scholar] [CrossRef]
- Bond-Lamberty, B.; Peckham, S.D.; Ahl, D.E.; Gower, S.T. The dominance of fire in determining carbon balance of the central Canadian boreal forest. Nature 2007, 450, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Nord-Larsen, T.; Vesterdal, L.; Bentsen, N.S.; Larsen, J.B. Ecosystem carbon stocks and their temporal resilience in a semi-natural beech-dominated forest. For. Ecol. Manag. 2019, 447, 67–76. [Google Scholar] [CrossRef]
- Wei, Y.; Li, M.; Chen, H.; Lewis, B.J.; Yu, D.; Zhou, L.; Zhou, W.; Fang, X.; Zhao, W.; Dai, L. Variation in carbon storage and its distribution by stand age and forest type in boreal and temperate forests in northeastern China. PLoS ONE 2013, 8, e72201. [Google Scholar] [CrossRef]
- Uri, V.; Kukumägi, M.; Aosaar, J.; Varik, M.; Becker, H.; Aun, K.; Krasnova, A.; Morozov, G.; Ostonen, I.; Mander, Ü.; et al. The carbon balance of a six-year-old Scots pine (Pinus sylvestris L.) ecosystem estimated by different methods. For. Ecol. Manag. 2019, 433, 248–262. [Google Scholar] [CrossRef]
- Liski, J.; Lehtonen, A.; Palosuo, T.; Peltoniemi, M.; Eggers, T.; Muukkonen, P.; Mäkipää, R. Carbon accumulation in Finland’s forests 1922–2004#x2014;An estimate obtained by combination of forest inventory data with modelling of biomass, litter and soil. Ann. For. Sci. 2006, 63, 687–697. [Google Scholar] [CrossRef]
- Jandl, R.; Lindner, M.; Vesterdal, L.; Bauwens, B.; Baritz, R.; Hagedorn, F.; Johnson, D.W.; Minkkinen, K.; Byrne, K.A. How strongly can forest management influence soil carbon sequestration? Geoderma 2007, 137, 253–268. [Google Scholar] [CrossRef]
- Bravo-Oviedo, A.; Ruiz-Peinado, R.; Modrego, P.; Alonso, R.; Montero, G. Forest thinning impact on carbon stock and soil condition in Southern European populations of P. sylvestris L. For. Ecol. Manag. 2015, 357, 259–267. [Google Scholar] [CrossRef]
- Moreno-Fernández, D.; Díaz-Pinés, E.; Barbeito, I.; Sánchez-González, M.; Montes, F.; Rubio, A.; Cañellas, I. Temporal carbon dynamics over the rotation period of two alternative management systems in Mediterranean mountain Scots pine forests. For. Ecol. Manag. 2015, 348, 186–195. [Google Scholar] [CrossRef]
- Jacob, M.; Bade, C.; Calvete, H.; Dittrich, S.; Leuschner, C.; Hauck, M. Significance of over-mature and decaying trees for carbon stocks in a Central European natural spruce forest. Ecosystems 2013, 16, 336–346. [Google Scholar] [CrossRef]
- Seedre, M.; Shrestha, B.M.; Chen, H.Y.H.; Colombo, S.; Jõgiste, K. Carbon dynamics of North American boreal forest after stand replacing wildfire and clearcut logging. J. For. Res. 2011, 16, 168–183. [Google Scholar] [CrossRef]
- Finér, L.; Mannerkoski, H.; Piirainen, S.; Starr, M. Carbon and nitrogen pools in an old-growth, Norway spruce mixed forest in eastern Finland and changes associated with clear-cutting. For. Ecol. Manag. 2003, 174, 51–63. [Google Scholar] [CrossRef]
- Sabatini, F.M.; Burrascano, S.; Keeton, W.S.; Levers, C.; Lindner, M.; Pötzschner, F.; Verkerk, P.J.; Bauhus, J.; Buchwald, E.; Chaskovsky, O.; et al. Where are Europe’s last primary forests? Divers. Distrib. 2018, 24, 1426–1439. [Google Scholar] [CrossRef]
- Kitenberga, M.; Drobyshev, I.; Elferts, D.; Matisons, R.; Adamovics, A.; Katrevics, J.; Niklasson, M.; Jansons, A. A mixture of human and climatic effects shapes the 250-year long fire history of a semi-natural pine dominated landscape of Northern Latvia. For. Ecol. Manag. 2019, 441, 192–201. [Google Scholar] [CrossRef]
- Seedre, M.; Taylor, A.R.; Brassard, B.W.; Chen, H.Y.H.; Jõgiste, K. Recovery of ecosystem carbon stocks in young boreal forests: A comparison of harvesting and wildfire disturbance. Ecosystems 2014, 17, 851–863. [Google Scholar] [CrossRef]
- Taylor, A.R.; Seedre, M.; Brassard, B.W.; Chen, H.Y.H. Decline in net ecosystem productivity following canopy transition to late-succession forests. Ecosystems 2014, 17, 778–791. [Google Scholar] [CrossRef]
- Ķēniņa, L.; Elferts, D.; Bāders, E.; Jansons, Ā. Carbon pools in a hemiboreal over-mature Norway Spruce stands. Forests 2018, 9, 435. [Google Scholar] [CrossRef]
- Seedre, M.; Kopáček, J.; Janda, P.; Bače, R.; Svoboda, M. Carbon pools in a montane old-growth Norway spruce ecosystem in Bohemian Forest: Effects of stand age and elevation. For. Ecol. Manag. 2015, 346, 106–113. [Google Scholar] [CrossRef]
- Kranabetter, J.M. Site carbon storage along productivity gradients of a late-seral southern boreal forest. Can. J. For. Res. 2008, 39, 1053–1060. [Google Scholar] [CrossRef]
- Zhou, G.; Liu, S.; Li, Z.; Zhang, D.; Tang, X.; Zhou, C.; Yan, J.; Mo, J. Old-growth forests can accumulate carbon in soils. Science 2006, 314, 1417. [Google Scholar] [CrossRef]
- Shanin, V.; Komarov, A.; Mäkipää, R. Tree species composition affects productivity and carbon dynamics of different site types in boreal forests. Eur. J. For. Res. 2014, 133, 273–286. [Google Scholar] [CrossRef][Green Version]
- Ahti, T.; Hämet-Ahti, L.; Jalas, J. Vegetation zones and their sections in north-western Europe. Ann. Bot. Fenn. 1968, 5, 169–211. [Google Scholar]
- Schelhaas, M.J.; Nabuurs, G.J.; Hengeveld, G.; Reyer, C.; Hanewinkel, M.; Zimmermann, N.E.; Cullmann, D. Alternative forest management strategies to account for climate change-induced productivity and species suitability changes in Europe. Reg. Environ. Chang. 2015, 15, 1581–1594. [Google Scholar] [CrossRef]
- Triviño, M.; Pohjanmies, T.; Mazziotta, A.; Juutinen, A.; Podkopaev, D.; Le Tortorec, E.; Mönkkönen, M. Optimizing management to enhance multifunctionality in a boreal forest landscape. J. Appl. Ecol. 2017, 54, 61–70. [Google Scholar] [CrossRef]
- Jansons, A.; Matisons, R.; Zadina, M.; Sisenis, L.; Jansons, J. The effect of climatic factors on height increment of Scots pine in sites differing by continentality in Latvia. Silva Fenn. 2015, 49, 14. [Google Scholar] [CrossRef]
- Bušs, K. Fundamentals of Forest Classification in Latvia SSR; LRZTIPI: Rīga, Latvia, 1976; p. 24. (In Latvian) [Google Scholar]
- Köster, K.; Metslaid, M.; Engelhart, J.; Köster, E. Dead wood basic density, and the concentration of carbon and nitrogen for main tree species in managed hemiboreal forests. For. Ecol. Manag. 2015, 354, 35–42. [Google Scholar] [CrossRef]
- Fleck, S.; Cools, N.; De Vos, B.; Meesenburg, H.; Fischer, R. The Level II aggregated forest soil condition database links soil physicochemical and hydraulic properties with long-term observations of forest condition in Europe. Ann. For. Sci. 2016, 73, 945–957. [Google Scholar] [CrossRef]
- Sharma, R.P.; Vacek, Z.; Vacek, S. Nonlinear mixed effect height-diameter model for mixed species forests in the central part of the Czech Republic. J. For. Sci. 2016, 62, 470–484. [Google Scholar] [CrossRef]
- Liepa, I. Tree Growth Study; LLU: Jelgava, Latvia, 1996; p. 31. (In Latvian) [Google Scholar]
- Liepiņš, J.; Lazdiņš, A.; Liepiņš, K. Equations for estimating above- and belowground biomass of Norway spruce, Scots pine, birch spp. and European aspen in Latvia. Scand. J. For. Res. 2018, 33, 58–70. [Google Scholar] [CrossRef]
- Thomas, S.C.; Martin, A.R. Carbon content of tree tissues: A synthesis. Forests 2012, 3, 332–352. [Google Scholar] [CrossRef]
- Neumann, M.; Moreno, A.; Mues, V.; Härkönen, S.; Mura, M.; Bouriaud, O.; Lang, M.; Achten, W.M.J.; Thivolle-Cazat, A.; Bronisz, K.; et al. Comparison of carbon estimation methods for European forests. For. Ecol. Manag. 2016, 361, 397–420. [Google Scholar] [CrossRef]
- Baritz, R.; Seufert, G.; Montanarella, L.; van Ranst, E. Carbon concentrations and stocks in forest soils of Europe. For. Ecol. Manag. 2010, 260, 262–277. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Pregitzer, K.S.; Euskirchen, E.S. Carbon cycling and storage in world forests: Biome patterns related to forest age. Glob. Chang. Biol. 2004, 10, 2052–2077. [Google Scholar] [CrossRef]
- Lee, J.; Tolunay, D.; Makineci, E.; Çömez, A.; Son, Y.M.; Kim, R.; Son, Y. Estimating the age-dependent changes in carbon stocks of Scots pine (Pinus sylvestris L.) stands in Turkey. Ann. For. Sci. 2016, 73, 523–531. [Google Scholar] [CrossRef]
- Kriiska, K.; Frey, J.; Asi, E.; Kabral, N.; Uri, V.; Aosaar, J.; Varik, M.; Napa, Ü.; Apuhtin, V.; Timmusk, T.; et al. Variation in annual carbon fluxes affecting the SOC pool in hemiboreal coniferous forests in Estonia. For. Ecol. Manag. 2019, 433, 419–430. [Google Scholar] [CrossRef]
- Krankina, O.N.; Harmon, M.E. Dynamics of the dead wood carbon pool in northwestern Russian boreal forests. Water Air Soil Pollut. 1995, 82, 227–238. [Google Scholar] [CrossRef]
- Rouvinen, S.; Kuuluvainen, T.; Siitonen, J. Tree mortality in a Pinus sylvestris dominated boreal forest landscape in Vienansalo wilderness, eastern Fennoscandia. Silva Fenn. 2002, 36, 554. [Google Scholar] [CrossRef]
- Kuuluvainen, T.; Aakala, T.; Várkonyi, G. Dead standing pine trees in a boreal forest landscape in the Kalevala National Park, northern Fennoscandia: Amount, population characteristics and spatial pattern. For. Ecosyst. 2017, 4. [Google Scholar] [CrossRef]
- Lutter, R.; Kõlli, R.; Tullus, A.; Tullus, H. Ecosystem carbon stocks of Estonian premature and mature managed forests: Effects of site conditions and overstorey tree species. Eur. J. For. Res. 2019, 138, 125–142. [Google Scholar] [CrossRef]
- Callesen, I.; Liski, J.; Raulund-Rasmussen, K.; Olsson, M.T.; Tau-Strand, L.; Vesterdal, L.; Westman, C.J. Soil carbon stores in Nordic well-drained forest soils relationships with climate and texture class. Glob. Chang. Biol. 2003, 9, 358–370. [Google Scholar] [CrossRef]
- Krasnova, A.; Kukumägi, M.; Mander, Ü.; Torga, R.; Krasnov, D.; Noe, M.; Ostonen, I.; Püttsepp, Ü.; Killian, H.; Uri, V.; et al. Carbon exchange in a hemiboreal mixed forest in relation to tree species composition. Agric. For. Meteorol. 2019, 275, 11–23. [Google Scholar] [CrossRef]
- Martin, M.; Fenton, N.; Morin, H. Structural diversity and dynamics of boreal old-growth forests case study in Eastern Canada. For. Ecol. Manag. 2018, 422, 125–136. [Google Scholar] [CrossRef]
- Ciais, P.; Janssens, I.; Shvidenko, A.; Wirth, C.; Malhi, Y.; Grace, J.; Schulze, E.D.; Heimann, M.; Phillips, O.; Dolman, A.J. The potential for rising CO2 to account for the observed uptake of carbon by tropical, temperate and boreal forest biomes. In The Carbon Balance of Forest Biomes; Griffith, H., Jarvis, P., Eds.; Taylor & Francis: London, UK, 2005; pp. 109–150. [Google Scholar]


| Carbon Pool | Mean ± CI | % |
|---|---|---|
| Above-ground tree biomass | 135.8 ± 4.9 | 47 |
| Below-ground tree biomass | 35.4 ± 1.2 | 12 |
| Living tree biomass (n = 143) | 171.2 ± 6.1 | 59 |
| Lying deadwood | 5.9 ± 1.5 | 2 |
| Standing dead trees | 7.1 ± 1.2 | 2 |
| Snags | 1.8 ± 0.4 | 1 |
| Total deadwood (n = 143) | 14.8 ± 2.1 | 5 |
| Forest floor (n = 18) | 15.2 ± 7.1 | 5 |
| Mineral soil (0–80 cm; n = 18) | 90.0 ± 38.9 | 31 |
| Total ecosystem carbon1 | 291.2 ± 54.2 | 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ķēniņa, L.; Jaunslaviete, I.; Liepa, L.; Zute, D.; Jansons, Ā. Carbon Pools in Old-Growth Scots Pine Stands in Hemiboreal Latvia. Forests 2019, 10, 911. https://doi.org/10.3390/f10100911
Ķēniņa L, Jaunslaviete I, Liepa L, Zute D, Jansons Ā. Carbon Pools in Old-Growth Scots Pine Stands in Hemiboreal Latvia. Forests. 2019; 10(10):911. https://doi.org/10.3390/f10100911
Chicago/Turabian StyleĶēniņa, Laura, Ieva Jaunslaviete, Līga Liepa, Daiga Zute, and Āris Jansons. 2019. "Carbon Pools in Old-Growth Scots Pine Stands in Hemiboreal Latvia" Forests 10, no. 10: 911. https://doi.org/10.3390/f10100911
APA StyleĶēniņa, L., Jaunslaviete, I., Liepa, L., Zute, D., & Jansons, Ā. (2019). Carbon Pools in Old-Growth Scots Pine Stands in Hemiboreal Latvia. Forests, 10(10), 911. https://doi.org/10.3390/f10100911






