Effect of Herbaceous Layer Interference on the Post-Fire Regeneration of a Serotinous Pine (Pinus pinaster Aiton) across Two Seedling Ages
Abstract
1. Introduction
2. Material and Methods
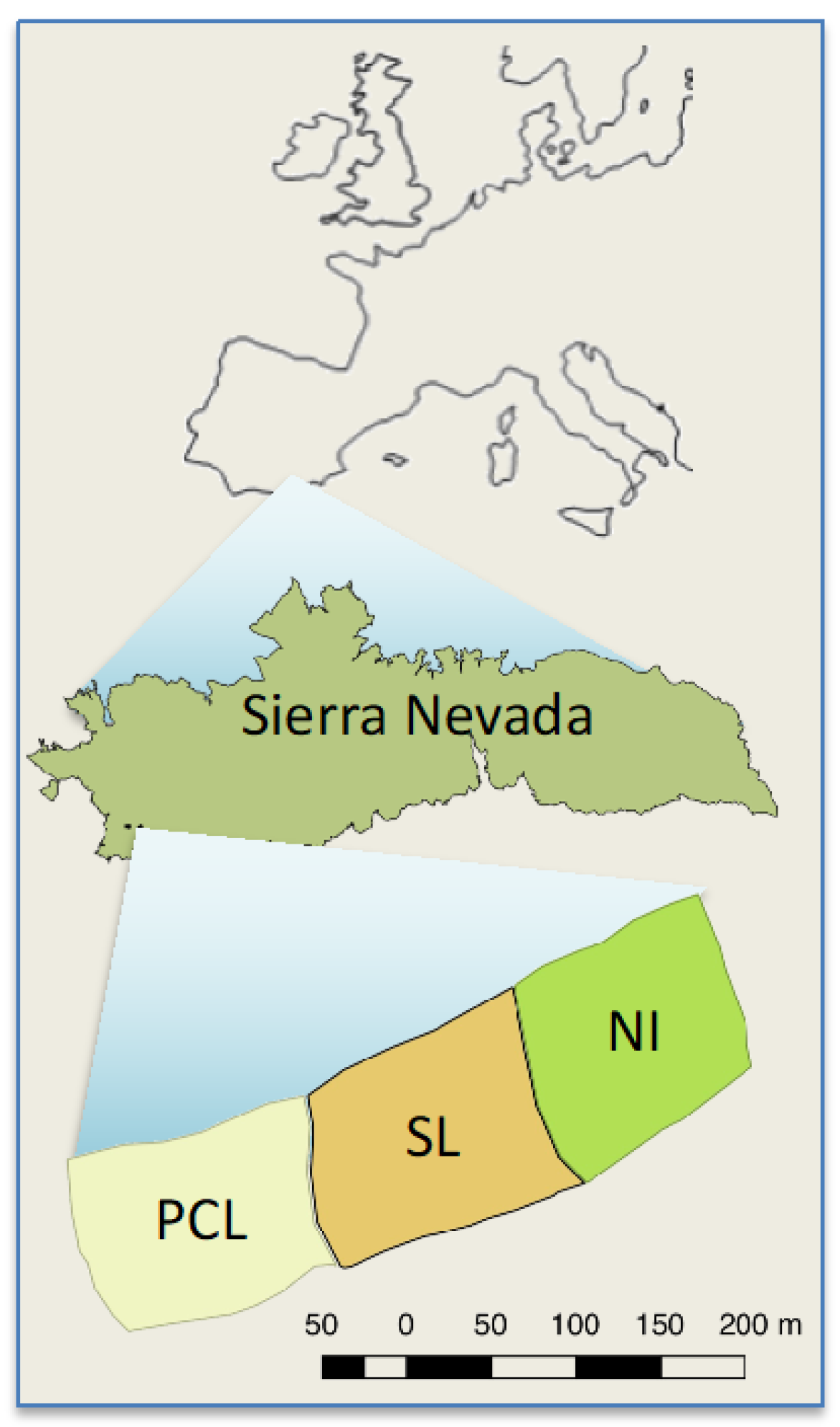
2.1. Study Site and Species
2.2. Experimental Design
- “Non-intervention” (NI), where no post-fire intervention was conducted and all burned trees were left standing.
- “Partial cut plus lopping” (PCL), generated by the felling of about 90% of the trees, with the main branches also lopped off, but leaving all the cut biomass (boles and branches) in situ on the ground. After treatment application, felled logs and branches covered 45% of the surface at 0–10 cm from the ground, 61% at 11–50 cm, and 9% at 51–100 cm [28].
- “Salvage logging” (SL), where trees were felled and limbed with the use of chainsaws. Woody debris was masticated using a tractor, and trunks were manually piled in groups of 10–15. The local Forest Service planned to remove the piled trunks with a log forwarder in this SL treatment, but this step was later cancelled due to difficulties in precisely operating machinery within the spatial arrangement of the plots.
2.3. Effect of Herbaceous Cover on Pine Seedling Survival and Growth
2.4. Data Analysis
3. Results
3.1. Initial Conditions
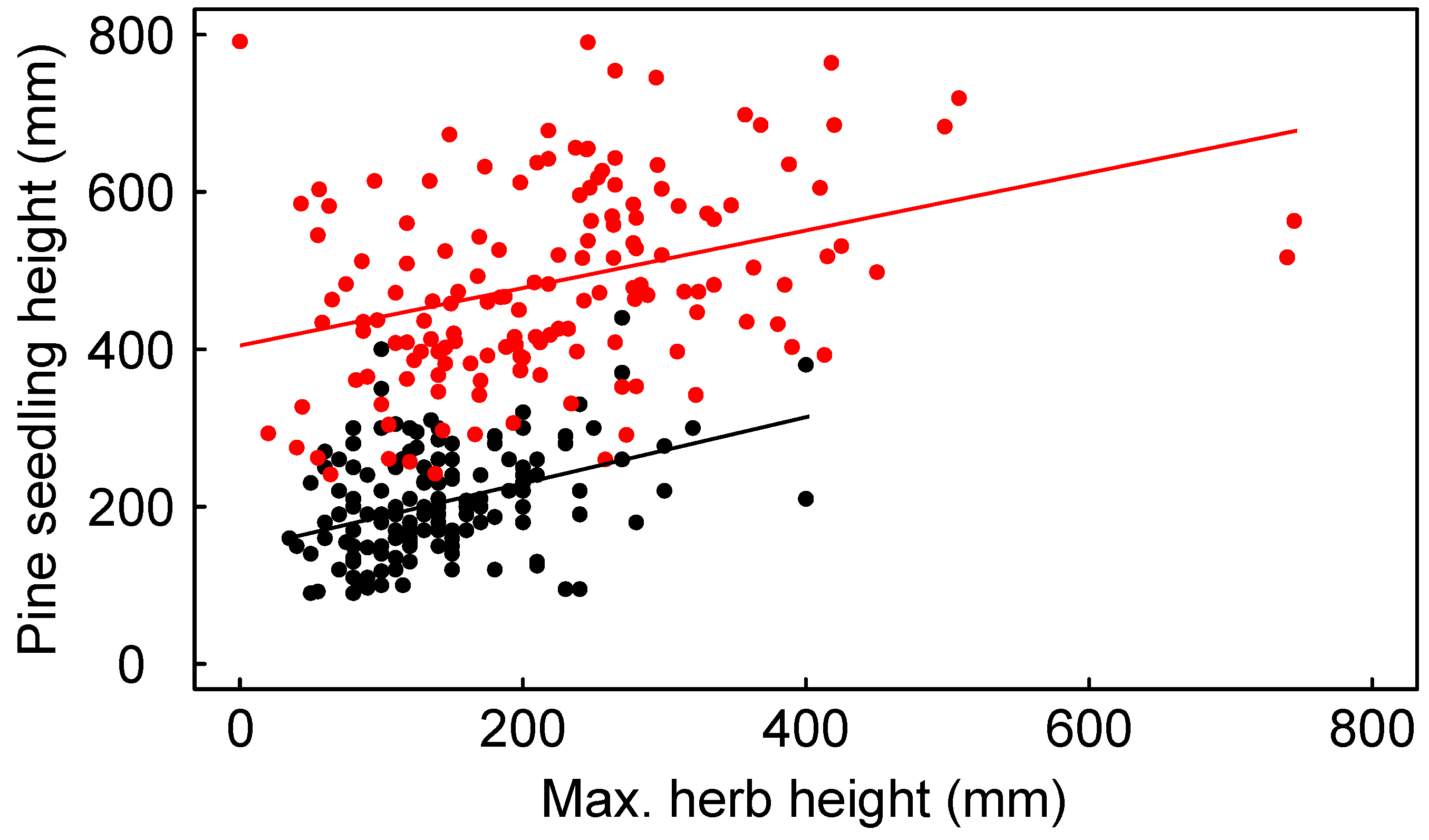
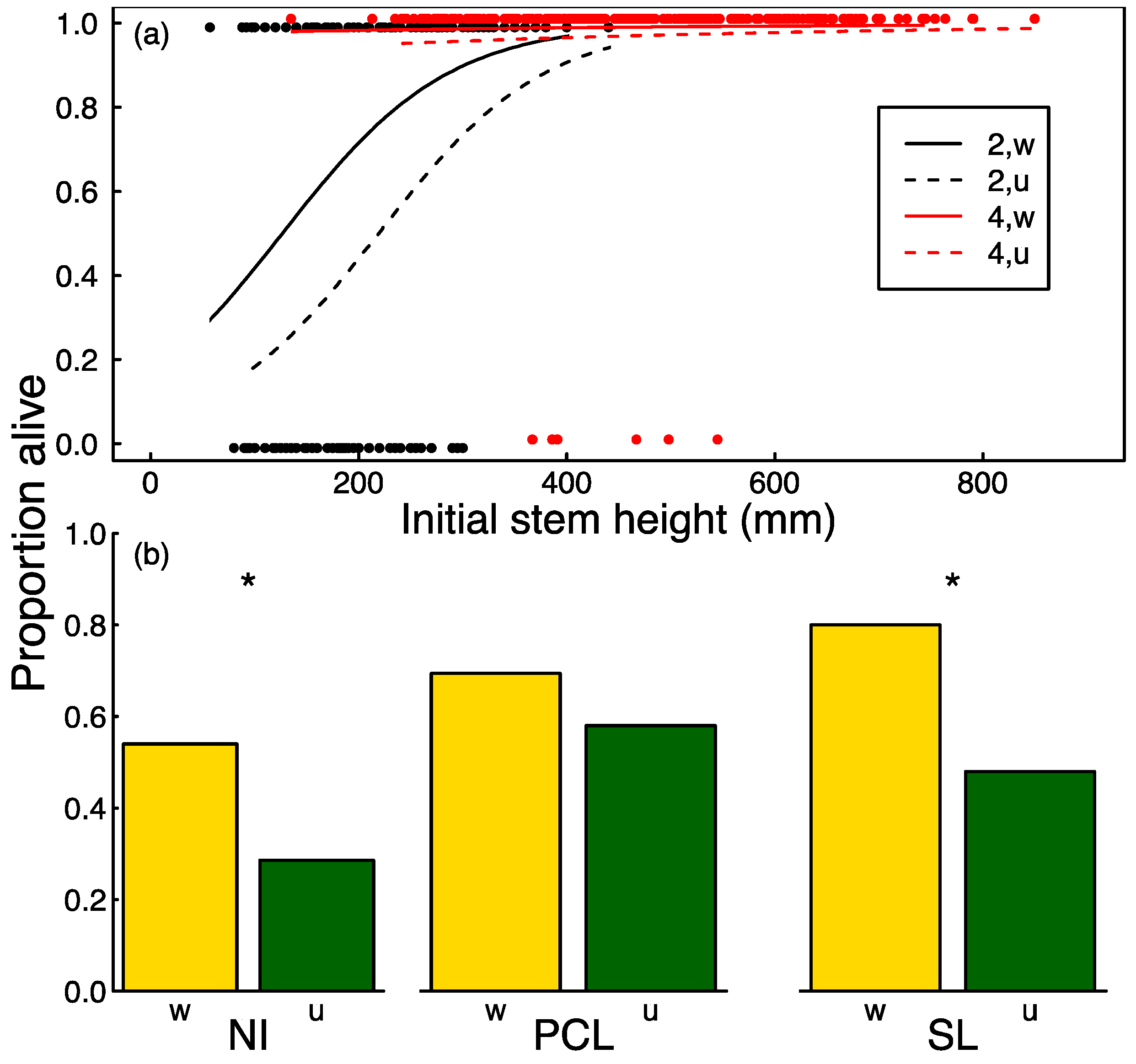
3.2. Seedling Survival
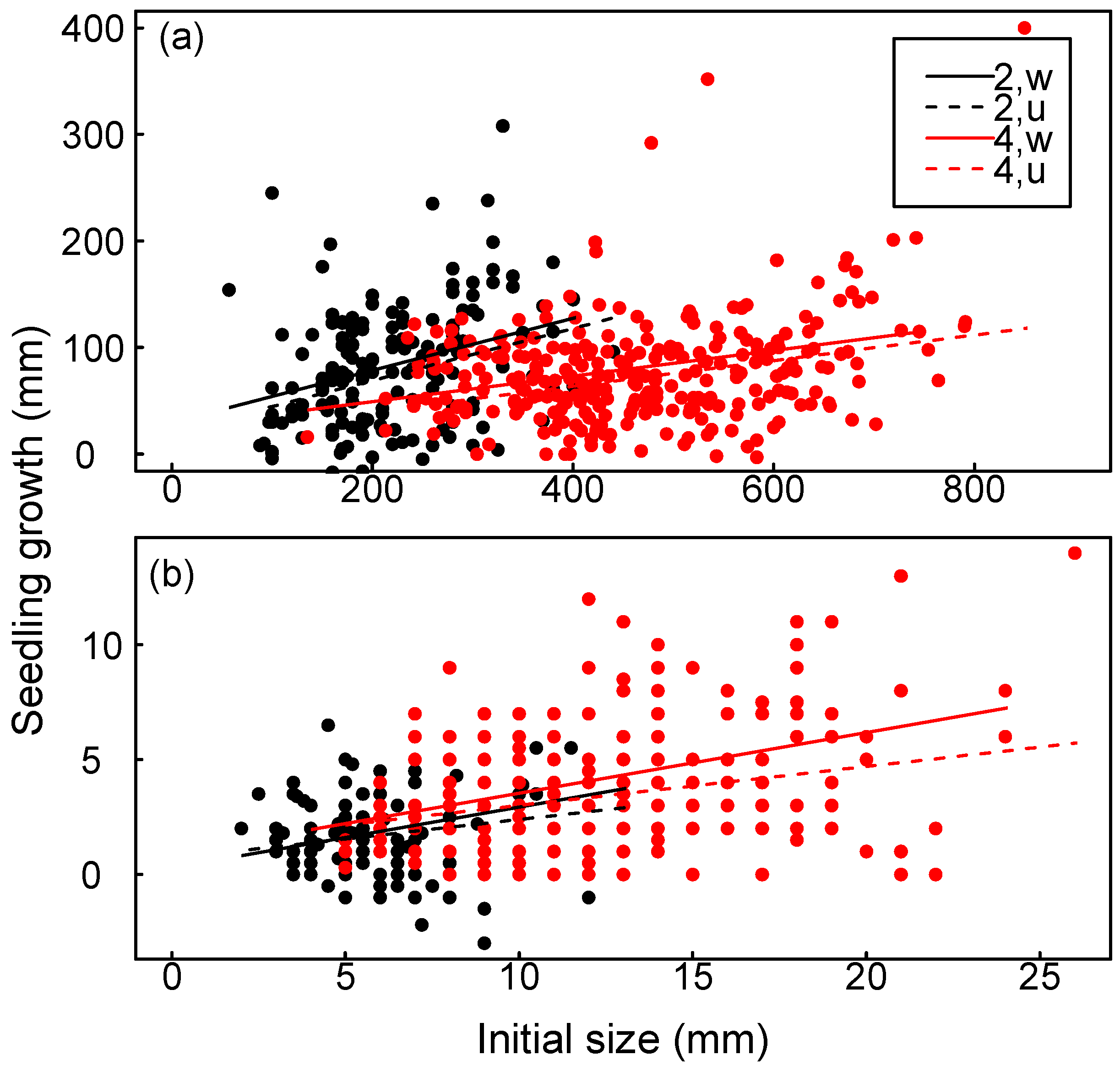
3.3. Seedling Growth
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kozlowski, T.T.; Kramer, P.J.; Pallardy, S.G. The Physiological Ecology of Woody Plants; Academic Press, Inc.: San Diego, CA, USA, 1991. [Google Scholar]
- Cain, M.D. Woody and herbaceous competition effects on the growth of naturally regenerated loblolly and shortleaf pines through 11 years. New For. 1996, 14, 107–125. [Google Scholar] [CrossRef]
- Elliot, K.J.; Vose, J.M.; Knoepp, J.D.; Clinton, B.D.; Kloeppel, B.D. Functional role of the herbaceous layer in Eastern deciduous forest ecosystes. Ecosystems 2015, 18, 221–236. [Google Scholar] [CrossRef]
- Savill, P.; Evans, J.; Auclair, D.; Falck, J. Plantation Silviculture in Europe; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Löf, M. Establishment and growth in seedlings of Fagus sylvatica and Quercus robur: Influence of interference from herbaceous vegetation. Can. J. For. Res. 2000, 30, 855–864. [Google Scholar] [CrossRef]
- Andrés, P.; Salgado, C.; Espelta, J.M. Optimizing nursery and plantation methods to grow Cedrela odorata seedlings in tropical dry agroecosystems. Agrofor. Syst. 2011, 83, 225–234. [Google Scholar] [CrossRef]
- Pitt, D.G.; Hoepting, M.K.; Paarker, W.C.; Morneault, A.E.; Lanteigne, L.; Stinson, A.; Farrell, J.C.G. Optimum vegetation conditions for successful establishment of planted eastern white pine (Pinus strobus, L.). Forests 2016, 7, 175. [Google Scholar] [CrossRef]
- Gärtner, S.M.; Bokalo, M.; Macdonald, S.E.; Stadt, K. Variation in post-wildfire regeneration of boreal mixedwood forests: Underlying factors and implications for natural disturbance-based management. New For. 2014, 45, 215–234. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Lorite, J.; Navarro, F.B.; Sánchez-Cañete, E.P.; Castro, J. Post-fire salvage logging alters species composition and reduces cover, richness, and diversity in Mediterranean plant communities. J. Environ. Manag. 2014, 133, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Klutsch, J.G.; Goodrich, B.A.; Jacobi, W.R. Post-fire regeneration dynamics in whitebark pine (Pinus albicaulis) forests in Wind River and Absaroka Mountains, Wyoming, USA. J. For. Res. 2015, 26, 719–733. [Google Scholar] [CrossRef]
- Navarro-Cerrillo, R.M.; Griffith, D.M.; Ramírez-Soria, M.J.; Pariona, W.; Golicher, D.; Palacios, G. Enrichment of big-leaf mahogany (Swietenia macrophylla King) in logging gaps in Bolivia: The effects of planting method and silvicultural treatments on long-term seedling survival and growth. For. Ecol. Manag. 2011, 262, 2271–2280. [Google Scholar] [CrossRef]
- Gordon, D.R.; Rice, K.J. Competitive suppression of Quercus douglasii (Fagaceae) seedling emergence and growth. Am. J. Bot. 2000, 87, 986–994. [Google Scholar] [CrossRef]
- Cramer, M.D.; van Cauter, A.; Bond, W.J. Growth of N2-fixing African savanna Acacia species is constrained by below-ground competition with grass. J. Ecol. 2010, 98, 156–167. [Google Scholar] [CrossRef]
- NeSmith, J.E.; Alba, C.; Flory, S.L. Experimental drought and plant invasion additively suppress primary pine species of southeastern US forests. For. Ecol. Manag. 2018, 411, 158–165. [Google Scholar] [CrossRef]
- Da Silva, I.; da Silva, M.; Rocha, J.S.; Bianchini, E.; Pimenta, J.A.; Stolf-Moreira, R.; Oliveira, H.C. Potential allelopathic effect of Brachiaria decumbens root exudates on neotropical tree seedlings. Theor. Exp. Plant Physiol. 2017, 29, 177–186. [Google Scholar] [CrossRef]
- Good, M.K.; Clarke, P.J.; Price, J.N.; Reid, N. Seasonality and facilitation drive tree establishment in a semi-arid floodplain savanna. Oecologia 2014, 175, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Loranger, H.; Zotz, G.; Bader, M.Y. Competitor or facilitator? The ambiguous role of alpine grassland for the early establishment of tree seedlings at treeline. Oikos 2017, 126, 1625–1636. [Google Scholar] [CrossRef]
- St-Denis, A.; Kneeshaw, D.; Messier, C. Effect of predation, competition, and facilitation on tree survival land growth in abandoned fields: Towards precision restoration. Forests 2018, 9, 692. [Google Scholar] [CrossRef]
- Provendier, D.; Balandier, P. Compared effects of competition by grasses (Gramonoids) and broom (Cytisus scoparius) on growth and functional traits of beech (Fagus sylvatica). Ann. For. Sci. 2008, 65, 510. [Google Scholar] [CrossRef]
- Meli, P.; Rey-Benayas, J.M.; Martínez-Ramos, M.; Carabias, J. Effects of grass clearing and soil tilling on establishment of planted tree seedlings in tropical riparian pastures. New For. 2015, 46, 507–525. [Google Scholar] [CrossRef]
- Vilà, M.; Sardans, J. Plant competition in mediterranean-type vegetation. J. Veg. Sci. 1999, 10, 281–294. [Google Scholar] [CrossRef]
- Sánchez, A.M.; Peco, B. Interference between perennial grassland and Lavandula stoechas subsp pedunculata seedlings: A case of spatial segregation caused by competition. Acta Oecol. 2004, 26, 39–44. [Google Scholar] [CrossRef]
- Hill, J.D.; Canham, C.D.; Wood, D.M. Patterns and causes of resistance to tree invasion in rights-of-way. Ecol. Appl. 1995, 5, 459–470. [Google Scholar] [CrossRef]
- Scholes, R.J.; Archer, S.R. Tree-grass interactions in savannas. Annu. Rev. Ecol. Syst. 1997, 28, 517–544. [Google Scholar] [CrossRef]
- Bailey, T.G.; Davidson, N.J.; Close, D.C. Understanding the regeneration niche: Microsite attributes and recruitment of eucalypts in dry forests. For. Ecol. Manag. 2012, 269, 229–238. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Lindenmayer, D.B.; Thorn, S.; Gustafsson, L. Salvage logging in the world’s forests: Interactions between natural disturbance and logging need recognition. Glob. Ecol. Biogeogr. 2018, 27, 1140–1154. [Google Scholar] [CrossRef]
- Müller, J.; Noss, R.; Thorn, S.; Bässler, C.; Leverkus, A.B.; Lindenmayer, D. Increasing disturbance demands new policies to conserve intact forest. Conserv. Lett. 2018, e12449. [Google Scholar] [CrossRef]
- Castro, J.; Allen, C.D.; Molina-Morales, M.; Marañón-Jiménez, S.; Sánchez-Miranda, A.; Zamora, R. Salvage logging versus the use of burnt wood as a nurse object to promote post-fire tree seedling establishment. Restor. Ecol. 2011, 19, 537–544. [Google Scholar] [CrossRef]
- Marzano, R.; Garbarino, M.; Marcolin, E.; Pividori, M.; Lingua, E. Deadwood anisotropic facilitation on seedling establishment after a stand-replacing wildfire in Aosta Valley (NW Italy). Ecol. Eng. 2013, 51, 117–122. [Google Scholar] [CrossRef]
- Leverkus, A.B.; Rey-Benayas, J.M.; Castro, J.; Boucher, D.; Brewer, S.; Collins, B.M.; Donato, D.; Fraver, S.; Kishchuck, B.E.; Lee, E.-J.; et al. Salvage logging effects on regulating and supporting ecosystem services—A systematic map. Can. J. For. Res. 2018, 48, 983–1000. [Google Scholar] [CrossRef]
- Flores, B.M.; Fagoaga, R.; Nelson, B.W.; Holmgren, M. Repeated fires trap Amazonian blackwater floodplains in an open vegetation state. J. Appl. Ecol. 2016, 53, 1597–1603. [Google Scholar] [CrossRef]
- Reyes, O.; Casal, M. Effect of high temperatures on cone opening and on the release and viability of Pinus pinaster and, P. radiata seeds in NW Spain. Ann. For. Sci. 2002, 59, 327–334. [Google Scholar]
- Martínez-Sánchez, J.J.; Marín, A.; Herranz, J.M.; Ferrandis, P.; de las Heras, J. Effects of high temperatures on germination of Pinus halepensis Mill. and, P. pinaster Aiton subsp. pinaster seeds in southeast Spain. Vegetatio 1995, 116, 69–72. [Google Scholar]
- Davies, G.M.; Gray, A. Don’t let spurious accusations of pseudoreplication limit our ability to learn from natural experiments (and other messy kinds of ecological monitoring). Ecol. Evol. 2015, 5, 5295–5304. [Google Scholar] [CrossRef] [PubMed]
- Colegrave, N.; Ruxton, G.D. Using biological insight and pragmatism when thinking about pseudoreplication. Trends Ecol. Evol. 2018, 33, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Molinas-González, C.R.; Leverkus, A.B.; Marañón-Jiménez, S.; Castro, J. Fall rate of burnt pines across an altitudinal gradient in a Mediterranean mountain. Eur. J. For. Res. 2017, 136, 401–409. [Google Scholar] [CrossRef]
- Marañón-Jiménez, S.; Castro, J.; Querejeta, J.I.; Fernández-Ondoño, E.; Allen, C.D. Post-fire wood management alters water stress, growth, and performance of pine regeneration in a Mediterranean ecosystem. For. Ecol. Manag. 2013, 308, 231–239. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2016. [Google Scholar]
- Crawley, M.J. The R Book, 2nd ed.; Wiley & Sons: New Delhi, India, 2013; ISBN 9780262232586. [Google Scholar]
- Hurlbert, S.H. Pseudoreplication and the design of ecological field experiments. Ecol. Monogr. 1984, 54, 187–211. [Google Scholar] [CrossRef]
- Schwartz, N.B.; Urban, D.L.; White, P.S.; Moody, A.; Klein, R.N. Vegetation dynamics vary across topographic and fire severity gradients following prescribed burning in Great Smoky Mountains National Park. For. Ecol. Manag. 2016, 365, 1–11. [Google Scholar] [CrossRef]
- Gilliam, F.S. The ecological significance of the herbaceous layer in temperate forest ecosystems. BioScience 2007, 57, 845–858. [Google Scholar] [CrossRef]
- Jensen, A.M.; Löf, M. Effects of interspecific competition from surrounding vegetation on mortality, growth and stem development in young oaks (Quercus robur). For. Ecol. Manag. 2017, 392, 176–183. [Google Scholar] [CrossRef]
- Miriti, M.N. Ontogenetic shift from facilitation to competition in a desert shrub. J. Ecol. 2006, 94, 973–979. [Google Scholar] [CrossRef]
- Niinemets, Ü. Responses of forest trees to single and multiple environmental stresses from seedlings to mature plants: Past stress history, stress interactions, tolerance and acclimation. For. Ecol. Manag. 2010, 260, 1623–1639. [Google Scholar] [CrossRef]
- Davis, M.A.; Wrage, K.J.; Reich, P.B.; Tjoelker, M.G.; Schaeffer, T.; Muermann, C. Survival, growth, and photosynthesis of tree seedlings competing with herbaceous vegetation along a water-light-nitrogen gradient. Plant Ecol. 1999, 145, 341–350. [Google Scholar] [CrossRef]
- Picon-Cochard, C.; Coll, L.; Balandier, P. The role of below-ground competition during early stages of secondary succession: The case of 3-year-old Scots pine (Pinus sylvestris, L.) seedlings in an abandoned grassland. Oecologia 2006, 148, 373–383. [Google Scholar] [CrossRef]
- Gómez-Aparicio, L.; Valladares, F.; Zamora, R. Differential light responses of Mediterranean tree saplings: Linking ecophysiology with regeneration niche in four co-occuring species. Tree Physiol. 2006, 26, 947–958. [Google Scholar] [CrossRef]
- Castro, J.; Zamora, R.; Hódar, J.A.; Gómez, J.M. Use of shrubs as nurse plants: A new technique for reforestation in Mediterranean mountains. Restor. Ecol. 2002, 10, 297–305. [Google Scholar] [CrossRef]
- Gómez-Aparicio, L.; Zamora, R.; Gómez, J.M.; Hódar, J.A.; Castro, J.; Baraza, E. Aplying plant facilitation to forest restoration: A meta-analysis of the use of shrubs as nurse plants. Ecol. Appl. 2004, 14, 1128–1138. [Google Scholar] [CrossRef]
- Bloor, J.M.G.; Leadley, P.W.; Barthes, L. Responses of Fraxinus excelsior seedlings to grass-induced above- and below-ground competition. Plant Ecol. 2008, 194, 293–304. [Google Scholar] [CrossRef]
- Gunaratne, A.M.T.A.; Gunatilleke, C.V.S.; Gunatilleke, I.A.U.N.; Weerasihghe, H.M.S.P.M.; Burslem, D.F.R.P. Release from root competition promotes tree seedling survival and growth following transplantation into human-induced grasslands in Sri Lanka. For. Ecol. Manag. 2011, 262, 229–236. [Google Scholar] [CrossRef]
- Salazar-Tortosa, D.; Castro, J.; Villar-Salvador, P.; Viñegla, B.; Matías, L.; Michelsen, A.; de Casas, R.R.; Querejeta, I. The “isohydric trap”: A proposed feed-back between water shortage, stomatal regulation and nutrient acquisition drives differential growth and survival of European pines under climatic dryness. Glob. Chang. Biol. 2018, 24, 4069–4083. [Google Scholar] [CrossRef]
- Bottero, A.; Garbarino, M.; Long, J.N.; Motta, R. The interacting ecological effects of large-scale disturbances and salvage logging on montane spruce forest regeneration in the western European Alps. For. Ecol. Manag. 2013, 292, 19–28. [Google Scholar] [CrossRef]
- Fidej, G.; Rozman, A.; Diaci, J. Drivers of regeneration dynamics following salvage logging and different silvicultural treatments in windthrow areas in Slovenia. For. Ecol. Manag. 2018, 409, 378–389. [Google Scholar] [CrossRef]
- Castro, J.; Zamora, R.; Hódar, J. Mechanisms blocking Pinus sylvestris colonization of Mediterranean mountains meadows. J. Veg. Sci. 2002, 13, 725–731. [Google Scholar] [CrossRef]
| Landscape Unit | ||||
|---|---|---|---|---|
| NI | PCL | SL | ||
| Sampling unit variables | Centroid coordinates (x, y) | 456,142 4,090,020 | 455,887 4,089,880 | 456,024 4,089,940 |
| Area (m2) | 18,798 | 14,586 | 26,157 | |
| Elevation (m a.s.l.) | 1533 | 1430 | 1474 | |
| Slope (%) a | 26.8 | 23.9 | 23.1 | |
| Pre-treatment tree density (individuals/ha) b | 1304 ± 95 | 1236 ± 73 | 1316 ± 89 | |
| Pre-treatment tree height (m) c | 5.8 ± 0.2 | 6.1 ± 0.2 | n.a. | |
| Pre-treatment tree basal diameter (cm) d | 18.9 ± 0.6 | 20.1 ± 0.4 | 18.8 ± 0.7 | |
| Initial values of herbaceous cover and pine seedlings | Herb height year 2 (cm) | 15.7 ± 1.0 | 15.3±0.9 | 11.8 ± 1.0 |
| Pine height year 2 (cm) | 17.6 ± 0.6 | 24.1±0.7 | 18.2 ± 0.6 | |
| Pine stem diameter year 2 (mm) | 4.3 ± 0.2 | 6.5 ± 0.2 | 5.9 ± 0.2 | |
| Herb height year 4 (cm) | 24.6 ± 1.6 | 25.8 ± 1.6 | 16.7 ± 1.6 | |
| Pine height year 4 (cm) | 50.5 ± 1.2 | 47.5 ± 1.2 | 40.5 ± 1.2 | |
| Pine stem diameter year 4 (mm) | 11.4 ± 0.3 | 12.1 ± 0.4 | 12.4 ± 0.4 | |
| Explanatory Variable a | Seedling Survival | Height Growth | Stem width Growth | ||||
|---|---|---|---|---|---|---|---|
| df | χ2 | p | F | p | F | p | |
| Weeding (W) | 1 | 23.02 | <0.001 | 4.00 | <0.05 | b | b |
| Age (A) | 1 | b | b | b | b | 4.87 | <0.05 |
| Initial size (S) | 1 | b, c | b | b | b | b | b |
| W:A | 1 | 1.80 | 0.18 | 0.01 | 0.92 | 0.23 | 0.63 |
| W:S | 1 | 0.02 | 0.88 | 0.65 | 0.42 | 4.49 | <0.05 |
| A:S | 1 | 4.88 | <0.05 | 8.5 | <0.01 | 1.28 | 0.26 |
| W:A:S | 1 | 0.06 | 0.81 | 0.03 | 0.85 | 0.10 | 0.75 |
| N seedlings | 596 | 460 | 460 | ||||
| Adjusted R2 d | 0.35 e | 0.10 | 0.22 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro, J.; Leverkus, A.B. Effect of Herbaceous Layer Interference on the Post-Fire Regeneration of a Serotinous Pine (Pinus pinaster Aiton) across Two Seedling Ages. Forests 2019, 10, 74. https://doi.org/10.3390/f10010074
Castro J, Leverkus AB. Effect of Herbaceous Layer Interference on the Post-Fire Regeneration of a Serotinous Pine (Pinus pinaster Aiton) across Two Seedling Ages. Forests. 2019; 10(1):74. https://doi.org/10.3390/f10010074
Chicago/Turabian StyleCastro, Jorge, and Alexandro B. Leverkus. 2019. "Effect of Herbaceous Layer Interference on the Post-Fire Regeneration of a Serotinous Pine (Pinus pinaster Aiton) across Two Seedling Ages" Forests 10, no. 1: 74. https://doi.org/10.3390/f10010074
APA StyleCastro, J., & Leverkus, A. B. (2019). Effect of Herbaceous Layer Interference on the Post-Fire Regeneration of a Serotinous Pine (Pinus pinaster Aiton) across Two Seedling Ages. Forests, 10(1), 74. https://doi.org/10.3390/f10010074





