A Fovea Localization Scheme Using Vessel Origin-Based Parabolic Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Illumination Correction
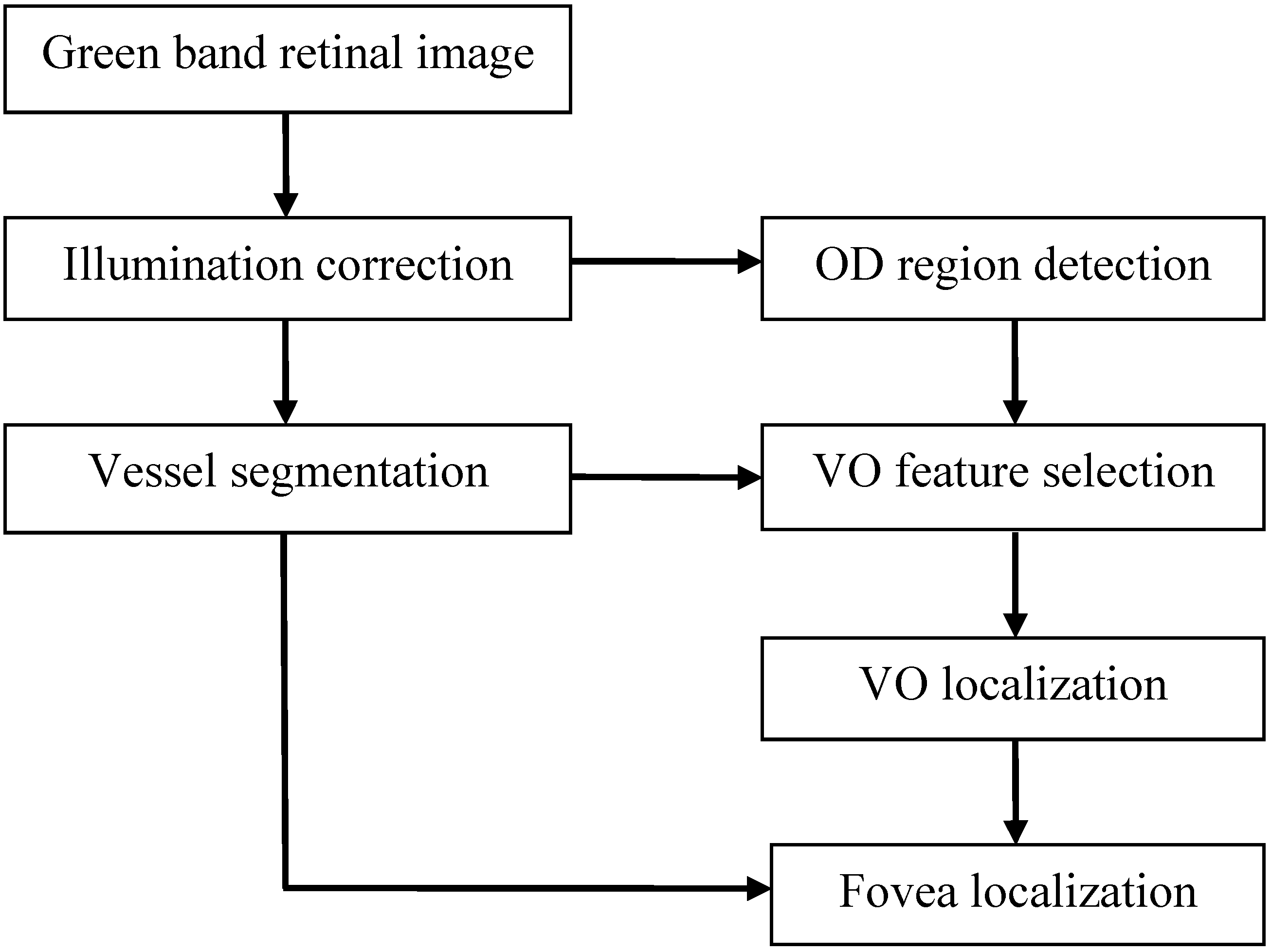

2.2. Optic Disc (OD) Localization
2.3. Retinal Vessel Segmentation
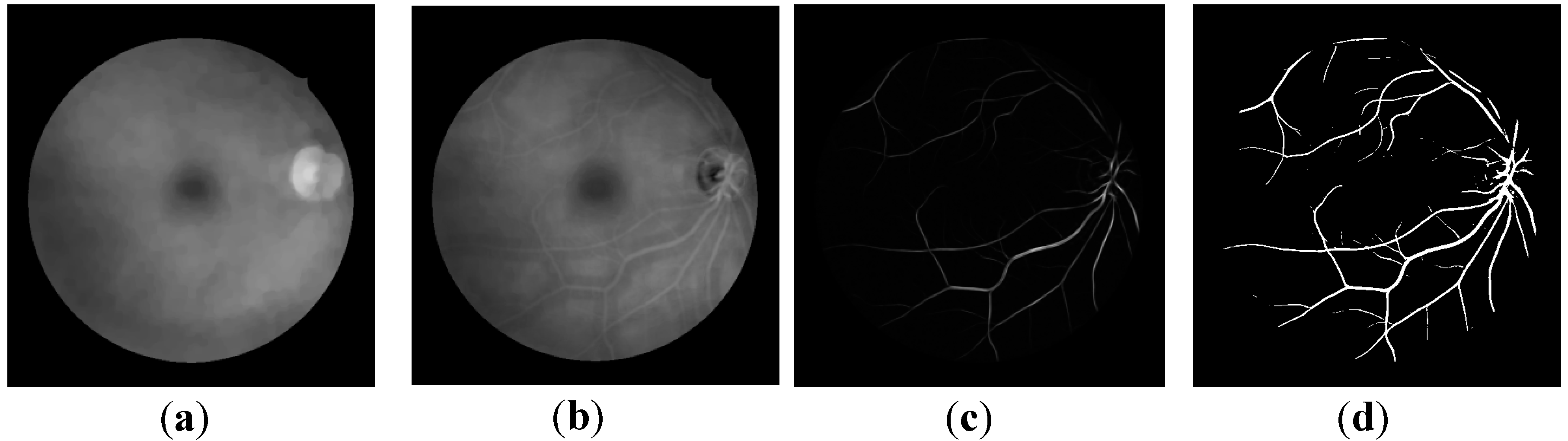
2.4. Vessel Origin (VO) Feature Selection
2.4.1. Vessel Thickness Rate
2.4.2. Vessel Density

2.4.3. Vessel Radiation
2.4.4. Vessel Average Intensity
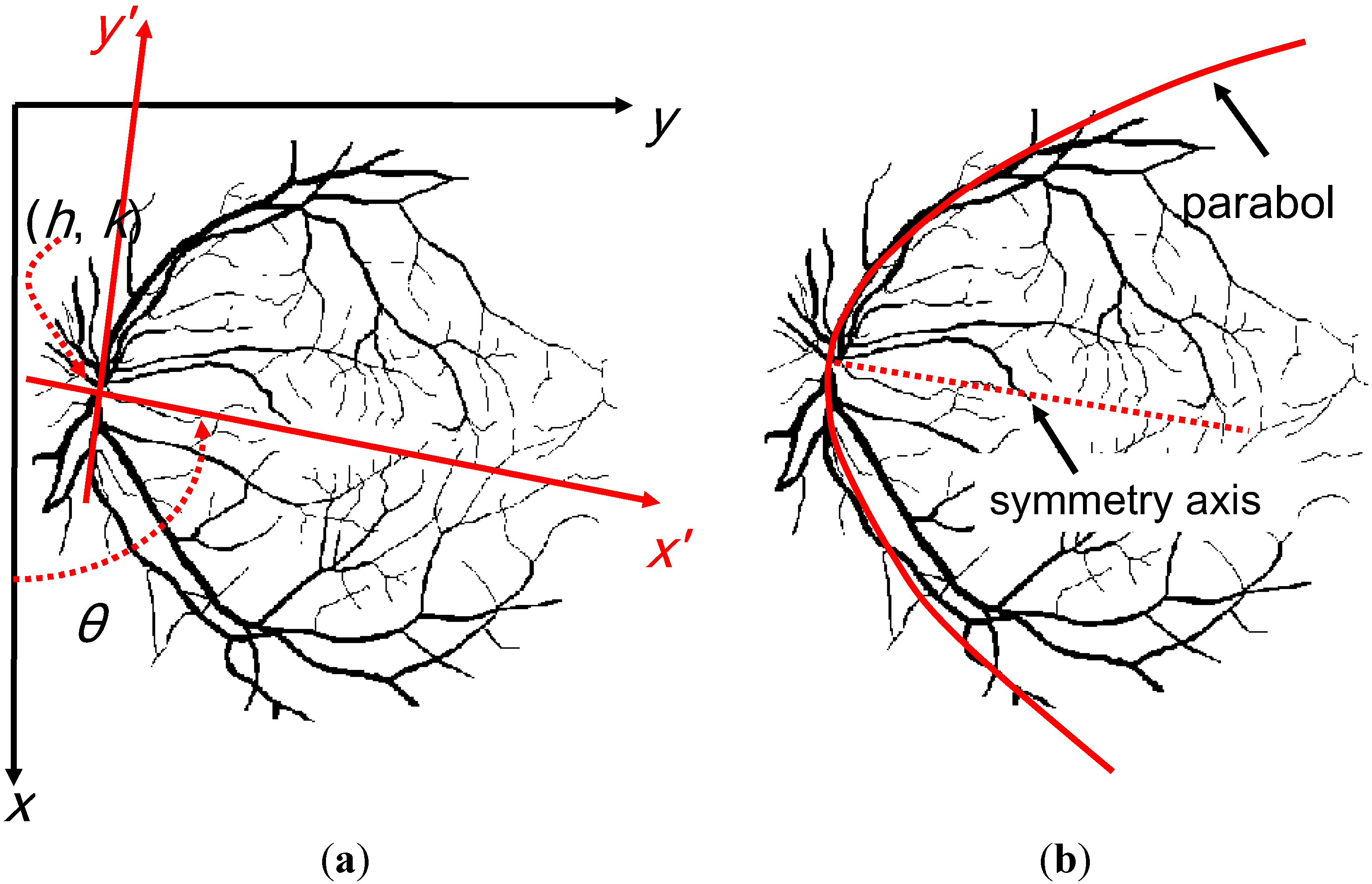
2.5. VO Localization
2.6. Fovea Localization
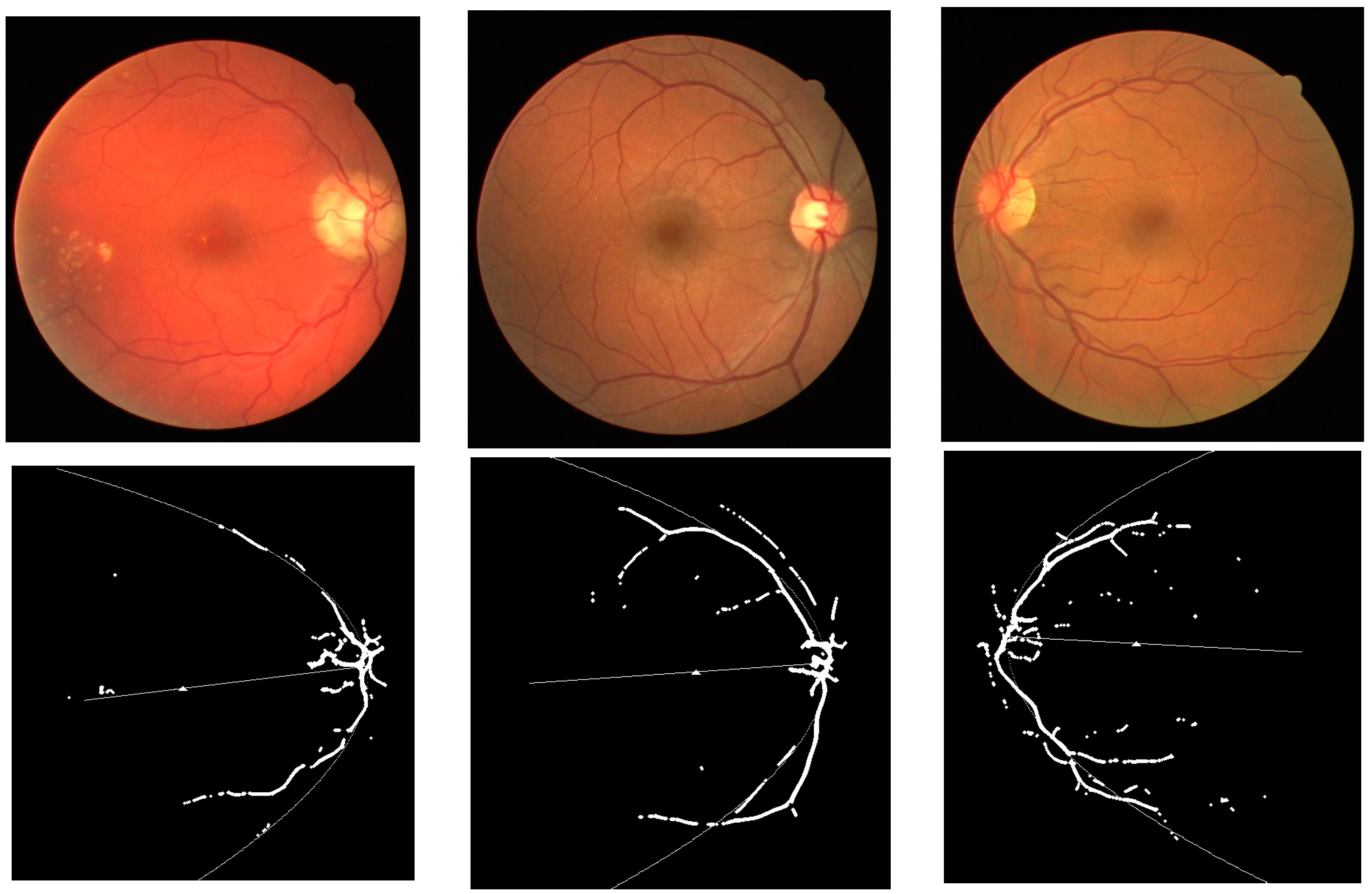
3. Results and Discussions
3.1. Results
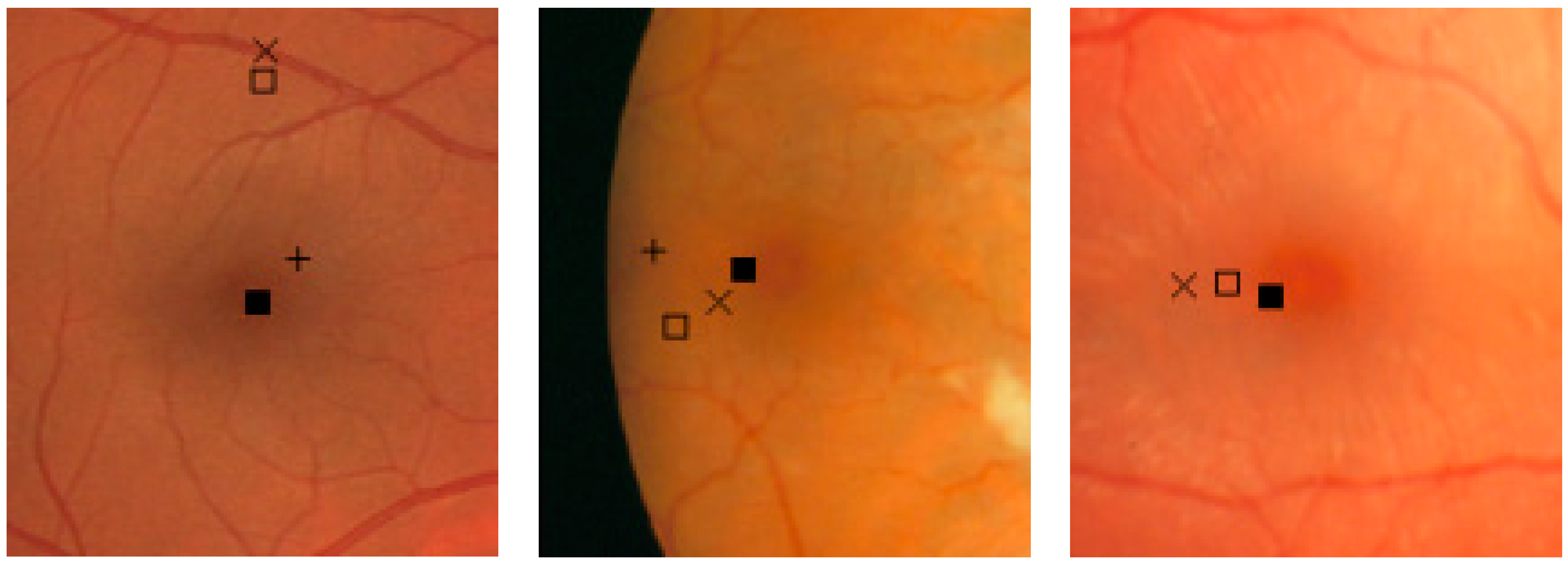
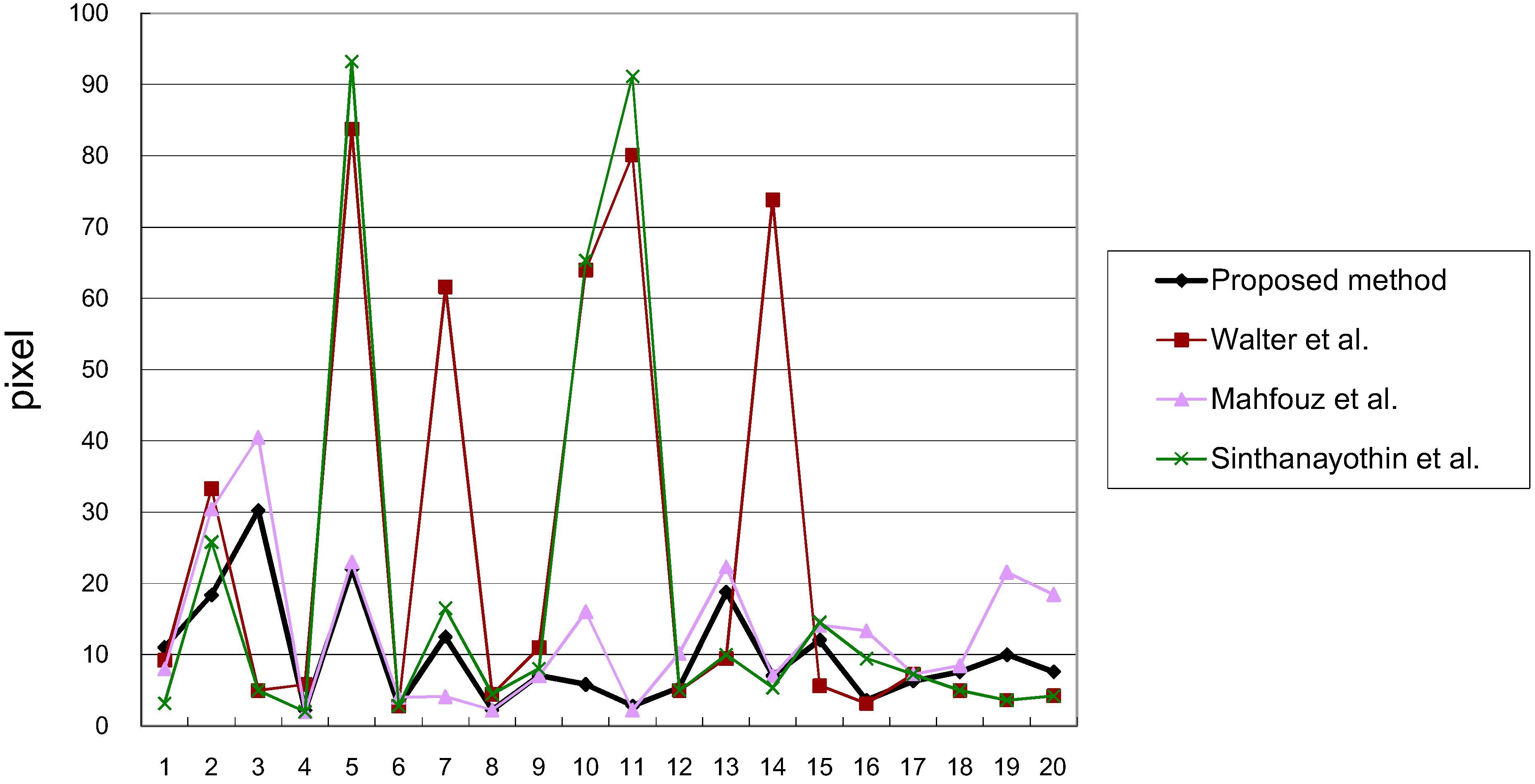
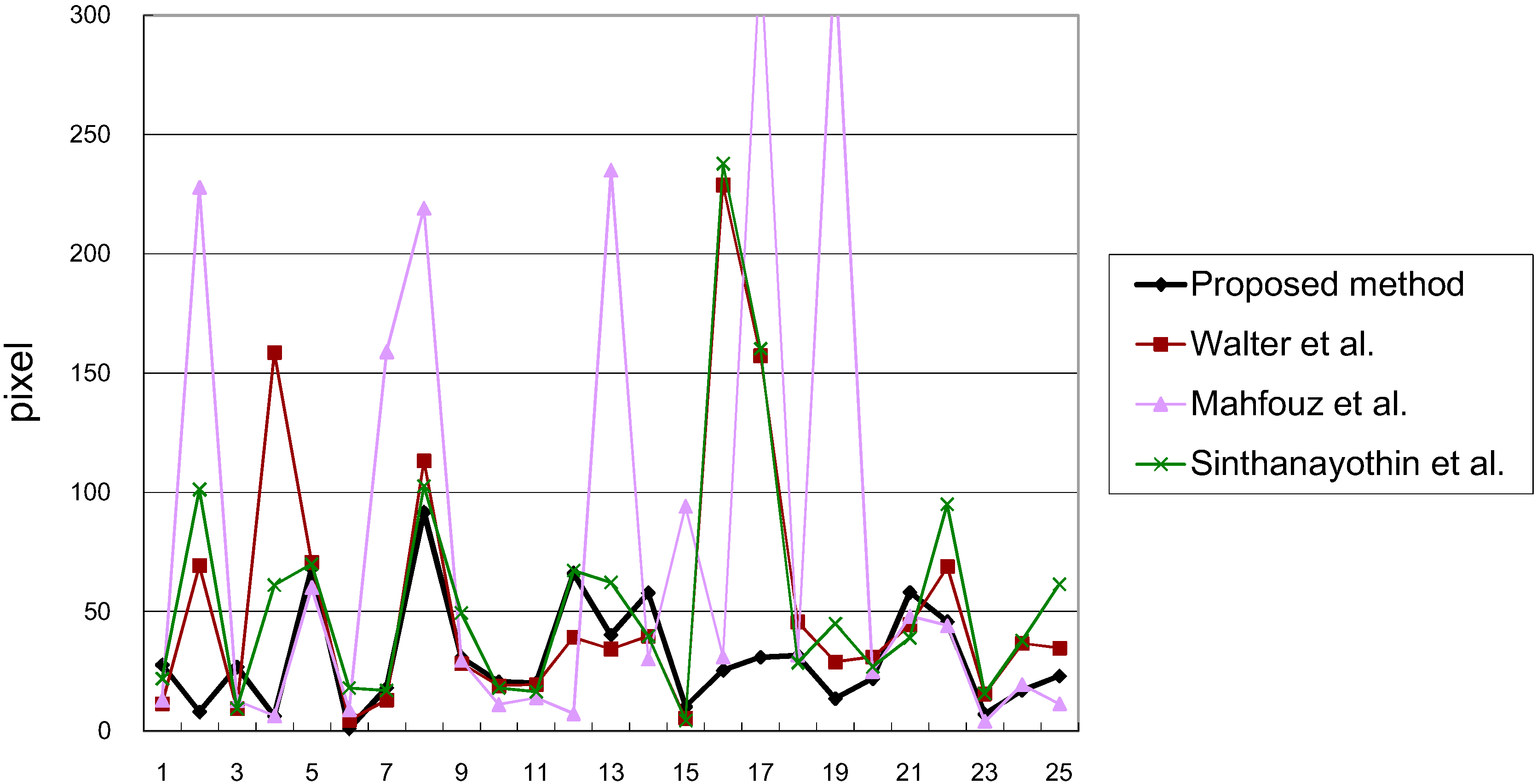
3.2. Discussions
4. Conclusions
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Gegundez-Arias, M.E.; Marin, D.; Bravo, J.M.; Suero, A. Locating the Fovea Center Position in Digital Fundus Images Using Thresholding and Feature Extraction Techniques. Comput. Med. Imaging Graph. 2013, 37, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, L.; Lalonde, M.; Beaulieu, M.; Boucher, M.C. Procedure to Detect Anatomical Structures in Optical Fundus Images. Proc. SPIE 2001. [Google Scholar] [CrossRef]
- Zhang, B.; Karray, F. Optic disc and fovea detection via multi-scale matched filters and a vessels’ directional matched filter. In Proceedings of the 2010 International Conference on Autonomous and Intelligent Systems (AIS), Povoa de Varzim, Portugal, 21–23 June 2010; pp. 1–5.
- Sinthanayothin, C.; Boyce, J.; Cook, H.; Williamson, T. Automated Localization of the Optic Disc, Fovea, and Retinal Blood Vessels from Digital Colour Fundus Images. Br. J. Ophthalmol. 1999, 83, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Chin, K.S.; Trucco, E.; Tan, L.; Wilson, P.J. Automatic Fovea Location in Retinal Images Using Anatomical Priors and Vessel Density. Pattern Recognit. Lett. 2013, 34, 1152–1158. [Google Scholar] [CrossRef]
- Li, H.; Chutatape, O. Automated Feature Extraction in Color Retinal Images by a Model Based Approach. IEEE Trans. Biomedical Eng. 2004, 51, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Foracchia, M.; Grisan, E.; Ruggeri, A. Detection of Optic Disc in Retinal Images by Means of a Geometrical Model of Vessel Structure. IEEE Trans. Med. Imaging 2004, 23, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Tobin, K.W.; Chaum, E.; Govindasamy, V.P. Detection of Anatomic Structures in Human Retinal Imagery. IEEE Trans. Med. Imaging 2007, 26, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.D.; Goatman, K.A.; Philip, S.; Olson, J.A.; Sharp, P.F. Automatic Detection of Retinal Anatomy to Assist Diabetic Retinopathy Screening. Phys. Med. Biol. 2007, 52, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Niemeijer, M.; Abràmoff, M.D.; van Ginneken, B. Automated Localization of the Optic Disc and the Fovea. In Proceedings of the IEEE EMBS Conference on 30th Annual International, Vancouver, BC, Canada, 2008; pp. 3538–3541.
- Goatman, K.A.; Fleming, A.D.; Philip, S.; Williams, G.J.; Olson, J.A.; Sharp, P.F. Detection of New Vessels on the Optic Disc Using Retinal Photographs. IEEE Trans. Med. Imaging 2011, 30, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.Y.; Liu, C.C.; Wu, J.L.; Yu, S.S.; Huang, J.Y. A Study of Vessel Origin Detection on Retinal Images. In Proceedings of IEA/AIE 2013 Conference, Amsterdam, Netherlands, 17–21 June 2013.
- University Medical Center Utrecht, Image Sciences Institute. Available online: http://www.isi.uu.nl/Research/ Databases/DRIVE (accessed on 20 October 2011).
- Hoover, A. Structured Analysis of the Retina Project Website. Available online: http://www.ces.clemson.edu/~ahoover/stare (accessed on 10 September 2009).
- Foracchia, M.; Grisan, E.; Ruggeri, A. Luminosity and Contrast Normalization in Retinal Images. Med. Image Anal. 2005, 9, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Ricci, E.; Perfetti, R. Retinal Blood Vessel Segmentation Using Line Operators and Support Vector Classification. IEEE Trans. Med. Imaging 2007, 26, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, H.K.; Liu, C.C.; Yu, C.Y.; Kuo, S.W.; Yu, S.S. A Novel Optic Disc Detection Scheme on Retinal Images. Expert Syst. Appl. 2012, 39, 10600–10606. [Google Scholar] [CrossRef]
- Zwiggelaar, R.; Astley, S.M.; Boggis, C.R.M.; Taylor, C.J. Linear Structures in Mammographic Images: Detection and Classification. IEEE Trans. Med. Imaging 2004, 23, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.C.; Woods, R.E. Digital Image Processing, 3rd ed.; Pearson: New Jersey, NJ, USA, 2010. [Google Scholar]
- Duda, R.O.; Hart, P.E.; Stork, D.G. Pattern Classification, 2nd ed.; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Niemeijer, M.; Abràmoff, M.D. Segmentation of the Optic Disc, Macula and Vascular Arch in Fundus Photographs. IEEE Trans. Med. Imaging 2007, 26, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Walter, T.; Klein, J.C.; Massin, P.; Erginay, A. A Contribution of Image Processing to the Diagnosis of Diabetic Retinopathy—Detection of Exudates in Color Fundus Images of the Human Retina. IEEE Trans. Med. Imaging 2002, 21, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Mahfouz, A.E.; Fahmy, A.S. Fast Localization of the Optic Disc Using Projection of Image Features. IEEE Trans. Image Process. 2010, 19, 3285–3289. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, L.; Qureshi, R.J.; Nagy, B.; Harangi, B.; Hajdu, A. Graph based detection of optic disc and fovea in retinal images. In Proceedings of the 2010 4th International Workshop on Soft Computing Applications (SOFA), Arad, Romania, 15–17 July 2010; pp. 143–148.
- Conversano, F.; Franchini, R.; Demitri, C.; Massoptier, L.; Montagna, F.; Maffezzoli, A.; Malvasi, A.; Casciaro, S. Hepatic vessel segmentation for 3D planning of liver surgery: Experimental evaluation of a new fully automatic algorithm. Acad. Radiol. 2011, 18, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, U.T.V.; Bhuiyan, A.; Park, L.A.F.; Ramamohanarao, K. An Effective Retinal Blood Vessel Segmentation Method using Multi-scale Line Detection. Pattern Recognit. 2013, 46, 703–715. [Google Scholar] [CrossRef]
- Bhuiyan, A.; Kawasaki, R.; Lamoureux, E.; Ramamohanarao, K.; Wong, T.Y. Retinal Artery-Vein Caliber Grading Using Color Fundus Imaging. Comput. Methods Programs Biomed. 2013, 111, 104–114. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yu, C.-Y.; Liu, C.-C.; Yu, S.-S. A Fovea Localization Scheme Using Vessel Origin-Based Parabolic Model. Algorithms 2014, 7, 456-470. https://doi.org/10.3390/a7030456
Yu C-Y, Liu C-C, Yu S-S. A Fovea Localization Scheme Using Vessel Origin-Based Parabolic Model. Algorithms. 2014; 7(3):456-470. https://doi.org/10.3390/a7030456
Chicago/Turabian StyleYu, Chun-Yuan, Chen-Chung Liu, and Shyr-Shen Yu. 2014. "A Fovea Localization Scheme Using Vessel Origin-Based Parabolic Model" Algorithms 7, no. 3: 456-470. https://doi.org/10.3390/a7030456
APA StyleYu, C.-Y., Liu, C.-C., & Yu, S.-S. (2014). A Fovea Localization Scheme Using Vessel Origin-Based Parabolic Model. Algorithms, 7(3), 456-470. https://doi.org/10.3390/a7030456




