Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification
Abstract
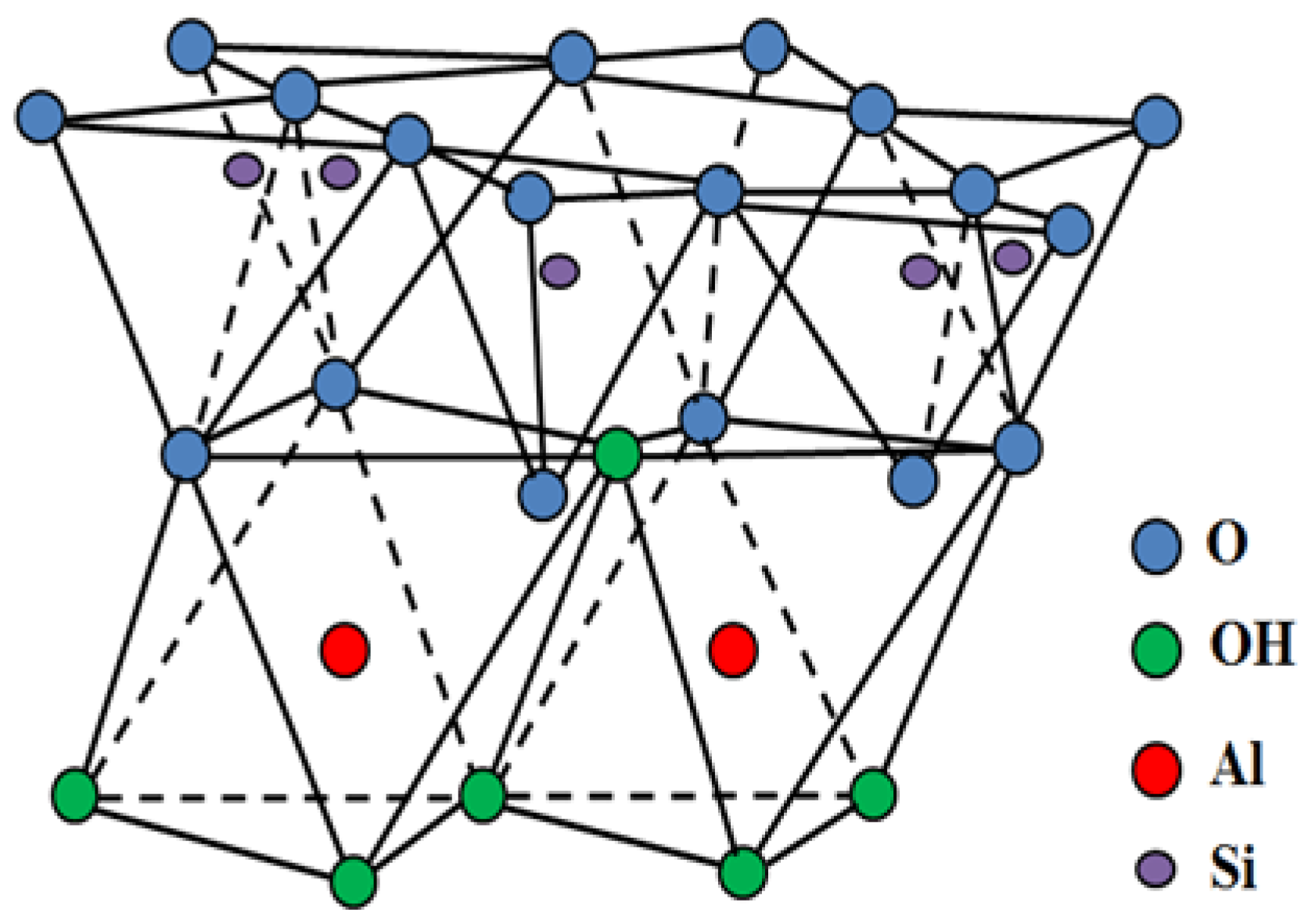
:1. Introduction
2. Results and Discussion
2.1. FTIR
2.2. XRD
2.3. TEM
2.4. FESEM
2.5. Mapping with EDS
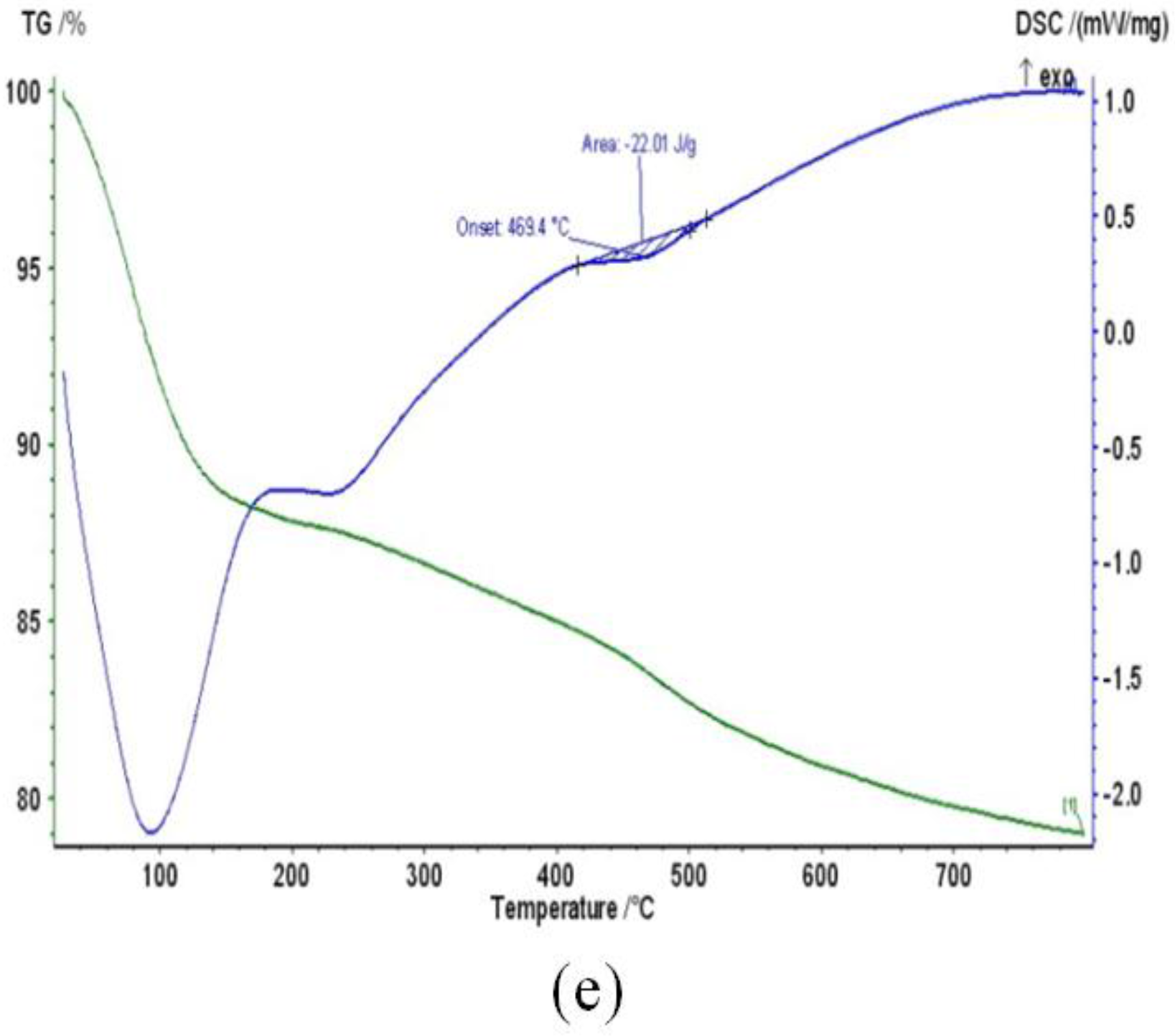
2.6. TGA
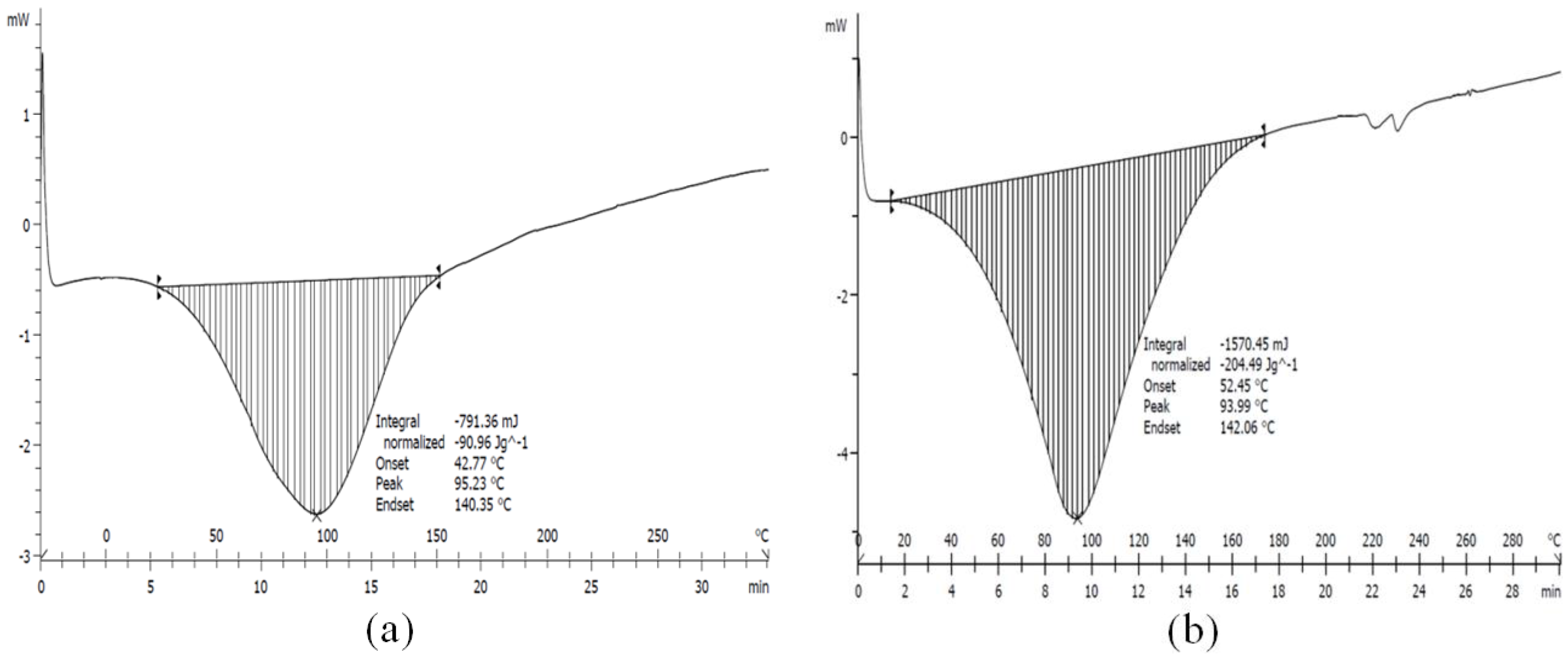
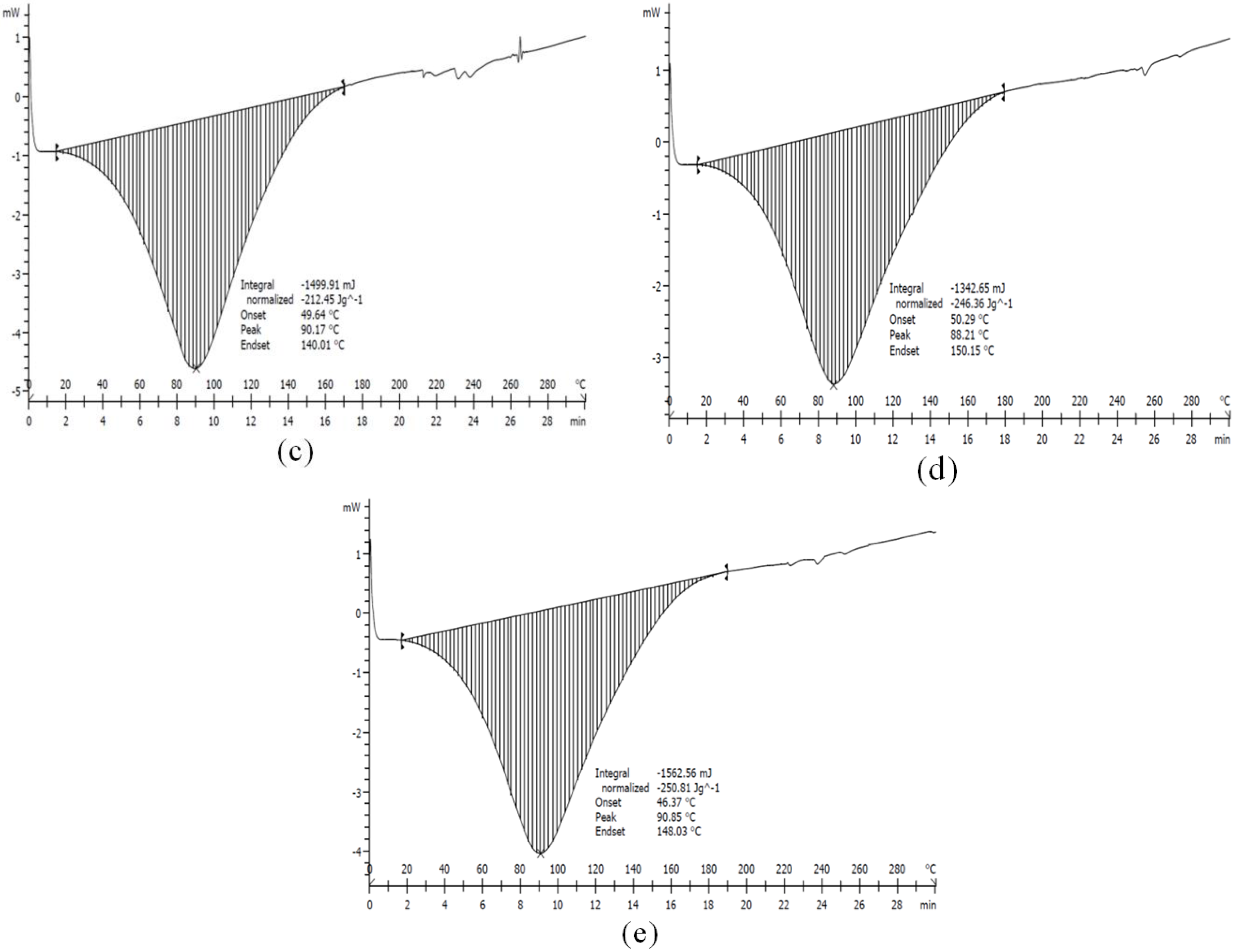
2.7. DSC
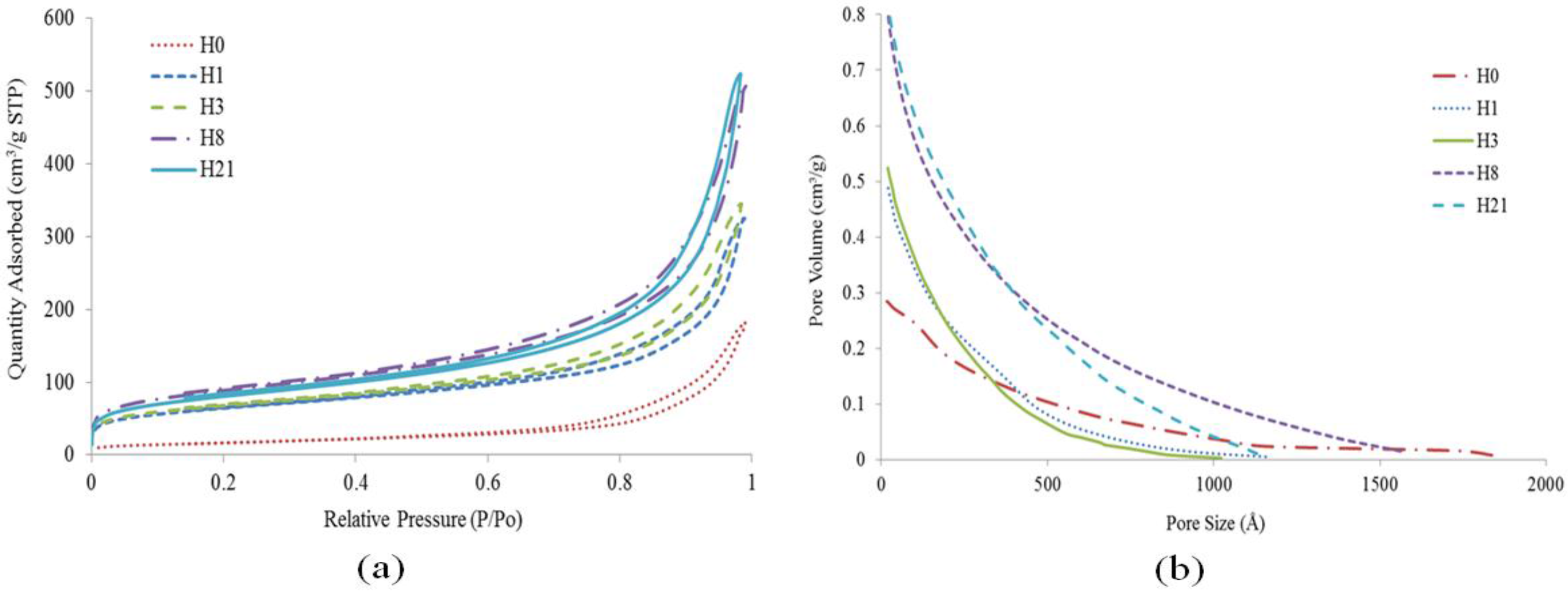
2.8. BET
3. Materials and Methods
Acid Treatment
4. Characteristics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaaz, T.S.; Sulong, A.B.; Akhtar, M.N.; Kadhum, A.A.H.; Mohamad, A.B.; Al-Amiery, A.A. Properties and Applications of Polyvinyl Alcohol, Halloysite Nanotubes and Their Nanocomposites. Molecules 2015, 20, 22833–22847. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.-B.; Pan, L.; Zhang, H.-Y.; Liu, S.-T.; Xia, M.-S.; Chen, X.-G. Effects of acid treatment on the physico-chemical and pore characteristics of halloysite. Colloid Surf. A Physicochem. Eng. Asp. 2012, 396, 182–188. [Google Scholar] [CrossRef]
- García, F.J.; García Rodríguez, S.K.; Andreas, K.; Reller, A. Study of natural halloysite from the Dragon Mine, Utah (USA). Z. Anorg. Allg. Chem. 2009, 635, 790–795. [Google Scholar]
- Ece, Ö.I.; Schroeder, P.A. Clay mineralogy and chemistry of halloysite and alunite deposits in the Turplu area, Balikesir, Turkey. Clay Clay Miner. 2007, 55, 18–35. [Google Scholar] [CrossRef]
- Singer, A.; Zarei, M.; Lange, F.M.; Stahr, K. Halloysite characteristics and formation in the northern Golan Heights. Geoderma 2004, 123, 279–295. [Google Scholar] [CrossRef]
- Joussein, E.; Petit, S.; Churchman, J.; Theng, B.; Righi, D.; Delvaux, B. Halloysite clay minerals—A review. Clay Miner. 2005, 40, 383–426. [Google Scholar] [CrossRef]
- Vergaro, V.; Abdullayev, E.; Lvov, Y.M.; Zeitoun, A.; Cingolani, R.; Rinaldi, R.; Leporatti, S. Cytocompatibility and uptake of halloysite clay nanotubes. Biomacromolecules 2010, 11, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Kautz, C.Q.; Ryan, P.C. The 10 Å to 7 Å halloysite transition in a tropical soil sequence, Costa Rica. Clay Clay Miner. 2003, 51, 252–263. [Google Scholar] [CrossRef]
- Lvov, Y.; Price, R.; Gaber, B.; Ichinose, I. Thin film nanofabrication via layer-by-layer adsorption of tubule halloysite, spherical silica, proteins and polycations. Colloid Surf. A Physicochem. Eng. Asp 2002, 198, 375–382. [Google Scholar] [CrossRef]
- Du, M.; Guo, B.; Jia, D. Newly emerging applications of halloysite nanotubes: A review. Poly. Int. 2010, 59, 574–582. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, J.; Zheng, Y.; Wang, A. Adsorption and release of ofloxacin from acid-and heat-treated halloysite. Colloid Surf. B Biointerfaces 2014, 113, 51–58. [Google Scholar] [PubMed]
- Belver, C.; Bañares Muñoz, M.A.; Vicente, M.A. Chemical activation of a kaolinite under acid and alkaline conditions. Chem. Mater. 2002, 14, 2033–2043. [Google Scholar] [CrossRef]
- Frini-Srasra, N.; Srasra, E. Effect of heating on palygorskite and acid treated palygorskite properties. Surf. Eng. Appl. Electr. 2008, 44, 43–49. [Google Scholar] [CrossRef]
- Kennedy Oubagaranadin, J.U.; Murthy, Z.V.P. Characterization and use of acid-activated montmorillonite-illite type of clay for lead (II) removal. AIChE J. 2010, 56, 2312–2322. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Q.; Chen, H.; Wang, A. XRF and nitrogen adsorption studies of acid-activated palygorskite. Clay Miner. 2010, 45, 145–156. [Google Scholar] [CrossRef]
- Bretti, C.; Cataldo, S.; Gianguzza, A.; Lando, G.; Lazzara, G.; Pettignano, A.; Sammatrano, S. Thermodynamics of Proton Binding of Halloysite Nanotubes. J. Phys. Chem. C 2016, 120, 7849–7859. [Google Scholar] [CrossRef]
- Panda, A.K.; Mishra, B.G.; Mishra, D.K.; Singh, R.K. Effect of sulphuric acid treatment on the physico-chemical characteristics of kaolin clay. Colloid Surf. A Physicochem. Eng. Asp. 2010, 363, 98–104. [Google Scholar] [CrossRef]
- Swapna, V.P.; Selvin Thomas, P.; Suresh, K.I.; Saranya, V.; Rahana, M.P.; Ranimol, S. Thermal properties of poly (vinyl alcohol)(PVA)/halloysite nanotubes reinforced nanocomposites. Int. J. Plastics Tech. 2015, 19, 1–13. [Google Scholar]
- Levis, S.; Deasy, P. Characterisation of halloysite for use as a microtubular drug delivery system. Int. J. Pharm. 2002, 243, 125–134. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X.; Zhang, B.; Zhao, Y.; Zhai, R.; Liu, J.; Chen, R. Rapid adsorption of Cr (VI) on modified halloysite nanotubes. Desalination 2010, 259, 22–28. [Google Scholar]
- Zhao, M.; Liu, P. Adsorption behavior of methylene blue on halloysite nanotubes. Microporous Mesoporous Mater. 2008, 112, 419–424. [Google Scholar] [CrossRef]
- Liu, S.; Cao, X.; Li, L.; Li, C.; Ji, Y.; Xiao, F.-S. Preformed zeolite precursor route for synthesis of mesoporous X zeolite. Colloid Surf. A Physicochem. Eng. Asp. 2008, 318, 269–274. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, B.; Mei, D.; Zhang, H.; Liu, J. Adsorption of methyl violet from aqueous solution by halloysite nanotubes. Desalination 2011, 268, 111–116. [Google Scholar] [CrossRef]
- Lenarda, M.; Storaro, L.; Talon, A.; Moretti, E.; Riello, P. Solid acid catalysts from clays: Preparation of mesoporous catalysts by chemical activation of metakaolin under acid conditions. J. Colloid Interface Sci. 2007, 311, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Frost, R.L.; Vassallo, A.M. The dehydroxylation of the kaolinite clay minerals using infrared emission spectroscopy. Clay Clay Miner. 1996, 44, 635–651. [Google Scholar] [CrossRef]
- Morterra, C.; Magnacca, G. A case study: Surface chemistry and surface structure of catalytic aluminas, as studied by vibrational spectroscopy of adsorbed species. Catal. Today 1996, 27, 497–532. [Google Scholar] [CrossRef]
- Koretsky, C.M.; Sverjensky, D.A.; Salisbury, J.W.; D’Aria, D.M. Detection of surface hydroxyl species on quartz, γ-alumina, and feldspars using diffuse reflectance infrared spectroscopy. Geochim. Cosmoc. Acta 1997, 61, 2193–2210. [Google Scholar] [CrossRef]
- Madejova, J.; Komadel, P. Baseline studies of the clay minerals society source clays: Infrared methods. Clay Clay Miner. 2001, 49, 410–432. [Google Scholar] [CrossRef]
- Luo, P.; Zhao, Y.; Zhang, B.; Liu, J.; Yang, Y.; Liu, J. Study on the adsorption of Neutral Red from aqueous solution onto halloysite nanotubes. Water Res. 2010, 44, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Tan, D.; Aannabi-Bergaya, F.; Yan, W.; Fan, M.; Liu, D.; He, H. Changes in structure, morphology, porosity, and surface activity of mesoporous halloysite nanotubes under heating. Clay Clay Miner. 2012, 60, 561–573. [Google Scholar] [CrossRef]
- Abdullayev, E.; Joshi, A.; Wei, W.; Zhao, Y.; Lvov, Y. Enlargement of halloysite clay nanotube lumen by selective etching of aluminum oxide. ACS Nano. 2012, 6, 7216–7226. [Google Scholar] [CrossRef] [PubMed]
- Bish, D.L.; Blake, D.F.; Vaniman, D.T.; Chipera, S.J.; Morris, R.V.; Ming, D.W.; Treiman, A.H.; Sarrazin, P.; Morrison, S.M.; Downs, R.T.; et al. X-ray diffraction results from Mars Science Laboratory: Mineralogy of Rocknest at Gale crater. Science 2013, 341. [Google Scholar] [CrossRef] [PubMed]
- Bujold, E.; Roberge, S.; Lacasse, Y.; Bureau, M.; François, A.; Sylvie, M.; Jean-Claude, F.; Yves, G. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: A meta-analysis. Obs. Gyn. 2010, 116, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Remškar, M. Inorganic nanotubes. Adv. Mater. 2004, 16, 1497–1504. [Google Scholar] [CrossRef]
- Vasutova, V.; Bezdička, P.; Lang, K.; Hradil, D. Mineralogy of halloysites and their interaction with porphyrine. Ceramics Silikáty 2013, 57, 243–250. [Google Scholar]
- Rybiński, P.; Janowska, G.; Jóźwiak, M.; Pająk, A. Thermal properties and flammability of nanocomposites based on diene rubbers and naturally occurring and activated halloysite nanotubes. J. Therm. Anal. Calorim. 2012, 107, 1243–1249. [Google Scholar] [CrossRef]
- Papoulis, D.; Komarneni, S.; Nikolopoulou, A.; Tsolis-Katagas, P.; Panagiotaras, D.; Kacandes, H.G.; Zhang, P.; Yin, S.; Sato, T.; Katsuki, H. Palygorskite-and Halloysite-TiO2 nanocomposites: Synthesis and photocatalytic activity. Appl. Clay Sci. 2010, 50, 118–124. [Google Scholar] [CrossRef]
- Pasbakhsh, P.; Churchman, G.J.; Keeling, J.L. Characterisation of properties of various halloysites relevant to their use as nanotubes and microfibre fillers. Appl. Clay Sci. 2013, 74, 47–57. [Google Scholar] [CrossRef]
- Markovska, I.; Lyubchev, L. A study on the thermal destruction of rice husk in air and nitrogen atmosphere. J. Therm. Anal. Calorim. 2007, 89, 809–814. [Google Scholar] [CrossRef]
- Fujii, K.; Nakagatio, A.N.; Takagi, H.; Yonekura, D. Sulfuric acid treatment of halloysite nanoclay to improve the mechanical properties of PVA/halloysite transparent composite films. Compos. Interfaces 2014, 21, 319–327. [Google Scholar] [CrossRef]
- Gregg, S.; Sing, K.S.W.; Salzberg, H.W. Water Adsorption, Surface Area and Porosity, 2nd ed.; Academic Press: London, UK, 1982. [Google Scholar]
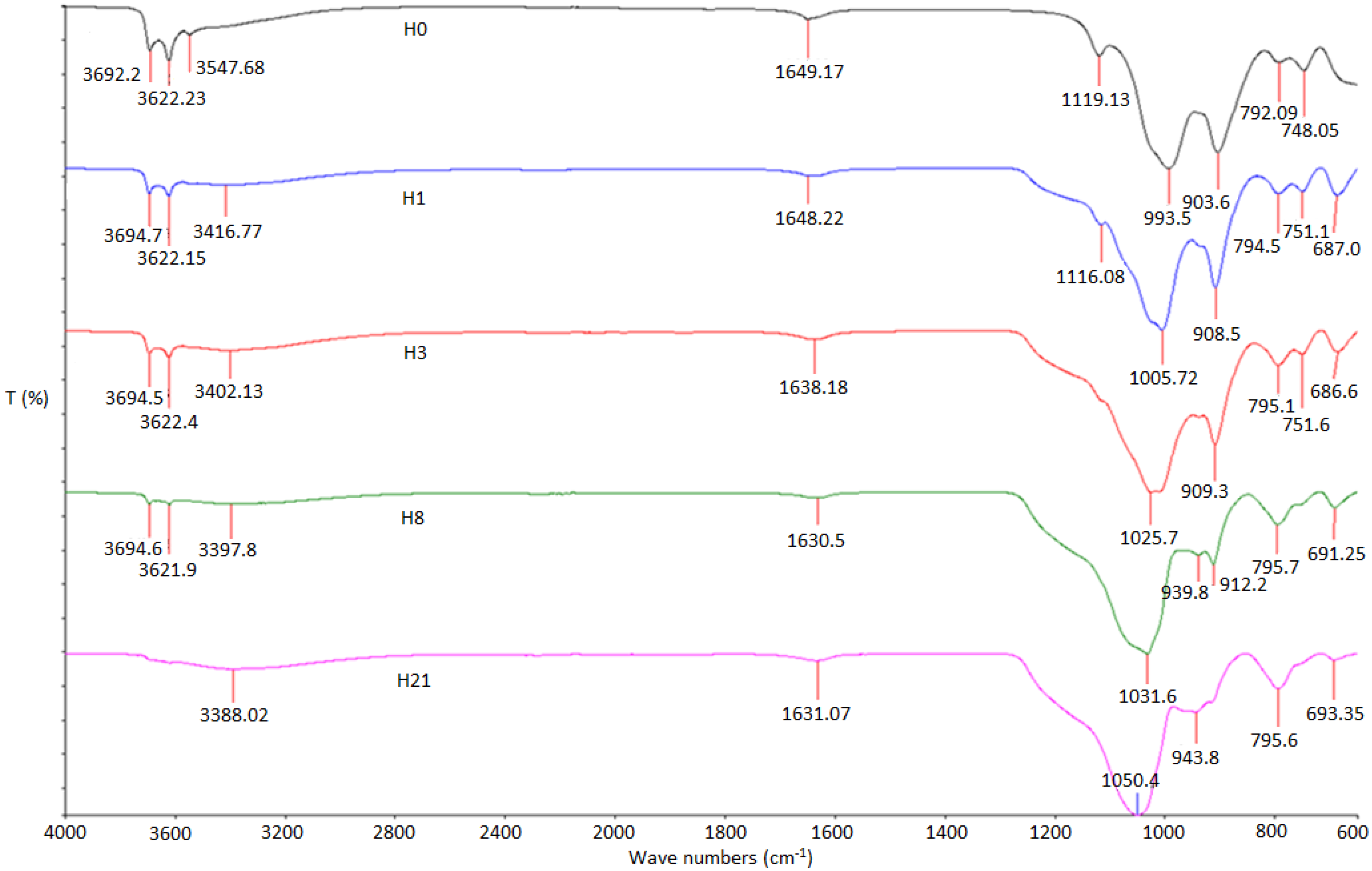
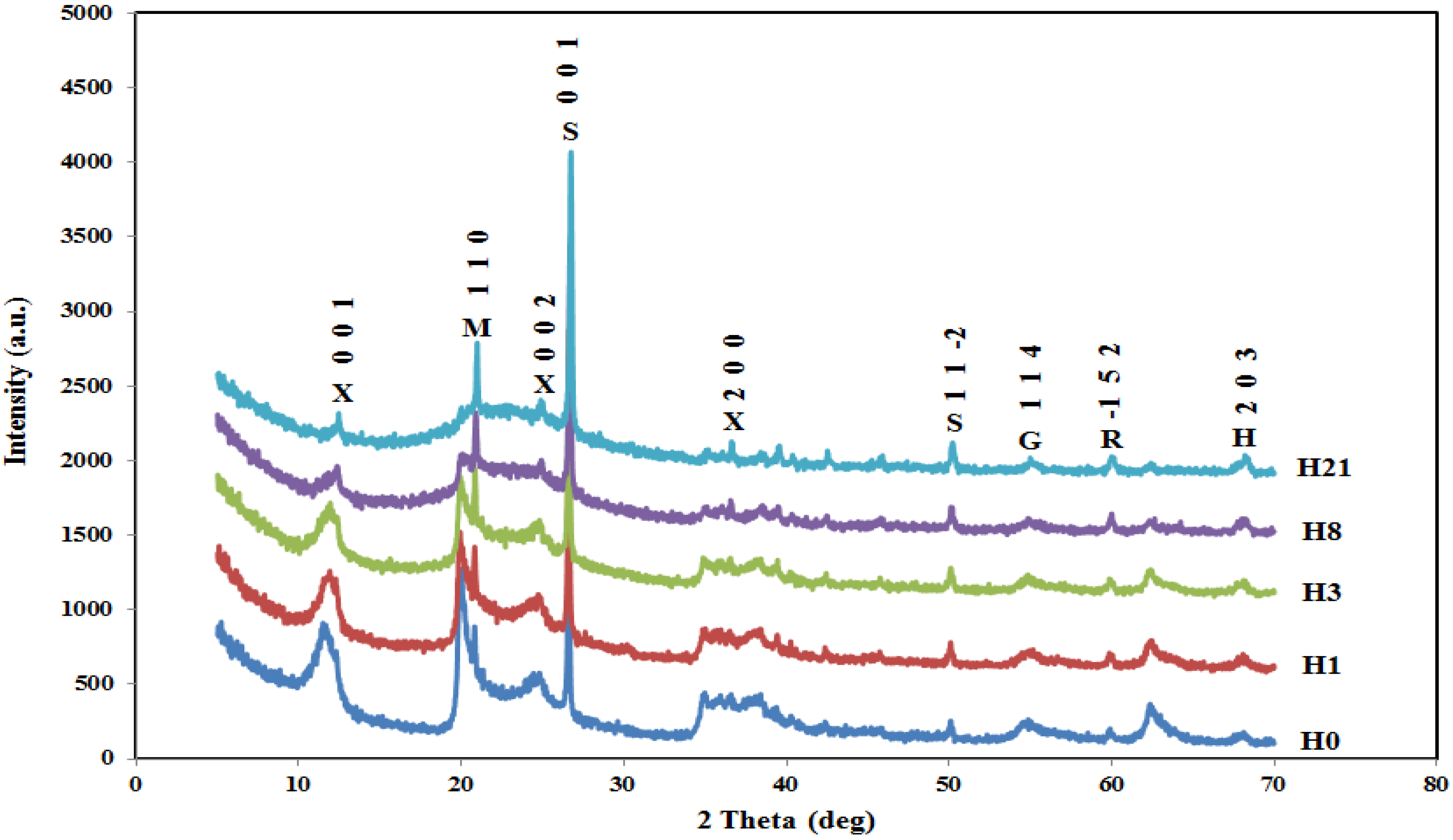





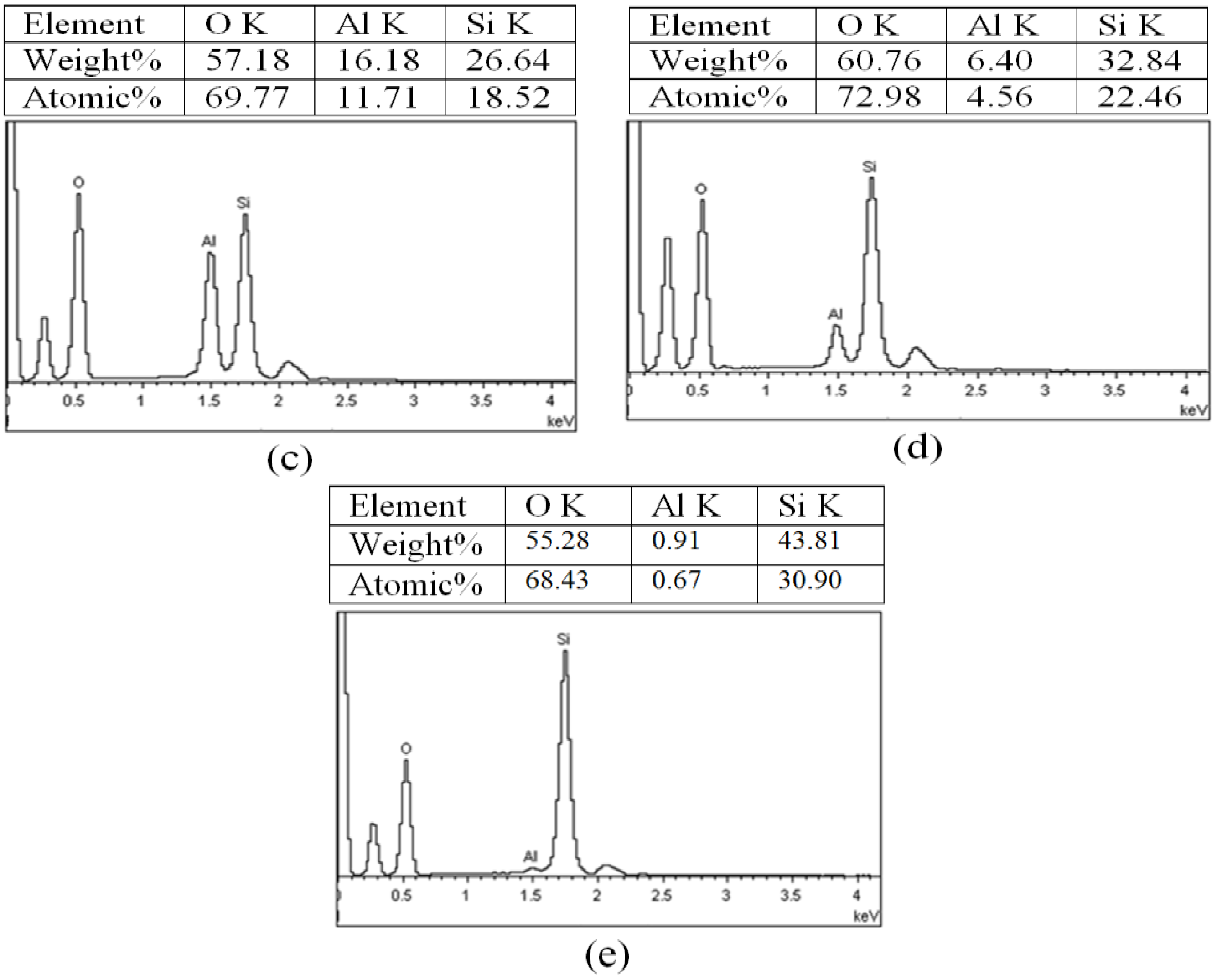

| Sample | H0 | H1 | H3 | H8 | H21 | |
|---|---|---|---|---|---|---|
| OH/O-H-Structure | O-H inner | 3692.2 | 3694.7 | 3694.5 | 3694.6 | - |
| OH-inner | 3622.23 | 3622.15 | 3622.4 | 3621.9 | - | |
| O-H intramolecular | 3547.68 | 3416.77 | 3402.13 | 3397.8 | 3388.02 | |
| C-C | 1649.17 | 1648.22 | 1638.18 | 1630.5 | 1631.07 | |
| Si-OH | 1119.13 | 1116.08 | - | - | - | |
| Si-O-Si | 993.5 | 1005.72 | 1025.7 | 1031.6 | 1050.4 | |
| Al-OH | 903.6 | 908.5 | 909.3 | 912.2 | 943.8 | |
| Al-O-OH | 792.09 | 794.5 | 795.1 | 795.7 | 795.6 | |
| 748.05 | 751.1 | 751.6 | - | - | ||
| Alcohol-OH-Out of Plane | - | 678.0 | 686.6 | 691.25 | 693.35 | |
| Sample | Elements | Zhang et al. [2] | Current Work | Percentage Error | |||
|---|---|---|---|---|---|---|---|
| Weight% | Atomic% | Weight% | Atomic% | Weight% | Atomic% | ||
| H0 | O | 61.6 | 73.4 | 59.59 | 71.76 | −3.31 | −2.28 |
| Al | 8.2 | 12.8 | 18.54 | 13.24 | +55.7 | +3.32 | |
| Si | 20.2 | 13.7 | 21.78 | 15.00 | +7.25 | +8.66 | |
| H8 | O | 60.4 | 72.8 | 60.76 | 72.98 | ||
| Al | 9.3 | 6.7 | 6.40 | 4.56 | −45.31 | −46.92 | |
| Si | 29.5 | 20.2 | 32.84 | 22.46 | +10.17 | +10.08 | |
| Nanotube/Treatment Time h (H) | Onset Temp. (°C) | Peak Temp. (°C) | End Temp. (°C) |
|---|---|---|---|
| Neat HNT (H0) | 42.77 | 95.23 | 140.35 |
| 1 h (H1) | 52.45 | 93.95 | 142.06 |
| 3 h (H3) | 49.64 | 90.17 | 140.01 |
| 8 h (H8) | 50.29 | 88.21 | 150.15 |
| 21 h (H21) | 46.37 | 90.85 | 140.30 |
| Sample | H0 | H1 | H3 | H8 | H21 |
|---|---|---|---|---|---|
| BET surface area (m2/g) | 59.04 | 222.55 | 234.53 | 306.43 | 279.47 |
| Total pore volume (cm3/g) | 0.26 | 0.45 | 0.48 | 0.71 | 0.74 |
| Micropore volume (cm3/g) | 0.001 | 0.018 | 0.019 | 0.022 | 0.018 |
| Mesopore volume (cm3/g) | 0.28 | 0.49 | 0.52 | 0.81 | 0.82 |
| Mesopore surface area (m2/g) | 67.99 | 199.66 | 215.01 | 303.57 | 281.16 |
| Average pore size (nm) | 16.73 | 9.94 | 9.84 | 10.66 | 11.74 |
| BET surface area (m2/g) [2] | 47.8 | 207.6 | 259.1 | 248.4 | 134.1 |
| Error% BET Surface area m2/g | −23.51 | −7.20 | +9.48 | −23.34 | −108.40 |
| Chemical composition | ||||
|---|---|---|---|---|
| Compound | O:SiO2 | Al:Al2O3 | Si:SiO2 | Impurities |
| Weight% | 61.19 | 18.11 | 20.11 | 0.59 |
| Physical properties | ||||
| Formula | Surface Area | Pore Volume | Density | Refractive Index |
| Al2Si2O5(OH)4·nH2O | 65 m2/g | ~1.25 mL/g | 2540 Kg/m3 | 1.54 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaaz, T.S.; Sulong, A.B.; Kadhum, A.A.H.; Nassir, M.H.; Al-Amiery, A.A. Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification. Materials 2016, 9, 620. https://doi.org/10.3390/ma9080620
Gaaz TS, Sulong AB, Kadhum AAH, Nassir MH, Al-Amiery AA. Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification. Materials. 2016; 9(8):620. https://doi.org/10.3390/ma9080620
Chicago/Turabian StyleGaaz, Tayser Sumer, Abu Bakar Sulong, Abdul Amir H. Kadhum, Mohamed H. Nassir, and Ahmed A. Al-Amiery. 2016. "Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification" Materials 9, no. 8: 620. https://doi.org/10.3390/ma9080620
APA StyleGaaz, T. S., Sulong, A. B., Kadhum, A. A. H., Nassir, M. H., & Al-Amiery, A. A. (2016). Impact of Sulfuric Acid Treatment of Halloysite on Physico-Chemic Property Modification. Materials, 9(8), 620. https://doi.org/10.3390/ma9080620







