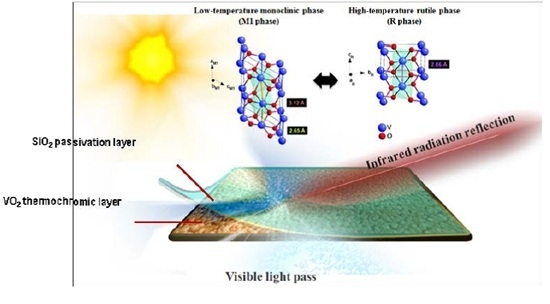
Enhanced Visible Transmittance of Thermochromic VO2 Thin Films by SiO2 Passivation Layer and Their Optical Characterization
Abstract
:1. Introduction
2. Materials and Methods
2.1. SiO2/VO2 Thin Film Preparation
2.2. Characterizations
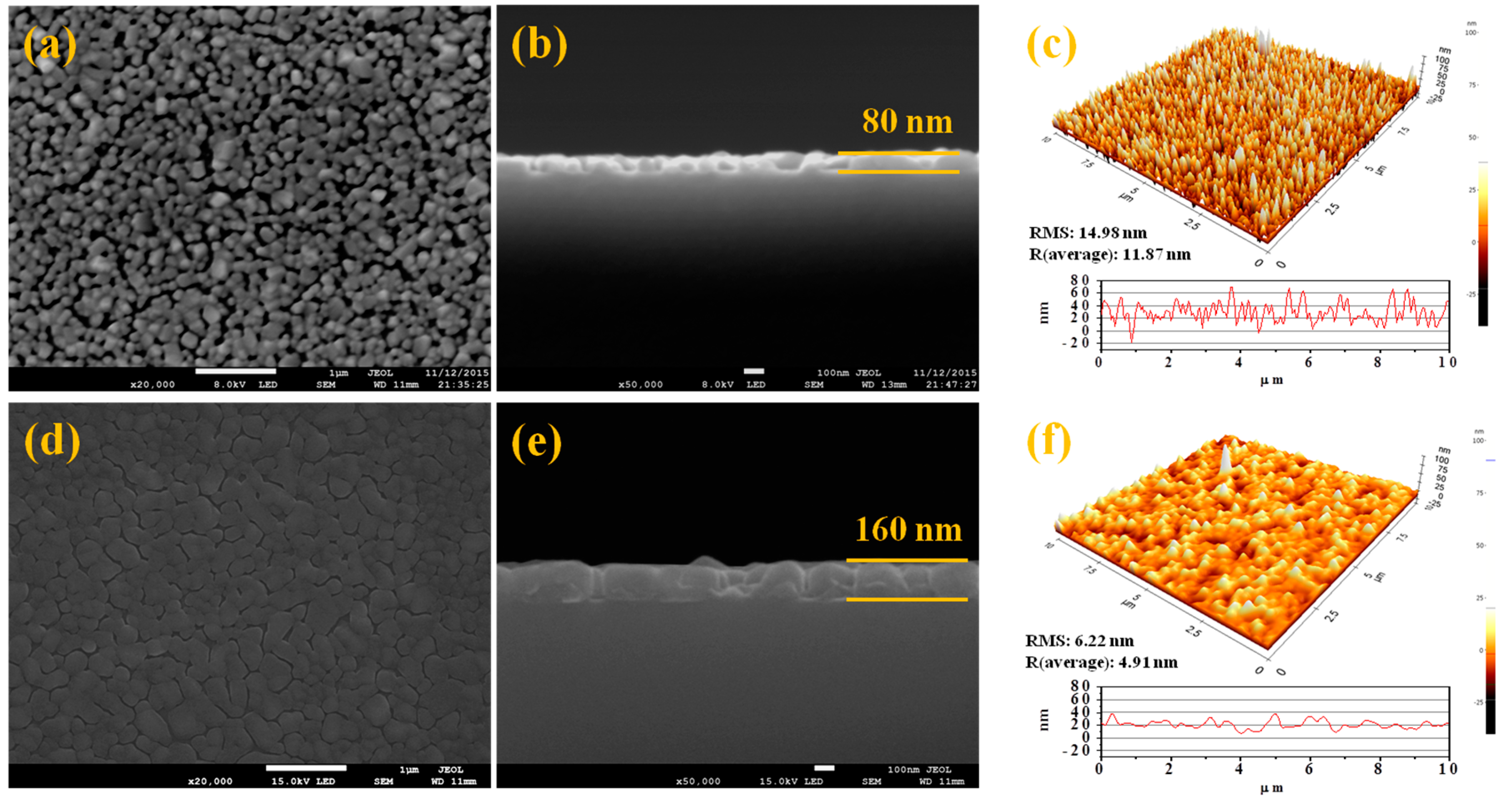
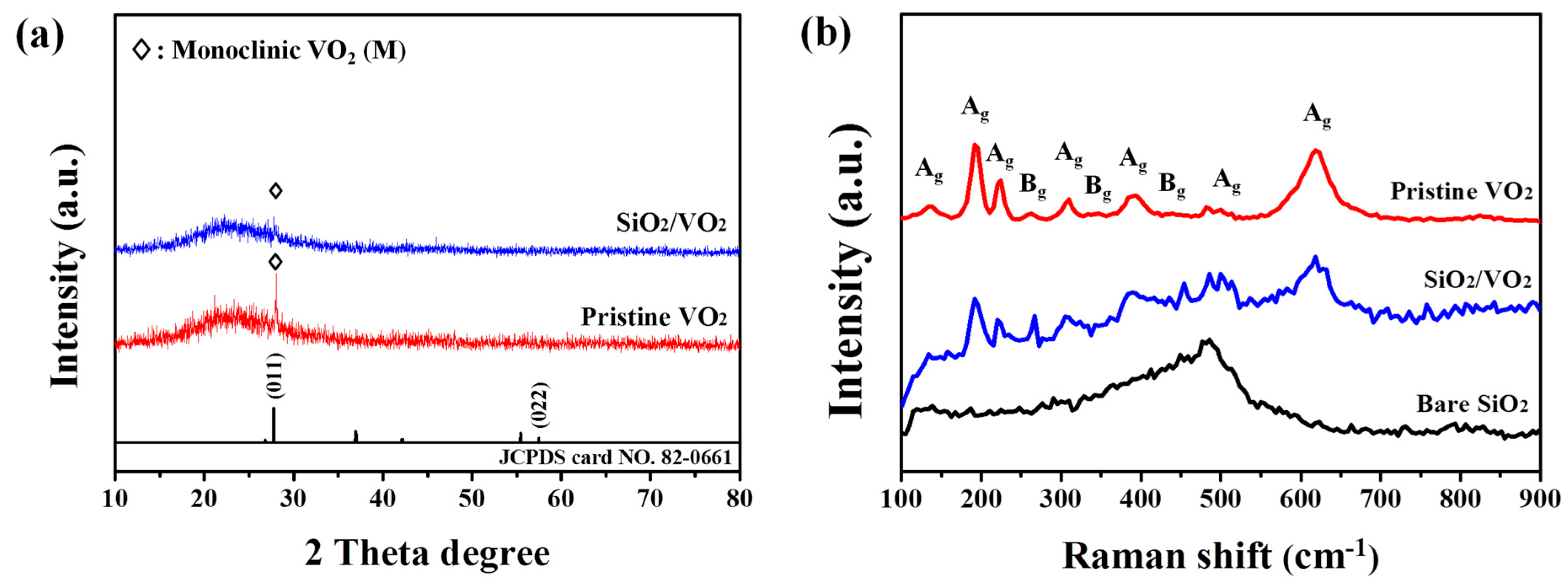
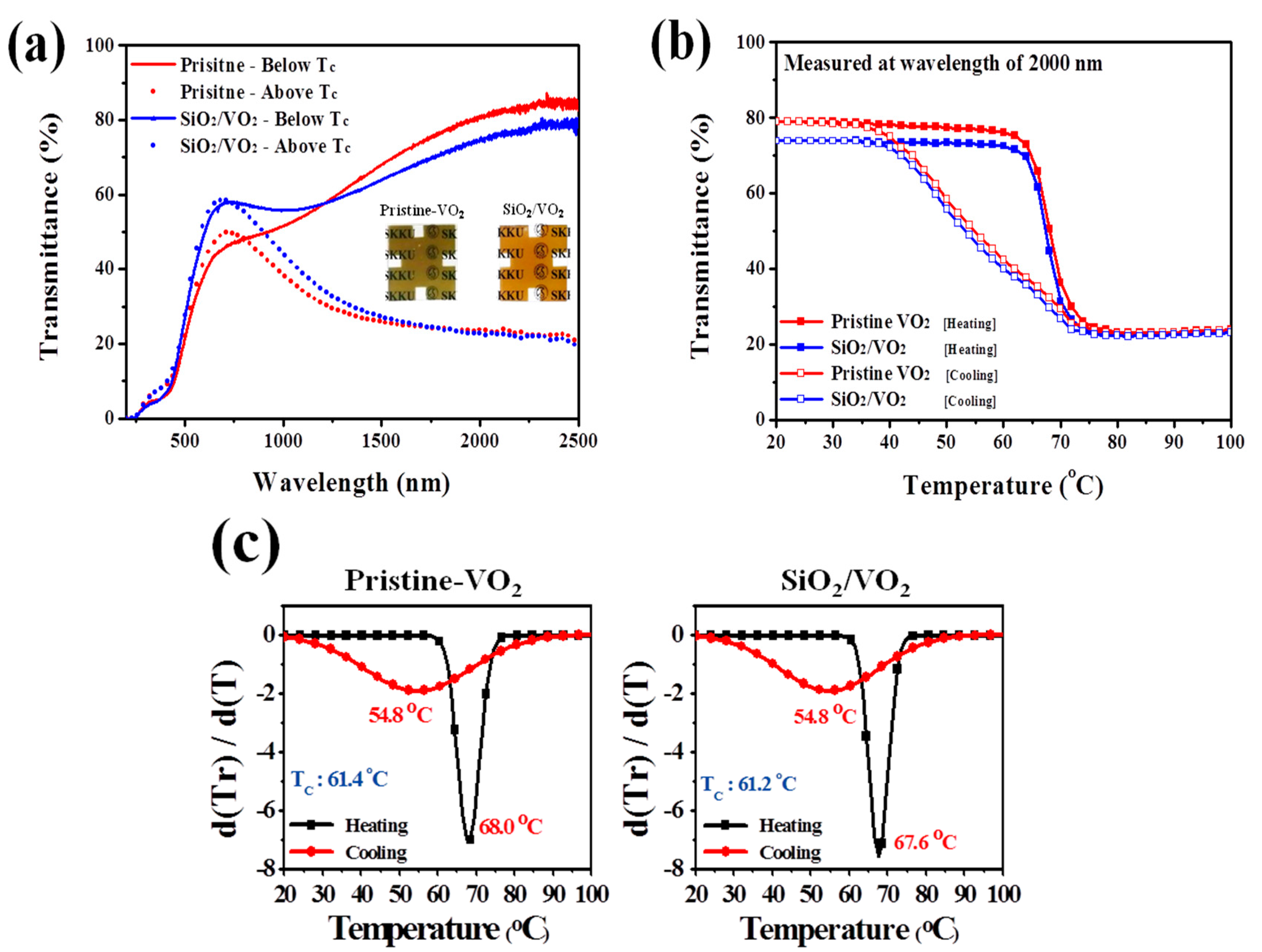
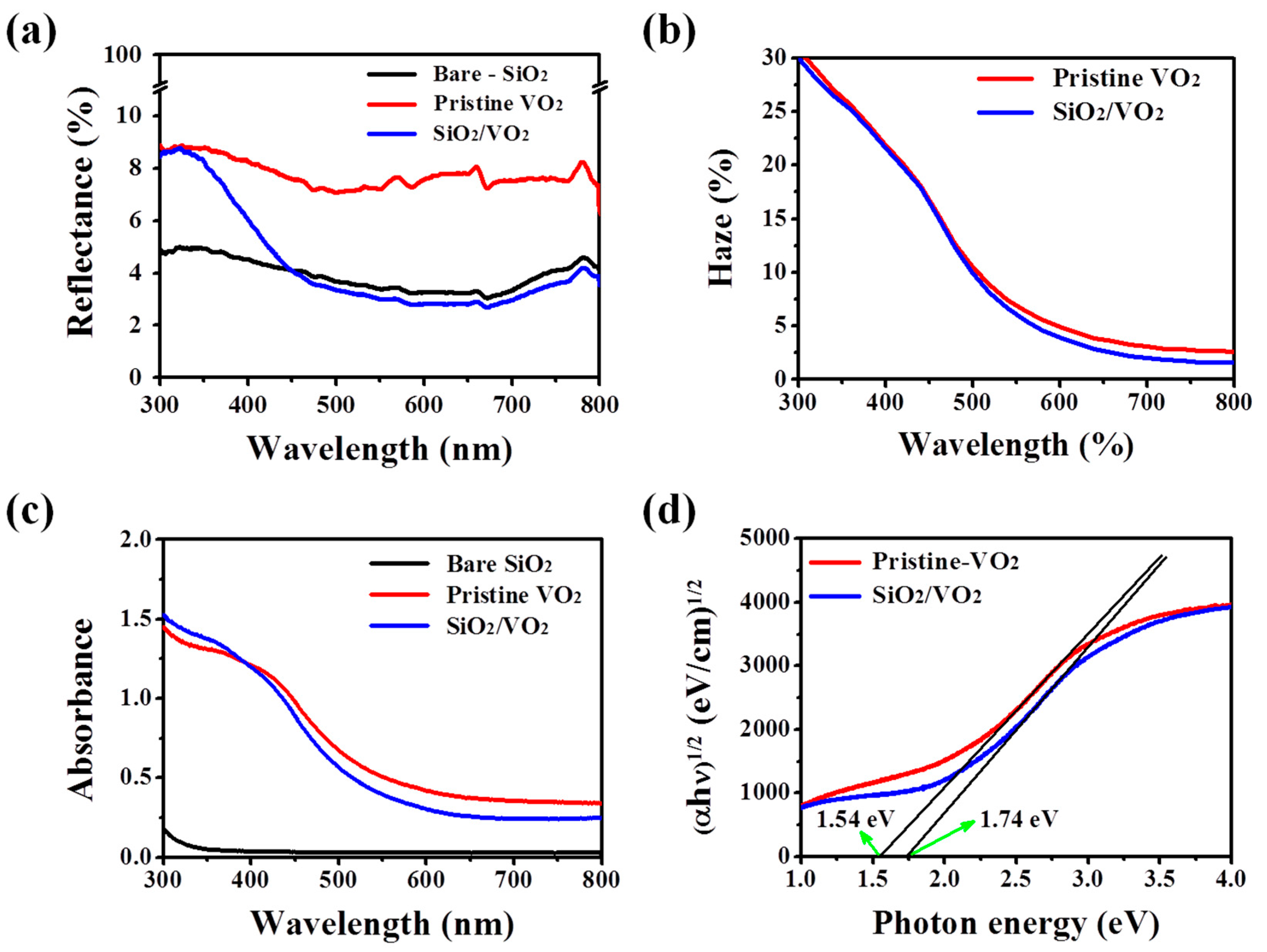
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PECVD | Plasma enhanced chemical vapor deposition |
| Tvis | Visible transmittance |
| Eos | Optical switching efficiency |
| VO2 | Vanadium dioxide |
| MIT | Metal-insulator transition |
| SEM | Scanning electron microscopy |
| XRD | X-ray diffraction |
References
- Hee, W.J.; Alghoul, M.A.; Bakhtyar, B.; OmKalthum, E.; Shameri, M.A.; Alrubaih, M.S.; Sopian, K. The role of window glazing on daylighting and energy saving in buildings. Renew. Sustain. Energy Rev. 2015, 42, 323–343. [Google Scholar] [CrossRef]
- Morin, F.J. Oxides which show a metal-to-insulator transition at the neel temperature. Phys. Rev. Lett. 1959, 3, 34–36. [Google Scholar] [CrossRef]
- Qazilbash, M.M.; Brehm, M.; Andreev, G.O.; Kim, B.J.; Yun, S.J.; Balatsky, A.V.; Maple, M.B.; Keilmann, F.; Kim, H.T.; Basov, D.N. Mott transition in VO2 revealed by infrared spectroscopy and nano-imaging. Science 2007, 318, 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Gao, Y.; Luo, H. Pure single-crystal rutile vanadium dioxide powders: Synthesis, mechanism and phase-transformation property. J. Phys. Chem. C 2008, 112, 18810–18814. [Google Scholar] [CrossRef]
- Kang, L.; Gao, Y.; Luo, H. A Novel solution process for the synthesis of VO2 thin films with excellent thermochromic properties. ACS Appl. Mater. Interfaces 2009, 1, 2211–2218. [Google Scholar] [CrossRef] [PubMed]
- Mlyuka, N.R.; Niklasson, G.A.; Granqvist, C.G. Thermochromic multilayer films of VO2 and TiO2 with enhanced transmittance. Sol. Energy Mater. Sol. Cells 2009, 93, 1685–1687. [Google Scholar] [CrossRef]
- Vernardou, D.; Louloudakis, D.; Spanakis, E.; Katsarakis, N.; Koudoumas, E. Thermochromic amorphous VO2 coatings grown by APCVD using a single-precursor. Sol. Energy Mater. Sol. Cells 2014, 128, 36–40. [Google Scholar] [CrossRef]
- Drosos, C.; Vernardou, D. Perspectives of energy materials grown by APCVD. Sol. Energy Mater. Sol. Cells 2015, 140, 1–8. [Google Scholar] [CrossRef]
- Wang, S.; Liu, M.; Kong, L.; Long, Y.; Jiang, X.; Yu, A. Recent progress in VO2 smart coating: Strategies to improve the thermochromic properties. Prog. Mater. Sci. 2016, 81, 1–54. [Google Scholar] [CrossRef]
- Zhou, Y.; Cai, Y.; Hu, X.; Long, Y. VO2/hydrogel hybrid nanothermochromic material with ultra-high solar modulation and luminous transmission. J. Mater. Chem. A 2015, 3, 1121–1126. [Google Scholar] [CrossRef]
- Burkhardt, W.; Christmann, T.; Franke, S.; Kriegseis, W.; Meister, D.; Meyer, B.K.; Niessner, W.; Schalch, D.; Scharmann, A. Tungsten and fluorine co-doping of VO2 films. Thin Sol. Films 2002, 402, 226–231. [Google Scholar] [CrossRef]
- Qazilbash, M.M.; Schafgans, A.A.; Burch, K.S.; Yun, S.J.; Chae, B.G.; Kim, B.J.; Kim, H.T.; Basov, D.N. Electrodynamics of the vanadium oxides VO2 and V2O3. Phys. Rev. B 2008, 77, 115121. [Google Scholar] [CrossRef]
- Zhou, J.; Gao, Y.; Liu, X.; Chen, Z.; Dai, L.; Cao, C.; Luo, H.; Kanahira, M.; Sunc, C.; Yanc, L. Mg-doped VO2 nanoparticles: Hydrothermal synthesis, enhanced visible transmittance and decreased metal–insulator transition temperature. Phys. Chem. Chem. Phys. 2013, 15, 7505–7511. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Dai, L.; Liu, J.; Gao, Y.; Liu, X.; Chen, Z.; Zhou, J.; Cao, C.; Han, P.; Kanahiraa, M. The visible transmittance and solar modulation ability of VO2 flexible foils simultaneously improved by Ti doping: An optimization and first principle study. Phys. Chem. Chem. Phys. 2013, 15, 17537–17543. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Bao, J.; Tao, M.; Zhu, R.; Lin, Y.; Xie, Y. Periodic porous thermochromic VO2(M) films with enhanced visible transmittance. Chem. Commun. 2013, 49, 6021–6023. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Jin, P.; Tazawa, M.; Yoshimura, K. Optimization of antireflection coating for VO2-based energy efficient window. Sol. Energy Mater. Sol. Cells 2004, 83, 29–37. [Google Scholar] [CrossRef]
- Jin, P.; Xu, G.; Tazawa, M.; Yoshimura, K. Design, formation and characterization of a novel multifunctional window with VO2 and TiO2 coatings. Appl. Phys. A 2003, 77, 455–459. [Google Scholar]
- Kusano, E.; Theil, J.A. Effects of microstructure and nonstoichiometry on electrical properties of vanadium dioxide films. J. Vac. Sci. Technol. A 1989, 7, 1314–1317. [Google Scholar] [CrossRef]
- Griffiths, C.H.; Eastwood, H.K. Influence of stoichiometry on the metal-semiconductor transition in vanadium dioxide. J. Appl. Phys. 1974, 45, 2201–2206. [Google Scholar] [CrossRef]
- Jin, S.B.; Lee, J.S.; Choi, Y.S.; Choi, I.S.; Han, J.G. High-rate deposition and mechanical properties of SiOx film at low temperature by plasma enhanced chemical vapor deposition with the dual frequencies ultra high frequency and high frequency. Thin Sol. Films 2011, 519, 6334–6338. [Google Scholar] [CrossRef]
- Wang, N.; Liu, S.; Zeng, X.T.; Magdassi, S.; Long, Y. Mg/W-codoped vanadium dioxide thin films with enhanced visible transmittance and low phase transition temperature. J. Mater. Chem. C 2015, 3, 6771–6777. [Google Scholar] [CrossRef]
- Li, J.; Dho, J. Anomalous optical switching and thermal hysteresis behaviors of VO2 films on glass substrate. Appl. Phys. Lett. 2011, 99, 231909. [Google Scholar] [CrossRef]
- Petrov, G.I.; Yakovlev, V.V.; Squier, J. Raman microscopy analysis of phase transformation mechanisms in vanadium dioxide. Appl. Phys. Lett. 2002, 81, 1023–1025. [Google Scholar] [CrossRef]
- Donev, E.U.; Lopez, R.; Feldman, L.C.; Haglund, R.F. Confocal raman microscopy across the metal-insulator transition of single vanadium dioxide nanoparticles. Nano Lett. 2009, 9, 702–106. [Google Scholar] [CrossRef] [PubMed]
- Vernardou, D.; Louloudakis, D.; Spanakis, E.; Katsarakis, N.; Koudoumas, E. Functional Properties of APCVD VO2 Layers. Int. J. Thin Films Sci. Technol. 2015, 4, 187–191. [Google Scholar]
- Du, J.; Gao, Y.; Luo, H.; Kang, L.; Zhang, Z.; Chen, Z.; Cao, C. Significant changes in phase-transition hysteresis for Ti-doped VO2 films prepared by polymer-assisted deposition. Sol. Energy Mater. Sol. Cells 2010, 95, 469–475. [Google Scholar] [CrossRef]
- Kim, E.; Jiang, Z.; No, K. Measurement and calculation of optical band gap of chromium aluminum oxide films. Jpn. J. Appl. Phys. 2000, 39, 4820–4825. [Google Scholar] [CrossRef]
- Jiang, M.; Li, Y.; Li, S.; Zhou, H.; Cao, X.; Bao, S.; Gao, Y.; Luo, H.; Jin, P. Room temperature optical constants and band gap evolution of phase pure M1-VO2 thin films deposited at different oxygen partial pressures by reactive magnetron sputtering. J. Nanomater. 2014, 2014, 183954. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.-H.; Nam, S.-H.; Lee, J.W.; Boo, J.-H. Enhanced Visible Transmittance of Thermochromic VO2 Thin Films by SiO2 Passivation Layer and Their Optical Characterization. Materials 2016, 9, 556. https://doi.org/10.3390/ma9070556
Yu J-H, Nam S-H, Lee JW, Boo J-H. Enhanced Visible Transmittance of Thermochromic VO2 Thin Films by SiO2 Passivation Layer and Their Optical Characterization. Materials. 2016; 9(7):556. https://doi.org/10.3390/ma9070556
Chicago/Turabian StyleYu, Jung-Hoon, Sang-Hun Nam, Ji Won Lee, and Jin-Hyo Boo. 2016. "Enhanced Visible Transmittance of Thermochromic VO2 Thin Films by SiO2 Passivation Layer and Their Optical Characterization" Materials 9, no. 7: 556. https://doi.org/10.3390/ma9070556
APA StyleYu, J.-H., Nam, S.-H., Lee, J. W., & Boo, J.-H. (2016). Enhanced Visible Transmittance of Thermochromic VO2 Thin Films by SiO2 Passivation Layer and Their Optical Characterization. Materials, 9(7), 556. https://doi.org/10.3390/ma9070556