Coating Methods for Surface Modification of Ammonium Nitrate: A Mini-Review
Abstract
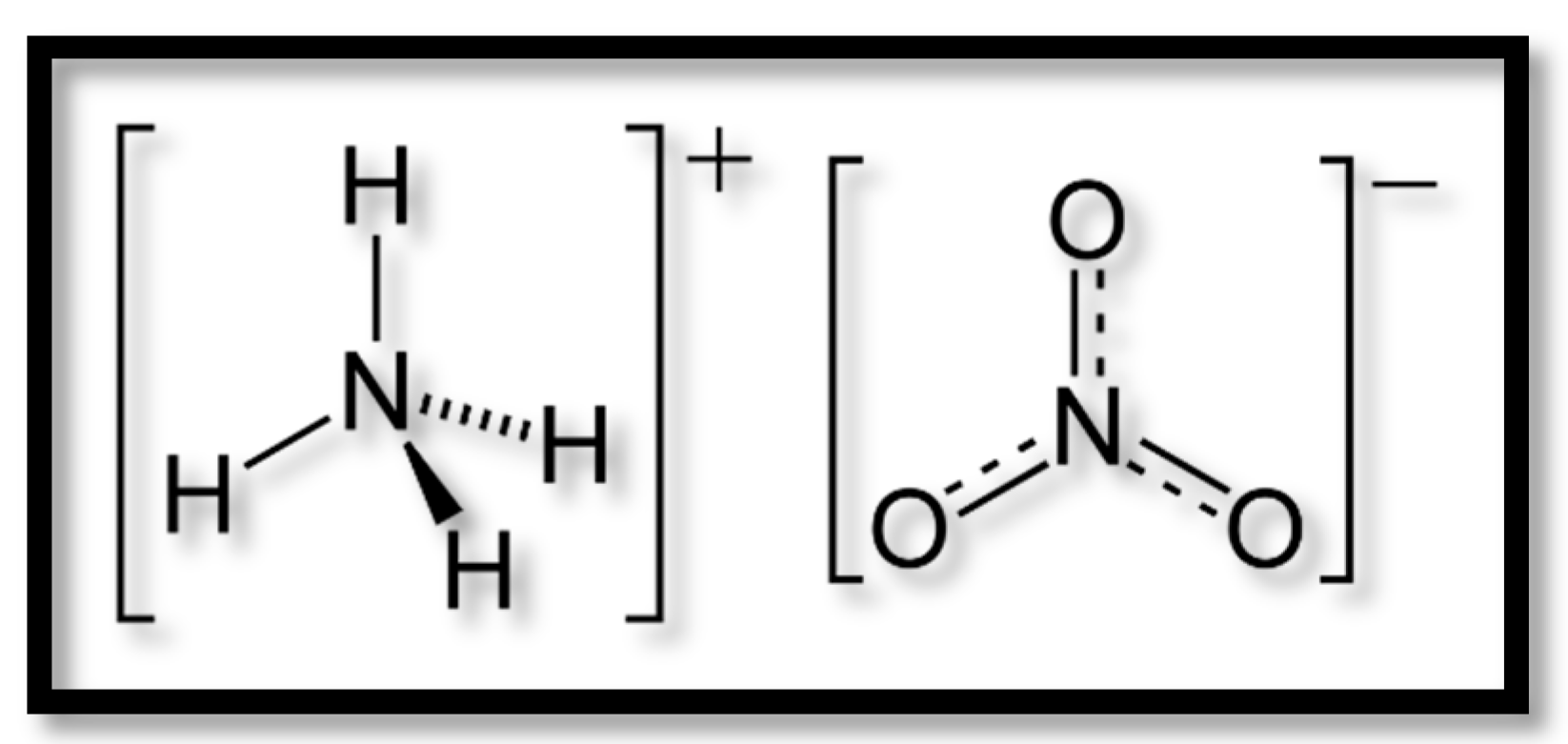
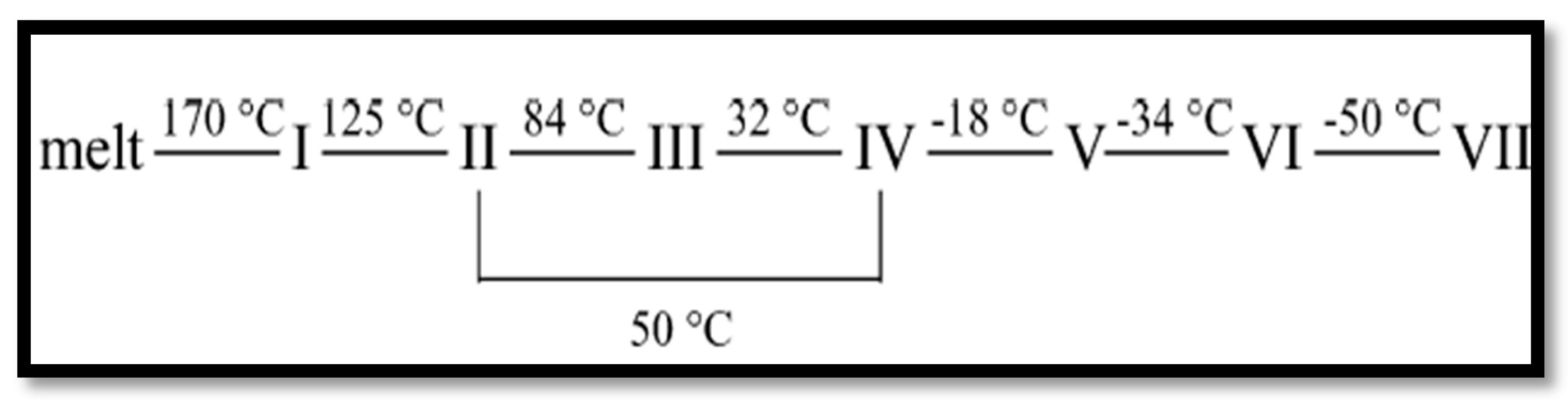
:1. Introduction
2. Coating Methods
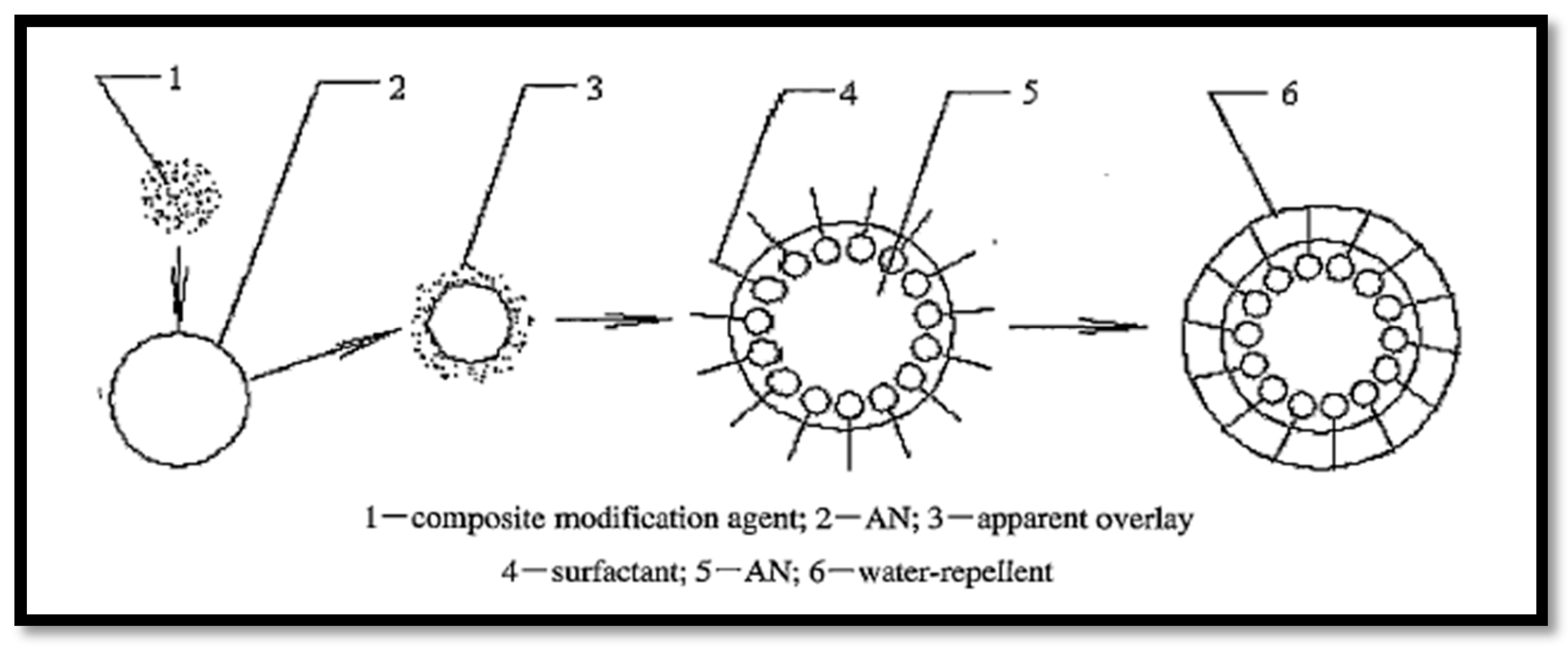
2.1. Physical Coating Method
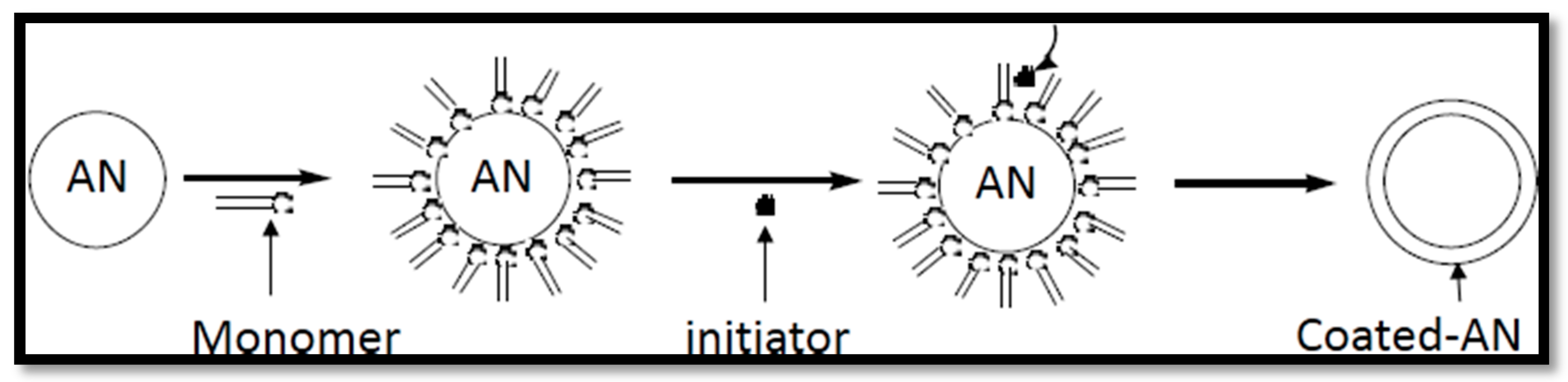
2.2. Chemical Coating Method
2.3. Encapsulation Methods
- (1)
- Cellulose derivatives, such as nitrocellulose, ethyl cellulose.
- (2)
- Resin and plastics, such as poly methyl methacrylate, acrylic acid resin, polyvinyl acetylate, polystyrene, etc.
- (3)
- Elastomeric polymer such as polyurethane elastomer.
- (1)
- Mechanical and physical methods such as the spray-drying method.
- (2)
- Physical methods such as the phase separation method.
- (3)
- Chemical methods such as the precipitation polymerization method.
3. Conclusions
Conflicts of Interest
References
- Singh, G.; Felix, S. Evaluation of transition metal salts of NTO as burning rate modifier for HRPB-AN composite solid propellants. Combust. Flame 2003, 132, 422–432. [Google Scholar] [CrossRef]
- Kim, J.-K.; Choi, S.-I.; Kim, E.J.; Kim, J.H.; Koo, K.-K. Preparation of Spherical Ammonium Nitrate Particles by Melt Spray. Ind. Eng. Chem. Res. 2010, 49, 12632–12637. [Google Scholar] [CrossRef]
- Oommen, C.; Jain, S. Ammonium nitrate: A promising rocket propellant oxidizer. J. Hazard. Mater. 1999, 67, 253–281. [Google Scholar] [CrossRef]
- Heller, A.N.; Cuffe, S.T.; Goodwin, D.R. Inorganic chemical industry. In Sources of Air Pollution and Their Control: Air Pollution; Academic Press: Cambridge, MA, USA, 2015; p. 191. [Google Scholar]
- Marlair, G.; Kordek, M.-A. Safety and security issues relating to low capacity storage of AN-based fertilizers. J. Hazard. Mater. 2005, 123, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Mellor, J.W. A Comprehensive Treatise on Inorganic and Theoretical Chemistry; Longmans, Green: London, UK, 1922; Volume 2. [Google Scholar]
- Oxley, J.C.; Smith, J.L.; Rogers, E.; Yu, M. Ammonium nitrate: Thermal stability and explosivity modifiers. Thermochim. Acta 2002, 384, 23–45. [Google Scholar] [CrossRef]
- Kajiyama, K.; Izato, Y.-I.; Miyake, A. Thermal characteristics of ammonium nitrate, carbon, and copper (II) oxide mixtures. J. Therm. Anal. Calorim. 2013, 113, 1475–1480. [Google Scholar] [CrossRef]
- Clay, R.B. Water-in-Oil Blasting Composition. U.S. Patent 4,111,727, 5 September 1978. [Google Scholar]
- Vargeese, A.A.; Muralidharan, K.; Krishnamurthy, V. Thermal stability of habit modified ammonium nitrate: Insights from isoconversional kinetic analysis. Thermochim. Acta 2011, 524, 165–169. [Google Scholar] [CrossRef]
- Kwok, Q.S.; Kruus, P.; Jones, D.E. Wettability of ammonium nitrate prills. J. Energetic Mater. 2004, 22, 127–150. [Google Scholar] [CrossRef]
- Davey, R.; Ruddick, A.J.; Guy, P.D.; Mitchell, B.; Maginn, S.J.; Polywka, L.A. The IV-III polymorphic phase transition in ammonium nitrate: A unique example of solvent mediation. J. Phys. D Appl. Phys. 1991, 24, 176. [Google Scholar] [CrossRef]
- Borman, S. Advanced energetic materials emerge for military and space applications. Chem. Eng. News 1994, 72, 18–22. [Google Scholar] [CrossRef]
- Matsunaga, H.; Habu, H.; Miyake, A. Thermal decomposition of the high-performance oxidizer ammonium dinitramide under pressure. J. Therm. Anal. Calorim. 2014, 116, 1227–1232. [Google Scholar] [CrossRef]
- Heintz, T.; Pontius, H.; Aniol, J.; Birke, C.; Leisinger, K.; Reinhard, W. Ammonium Dinitramide (ADN)—Prilling, Coating, and Characterization. Propellants Explos. Pyrotech. 2009, 34, 231–238. [Google Scholar] [CrossRef]
- Cui, J.; Han, J.; Wang, J.; Huang, R. Study on the crystal structure and hygroscopicity of ammonium dinitramide. J. Chem. Eng. Data 2010, 55, 3229–3234. [Google Scholar] [CrossRef]
- Mathew, S.; Krishnan, K.; Ninan, K. Effect of energetic materials on thermal decomposition of phase-stabilised ammonium nitrate-An eco-friendly oxidiser. Def. Sci. J. 1999, 49, 65–69. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Dave, P.N. Review on thermal decomposition of ammonium nitrate. J. Energetic Mater. 2013, 31, 1–26. [Google Scholar] [CrossRef]
- Gunawan, R.; Freij, S.; Zhang, D.K.; Beach, F.; Littlefair, M. A mechanistic study into the reactions of ammonium nitrate with pyrite. Chem. Eng. Sci. 2006, 61, 5781–5790. [Google Scholar] [CrossRef]
- Komunjer, L.; Affolter, C. Absorption-evaporation kinetics of water vapour on highly hygroscopic powder: Case of ammonium nitrate. Powder Technol. 2005, 157, 67–71. [Google Scholar] [CrossRef]
- Harris, J. Hygroscopicity of Ammonium Nitrate Samples; DTIC Document: Fort Belvoir, VA, USA, 1970. [Google Scholar]
- Sinditskii, V.P.; Egorshev, V.Y.; Levshenkov, A.I.; Serushkin, V.V. Ammonium nitrate: Combustion mechanism and the role of additives. Propellants Explos. Pyrotech. 2005, 30, 269–280. [Google Scholar] [CrossRef]
- Komunjer, L.; Pezron, I. A new experimental method for determination of solubility and hyper-solubility of hygroscopic solid. Powder Technol. 2009, 190, 75–78. [Google Scholar] [CrossRef]
- Ye, Z.-W. Research on Improvement in Hydroscopicity of Ammonium Nitrate with Surfactants. Fine Chem. Dalian 2001, 18, 70–71. [Google Scholar]
- Rosen, M.J.; Kunjappu, J.T. Surfactants and Interfacial Phenomena; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Myers, D. Surfactant Science and Technology; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Chen, T.; Lv, C. Study on Reducing AN Absorptivity with Surface Active Agents and Additives. Chin. J. Explos. Propellants 1998, 4, 19–21. [Google Scholar]
- Chattopadhyay, A.K. Coating for Ammonium Nitrate Prills. U.S. Patent 5,567,910, 22 October 1996. [Google Scholar]
- Patrick, F.D.; Rapstein, K.T.; Schrieber, C.F.; Wilson, J.S. Coated Explosive Comprising Ammonium Nitrate. U.S. Patent 3,287,189, 22 November 1966. [Google Scholar]
- Huang, W.Y.; Yan, S.L.; Xie, X.H.; Guo, Z. The production and Implication of Composite Modified Water-Resistant Powdery Ammonium Nitrate. In Proceedings of the International Conference on Theory and Practice of Energetic Materials (IASPEP 2003), Guilin, China, 15–18 Octomber 2003; pp. 68–73.
- Damse, R. Waterproofing materials for ammonium nitrate. Def. Sci. J. 2004, 54, 483–492. [Google Scholar] [CrossRef]
- Hu, K.L.; Luo, N.; Huang, W.Y.; Ma, X.M.; Xu, X.F. Test and Study on the Modification of Ammonium N itrate by Coating Its Surface. Explos. Mater. 2006, 35, 14–17. [Google Scholar]
- Ye, W. Research on properties of ammonium nitrate modified by coupling cationic surfactant. Inorg. Chem. Ind. 2007, 39, 26. [Google Scholar]
- Xiong, Y.-T.; Liu, Z.-L. Improvement of the Hygroscopicity of AN by Modified Paraffin. Chin. J. Explos. Propellants 2013, 4, 50–53. [Google Scholar]
- Lu, L.Y.; Yang, L.; Zhou, Y.K. Study on Contact Angle for Characterizing the Modifying Effect of Powder Ammonium Nitrate. Coal Mine Blasting 2009, 4, 4–6. [Google Scholar]
- Lu, L.-Y.; Yang, L. Study on the Interaction Mechanism of Modified Ammonium Nitrate by O tadecylamine. Initiat. Pyrotech. 2010, 2, 41–43. [Google Scholar]
- Wen, Y. Study on The Technics of Surface Modification of Ammonium Nitrate. Master’s Thesis, Hunan University, Changsha, China, 2003. [Google Scholar]
- Malash, G.; Hashem, H. Improving the properties of ammonium nitrate fertilizer using additives. Alex. Eng. J. 2005, 44, 685–693. [Google Scholar]
- Zhang, X.D.; Li, J.M.; Yang, R.J.; Zhao, X.Q. Surface Modification of Phase Stabilized Ammonium Nitrate and Its Application in Solid Composite Propellants. Chin. J. Explos. Propellants 2009, 32, 5–9. [Google Scholar]
- Masao, K.; Asaji, K.; Noriynki, M.; Tsutomu, Y. Encapsulation Method. U.S. Patent 3,691,090, 23 Octomber 2014. [Google Scholar]
- Umer, H.; Nigam, H.; Tamboli, A.M.; Nainar, M.S.M. Microencapsulation: Process, techniques and applications. Int. J. Res. Pharm. Biomed. Sci. 2011, 2, 474–480. [Google Scholar]
- Gang, Y.P.; Cang, S.W.; Zhang, Y.H.; Li, Q.S. Self assembling Monolayers for Anti-moisture Absorption of Ammonium Nitrate. Chin. J. Appl. Chem. 2000, 17, 186–188. [Google Scholar]
- Yin, P.-G.; Jin, G.-L.; Zhang, Y.-H.; Li, Q.-S. Structure Characterization of Self-Assembled Monolayer Film on the Surface of Ammonium Nitrate. J. Beijing Inst. Technol. 2000, 20, 643–646. [Google Scholar]
- Yin, Y.-X.; Yang, R.-J. Modification of AN by using microencapsulation technology. Liaoning Chem. Ind. 2001, 30, 139. [Google Scholar] [CrossRef]
- Xu, J.-M.; Liu, Z.-L.; Zhai, D.-D. Modification of inorganic salts by encapsulation method. Liaoning Chem. Ind. 2003, 32, 142. [Google Scholar]
- Zhang, J. Study on Properties of the Coated AN with Polyvinyl Butyral. Chin. J. Explos. Propellants 2001, 24, 41–43. [Google Scholar]
- Wei, Y.; Cai, B.H. Study on Surface Modification of Ammonium Nitrate. In Advanced Materials Research; Trans Tech Publications: Pfaffikon, Switzerland, 2011. [Google Scholar]
- Zhang, J.; Yang, R.-J. Study on Surface Properties of Coated Ammonium Nitrate. Energetic Mater. 2004, 12, 1–5. [Google Scholar]
- Zhang, J.Z.; Wang, X.Q.; Lin, D.-H. Study on Hygroscopicity of Ammonium Nitrate Particle Coated by Precipitation Polymerization of Styrene. Adv. Fine Petrochem. 2008, 12, 35–37. [Google Scholar]
- Yu, Z.C.; Zhang, Y.J. Coating and modification of ammonium nitrate particles. Master’s Thesis, Nanjing University of Science & Technology, Nanjing, China, 2013. [Google Scholar]
| No. | Year | Absorption Moisture Rate (%) | Mass Ratio of Coating Layer (%) | Absorption Moisture Test Condition | Decline of Absorption Moisture (%) | Ref. |
|---|---|---|---|---|---|---|
| 1 | 2003 | 1.62% | 4% | T = 20 °C, RH = 83%, t = 48 h | 64.78% | [30] |
| 2 | 2004 | 7% | 1% | T = 20 °C, RH = 65%, t = 96 h | 80% | [31] |
| 3 | 2006 | 2.174% | 2.17% | T = 25 °C, RH = 90%, t = 8 h | 65.28% | [32] |
| 4 | 2007 | 5.27% | - | T = 25 °C, RH = 90%, t = 30 h | 69.02% | [33] |
| 5 | 2013 | 3.04% | 2.5% | T = 30 °C, RH = 90%, t = 24 h | 88% | [34] |
| No. | Year | Absorption Moisture Rate | Mass Ratio of Coating Layer | Absorption Moisture Test Condition | Decline of Absorption Moisture | Ref. |
|---|---|---|---|---|---|---|
| 1 | 2003 | 11.4% | - | T = 35 °C, RH = 92%, t = 24 h | 38.7% | [37] |
| 2 | 2005 | 0.21% | 0.3% | T = 35 °C, RH = 55%, t = 24 h | 56.25% | [38] |
| 3 | 2009 | 10.92% | - | T = 20 °C, RH = 88%, t = 8 h | 15.08% | [39] |
| No. | Year | Absorption Moisture Rate | Mass Ratio of Coating Layer | Absorption Moisture Test Condition | Decline of Absorption Moisture | Ref. |
|---|---|---|---|---|---|---|
| 1 | 2003 | 9.91% | - | T = 32 °C, RH = 90%, t = 24 h | 22.50% | [37] |
| 2 | 2004 | 2.8427% | 1.5% | T = 30 °C, RH = 94%, t = 8 h | 49% | [48] |
| 3 | 2008 | 0.623% | - | T = 20 °C, RH = 75%, t = 12 h | 88% | [49] |
| 4 | 2011 | 0.33% | - | T = 30 °C, RH = 75%, t = 24 h | 30.6% | [47] |
| 5 | 2013 | 8.78% | 0.48% | T = 35 °C, RH = 92%, t = 24 h | 28.35% | [50] |
| Method | Advantages | Disadvantages |
|---|---|---|
| Physical coating |
|
|
| Chemical coating |
|
|
| Encapsulation coating |
|
|
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elzaki, B.I.; Zhang, Y.J. Coating Methods for Surface Modification of Ammonium Nitrate: A Mini-Review. Materials 2016, 9, 502. https://doi.org/10.3390/ma9070502
Elzaki BI, Zhang YJ. Coating Methods for Surface Modification of Ammonium Nitrate: A Mini-Review. Materials. 2016; 9(7):502. https://doi.org/10.3390/ma9070502
Chicago/Turabian StyleElzaki, Baha I., and Yue Jun Zhang. 2016. "Coating Methods for Surface Modification of Ammonium Nitrate: A Mini-Review" Materials 9, no. 7: 502. https://doi.org/10.3390/ma9070502
APA StyleElzaki, B. I., & Zhang, Y. J. (2016). Coating Methods for Surface Modification of Ammonium Nitrate: A Mini-Review. Materials, 9(7), 502. https://doi.org/10.3390/ma9070502





