Immobilization of Magnetic Nanoparticles onto Amine-Modified Nano-Silica Gel for Copper Ions Remediation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
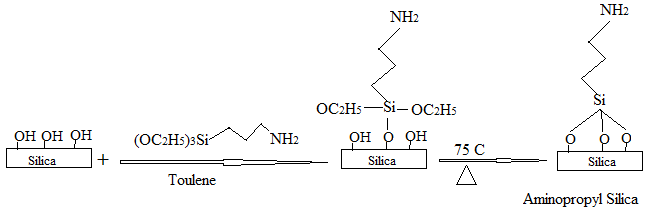
2.2.1. Preparation of Amine-Functionalized Nano-Silica Gel
2.2.2. Preparation of Magnetic Amine-Functionalized Nano-Silica Gel
2.2.3. Characterization of the Magnetic Amine-Functionalized Nano-Silica Gel
2.2.4. Adsorption Technique for Copper Remediation from Polluted Water Using the MANSG Nano-Hybrid
2.2.5. Desorption of Copper Ions from the Contaminated MANSG Nano-Hybrid after the Treatment Process
3. Results and Discussion
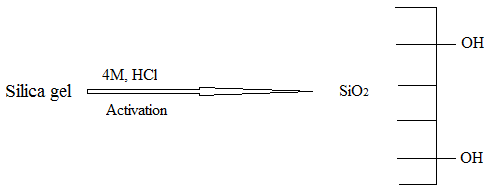
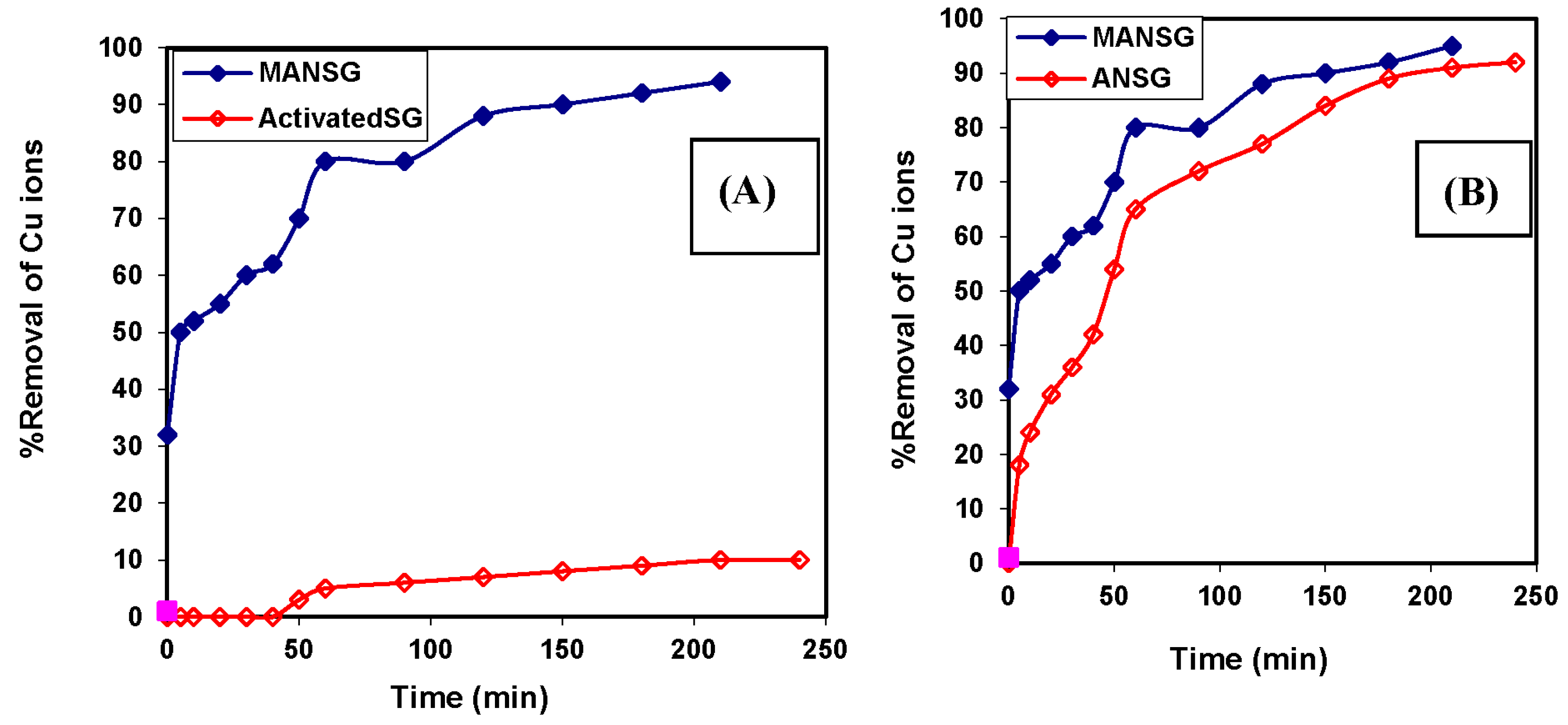
3.1. Preparation of the Amine-Functionalized Nano-Silica Gel Silica Gel Activation Process
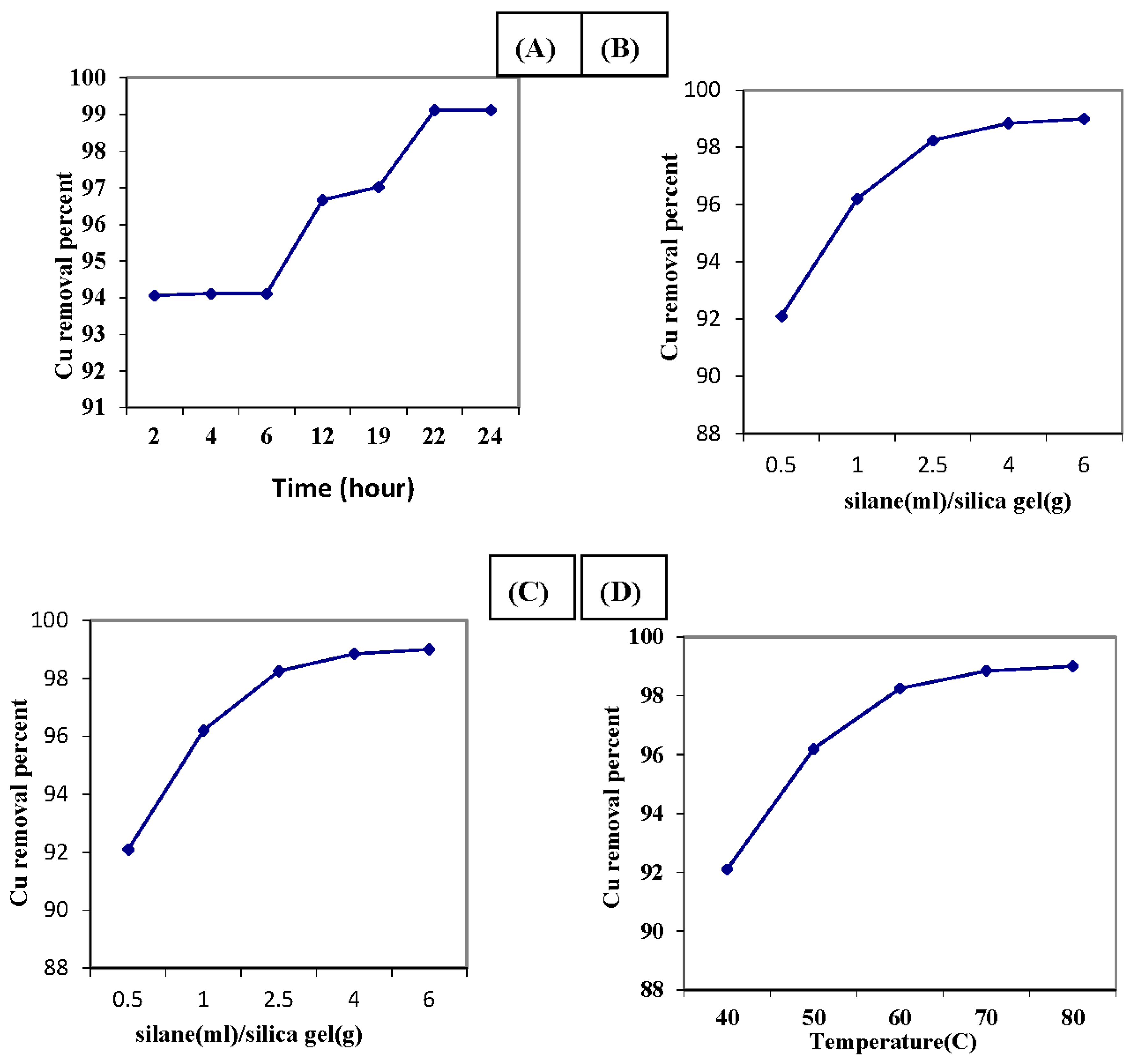
3.2. Optimization of the Chemical Modification Process of the Activated Silica Gel
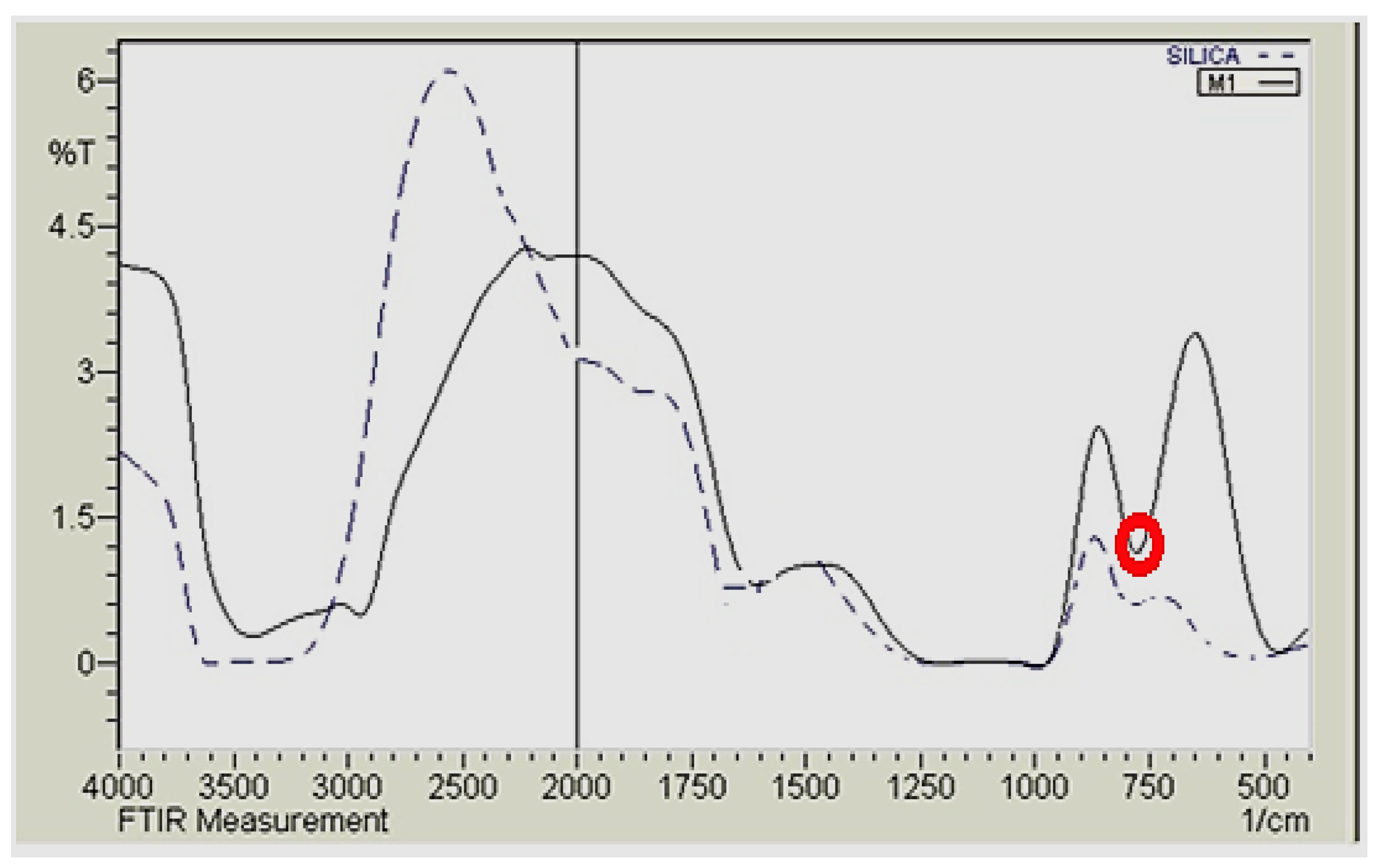
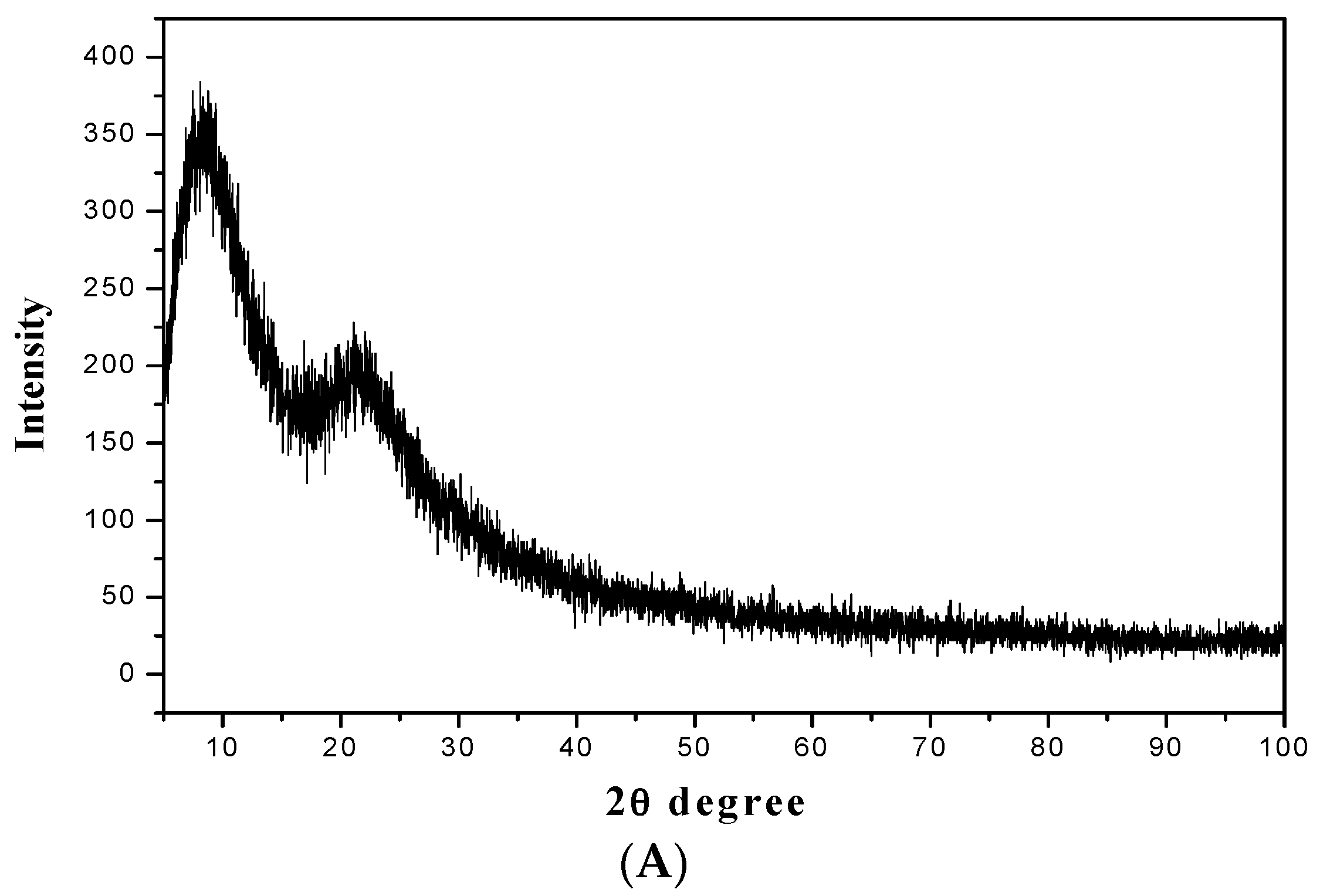
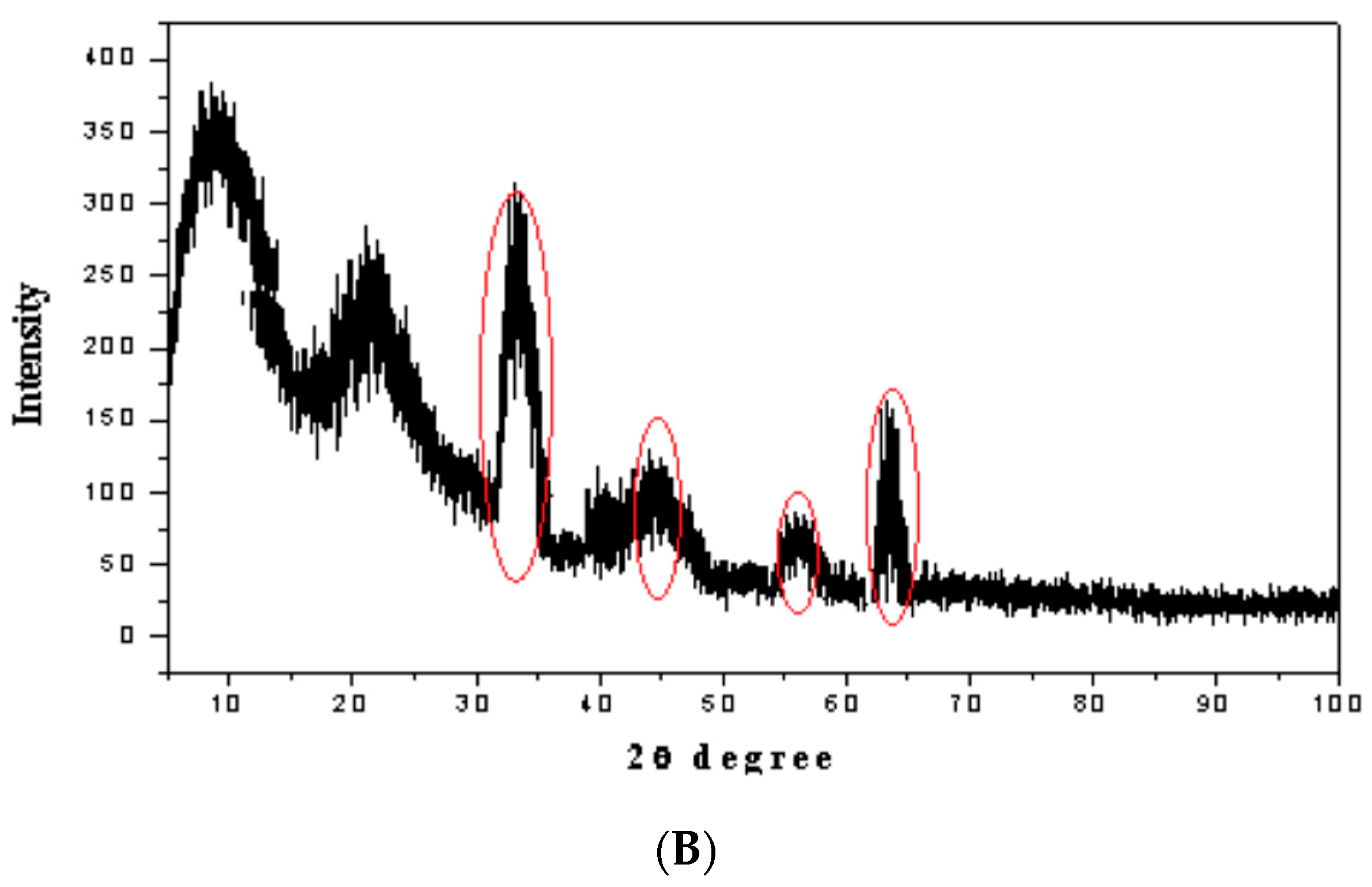
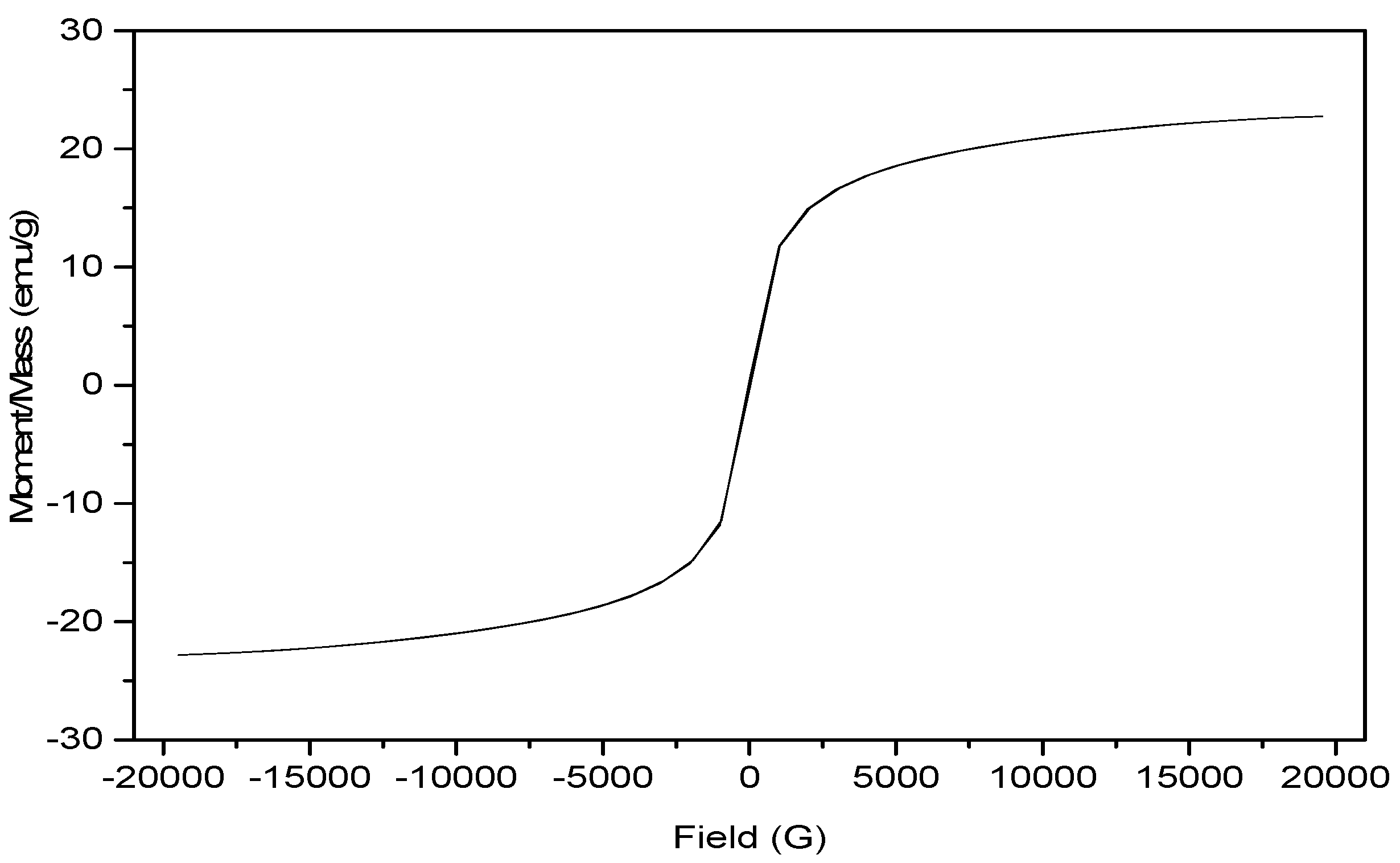
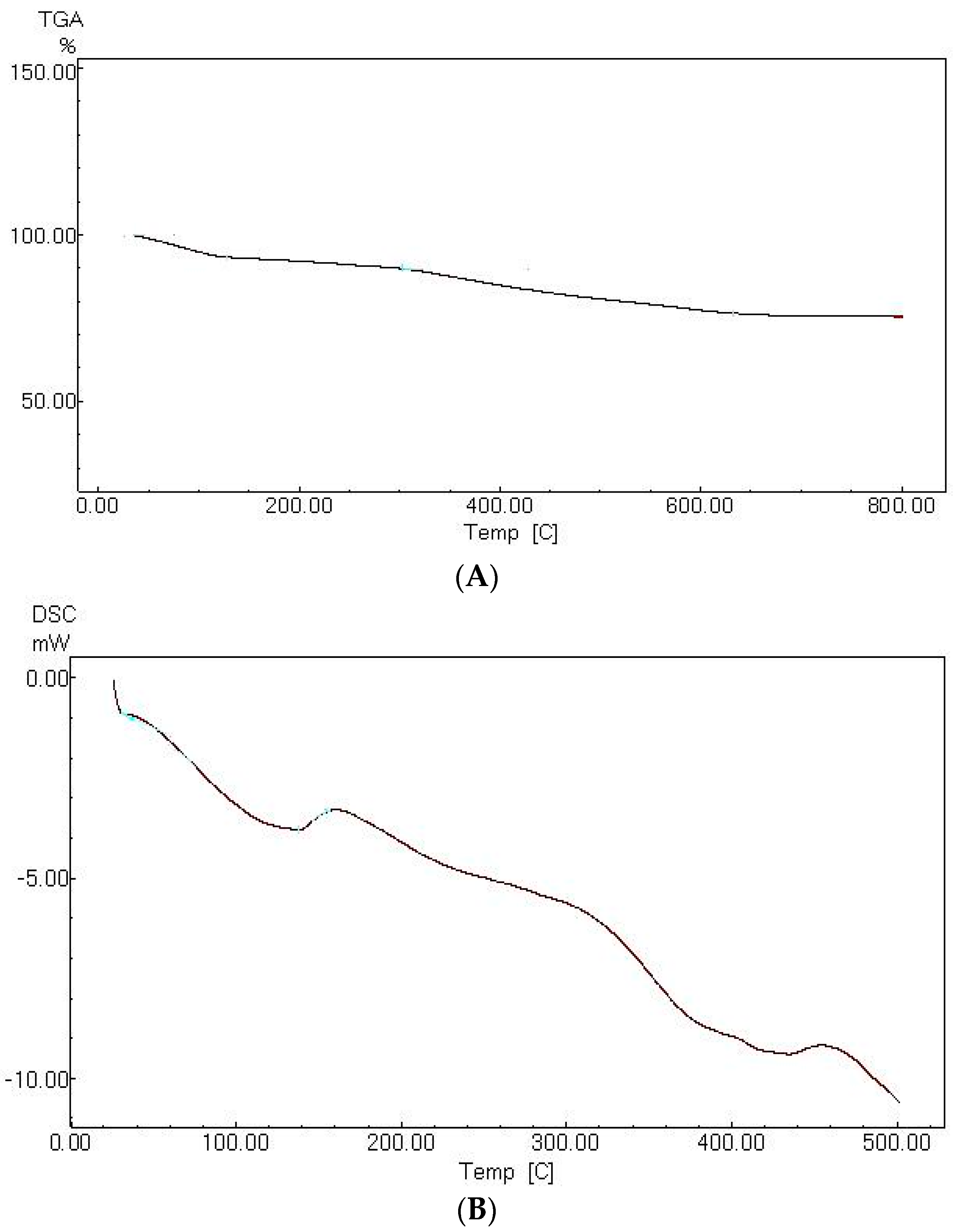
3.3. Characterization of the Magnetic Amine-Functionalized Nano-Silica Gel
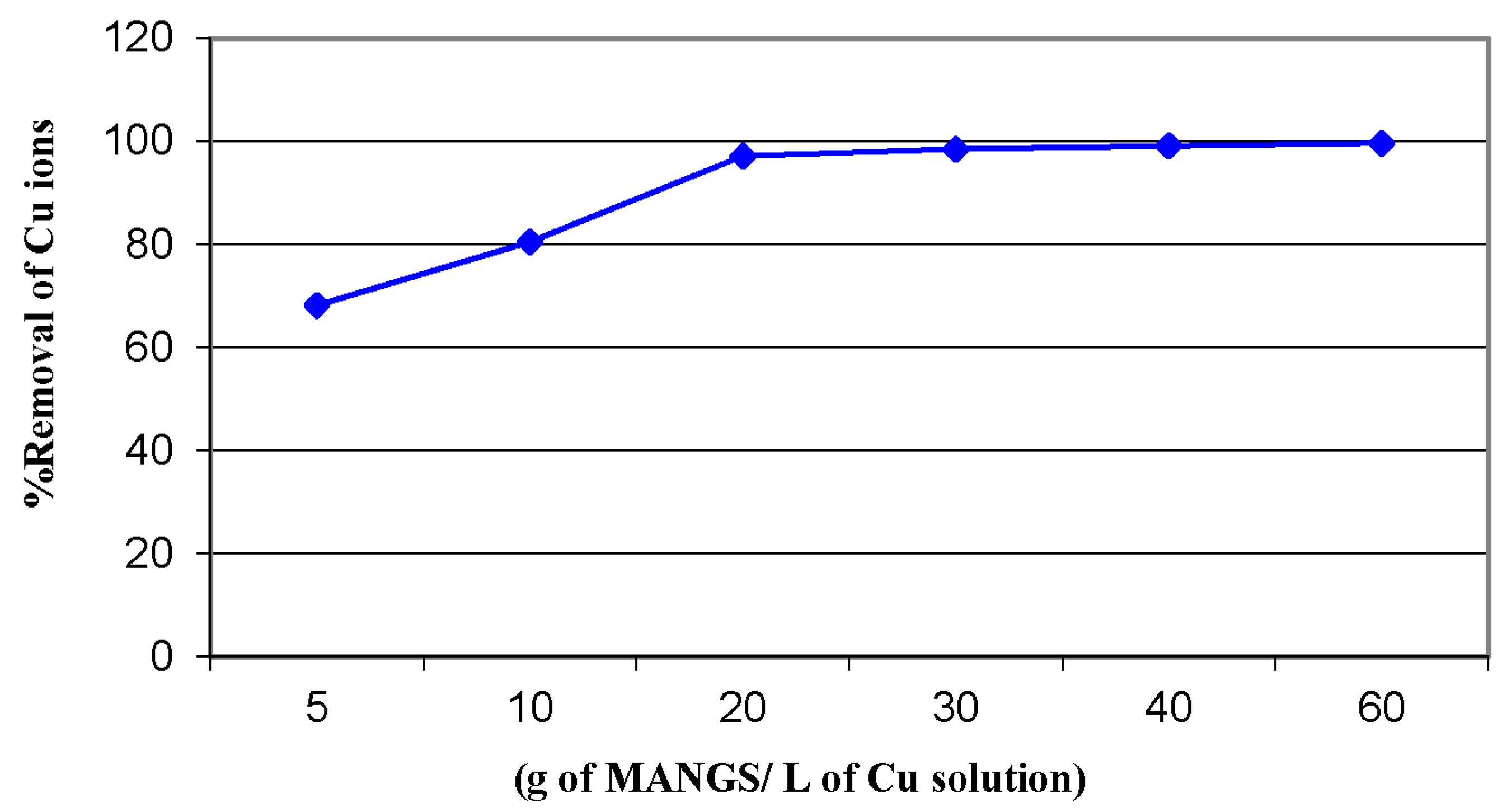
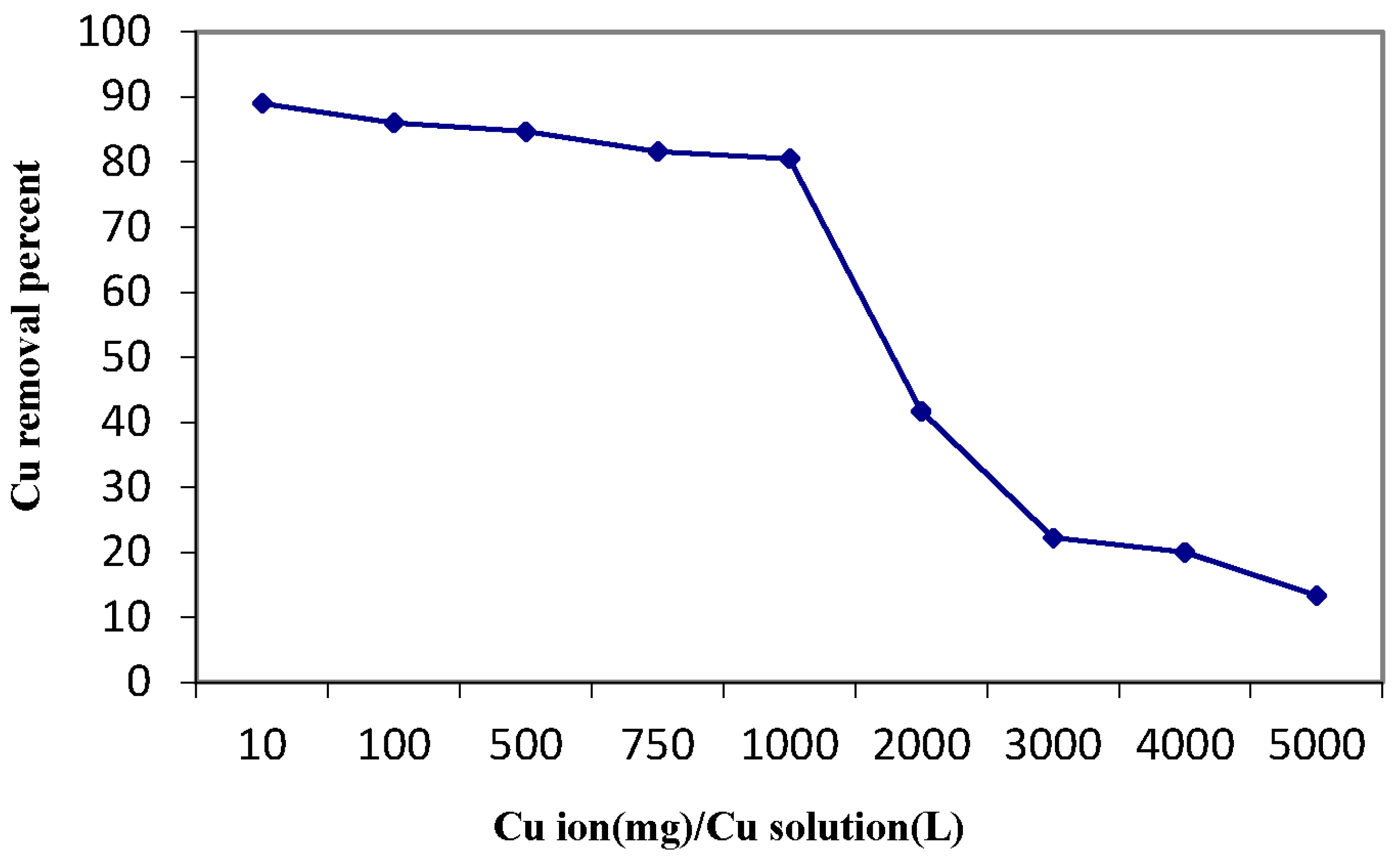
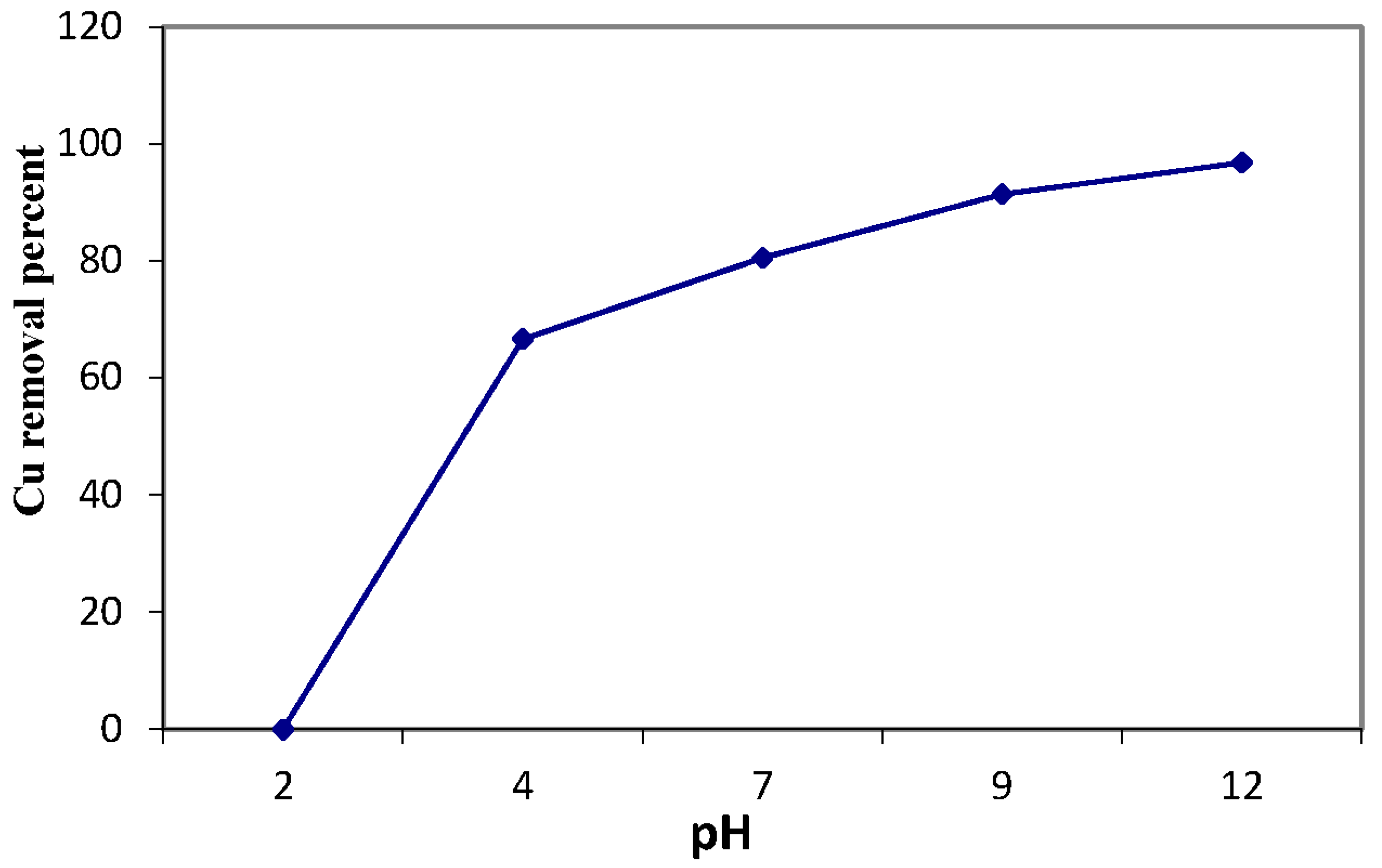
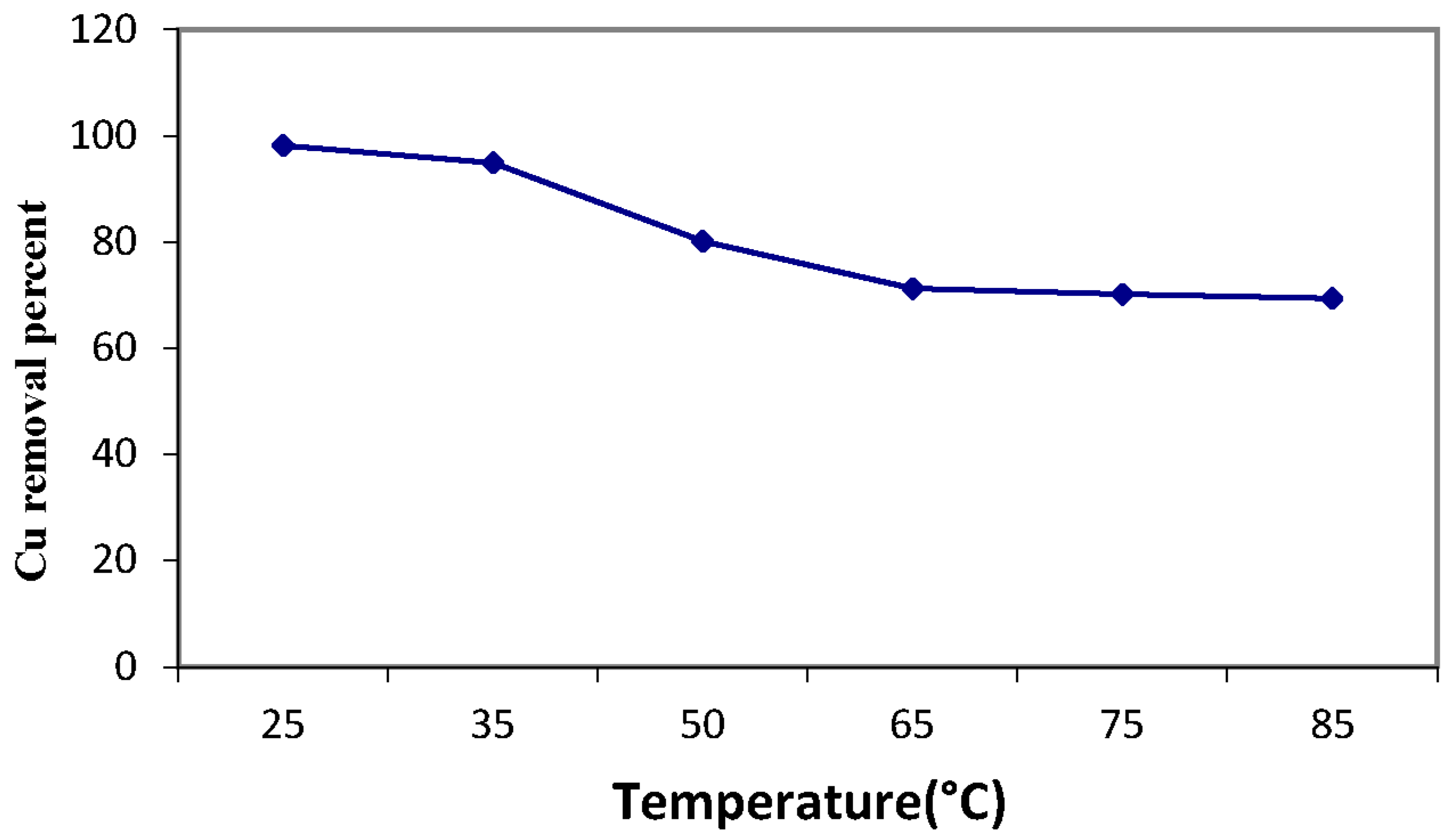
3.4. Copper Sorption Profile of the Magnetic Amine-Functionalized Nano-Silica Gel
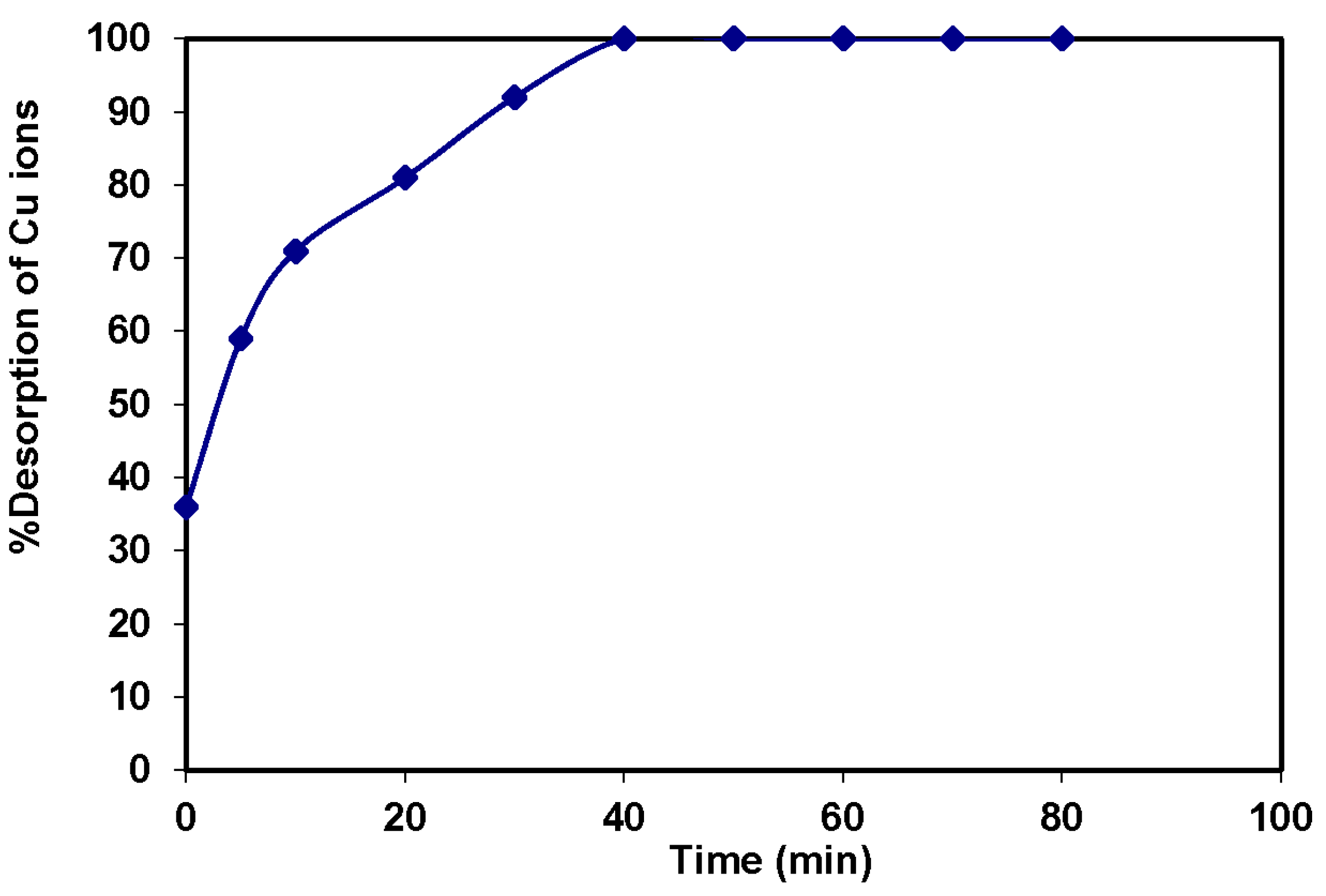
3.5. Regeneration of the Magnetic Amine-Functionalized Nano-Silica Gel
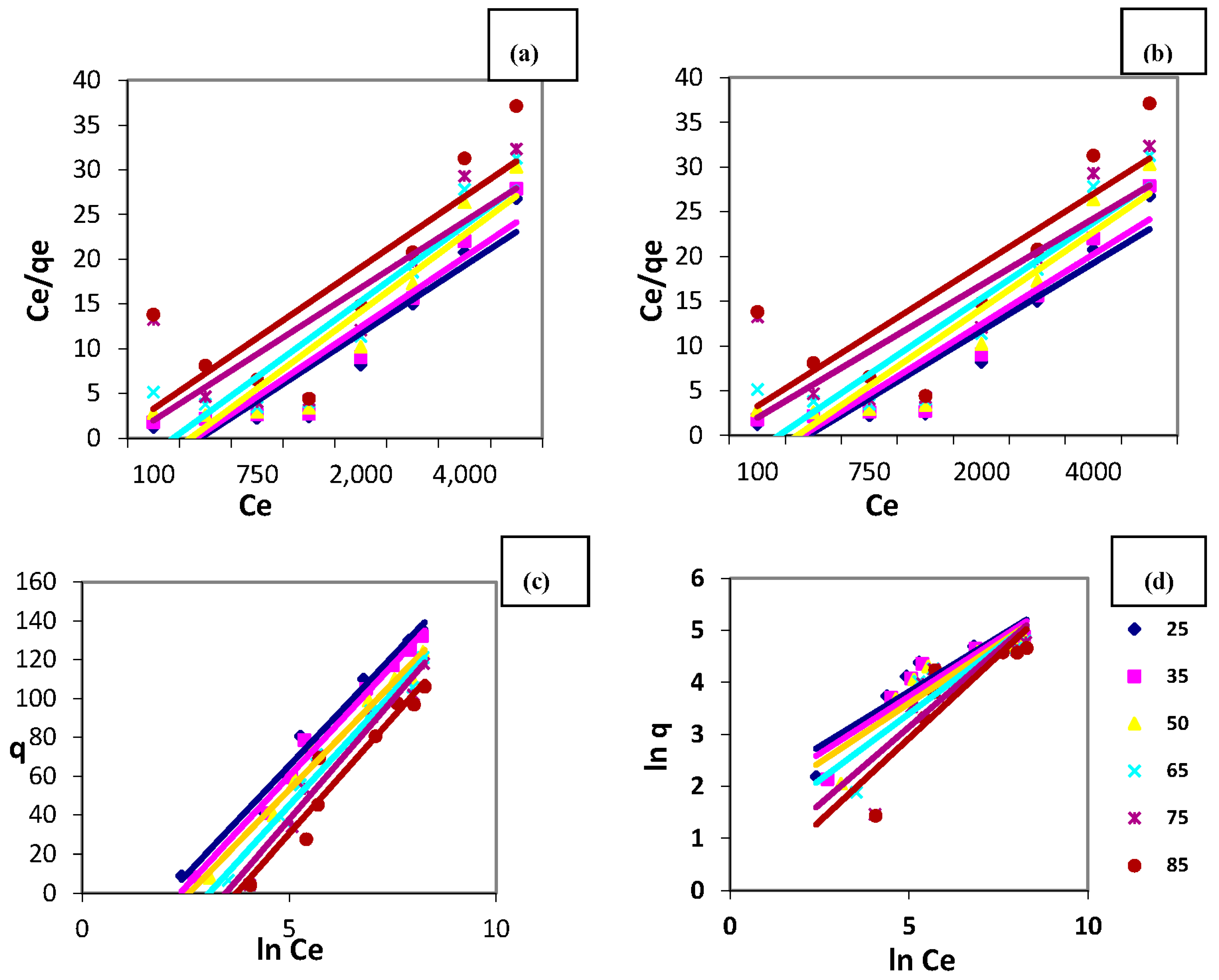
3.6. Equilibrium Isotherm Modelling for Copper Decontamination Using Magnetic Amine-Functionalized Nano-Silica Gel
3.6.1. Langmuir Isotherm
3.6.2. Freundlich Isotherm
3.6.3. Temkin Isotherm
3.6.4. Elovich Isotherm
3.6.5. Dubinin-Radushkevich Isotherm
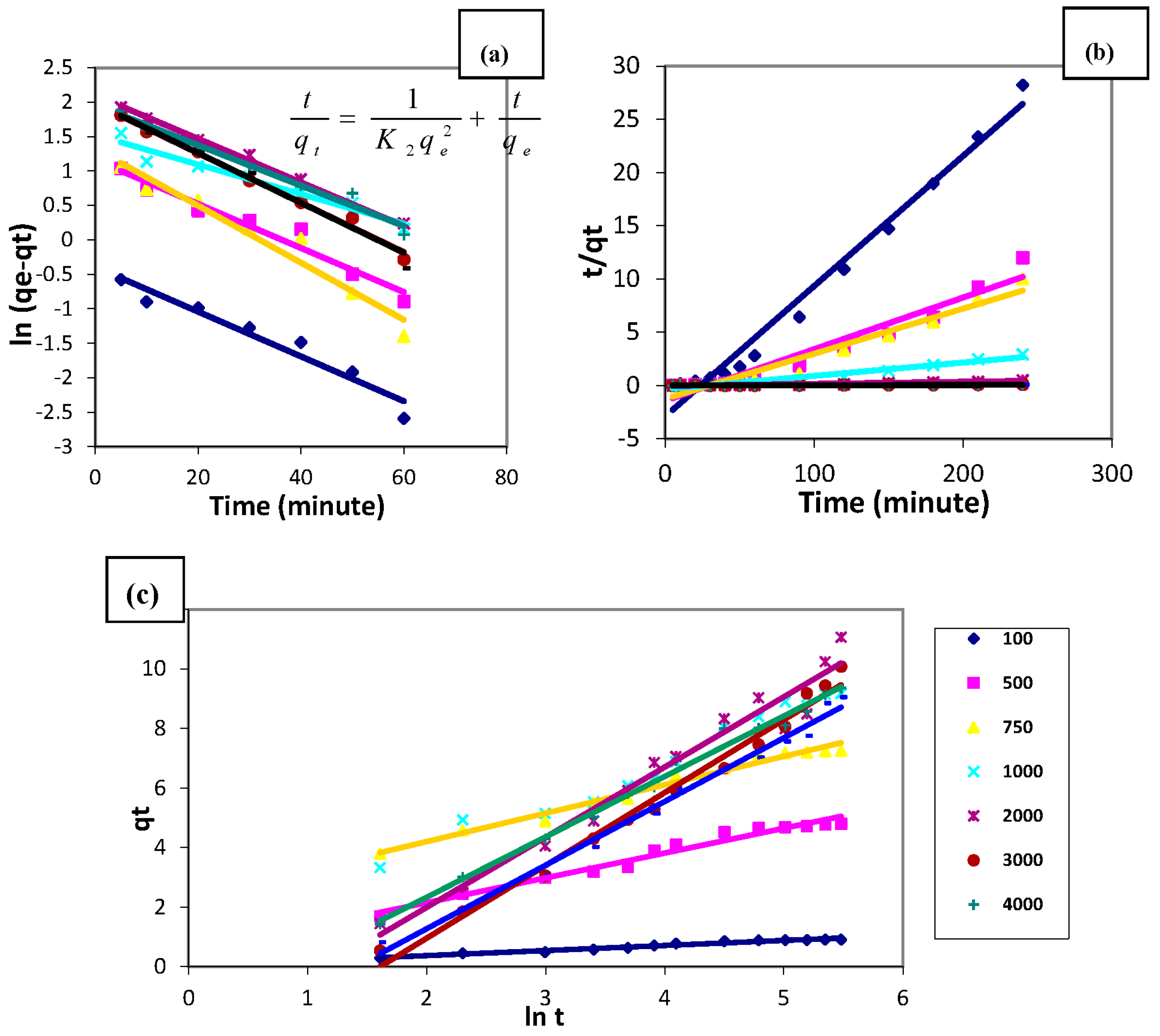
3.7. Copper Sorption Kinetic Modelling
3.7.1. Pseudo-First-Order Rate Model
3.7.2. Pseudo-Second-Order Rate Model
3.7.3. Simple Elovich Model
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zaporozhets, O.; Petruniock, N.; Sukhan, V. Determination of Ag(I), Hg(II) and Pb(II) by using silica gel loaded with dithizone and zinc dithizonate. Talanta 1999, 50, 865–873. [Google Scholar] [CrossRef]
- Caroli, C.; Alimonti, A.; Petrucci, F.; Horvth, Z. Determination of trace elements in analytical-reagent grade sodium salts by atomic absorption spectrometry and inductively coupled plasma atomic emission spectrometry after preconcentration by column solid phase extraction. Anal. Chim. Acta 1991, 360, 241–245. [Google Scholar] [CrossRef]
- Rao, T.P.; Metilda, P.; Gladis, J.M. Preconcentration techniques for uranium(VI) and thorium(IV) prior to analytical determination—An overview. Talanta 2006, 68, 1047–1064. [Google Scholar] [CrossRef] [PubMed]
- Elkady, M.F.; Hassan, H.S. Invention of hollow zirconium tungesto-Vanadate at Nanotube Morphological Structure for Radionuclides and Heavy Metal Pollutants Decontamination from Aqueous Solutions. Nanoscale Res. Lett. 2015, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Luan, J.; Li, Y. Photocatalytic water splitting for hydrogen production with Gd2MSbO7 (M = Fe, In, Y) photocatalysts under visible light irradiation. Materials 2015, 8, 16–30. [Google Scholar] [CrossRef]
- Sharma, R.K.; Mittal, S.; Koel, M. Analysis of trace amounts of metal ions using silica-based chelating resins: A green analytical method. Crit. Rev. Anal. Chem. 2003, 33, 183–197. [Google Scholar] [CrossRef]
- Sales, J.A.A.; Faria, F.P.; Prado, A.G.S.; Airoldi, C. Attachment of 2-aminomethylpyridine molecule onto grafted silica gel surface and its ability in chelating cations. Polyhedron 2004, 23, 719–725. [Google Scholar] [CrossRef]
- Harry, B.; Fransiska, S.H.K.; Achmad, H.S. Preparation of silica modified with 2-mercaptoimidazole and its SorptionProperties of Chromium(III). J. Chem. 2009, 6, 141–150. [Google Scholar]
- Luechinger, M.; Prins, R.; Pirngruber, G.D. Functionalization of silica surfaces with mixtures of 3-aminopropyl and methyl groups. Microporous Mesoporous Mater. 2005, 85, 111–118. [Google Scholar] [CrossRef]
- Hassan, H.S.; Kashyout, A.B.; Morsi, I.; Nasser, A.A.A.; Raafat, A. Fabrication and characterization of gas sensor micro-arrays. Sens. Bio-Sens. Res. 2014, 1, 34–40. [Google Scholar] [CrossRef]
- Cui, Y.; Chang, X.; Zhu, X.; Luo, H.; Hu, Z.; Zou, X.; He, Q. Chemically modified silica gel with p-dimethylaminobenzaldehyde for selective solid-phase extraction and preconcentration of Cr(III), Cu(II), Ni(II), Pb(II) and Zn(II) by ICP-OES. Microchem. J. 2007, 87, 20–26. [Google Scholar] [CrossRef]
- Scott, G. Gas Phase studies of the competition between substitution and elimination reactions. Acc. Chem. Res. 2003, 36, 848–857. [Google Scholar]
- Andrade, G.F.; Soares, D.C.F.; de Sousa Almeida, R.K.; Sousa, E.M.B. Mesoporous silica SBA-16 functionalized with alkoxysilane groups: Preparation, characterization, and release profile study. J. Nanomater. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Elkady, M.F.; Hassan, H.S.; Hafez, E.E.; Fouad, A. Construction of zinc oxide into different morphological structures to be utilized as antimicrobial agent against multidrug resistant bacteria. Bioinorg. Chem. Appl. 2015, 2015, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.; Souza, D.; Pereira, M.; Fabris, J.; Domingues, R. Synthesis and characterization of magnetic nanoparticles coated with silica through a sol-gel approach. Cerâmica 2009, 55, 420–424. [Google Scholar] [CrossRef]
- Elkady, M.F.; El-Aassar, R.; Shokry Hassan, H. Adsorption Profile of Basic Dye onto Novel Fabricated Carboxylated Functionalized Co-Polymer Nanofibers. Polymers 2016, 177, 1–13. [Google Scholar] [CrossRef]
- Camila, G.P.; Fernanda, S.R.; Nathália, M.S.; Araci, A.S.; Vaghetti, C.P.J.; Edilson, V.B.; Éder, C.L. Use of statistical design of experiments to evaluate the sorption capacity of 7-amine-4-azaheptylsilica and 10-amine-4-azadecylsilica for Cu(II), Pb(II), and Fe(III) adsorption. J. Colloid Interface Sci. 2006, 302, 396. [Google Scholar]
- Salah El-Din, T.A.; Elzatahry, A.; Aldhayan, D.; Al-Enizi, A.; Al-Deyab, S. Synthesis and characterization of magnetite zeolite nano composite. Int. J. Electrochem. Sci. 2011, 6, 6177–6183. [Google Scholar]
- El Ghandoor, H.; Zidan, H.; Mostafa, M.; Ismail, M. Synthesis and some physical properties of magnetite (Fe3O4) nanoparticles. Int. J. Electrochem. Sci. 2012, 7, 5734–5757. [Google Scholar]
- Kharroubia, M.; Balme, S.; Henn, F.; Giuntini, J.C.; Belarbi, H.; Haouzi, A. Dehydration enthalpy of alkali-cations-exchanged montmorillonite from thermogravimetric analysis. J. Colloid Interface Sci. 2009, 329, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Mahitti, P.; Fuangfa, U. Preparation and use of chemically modified MCM-41 and silica gel as selective adsorbents for Hg(II) ions. J. Hazard. Mater. 2008, 154, 578–587. [Google Scholar]
- Salleh, M.A.M.; Mahmoud, D.K.; Abdul Karim, W.A.W.; Idris, A. Cationic and anionic dye adsorption by agricultural solid wastes: A comprehensive review. Desalination 2011, 280, 1–13. [Google Scholar] [CrossRef]
- Wahi, R.; Ngaini, Z.; Jok, U. Removal of mercury, lead and copper from aqueous solution by activated carbon of palm oil empty fruit bunch. World Appl. Sci. J. 2009, 5, 84–91. [Google Scholar]
- Elkady, M.F.; EL-Sayed, E.; Farag, H.; Zaatout, A. Assessment of novel synthetized nanozirconium tungstovanadate as cation exchanger for lead ion decontamination. J. Nanomater. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Xue, X.; Li, F. Removal of Cu(II) from aqueous solution by adsorption onto functionalized SBA-16 mesoporous silica. Microporous Mesoporous Mater. 2008, 116, 116–122. [Google Scholar] [CrossRef]
- Hassan, H.S.; Elkady, M.F.; El-Shazly, A.; Hesham, B. Formulation of synthesized zinc oxide nanopowder into hybrid beads for dye separation. J. Nanomater. 2014, 2014, 1–14. [Google Scholar]
- Mohammad, A.; Rifaqat, A.K.R.; Rais, A.; Jameel, A. Adsorption studies on Citrus reticulata (fruit peel of orange): Removal and recovery of Ni(II) from electroplating wastewater. J. Hazard. Mater. 2000, 79, 117–131. [Google Scholar]
- Elkady, M.F.; Shokry Hassan, H. Equilibrium and dynamic profiles of azo dye sorption onto innovative nano-zinc oxide biocomposite. Curr. Nanosci. 2015, 11, 805–814. [Google Scholar] [CrossRef]
- Ho, Y.S.; Porter, J.F.; Mckay, G. Equilibrium isotherm studies for the sorption of divalent metal ions onto peat: Copper, nickel and lead single component systemsg. Water Air Soil Pollut. 2002, 141, 1–33. [Google Scholar] [CrossRef]
- Mohy-Eldin, M.S.; Elkady, M.F.; Abu-Saied, M.A.; Abdel Rahman, A.M.; Soliman, E.A.; Elzatahry, A.A.; Youssef, M.E. Removal of cadmium ions from synthetic aqueous solutions using novel nano-sulphonated poly glycidylmethacrylate cation exchanger: Kinetic and equilibrium studies. J. Appl. Polym. Sci. 2010, 6, 280–292. [Google Scholar]
- Jeon, C.; Cha, J.-H. Removal of cesium ions from waste solution using sericite incorporated into nickel hexacyanoferrate. Korean J. Chem. Eng. 2015, 32, 2273–2279. [Google Scholar] [CrossRef]
- Subbaiah, M.V.; Vijaya, Y.; Subba, R.A.; Yuvaraja, G.; Krishnaiah, A. Equilibrium, kinetic and thermodynamic studies on the biosorption of Cu(II) onto Trametes versicolor biomass. Desalination 2011, 276, 310–316. [Google Scholar] [CrossRef]
- Abd El-Latif, M.F.; Elkady, M.F. Synthesis, characterization and evaluation of nano-zirconium vanadate ion exchanger by using three different preparation techniques. Mater. Res. Bull. 2011, 46, 105–118. [Google Scholar] [CrossRef]
- Elkady, M.F.; Hassan, H.S.; El-Sayed, E. Basic violet decolourization using alginate immobilized nanozirconium tungestovanadate matrix as cation exchanger. J. Chem. 2015, 2015, 1–10. [Google Scholar] [CrossRef]



| Solution Temperature (°C) | qm (mg/g) | b (L/mg) | R2 | RL |
|---|---|---|---|---|
| 25 | 140.85 | 0.0052 | 0.9981 | 0.951–0.037 |
| 35 | 136.99 | 0.0042 | 0.9981 | 0.957–0.043 |
| 50 | 126.58 | 0.0039 | 0.9943 | 0.962–0.049 |
| 65 | 122.21 | 0.0026 | 0.9875 | 0.975–0.072 |
| 75 | 115.135 | 0.0014 | 0.9028 | 0.986–0.123 |
| 85 | 109.87 | 0.001 | 0.9223 | 0.988–0.142 |
| Solution Temperature (°C) | Kf | n | R2 |
|---|---|---|---|
| 25 | 5.55 | 2.37 | 0.88 |
| 35 | 4.6 | 2.27 | 0.86 |
| 50 | 3.74 | 2.2 | 0.82 |
| 65 | 2.31 | 1.99 | 0.8 |
| 75 | 1.18 | 1.68 | 0.71 |
| 85 | 0.76 | 1.57 | 0.76 |
| Solution Temperature (°C) | KT (L/mg) | B | b (J/mol) | R2 |
|---|---|---|---|---|
| 25 | 0.12 | 22.497 | 0.11 | 0.99 |
| 35 | 0.09 | 22.794 | 0.11 | 0.99 |
| 50 | 0.08 | 21.91 | 0.12 | 0.98 |
| 65 | 0.05 | 23.192 | 0.12 | 0.97 |
| 75 | 0.03 | 24.601 | 0.12 | 0.95 |
| 85 | 0.02 | 23.875 | 0.12 | 0.93 |
| Solution Temperature (°C) | Ke (L/mg) | qm (mg/g) | R2 |
|---|---|---|---|
| 25 | 0.04 | 39.68 | 0.89 |
| 35 | 0.023 | 41.67 | 0.88 |
| 50 | 0.02 | 42.37 | 0.8 |
| 65 | 0.01 | 51.55 | 0.69 |
| 75 | 0.003 | 76.92 | 0.35 |
| 85 | 0.002 | 85.47 | 0.33 |
| Initial Concentration of Cu Ion (ppm) | R2 | K1 (min−1) | qe (cal.) (mg/g) | qe (exp.) (mg/g) |
|---|---|---|---|---|
| 100 | 0.93 | 0.025 | 0.616 | 0.86 |
| 500 | 0.95 | 0.032 | 3.194 | 4.52 |
| 750 | 0.95 | 0.041 | 3.758 | 6.667 |
| 1000 | 0.94 | 0.021 | 4.559 | 8.05 |
| 2000 | 0.99 | 0.03 | 7.798 | 8.333 |
| 3000 | 0.98 | 0.037 | 7.53 | 6.667 |
| 4000 | 0.97 | 0.03 | 7.464 | 8 |
| 5000 | 0.95 | 0.037 | 7.716 | 6.663 |
| Initial Concentration of Cu Ion (ppm) | R2 | K2 (g/mg·min) | qe (cal.) (mg/g) | qe (exp.) (mg/g) |
|---|---|---|---|---|
| 100 | 0.98 | 0.0051 | 0.82 | 0.86 |
| 500 | 0.98 | 0.0016 | 2.06 | 4.52 |
| 750 | 0.98 | 0.0014 | 2.36 | 6.67 |
| 1000 | 0.98 | 0.0092 | 8.00 | 8.05 |
| 2000 | 0.97 | 0.0005 | 8.85 | 8.33 |
| 3000 | 0.99 | 0.0062 | 6.67 | 6.67 |
| 4000 | 0.99 | 0.0575 | 7.4 | 8 |
| 5000 | 0.99 | 0.0001 | 6.33 | 6.66 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elkady, M.; Hassan, H.S.; Hashim, A. Immobilization of Magnetic Nanoparticles onto Amine-Modified Nano-Silica Gel for Copper Ions Remediation. Materials 2016, 9, 460. https://doi.org/10.3390/ma9060460
Elkady M, Hassan HS, Hashim A. Immobilization of Magnetic Nanoparticles onto Amine-Modified Nano-Silica Gel for Copper Ions Remediation. Materials. 2016; 9(6):460. https://doi.org/10.3390/ma9060460
Chicago/Turabian StyleElkady, Marwa, Hassan Shokry Hassan, and Aly Hashim. 2016. "Immobilization of Magnetic Nanoparticles onto Amine-Modified Nano-Silica Gel for Copper Ions Remediation" Materials 9, no. 6: 460. https://doi.org/10.3390/ma9060460
APA StyleElkady, M., Hassan, H. S., & Hashim, A. (2016). Immobilization of Magnetic Nanoparticles onto Amine-Modified Nano-Silica Gel for Copper Ions Remediation. Materials, 9(6), 460. https://doi.org/10.3390/ma9060460







