Optimizing the Pore Structure of Bio-Based ACFs through a Simple KOH–Steam Reactivation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Subsection Surface Morphology
2.2. Pore Structure
2.3. Chemical and Crystal Structure
2.4. Iodine Adsorption
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Activated Carbon Fibers
3.3. Characterization of ACFs
3.4. Liquid-Phase Adsorption of Iodine
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Feng, B.; Xu, X.; Xu, W.; Zhou, G.; Hu, J.; Wang, Y.; Bao, Z. Self-assembled 3D ACF-rGO-TiO2 composite as efficient and recyclable spongy adsorbent for organic dye removal. Mater. Des. 2015, 83, 522–527. [Google Scholar] [CrossRef]
- Feng, H.M.; Zhang, S.J.; Chen, Y.Z.; Ding, Y.W.; Yu, H.Q.; Lam, M.H.W. Fabrication and Evaluation of Mesoporous Poly(vinyl alcohol)-Based Activated Carbon Fibers. Ind. Eng. Chem. Res. 2009, 48, 3398–3402. [Google Scholar] [CrossRef]
- Zhang, S.J.; Yu, H.Q.; Feng, H.M. PVA-based activated carbon fibers with lotus root-like axially porous structure. Carbon 2006, 44, 2059–2068. [Google Scholar] [CrossRef]
- Lopez, M.C.B.; Villar-Rodil, S.M.; Martinez-Alonso, A.; Tascon, J.M.D. Effect of some precursor characteristics on the porous texture of activated carbon fibres prepared from Nomex aramid fibres. Microporous Mesoporous Mater. 2000, 41, 319–321. [Google Scholar] [CrossRef]
- Basova, Y.V.; Edie, D.D.; Badheka, P.Y.; Bellam, H.C. The effect of precursor chemistry and preparation conditions on the formation of pore structure in metal-containing carbon fibers. Carbon 2005, 43, 1533–1545. [Google Scholar] [CrossRef]
- Dai, D.; Fan, M. Preparation of bio-composite from wood sawdust and gypsum. Ind. Crop. Prod. 2015, 74, 417–424. [Google Scholar] [CrossRef]
- Ma, X.J.; Zhao, G.J. Preparation of carbon fibers from liquefied wood. Wood Sci. Technol. 2010, 44, 3–11. [Google Scholar]
- Liu, W.J.; Zhao, G.J. Effect of temperature and time on microstructure and surface functional groups of activated carbon fibers prepared from liquefied wood. Bioresources 2012, 7, 5552–5567. [Google Scholar] [CrossRef]
- Huang, Y.X.; Ma, E.N.; Zhao, G.J. Thermal and structure analysis on reaction mechanisms during the preparation of activated carbon fibers by KOH activation from liquefied wood-based fibers. Ind. Crop. Prod. 2015, 69, 447–455. [Google Scholar] [CrossRef]
- Yuan, R.S.; Guan, R.B.; Zheng, J.T. Effect of the pore size of TiO2-loaded activated carbon fiber on its photocatalytic activity. Scr. Mater. 2005, 52, 1329–1334. [Google Scholar] [CrossRef]
- Pandolfo, A.G.; Hollenkamp, A.F. Carbon properties and their role in supercapacitors. J. Power Sources 2006, 157, 11–27. [Google Scholar] [CrossRef]
- Huang, Y.X.; Zhao, G.J. Preparation and characterization of activated carbon fibers from liquefied wood by KOH activation. Holzforschung 2016, 70, 195–202. [Google Scholar] [CrossRef]
- Miyamoto, J.; Kanoh, K.; Kaneko, K. The addition of mesoporosity to activated carbon fibers by a simple reactivation process. Carbon 2005, 43, 855–857. [Google Scholar] [CrossRef]
- Lei, S.; Miyamoto, J.; Kanoh, H.; Nakahigashi, Y.; Kaneko, K. Enhancement of the methylene blue adsorption rate for ultramicroporous carbon fibers by addition of mesopores. Carbon 2006, 44, 1884–1890. [Google Scholar] [CrossRef]
- Wang, P.H.; Hong, K.L.; Zhu, Q.R. Surface analyses of polyacrylonitrile-based activated carbon fibers by X-ray photoelectron spectroscopy. J. Appl. Polym. Sci. 1996, 62, 1987–1991. [Google Scholar] [CrossRef]
- Huang, Y.X.; Ma, E.N.; Zhao, G.J. Preparation of liquefied wood-based activated carbon fibers by different activation methods for methylene blue adsorption. RSC Adv. 2015, 5, 70287–70296. [Google Scholar] [CrossRef]
- Huang, Y.X.; Zhao, G.J. A novel method for the production of mesoporous activated carbon fibers from liquefied wood. Mater. Lett. 2016, 178, 190–192. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas solid systems with special reference to the determination of surface-area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Zhu, L.; Qi, H.-Y.; Lv, M.-L.; Kong, Y.; Yu, Y.-W.; Xu, X.-Y. Component analysis of extracellular polymeric substances (EPS) during aerobic sludge granulation using FTIR and 3D-EEM technologies. Bioresour. Technol. 2012, 124, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Lei, H.; Chen, D.; Huo, J. Blowing and in-situ activation of carbonaceous “lather” from starch: Preparation and potential application. Mater. Des. 2016, 92, 362–370. [Google Scholar] [CrossRef]
- Deng, L.Y.; Xu, G.R.; Li, G.B. Surface properties and adsorption characteristics to methylene blue and iodine of adsorbents from sludge. Water Sci. Technol. 2010, 62, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Fang, F.; Xiao, H.-M.; Feng, Q.-P.; Xiong, L.-Y.; Fu, S.-Y. Preparation of pore-size controllable activated carbon fibers from bamboo fibers with superior performance for xenon storage. Chem. Eng. J. 2015, 270, 528–534. [Google Scholar] [CrossRef]
- Xu, J.; Chen, L.; Qu, H.; Jiao, Y.; Xie, J.; Xing, G. Preparation and characterization of activated carbon from reedy grass leaves by chemical activation with H3PO4. Appl. Surf. Sci. 2014, 320, 674–680. [Google Scholar] [CrossRef]
- Saka, C. BET, TG-DTG, FT-IR, SEM, iodine number analysis and preparation of activated carbon from acorn shell by chemical activation with ZnCl2. J. Anal. Appl. Pyrolysis 2012, 95, 21–24. [Google Scholar] [CrossRef]
- Maddah, B.; Nasouri, K. Fabrication of High Surface Area PAN-based Activated Carbon Fibers Using Response Surface Methodology. Fibers Polym. 2015, 16, 2141–2147. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- De Boer, J.H.; Lippens, B.C.; Linsen, B.G.; Broekhoff, J.C.P.; van den Heuvel, A.; Osinga, T.J. T-curve of multimolecular N2-adsorption. J. Colloid Interface Sci. 1966, 21, 405–414. [Google Scholar] [CrossRef]
- American Society for Testing Materials. Standard Test Method for Determination of Iodine Number of Activated Carbon; ASTM D4607-94; ASTM International: West Conshohocken, PA, USA, 1999. [Google Scholar]

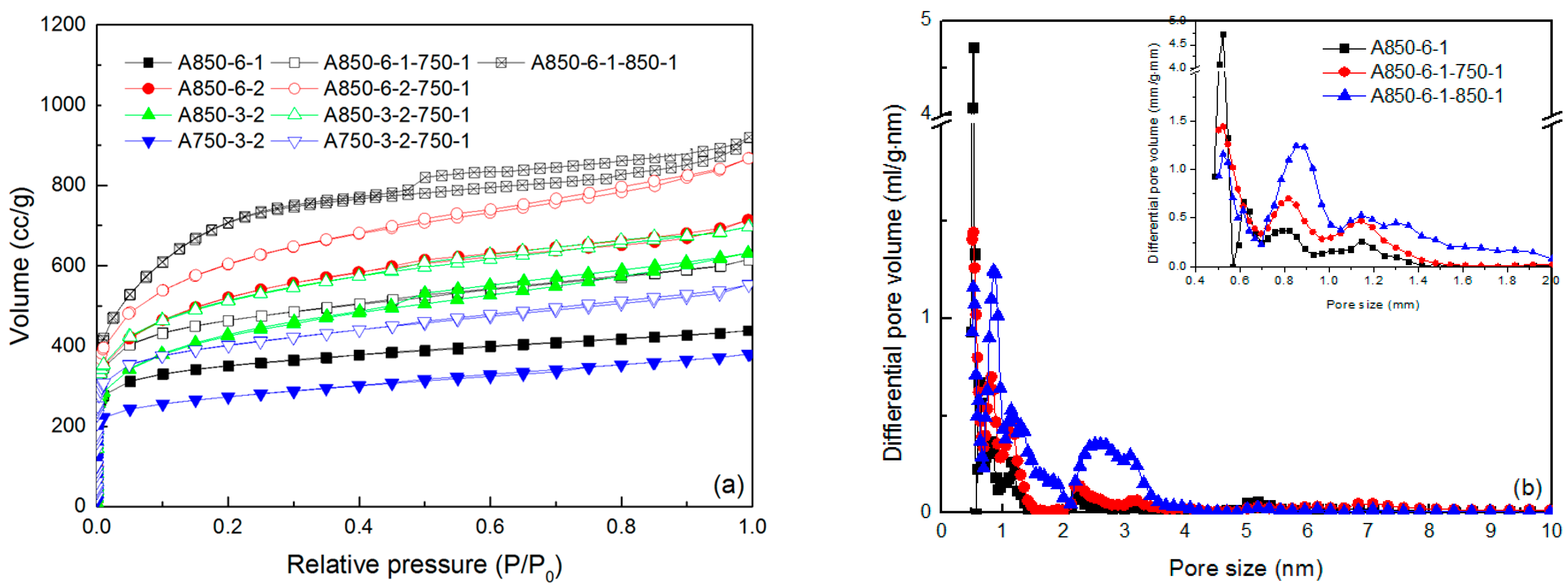
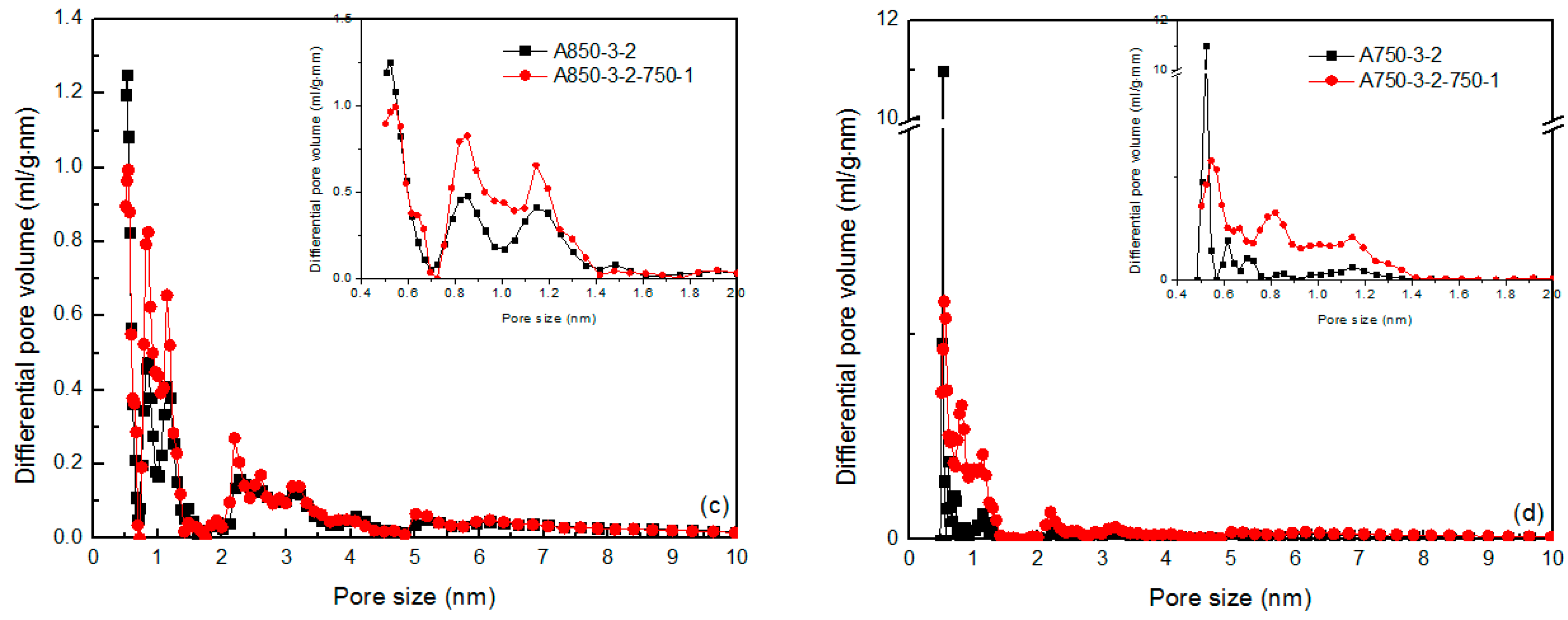
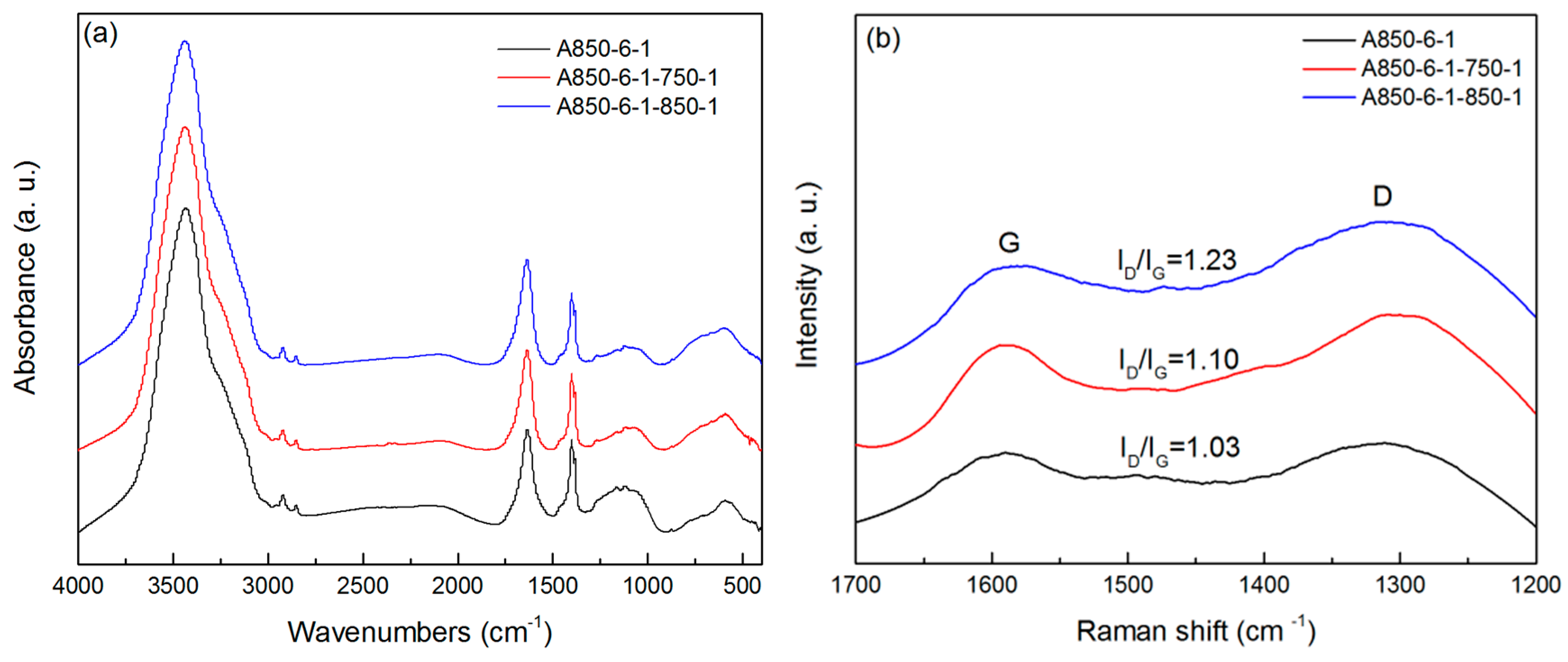
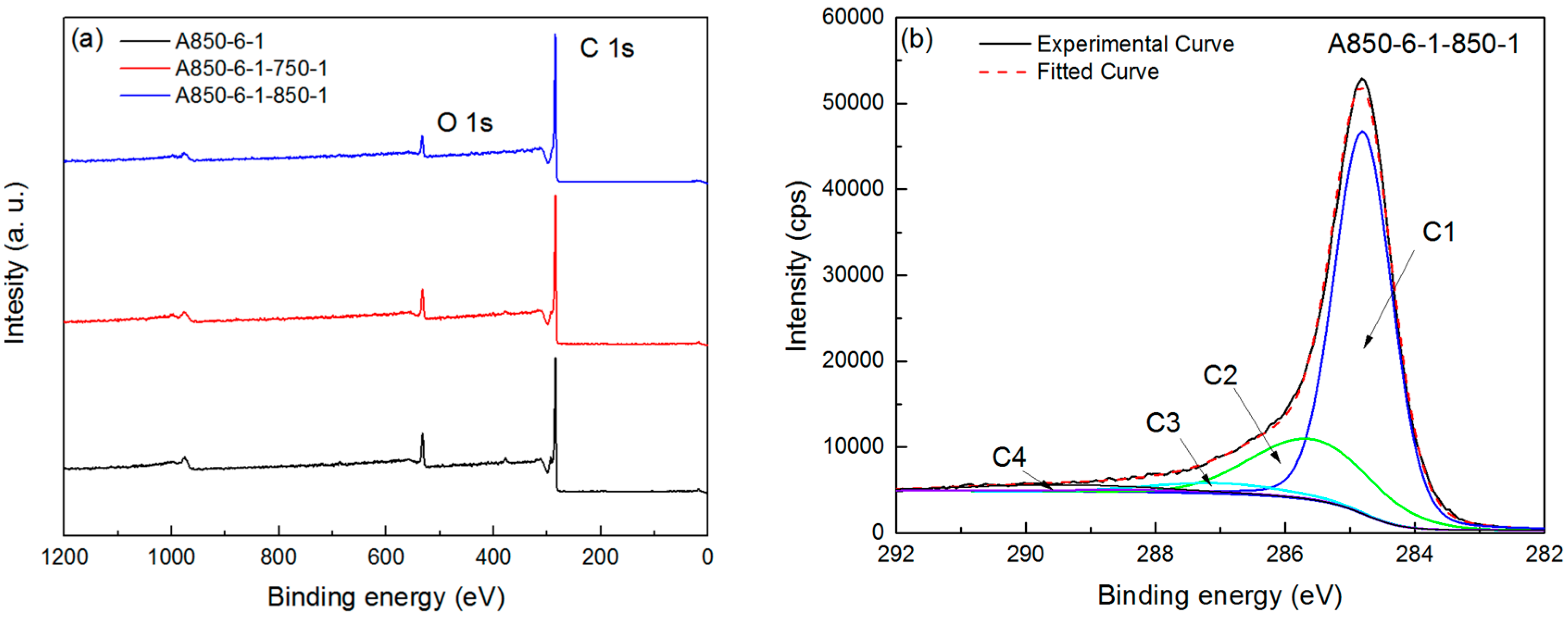
| Sample | SBET | Smicro | Smeso | Vtot | Vmicro | Vmeso | Vmeso/Vtot | Yield | Iodine Adsorption |
|---|---|---|---|---|---|---|---|---|---|
| (m2·g−1) | (m2·g−1) | (m2·g−1) | (cm3·g−1) | (cm3·g−1) | (cm3·g−1) | (%) | (%) | (mg·g−1) | |
| A850-6-1 | 1301 | 976 | 209 | 0.679 | 0.4 | 0.216 | 31.8 | 43.2 | 1237 |
| A850-6-1-750-1 | 1715 | 1295 | 316 | 0.952 | 0.538 | 0.354 | 37.2 | 17.5 | 1628 |
| A850-6-1-850-1 | 2578 | 2196 | 452 | 1.425 | 0.966 | 0.424 | 29.7 | 9.6 | 1957 |
| A850-6-2 | 1861 | 1115 | 466 | 1.105 | 0.484 | 0.473 | 42.8 | 29.6 | 1713 |
| A850-6-2-750-1 | 2153 | 1463 | 581 | 1.342 | 0.656 | 0.627 | 46.7 | 10.3 | 1895 |
| A850-3-2 | 1512 | 909 | 572 | 0.977 | 0.402 | 0.549 | 56.2 | 32.4 | 1597 |
| A850-3-2-750-1 | 1845 | 1241 | 586 | 1.08 | 0.542 | 0.519 | 48 | 16.7 | 1764 |
| A750-3-2 | 1013 | 694 | 261 | 0.589 | 0.285 | 0.27 | 45.8 | 45.2 | 1044 |
| A750-3-2-750-1 | 1493 | 1083 | 300 | 0.857 | 0.447 | 0.348 | 40.6 | 21.6 | 1566 |
| Sample | Atomic Concentration (%) | Data Derived of C 1s Peaks (%) | |||||
|---|---|---|---|---|---|---|---|
| C | O | C1 | C2 | C3 | C4 | C5 | |
| A850-6-1 | 85.0 | 15.0 | 67.1 | 16.6 | 6.1 | 4.3 | 5.9 |
| A850-6-1-750-1 | 87.3 | 12.7 | 62.5 | 18.7 | 7.8 | 5.6 | 5.4 |
| A850-6-1-850-1 | 91.9 | 8.1 | 59.6 | 20.1 | 10.8 | 9.5 | - |
| A850-6-2 | 92.8 | 7.2 | 65.0 | 13.3 | 8.3 | 7.7 | 5.7 |
| A850-6-2-750-1 | 93.5 | 6.5 | 61.4 | 17.6 | 11.8 | - | 9.2 |
| A850-3-2 | 92.2 | 7.8 | 67.1 | 10.5 | 9.0 | 8.6 | 4.9 |
| A850-3-2-750-1 | 93.1 | 6.9 | 63.2 | 15.6 | 9.2 | 7.5 | 4.5 |
| A750-3-2 | 86.5 | 13.5 | 67.9 | 15.8 | 7.7 | 4.7 | 3.9 |
| A750-3-2-750-1 | 88.4 | 11.6 | 65.3 | 14.7 | 8.5 | 5.6 | 5.9 |
| Sample | KOH Activation | Steam Activation | |||
|---|---|---|---|---|---|
| KOH/Fiber Ratio | Temperature (°C) | Time (h) | Temperature (°C) | Time (h) | |
| A850-6-1 | 6 | 850 | 1 | - | - |
| A850-6-1-750-1 | 6 | 850 | 1 | 750 | 1 |
| A850-6-1-850-1 | 6 | 850 | 1 | 850 | 1 |
| A850-6-2 | 6 | 850 | 2 | - | - |
| A850-6-2-750-1 | 6 | 850 | 2 | 750 | 1 |
| A850-3-2 | 3 | 850 | 2 | - | - |
| A850-3-2-750-1 | 3 | 850 | 2 | 750 | 1 |
| A750-3-2 | 3 | 750 | 2 | - | - |
| A750-3-2-750-1 | 3 | 750 | 2 | 750 | 1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Ma, E.; Zhao, G. Optimizing the Pore Structure of Bio-Based ACFs through a Simple KOH–Steam Reactivation. Materials 2016, 9, 432. https://doi.org/10.3390/ma9060432
Huang Y, Ma E, Zhao G. Optimizing the Pore Structure of Bio-Based ACFs through a Simple KOH–Steam Reactivation. Materials. 2016; 9(6):432. https://doi.org/10.3390/ma9060432
Chicago/Turabian StyleHuang, Yuxiang, Erni Ma, and Guangjie Zhao. 2016. "Optimizing the Pore Structure of Bio-Based ACFs through a Simple KOH–Steam Reactivation" Materials 9, no. 6: 432. https://doi.org/10.3390/ma9060432
APA StyleHuang, Y., Ma, E., & Zhao, G. (2016). Optimizing the Pore Structure of Bio-Based ACFs through a Simple KOH–Steam Reactivation. Materials, 9(6), 432. https://doi.org/10.3390/ma9060432






