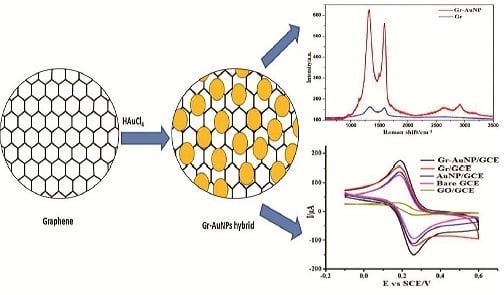
Graphene–Gold Nanoparticles Hybrid—Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor
Abstract
:1. Introduction
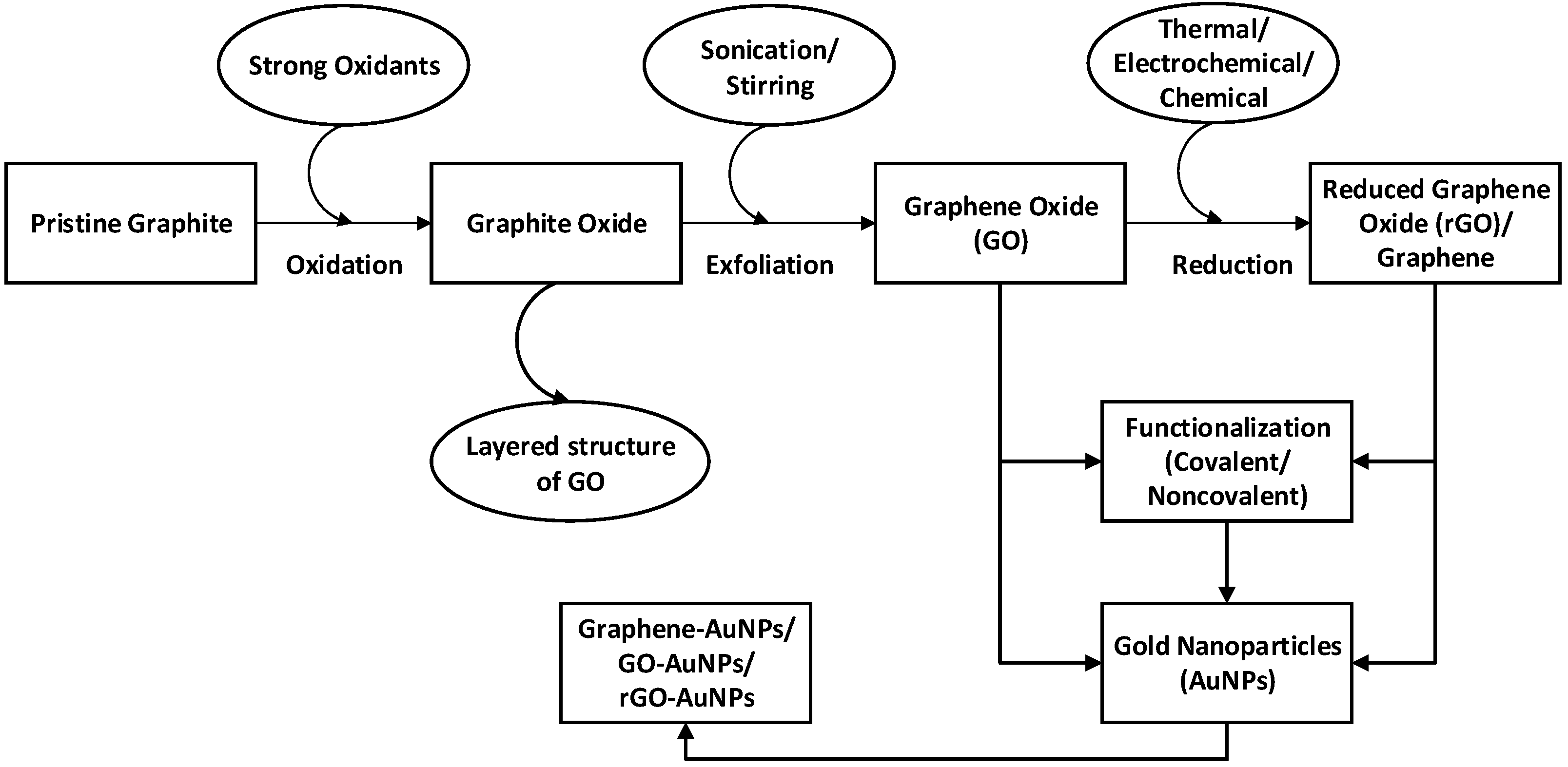
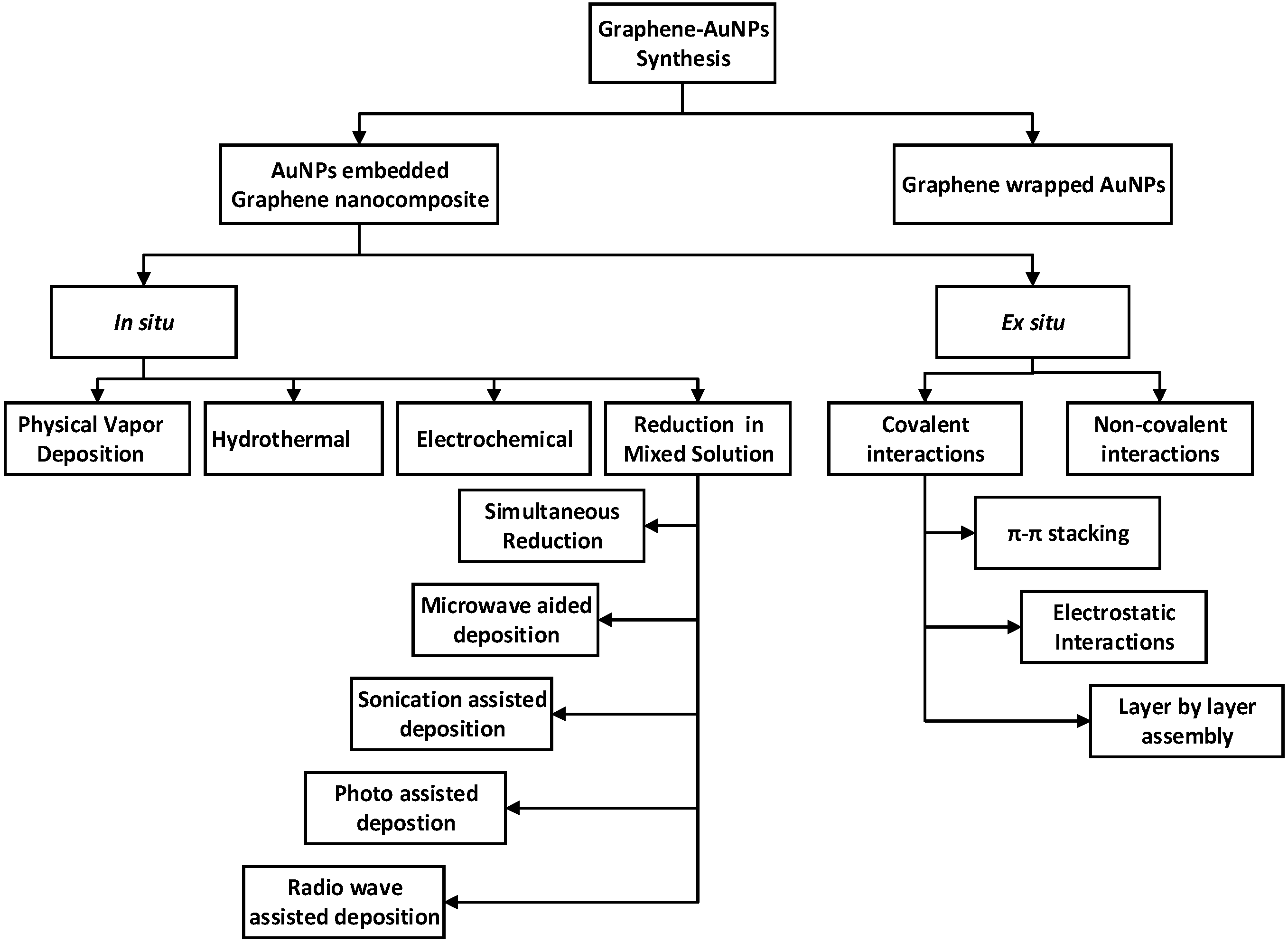
2. Fabrication of Graphene–Gold Nanocomposite
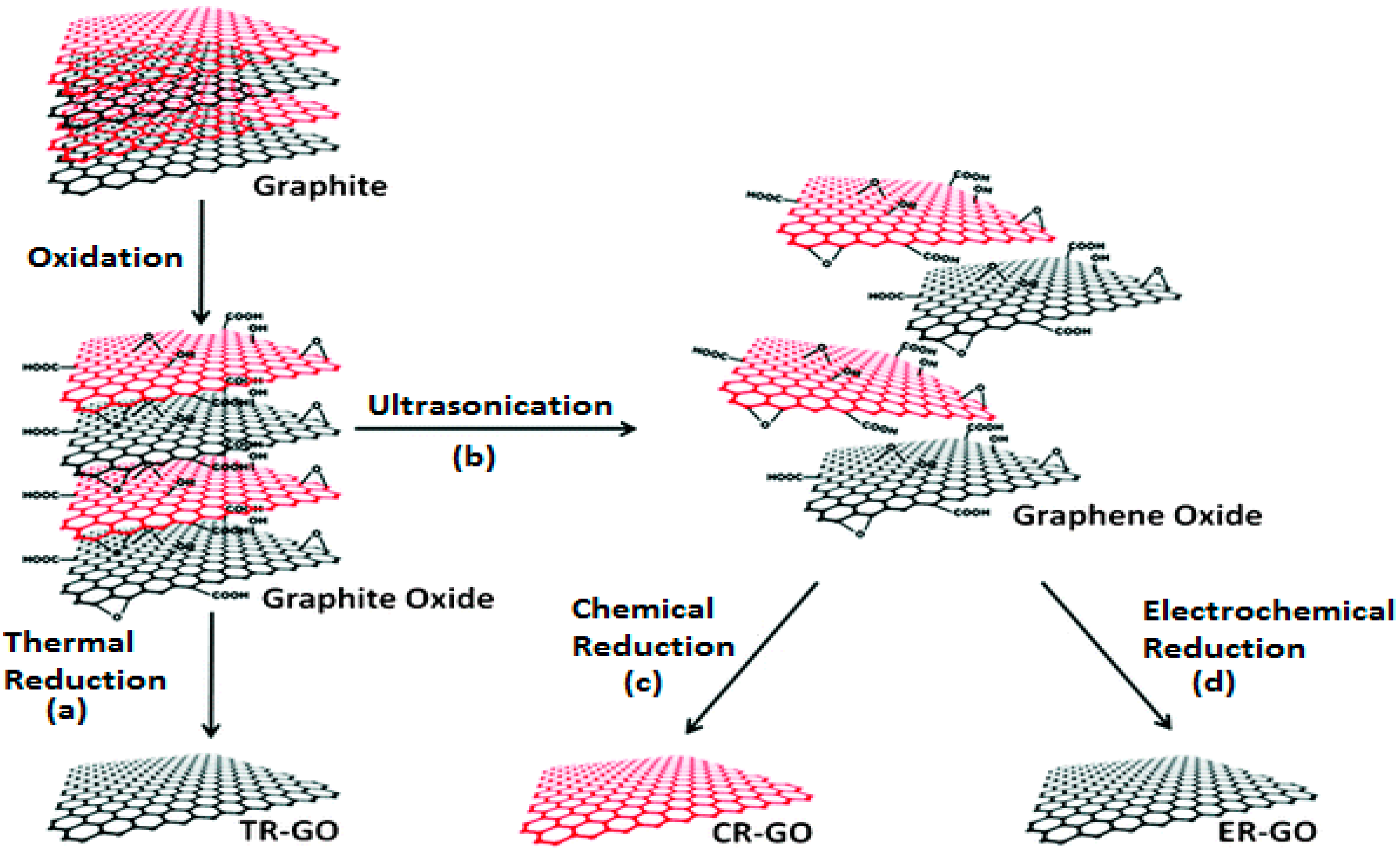
2.1. Synthesis and Functionalization of Graphene
2.2. Synthesis and Functionalization of Graphene–Gold Nanoparticles
2.2.1. In situ Synthesis of Graphene–Gold Nanoparticles
Simultaneous Reduction
Microwave-Assisted Deposition
Sonication-Assisted Deposition
Photo-Assisted Deposition
Radio Wave-Assisted Deposition
2.2.2. Electrochemical Method
2.2.3. Hydrothermal Reduction Method
2.2.4. Physical Vapor Deposition Method
2.2.5. Ex Situ Method
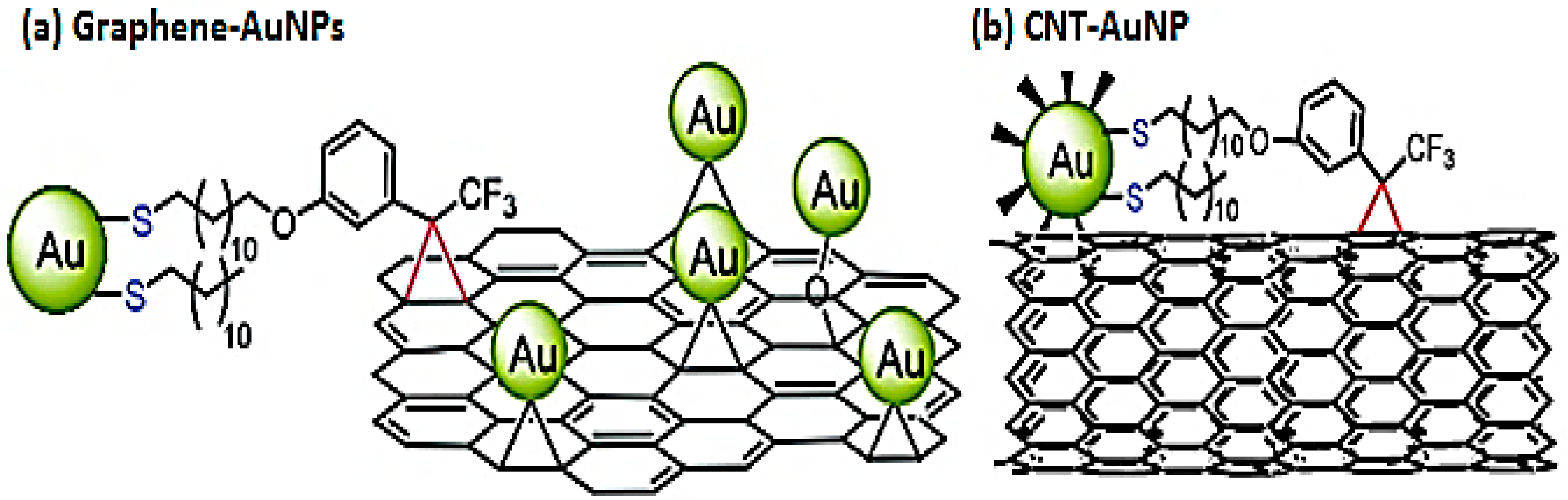
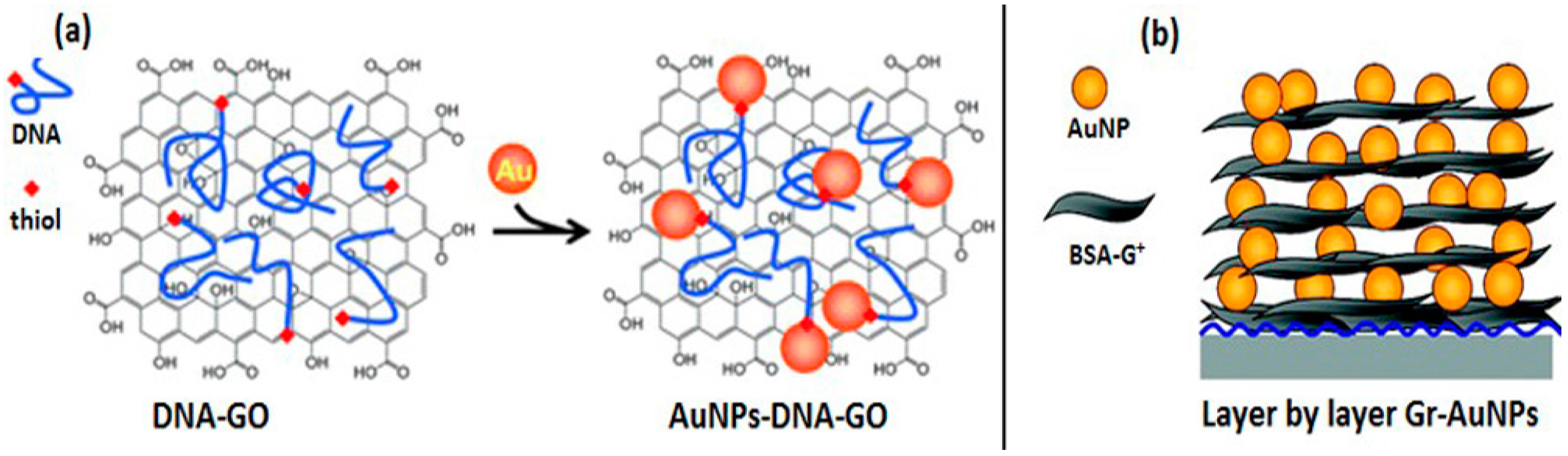
Covalent Interactions
Noncovalent Interactions
- π-π stacking
- Electrostatic interactions
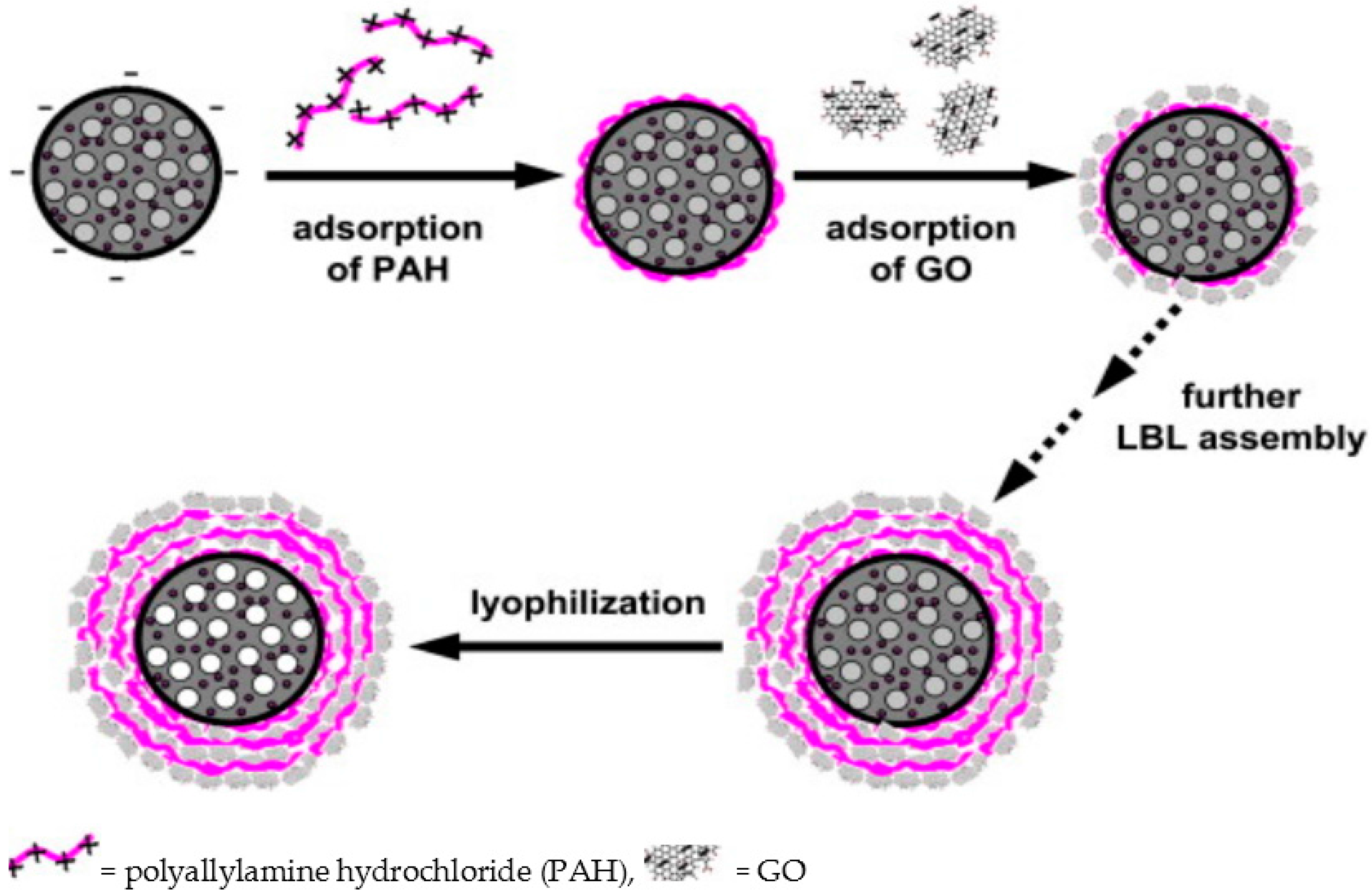
- Layer-by-layer self-assembly
2.2.6. Graphene-Wrapped Gold Nanoparticles
3. Graphene–Gold Nanoparticle Hybrid for Biosensing and Bioimaging Application
3.1. Electrochemical Biosensor
3.2. SERS Biosensor
3.3. SERS Bioimaging
4. Conclusions, Challenges and Perspectives
Acknowledgments
Conflicts of Interest
Abbreviations
| APTES | Aminopropyltriethoxysilane |
| ATP | 4-aminothiphenol |
| Arg | Arginine |
| Ar | Argon |
| CBZ | Carbamazepine |
| CILE | Carbon Ionic Liquid Electrode |
| CNT | Carbon Nanotube |
| CEA | Carcinoembryonic Antigen |
| CTAB | Cetyl-trimethylammonium bromide |
| CR-GO | Chemically Reduced Graphene Oxide |
| CVD | Chemical Vapor Deposition |
| cfu | Colony Forming Unit |
| RDX | Cyclotrimethylenetrinitramine |
| Cy3 | Cysteamine |
| DES | Diethylstilboestrol |
| DHB | Dihydroxybenzoic Acid |
| EIS | Electrochemical Impedance Spectroscopy |
| EPA | Environmental Protection Agency |
| fM | Femtomolar |
| FAO | Food and Agriculture Organization |
| FA | Functionalizing Agent |
| GCE | Glassy Carbon Electrode |
| GOD | Glucose Oxidase |
| AuNC | Gold Nanocrystal |
| AuNP | Gold Nanoparticle |
| AuNS | Gold Nanostructure |
| GA | Graphene Aerogel |
| GNs | Graphene Nanosheets |
| GO | Graphene Oxide |
| Hb | Hemoglobin |
| H2O2 | Hydrogen Peroxide |
| ITO | Indium Titanium Oxide |
| kW | Kilowatt |
| LBL | Layer by layer |
| LOD | Limit of Detection |
| MHz | Megahertz |
| MBA | Mercaptophenyl Boronic Acid |
| MPTMS | 3-mercaptopropyltrimethoxysilane |
| MRSA | Methicillin Resistant Staphylococcus aureus |
| μM | Micromolar |
| mM | Milimolar |
| MDR | Multidrug Resistant |
| NDs | Nanodots |
| nM | Nanomolar |
| nGO | Nano graphene oxide |
| NR | Nano Rod |
| NADH | Nicotinamide Adenine Dinucleotide |
| N2 | Nitrogen |
| ODT | Octadecanethiol |
| ODA | Octadecylamine |
| O2 | Oxygen |
| ppm | Parts per million |
| PEG | Pegylated |
| pM | Picomolar |
| Pt | Platinum |
| PANI | Polyaniline |
| PAH | Poly allylamine hydrochloride |
| PLA | Poly (lactic acid) |
| PVP | Poly(vinylpyrrolidone) |
| KClsat | Potassium Chloride saturated |
| rGO | Reduced Graphene Oxide |
| RA | Reducing Agent |
| RP | Reducing Process |
| Ref. | Reference |
| SCE | Saturated Calomel Electrode |
| SEM | Scanning Electron Microscopy |
| Si | Silica |
| SiO2 | Silica Oxide |
| Ag | Silver |
| AgCl | Silver Chloride |
| AgNS | Silver nanostructures |
| ss | Single Stranded |
| SDS | Sodium Dodecyl Sulfate |
| SA | Stabilizing Agent |
| SERS | Surface Enhanced Raman Spectroscopy/Scattering |
| TR-GO | Thermally Reduced Graphene Oxide |
| 3D | Three-dimensional |
| TB | Toluidine Blue |
| TEM | Transmission Electron Microscopy |
| 2D | Two dimensional |
| wt | Weight |
| WHO | World Health Organization |
References
- Novoselov, K.S.; Geim, A.K.; Morozov, S.; Jiang, D.; Zhang, Y.; Dubonos, S.A.; Grigorieva, I.; Firsov, A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.L.; Kubista, K.D.; Rutter, G.M.; Ruan, M.; de Heer, W.A.; First, P.N.; Stroscio, J.A. Observing the quantization of zero mass carriers in graphene. Science 2009, 324, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, A. The era of carbon allotropes. Nat. Mater. 2010, 9, 868–871. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tan, Y.-W.; Stormer, H.L.; Kim, P. Experimental observation of the quantum hall effect and berry’s phase in graphene. Nature 2005, 438, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorso, F.; Sun, Z.; Hasan, T.; Ferrari, A.C. Graphene photonics and optoelectronics. Nat. Photonics 2010, 4, 611–622. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.V.; Dubonos, S.V.; Firsov, A.A. Two-dimensional gas of massless dirac fermions in graphene. Nature 2005, 438, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Hong, T.-K.; Lee, D.W.; Choi, H.J.; Shin, H.S.; Kim, B.-S. Transparent, flexible conducting hybrid multilayer thin films of multiwalled carbon nanotubes with graphene nanosheets. ACS Nano 2010, 4, 3861–3868. [Google Scholar] [CrossRef] [PubMed]
- Tung, V.C.; Chen, L.-M.; Allen, M.J.; Wassei, J.K.; Nelson, K.; Kaner, R.B.; Yang, Y. Low-temperature solution processing of graphene-carbon nanotube hybrid materials for high-performance transparent conductors. Nano Lett. 2009, 9, 1949–1955. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Bhattacharyya, P. Recent developments on graphene and graphene oxide based solid state gas sensors. Sens. Actuators B Chem. 2012, 173, 1–21. [Google Scholar] [CrossRef]
- Chowdhury, S.; Balasubramanian, R. Recent advances in the use of graphene-family nanoadsorbents for removal of toxic pollutants from wastewater. Adv. Colloid Interface Sci. 2014, 204, 35–56. [Google Scholar] [CrossRef] [PubMed]
- Stoller, M.D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R.S. Graphene-based ultracapacitors. Nano Lett. 2008, 8, 3498–3502. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Bolotin, K.I.; Sikes, K.; Jiang, Z.; Klima, M.; Fudenberg, G.; Hone, J.; Kim, P.; Stormer, H. Ultrahigh electron mobility in suspended graphene. Solid State Commun. 2008, 146, 351–355. [Google Scholar] [CrossRef]
- Balasubramanian, K.; Burghard, M. Biosensors based on carbon nanotubes. Anal. Bioanal. Chem. 2006, 385, 452–468. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Carbon-nanotube based electrochemical biosensors: A review. Electroanalysis 2005, 17, 7–14. [Google Scholar] [CrossRef]
- Ma, H.; Wu, D.; Cui, Z.; Li, Y.; Zhang, Y.; Du, B.; Wei, Q. Graphene-based optical and electrochemical biosensors: A review. Anal. Lett. 2013, 46, 1–17. [Google Scholar] [CrossRef]
- Zhou, K.; Zhu, Y.; Yang, X.; Li, C. One-pot preparation of graphene/fe 3 o 4 composites by a solvothermal reaction. New J. Chem. 2010, 34, 2950–2955. [Google Scholar] [CrossRef]
- Nethravathi, C.; Rajamathi, M.; Ravishankar, N.; Basit, L.; Felser, C. Synthesis of graphene oxide-intercalated α-hydroxides by metathesis and their decomposition to graphene/metal oxide composites. Carbon 2010, 48, 4343–4350. [Google Scholar] [CrossRef]
- Jiang, B.; Tian, C.; Zhou, W.; Wang, J.; Xie, Y.; Pan, Q.; Ren, Z.; Dong, Y.; Fu, D.; Han, J.; et al. In situ growth of TiO2 in interlayers of expanded graphite for the fabrication of TiO2-graphene with enhanced photocatalytic activity. Chemistry 2011, 17, 8379–8387. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Nackashi, D.; Lu, W.; Kittrell, C.; Tour, J.M. Decoration, migration, and aggregation of palladium nanoparticles on graphene sheets. Chem. Mater. 2010, 22, 5695–5699. [Google Scholar] [CrossRef]
- Ismaili, H.; Geng, D.; Sun, A.X.; Kantzas, T.T.; Workentin, M.S. Light-activated covalent formation of gold nanoparticle–graphene and gold nanoparticle–glass composites. Langmuir 2011, 27, 13261–13268. [Google Scholar] [CrossRef] [PubMed]
- Ismaili, H.; Lagugne-Labarthet, F.; Workentin, M.S. Covalently assembled gold nanoparticle-carbon nanotube hybrids via a photoinitiated carbene addition reaction. Chem. Mater. 2011, 23, 1519–1525. [Google Scholar] [CrossRef]
- Bai, S.; Shen, X. Graphene–inorganic nanocomposites. Rsc Adv. 2012, 2, 64–98. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.T.; Ruoff, R.S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 2007, 45, 1558–1565. [Google Scholar] [CrossRef]
- Si, Y.; Samulski, E.T. Exfoliated graphene separated by platinum nanoparticles. Chem. Mater. 2008, 20, 6792–6797. [Google Scholar] [CrossRef]
- Tien, H.-W.; Huang, Y.-L.; Yang, S.-Y.; Wang, J.-Y.; Ma, C.-C.M. The production of graphene nanosheets decorated with silver nanoparticles for use in transparent, conductive films. Carbon 2011, 49, 1550–1560. [Google Scholar] [CrossRef]
- Chen, R.J.; Bangsaruntip, S.; Drouvalakis, K.A.; Kam, N.W.S.; Shim, M.; Li, Y.; Kim, W.; Utz, P.J.; Dai, H. Noncovalent functionalization of carbon nanotubes for highly specific electronic biosensors. Proc. Natl. Acad. Sci. USA 2003, 100, 4984–4989. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, N.; Berry, V. Graphene-based single-bacterium resolution biodevice and DNA transistor: Interfacing graphene derivatives with nanoscale and microscale biocomponents. Nano Lett. 2008, 8, 4469–4476. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.-S.; Zhou, G.; Yin, L.-C.; Ren, W.; Li, F.; Cheng, H.-M. Graphene/metal oxide composite electrode materials for energy storage. Nano Energy 2012, 1, 107–131. [Google Scholar] [CrossRef]
- Zhong, Z.; Patskovskyy, S.; Bouvrette, P.; Luong, J.H.; Gedanken, A. The surface chemistry of Au colloids and their interactions with functional amino acids. J. Phys. Chem. B 2004, 108, 4046–4052. [Google Scholar] [CrossRef]
- Han, J.; Liu, Y.; Guo, R. Facile synthesis of highly stable gold nanoparticles and their unexpected excellent catalytic activity for suzuki−miyaura cross-coupling reaction in water. J. Am. Chem. Soc. 2009, 131, 2060–2061. [Google Scholar] [CrossRef] [PubMed]
- Connor, E.E.; Mwamuka, J.; Gole, A.; Murphy, C.J.; Wyatt, M.D. Gold nanoparticles are taken up by human cells but do not cause acute cytotoxicity. Small 2005, 1, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Elghanian, R.; Storhoff, J.J.; Mucic, R.C.; Letsinger, R.L.; Mirkin, C.A. Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 1997, 277, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Rothberg, L. Colorimetric detection of DNA sequences based on electrostatic interactions with unmodified gold nanoparticles. Proc. Natl. Acad. Sci. USA 2004, 101, 14036–14039. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Vishakante, G.D.; Siddaramaiah, H. Gold nanoparticles: A paradigm shift in biomedical applications. Adv. Colloid Interface Sci. 2013, 199, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Li, B.; Li, J.; Wang, E.; Dong, S. Simple and sensitive aptamer-based colorimetric sensing of protein using unmodified gold nanoparticle probes. Chem. Commun. 2007, 3735–3737. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.L.; Miranda, O.R.; You, C.C.; Rotello, V.M.; Bunz, U.H. Rapid and efficient identification of bacteria using gold-nanoparticle–poly (para-phenyleneethynylene) constructs. Angew. Chem. Int. Ed. 2008, 47, 2590–2594. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Tisch, U.; Adams, O.; Hakim, M.; Shehada, N.; Broza, Y.Y.; Billan, S.; Abdah-Bortnyak, R.; Kuten, A.; Haick, H. Diagnosing lung cancer in exhaled breath using gold nanoparticles. Nat. Nano 2009, 4, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Govindhan, M.; Amiri, M.; Chen, A. Au nanoparticle/graphene nanocomposite as a platform for the sensitive detection of NADH in human urine. Biosens. Bioelectron. 2015, 66, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; He, D.; Duan, J.; Wang, S.; Peng, H.; Wu, H.; Fu, M.; Wang, Y.; Zhang, X. Simple synthesis method of reduced graphene oxide/gold nanoparticle and its application in surface-enhanced raman scattering. Chem. Phys. Lett. 2013, 582, 119–122. [Google Scholar] [CrossRef]
- Sidorov, A.N.; Sławiński, G.W.; Jayatissa, A.; Zamborini, F.P.; Sumanasekera, G.U. A surface-enhanced raman spectroscopy study of thin graphene sheets functionalized with gold and silver nanostructures by seed-mediated growth. Carbon 2012, 50, 699–705. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Paredes, J.; Villar-Rodil, S.; Martinez-Alonso, A.; Tascon, J. Graphene oxide dispersions in organic solvents. Langmuir 2008, 24, 10560–10564. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Piner, R.D.; Chen, X.; Wu, N.; Nguyen, S.T.; Ruoff, R.S. Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly (sodium 4-styrenesulfonate). J. Mater. Chem. 2006, 16, 155–158. [Google Scholar] [CrossRef]
- Jung, I.; Pelton, M.; Piner, R.; Dikin, D.A.; Stankovich, S.; Watcharotone, S.; Hausner, M.; Ruoff, R.S. Simple approach for high-contrast optical imaging and characterization of graphene-based sheets. Nano Lett. 2007, 7, 3569–3575. [Google Scholar] [CrossRef]
- Si, Y.; Samulski, E.T. Synthesis of water soluble graphene. Nano Lett. 2008, 8, 1679–1682. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Fal, V.; Colombo, L.; Gellert, P.; Schwab, M.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Choi, W.; Lahiri, I.; Seelaboyina, R.; Kang, Y.S. Synthesis of graphene and its applications: A review. Crit. Rev. Solid State Mater. Sci. 2010, 35, 52–71. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Reina, A.; Jia, X.; Ho, J.; Nezich, D.; Son, H.; Bulovic, V.; Dresselhaus, M.S.; Kong, J. Large area, few-layer graphene films on arbitrary substrates by chemical vapor deposition. Nano Lett. 2008, 9, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Mattevi, C.; Kim, H.; Chhowalla, M. A review of chemical vapour deposition of graphene on copper. J. Mater. Chem. 2011, 21, 3324–3334. [Google Scholar] [CrossRef]
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Li, T.; Hass, J.; Marchenkov, A.N. Electronic confinement and coherence in patterned epitaxial graphene. Science 2006, 312, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- De Heer, W.A.; Berger, C.; Wu, X.; First, P.N.; Conrad, E.H.; Li, X.; Li, T.; Sprinkle, M.; Hass, J.; Sadowski, M.L. Epitaxial graphene. Solid State Commun. 2007, 143, 92–100. [Google Scholar] [CrossRef]
- Shivaraman, S.; Barton, R.A.; Yu, X.; Alden, J.; Herman, L.; Chandrashekhar, M.; Park, J.; McEuen, P.L.; Parpia, J.M.; Craighead, H.G. Free-standing epitaxial graphene. Nano Lett. 2009, 9, 3100–3105. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; An, J.; Piner, R.D.; Jung, I.; Yang, D.; Velamakanni, A.; Nguyen, S.T.; Ruoff, R.S. Aqueous suspension and characterization of chemically modified graphene sheets. Chem. Mater. 2008, 20, 6592–6594. [Google Scholar] [CrossRef]
- Kosynkin, D.V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J.M. Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nature 2009, 458, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Zhang, L.; Wang, X.; Diankov, G.; Dai, H. Narrow graphene nanoribbons from carbon nanotubes. Nature 2009, 458, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Yang, J.; Park, J.; Gou, X.; Wang, B.; Liu, H.; Yao, J. Facile synthesis and characterization of graphene nanosheets. J. Phys. Chem. C 2008, 112, 8192–8195. [Google Scholar] [CrossRef]
- McAllister, M.J.; Li, J.-L.; Adamson, D.H.; Schniepp, H.C.; Abdala, A.A.; Liu, J.; Herrera-Alonso, M.; Milius, D.L.; Car, R.; Prud’homme, R.K. Single sheet functionalized graphene by oxidation and thermal expansion of graphite. Chem. Mater. 2007, 19, 4396–4404. [Google Scholar] [CrossRef]
- Wong, C.H.A.; Pumera, M. Stripping voltammetry at chemically modified graphenes. RSC Adv. 2012, 2, 6068–6072. [Google Scholar] [CrossRef]
- Wang, H.; Yuan, X.; Wu, Y.; Huang, H.; Peng, X.; Zeng, G.; Zhong, H.; Liang, J.; Ren, M. Graphene-based materials: Fabrication, characterization and application for the decontamination of wastewater and wastegas and hydrogen storage/generation. Adv. Colloid Interface Sci. 2013, 195, 19–40. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, X.; Liu, Z.; Ma, Y.; Huang, Y.; Chen, Y. High-efficiency loading and controlled release of doxorubicin hydrochloride on graphene oxide. J. Phys. Chem. C 2008, 112, 17554–17558. [Google Scholar] [CrossRef]
- Lv, W.; Guo, M.; Liang, M.-H.; Jin, F.-M.; Cui, L.; Zhi, L.; Yang, Q.-H. Graphene-DNA hybrids: Self-assembly and electrochemical detection performance. J. Mater. Chem. 2010, 20, 6668–6673. [Google Scholar] [CrossRef]
- Patil, A.J.; Vickery, J.L.; Scott, T.B.; Mann, S. Aqueous stabilization and self-assembly of graphene sheets into layered bio-nanocomposites using DNA. Adv. Mater. 2009, 21, 3159–3164. [Google Scholar] [CrossRef]
- Bai, H.; Xu, Y.; Zhao, L.; Li, C.; Shi, G. Non-covalent functionalization of graphene sheets by sulfonated polyaniline. Chem. Commun. 2009. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K.; Na, H.-K.; Min, D.-H. Influence of surface functionalization on the growth of gold nanostructures on graphene thin films. Langmuir 2010, 26, 13065–13070. [Google Scholar] [CrossRef] [PubMed]
- Hao, R.; Qian, W.; Zhang, L.; Hou, Y. Aqueous dispersions of tcnq-anion-stabilized graphene sheets. Chem. Commun. 2008. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Bai, H.; Lu, G.; Li, C.; Shi, G. Flexible graphene films via the filtration of water-soluble noncovalent functionalized graphene sheets. J. Am. Chem. Soc. 2008, 130, 5856–5857. [Google Scholar] [CrossRef] [PubMed]
- Niyogi, S.; Bekyarova, E.; Itkis, M.E.; McWilliams, J.L.; Hamon, M.A.; Haddon, R.C. Solution properties of graphite and graphene. J. Am. Chem. Soc. 2006, 128, 7720–7721. [Google Scholar] [CrossRef] [PubMed]
- Quintana, M.; Spyrou, K.; Grzelczak, M.; Browne, W.R.; Rudolf, P.; Prato, M. Functionalization of graphene via 1, 3-dipolar cycloaddition. ACS Nano 2010, 4, 3527–3533. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liu, Q.; Huang, Y.; Ma, Y.; Yin, S.; Zhang, X.; Sun, W.; Chen, Y. Organic photovoltaic devices based on a novel acceptor material: Graphene. Adv. Mater. 2008, 20, 3924–3930. [Google Scholar] [CrossRef]
- Cooper, D.R.; D’Anjou, B.; Ghattamaneni, N.; Harack, B.; Hilke, M.; Horth, A.; Majlis, N.; Massicotte, M.; Vandsburger, L.; Whiteway, E. Experimental review of graphene. ISRN Condens. Matter Phys. 2012. [Google Scholar] [CrossRef]
- Yin, P.T.; Shah, S.; Chhowalla, M.; Lee, K.-B. Design, synthesis, and characterization of graphene–nanoparticle hybrid materials for bioapplications. Chem. Rev. 2015, 115, 2483–2531. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.-H.; Kim, P.; Choi, J.-Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Sutter, P.W.; Flege, J.-I.; Sutter, E.A. Epitaxial graphene on ruthenium. Nat. Mater. 2008, 7, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Ruoff, R.S. Chemical methods for the production of graphenes. Nat. Nanotechnol. 2009, 4, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.R.; Hamid, S.B.A.; Basirun, W.J.; Chowdhury, Z.Z.; Kandjani, A.E.; Bhargava, S.K. Ga doped RGO–TiO2 composite on an ito surface electrode for investigation of photoelectrocatalytic activity under visible light irradiation. New J. Chem. 2015, 39, 369–376. [Google Scholar] [CrossRef]
- Song, J.; Xu, L.; Xing, R.; Li, Q.; Zhou, C.; Liu, D.; Song, H. Synthesis of au/graphene oxide composites for selective and sensitive electrochemical detection of ascorbic acid. Sci. Rep. 2014. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, L.; Chen, B.; Ji, N.; Chen, F.; Zhang, Y.; Zhang, Z. Nanocomposites of size-controlled gold nanoparticles and graphene oxide: Formation and applications in sers and catalysis. Nanoscale 2010, 2, 2733–2738. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.A.; Choi, B.C.; Lim, K.T.; Jeong, Y.T. A simple approach for immobilization of gold nanoparticles on graphene oxide sheets by covalent bonding. Appl. Surf. Sci. 2011, 257, 3350–3357. [Google Scholar] [CrossRef]
- Zhuo, Q.; Ma, Y.; Gao, J.; Zhang, P.; Xia, Y.; Tian, Y.; Sun, X.; Zhong, J.; Sun, X. Facile synthesis of graphene/metal nanoparticle composites via self-catalysis reduction at room temperature. Inorg. Chem. 2013, 52, 3141–3147. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Li, J.; Kong, Y.; Li, X.; Tao, Y.; Li, S.; Wang, Y. In situ growth of Au nanocrystals on graphene oxide sheets. Nanoscale 2014, 6, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Bao, Q.; Tang, L.A.L.; Zhong, Y.; Loh, K.P. Hydrothermal dehydration for the “green” reduction of exfoliated graphene oxide to graphene and demonstration of tunable optical limiting properties. Chem. Mater. 2009, 21, 2950–2956. [Google Scholar] [CrossRef]
- Liu, H.; Su, X.; Duan, C.; Dong, X.; Zhou, S.; Zhu, Z. Microwave-assisted hydrothermal synthesis of Au NPs–graphene composites for H2O2 detection. J. Electroanal. Chem. 2014, 731, 36–42. [Google Scholar] [CrossRef]
- Hu, Y.; Jin, J.; Wu, P.; Zhang, H.; Cai, C. Graphene–gold nanostructure composites fabricated by electrodeposition and their electrocatalytic activity toward the oxygen reduction and glucose oxidation. Electrochim. Acta 2010, 56, 491–500. [Google Scholar] [CrossRef]
- Sun, X.; Jia, M.; Guan, L.; Ji, J.; Zhang, Y.; Tang, L.; Li, Z. Multilayer graphene–gold nanocomposite modified stem-loop DNA biosensor for peanut allergen-Ara h1 detection. Food Chem. 2015, 172, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Benvidi, A.; Dehghani-Firouzabadi, A.; Mazloum-Ardakani, M.; Mirjalili, B.-B.F.; Zare, R. Electrochemical deposition of gold nanoparticles on reduced graphene oxide modified glassy carbon electrode for simultaneous determination of levodopa, uric acid and folic acid. J. Electroanal. Chem. 2015, 736, 22–29. [Google Scholar] [CrossRef]
- Ma, X.; Chen, M. Electrochemical sensor based on graphene doped gold nanoparticles modified electrode for detection of diethylstilboestrol. Sens. Actuators B Chem. 2015, 215, 445–450. [Google Scholar] [CrossRef]
- Li, S.-J.; Deng, D.-H.; Shi, Q.; Liu, S.-R. Electrochemical synthesis of a graphene sheet and gold nanoparticle-based nanocomposite, and its application to amperometric sensing of dopamine. Microchim. Acta 2012, 177, 325–331. [Google Scholar] [CrossRef]
- Kong, B.-S.; Geng, J.; Jung, H.-T. Layer-by-layer assembly of graphene and gold nanoparticles by vacuum filtration and spontaneous reduction of gold ions. Chem. Commun. 2009. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.-Q.; Wu, J.-F.; Zhao, G.-C. Direct electrochemistry of hemoglobin at a graphene gold nanoparticle composite film for nitric oxide biosensing. Sensors 2013, 13, 7492–7504. [Google Scholar] [CrossRef] [PubMed]
- Muszynski, R.; Seger, B.; Kamat, P.V. Decorating graphene sheets with gold nanoparticles. J. Phys. Chem. C 2008, 112, 5263–5266. [Google Scholar] [CrossRef]
- Mao, S.; Lu, G.; Yu, K.; Bo, Z.; Chen, J. Specific protein detection using thermally reduced graphene oxide sheet decorated with gold nanoparticle-antibody conjugates. Adv. Mater. 2010, 22, 3521–3526. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, S.; Lu, W.; Wang, L.; Tian, J.; Sun, X. In situ green synthesis of Au nanostructures on graphene oxide and their application for catalytic reduction of 4-nitrophenol. Catal. Sci. Technol. 2011, 1, 1142–1144. [Google Scholar] [CrossRef]
- Shervedani, R.K.; Amini, A. Novel graphene-gold hybrid nanostructures constructed via sulfur modified graphene: Preparation and characterization by surface and electrochemical techniques. Electrochim. Acta 2014, 121, 376–385. [Google Scholar] [CrossRef]
- Lin, Y.; Baggett, D.W.; Kim, J.-W.; Siochi, E.J.; Connell, J.W. Instantaneous formation of metal and metal oxide nanoparticles on carbon nanotubes and graphene via solvent-free microwave heating. ACS Appl. Mater. Interfaces 2011, 3, 1652–1664. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Wang, X.; Xu, C.; Wang, J.; Wan, L.; Zhang, M.; Shang, X. Microwave-assisted synthesis of graphene nanosheets–gold nanocomposites with enhancing electrochemical response. Fuller. Nanotub. Carbon Nanostruct. 2012, 20, 31–40. [Google Scholar] [CrossRef]
- Jasuja, K.; Linn, J.; Melton, S.; Berry, V. Microwave-reduced uncapped metal nanoparticles on graphene: Tuning catalytic, electrical, and raman properties. J. Phys. Chem. Lett. 2010, 1, 1853–1860. [Google Scholar] [CrossRef]
- Sharma, P.; Darabdhara, G.; Reddy, T.M.; Borah, A.; Bezboruah, P.; Gogoi, P.; Hussain, N.; Sengupta, P.; Das, M.R. Synthesis, characterization and catalytic application of Au NPs-reduced graphene oxide composites material: An eco-friendly approach. Catal. Commun. 2013, 40, 139–144. [Google Scholar] [CrossRef]
- Ponnusamy, V.K.; Mani, V.; Chen, S.-M.; Huang, W.-T.; Jen, J.-F. Rapid microwave assisted synthesis of graphene nanosheets/polyethyleneimine/gold nanoparticle composite and its application to the selective electrochemical determination of dopamine. Talanta 2014, 120, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Lee, K.G.; Lee, S.J.; Park, T.J.; Wi, R.; Kim, D.H. Synthesis of graphene-gold nanocomposites via sonochemical reduction. J. Nanosci. Nanotechnol. 2011, 11, 6095–6101. [Google Scholar] [CrossRef] [PubMed]
- Vinodgopal, K.; Neppolian, B.; Lightcap, I.V.; Grieser, F.; Ashokkumar, M.; Kamat, P.V. Sonolytic design of graphene–Au nanocomposites. Simultaneous and sequential reduction of graphene oxide and Au (iii). J. Phys. Chem. Lett. 2010, 1, 1987–1993. [Google Scholar] [CrossRef]
- Caires, A.; Alves, D.; Fantini, C.; Ferlauto, A.; Ladeira, L. One-pot in situ photochemical synthesis of graphene oxide/gold nanorod nanocomposites for surface-enhanced raman spectroscopy. RSC Adv. 2015, 5, 46552–46557. [Google Scholar] [CrossRef]
- Huang, X.; Zhou, X.; Wu, S.; Wei, Y.; Qi, X.; Zhang, J.; Boey, F.; Zhang, H. Reduced graphene oxide-templated photochemical synthesis and in situ assembly of Au nanodots to orderly patterned Au nanodot chains. Small 2010, 6, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cui, X.; Wang, Q.; Wang, H.; Zheng, X.; Liu, C.; Xue, T.; Wang, S.; Zheng, W. One-pot photochemical synthesis of ultrathin Au nanocrystals on co-reduced graphene oxide and its application. J. Colloid Interface Sci. 2012, 383, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.-E.; Lin, C.-Y.; Chang, H.-Y.; Huang, C.-H.; Lin, H.-Y.; Chen, C.-H.; Hsu, C.-C.; Chang, C.-S.; Sen Chien, F.S. Surface-enhanced raman scattering of graphene with photo-assisted-synthesized gold nanoparticles. Opt. Express 2013, 21, 6547–6554. [Google Scholar] [CrossRef] [PubMed]
- Pruneanu, S.; Pogacean, F.; Biris, A.R.; Ardelean, S.; Canpean, V.; Blanita, G.; Dervishi, E.; Biris, A.S. Novel graphene-gold nanoparticle modified electrodes for the high sensitivity electrochemical spectroscopy detection and analysis of carbamazepine. J. Phys. Chem. C 2011, 115, 23387–23394. [Google Scholar] [CrossRef]
- Zhang, H.; Hines, D.; Akins, D.L. Synthesis of a nanocomposite composed of reduced graphene oxide and gold nanoparticles. Dalton Trans. 2014, 43, 2670–2675. [Google Scholar] [CrossRef] [PubMed]
- Borowiec, J.; Wang, R.; Zhu, L.; Zhang, J. Synthesis of nitrogen-doped graphene nanosheets decorated with gold nanoparticles as an improved sensor for electrochemical determination of chloramphenicol. Electrochim. Acta 2013, 99, 138–144. [Google Scholar] [CrossRef]
- Wojnicki, M.; Luty-Błocho, M.; Grzonka, J.; Pacławski, K.; Kurzydłowski, K.J.; Fitzner, K. Micro-continuous flow synthesis of gold nanoparticles and integrated deposition on suspended sheets of graphene oxide. Chem. Eng. J. 2013, 225, 597–606. [Google Scholar] [CrossRef]
- Tabrizi, M.A.; Varkani, J.N. Green synthesis of reduced graphene oxide decorated with gold nanoparticles and its glucose sensing application. Sens. Actuators B Chem. 2014, 202, 475–482. [Google Scholar] [CrossRef]
- Goncalves, G.; Marques, P.A.; Granadeiro, C.M.; Nogueira, H.I.; Singh, M.; Gracio, J. Surface modification of graphene nanosheets with gold nanoparticles: The role of oxygen moieties at graphene surface on gold nucleation and growth. Chem. Mater. 2009, 21, 4796–4802. [Google Scholar] [CrossRef]
- Iliut, M.; Leordean, C.; Canpean, V.; Teodorescu, C.-M.; Astilean, S. A new green, ascorbic acid-assisted method for versatile synthesis of Au–graphene hybrids as efficient surface-enhanced raman scattering platforms. J. Mater. Chem. C 2013, 1, 4094–4104. [Google Scholar] [CrossRef]
- Fu, W.L.; Zhen, S.J.; Huang, C.Z. One-pot green synthesis of graphene oxide/gold nanocomposites as sers substrates for malachite green detection. Analyst 2013, 138, 3075–3081. [Google Scholar] [CrossRef] [PubMed]
- Xi, Q.; Chen, X.; Evans, D.G.; Yang, W. Gold nanoparticle-embedded porous graphene thin films fabricated via layer-by-layer self-assembly and subsequent thermal annealing for electrochemical sensing. Langmuir 2012, 28, 9885–9892. [Google Scholar] [CrossRef] [PubMed]
- Li, R.-Y.; Zhang, J.-J.; Wang, Z.-P.; Li, Z.-J.; Liu, J.-K.; Gu, Z.-G.; Wang, G.-L. Novel graphene-gold nanohybrid with excellent electrocatalytic performance for the electrochemical detection of glucose. Sens. Actuators B Chem. 2015, 208, 421–428. [Google Scholar]
- Dharuman, V.; Hahn, J.H.; Jayakumar, K.; Teng, W. Electrochemically reduced graphene–gold nano particle composite on indium tin oxide for label free immuno sensing of estradiol. Electrochim. Acta 2013, 114, 590–597. [Google Scholar] [CrossRef]
- Zhou, L.-T.; Li, R.-Y.; Li, Z.-J.; Xia, Q.-F.; Fang, Y.-J.; Liu, J.-K. An immunosensor for ultrasensitive detection of aflatoxin B1 with an enhanced electrochemical performance based on graphene/conducting polymer/gold nanoparticles/the ionic liquid composite film on modified gold electrode with electrodeposition. Sens. Actuators B Chem. 2012, 174, 359–365. [Google Scholar]
- Niu, X.; Yang, W.; Wang, G.; Ren, J.; Guo, H.; Gao, J. A novel electrochemical sensor of bisphenol a based on stacked graphene nanofibers/gold nanoparticles composite modified glassy carbon electrode. Electrochim. Acta 2013, 98, 167–175. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, J.; Li, D.; Li, Y. An electrochemical aptasensor based on gold nanoparticles dotted graphene modified glassy carbon electrode for label-free detection of bisphenol a in milk samples. Food Chem. 2014, 162, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, X. Electrochemical co-reduction synthesis of graphene/nano-gold composites and its application to electrochemical glucose biosensor. Electrochim. Acta 2013, 112, 774–782. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, J.; Yin, Z.; Wu, S.; Mandler, D.; Zhang, H. Fabrication of nanoelectrode ensembles by electrodepositon of Au nanoparticles on single-layer graphene oxide sheets. Nanoscale 2012, 4, 2728–2733. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; You, Z.; Cheng, Y.; Sha, H.; Li, G.; Zhu, H.; Sun, W. Application of nanosized gold and graphene modified carbon ionic liquid electrode for the sensitive electrochemical determination of folic acid. J. Mol. Liq. 2015, 204, 112–117. [Google Scholar] [CrossRef]
- Zaretski, A.V.; Root, S.E.; Savchenko, A.; Molokanova, E.; Printz, A.D.; Jibril, L.; Arya, G.; Mercola, M.; Lipomi, D.J. Metallic nanoislands on graphene as highly sensitive transducers of mechanical, biological, and optical signals. Nano Lett. 2016, 16, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.A.; Bell, G.R.; Rourke, J.P.; Sanchez, A.M.; Elkin, M.D.; Hickey, B.J.; Wilson, N.R. Physical vapor deposition of metal nanoparticles on chemically modified graphene: Observations on metal–graphene interactions. Small 2011, 7, 3202–3210. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Somers, L.A.; Dan, Y.; Ly, T.; Kybert, N.J.; Mele, E.; Johnson, A.C. Size-selective nanoparticle growth on few-layer graphene films. Nano Lett. 2010, 10, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Vélez, G.Y.; Encinas, A.; Quintana, M. Immobilization of metal and metal oxide nanoparticles on graphene. Funct. Gr. 2014. [Google Scholar] [CrossRef]
- Vilatela, J.J.; Eder, D. Nanocarbon composites and hybrids in sustainability: A review. ChemSusChem 2012, 5, 456–478. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, Y.; Li, Y.; Li, J.; Deng, Z. Noncovalent DNA decorations of graphene oxide and reduced graphene oxide toward water-soluble metal–carbon hybrid nanostructures via self-assembly. J. Mater. Chem. 2010, 20, 900–906. [Google Scholar] [CrossRef]
- YoungáChoi, J.; SeokáSeo, T. DNA mediated water-dispersible graphene fabrication and gold nanoparticle-graphene hybrid. Chem. Commun. 2010, 46, 2844–2846. [Google Scholar]
- Wang, Y.; Zhen, S.J.; Zhang, Y.; Li, Y.F.; Huang, C.Z. Facile fabrication of metal nanoparticle/graphene oxide hybrids: A new strategy to directly illuminate graphene for optical imaging. J. Phys. Chem. C 2011, 115, 12815–12821. [Google Scholar] [CrossRef]
- Liu, S.; Yan, J.; He, G.; Zhong, D.; Chen, J.; Shi, L.; Zhou, X.; Jiang, H. Layer-by-layer assembled multilayer films of reduced graphene oxide/gold nanoparticles for the electrochemical detection of dopamine. J. Electroanal. Chem. 2012, 672, 40–44. [Google Scholar] [CrossRef]
- Turcheniuk, K.; Boukherroub, R.; Szunerits, S. Gold–graphene nanocomposites for sensing and biomedical applications. J. Mater. Chem. B 2015, 3, 4301–4324. [Google Scholar] [CrossRef]
- Chopra, N.; Bachas, L.G.; Knecht, M.R. Fabrication and biofunctionalization of carbon-encapsulated Au nanoparticles. Chem. Mater. 2009, 21, 1176–1178. [Google Scholar] [CrossRef]
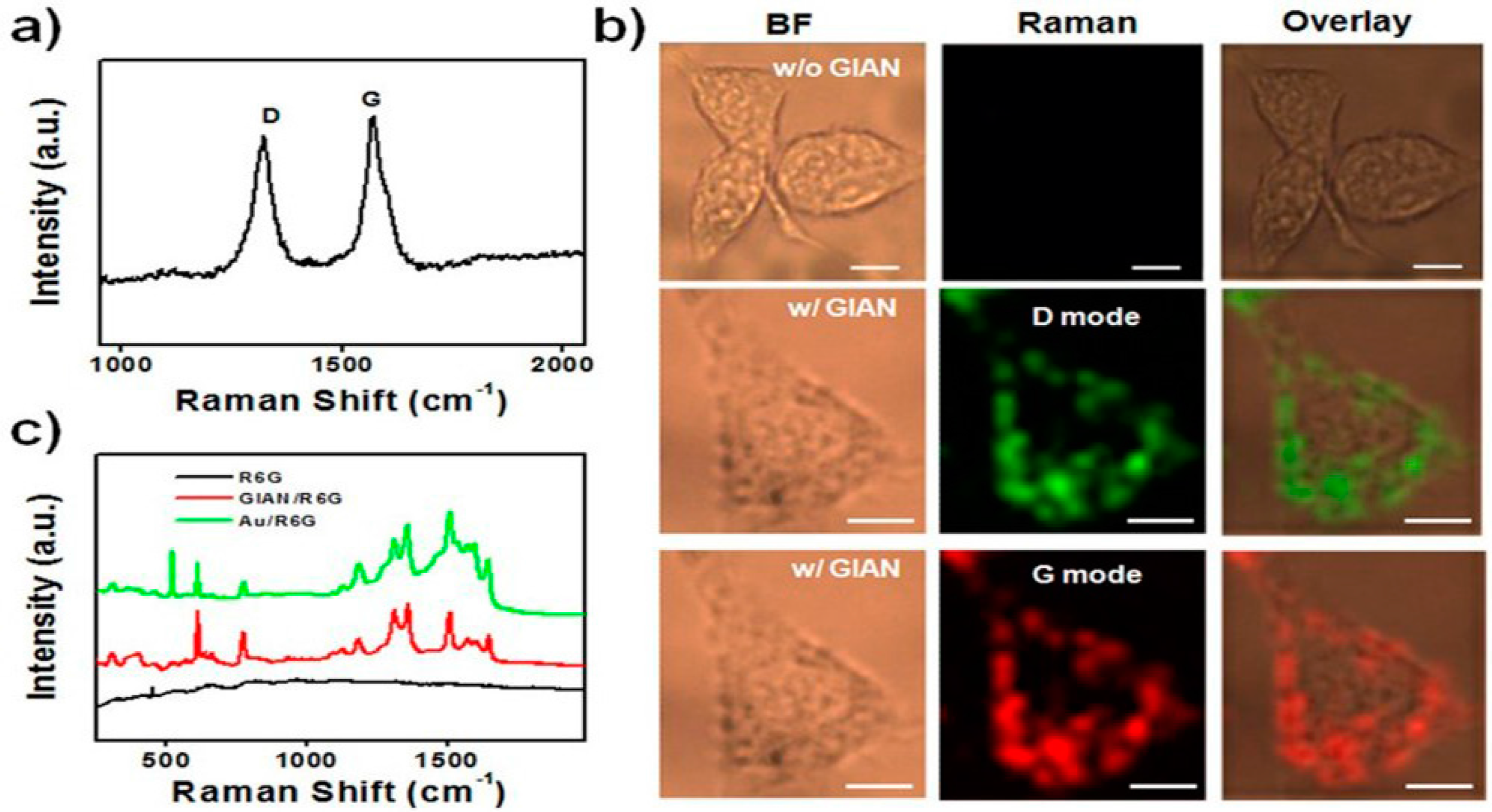
- Bian, X.; Song, Z.-L.; Qian, Y.; Gao, W.; Cheng, Z.-Q.; Chen, L.; Liang, H.; Ding, D.; Nie, X.-K.; Chen, Z. Fabrication of graphene-isolated-au-nanocrystal nanostructures for multimodal cell imaging and photothermal-enhanced chemotherapy. Sci. Rep. 2014. [Google Scholar] [CrossRef] [PubMed]
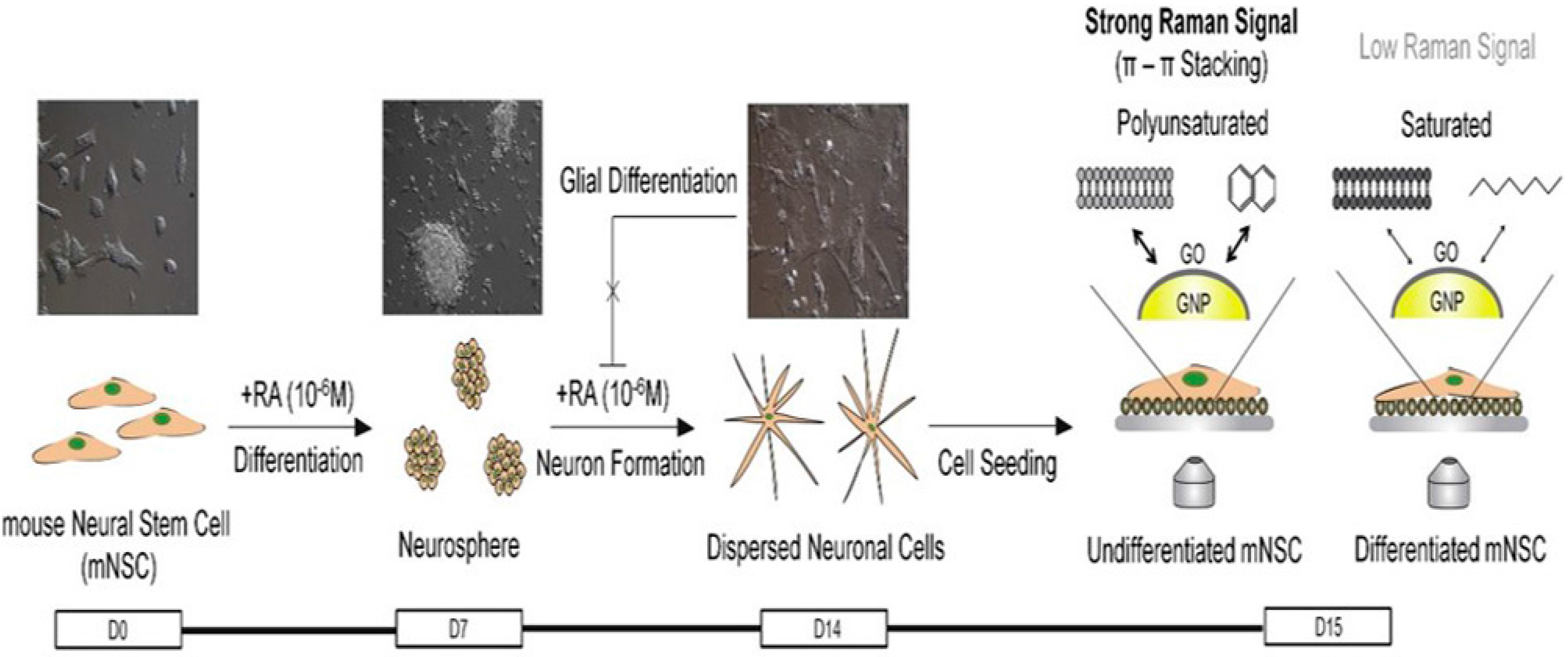
- Kim, T.-H.; Lee, K.-B.; Choi, J.-W. 3D graphene oxide-encapsulated gold nanoparticles to detect neural stem cell differentiation. Biomaterials 2013, 34, 8660–8670. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Qu, Q.; Zhao, Y.; Luo, Z.; Zhao, Y.; Ng, K.W.; Zhao, Y. Graphene oxide wrapped gold nanoparticles for intracellular raman imaging and drug delivery. J. Mater. Chem. B 2013, 1, 6495–6500. [Google Scholar] [CrossRef]
- Cui, Y.; Zhou, D.; Sui, Z.; Han, B. Sonochemical synthesis of graphene oxide-wrapped gold nanoparticles hybrid materials: Visible light photocatalytic activity. Chin. J. Chem. 2015, 33, 119–124. [Google Scholar] [CrossRef]
- Xu, C.; Yang, D.; Mei, L.; Lu, B.; Chen, L.; Li, Q.; Zhu, H.; Wang, T. Encapsulating gold nanoparticles or nanorods in graphene oxide shells as a novel gene vector. ACS Appl. Mater. Interfaces 2013, 5, 2715–2724. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.-K.; Barhoumi, A.; Wylie, R.G.; Reznor, G.; Langer, R.S.; Kohane, D.S. Enhanced photothermal effect of plasmonic nanoparticles coated with reduced graphene oxide. Nano Lett. 2013, 13, 4075–4079. [Google Scholar] [CrossRef] [PubMed]
- Turcheniuk, K.; Hage, C.-H.; Spadavecchia, J.; Serrano, A.Y.; Larroulet, I.; Pesquera, A.; Zurutuza, A.; Pisfil, M.G.; Héliot, L.; Boukaert, J. Plasmonic photothermal destruction of uropathogenic E. coli with reduced graphene oxide and core/shell nanocomposites of gold nanorods/reduced graphene oxide. J. Mater. Chem. B 2015, 3, 375–386. [Google Scholar] [CrossRef]
- Yun, Y.J.; Song, K.-B. Preparation and characterization of graphene oxide encapsulated gold nanoparticles. J. Nanosci. Nanotechnol. 2013, 13, 7376–7380. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zhang, W.; Liang, O.; Pantoja, M.; Katzer, J.; Schroeder, T.; Xie, Y.-H. Giant optical response from graphene–plasmonic system. ACS Nano 2012, 6, 6244–6249. [Google Scholar] [CrossRef] [PubMed]
- Heeg, S.; Fernandez-Garcia, R.; Oikonomou, A.; Schedin, F.; Narula, R.; Maier, S.A.; Vijayaraghavan, A.; Reich, S. Polarized plasmonic enhancement by Au nanostructures probed through raman scattering of suspended graphene. Nano Lett. 2012, 13, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wang, J.; Ke, H.; Wang, S.; Dai, Z. Graphene oxide modified pla microcapsules containing gold nanoparticles for ultrasonic/ct bimodal imaging guided photothermal tumor therapy. Biomaterials 2013, 34, 4794–4802. [Google Scholar] [CrossRef] [PubMed]
- Ronkainen, N.J.; Halsall, H.B.; Heineman, W.R. Electrochemical biosensors. Chem. Soc. Rev. 2010, 39, 1747–1763. [Google Scholar] [CrossRef] [PubMed]
- Chikkaveeraiah, B.V.; Bhirde, A.A.; Morgan, N.Y.; Eden, H.S.; Chen, X. Electrochemical immunosensors for detection of cancer protein biomarkers. ACS Nano 2012, 6, 6546–6561. [Google Scholar] [CrossRef] [PubMed]
- Pohanka, M.; Skládal, P. Electrochemical biosensors–principles and applications. J. Appl. Biomed 2008, 6, 57–64. [Google Scholar]
- Shao, Y.; Wang, J.; Wu, H.; Liu, J.; Aksay, I.A.; Lin, Y. Graphene based electrochemical sensors and biosensors: A review. Electroanalysis 2010, 22, 1027–1036. [Google Scholar] [CrossRef]
- Yin, P.T.; Kim, T.-H.; Choi, J.-W.; Lee, K.-B. Prospects for graphene–nanoparticle-based hybrid sensors. Phys. Chem. Chem. Phys. 2013, 15, 12785–12799. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, A.; Pumera, M. Nanographite impurities dominate electrochemistry of carbon nanotubes. Chem. Eur. J. 2010, 16, 10946–10949. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Morrin, A.; Killard, A.J.; Smyth, M.R. Application of nanoparticles in electrochemical sensors and biosensors. Electroanalysis 2006, 18, 319–326. [Google Scholar] [CrossRef]
- Plowman, B.J.; Bhargava, S.K.; O’Mullane, A.P. Electrochemical fabrication of metallic nanostructured electrodes for electroanalytical applications. Analyst 2011, 136, 5107–5119. [Google Scholar] [CrossRef] [PubMed]
- Plowman, B.J.; O’Mullane, A.P.; Bhargava, S.K. The active site behaviour of electrochemically synthesised gold nanomaterials. Faraday Discuss. 2011, 152, 43–62. [Google Scholar] [CrossRef] [PubMed]
- Plowman, B.J.; Field, M.R.; Bhargava, S.K.; O’Mullane, A.P. Exploiting the facile oxidation of evaporated gold films to drive electroless silver deposition for the creation of bimetallic Au/Ag surfaces. ChemElectroChem 2014, 1, 76–82. [Google Scholar] [CrossRef]
- Shan, C.; Yang, H.; Han, D.; Zhang, Q.; Ivaska, A.; Niu, L. Graphene/AuNPs/chitosan nanocomposites film for glucose biosensing. Biosens. Bioelectron. 2010, 25, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, Y.; Sun, D.; Tian, D.; Zhang, J.; Zhu, J.-J. Fabrication of gold nanoparticles on bilayer graphene for glucose electrochemical biosensing. J. Mater. Chem. 2011, 21, 7604–7611. [Google Scholar] [CrossRef]
- Xu, Q.; Gu, S.-X.; Jin, L.; Zhou, Y.-E.; Yang, Z.; Wang, W.; Hu, X. Graphene/polyaniline/gold nanoparticles nanocomposite for the direct electron transfer of glucose oxidase and glucose biosensing. Sens. Actuators B Chem. 2014, 190, 562–569. [Google Scholar] [CrossRef]
- German, N.; Ramanavicius, A.; Ramanaviciene, A. Electrochemical deposition of gold nanoparticles on graphite rod for glucose biosensing. Sens. Actuators B Chem. 2014, 203, 25–34. [Google Scholar] [CrossRef]
- Cao, X.; Ye, Y.; Li, Y.; Xu, X.; Yu, J.; Liu, S. Self-assembled glucose oxidase/graphene/gold ternary nanocomposites for direct electrochemistry and electrocatalysis. J. Electroanal. Chem. 2013, 697, 10–14. [Google Scholar] [CrossRef]
- Ekram, H.; Galal, A.; Atta, N.F. Electrochemistry of glucose at gold nanoparticles modified graphite/srpdo 3 electrode–towards a novel non-enzymatic glucose sensor. J. Electroanal. Chem. 2015, 749, 42–52. [Google Scholar]
- Fang, Y.; Guo, S.; Zhu, C.; Zhai, Y.; Wang, E. Self-assembly of cationic polyelectrolyte-functionalized graphene nanosheets and gold nanoparticles: A two-dimensional heterostructure for hydrogen peroxide sensing. Langmuir 2010, 26, 11277–11282. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Wang, X.; Shiu, K.-K.; Zhu, Y.; Wang, J.; Li, Q.; Chen, B.; Jiang, H. Layer-by-layer assembly of graphene, Au and poly (toluidine blue O) films sensor for evaluation of oxidative stress of tumor cells elicited by hydrogen peroxide. Biosens. Bioelectron. 2013, 41, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shi, A.; Fang, X.; Han, X.; Zhang, Y. An ultrasensitive supersandwich electrochemical DNA biosensor based on gold nanoparticles decorated reduced graphene oxide. Anal. Biochem. 2015, 469, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.-P.; Hu, Y.; Liu, P.; Deng, Y.-N.; Wang, P.; Chen, W.; Liu, A.-L.; Chen, Y.-Z.; Lin, X.-H. Label-free electrochemical DNA biosensor for rapid detection of mutidrug resistance gene based on Au nanoparticles/toluidine blue–graphene oxide nanocomposites. Sens. Actuators B Chem. 2015, 207, 269–276. [Google Scholar] [CrossRef]
- Liu, C.; Jiang, D.; Xiang, G.; Liu, L.; Liu, F.; Pu, X. An electrochemical DNA biosensor for the detection of mycobacterium tuberculosis, based on signal amplification of graphene and a gold nanoparticle–polyaniline nanocomposite. Analyst 2014, 139, 5460–5465. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ping, J.; Ye, Z.; Wu, J.; Ying, Y. Impedimetric immunosensor based on gold nanoparticles modified graphene paper for label-free detection of Escherichia coli O157:H7. Biosens. Bioelectron. 2013, 49, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Cao, X.; Yu, M. Electrochemical immunoassay based on gold nanoparticles and reduced graphene oxide functionalized carbon ionic liquid electrode. Microchem. J. 2012, 103, 125–130. [Google Scholar] [CrossRef]
- Jiang, J.; Fan, W.; Du, X. Nitrite electrochemical biosensing based on coupled graphene and gold nanoparticles. Biosens. Bioelectron. 2014, 51, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Yola, M.L.; Qureshi, M.S.; Solak, A.O.; Atar, N.; Üstündağ, Z. A novel impedimetric biosensor based on graphene oxide/gold nanoplatform for detection of DNA arrays. Sens. Actuators B Chem. 2013, 188, 1201–1211. [Google Scholar] [CrossRef]
- Plowman, B.; Ippolito, S.J.; Bansal, V.; Sabri, Y.M.; O’Mullane, A.P.; Bhargava, S.K. Gold nanospikes formed through a simple electrochemical route with high electrocatalytic and surface enhanced raman scattering activity. Chem. Commun. 2009. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.K.; Ott, A.; O’Mullane, A.P.; Bhargava, S.K. The facile formation of silver dendritic structures in the absence of surfactants and their electrochemical and sers properties. Colloids Surf. A Physicochem. Eng. Asp. 2011, 386, 98–106. [Google Scholar] [CrossRef]
- Selvakannan, P.; Ramanathan, R.; Plowman, B.J.; Sabri, Y.M.; Daima, H.K.; O’Mullane, A.P.; Bansal, V.; Bhargava, S.K. Probing the effect of charge transfer enhancement in off resonance mode sers via conjugation of the probe dye between silver nanoparticles and metal substrates. Phys. Chem. Chem. Phys. 2013, 15, 12920–12929. [Google Scholar] [CrossRef] [PubMed]
- Kandjani, A.E.; Mohammadtaheri, M.; Thakkar, A.; Bhargava, S.K.; Bansal, V. Zinc oxide/silver nanoarrays as reusable sers substrates with controllable ‘hot-spots’ for highly reproducible molecular sensing. J. Colloid Interface Sci. 2014, 436, 251–257. [Google Scholar] [CrossRef] [PubMed]
- O’Mullane, A.P.; Ippolito, S.J.; Sabri, Y.M.; Bansal, V.; Bhargava, S.K. Premonolayer oxidation of nanostructured gold: An important factor influencing electrocatalytic activity. Langmuir 2009, 25, 3845–3852. [Google Scholar] [CrossRef] [PubMed]
- Pearson, A.; O’Mullane, A.P.; Bhargava, S.K.; Bansal, V. Comparison of nanostructures obtained from galvanic replacement in water and an ionic liquid for applications in electrocatalysis and sers. Electrochem. Commun. 2012, 25, 87–90. [Google Scholar] [CrossRef]
- Abalde-Cela, S.; Carregal-Romero, S.; Coelho, J.P.; Guerrero-Martínez, A. Recent progress on colloidal metal nanoparticles as signal enhancers in nanosensing. Adv. Colloid Interface Sci. 2015, in press. [Google Scholar]
- Kiefer, W.; Schlücker, S. Surface Enhanced Raman Spectroscopy: Analytical, Biophysical and Life Science Applications; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Hicks, C.J. Surface Enhanced Raman Spectroscopy (SERS). Available online: http://www.cem.msu.edu/~cem924sg/ChristineHicks.pdf (accessed on 19 May 2016).
- Zong, C.; Chen, C.-J.; Zhang, M.; Wu, D.-Y.; Ren, B. Transient electrochemical surface-enhanced raman spectroscopy (tec-sers): A millisecond time-resolved study of an electrochemical redox process. J. Am. Chem. Soc. 2015, 137, 11768–11774. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Li, H.; Liusman, C.; Yin, Z.; Wu, S.; Zhang, H. Surface enhanced raman scattering of Ag or Au nanoparticle-decorated reduced graphene oxide for detection of aromatic molecules. Chem. Sci. 2011, 2, 1817–1821. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced raman scattering (SERS). Phys. Rev. Lett. 1997. [Google Scholar] [CrossRef]
- Nie, S.; Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced raman scattering. Science 1997, 275, 1102–1106. [Google Scholar] [CrossRef] [PubMed]
- Le Ru, E.C.; Etchegoin, P.G. Single-molecule surface-enhanced raman spectroscopy. Ann. Rev. Phys. Chem. 2012, 63, 65–87. [Google Scholar] [CrossRef] [PubMed]
- Sonntag, M.D.; Klingsporn, J.M.; Zrimsek, A.B.; Sharma, B.; Ruvuna, L.K.; van Duyne, R.P. Molecular plasmonics for nanoscale spectroscopy. Chem. Soc. Rev. 2014, 43, 1230–1247. [Google Scholar] [CrossRef] [PubMed]
- Ling, X.; Xie, L.; Fang, Y.; Xu, H.; Zhang, H.; Kong, J.; Dresselhaus, M.S.; Zhang, J.; Liu, Z. Can graphene be used as a substrate for raman enhancement? Nano Lett. 2009, 10, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Mao, N.; Zhang, J. Graphene: A platform for surface-enhanced raman spectroscopy. Small 2013, 9, 1206–1224. [Google Scholar] [CrossRef] [PubMed]
- Jasuja, K.; Berry, V. Implantation and growth of dendritic gold nanostructures on graphene derivatives: Electrical property tailoring and raman enhancement. Acs Nano 2009, 3, 2358–2366. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Rong, J.; Cui, J.; Yang, Y.; Yang, L.; Wang, Y.; Liu, Y. Fabrication of a graphene oxide–gold nanorod hybrid material by electrostatic self-assembly for surface-enhanced raman scattering. Carbon 2013, 51, 255–264. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, C.; Zhang, Z. Graphene oxide embedded sandwich nanostructures for enhanced raman readout and their applications in pesticide monitoring. Nanoscale 2013, 5, 3773–3779. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.U.; Lee, W.; Yoon, S.S.; Kim, J.; Byun, J.H. Site-selective immobilization of gold nanoparticles on graphene sheets and its electrochemical properties. Appl. Surf. Sci. 2014, 315, 73–80. [Google Scholar] [CrossRef]
- Qiu, C.; Zhou, H.; Cao, B.; Sun, L.; Yu, T. Raman spectroscopy of morphology-controlled deposition of Au on graphene. Carbon 2013, 59, 487–494. [Google Scholar] [CrossRef]
- Zhu, J.; Gao, J.; Li, J.-J.; Zhao, J.-W. Improve the surface-enhanced raman scattering from rhodamine 6g adsorbed gold nanostars with vimineous branches. Appl. Surf. Sci. 2014, 322, 136–142. [Google Scholar] [CrossRef]
- Wang, Y.; Ni, Z.; Hu, H.; Hao, Y.; Wong, C.P.; Yu, T.; Thong, J.T.; Shen, Z.X. Gold on graphene as a substrate for surface enhanced raman scattering study. Appl. Phys. Lett. 2010, 97, 163111. [Google Scholar] [CrossRef]
- Xu, W.; Xiao, J.; Chen, Y.; Chen, Y.; Ling, X.; Zhang, J. Graphene-veiled gold substrate for surface-enhanced raman spectroscopy. Adv. Mater. 2013, 25, 928–933. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Shim, S.; Kim, B.; Shin, H.S. Surface-enhanced raman scattering of single- and few-layer graphene by the deposition of gold nanoparticles. Chem. A Eur. J. 2011, 17, 2381–2387. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-J.; An, H.-Q.; Zhu, J.; Zhao, J.-W. Improve the surface enhanced raman scattering of gold nanorods decorated graphene oxide: The effect of ctab on the electronic transition. Appl. Surf. Sci. 2015, 347, 856–860. [Google Scholar] [CrossRef]
- Biroju, R.K.; Giri, P. Defect enhanced efficient physical functionalization of graphene with gold nanoparticles probed by resonance raman spectroscopy. J. Phys. Chem. C 2014, 118, 13833–13843. [Google Scholar] [CrossRef]
- Nguyen, T.H.; Zhang, Z.; Mustapha, A.; Li, H.; Lin, M. Use of graphene and gold nanorods as substrates for the detection of pesticides by surface enhanced raman spectroscopy. J. Agric. Food Chem. 2014, 62, 10445–10451. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Kanchanapally, R.; Ray, P.C. Hybrid graphene oxide based ultrasensitive sers probe for label-free biosensing. J. Phys. Chem. Lett. 2013, 4, 3813–3818. [Google Scholar] [CrossRef]
- Das, G.; Patra, N.; Gopalakrishnan, A.; Zaccaria, R.P.; Toma, A.; Thorat, S.; Di Fabrizio, E.; Diaspro, A.; Salerno, M. Fabrication of large-area ordered and reproducible nanostructures for sers biosensor application. Analyst 2012, 137, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, J.; Suri, S.; Sooter, L.J.; Ma, D.; Wu, N. Detection of adenosine triphosphate with an aptamer biosensor based on surface-enhanced raman scattering. Anal. Chem. 2012, 84, 2837–2842. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Liu, K.-K.; Su, S.; Yan, J.; Mao, X.; Wang, D.; He, Y.; Li, L.-J.; Song, S.; Fan, C. Graphene-based high-efficiency surface-enhanced raman scattering-active platform for sensitive and multiplex DNA detection. Anal. Chem. 2012, 84, 4622–4627. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, M.; Abdelhamid, H.N.; Talib, A.; Wu, H.-F. Facile synthesis of gold nanohexagons on graphene templates in raman spectroscopy for biosensing cancer and cancer stem cells. Biosens. Bioelectron. 2014, 55, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Hakonen, A.; Andersson, P.O.; Schmidt, M.S.; Rindzevicius, T.; Käll, M. Explosive and chemical threat detection by surface-enhanced raman scattering: A review. Anal. Chim. Acta 2015, 893, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Kong, L.; Wang, J.; Fang, F.; Li, D.; Liu, J. Highly sensitive sers detection of Hg2+ ions in aqueous media using gold nanoparticles/graphene heterojunctions. ACS Appl. Mater. Interfaces 2013, 5, 7072–7078. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Atar, N.; Yola, M.L.; Eryılmaz, M.; Torul, H.; Tamer, U.; Boyacı, İ.H.; Üstündağ, Z. A novel glucose biosensor platform based on Ag@AuNPs modified graphene oxide nanocomposite and sers application. J. Colloid Interface Sci. 2013, 406, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Kanchanapally, R.; Sinha, S.S.; Fan, Z.; Dubey, M.; Zakar, E.; Ray, P.C. Graphene oxide–gold nanocage hybrid platform for trace level identification of nitro explosives using a raman fingerprint. J. Phys. Chem. C 2014, 118, 7070–7075. [Google Scholar] [CrossRef]
- Kong, X.-K.; Chen, Q.-W.; Sun, Z.-Y. Enhanced sers of the complex substrate using Au supported on graphene with pyridine and r6g as the probe molecules. Chem. Phys. Lett. 2013, 564, 54–59. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, D.; Zhang, L.; Fang, Y. The sers study of graphene deposited by gold nanoparticles with 785 nm excitation. Chem. Phys. Lett. 2013, 556, 146–150. [Google Scholar] [CrossRef]
- Jalani, G.; Cerruti, M. Nano graphene oxide-wrapped gold nanostars as ultrasensitive and stable sers nanoprobes. Nanoscale 2015, 7, 9990–9997. [Google Scholar] [CrossRef] [PubMed]
- Benítez-Martínez, S.; López-Lorente, Á.I.; Valcárcel, M. Multilayer graphene–gold nanoparticle hybrid substrate for the sers determination of metronidazole. Microchem. J. 2015, 121, 6–13. [Google Scholar] [CrossRef]
- Lu, R.; Konzelmann, A.; Xu, F.; Gong, Y.; Liu, J.; Liu, Q.; Xin, M.; Hui, R.; Wu, J.Z. High sensitivity surface enhanced raman spectroscopy of r6g on in situ fabricated Au nanoparticle/graphene plasmonic substrates. Carbon 2015, 86, 78–85. [Google Scholar] [CrossRef]
- Liu, A.; Xu, T.; Tang, J.; Wu, H.; Zhao, T.; Tang, W. Sandwich-structured Ag/graphene/Au hybrid for surface-enhanced raman scattering. Electrochim. Acta 2014, 119, 43–48. [Google Scholar] [CrossRef]
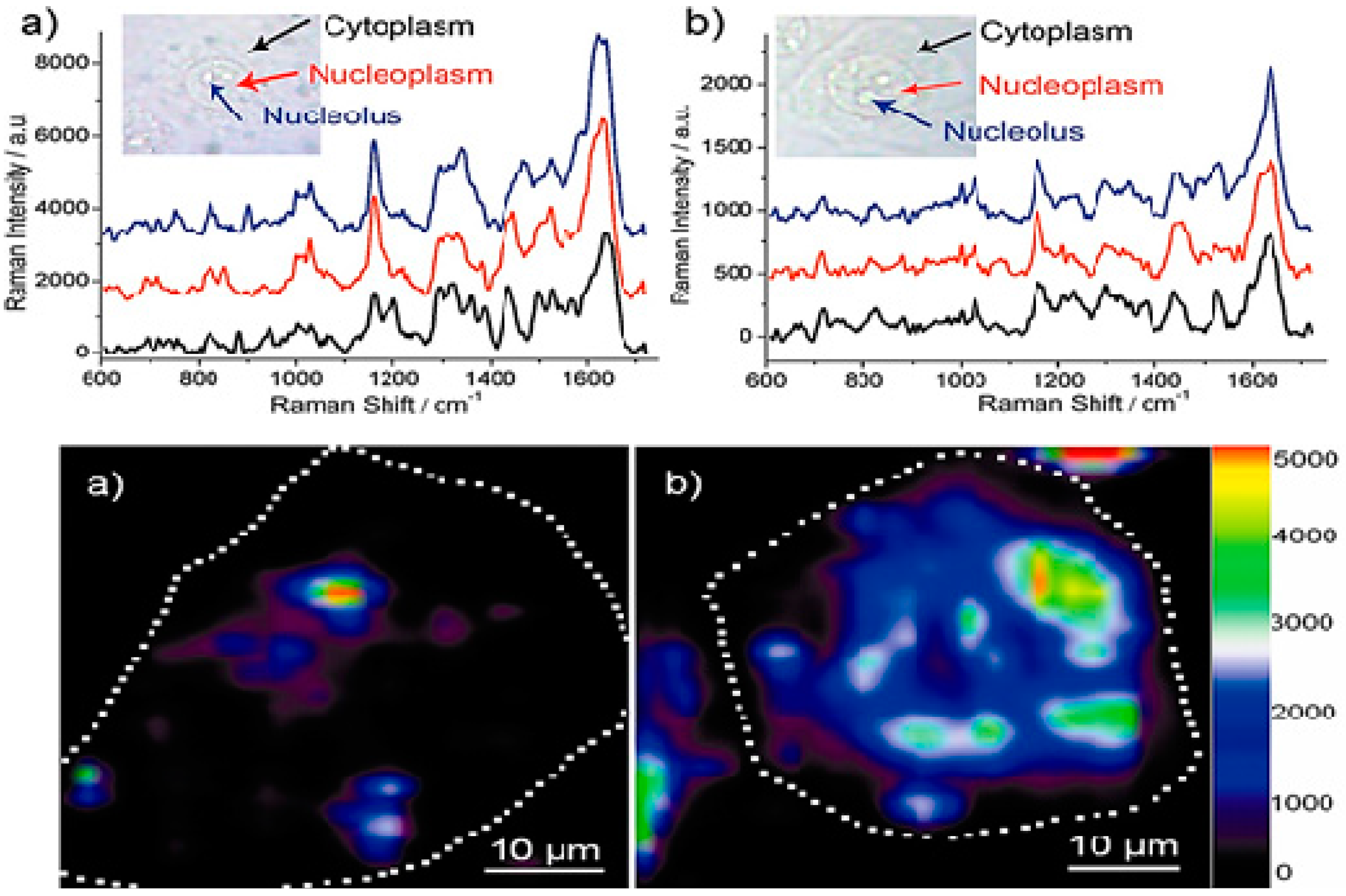
- Liu, Q.; Wei, L.; Wang, J.; Peng, F.; Luo, D.; Cui, R.; Niu, Y.; Qin, X.; Liu, Y.; Sun, H. Cell imaging by graphene oxide based on surface enhanced raman scattering. Nanoscale 2012, 4, 7084–7089. [Google Scholar] [CrossRef] [PubMed]
- Nergiz, S.Z.; Gandra, N.; Tadepalli, S.; Singamaneni, S. Multifunctional hybrid nanopatches of graphene oxide and gold nanostars for ultraefficient photothermal cancer therapy. ACS Appl. Mater. Interfaces 2014, 6, 16395–16402. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Hu, C.; Li, S.; Zhang, W.; Guo, Z. Rapid intracellular growth of gold nanostructures assisted by functionalized graphene oxide and its application for surface-enhanced raman spectroscopy. Anal. chem. 2012, 84, 10338–10344. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Xu, T.; Ren, Q.; Yuan, M.; Dong, W.; Tang, W. Graphene modulated 2D assembly of plasmonic gold nanostructure on diamond-like carbon substrate for surface-enhanced raman scattering. Electrochem. Commun. 2012, 25, 74–78. [Google Scholar] [CrossRef]
- Huang, J.; Zong, C.; Shen, H.; Liu, M.; Chen, B.; Ren, B.; Zhang, Z. Mechanism of cellular uptake of graphene oxide studied by surface-enhanced raman spectroscopy. Small 2012, 8, 2577–2584. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Polavarapu, L.; Liz-Marzán, L.M. Reduced graphene oxide-supported gold nanostars for improved sers sensing and drug delivery. ACS Appl. Mater. Interfaces 2014, 6, 21798–21805. [Google Scholar] [CrossRef] [PubMed]
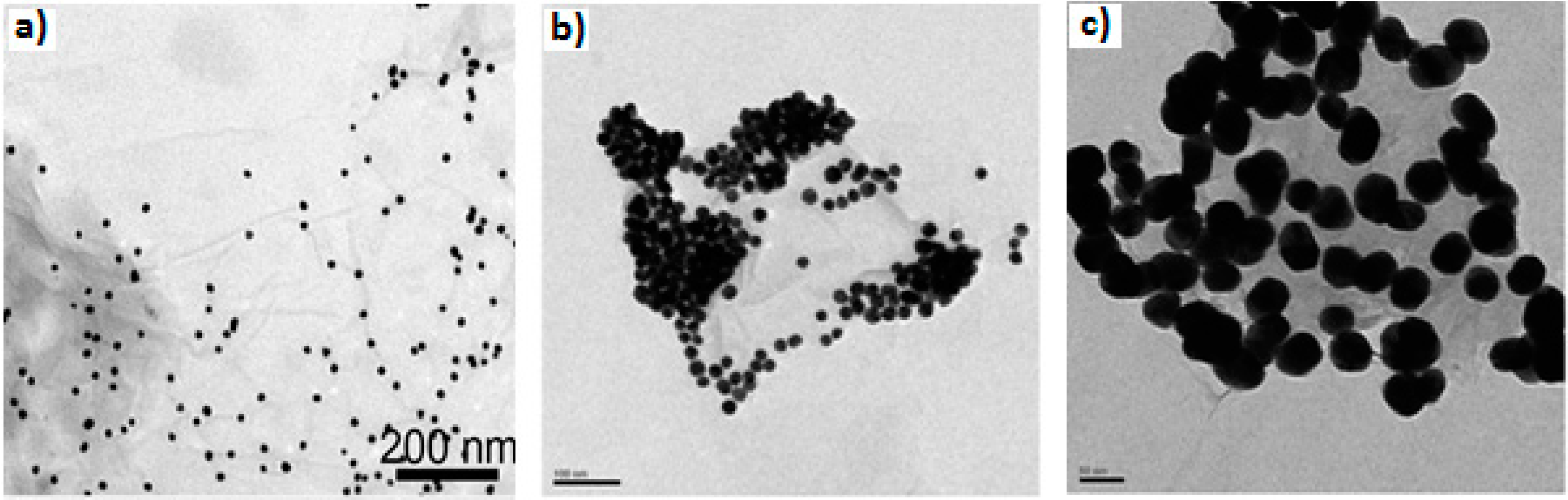

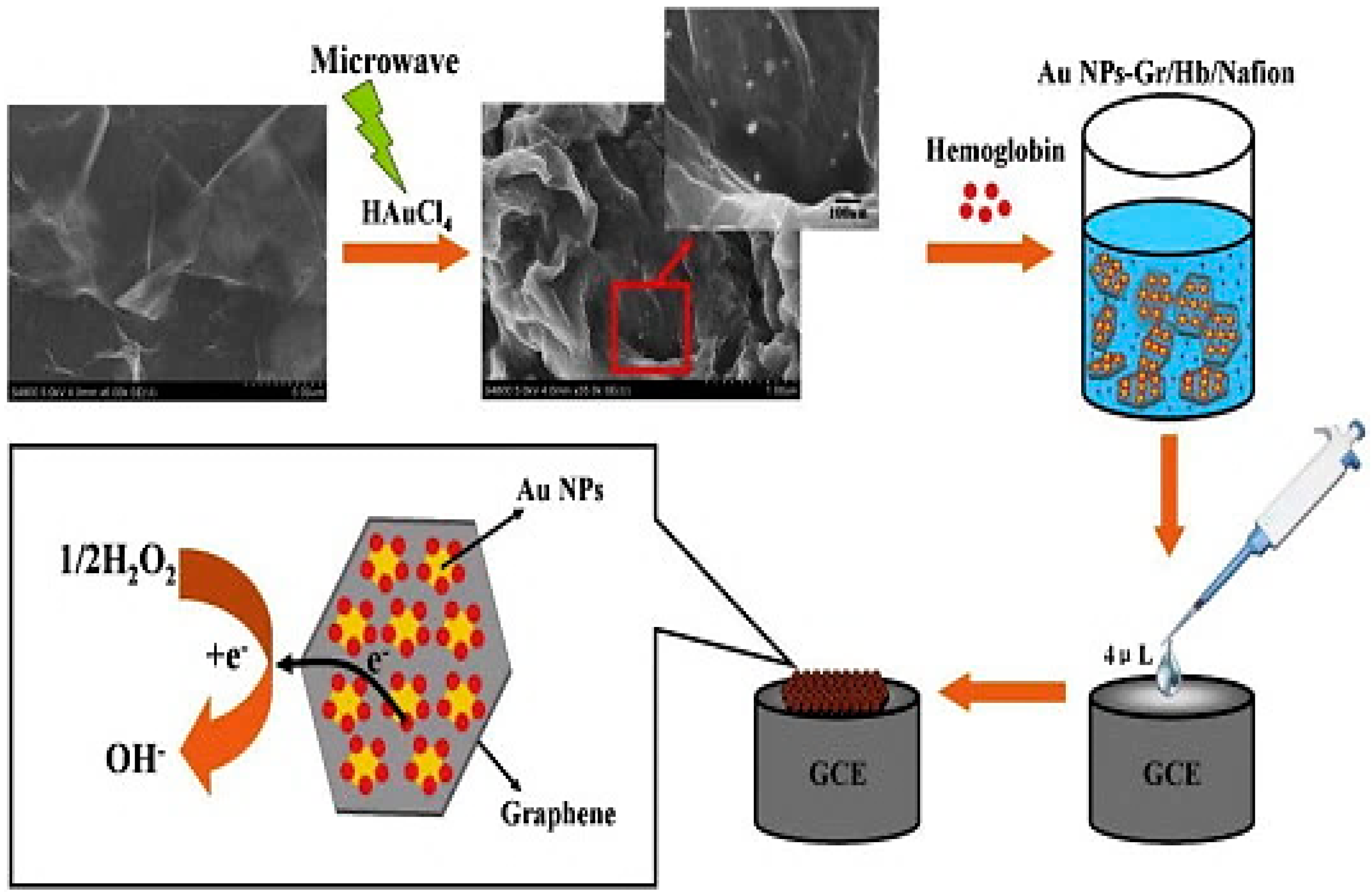
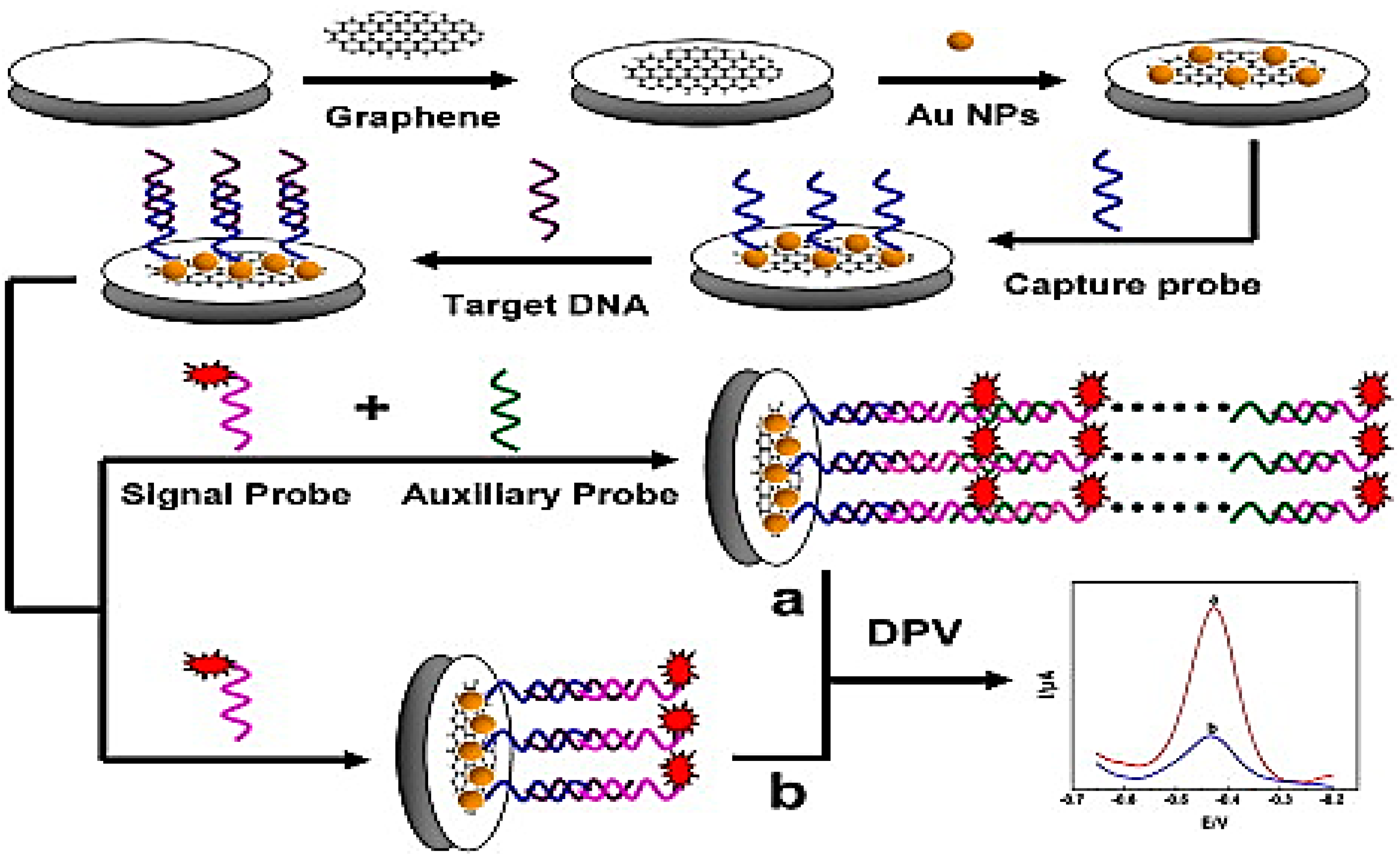
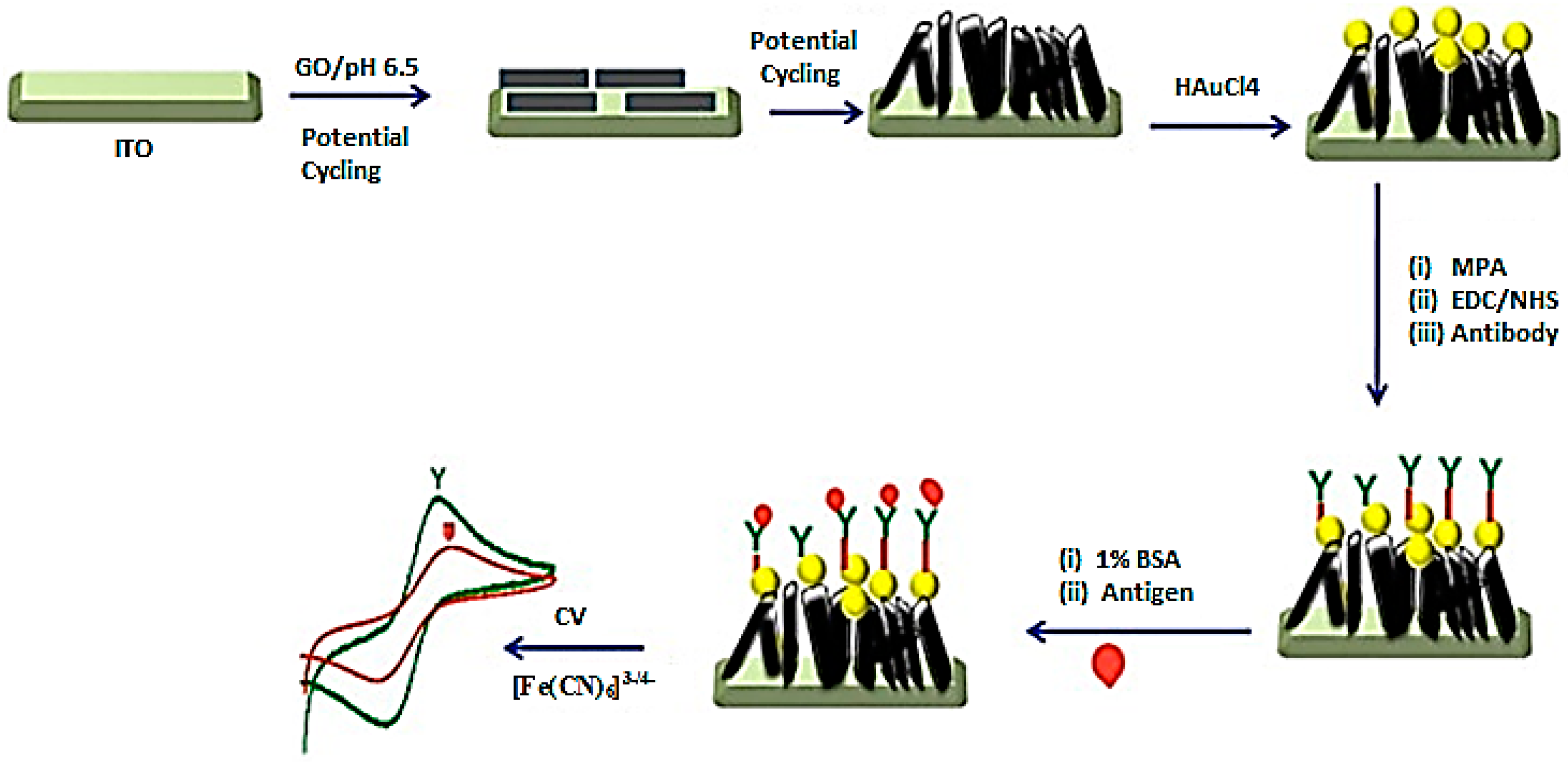
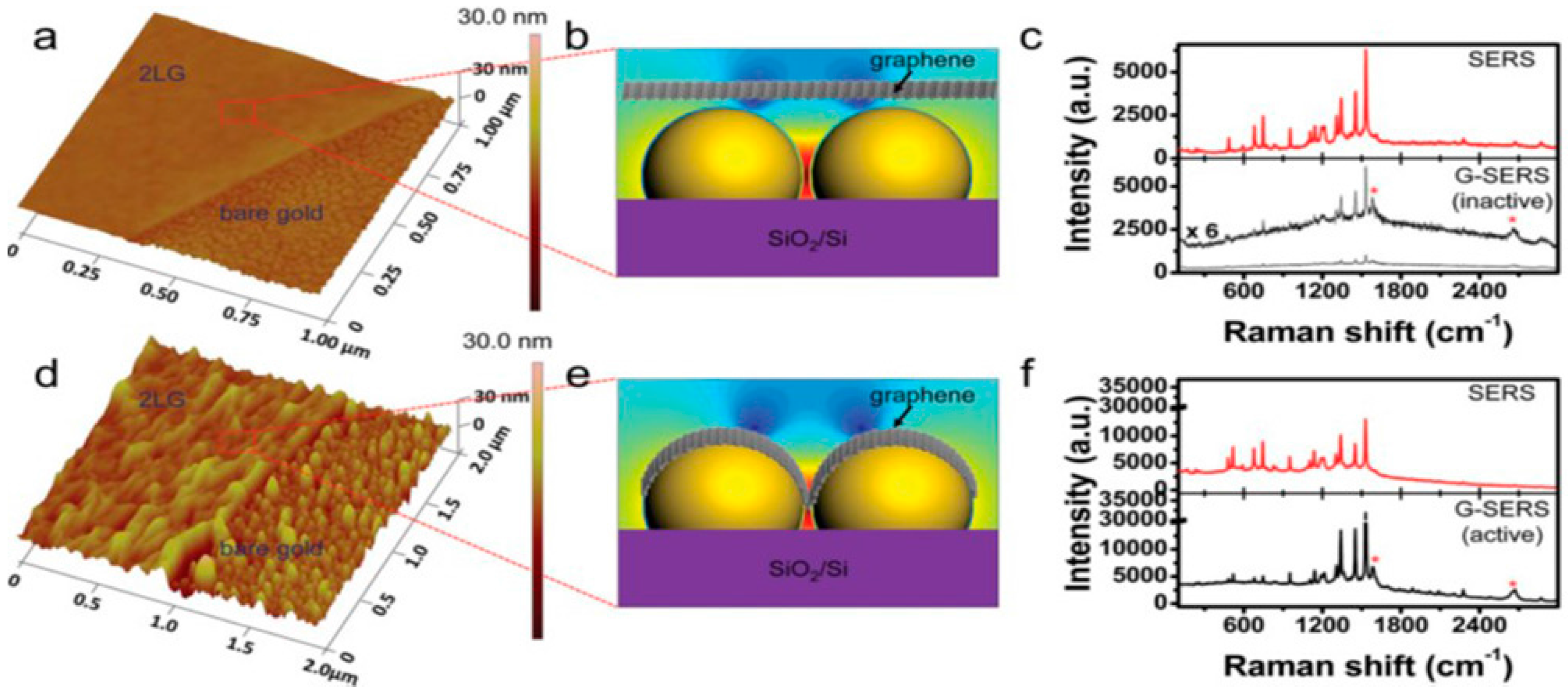
| Properties | Value | References |
|---|---|---|
| Optical transmittance | ~97.7% | [6] |
| Density | 0.77 mg·m−2 | [10,11] |
| Career density | 1012 cm−2 | [10,11] |
| Resistivity | 10−6 Ω·cm | [10,11] |
| Planar surface area | 2630 m2·g−1 | [12] |
| Mechanical strength of its Young Modulus | 1100 GPa | [13] |
| Fracture strength | 125 GPa | [13] |
| Thermal conductivity | ~5000 W·m−1·K−1 | [14] |
| Mobility charge carrier | 200,000 cm2·V−1·s−1 | [15] |
| Synthesis Procedure | Beneficial Aspects | Limitations | References |
|---|---|---|---|
| Micromechanical exfoliation |
|
| [49,75,76] |
| CVD |
|
| [49,53,77,78] |
| Epitaxial growth |
|
| [49] |
| Colloidal suspension |
|
| [79] |
| Unzipping of CNTs |
|
| [49,60] |
| Reduction of GO |
|
| [76,79,80] |
| Synthesis Techniques | Advantages | Limitations | References |
|---|---|---|---|
| In situ reduction |
|
| [25,83,84] |
| Ex situ |
|
| [76,82,84] |
| Hydro-thermal |
|
| [85,86,87] |
| Electro-chemical |
|
| [41,88,89,90,91,92] |
| Name of the Synthesis Process | Name of the Final Graphene–Gold Hybrid | Gold—Functionalizing Agent (FA)/Stabilizing Agent (SA)/Reducing Agent (RA) | Graphene—Functionalizing Agent (FA)/Stabilizing Agent (SA)/Reducing Agent (RA)/Reduction Process (RP) | References |
|---|---|---|---|---|
| Seeded-growth simultaneous reduction | rGO-AuNPs | Sodium citrate (FA), NaBH4 (RA) | GO–rGO via redox chemistry of GO & Au Precursors (RP) | [111] |
| Sequential reduction method | N2 doped graphene-AuNPs | Ethylene glycol (RA) | Hydrazine hydrate & NH3 (RA) | [112] |
| Chemical reduction in micro flow reactor | GO-AuNPs | Dimethylamina borane (RA) | No agent | [113] |
| Eco-friendly chemical reduction method | rGO-AuNPs | Rose water (RA) | Rose water (RA) | [114] |
| Reductive deposition process | rGO-AuNPs | No agent | Hydrazine and NH3 | [93] |
| Chemical reduction | Graphene-AuNPs | NaBH4 (RA) in presence of GO | Hydrazine hydrate (RA); SDS as a protector and disperser | [94] |
| Solution-based chemical reduction | Graphene-AuNPs | NaBH4 (RA) | ODA (FA) | [95] |
| Green synthesis method | GO-AuNPs | Tannic acid as RA and immobilizing agent | Tannic Acid (FA) | [97] |
| Electrostatic self- assembly | Graphene-AuNPs | NaBH4 (RA) | 1-pyrene butyric acid (FA) | [96] |
| Seed-assisted reduction method | rGO-AuNPs | NaBH4 (RA) &Trisodium citrate (SA) | Pyrene ethylene glycol amine or decyl pyrene (FA) | [69] |
| Wet impregnation thermal reduction method | Graphene-AuNPs | A flow of H2/Ar (RA) | Hydrazine hydrate & NH3 (RA); Sulphur (FA) | [98] |
| Reduction via amidation reaction | GO-AuNPs | 4 amino-thiophenol (FA) | Thionyl chloride (FA) | [83] |
| Chemical Reduction | Graphene-AuNPs | Sodium citrate (RA) | 1050 °C for 30 s in furnace (RP); Hydrazine hydrate (RA) | [115] |
| Chemical reduction | GO-AuNPs | Sodium citrate (RA) | - | [81] |
| Green dual reduction method | rGO-AuNP | Ascorbic acid (RA) | Ascorbic acid (RA) Polyvinylpyrrolidone (SA) | [116] |
| One-pot green synthesis | GO-AuNPs | Tyrosine (RA) | No agent | [117] |
| Wet impregnation–thermal reduction method | GNs-AuNPs | Flow of H2/Ar (10% H2) by ramping temp. From room temp. to 350 °C (10 °C/min) and holding at 350 °C for 3 h | GO–GNs by Hydrazine hydrate and NH3 | [98] |
| Microwave reduction | GO-AuNPs | Microwave exposure (1.05 kW, 2450 MHz) | No agent | [101] |
| Microwave-assisted simultaneous reduction | Graphene-AuNPs | Microwave exposure (0.8 kw) at 80 °C for 5 min under vigorous stirring | Hydrazine hydrate (RA) | [100] |
| Microwave irradiation—simultaneous reduction | Graphene-AuNPs | Microwave irradiation for 5 min | Ascorbic acid (RA) | [102] |
| Microwave-assisted simultaneous reduction | Graphene-AuNPs | Microwave exposure (0.2 kw) for 2 min | Polyethyleneimine (FA) | [103] |
| Sonolytic simultaneous and sequential reduction | Graphene-AuNPs | Ultrasonic frequency of 211 kHz | No agent | [105] |
| Sonochemical reduction | Graphene-AuNPs | Ultrasound irradiation | No agent | [104] |
| One-pot one step photochemical method | GO-AuNRs | UV-irradiation (256 nm, 30 W) for 25 min in a quartz tube | No agent | [106] |
| Photochemical reduction | Graphene-AuNDs | Photochemistry (RA) in presence of octadecanethiol | No agent | [107] |
| Photochemical reduction | Graphene-AuNS | Photo (LED) irradiation | No agent | [108] |
| Photo-assisted chemical reduction | Graphene-AuNPs | Laser light in presence of AuCl4− electrolyte | No agent | [109] |
| Light-induced covalent interactions | rGO-AuNPs | 3-aryl-3-(trifluoromethyl) diazirine (FA) | GO to rGO by high temp. (1050 °C for 30 s) reduction in an argon flow environment (RP) | [23] |
| Self-catalysis reduction | rGO-AuNPs | NaBH4 (RA) CTAB (SA) | NaBH4 (RA), GO–AuNPs (catalyst) | [84] |
| Self-assembly/Noncovalent attachment | rGO-AuNPs & GO-AuNPs | 2-mercaptopyridine (FA), Trisodium citrate (SA/RA) | - | [82] |
| Thermal reduction of GO/electrostatic attractions | rGO-AuNPs | - | GO to rGO by thermal (200 °C) reduction in an argon flow environment | [96] |
| LBL self-assembly/electrostatic interactions | Graphene-AuNPs | Trisodium citrate (SA) | BSA (RA & SA) | [118] |
| - | Graphene-aerogel (GA)@AuNPs/AuNPs | Citric Acid (RA) | Ascorbic acid (RA) for GO to GA; freezing drying and thermal annealing at 180 °C for 6 h for final products | [119] |
| Electrode | Composition of Electrolytic Solution | Applied Electrochemical Potential and Reaction Condition | References |
|---|---|---|---|
| AuNPs/rGO/GCE | 10 mM AuCl3, Nafion (0.5%), and 0.1 M H2SO4 | −1.0 V for 500 s. | [41] |
| Au film/graphene–Au nanocomposite/GCE | 0.1 mM HAuCl4 | −1.2 V for 50 s for graphene; −0.25 V for 50 s for Au electrodeposition. Run the process using alternate graphene and AuNPs for 3 cycles. | [89] |
| DHB/AuNPs/rGO/GCE | 0.3 mM HAuCl4 | −0.2 V for 24 h at room temp. | [90] |
| Graphene/nano-Au/GCE | 0.1 M Kn and 5 mM HAuCl4 | −0.2 to 1.0 V for 2 cycles at scan rate of 50 mV∙s−1. | [91] |
| AuNP/electro reduced graphene (eGr)/Indium titanium oxide (ITO) | 0.5 mM HAuCl4.nH2O in phosphate buffer | 0 to −1.6 V continuously for 75 cycles at a scan rate 50 mV·s−1 to electrodeposit eGr on ITO and 25 cycles for AuNPs electrodeposition. | [120] |
| AuNPs/2,5-di-(2-thienyl)-1-pyrrole-1-(p-benzoic acid) (DPB)/graphene/Au electrode | 3 mM of HAuCl4 containing 0.5 M H2SO4 | −1.2 V for 200 s for electrochemical reduction and deposition of GO on Au electrode; −0.25 V for 25 s at 10 °C for the electrodeposition of AuNPs. | [121] |
| AuNPs/graphene-nanofibers/GCE | 25 mM of HAuCl4 containing 0.1 M Na2SO4 solution | −0.4 V for 300 s for the electrochemical deposition of AuNPs. | [122] |
| AuNPs/graphene/GCE | mM HAuCl4 solution containing 0.5 M H2SO4 | −1.2 V for 1200 s for the electrochemical reduction of GO on the electrode surface; −0.25 V for 30 s for the electrodeposition of AuNPs. | [123] |
| Graphene/nano-Au/GCE | 0.1 M phosphate buffer (pH 7.0) containing 6.5 mM HAuCl4 | 0 to −2 V at a scan rate of 100 mV·s−1 for continuous cyclic voltammetric sweep of 40 cycles. | [124] |
| AuNPs/GO/GCE | HAuCl4 solution | Electrodeposition of AuNPs by pulse voltammetry with a pulse width of 0.1 s, potential 1.1 and −0.2 V, respectively. | [125] |
| AuNPs/ERGO/carbon ionic liquid electrode (CLIE) | 5.0 mM HAuCl4 solution | −1.3 V for 300 s to from a stable ERGO on the surface of CILE; −0.4 V for 300 s for electrodeposition of AuNPs on ERGO/CILE. | [126] |
| Composition of the Sensors | Detected Analyte | Linear Range of Detection | LOD | References |
|---|---|---|---|---|
| GOD/rGO–AuNPs/GCE | Glucose | 1–8 mM | 10 μM | [114] |
| Graphene/nano–Au/GOD/GCE | Glucose | 0.2–2 and 2–20 mM | 17 μM | [124] |
| Graphene/AuNPs/chitosan/GOD | Glucose | 2–10 mM | 180 μM | [159] |
| GOD/graphene–AuNPs/GCE | Glucose | 0.1–10 mM | 35 mM | [160] |
| Graphene Rod/AuNPs/GOD | Glucose | 0.1–10 mM | 83 μM | [162] |
| GCE–ATP–GNs–AuNPs–GOD | Glucose | 1–12 mM (voltammetry) | 9.3 μM | [98] |
| GCE–ATP–GNs–AuNPs–GOD | Glucose | 1–8 mM (EIS) | 4.1 μM | [98] |
| GOD/graphene–AuNPs | Glucose | 0.02–2.26 | 4.1 μM | [163] |
| GA@AuNPs/AuNPs | Glucose | 0.01–16 mM | 4.0 μM | [119] |
| Graphite/SrPdO3/AuNPs | Glucose | 0.1–6 mM | 10.1 μM | [164] |
| GOD–graphene/PANI/AuNPs/GCE | Glucose | 0.004–1.12 mM | 0.6 μM | [161] |
| Graphene/Au–NPs/GCE | H2O2 | 0.0005–0.5 mM | 0.44 μM | [165] |
| Nafion/Hb/AuNPs–graphene/GCE | H2O2 | 0.0001–0.07 mM | 0.03 μM | [87] |
| GCE–GO–AuNP–ssDNA | DNA | - | 100 fM | [125] |
| ssDNA/AuNPs–ATPGO/GCE | DNA | 1.0 × 10−13 to 1.0 × 10−9 M | 1.13 × 10−14 M | [173] |
| ssDNA/AuNPs/TB–GO/GCE | MDR gene (DNA) | 1.0 × 10−11 to 1.0 × 10−9 M | 2.95 × 10−12 M | [168] |
| Capture probe (cDNA)/AuNPs–rGO/GCE | DNA | 0.1 μM to 0.1 fM | 35 aM | [167] |
| Au film/graphene–Au nanocomposite/GCE | Peanut allergen Ara h1 gene | 10−16 to 10−13 M | 0.041 fM | [89] |
| DHB/AuNPs/rGO/GCE | levodopa (LD) | 0.05–1200.0 μM | 18 nM | [90] |
| Au NP/GO/GCEs | Ascorbic Acid | 0.11–0.6 mM | 100 nM | [81] |
| Graphene/AuNPs/GCE | DES | 1.20 × 10−8 to 1.20 × 10−5 M | 9.80 × 10−9 M | [91] |
| AuNPs/rGO/GCE | NADH in human urine | 50 nM to 500 μM | 1.13 nM | [41] |
| AuNPs/ERGO/CILE | Folic Acid | 0.01 μM to 50.0 μM | 2.7 nM | [126] |
| AuNPs/1-pyrene butyric acid-functionalized graphene/GCE | Uric acid | 2.6 × 10−6 to 6.2 × 10−5 M | 2.0 × 10−7 M | [96] |
| Graphene nanosheet–PEI/AuNPs/GCE | Dopamine | 2.0 to 48.0 μM | 0.2 μM | [103] |
| [AuNPs/rGO]20/GCE | Dopamine | 1.0 to 60.0 μM | 0.02 μM | [135] |
| Hb/AuNPs–graphene–SDS/BPG | Nitric oxide | 7.2 × 10−7 to 7.92 × 10−6 M | 1.2 × 10−8 M | [94] |
| Hb/AuNPs/GACS/GRE | Nitrite | 0.05 to 1000 μM | 0.01 μM | [172] |
| AuNPs/graphene nanofibers/GCE | Bisphenol A in baby bottle | 8.0 × 10−8 to 2.5 × 10−4 M | 3.5 × 10−8 M | [122] |
| Anti-BPA/MCH/AuNPs/graphene/GCE | Bisphenol A in milk sample | 0.01–10.0 μM | 5 nM | [123] |
| Aflatoxin B1 antibody-AuNPs/DPB/graphene/Au electrode | Aflatoxin B1 in spiked food | 3.2 fM–0.32 pM | 1 fM | [121] |
| AuNP/N2-doped graphene/GCE | Chloramphenicol | 2.0 × 10−6 to 8.0 × 10−5 M | 5.9 × 10−7 M | [112] |
| Anti-estradiol antibody-AuNP–eGr/ITO | 17 β-estradiol | 1 × 10−3 to 0.1 × 10−12 M | 0.1 fM | [120] |
| rGO–AuNPs-modified GCE | M. tuberculosis | 1.0 × 10−15 and 1.0 × 10−9 M | fM level | [169] |
| E. coli O157:H7 antibodies-AuNPs/rGOPE | E. coli O157:H7 | 1.5 × 102 to 1.5 × 107 cfu/mL | 1.5 × 102 cfu/mL | [170] |
| Au-graphene–AuNPs electrode | Carbamazepine | 5 × 10−6 to 10−2 M | 3.03 × 10−6 M | [110] |
| anti-CEA/AuNPs/Arg/rGO/CILE | CEA | 0.5 to 200 ng·mL−1 | 0.03 ng·mL−1 | [171] |
| AuNP–graphene/CILE | Hydroquinone | 0.06 μM to 800.0 μM | 0.018 μM | [100] |
| Name of the Hybrid Substrate | SERS—Order of Magnitude | Compared Material | References |
|---|---|---|---|
| rGO–AuNPs | 100 | Pure AuNPs | [42] |
| Graphene–AuNS | 45 | Graphene | [43] |
| Graphene–AgNS | 150 | Graphene | [43] |
| AuNPs/graphene/SiO2/Si | 120 | Graphene/SiO2/Si | [199] |
| Graphene–AuNPs | 10–100 | AuNPs | [212] |
| Graphene–AuNPs | 77.6 | Graphene | [213] |
| Pyrene ethylene glycol amine-functionalized rGO/AuNRs | 14.7 | Bare rGO | [69] |
| R6G/GO–AuNR with CTAB | 10 | Pure AuNRs | [200] |
| Nano GO (nGO)–Au nanostars | 5.3 | nGO | [214] |
| Graphene–AuNPs | 3.3 | AuNPs | [215] |
| R6G/AuNP/graphene/SiO2/Si | 86 | Graphene/SiO2/Si with R6G | [216] |
| Ag/rGO/Au for rhodamine B (RhB) | 8.8 | Pristine Ag dendrites | [217] |
| AuNPs/rGO/SiO2/Si | 40 | Blank substrate | [209] |
| GO–AuNPs | ~4 | GO | [218] |
| Neural Stem Cells on GO encapsulated AuNPs | 3.5 | AuNPs | [139] |
| GO–AuNS | 3 | GO | [219] |
| GO/PVP/intracellularly grown AuNPs (IGAuNPs) | 5 | IGAuNPs | [220] |
| Carbaryl on graphene–Au film–AuNR | 2 | Au film–AuNR | [202] |
| Carbaryl on graphene–Au film–AuNR | 100 | Graphene–AuNR | [202] |
| Si/N2 doped diamond-like carbon (DLC-N)/Au/rGO/Au for RhB | 860 | Si/DLC-N | [221] |
| GO-popcorn shaped AuNPs hybrid for R6G | 11 | GO | [203] |
| GO-Au nanocage for RDX | 4 | Au nanocage | [211] |
| Graphene-isolated AuNC (GIAN) nanostructures for R6G | More than 100 | R6G | [138] |
| Au@AgNPs/GO/Au@AgNPs sandwich for R6G | Enhancement factor of ~7.0 × 107 | - | [193] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, I.; Julkapli, N.M.; Yehye, W.A.; Basirun, W.J.; Bhargava, S.K. Graphene–Gold Nanoparticles Hybrid—Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor. Materials 2016, 9, 406. https://doi.org/10.3390/ma9060406
Khalil I, Julkapli NM, Yehye WA, Basirun WJ, Bhargava SK. Graphene–Gold Nanoparticles Hybrid—Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor. Materials. 2016; 9(6):406. https://doi.org/10.3390/ma9060406
Chicago/Turabian StyleKhalil, Ibrahim, Nurhidayatullaili Muhd Julkapli, Wageeh A. Yehye, Wan Jefrey Basirun, and Suresh K. Bhargava. 2016. "Graphene–Gold Nanoparticles Hybrid—Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor" Materials 9, no. 6: 406. https://doi.org/10.3390/ma9060406
APA StyleKhalil, I., Julkapli, N. M., Yehye, W. A., Basirun, W. J., & Bhargava, S. K. (2016). Graphene–Gold Nanoparticles Hybrid—Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor. Materials, 9(6), 406. https://doi.org/10.3390/ma9060406