One-Step Method to Prepare PLLA Porous Microspheres in a High-Voltage Electrostatic Anti-Solvent Process
Abstract
:1. Introduction
2. Results and Discussion
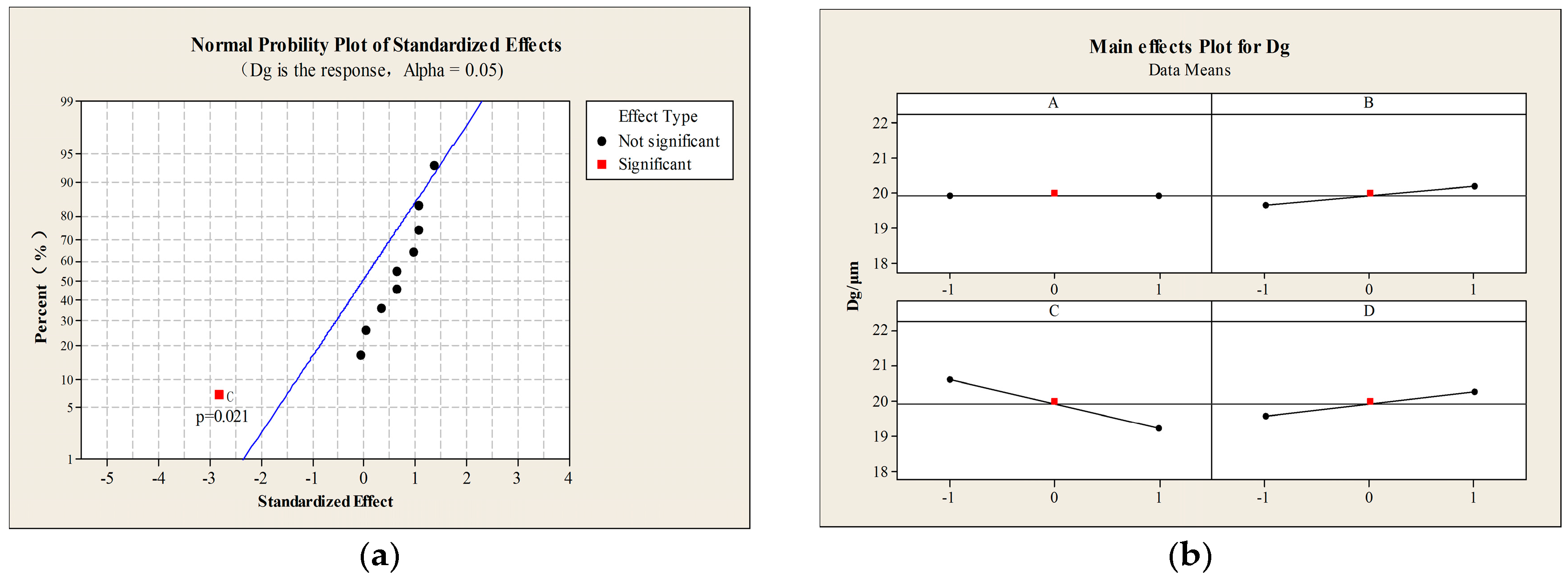
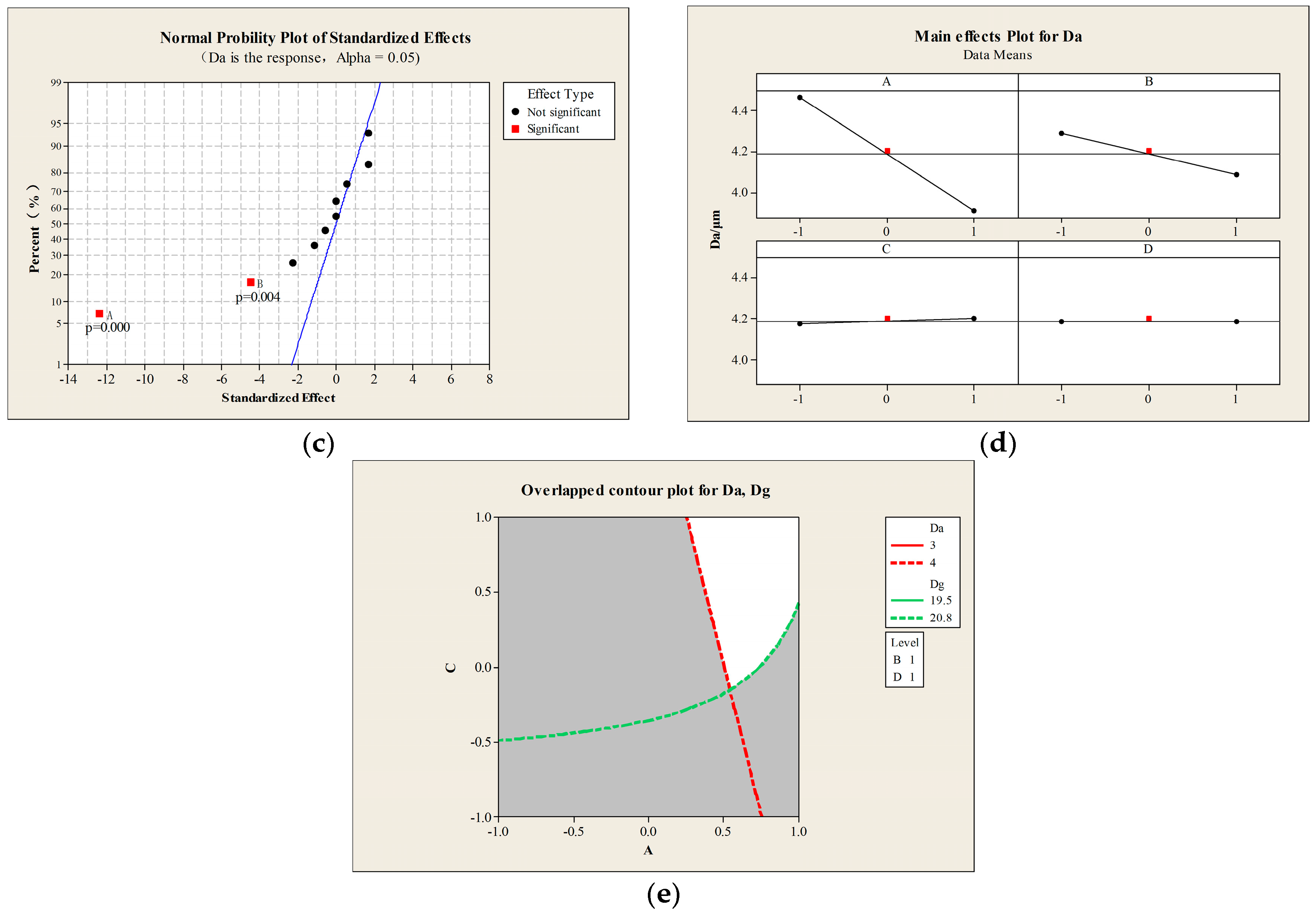
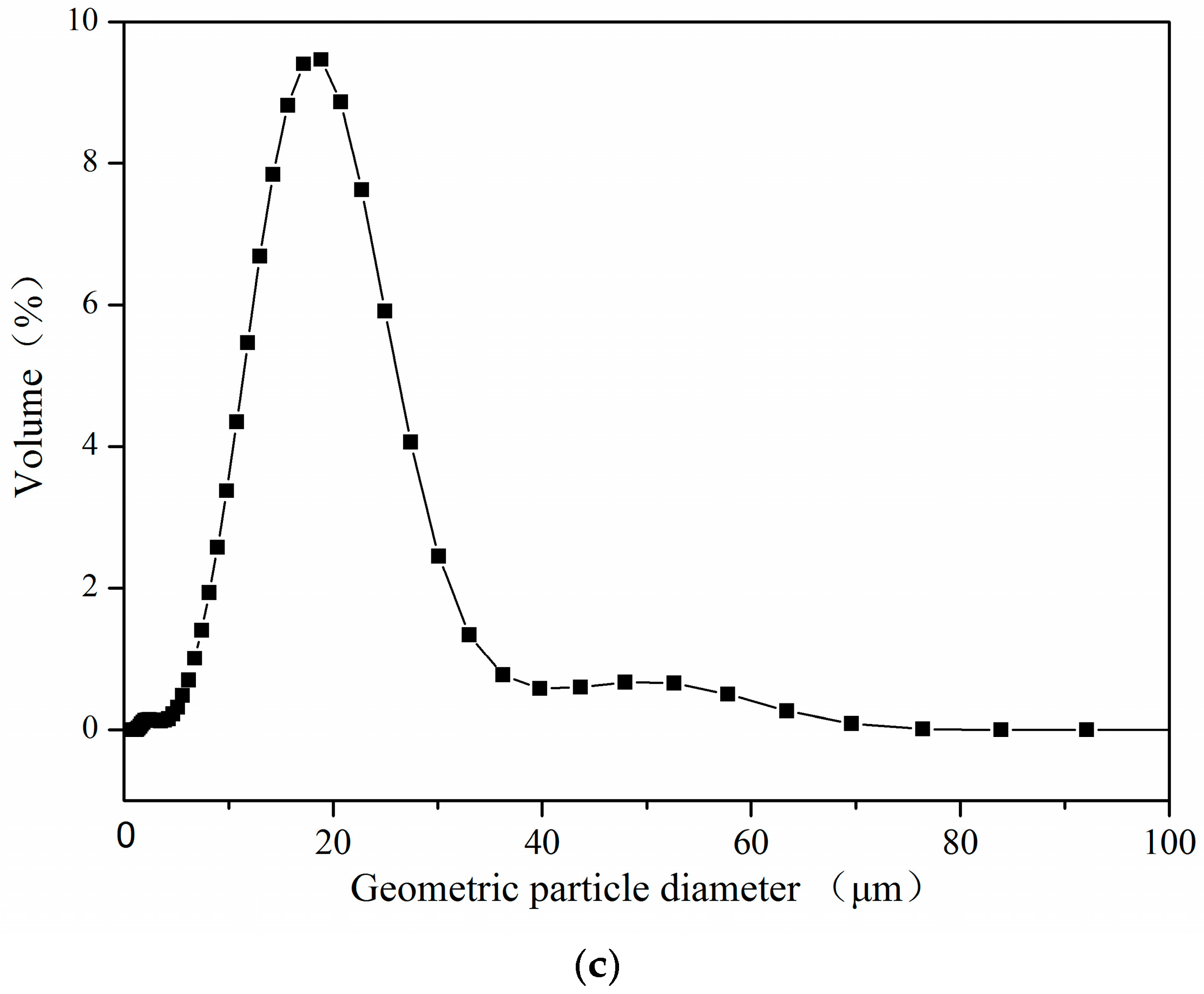
2.1. Optimization of the Process and Aerodynamic Properties of the PMs
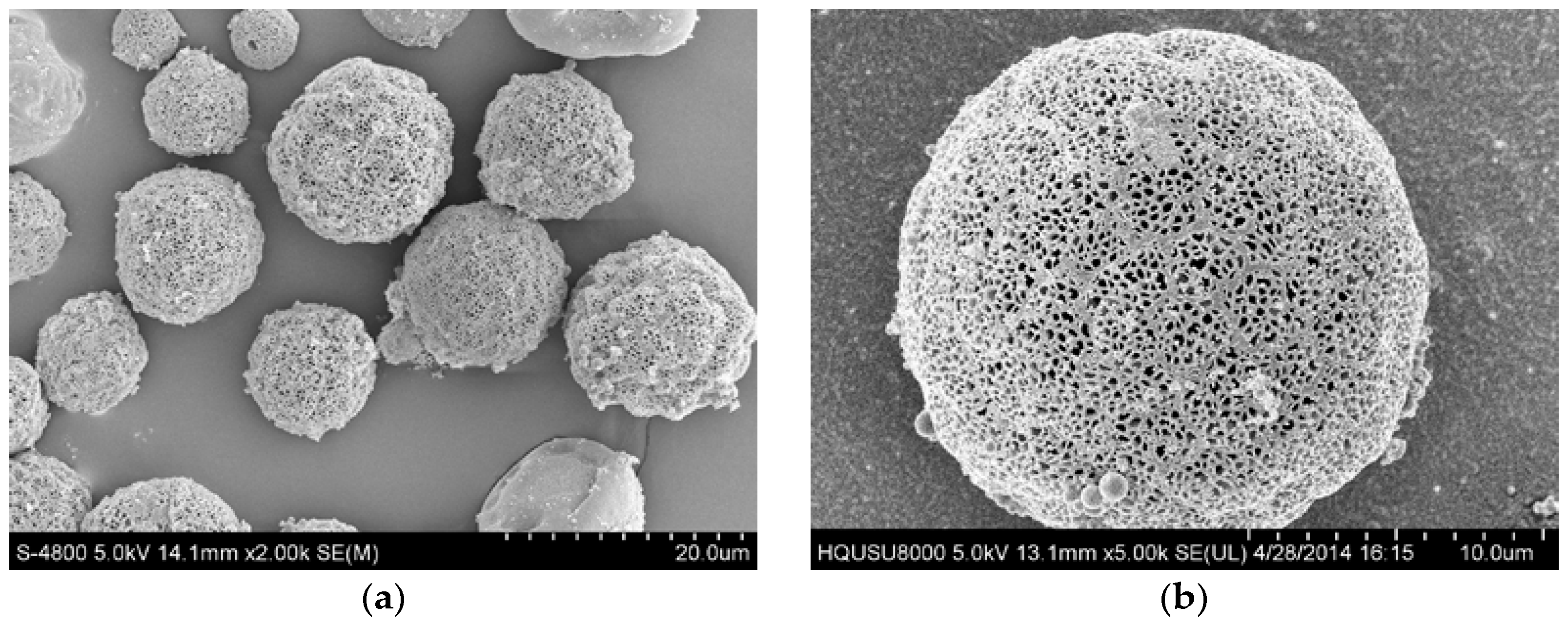
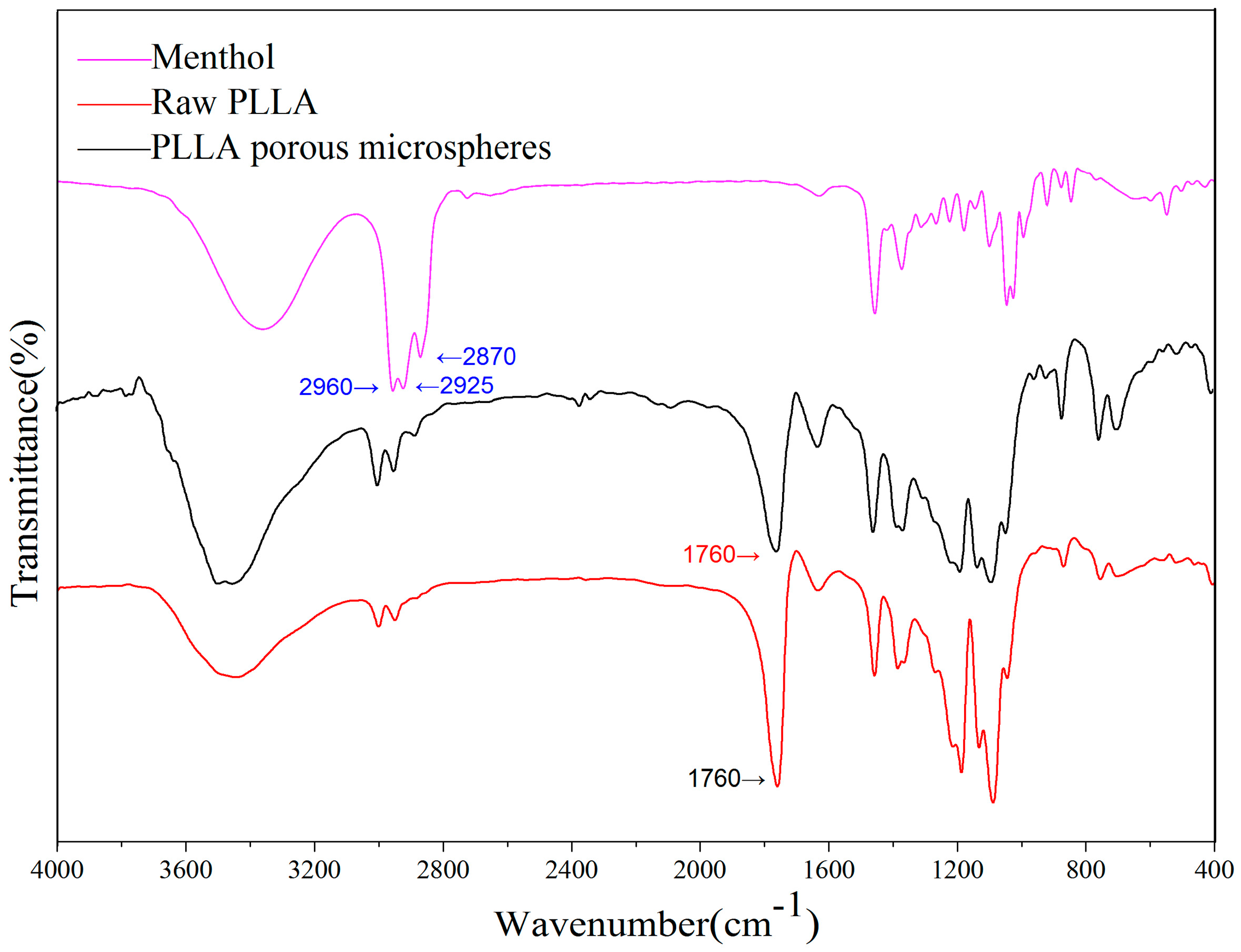
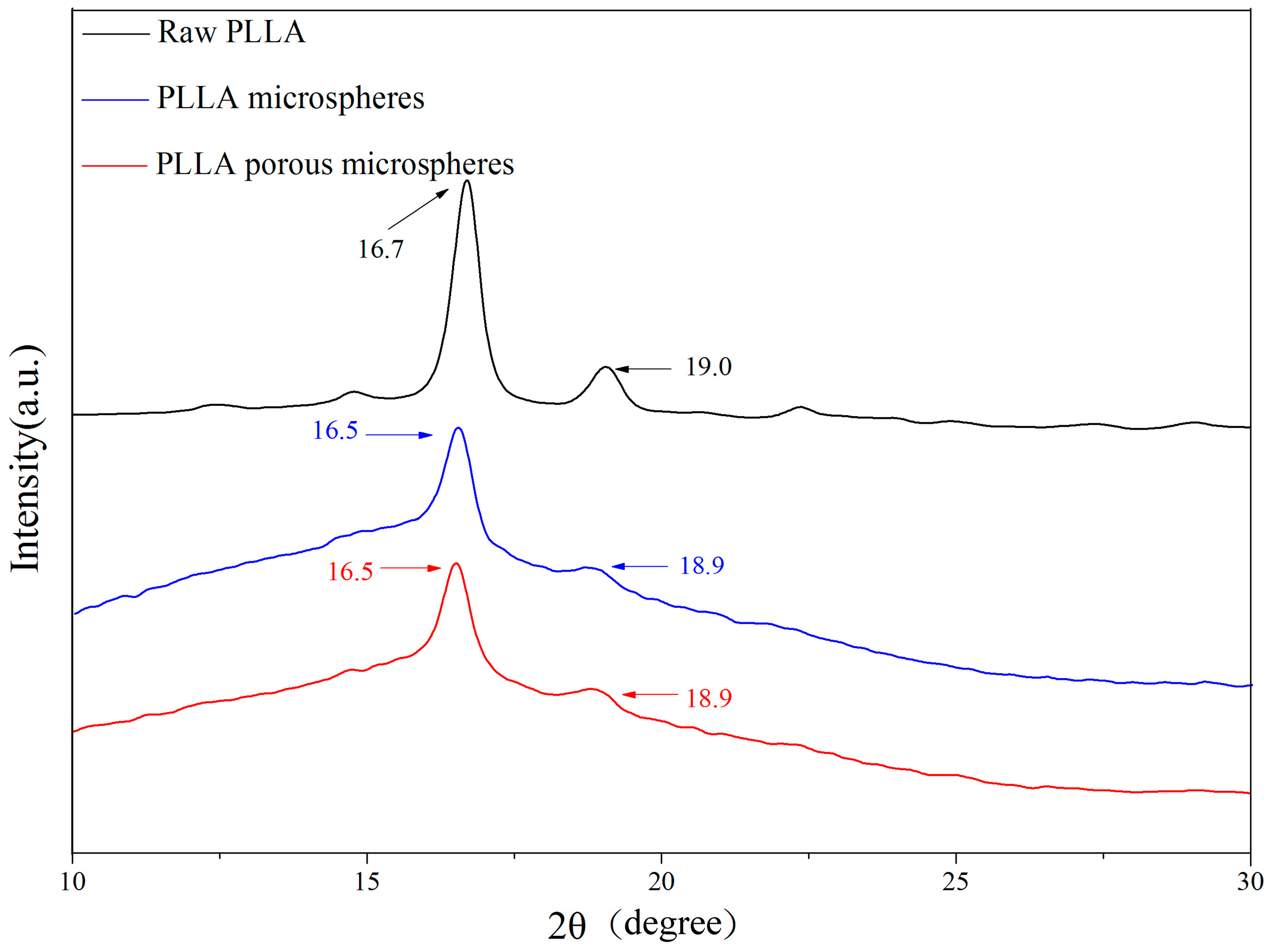
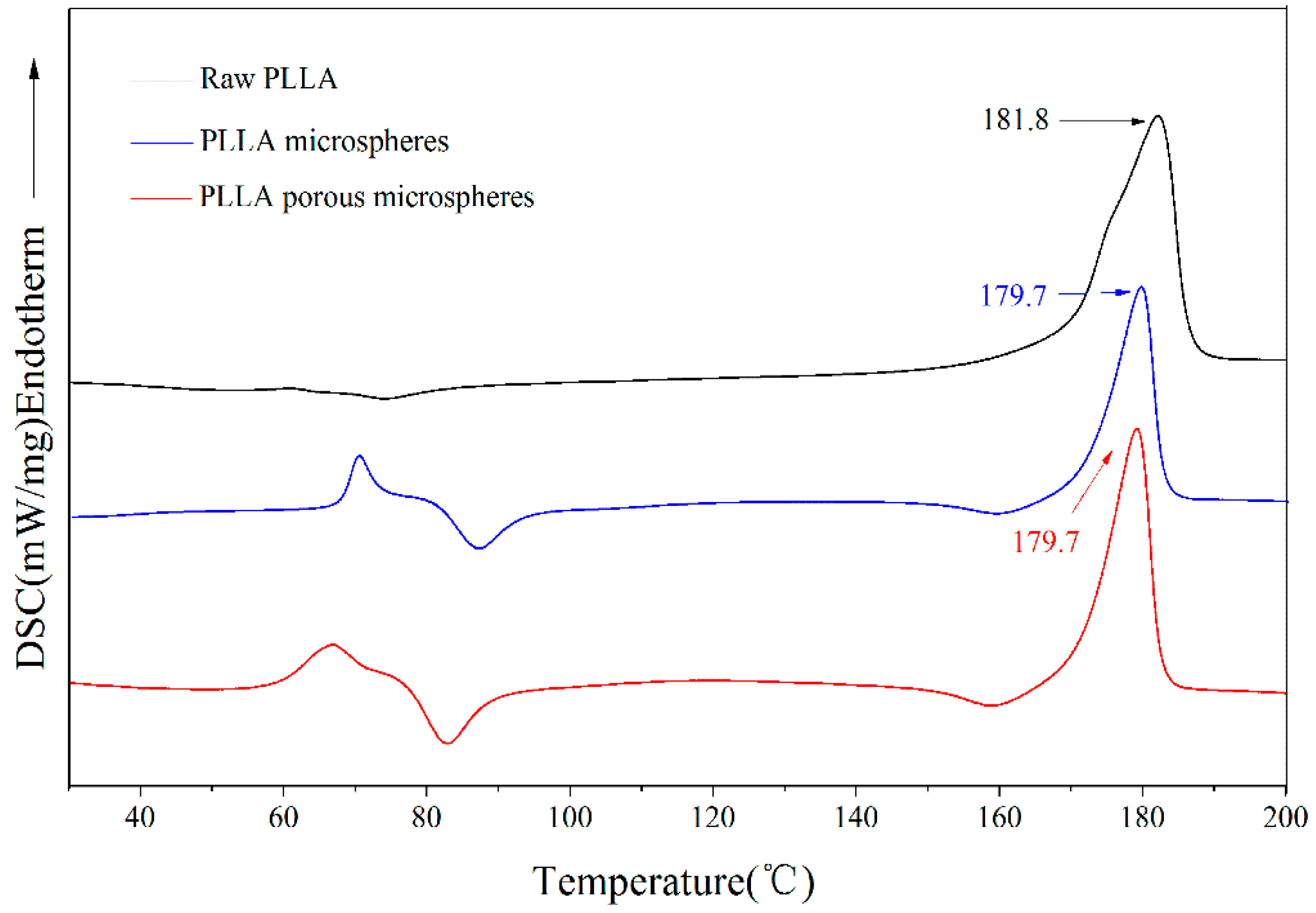
2.2. Physicochemical Properties of the PLLA PMs
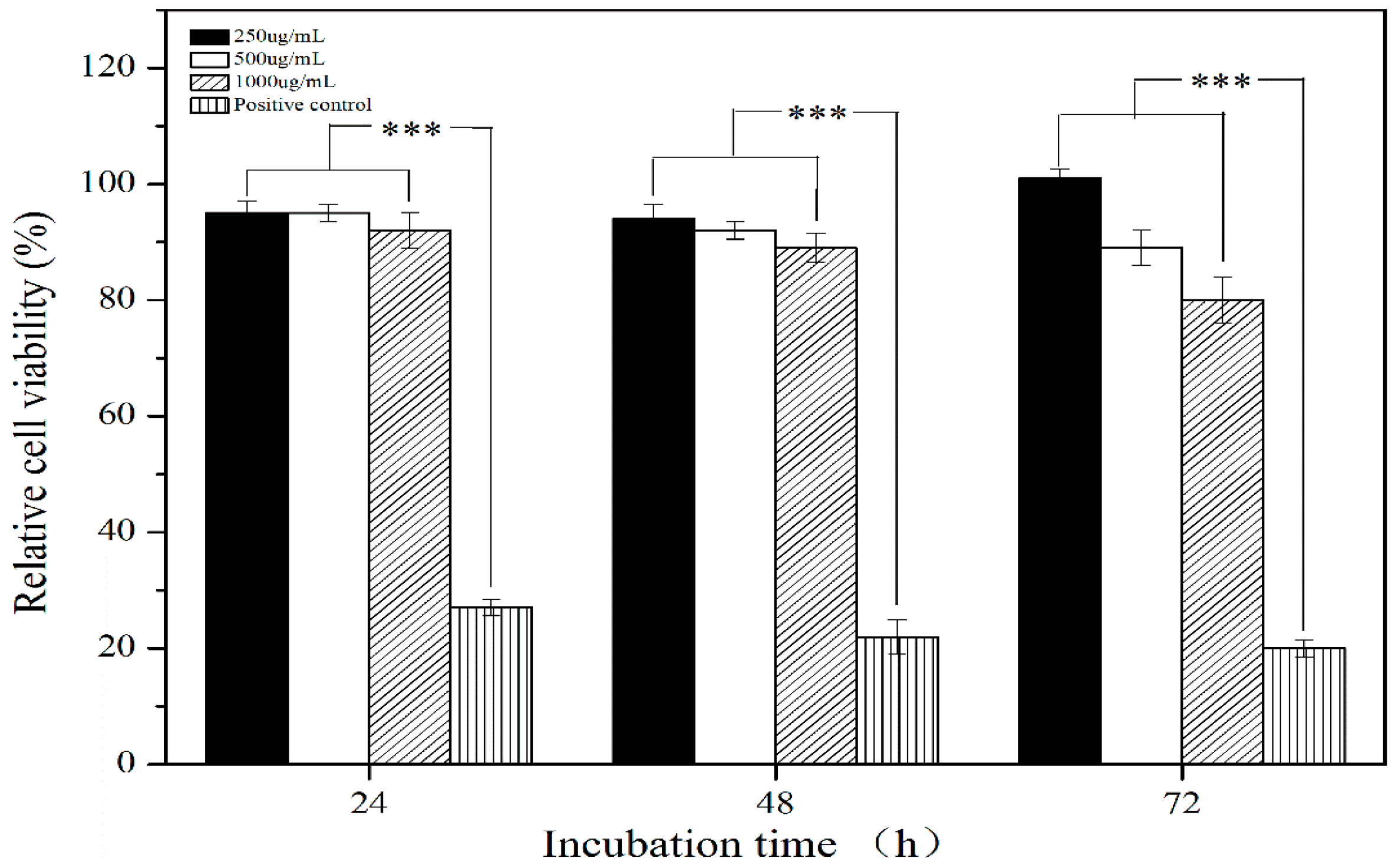
2.3. In Vitro Cytotoxicity Study
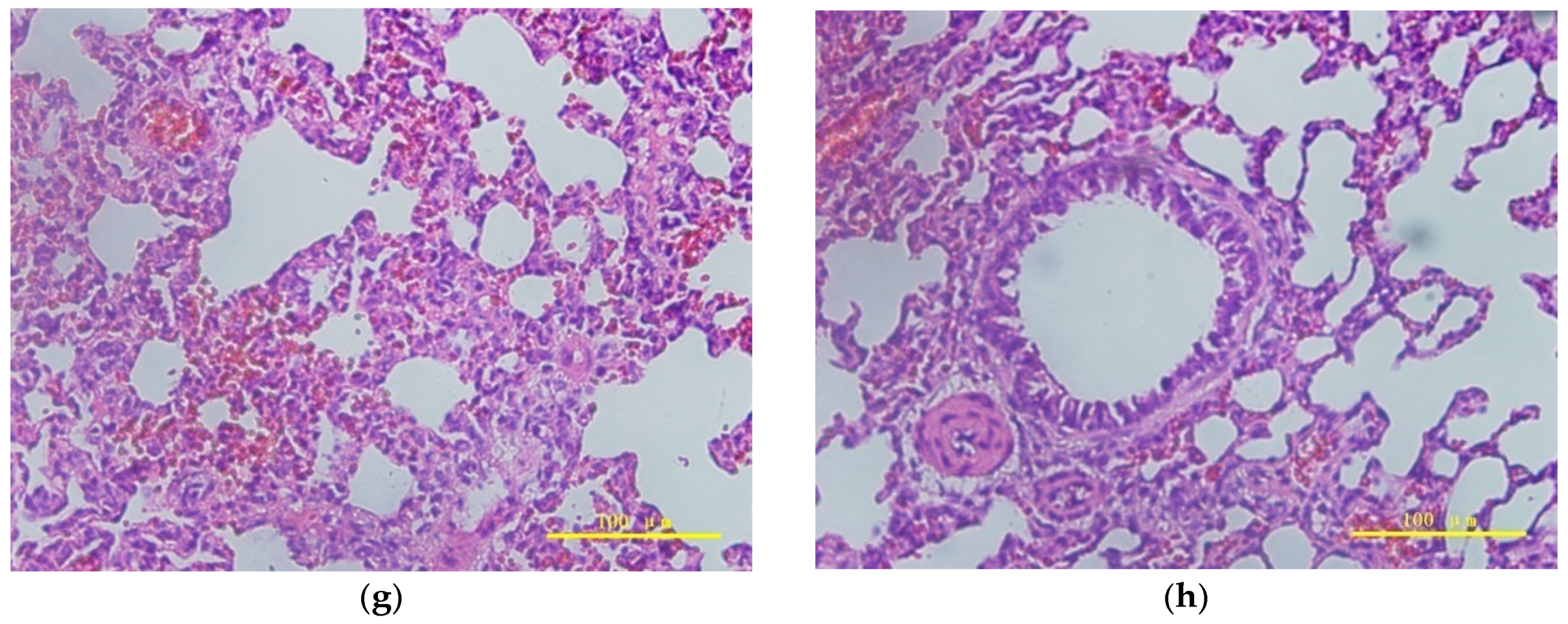
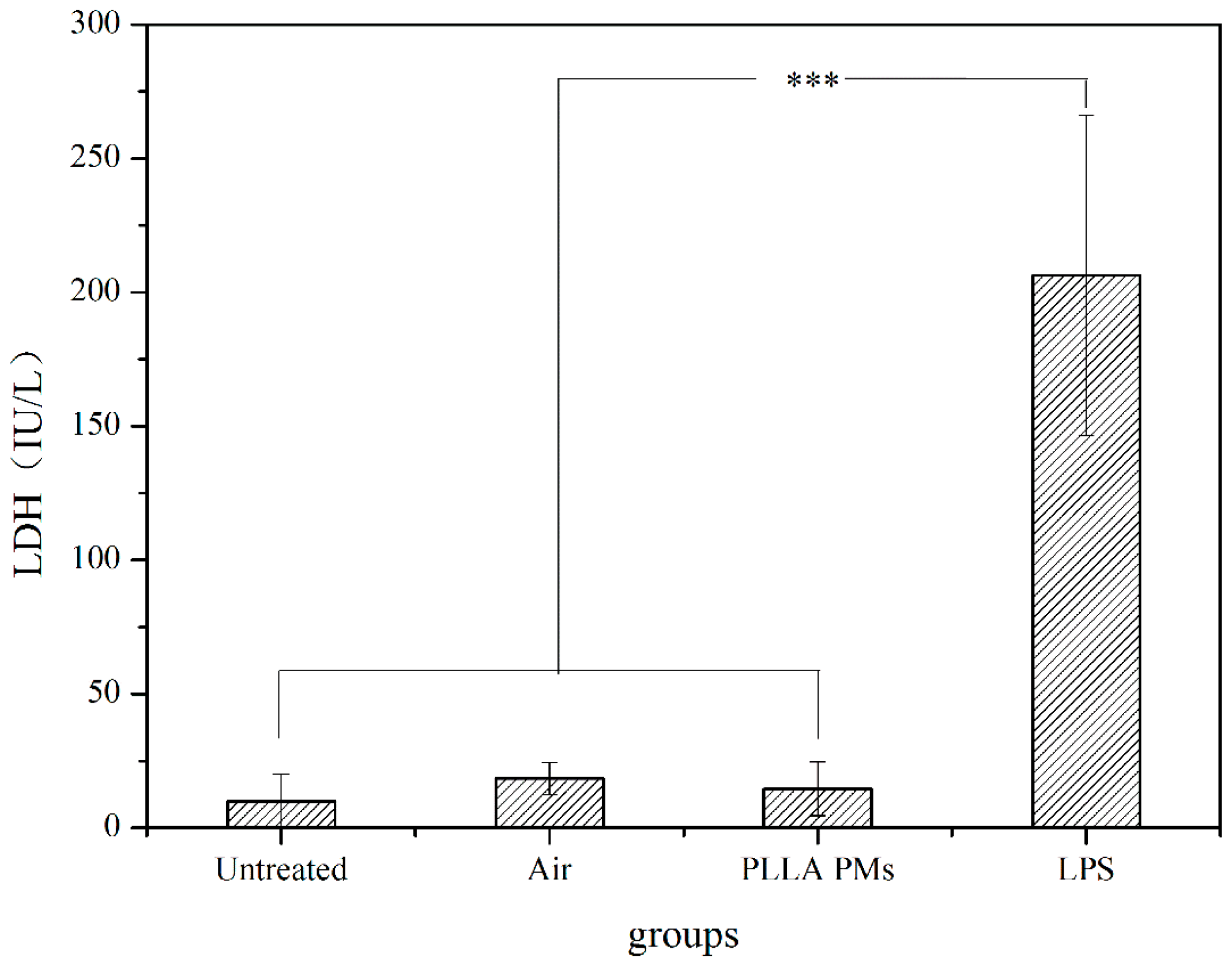
2.4. Pulmonary Toxicity Assessment
3. Materials and Methods
3.1. Materials
3.2. Methods
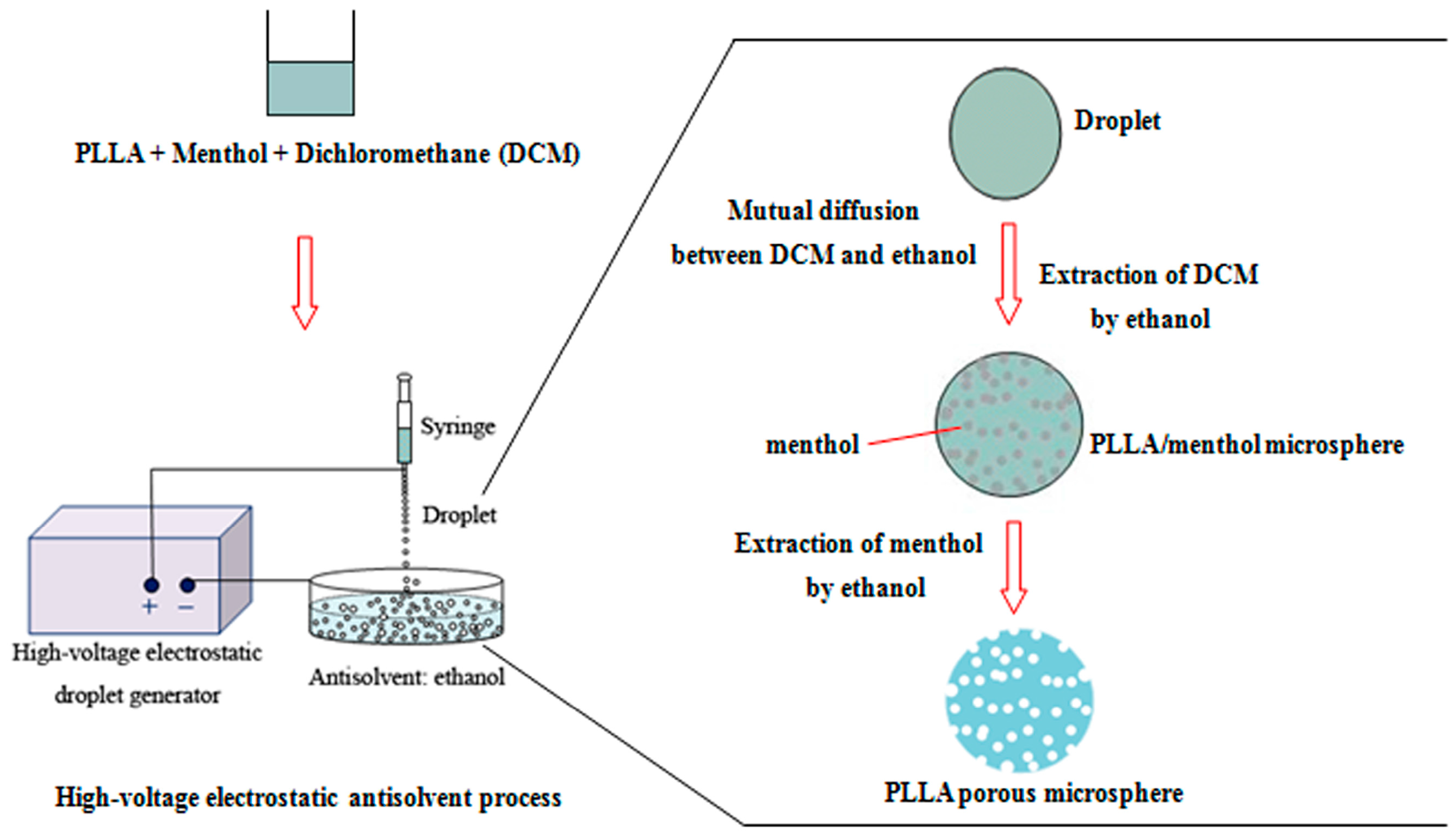
3.2.1. High-Voltage Electrostatic Anti-Solvent Process
3.2.2. Full Factorial Experimental Design
3.2.3. Surface Morphology Characterization
3.2.4. Aerodynamic Property Investigation
3.2.5. Physicochemical Properties of the PLLA PMs
3.2.6. Analysis of DCM Residue
3.2.7. In Vitro Cytotoxicity Investigation
3.2.8. Pulmonary Toxicity Assessment
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yang, Y.; Bajaj, N.; Xu, P.; Ohn, K.; Tsifansky, M.D.; Yeo, Y. Development of highly porous large PLGA microparticles for pulmonary drug delivery. Biomaterials 2009, 30, 1947–1953. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, K.; Dhanda, D.S.; Cheruvu, N.P.S.; Kompella, U.B. Pulmonary delivery of deslorelin: Large-porous plga particles and hpbetacd complexes. Pharm. Res. 2004, 21, 1119–1126. [Google Scholar]
- Chen, A.Z.; Zhao, C.; Wang, S.B.; Liu, Y.G.; Lin, D.L. Generation of porous poly-l-lactide microspheres by emulsion-combined precipitation with a compressed CO2 antisolvent process. J. Mater. Chem. B 2013, 1, 2967–2975. [Google Scholar] [CrossRef]
- Kim, I.; Byeon, H.J.; Kim, T.H.; Lee, E.S.; Oh, K.T.; Shin, B.S.; Lee, K.C.; Youn, Y.S. Doxorubicin-loaded highly porous large plga microparticles as a sustained- release inhalation system for the treatment of metastatic lung cancer. Biomaterials 2012, 33, 5574–5583. [Google Scholar] [CrossRef] [PubMed]
- Insoo, K.; Hyeong Jun, B.; Tae Hyung, K.; Eun Seong, L.; Kyung Taek, O.; Beom Soo, S.; Choon, L.K.; Seok, Y.Y. Doxorubicin-loaded porous PLGA microparticles with surface attached trail for the inhalation treatment of metastatic lung cancer. Biomaterials 2013, 34, 6444–6453. [Google Scholar]
- Francesca, U.; Concetta, G.; Agnese, M.; Raffaella, S.; Fabiana, Q.; Maria Immacolata, L.R. Insulin-loaded PLGA/cyclodextrin large porous particles with improved aerosolization properties: In vivo deposition and hypoglycaemic activity after delivery to rat lungs. J. Control. Release 2009, 135, 25–34. [Google Scholar]
- Hyunuk, K.; Hongil, P.; Juho, L.; Tae Hyung, K.; Eun Seong, L.; Kyung Taek, O.; Choon, L.K.; Seok, Y.Y. Highly porous large poly(lactic-co-glycolic acid) microspheres adsorbed with palmityl-acylated exendin-4 as a long-acting inhalation system for treating diabetes. Biomaterials 2011, 32, 1685–1693. [Google Scholar]
- Hyunuk, K.; Juho, L.; Tae Hyung, K.; Eun Seong, L.; Kyung Taek, O.; Don Haeng, L.; Eun-Seok, P.; Han, B.Y.; Choon, L.K.; Seok, Y.Y. Albumin-coated porous hollow poly (lactic-co-glycolic acid) microparticles bound with palmityl-acylated exendin-4 as a long-acting inhalation delivery system for the treatment of diabetes. Pharm. Res. 2011, 28, 2008–2019. [Google Scholar]
- Kang, Y.Q.; Zhao, C.; Chen, A.Z.; Wang, S.B.; Liu, Y.G.; Wu, W.G.; Su, X.Q. Study of lysozyme-loaded poly-l-lactide (PLLA) porous microparticles in a compressed CO2 antisolvent process. Materials 2013, 6, 3571–3583. [Google Scholar] [CrossRef]
- Na, Y.Y.; Yu, S.Y.; Oh, N.M.; Oh, K.T.; Lee, D.K.; Cha, K.H.; Oh, Y.T.; Lee, E.S. Antioxidant encapsulated porous poly (lactide-co-glycolide) microparticles for developing long acting inhalation system. Colloids Surf. B 2011, 88, 419–424. [Google Scholar]
- Francesca, U.; Concetta, G.; Ciro, C.; Raffaella, S.; Agnese, M.; Fabiana, Q. Engineering gas-foamed large porous particles for efficient local delivery of macromolecules to the lung. Eur. J. Pharm. Sci. 2010, 41, 60–70. [Google Scholar]
- Min, J.K.; Bae, J.H.; Kim, J.J.; Na, K.; Lee, E.S. Long acting porous microparticle for pulmonary protein delivery. Int. J. Pharm. 2007, 333, 5–9. [Google Scholar]
- Brijeshkumar, P.; Vivek, G.; Fakhrul, A. PEG-PLGA based large porous particles for pulmonary delivery of a highly soluble drug, low molecular weight heparin. J. Control. Release 2012, 162, 310–320. [Google Scholar]
- Zhu, W.; Ma, W.; Li, C.; Pan, J.; Dai, X. Well-designed multihollow magnetic imprinted microspheres based on cellulose nanocrystals (CNCS) stabilized pickering double emulsion polymerization for selective adsorption of bifenthrin. Chem. Eng. J. 2015, 276, 249–260. [Google Scholar] [CrossRef]
- Fidalgo, J.; Dias, Â.; Mendes, A.M.; Magalhães, F.D. Production of monodisperse multivesiculated polyester particles with a T-junction microfluidic device. Chem. Eng. J. 2013, 233, 323–330. [Google Scholar] [CrossRef]
- Liu, J.; Qiu, Z.; Wang, S.; Zhou, L.; Zhang, S. A modified double-emulsion method for the preparation of daunorubicin-loaded polymeric nanoparticle with enhanced in vitro anti-tumor activity. Biomed. Mater. 2010, 5, 065002. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Fu, D.; Xu, F.; Long, T.; Hong, F.; Wang, J. The design and features of apatite-coated chitosan microspheres as injectable scaffold for bone tissue engineering. Biomed. Mater. 2013, 8, 025007. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kanjwal, M.A.; Stephansen, K.; Chronakis, I.S. Preparing poly (caprolactone) micro-particles through solvent-induced phase separation. Mater. Lett. 2012, 75, 189–191. [Google Scholar] [CrossRef]
- Yang, J.; Hu, D.; Li, W.; Yi, S. Highly efficient microreactors with simultaneous separation of catalysts and products in deep desulfurization. Chem. Eng. J. 2015, 267, 93–101. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, D.; Chang, M.-W.; Ahmad, Z.; Li, X.; Suo, H.; Li, J.-S. Morphology control of electrosprayed core–shell particles via collection media variation. Mater. Lett. 2015, 146, 59–64. [Google Scholar] [CrossRef]
- Gao, Y.; Bai, Y.; Zhao, D.; Chang, M.W.; Ahmad, Z.; Li, J.S. Tuning microparticle porosity during single needle electrospraying synthesis via a non-solvent-based physicochemical approach. Polymers 2015, 7, 2701–2710. [Google Scholar] [CrossRef]
- Lee, E.S.; Min, J.K.; Na, K.; Bae, J.H. Protein release behavior from porous microparticle with lysozyme/hyaluronate ionic complex. Colloids Surf. B 2007, 55, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Jangwook, L.; Jin, O.Y.; Kyung, L.S.; Kuen Yong, L. Facile control of porous structures of polymer microspheres using an osmotic agent for pulmonary delivery. J. Control. Release 2010, 146, 61–67. [Google Scholar]
- Soon Eon, B.; Jun Sik, S.; Kwideok, P.; Dong Keun, H. Fabrication of covered porous PLGA microspheres using hydrogen peroxide for controlled drug delivery and regenerative medicine. J. Control. Release 2008, 133, 37–43. [Google Scholar]
- Taek Kyoung, K.; Jun Jin, Y.; Doo Sung, L.; Tae Gwan, P. Gas foamed open porous biodegradable polymeric microspheres. Biomaterials 2006, 27, 152–159. [Google Scholar]
- Yu, J.O.; Lee, J.; Ji, Y.S.; Rhim, T.; Kim, S.H.; Yoon, H.J.; Lee, K.Y. Preparation of budesonide-loaded porous PLGA microparticles and their therapeutic efficacy in a murine asthma model. J. Control. Release 2011, 150, 56–62. [Google Scholar]
- Ahmed, A.R.; Bodmeier, R. Preparation of preformed porous PLGA microparticles and antisense oligonucleotides loading. Eur. J. Pharm. Biopharm. 2008, 71, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.M.; Gorman, E.M.; Schieber, L.J.; Munson, E.J.; Berkland, C. Nanocipro encapsulation in monodisperse large porous plga microparticles. J. Control. Release 2007, 121, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Zhou, S.; Wang, W.; Li, X.; Wang, J.; Wang, J. Preparation and characterization of porous biodegradable microspheres used for controlled protein delivery. Colloids Surf. A 2009, 345, 173–181. [Google Scholar] [CrossRef]
- Wu, Y.; Clark, R.L. Controllable porous polymer particles generated by electrospraying. J. Colloid Interface Sci. 2007, 310, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Bock, N.; Woodruff, M.A.; Hutmacher, D.W.; Dargaville, T.R. Electrospraying, a reproducible method for production of polymeric microspheres for biomedical applications. Polymers 2011, 3, 131–149. [Google Scholar] [CrossRef]
- Xie, J.; Liang, K.L.; Phua, Y.; Hua, J.; Wang, C.H. Electrohydrodynamic atomization for biodegradable polymeric particle production. J. Colloid Interface Sci. 2006, 302, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Nangrejo, M.; Ahmad, Z.; Stride, E.; Edirisinghe, M.; Colombo, P. Preparation of polymeric and ceramic porous capsules by a novel electrohydrodynamic process. Pharm. Dev. Technol. 2008, 13, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.J.; Niu, G.C.; Kuo, S.M.; Chen, S.F. Preparation and preliminary characterization of concentric multi-walled chitosan microspheres. J. Biomed. Mater. Res. A 2007, 81, 554–566. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Z.; Yang, Y.M.; Wang, S.B.; Wang, G.Y.; Liu, Y.G.; Sun, Q.Q. Preparation of methotrexate-loaded, large, highly-porous PLLA microspheres by a high-voltage electrostatic antisolvent process. Mater. Sci. Mater. Med. 2013, 24, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Pfeffer, R.; Dave, R.; Enick, R. Polymer encapsulation of fine particles by a supercritical antisolvent process. Aiche J. 2005, 51, 440–455. [Google Scholar] [CrossRef]
- Dixon, D.J.; Johnston, K.P.; Bodmeier, R.A. Polymeric materials formed by precipitation with a compressed fluid antisolvent. Aiche J. 1993, 39, 127–139. [Google Scholar] [CrossRef]
- Ignjatović, N.; Savić, V.; Najman, S.; Plavšić, M.; Uskoković, D. A study of HAp/PLLA composite as a substitute for bone powder, using FT-IR spectroscopy. Biomaterials 2001, 22, 571–575. [Google Scholar] [CrossRef]
- Trasarti, A.F.; Marchi, A.J.; Apesteguía, C.R. Synthesis of menthols from citral on Ni/SiO2–Al2O3 catalysts. Catal. Commun. 2013, 32, 62–66. [Google Scholar] [CrossRef]
- Cortés, C.B.; Galván, V.T.; Pedro, S.S.; García, T.V. One pot synthesis of menthol from (±)-citronellal on nickel sulfated zirconia catalysts. Catal. Today 2011, 172, 21–26. [Google Scholar] [CrossRef]
- Pan, P.; Kai, W.; Zhu, B.; Tungalag Dong, A.; Inoue, Y. Polymorphous crystallization and multiple melting behavior of poly (l-lactide): Molecular weight dependence. Macromolecules 2007, 40, 6898–6905. [Google Scholar] [CrossRef]
- Zhou, Q.; Bao, M.; Yuan, H.; Zhao, S.; Dong, W.; Zhang, Y. Implication of stable jet length in electrospinning for collecting well-aligned ultrafine plla fibers. Polymer 2013, 54, 6867–6876. [Google Scholar] [CrossRef]
- Rangari, D.; Vasanthan, N. Study of strain-induced crystallization and enzymatic degradation of drawn poly (l-lactic acid) (PLLA) films. Macromolecules 2012, 45, 7397–7403. [Google Scholar] [CrossRef]
- Vanbever, R.; Mintzes, J.D.; Wang, J.; Nice, J.; Chen, D.; Batycky, R.; Langer, R.; Edwards, D.A. Formulation and physical characterization of large porous particles for inhalation. Pharm. Res. 1999, 16, 1735–1742. [Google Scholar] [CrossRef] [PubMed]

| Run Order | Factors | Particle Sizes (μm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | D10 | D50 | D90 | Dg | Span | Da | |
| 1 | −1 | 1 | −1 | 1 | 8.6 | 21.1 | 31.2 | 20.6 | 1.1 | 4.4 |
| 2 | −1 | −1 | −1 | −1 | 12.0 | 20.0 | 31.5 | 20.9 | 1.0 | 4.5 |
| 3 | −1 | −1 | 1 | 1 | 10.6 | 18.2 | 28.6 | 18.8 | 1.0 | 4.6 |
| 4 | 1 | −1 | −1 | −1 | 10.0 | 19.5 | 31.5 | 20.2 | 1.0 | 3.9 |
| 5 | 1 | 1 | 1 | 1 | 11.7 | 19.6 | 29.8 | 20.1 | 0.9 | 3.9 |
| 6 | −1 | 1 | 1 | −1 | 9.9 | 17.7 | 27.8 | 18.2 | 1.0 | 4.3 |
| 7 | −1 | 1 | −1 | −1 | 12.7 | 20.8 | 31.2 | 21.4 | 0.9 | 4.3 |
| 8 | 1 | 1 | −1 | 1 | 13.2 | 21.3 | 30.4 | 21.5 | 0.8 | 3.9 |
| 9 | −1 | −1 | −1 | 1 | 10.9 | 19.8 | 31.3 | 20.4 | 1.0 | 4.6 |
| 10 | 0 | 0 | 0 | 0 | 10.1 | 19.5 | 30.9 | 20.0 | 1.0 | 4.2 |
| 11 | 1 | 1 | 1 | −1 | 11.6 | 19.9 | 29.6 | 20.2 | 0.9 | 3.8 |
| 12 | 1 | −1 | −1 | 1 | 9.1 | 19.9 | 32.8 | 20.9 | 1.2 | 4.0 |
| 13 | −1 | 1 | 1 | 1 | 11.5 | 20.0 | 30.4 | 20.5 | 0.9 | 4.3 |
| 14 | −1 | −1 | 1 | −1 | 9.0 | 17.6 | 29.0 | 18.4 | 1.1 | 4.7 |
| 15 | 1 | −1 | 1 | 1 | 10.3 | 18.5 | 29.1 | 19.2 | 1.0 | 3.8 |
| 16 | 1 | −1 | 1 | −1 | 9.1 | 17.4 | 29.0 | 18.4 | 1.1 | 4.2 |
| 17 | 1 | 1 | −1 | −1 | 10.0 | 17.8 | 30.0 | 18.9 | 1.1 | 3.8 |
| Sample Name | Dg (μm) | Da (μm) | FPF (%) | Porosity (%) | Density (g/cm3) | Pore Size (μm) |
|---|---|---|---|---|---|---|
| PLLA PMs | 19.1 ± 0.4 | 4.2 ± 0.2 | 56.3 ± 1.2 | 76.2 | 0.289 ± 0.004 | 0.2 |
| Level | Code | A | B | C | D |
|---|---|---|---|---|---|
| Menthol: PLLA (w/w) | PLLA Concentration (wt/v, %) | Voltage (kV) | Push Speed (mm/h) | ||
| High | +1 | 1:1 | 4.0% | 10 | 50 |
| Centre | 0 | 3:4 | 3.5% | 9 | 45 |
| Low | −1 | 1:2 | 3.0% | 8 | 40 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhu, L.-H.; Chen, A.-Z.; Xu, Q.; Hong, Y.-J.; Wang, S.-B. One-Step Method to Prepare PLLA Porous Microspheres in a High-Voltage Electrostatic Anti-Solvent Process. Materials 2016, 9, 368. https://doi.org/10.3390/ma9050368
Wang Y, Zhu L-H, Chen A-Z, Xu Q, Hong Y-J, Wang S-B. One-Step Method to Prepare PLLA Porous Microspheres in a High-Voltage Electrostatic Anti-Solvent Process. Materials. 2016; 9(5):368. https://doi.org/10.3390/ma9050368
Chicago/Turabian StyleWang, Ying, Li-Hui Zhu, Ai-Zheng Chen, Qiao Xu, Yu-Juan Hong, and Shi-Bin Wang. 2016. "One-Step Method to Prepare PLLA Porous Microspheres in a High-Voltage Electrostatic Anti-Solvent Process" Materials 9, no. 5: 368. https://doi.org/10.3390/ma9050368
APA StyleWang, Y., Zhu, L.-H., Chen, A.-Z., Xu, Q., Hong, Y.-J., & Wang, S.-B. (2016). One-Step Method to Prepare PLLA Porous Microspheres in a High-Voltage Electrostatic Anti-Solvent Process. Materials, 9(5), 368. https://doi.org/10.3390/ma9050368





