Collagen Type I Conduits for the Regeneration of Nerve Defects
Abstract
:1. Introduction
2. Results
2.1. Implant Biocompability
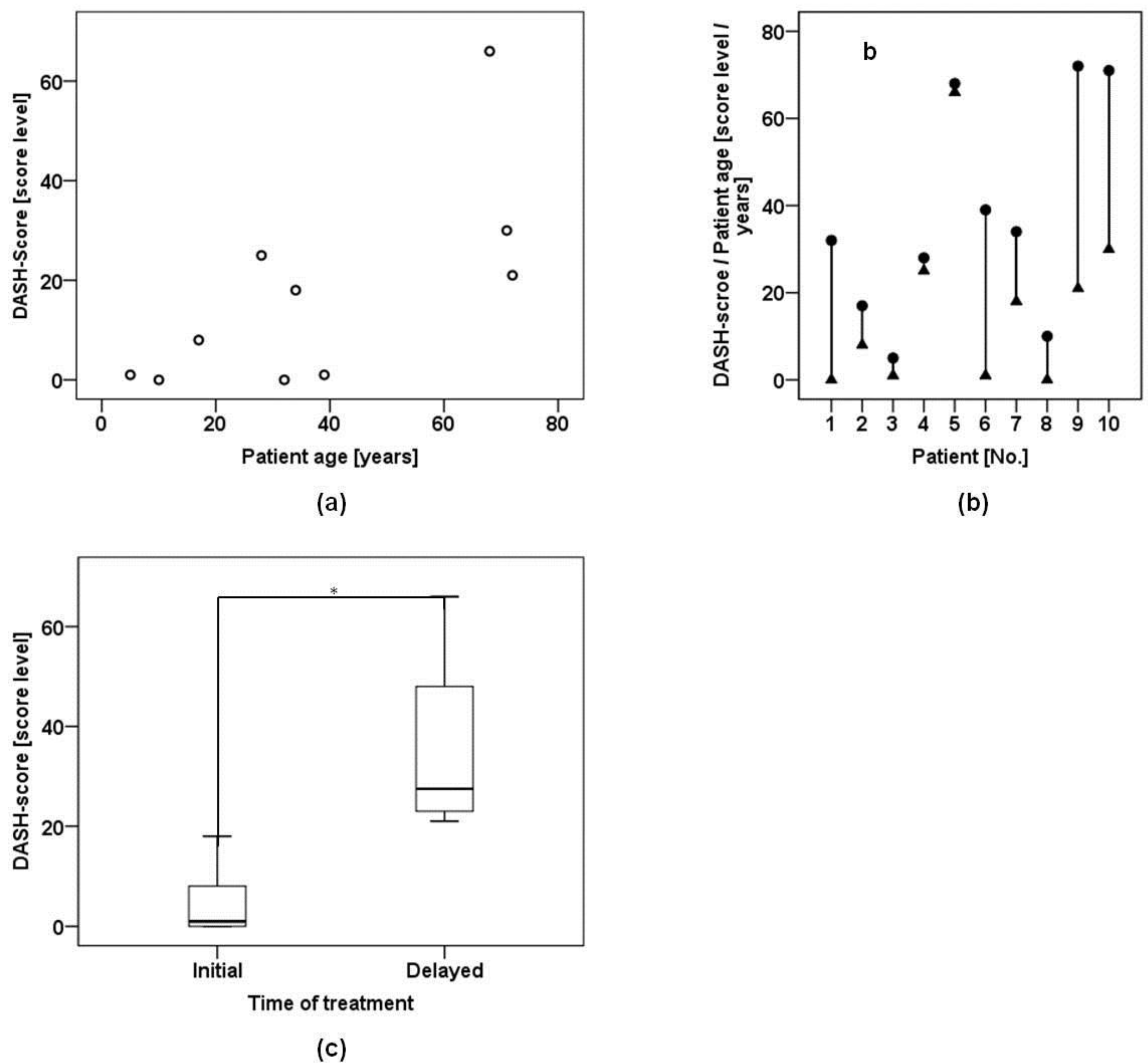
2.2. Disability of Arm Shoulder and Hand (DASH) and Visual Analogue Scale (VAS)
2.3. Nerve Conduction and Static Two Point Discrimination (S2PD)
2.4. Patient-Related Confounding Factors
3. Discussion
4. Materials and Methods
4.1. Patient Cohort
4.2. Nerve Conduit
4.3. Disability of the Arm, Shoulder and Hand Score
4.4. Nerve Conduction Velocities
4.5. Surgical Technique
4.6. Pain Assessment
4.7. Static Two-Point Discrimination
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thorsén, F.; Rosberg, H.-E.; Steen Carlsson, K.; Dahlin, L.B. Digital nerve injuries: Epidemiology, results, costs, and impact on daily life. J. Plast. Surg. Hand Surg. 2012, 46, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Boyd, J.B.; Olsavsky, A.; Olsavski, A.; Gelfand, M.; Putnam, B. Outcomes of complex gunshot wounds to the hand and wrist: A 10-year level I urban trauma center experience. Ann. Plast. Surg. 2012, 68, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Novak, C.B.; Anastakis, D.J.; Beaton, D.E.; Katz, J. Patient-reported outcome after peripheral nerve injury. J. Hand Surg. 2009, 34, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Rosberg, H.E.; Carlsson, K.S.; Höjgård, S.; Lindgren, B.; Lundborg, G.; Dahlin, L.B. Injury to the human median and ulnar nerves in the forearm—Analysis of costs for treatment and rehabilitation of 69 patients in southern Sweden. J. Hand Surg. Edinb. Scotl. 2005, 30, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Müller, H.; Grubel, G. Factors influencing peripheral nerve suture results. Arch. Orthop. Trauma. Surg. Arch. Für Orthop. Unf. Chir. 1983, 102, 51–55. [Google Scholar] [CrossRef]
- Millesi, H. Factors affecting the outcome of peripheral nerve surgery. Microsurgery 2006, 26, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Ray, W.Z.; Mackinnon, S.E. Management of nerve gaps: Autografts, allografts, nerve transfers, and end-to-side neurorrhaphy. Exp. Neurol. 2010, 223, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Ducic, I.; Maloney, C.T.; Dellon, A.L. Reconstruction of the spinal accessory nerve with autograft or neurotube? Two case reports. J. Reconstr. Microsurg. 2005, 21, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.N.; Weber, R.V.; Chao, J.D.; Rinker, B.D.; Zoldos, J.; Robichaux, M.R.; Ruggeri, S.B.; Anderson, K.A.; Bonatz, E.E.; Wisotsky, S.M.; et al. Processed nerve allografts for peripheral nerve reconstruction: A multicenter study of utilization and outcomes in sensory, mixed, and motor nerve reconstructions. Microsurgery 2012, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lohmeyer, J.A.; Siemers, F.; Machens, H.-G.; Mailänder, P. The clinical use of artificial nerve conduits for digital nerve repair: A prospective cohort study and literature review. J. Reconstr. Microsurg. 2009, 25, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Lundborg, G.; Rosén, B.; Dahlin, L.; Danielsen, N.; Holmberg, J. Tubular versus conventional repair of median and ulnar nerves in the human forearm: Early results from a prospective, randomized, clinical study. J. Hand Surg. 1997, 22, 99–106. [Google Scholar] [CrossRef]
- Hasegawa, T.; Nakamura, S.; Manabe, T.; Mikawa, Y. Vascularized nerve grafts for the treatment of large nerve gap after severe trauma to an upper extremity. Arch. Orthop. Trauma Surg. 2004, 124, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Hall, S. Nerve repair: A neurobiologist’s view. J. Hand Surg. Edinb. Scotl. 2001, 26, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Stang, F.; Stollwerck, P.; Prommersberger, K.J.; van Schoonhoven, J. Posterior interosseus nerve vs. medial cutaneous nerve of the forearm: Differences in digital nerve reconstruction. Arch. Orthop. Trauma Surg. 2013, 133, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Konofaos, P.; Ver Halen, J.P. Nerve repair by means of tubulization: Past, present, future. J. Reconstr. Microsurg. 2013, 29, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Daly, W.; Yao, L.; Zeugolis, D.; Windebank, A.; Pandit, A. A biomaterials approach to peripheral nerve regeneration: Bridging the peripheral nerve gap and enhancing functional recovery. J. R. Soc. Interface R. Soc. 2012, 9, 202–221. [Google Scholar] [CrossRef] [PubMed]
- Siemionow, M.; Bozkurt, M.; Zor, F. Regeneration and repair of peripheral nerves with different biomaterials: Review. Microsurgery 2010, 30, 574–588. [Google Scholar] [CrossRef] [PubMed]
- Stang, F.; Keilhoff, G.; Fansa, H. Biocompatibility of different nerve tubes. Materials 2009, 2, 1480–1507. [Google Scholar] [CrossRef]
- Taras, J.S.; Jacoby, S.M.; Lincoski, C.J. Reconstruction of digital nerves with collagen conduits. J. Hand Surg. 2011, 36, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Dienstknecht, T.; Klein, S.; Vykoukal, J.; Gehmert, S.; Koller, M.; Gosau, M.; Prantl, L. Type I collagen nerve conduits for median nerve repairs in the forearm. J. Hand Surg. 2013, 38, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.M.; Kasukurthi, R.; Magill, C.K.; Farhadi, H.F.; Borschel, G.H.; Mackinnon, S.E. Limitations of conduits in peripheral nerve repairs. Hand N. Y. 2009, 4, 180–186. [Google Scholar] [CrossRef] [PubMed]
- De Smet, L. The DASH questionnaire and score in the evaluation of hand and wrist disorders. Acta Orthop. Belg. 2008, 74, 575–581. [Google Scholar] [PubMed]
- Weber, R.A.; Breidenbach, W.C.; Brown, R.E.; Jabaley, M.E.; Mass, D.P. A randomized prospective study of polyglycolic acid conduits for digital nerve reconstruction in humans. Plast. Reconstr. Surg. 2000, 106, 1036–1045. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, B.D.; McWilliams, A.D.; Whitener, G.B.; Messer, T.M. Early clinical experience with collagen nerve tubes in digital nerve repair. J. Hand Surg. 2008, 33, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, S.E.; Dellon, A.L. Clinical nerve reconstruction with a bioabsorbable polyglycolic acid tube. Plast. Reconstr. Surg. 1990, 85, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Post, R.; de Boer, K.S.; Malessy, M.J.A. Outcome following nerve repair of high isolated clean sharp injuries of the ulnar nerve. PLoS ONE 2012, 7, e47928. [Google Scholar] [CrossRef] [PubMed]
- Archibald, S.J.; Krarup, C.; Shefner, J.; Li, S.T.; Madison, R.D. A collagen-based nerve guide conduit for peripheral nerve repair: An electrophysiological study of nerve regeneration in rodents and nonhuman primates. J. Comp. Neurol. 1991, 306, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Deal, D.N.; Griffin, J.W.; Hogan, M.V. Nerve conduits for nerve repair or reconstruction. J. Am. Acad. Orthop. Surg. 2012, 20, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Hudson, T.W.; Evans, G.R.; Schmidt, C.E. Engineering strategies for peripheral nerve repair. Orthop. Clin. North Am. 2000, 31, 485–498. [Google Scholar] [CrossRef]
- Tan, A.; Rajadas, J.; Seifalian, A.M. Biochemical engineering nerve conduits using peptide amphiphiles. J. Control. Release Off. J. Control. Release Soc. 2012, 163, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Nectow, A.R.; Marra, K.G.; Kaplan, D.L. Biomaterials for the development of peripheral nerve guidance conduits. Tissue Eng. Part B Rev. 2012, 18, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Lim, S.H.; Mao, H.-Q.; Chew, S.Y. Current applications and future perspectives of artificial nerve conduits. Exp. Neurol. 2010, 223, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Kingham, P.J.; Reid, A.J.; Wiberg, M. Adipose-derived stem cells for nerve repair: Hype or reality? Cells Tissues Organs 2014, 200, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kappos, E.A.; Engels, P.E.; Tremp, M.; Meyer zu Schwabedissen, M.; di Summa, P.; Fischmann, A.; von Felten, S.; Scherberich, A.; Schaefer, D.J.; Kalbermatten, D.F. Peripheral nerve repair: Multimodal comparison of the long-term regenerative potential of adipose tissue-derived cells in a biodegradable conduit. Stem Cells Dev. 2015, 24, 2127–2141. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Zhu, Z.; Zhu, Q.; Zhou, X.; Zheng, C.; Li, P.; Zhu, S.; Liu, X.; Zhu, J. Factors predicting sensory and motor recovery after the repair of upper limb peripheral nerve injuries. Neural Regen. Res. 2014, 9, 661–672. [Google Scholar] [PubMed]
- Battiston, B.; Geuna, S.; Ferrero, M.; Tos, P. Nerve repair by means of tubulization: Literature review and personal clinical experience comparing biological and synthetic conduits for sensory nerve repair. Microsurgery 2005, 25, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Jardin, E.; Huard, S.; Chastel, R.; Uhring, J.; Obert, L. Large diameter nerve conduits use in the upper limb: Report of four cases and literature review. Chir. Main 2011, 30, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Şahin, F.; Atalay, N.Ş.; Akkaya, N.; Ercidoğan, Ö.; Başakçi, B.; Kuran, B. The correlation of neurophysiological findings with clinical and functional status in patients following traumatic nerve injury. J. Hand Surg. Eur. Vol. 2014, 39, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Lundborg, G. A 25-year perspective of peripheral nerve surgery: Evolving neuroscientific concepts and clinical significance. J. Hand Surg. 2000, 25, 391–414. [Google Scholar] [CrossRef] [PubMed]
- Gummesson, C.; Atroshi, I.; Ekdahl, C. The disabilities of the arm, shoulder and hand (DASH) outcome questionnaire: Longitudinal construct validity and measuring self-rated health change after surgery. BMC Musculoskelet. Disord. 2003, 4, 11. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klein, S.; Vykoukal, J.; Felthaus, O.; Dienstknecht, T.; Prantl, L. Collagen Type I Conduits for the Regeneration of Nerve Defects. Materials 2016, 9, 219. https://doi.org/10.3390/ma9040219
Klein S, Vykoukal J, Felthaus O, Dienstknecht T, Prantl L. Collagen Type I Conduits for the Regeneration of Nerve Defects. Materials. 2016; 9(4):219. https://doi.org/10.3390/ma9040219
Chicago/Turabian StyleKlein, Silvan, Jody Vykoukal, Oliver Felthaus, Thomas Dienstknecht, and Lukas Prantl. 2016. "Collagen Type I Conduits for the Regeneration of Nerve Defects" Materials 9, no. 4: 219. https://doi.org/10.3390/ma9040219
APA StyleKlein, S., Vykoukal, J., Felthaus, O., Dienstknecht, T., & Prantl, L. (2016). Collagen Type I Conduits for the Regeneration of Nerve Defects. Materials, 9(4), 219. https://doi.org/10.3390/ma9040219






