Comparison of Electrochemical Methods for the Evaluation of Cast AZ91 Magnesium Alloy
Abstract
:1. Introduction
2. Experimental Material and Methods
2.1. Material
2.2. Microstructure Analysis
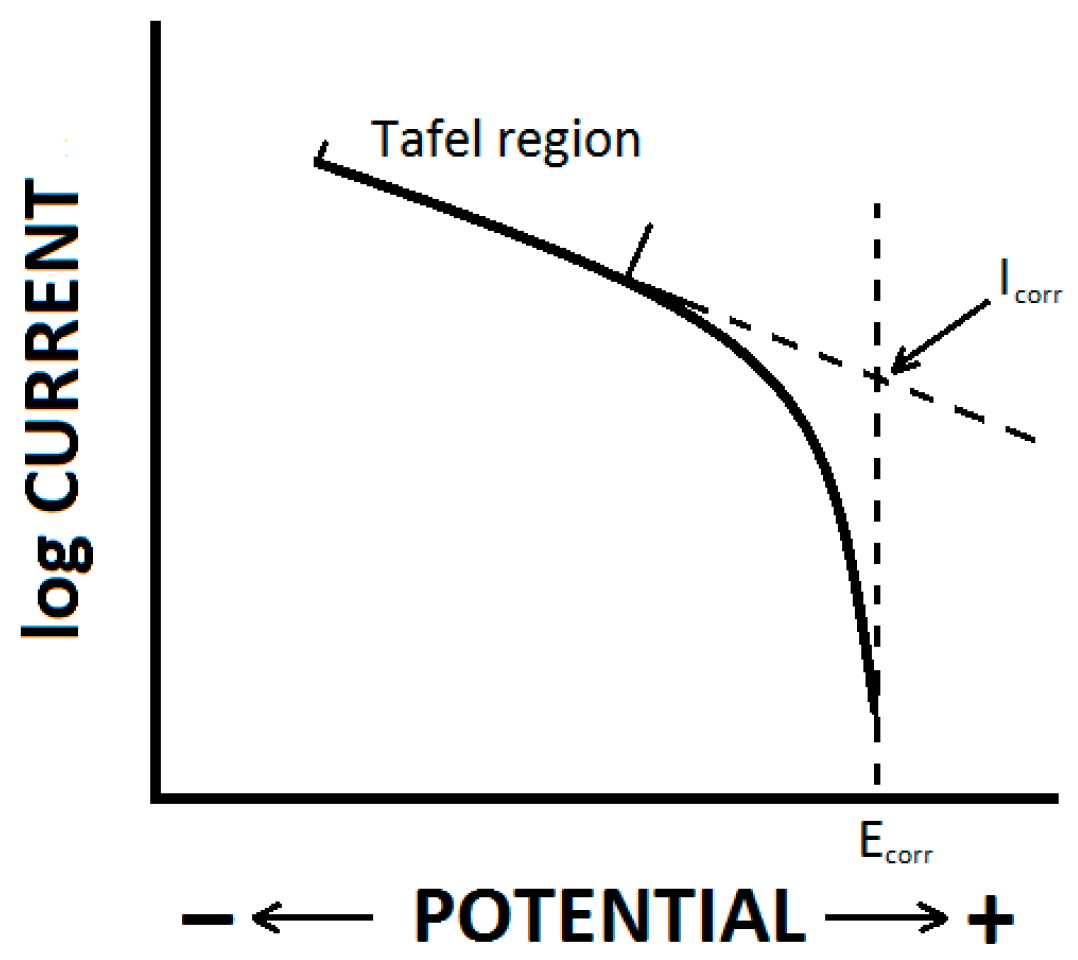
2.3. Electrochemical Measurements
3. Results
3.1. Microstructural Analysis
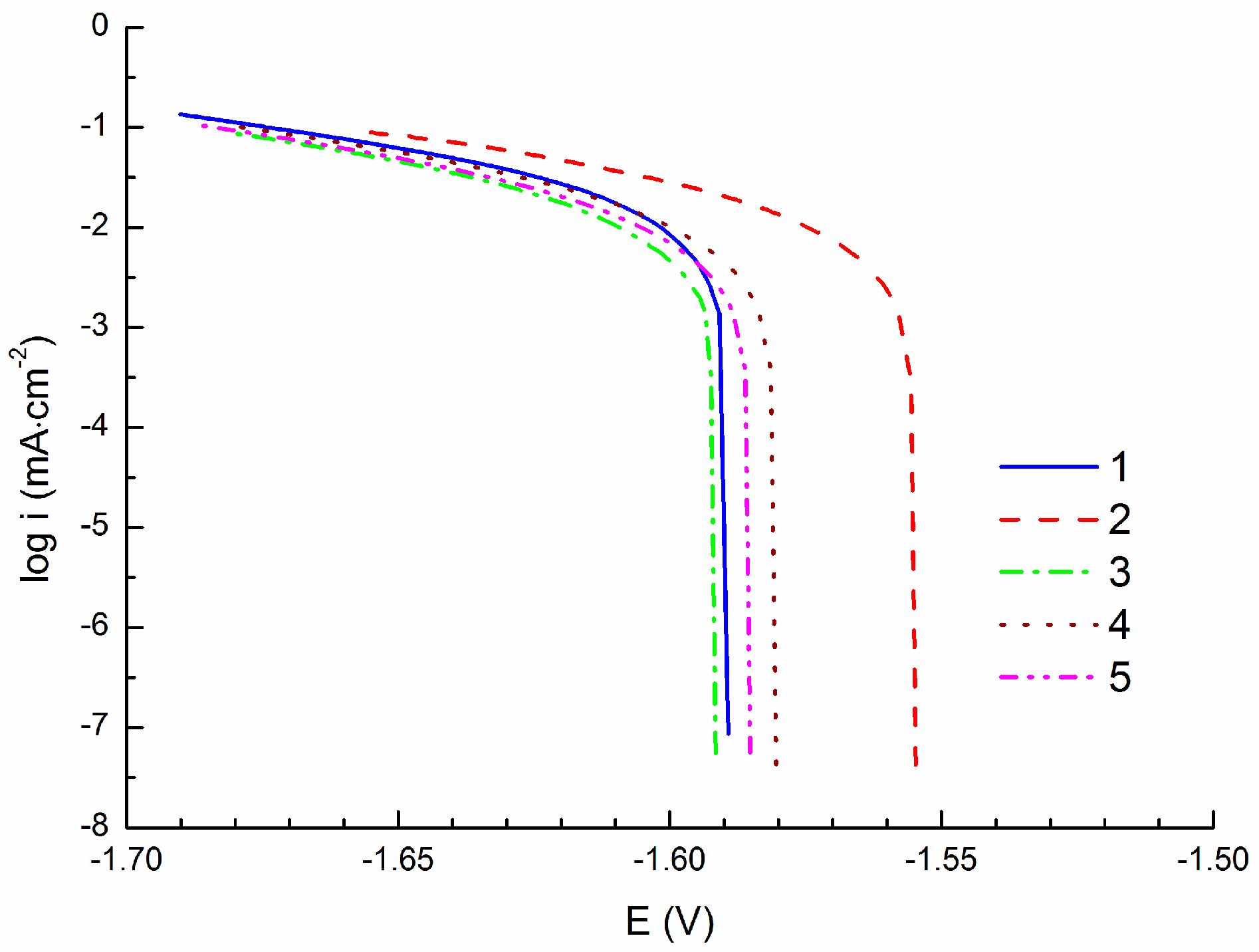
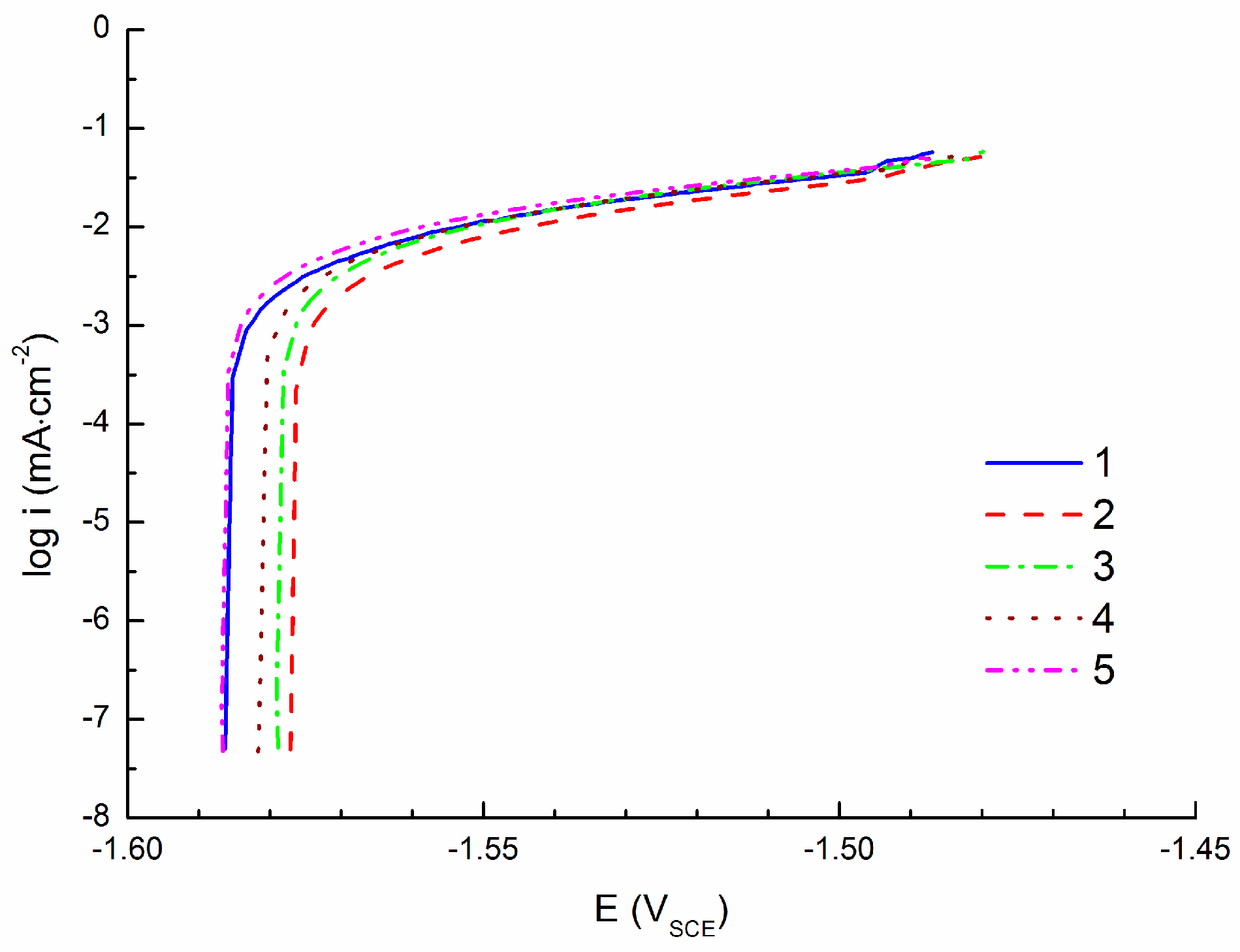
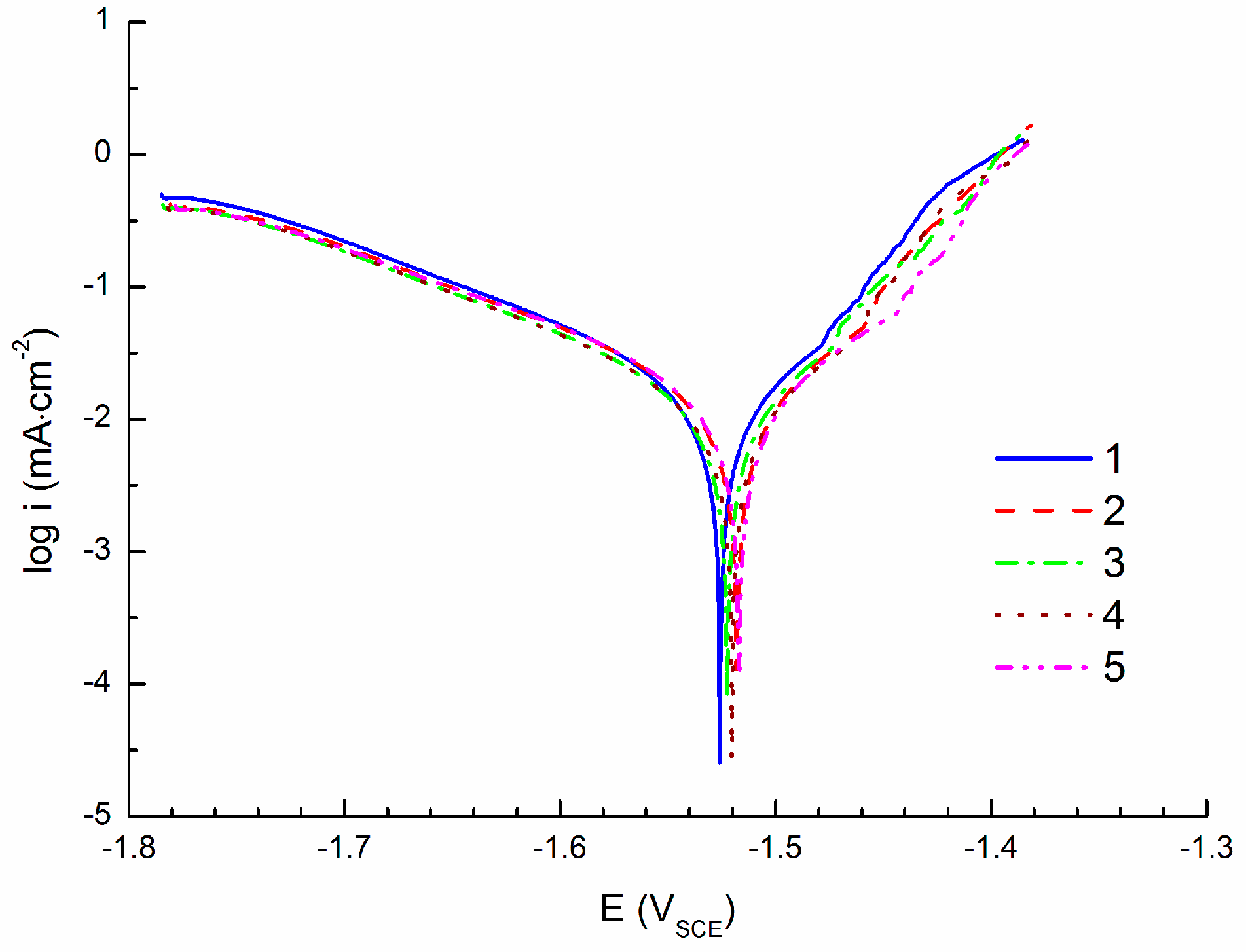
3.2. Linear Polarization Measurements
4. Discussion
5. Conclusions
- The microstructure of the examined AZ91 cast magnesium alloy was considered to be homogenous from the macroscopic point of view and homogenous corrosion behavior was observed.
- Corrosion current density of icorr = 15.4 ± 1.0 µA·cm−2 obtained by cathodic polarization is similar to the corrosion current density icorr = 14.7 ± 0.6 µA·cm−2 obtained by linear polarization. On the other hand, corrosion current density icorr = 8.0 ± 0.6 µA·cm−2 obtained by anodic polarization is significantly lower comparing to the previous methods.
- Corrosion potentials obtained from PC are shifted to more positive values of potential compared to the ACP and CPC.
- Significant pitting corrosion was observed only in the case of linear polarization measurements. Localized corrosion product areas were observed on PC and APC specimens.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cui, Z.; Li, X.; Xiao, K.; Dong, C. Atmospheric corrosion of field-exposed AZ31 magnesium in a tropical marine environment. Corros. Sci. 2013, 76, 243–256. [Google Scholar] [CrossRef]
- Jian, S.Y.; Chu, Y.R.; Lin, C.S. Permanganate conversion coating on AZ31 magnesium alloys with enhanced corrosion resistance. Corros. Sci. 2015, 93, 301–309. [Google Scholar] [CrossRef]
- Tkacz, J.; Němcová, A.; Zmrzlý, M.; Pacal, B. Improvement of corrosion resistance of AZ61 magnesium alloy. Koroze Ochr. Mater. 2013, 56, 100–103. [Google Scholar]
- Kulecki, M.K. Magnesium and its alloys applications in automotive industry. Int. J. Adv. Manuf. Technol. 2008, 39, 851–865. [Google Scholar]
- Mordike, B.L.; Ebert, T. Magnesium: Properties-applications-potential. Mater. Sci. Eng. A 2001, 302, 37–45. [Google Scholar] [CrossRef]
- Friedrich, H.; Schumann, S. Research for a “new age of magnesium” in the automotive industry. J. Mater. Process. Technol. 2001, 117, 276–281. [Google Scholar] [CrossRef]
- Gray, J.E.; Luan, B. Protective coatings on magnesium and its alloys—A critical review. J. Alloys Compd. 2002, 336, 88–113. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, C. Corrosion and Protection of Magnesium Alloys—A Review of the Patent Literature. Recent Pat. Corros. Sci. 2010, 2, 55–64. [Google Scholar] [CrossRef]
- Mousa, H.M.; Hussein, K.H.; Woo, H.M.; Park, C.H.; Kim, C.S. One-step anodization deposition of anticorrosive bioceramic compounds on AZ31B magnesium alloy for biomedical application. Ceram. Int. 2015, 61, 10861–10870. [Google Scholar] [CrossRef]
- Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Meyer-Lindenberg, A.; Wirth, C.J.; Windhagen, H. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 2005, 26, 3557–3563. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.; Qu, X.; Li, H.; Yang, K.; Wan, P.; Tan, L.; Ouyang, Z.; Liu, X.; Tian, B.; Xiao, F.; et al. The effect of metallic magnesium degradation products on osteoclast-induced osteolysis and attenuation of NF-κB and NFATc1 signaling. Biomaterials 2014, 35, 6299–6310. [Google Scholar] [CrossRef] [PubMed]
- Amaravathy, P.; Sathyanarayanan, S.; Sowndarya, S.; Rajendran, N. Bioactive HA/TiO2 coating on magnesium alloy for biomedical applications. Ceram. Int. 2014, 40, 6617–6630. [Google Scholar] [CrossRef]
- Razavi, M.; Fathi, M.; Savabi, O.; Razavi, S.M.; Beni, B.H.; Vashaee, D.; Tayebi, L. Controlling the degradation rate of bioactive magnesium implants by electrophoretic deposition of akermanite coating. Ceram. Int. 2014, 40, 3865–3872. [Google Scholar] [CrossRef]
- Wu, F.; Wei, J.; Guo, H.; Chen, F.; Hong, H.; Liu, C. Self-setting bioactive calcium–magnesium phosphate cement with high strength and degradability for bone regeneration. Acta Biomater. 2008, 4, 1873–1884. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Jin, X.; Zhang, X.; Sun, H.; Tu, J.; Tang, T.; Chang, J.; Dai, K. In vitro and in vivo evaluation of akermanite bioceramics for bone regeneration. Biomaterials 2009, 30, 5041–5048. [Google Scholar] [CrossRef] [PubMed]
- Barchiche, C.E.; Rocca, E.; Juers, C.; Hazan, J.; Steinmetz, J. Corrosion resistance of plasma-anodized AZ91D magnesium alloy by electrochemical methods. Electrochim. Acta 2007, 53, 417–425. [Google Scholar] [CrossRef]
- Ardelean, H.; Frateur, I.; Zanna, S.; Atrens, A.; Marcus, P. Corrosion protection of AZ91 magnesium alloy by anodizing in niobium and zirconium-containing electrolytes. Corros. Sci. 2009, 51, 3030–3038. [Google Scholar] [CrossRef]
- Lin, X.; Wang, X.; Tan, L.; Wan, P.; Yu, X.; Li, Q.; Yang, K. Effect of preparation parameters on the properties of hydroxyapatite containing micro-arc oxidation coating on biodegradable ZK60 magnesium alloy. Ceram. Int. 2014, 40, 10043–10051. [Google Scholar] [CrossRef]
- Hiromoto, S.; Inoue, M.; Taguchi, T.; Yamane, M.; Ohtsu, N. In vitro and in vivo biocompatibility and corrosion behaviour of a bioabsorbable magnesium alloy coated with octacalcium phosphate and hydroxyapatite. Acta Biomater. 2015, 11, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Heublein, B.; Rohde, R.; Kaese, V.; Niemeyer, M.; Hartung, W.; Haverich, A. Biocorrosion of magnesium alloys: A new principle in cardiovascular implant technology? Heart 2003, 89, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Calliess, T.; Windhagen, H. Degradable synthetische Implantatmaterialien. Orthopäde 2008, 37, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Dietzel, W.; Witte, F.; Hort, N.; Blawert, C. Progress and Challenge for Magnesium Alloys as Biomaterials. Adv. Eng. Mater. 2008, 10, B3–B14. [Google Scholar] [CrossRef]
- Witte, F. The history of biodegradable magnesium implants: A review. Acta Biomater. 2010, 6, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Waksman, R.; Pakala, R. Biodegradable and Bioabsorbable Stents. Curr. Pharm. Des. 2010, 16, 4041–4051. [Google Scholar] [CrossRef] [PubMed]
- Zartner, P.; Buettner, M.; Singer, H.; Sigler, M. First biodegradable metal stent in a child with congenital heart disease: Evaluation of macro and histopathology. Catheter. Cardiovasc. Interv. 2007, 69, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Erbel, R.; Mario, C.D.; Bartunek, J.; Bonnier, J.; Bruyne, B.D.; Eberli, F.R.; Erne, P.; Haude, M.; Heublein, B.; Horrigan, M.; et al. Temporary scaffolding of coronary arteries with bioabsorbable magnesium stents: A prospective, non-randomised multicentre trial. Lancet 2007, 369, 1869–1875. [Google Scholar] [CrossRef]
- Haude, M.; Erbel, R.; Varheye, S.; Degen, H.; Böse, D.; Vermeersch, P.; Wijnbergen, I.; Wiessman, N.; Prati, F.; Waksman, R.; et al. Safety and performance of the drug-eluting absorbable metal scaffold (DREAMS) in patients with de-novo coronary lesions: 12 month results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. Lancet 2013, 381, 836–844. [Google Scholar] [CrossRef]
- Harrison, R.; Maradze, D.; Lyons, S.; Zheng, Y.; Liu, Y. Corrosion of magnesium and magnesium–calcium alloy in biologically-simulated environment. Prog. Nat. Sci. 2014, 24, 539–546. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Birbilis, N.; Staiger, M.P. Assessing the corrosion of biodegradable magnesium implants: A critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Oyen, M.L.; Ferguson, V.L.; Bembey, A.K.; Bushby, A.J.; Boyde, A. Composite bounds on the elastic modulus of bone. J. Biomech. 2008, 41, 2585–2588. [Google Scholar] [CrossRef] [PubMed]
- Rho, J.Y.; Ashman, R.B.; Turner, C.H. Young’s modulus of trabecular and cortical bone material: Ultrasonic and microtensile measurements. J. Biomech. 1993, 26, 111–119. [Google Scholar] [CrossRef]
- Castellani, C.; Lindtner, R.A.; Hausbrandt, P.; Tschegg, E.; Stanzl-Tschegg, S.E.; Zanoni, G.; Beck, S.; Weinberg, A.M. Bone–implant interface strength and osseointegration: Biodegradable magnesium alloy versus standard titanium control. Acta Biomater. 2011, 7, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Feyerabend, F.; Maier, P.; Fischer, J.; Störmer, M.; Blawert, C.; Dietzel, W.; Hort, N. Biodegradable magnesium–hydroxyapatite metal matrix composites. Biomaterials 2007, 28, 2163–2174. [Google Scholar] [CrossRef] [PubMed]
- Waizy, H.; Seitz, J.M.; Reifenrath, J.; Weizbauer, A.; Bach, F.W.; Meyer-Lindenberg, A.; Denkena, B.; Windhagen, H. Biodegradable magnesium implants for orthopedic applications. J. Mater. Sci. 2013, 48, 39–50. [Google Scholar] [CrossRef]
- Grubbs, R.D.; Maguire, M.E. Magnesium as a regulatory cation: Criteria and evaluation. Magnesium 1987, 6, 113–127. [Google Scholar] [PubMed]
- Beyenbach, K.W. Transport of magnesium across biological membranes. Magnes. Trace Elem. 1990, 9, 223–254. [Google Scholar]
- Saris, N.E.L.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium: An update on physiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Wacker, W.E.C.; Parisi, A.F. Magnesium metabolism. Med. Prog. 1968, 278, 772–776. [Google Scholar]
- Flatman, P.W. Mechanisms of Magnesium Transport. Physiology 1991, 53, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Romani, A.M.; Scarpa, A. Regulation of cellular magnesium. Front. Biosci. 2000, 5, D720–D734. [Google Scholar] [CrossRef] [PubMed]
- Agus, Z.S.; Morad, M. Modulation of Cardiac ION Channels by Magnesium. Physiology 1991, 53, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Song, G. Control of biodegradation of biocompatable magnesium alloys. Corros. Sci. 2007, 49, 1696–1701. [Google Scholar] [CrossRef]
- Hong, D.; Saha, P.; Chou, D.T.; Lee, B.; Collins, B.E.; Tan, Z.; Dong, Z.; Kumta, P.N. In vitro degradation and cytotoxicity response of Mg–4% Zn–0.5% Zr (ZK40) alloy as a potential biodegradable material. Acta Biomater. 2013, 9, 8534–8547. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.; Liu, H. A Study on Factors Affecting the Degradation of Magnesium and a Magnesium-Yttrium Alloy for Biomedical Applications. PLoS ONE 2013, 8, e65603. [Google Scholar] [CrossRef] [PubMed]
- Guan, R.G.; Johnson, I.; Cui, T.; Zhao, T.; Zhao, Z.Y.; Li, X.; Liu, H. Electrodeposition of hydroxyapatite coating on Mg–4.0Zn–1.0Ca–0.6Zr alloy and in vitro evaluation of degradation, hemolysis, and cytotoxicity. J. Biomed. Mater. Res. Part A 2012, 100, 999–1015. [Google Scholar] [CrossRef] [PubMed]
- Liu, H. The effects of surface and biomolecules on magnesium degradation and mesenchymal stem cell adhesion. J. Biomed. Mater. Res. Part A 2011, 99, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Birbilis, N.; Williams, G.; Gusieva, K.; Samaniego, A.; Gibson, M.A.; McMurray, H.N. Poisoning the corrosion of magnesium. Electrochem. Commun. 2013, 34, 295–298. [Google Scholar] [CrossRef]
- Hadzima, B.; Mhaede, M.; Pastorek, F. Electrochemical characteristics of calcium-phosphatized AZ31 magnesium alloy in 0.9% NaCl solution. J. Mater. Sci. Mater. Med. 2014, 25, 1217–1237. [Google Scholar] [CrossRef] [PubMed]
- Altun, H.; Sen, S. The effect of PVD coatings on the corrosion behaviour of AZ91 magnesium alloy. Mater. Des. 2006, 27, 1174–1179. [Google Scholar] [CrossRef]
- Wu, C.S.; Zhang, Z.; Cao, F.H.; Zhang, L.J.; Zhang, J.Q.; Cao, C.N. Study on the anodizing of AZ31 magnesium alloys in alkaline borate solutions. Appl. Surf. Sci. 2007, 253, 3893–3898. [Google Scholar] [CrossRef]
- Chai, L.; Yu, X.; Yang, Z.; Wang, Y.; Okido, M. Anodizing of magnesium alloy AZ31 in alkaline solutions with silicate under continuous sparking. Corros. Sci. 2008, 50, 3274–3279. [Google Scholar] [CrossRef]
- Brusciotti, F.; Snihirova, D.V.; Xue, H.; Montemor, M.F.; Lamaka, S.V.; Ferreira, M.G.S. Hybrid epoxy–silane coatings for improved corrosion protection of Mg alloy. Corros. Sci. 2008, 67, 82–90. [Google Scholar] [CrossRef]
- Lamaka, S.V.; Montemor, M.F.; Galio, A.F.; Zheludkevich, M.L.; Trindade, C.; Dick, L.F.; Ferreira, M.G.S. Novel hybrid sol–gel coatings for corrosion protection of AZ31B magnesium alloy. Electrochim. Acta 2007, 53, 4773–4783. [Google Scholar] [CrossRef]
- Montemor, M.F.; Trabelsi, W.; Lamaka, S.V.; Yasakau, K.A.; Zheludkevich, M.L.; Bastos, A.C.; Ferreira, M.G.S. The synergistic combination of bis-silane and CeO2·ZrO2 nanoparticles on the electrochemical behaviour of galvanised steel in NaCl solutions. Electrochim. Acta 2008, 53, 5913–5922. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Huang, S.; Zhou, X. Study of the corrosion behavior and the corrosion films formed on the surfaces of Mg–xSn alloys in 3.5 wt. % NaCl solution. Appl. Surf. Sci. 2014, 317, 1143–1150. [Google Scholar] [CrossRef]
- Arrabal, R.; Pardo, A.; Merino, M.C.; Mohedano, M.; Casajús, P.; Paucar, K.; Garcés, G. Effect of Nd on the corrosion behaviour of AM50 and AZ91D magnesium alloys in 3.5 wt. % NaCl solution. Corros. Sci. 2012, 55, 301–312. [Google Scholar] [CrossRef]
- Rosalbino, F.; Angelini, E.; Negri, S.D.; Saccone, A.; Delfino, S. Effect of erbium addition on the corrosion behaviour of Mg–Al alloys. Intermetallics 2005, 13, 55–60. [Google Scholar] [CrossRef]
- Zhang, J.; Niu, X.; Qiu, X.; Liu, K.; Nan, C.; Tang, D.; Meng, J. Effect of yttrium-rich misch metal on the microstructures, mechanical properties and corrosion behavior of die cast AZ91 alloy. J. Alloys Compd. 2009, 471, 322–330. [Google Scholar] [CrossRef]
- Zhou, X.; Huang, Y.; Wei, Z.; Chen, Q.; Gan, F. Improvement of corrosion resistance of AZ91D magnesium alloy by holmium addition. Corros. Sci. 2006, 46, 4223–4233. [Google Scholar] [CrossRef]
- Chu, P.W.; Marquis, E.A. Linking the microstructure of a heat-treated WE43 Mg alloy with its corrosion behavior. Corros. Sci. 2015, 101, 94–104. [Google Scholar] [CrossRef]
- Gusieva, K.; Davies, C.H.J.; Scully, J.R.; Birbilis, N. Corrosion of magnesium alloys: The role of alloying. Int. Mater. Rev. 2014, 60, 169–194. [Google Scholar] [CrossRef]
- Song, D.; Ma, A.B.; Jiang, J.H.; Lin, P.H.; Yang, D.H.; Fan, J.F. Corrosion behaviour of bulk ultra-fine grained AZ91D magnesium alloy fabricated by equal-channel angular pressing. Corros. Sci. 2011, 53, 362–373. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.C.; Coy, A.E.; Arrabal, R.; Viejo, F.; Matykina, E. Corrosion behaviour of magnesium/aluminium alloys in 3.5 wt. % NaCl. Corros. Sci. 2008, 50, 823–834. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, W.J. Enhanced corrosion resistance of ultrafine-grained AZ61 alloy containingvery fine particles of Mg17Al12 phase. Corros. Sci. 2013, 75, 228–238. [Google Scholar] [CrossRef]
- Ambat, R.; Aung, N.N.; Zhou, W. Evaluation of microstructural effects on corrosion behaviour of AZ91D magnesium alloy. Corros. Sci. 2013, 42, 1433–1455. [Google Scholar] [CrossRef]
- Hadzima, B.; Janeček, M.; Suchý, P.; Müller, J.; Wagner, L. Microstructure and Corrosion Properties of Fine-Grained Mg-Based Alloys. Mater. Sci. Forum 2008, 584–586, 994–999. [Google Scholar] [CrossRef]
- Pardo, A.; Feliu, S., Jr.; Merino, M.C.; Arrabal, R.; Matykina, E. Electrochemical Estimation of the Corrosion Rate of Magnesium/Aluminium Alloys. Int. J. Corros. 2010, 2010, 953850. [Google Scholar] [CrossRef]
- King, A.D.; Birbilis, N.; Scully, J.R. Accurate Electrochemical Measurement of Magnesium Corrosion Rates; a Combined Impedance, Mass-Loss and Hydrogen Collection Study. Electrochim. Acta 2014, 121, 394–406. [Google Scholar] [CrossRef]
- Fontana, M.G. Corrosion Engineering, 3rd ed.; McGraw-Hill: New York, NY, USA, 1986. [Google Scholar]
- Stansbury, E.; Buchanan, R. Fundamentals of Electrochemical Corrosion, 1st ed.; ASM International: Materials Park, OH, USA, 2000. [Google Scholar]
- Kubatík, T.F.; Pala, Z.; Neufuss, K.; Vilémová, M.; Mušálek, R.; Stoulil, J.; Slepička, P.; Chráska, T. Metallurgical bond between magnesium AZ91 alloy and aluminium plasma sprayed coatings. Surf. Coat. Technol. 2015, 282, 163–170. [Google Scholar] [CrossRef]
- Geckili, O.; Bilhan, H.; Bilgin, T.; Anthony von Fraunhofer, J. The effect of urea on the corrosion behavior of different dental alloys. Indian J. Dent. Res. 2012, 23, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.-Y.; Kim, H.J.; Baek, S.-M.; Kim, B.; Sohn, S.-D.; Shin, H.-J.; Jeong, H.Y.; Park, S.H.; Yim, C.D.; You, B.S.; et al. Improved corrosion resistance of extruded Mg–8Sn–1Zn–1Al alloy by microalloying with Mn. Scr. Mater. 2015, 109, 38–43. [Google Scholar] [CrossRef]
- Gu, C.D.; Yan, W.; Zhang, J.L.; Tu, J.P. Corrosion resistance of AZ31B magnesium alloy with a conversion coating produced from a choline chloride—Urea based deep eutectic solvent. Corros. Sci. 2016, 106, 108–116. [Google Scholar] [CrossRef]
- Coy, A.E.; Viejo, F.; Garcia-Garcia, F.J.; Liu, Z.; Skeldon, P.; Thompson, G.E. Effect of excimer laser surface melting on the microstructure and corrosion performance of the die cast AZ91D magnesium alloy. Corros. Sci. 2010, 52, 387–397. [Google Scholar] [CrossRef]



| Alloy | Chemical Composition (wt%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Al | Zn | Mn | Si | Fe | Be | Ni | Cu | Mg | |
| AZ91 | 8.7 | 0.65 | 0.25 | 0.006 | 0.003 | 0.0008 | 0.0006 | 0.0005 | rest |
| Sample | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| EOCP (V) | −1.585 | −1.555 | −1.585 | −1.579 | −1.580 |
| Range of Tafel region (mV) | ~50 | ~50 | ~50 | ~50 | ~50 |
| icorr (µA·cm−2) | 15.0 | 14.2 | 16.5 | 14.5 | 16.7 |
| Sample | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| EOCP (V) | −1.586 | −1.577 | −1.579 | −1.582 | −1.587 |
| Epit (V) | −1.496 | −1.497 | −1.481 | −1.493 | −1.494 |
| Range of Tafel region (mV) | 40 | 30 | 48 | 39 | 43 |
| icorr (µA·cm−2) | 7.7 | 7.0 | 8.6 | 8.2 | 8.6 |
| Sample | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| EOCP (V) | −1.576 | −1.577 | −1.575 | −1.573 | −1.574 |
| Ecorr (V) | −1.551 | −1.552 | −1.550 | −1.550 | −1.549 |
| Epit (V) | −1.511 | −1.518 | −1.524 | −1.522 | −1.515 |
| Range of ACP Tafel region (mV) | −10 | −16 | −24 | −22 | −16 |
| Range of CPC Tafel region (mV) | >50 | >50 | >50 | >50 | >50 |
| icorr (µA·cm−2) | 14.9 | 13.4 | 15.1 | 14.9 | 15.0 |
| Technique | EOCP (V) | Ecorr (V) | icorr (µA·cm−2) |
|---|---|---|---|
| CPC | −1.577 ± 0.011 | −1.577 ± 0.011 1 | 15.4 ± 1.0 |
| APC | −1.582 ± 0.004 | −1.582 ± 0.004 1 | 8.0 ± 0.6 |
| PC | −1.575 ± 0.001 | −1.550 ± 0.001 | 14.7 ± 0.6 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tkacz, J.; Minda, J.; Fintová, S.; Wasserbauer, J. Comparison of Electrochemical Methods for the Evaluation of Cast AZ91 Magnesium Alloy. Materials 2016, 9, 925. https://doi.org/10.3390/ma9110925
Tkacz J, Minda J, Fintová S, Wasserbauer J. Comparison of Electrochemical Methods for the Evaluation of Cast AZ91 Magnesium Alloy. Materials. 2016; 9(11):925. https://doi.org/10.3390/ma9110925
Chicago/Turabian StyleTkacz, Jakub, Jozef Minda, Stanislava Fintová, and Jaromír Wasserbauer. 2016. "Comparison of Electrochemical Methods for the Evaluation of Cast AZ91 Magnesium Alloy" Materials 9, no. 11: 925. https://doi.org/10.3390/ma9110925
APA StyleTkacz, J., Minda, J., Fintová, S., & Wasserbauer, J. (2016). Comparison of Electrochemical Methods for the Evaluation of Cast AZ91 Magnesium Alloy. Materials, 9(11), 925. https://doi.org/10.3390/ma9110925






