An In-Depth Investigation into the Physicochemical, Thermal, Microstructural, and Rheological Properties of Petroleum and Natural Asphalts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Analytical Methods
2.2.1. Elemental Analysis (EA)
2.2.2. Thin-Layer Chromatography with Flame Ionization Detection Procedure (TLC-FID)
2.2.3. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectroscopy (MALDI-TOF-MS)
2.2.4. Fourier Transform Infrared Spectroscopy (FT-IR)
2.2.5. Raman Spectroscopy (RS)
2.2.6. Nuclear Magnetic Resonance Spectroscopy (1H-NMR)
2.2.7. Ultraviolet and Visible Spectroscopy (UV-VIS)
2.2.8. X-ray Diffraction (XRD)
2.2.9. Scanning Electron Microscopy (SEM)
2.2.10. Thermogravimetric Analysis (TGA)
2.2.11. Differential Scanning Calorimetry (DSC)
2.3. Determination of Rheological Properties
3. Results and Discussion
3.1. Elemental Analysis (EA)
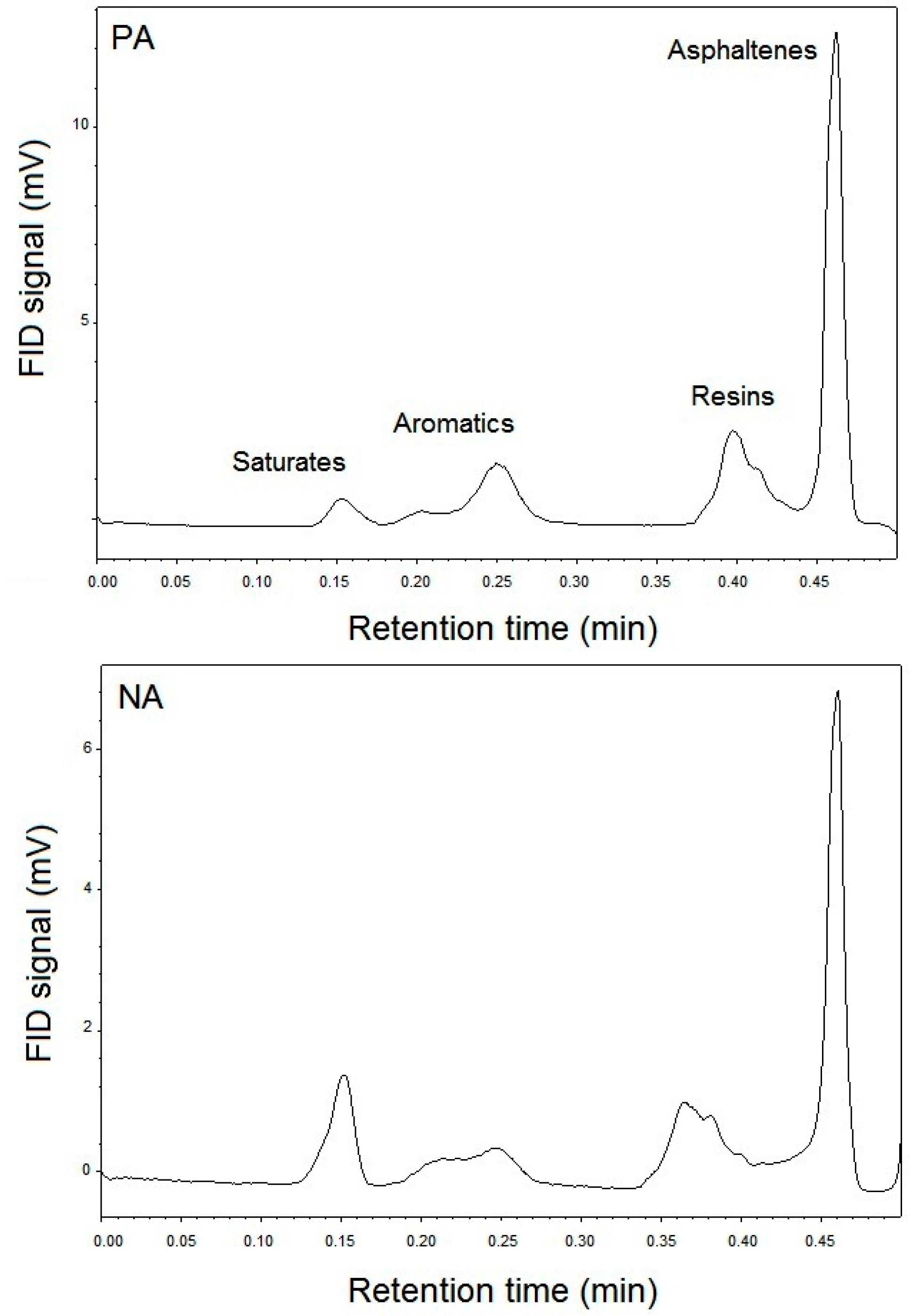
3.2. Thin-Layer Chromatography with Flame Ionization Detection (TLC-FID)
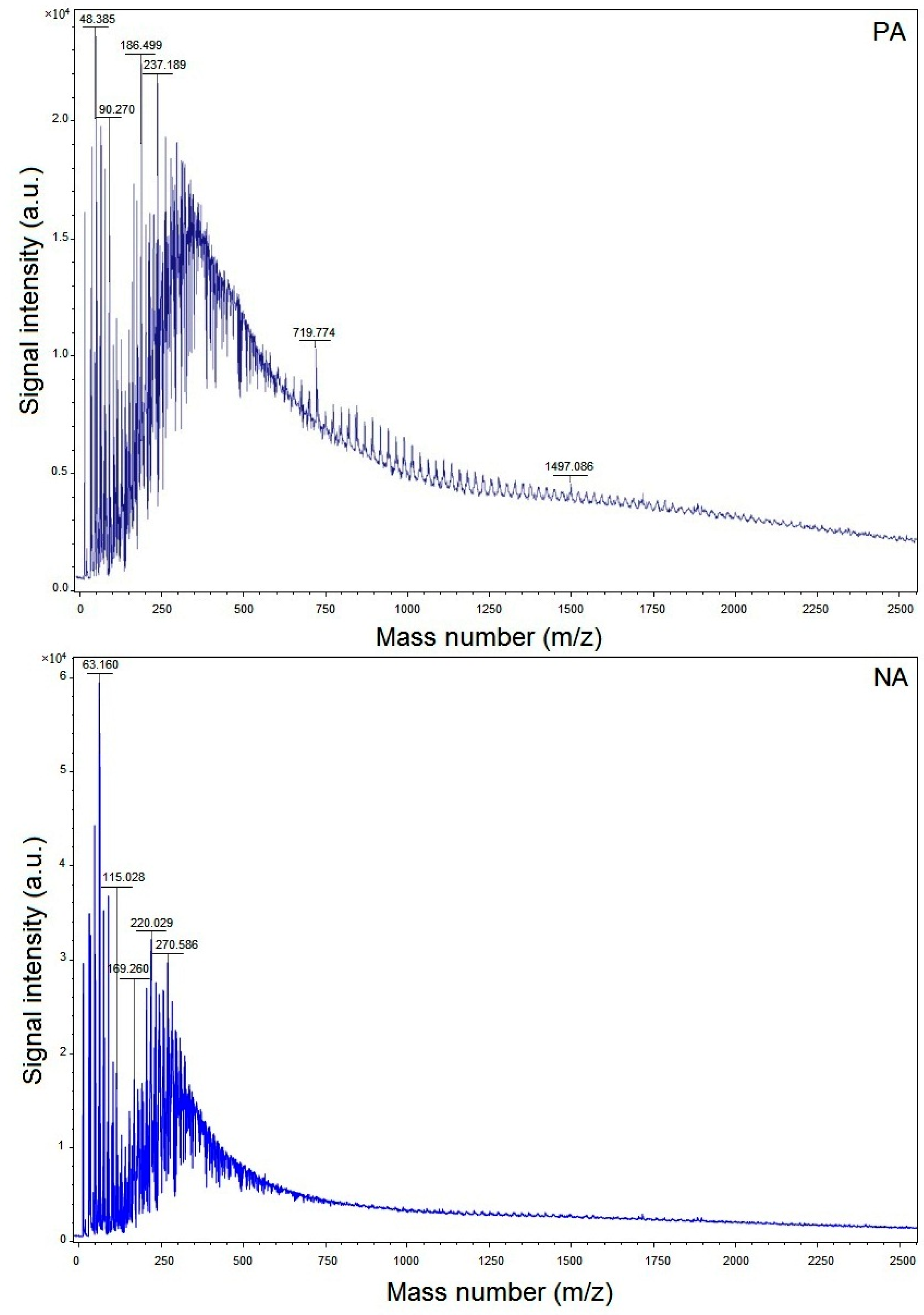
3.3. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectroscopy (MALDI-TOF-MS)
3.4. FT-IR Analysis
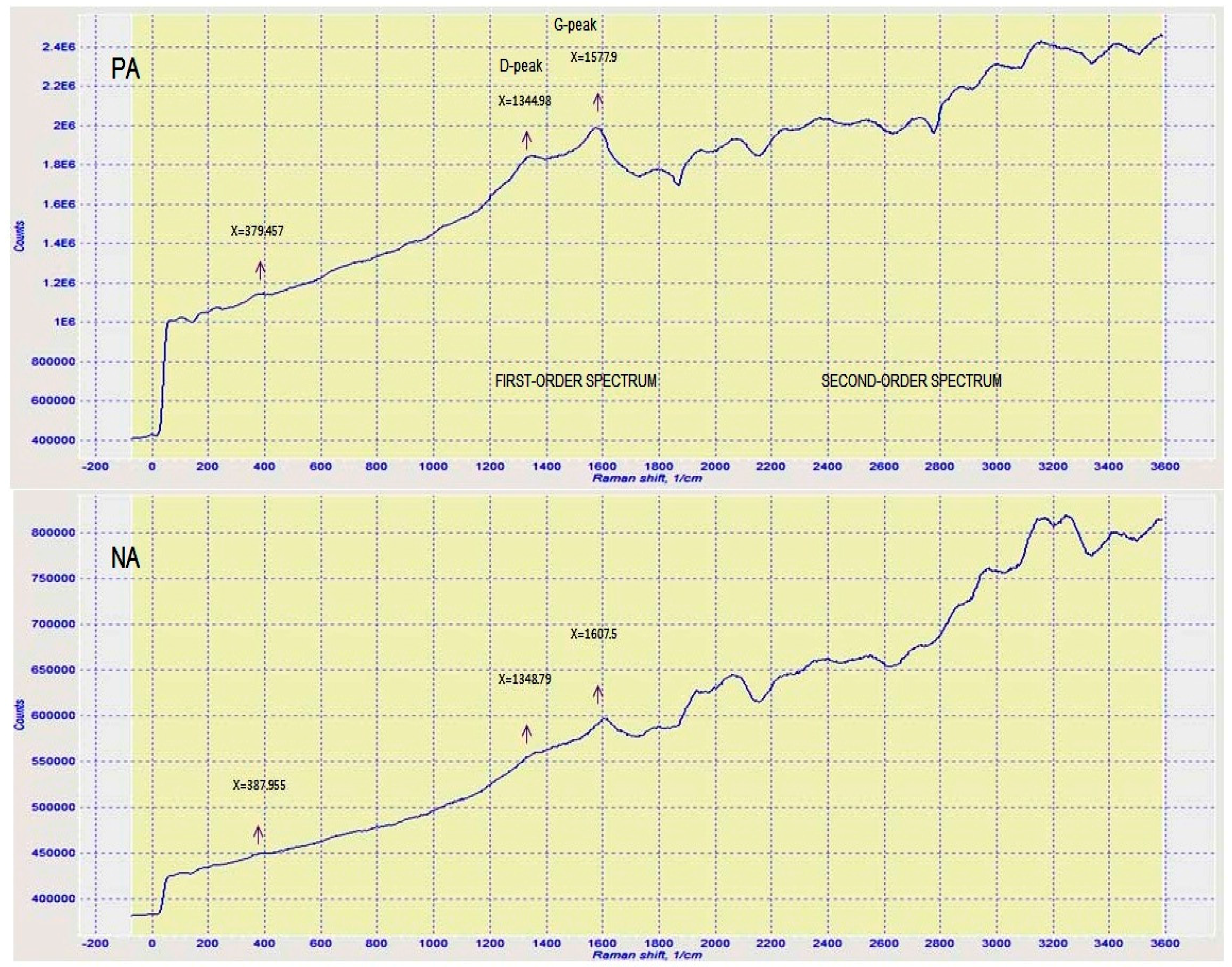
3.5. Raman Spectroscopy (RS)
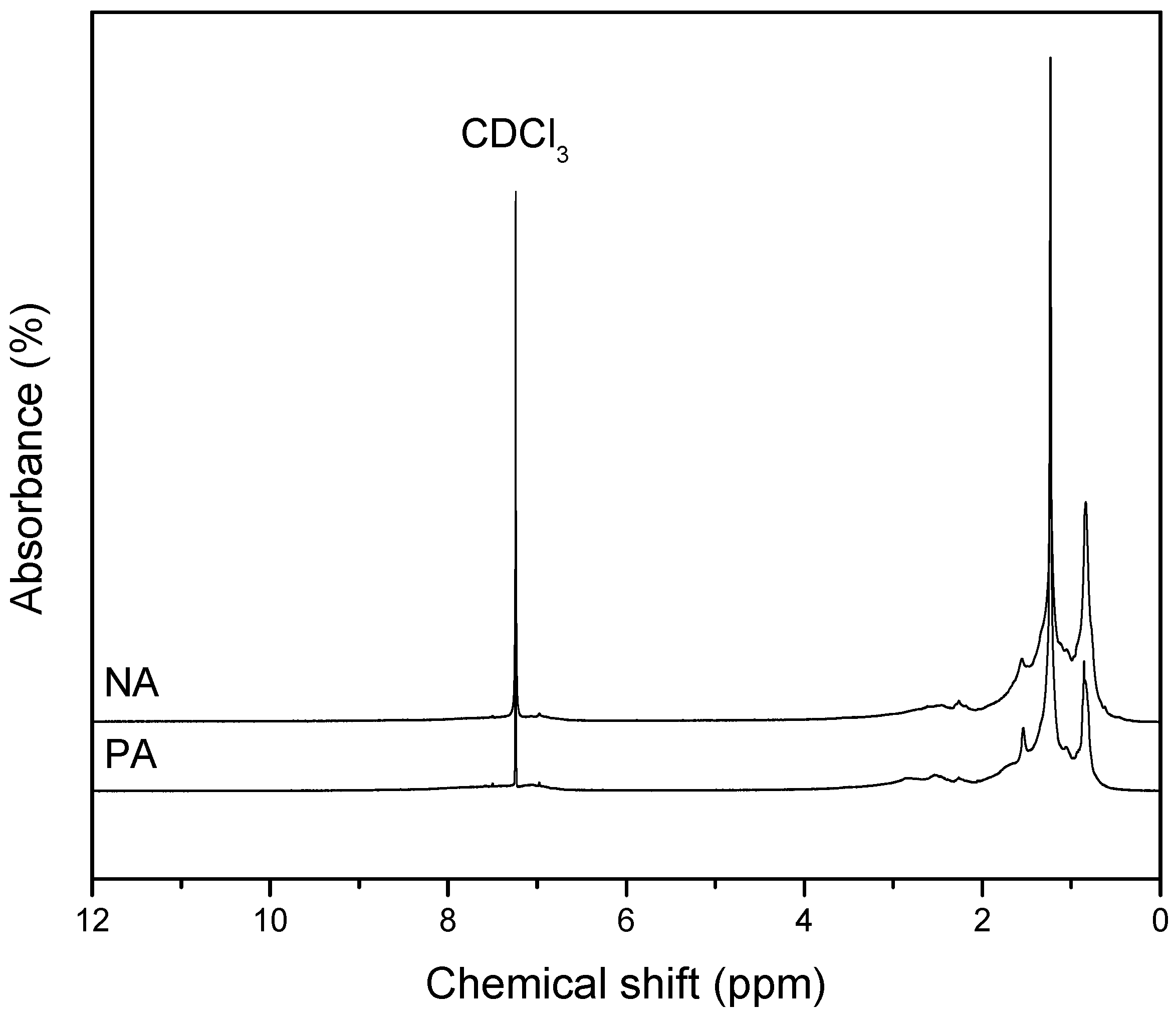
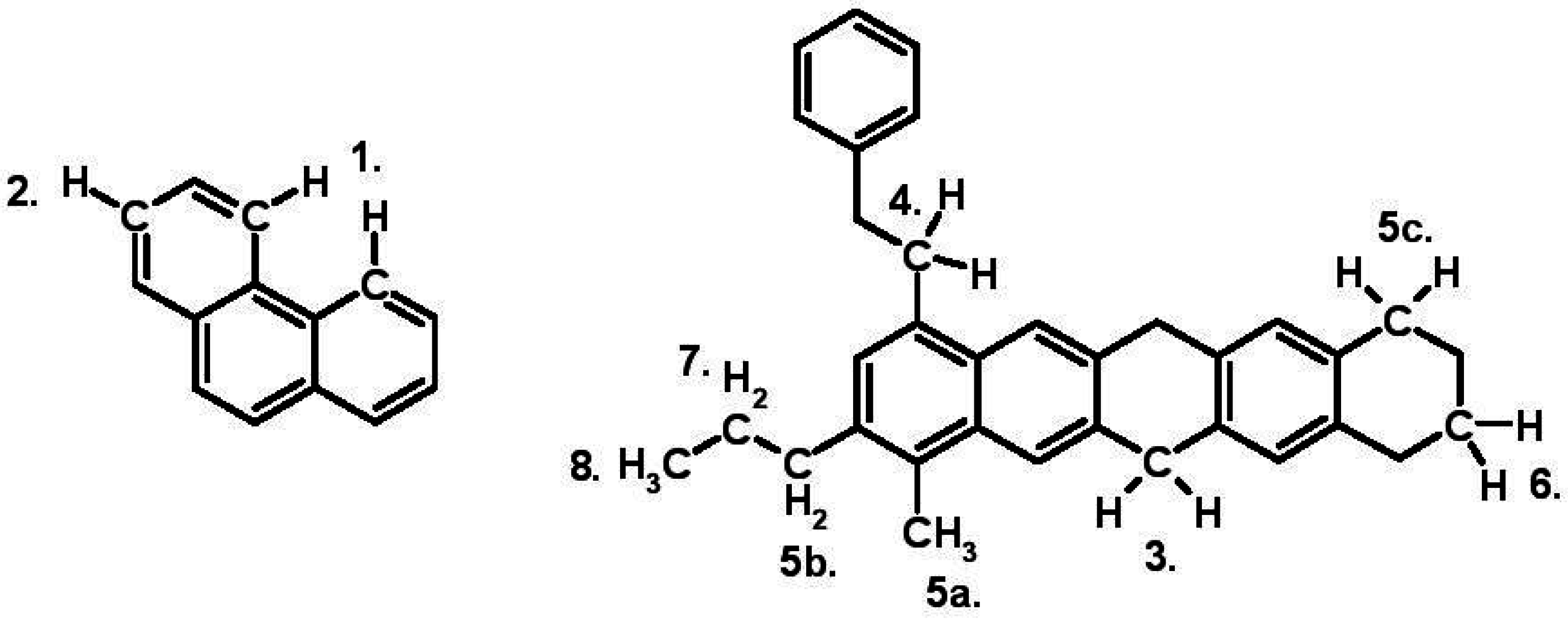
3.6. Nuclear Magnetic Resonance Spectroscopy (1H-NMR)
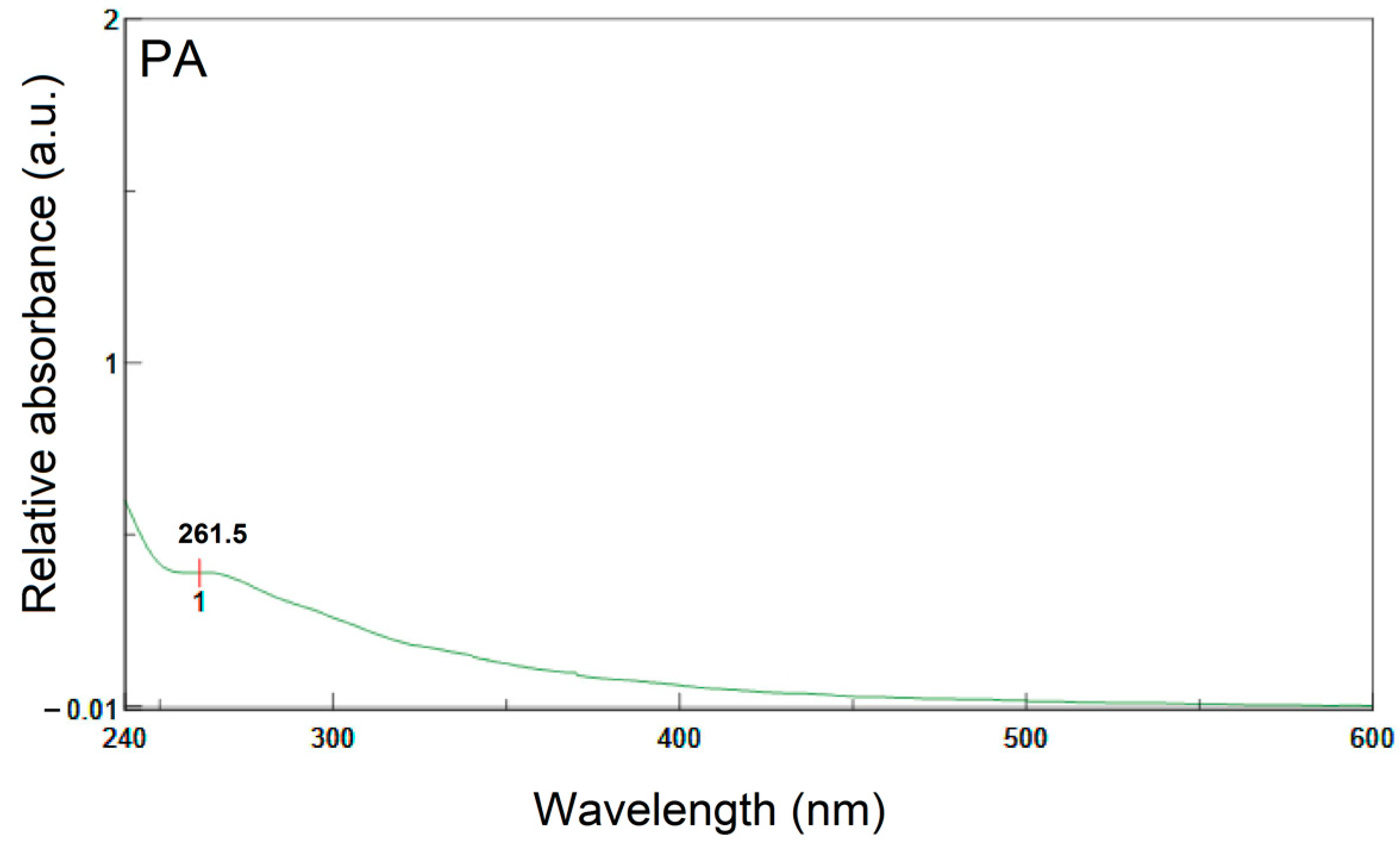
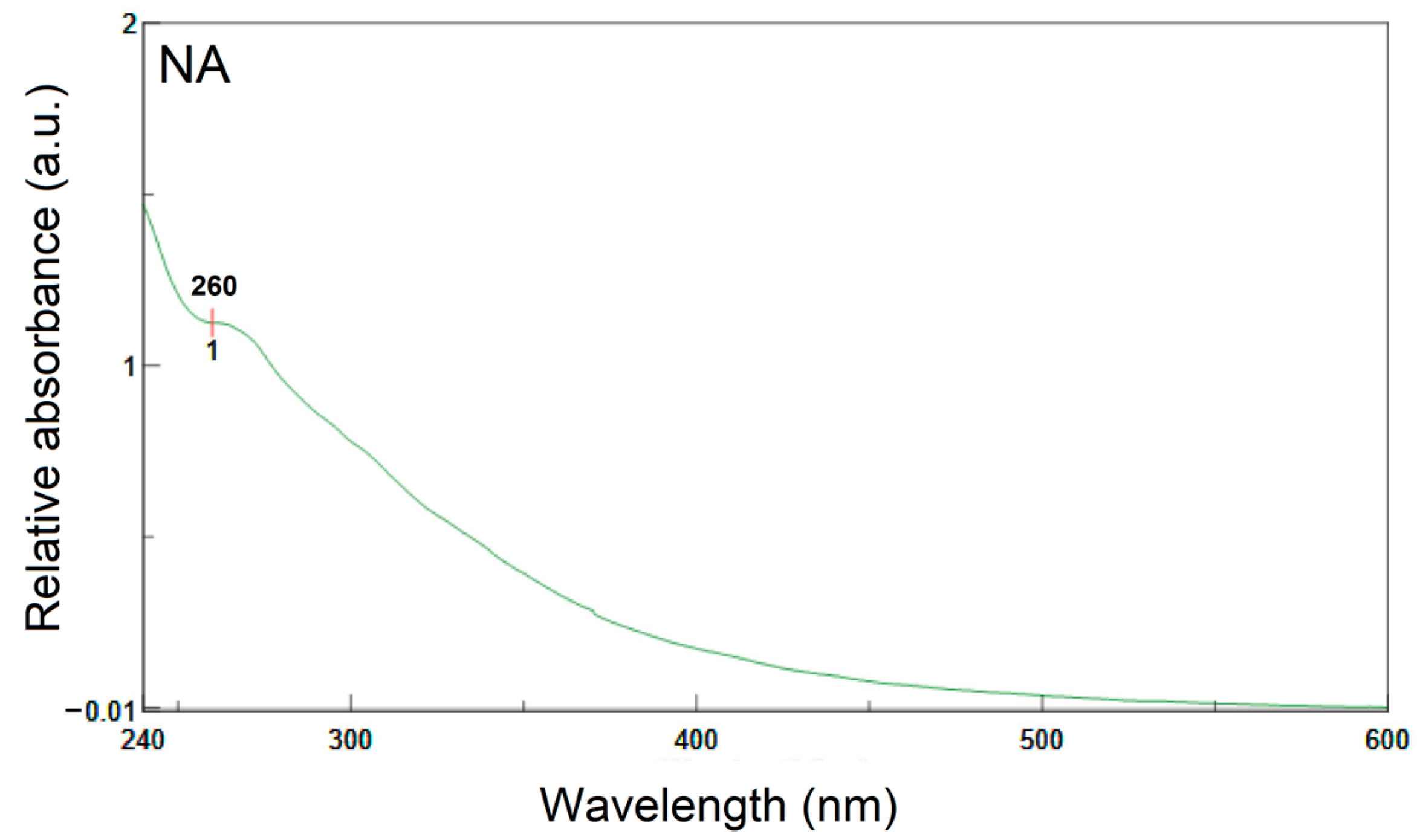
3.7. Ultraviolet and Visible Spectroscopy (UV-VIS)
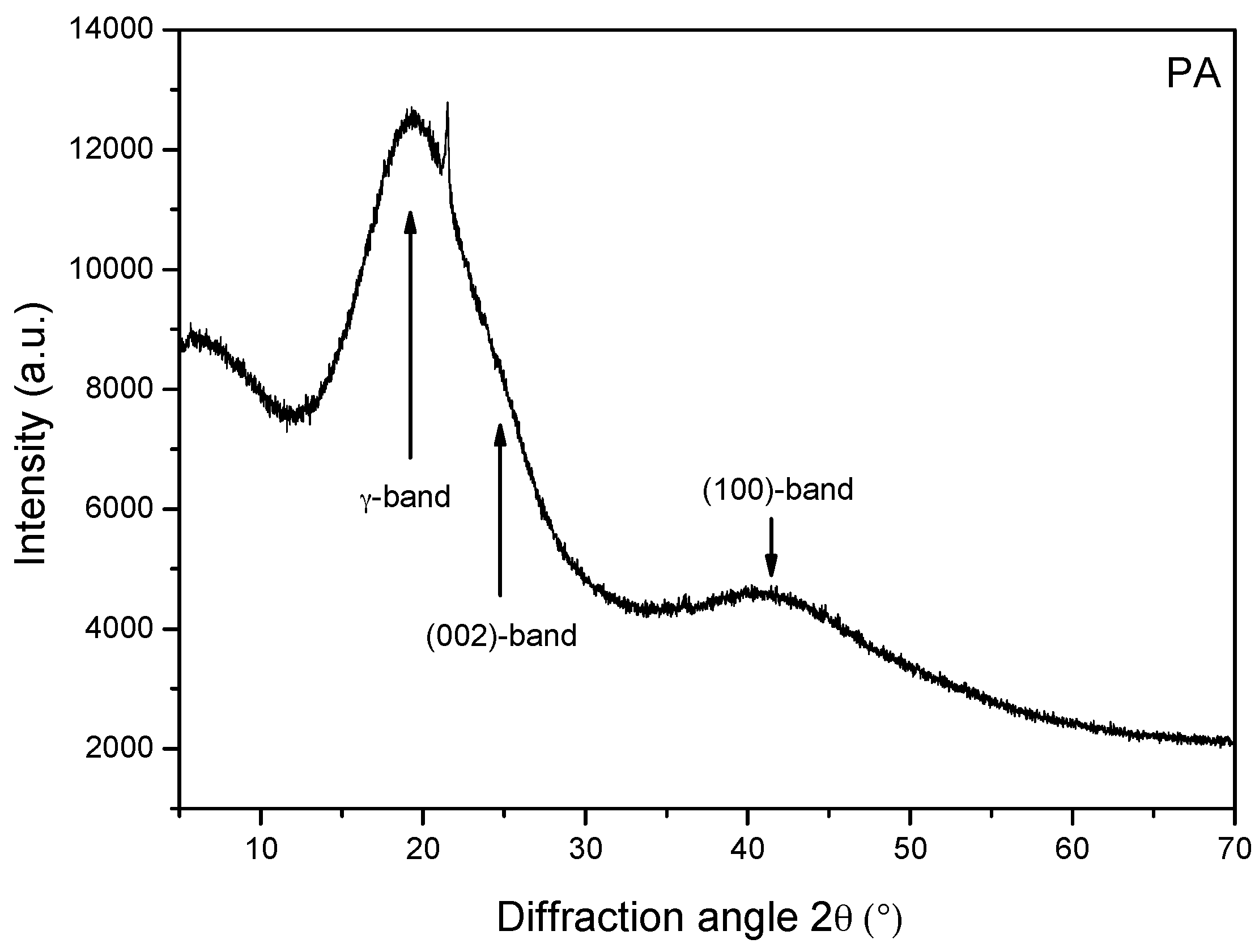
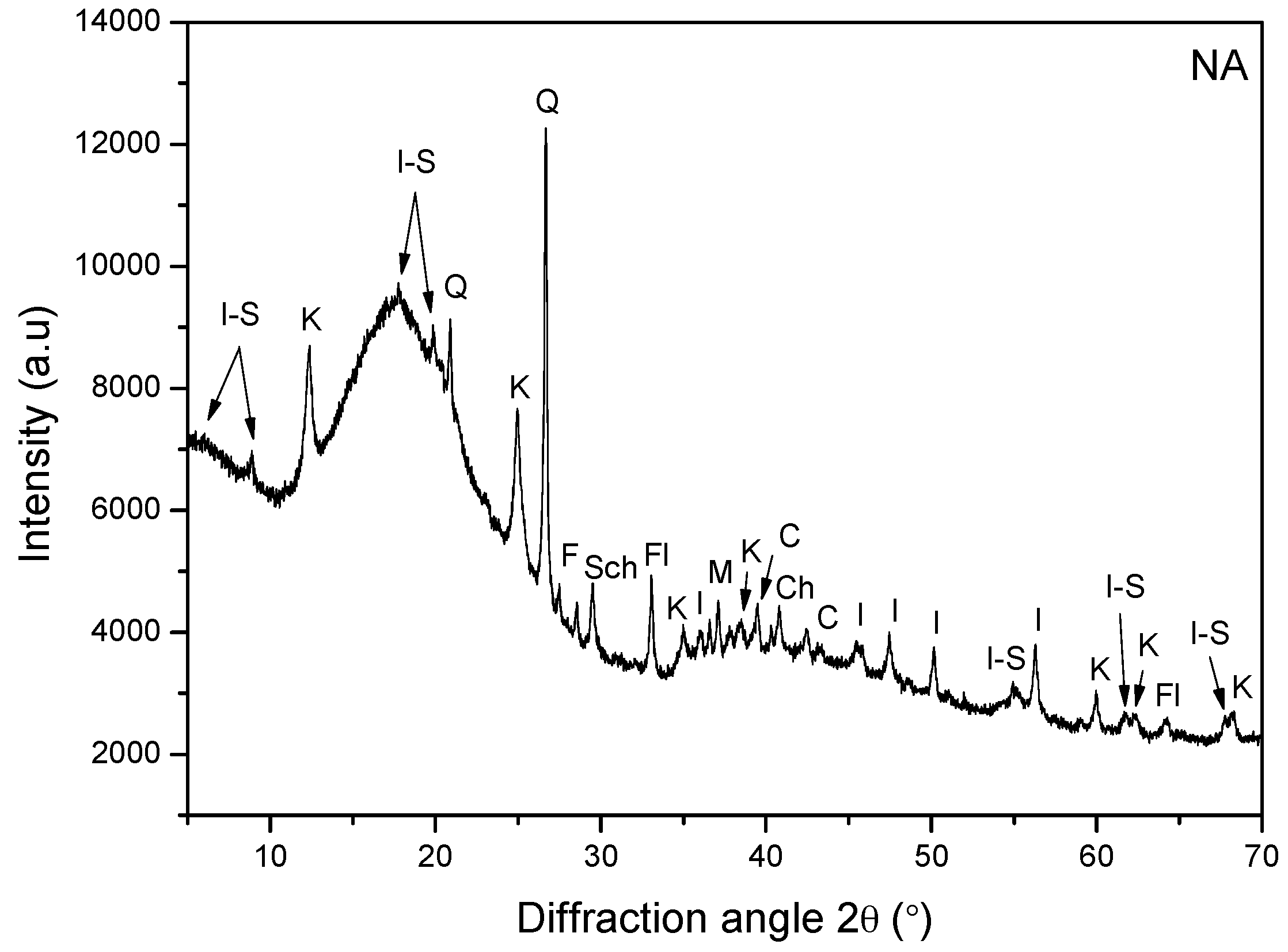
3.8. X-ray Diffraction (XRD)
3.9. Scanning Electron Microscopy (SEM)
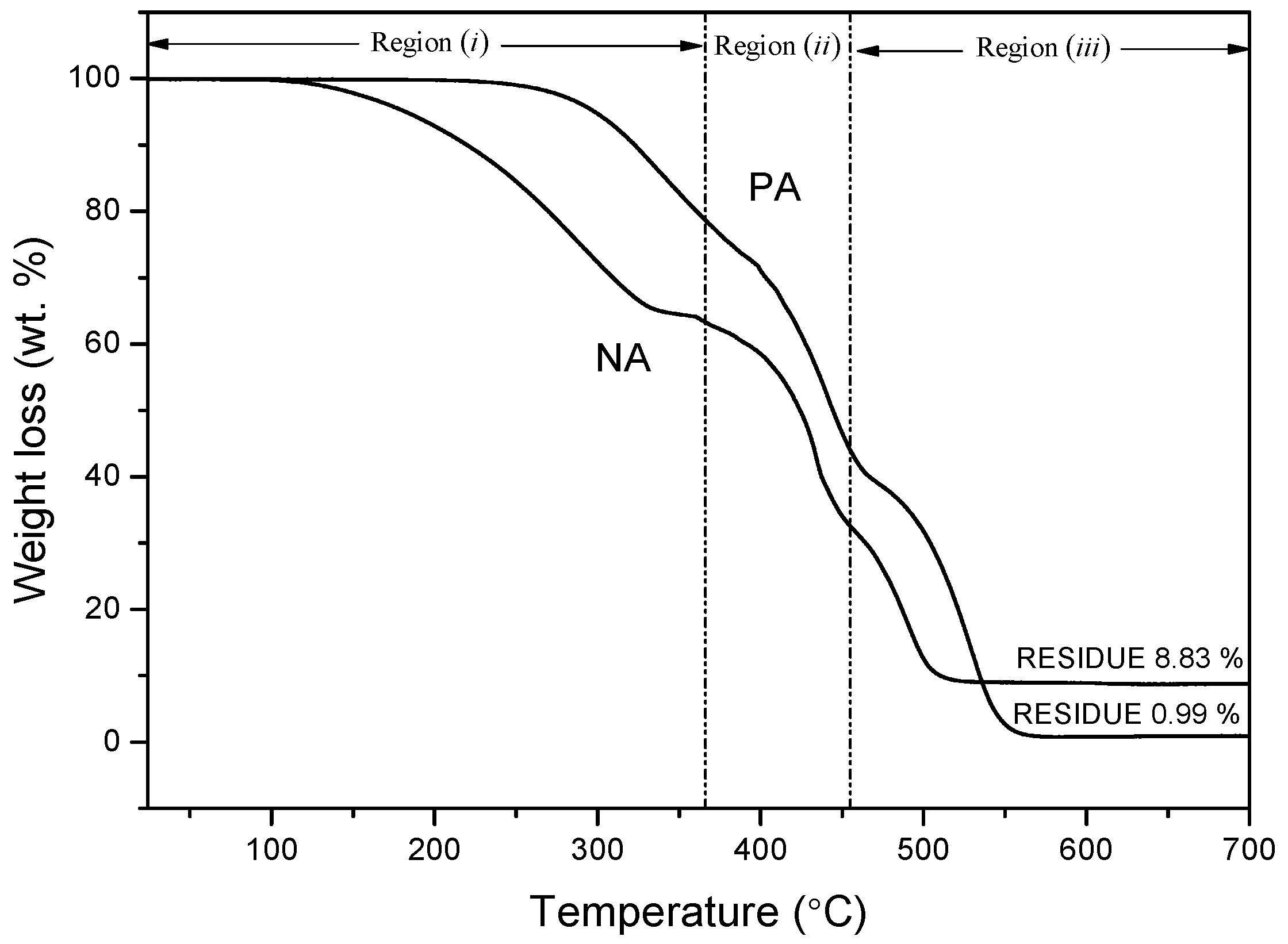
3.10. Thermogravimetric Analysis (TGA)
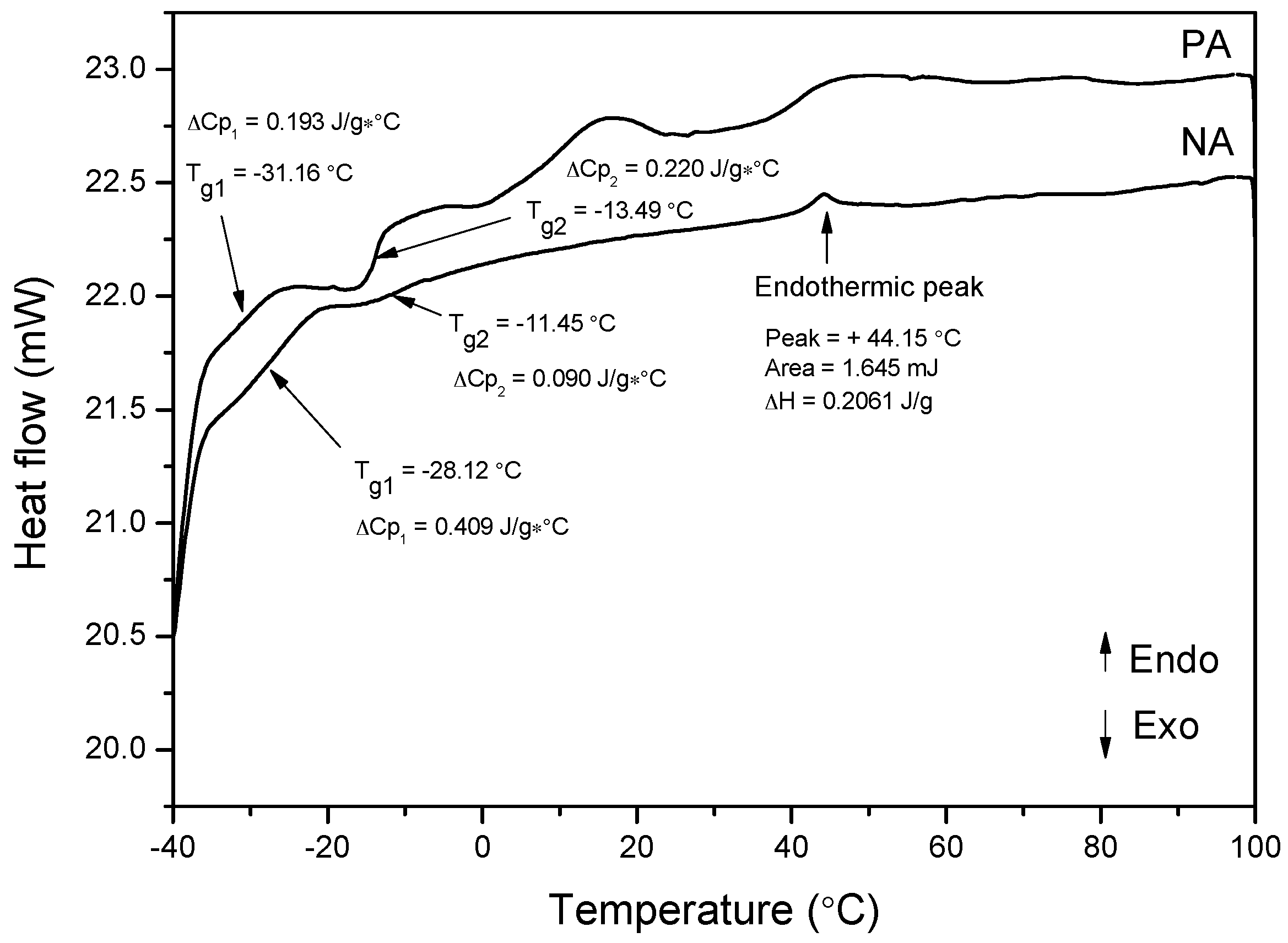
3.11. Differential Scanning Calorimetry (DSC)
3.12. Physical and Rheological Analysis
3.12.1. Penetration
3.12.2. Softening Point
3.12.3. Ductility Test
3.12.4. Viscosity
4. Conclusions
- -
- The results of this study revealed a clear distinction between the elemental compositions of PA and NA.
- -
- The SARA composition and the colloidal index stability were highly affected by the chemical composition of the feedstock and the technology of asphalt production.
- -
- Whereas the average value of the molecular weight of PA was found to be 328 Da, it was 252 Da for NA. Therefore, the compound types existent in the asphalts have diverse molecular weight distribution.
- -
- UV-Vis spectroscopy indicates the existence of more highly conjugated systems in NA than in PA.
- -
- NA has a lower carbon aromaticity than PA as observed from the analyses of FT-IR, Raman, and NMR tests. Also, NA has comprehensive alkylated aromatic structures with large amount of oxygenates.
- -
- Whereas clay minerals are present in NA, they are absent in PA. This result was obtained from the FT-IR, XRD, and SEM analyses.
- -
- Thermogravimetric analysis performed in air atmosphere indicated that the PA sample was thermally more stable than the NA sample.
- -
- When performing the differential scanning calorimetry of NA and PA, the former binder demonstrated a narrow endothermic peak at about 40~50 °C, while the latter displayed two broad endothermic bands at 14.50 °C and 48.5 °C. This revelation corresponds to the melting of different crystallizing wax fractions present in the asphalt.
- -
- Due to the different exhibition of chemical composition and structure in both the asphalts, PA and NA performed differently in the rheological tests.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Petersen, J.C. Chemical composition of asphalt as related to asphalt durability. Dev. Pet. Sci. 2000, 40, 363–399. [Google Scholar]
- Oyekunle, L.O. Influence of chemical composition on the physical characteristics of paving asphalts. Pet. Sci. Technol. 2007, 25, 1401–1414. [Google Scholar] [CrossRef]
- Hardin, J.C. Physical Properties of Asphalt Cement Binders; ASTM International: Philadelphia, PA, USA, 1995; p. 231. [Google Scholar]
- Oyekunle, L.O. Certain relationships between chemical composition and properties of petroleum asphalts from different origin. Oil Gas Sci. Technol. Rev. IFP 2006, 61, 433–441. [Google Scholar] [CrossRef]
- Ecker, A. The application of iatroscan-technique for analysis of bitumen. Pet. Coal 2001, 43, 51–53. [Google Scholar]
- Lin, Q.; Li, T.; Ji, Y.; Wang, W.; Wang, X. Study of the modification of coal-tar pitch with p-methyl benzaldehyde. Fuel 2005, 84, 177–182. [Google Scholar] [CrossRef]
- Jiang, T.; Zhao, Q.; Yin, H. Synthesis of highly stabilized mesoporous molecular sieves using natural clay as raw material. Appl. Clay Sci. 2007, 35, 155–161. [Google Scholar] [CrossRef]
- Zhang, F.; Yu, J.; Han, J. Effects of thermal oxidative ageing on dynamic viscosity, TG/DTG, DTA, and FTIR of SBS- and SBS/sulfur-modified asphalts. Constr. Build. Mater. 2011, 25, 129–137. [Google Scholar] [CrossRef]
- Shoute, L.C.T.; Schmidt, K.J.; Hall, R.H.; Webb, M.A.; Rifai, S.; Abel, P.; Arboleda, P.H.; Savage, A.; Bulmer, J.T.; Loppnow, G.R. UV raman spectroscopy of oil sands-derived bitumen and commercial petroleum products. Appl. Spectrosc. 2002, 56, 1308–1313. [Google Scholar] [CrossRef]
- Zhou, Q.; Xiao, X.; Pan, L.; Tian, H. The relationship between micro-Raman spectral parameters and reflectance of solid bitumen. Int. J. Coal Geol. 2014, 121, 19–25. [Google Scholar] [CrossRef]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cançado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2006, 9, 1276–1291. [Google Scholar] [CrossRef] [PubMed]
- Guillén, M.D.; Díaz, C.; Blanco, C.G. Characterization of coal tar pitches with different softening points by 1H-NMR: Role of the different kinds of protons in the thermal process. Fuel Process. Technol. 1998, 58, 1–15. [Google Scholar] [CrossRef]
- Abbas, O.; Rebufa, C.; Dupuy, N.; Permanyer, A.; Kister, J.; Azevedo, D.A. Application of chemometric methods to synchronous UV fluorescence spectra of petroleum oils. Fuel 2006, 85, 2653–2661. [Google Scholar] [CrossRef]
- Babu, V.S.; Farinash, L.; Seehra, M.S. Carbon in diesel particulate matter: Structure, microwave absorption, and oxidation. J. Mater. Res. 1995, 10, 1075–1078. [Google Scholar] [CrossRef]
- Babu, V.S.; Seehra, M.S. Modeling of disorder and X-ray diffraction in coal-based graphitic carbons. Carbon 1996, 34, 1259–1265. [Google Scholar] [CrossRef]
- Lewis, I.C. Chemistry of pitch carbonization. Fuel 1987, 66, 1527–1531. [Google Scholar] [CrossRef]
- Aske, N.; Kallevik, H.; Sjöblom, J. Determination of saturate, aromatic, resin, and asphaltenic (SARA) components in crude oils by means of infrared and near-infrared spectroscopy. Energ. Fuel 2001, 15, 1304–1312. [Google Scholar] [CrossRef]
- Guo, A.; Zhang, X.; Wang, Z. Simulated delayed coking characteristics of petroleum residues and fractions by thermogravimetry. Fuel Process. Technol. 2008, 89, 643–650. [Google Scholar] [CrossRef]
- Kok, M.V.; Karacan, C.O. Behavior and effect of SARA fractions of oil during combustion. SPE Reserv. Eval. Eng. 2000, 3, 380–385. [Google Scholar] [CrossRef]
- Gonçalves, M.L.A.; Teixeira, M.A.G.; Pereira, R.C.L.; Mercury, R.L.P.; Matos, J.R. Contribution of thermal analyses for characterization of asphaltenes from Brazilian crude oil. J. Therm. Anal. Calorim. 2001, 64, 697–706. [Google Scholar] [CrossRef]
- Masson, J.F.; Polomark, G.; Collins, P. Glass transitions and amorphous phases in SBS-bitumen blends. Thermochim. Acta 2005, 436, 96–100. [Google Scholar] [CrossRef]
- Lesueur, D. The colloidal structure of bitumen: Consequences on the rheology and the mechanisms of bitumen modification. Adv. Colloid Interface 2009, 145, 42–82. [Google Scholar] [CrossRef] [PubMed]
- Das, P.K.; Kringos, N.; Wallqvist, V.; Birgisson, B. Micromechanical investigation of phase separation in bitumen by combining atomic force microscopy with differential scanning results. Road Mater. Pavement 2013, 14, 25–37. [Google Scholar] [CrossRef]
- Department of the Army. Materials Testing Field Manual (FM) 5-472; Net Reprint: Washington, DC, USA, 1999. [Google Scholar]
- Akbulut, H. Rheological properties of cellulose fibre modified bitumen. Eng. Sci. Technol. 2011, 14, 59–66. [Google Scholar]
- Chakroborty, P.; Das, A. Principles of Transportation Engineering; Prentice-Hall of India Private Limited: New Delhi, India, 2003; p. 536. [Google Scholar]
- Kim, Y. Modeling of Asphalt Concrete, 1st ed.; McGraw-Hill Professional: New York, NY, USA, 2009; p. 460. [Google Scholar]
- US Army Corps of Engineers. Hot-Mix Asphalt Paving, AC 150/5370−14B, Appendix 1; Library of Congress: Washington, DC, USA, 2000.



| Elements and Ratios | Asphalt | |
|---|---|---|
| PA | NA | |
| Carbon, wt % | 87.66 | 79.01 |
| Hydrogen, wt % | 10.14 | 9.11 |
| Nitrogen, wt % | 0.44 | 0.95 |
| Sulfur, wt % | 6.29 | 0.72 |
| Oxygen, wt % | 0.29 | 1.38 |
| H/C, atomic ratio | 1.38 | 1.38 |
| Organic matter, wt % | 99.01 | 91.17 |
| Inorganic matter, wt % | 0.99 | 8.83 |
| Asphalt | Saturates wt % | Aromatics wt % | Resins wt % | Asphaltenes wt % | IC † |
|---|---|---|---|---|---|
| PA | 4.40 | 18.97 | 23.55 | 53.07 | 1.35 |
| NA | 14.17 | 13.92 | 23.36 | 48.52 | 1.68 |
| Asphalt | Hydrogen Distribution, % | |||
|---|---|---|---|---|
| Hα | Hβ | Hγ | Har | |
| PA | 15.72 | 66.13 | 18.15 | 10.81 |
| NA | 13.75 | 56.97 | 29.28 | 8.21 |
| Chemical Shift Range (ppm) | Symbol | Description |
|---|---|---|
| 9.5–8.36 | Har2 | Aliphatic hydrogens in sterically hindered positions, highly pericondensed polycyclic aromatic compounds (PACs), next to heteroatoms and some hydrogens joined to nitrogen |
| 8.36–6.3 | Har1 | All other aromatic hydrocarbons |
| 4.5–3.69 | HF | Aliphatic hydrogens in methylene groups α to two aromatic rings |
| 3.69–3.0 | HA | Aliphatic hydrogens in methylene groups α to an aromatic ring and β to another |
| 3.0–2.0 | Hα1 | Aliphatic hydrogens in methyl or methylene groups α to an aromatic ring which can also be attached in γ position or further to another or the same aromatic ring |
| 2.0–1.6 | Hβ2 | Alicyclic hydrogens in β position to an aromatic ring |
| 1.6–1.0 | Hβ1 | Aliphatic hydrogens in methyl or methylene groups β to an aromatic ring |
| 1.0–0.5 | Hγ | Aliphatic hydrogens in methyl or methylene groups γ to an aromatic ring |
| Asphaltic Samples | Tonset (°C) | Toffset (°C) | Tmax (°C) | Residue at 700 °C (wt %) |
|---|---|---|---|---|
| PA | 286.63 | 548.28 | 333.30/441.62/546.62 | 0.99 |
| NA | 189.96 | 504.99 | 286.63/433.30/489.96 | 8.83 |
| Property | Asphalt | |
|---|---|---|
| PA | NA | |
| Penetration at 25 °C, 1/10 mm (dmm) | 63.33 | 147.00 |
| Softening point, (°C) | 49.00 | 40.00 |
| Ductility at 25 °C, (cm) | >140 | >140 |
| Viscosity at 135 °C, (Pa·s) | 0.50 | 0.35 |
| Viscosity at 180 °C, (Pa·s) | 0.10 | 0.08 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nciri, N.; Kim, J.; Kim, N.; Cho, N. An In-Depth Investigation into the Physicochemical, Thermal, Microstructural, and Rheological Properties of Petroleum and Natural Asphalts. Materials 2016, 9, 859. https://doi.org/10.3390/ma9100859
Nciri N, Kim J, Kim N, Cho N. An In-Depth Investigation into the Physicochemical, Thermal, Microstructural, and Rheological Properties of Petroleum and Natural Asphalts. Materials. 2016; 9(10):859. https://doi.org/10.3390/ma9100859
Chicago/Turabian StyleNciri, Nader, Jeonghyun Kim, Namho Kim, and Namjun Cho. 2016. "An In-Depth Investigation into the Physicochemical, Thermal, Microstructural, and Rheological Properties of Petroleum and Natural Asphalts" Materials 9, no. 10: 859. https://doi.org/10.3390/ma9100859
APA StyleNciri, N., Kim, J., Kim, N., & Cho, N. (2016). An In-Depth Investigation into the Physicochemical, Thermal, Microstructural, and Rheological Properties of Petroleum and Natural Asphalts. Materials, 9(10), 859. https://doi.org/10.3390/ma9100859







