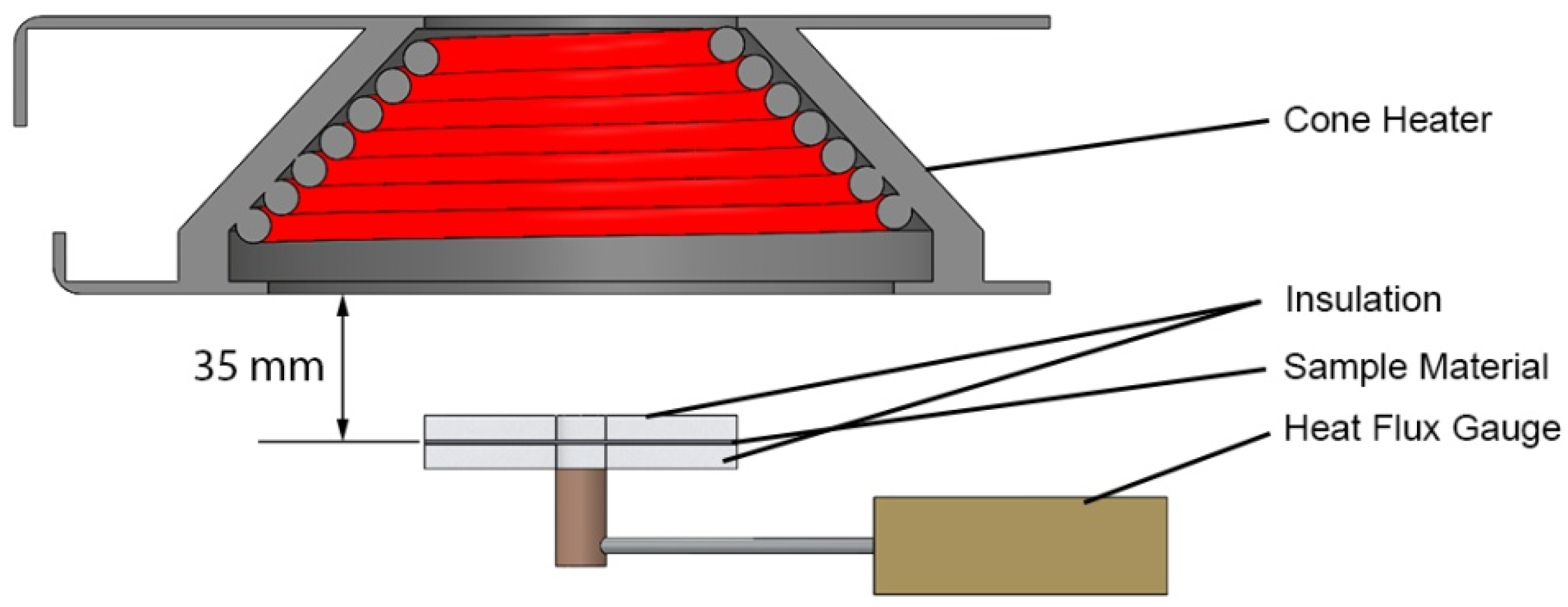
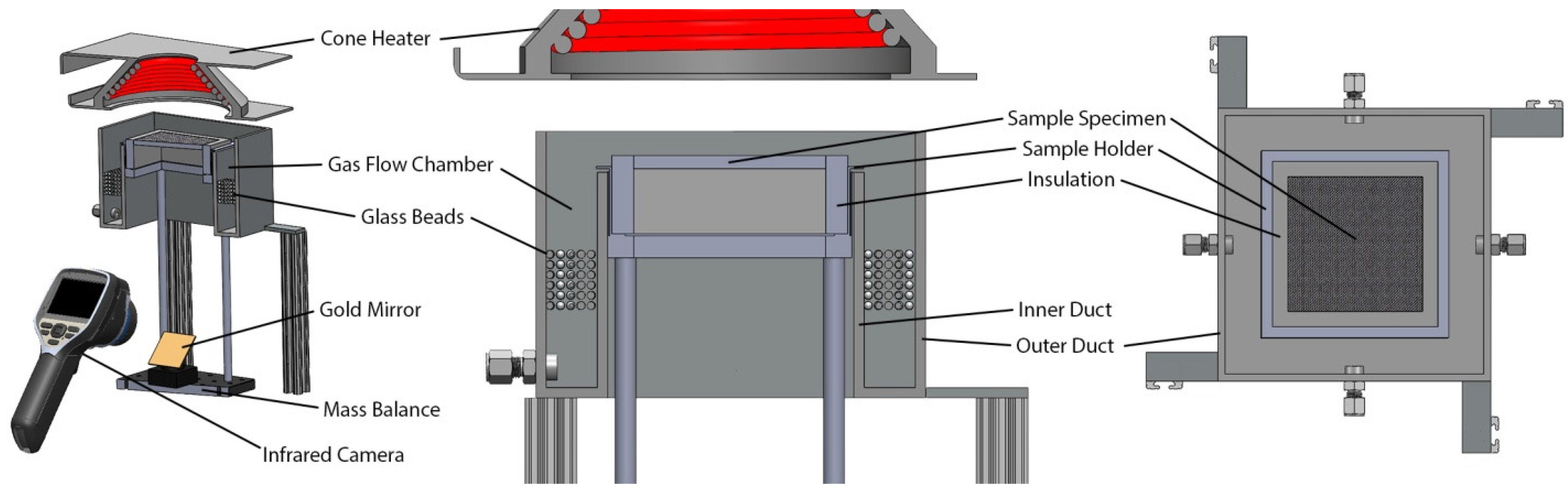
4.1.1. Thermal Degradation Kinetics and Energetics Determination
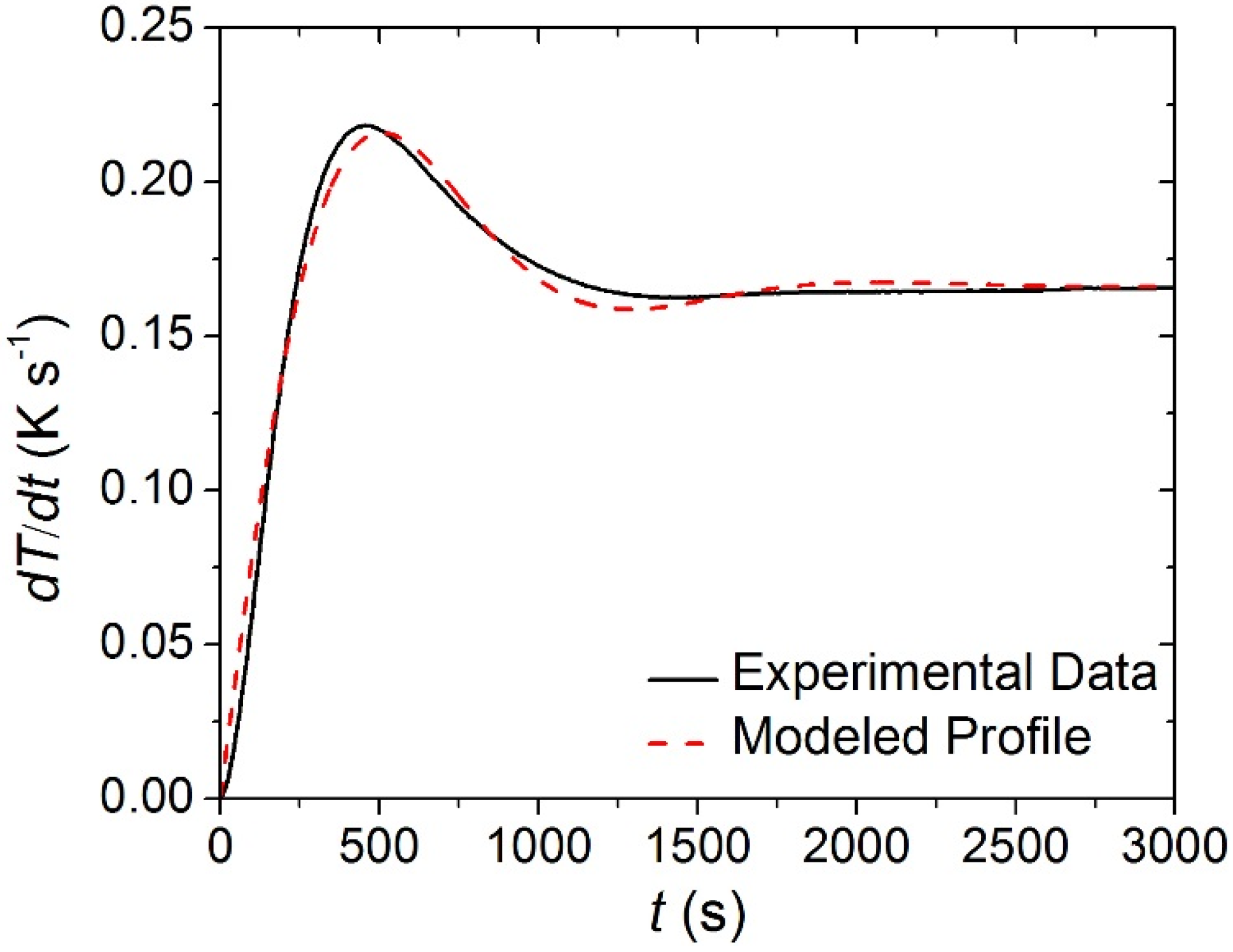
The STA data collected on each of the individual carpet layers was analyzed to determine the kinetics and energetics of the thermal degradation process as well as the heat capacity of the condensed phase components. Though the STA followed a well-defined temperature program, the set point heating rate (0.167 K·s
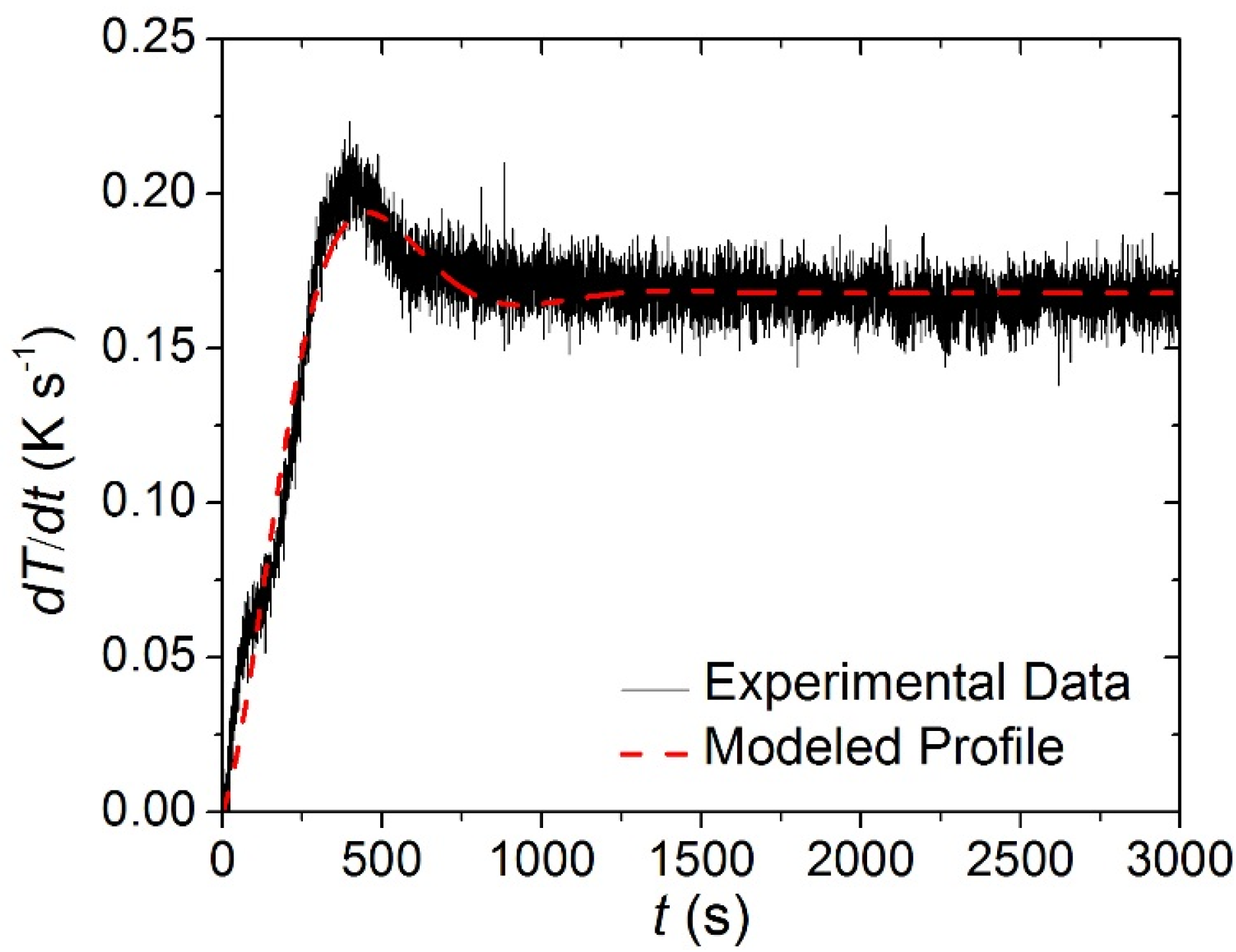
−1) was not achieved instantaneously. Rather, the heating rate measured in each of the tests had reproducible time-dependency that was approximated in ThermaKin by an exponentially-decaying sinusoid, Equation (11). The parameters of this equation were adjusted until it matched the experimental data. The agreement between the experimentally observed and modeled heating rate profiles is evident in
Figure 6.
Figure 6.
Experimentally observed and modeled heating rate histories typical of the Simultaneous Thermal Analysis (STA). The coefficients for Equation (11) that describe the modeled curve are the following: a = 0.166 K s−1, b = 0.0024 s−1, f = 0.004 s−1, g = −0.0623.
Figure 6.
Experimentally observed and modeled heating rate histories typical of the Simultaneous Thermal Analysis (STA). The coefficients for Equation (11) that describe the modeled curve are the following: a = 0.166 K s−1, b = 0.0024 s−1, f = 0.004 s−1, g = −0.0623.
It was assumed that a mechanism with consecutive reactions would be suitable for the face yarn and the base layer. Analysis of the face yarn layer resulted in a mechanism with one phase transition reaction and two thermal degradation reactions. The mechanism determined for the base layer also featured a single phase transition and two thermal degradation reactions. A parallel scheme was assumed for the middle layer because it was known prior to the analysis that the middle layer was composed of at least four distinct polymers that degraded independently. The pyrolysis mechanism for the middle layer included two parallel phase transitions and four parallel thermal degradation reactions. The effective reaction mechanism determined for each layer is provided in
Table 1.
Table 1.
Effective reaction mechanisms for each layer of the carpet composite and the heats of reactions. Positive heats represent endothermic processes.
Table 1.
Effective reaction mechanisms for each layer of the carpet composite and the heats of reactions. Positive heats represent endothermic processes.
| # | Reaction Equation | A (s−1) | E (J·mol−1) | h (J·kg−1) |
|---|
| Face Yarn |
| 1 | | 6.0 × 1038 | 3.80 × 105 | 6.1 × 104 |
| 2 | | 1.0 × 109 | 1.41 × 105 | 5.3 × 104 |
| 3 | | 3.0 × 1014 | 2.30 × 105 | 5.3 × 105 |
| Middle Layer |
| 1 | | 9.0 × 1038 | 3.84 × 105 | 8.0 × 104 |
| 2 | | 1.0 × 1028 | 3.00 × 105 | 6.0 × 104 |
| 3 | | 1.0 × 1012 | 1.55 × 105 | 2.7 × 106 |
| 4 | | 1.0 × 1020 | 2.62 × 105 | 0 |
| 5 | | 5.0 × 108 | 1.42 × 105 | 3.5 × 105 |
| 6 | | 1.0 × 1010 | 1.70 × 105 | 2.0 × 105 |
| Base Layer |
| 1 | | 1.0 × 1021 | 1.72 × 105 | 6.0 × 103 |
| 2 | | 5.0 × 106 | 1.15 × 105 | 4.0 × 104 |
| 3 | | 1.0 × 1016 | 2.58 × 105 | 1.5 × 105 |
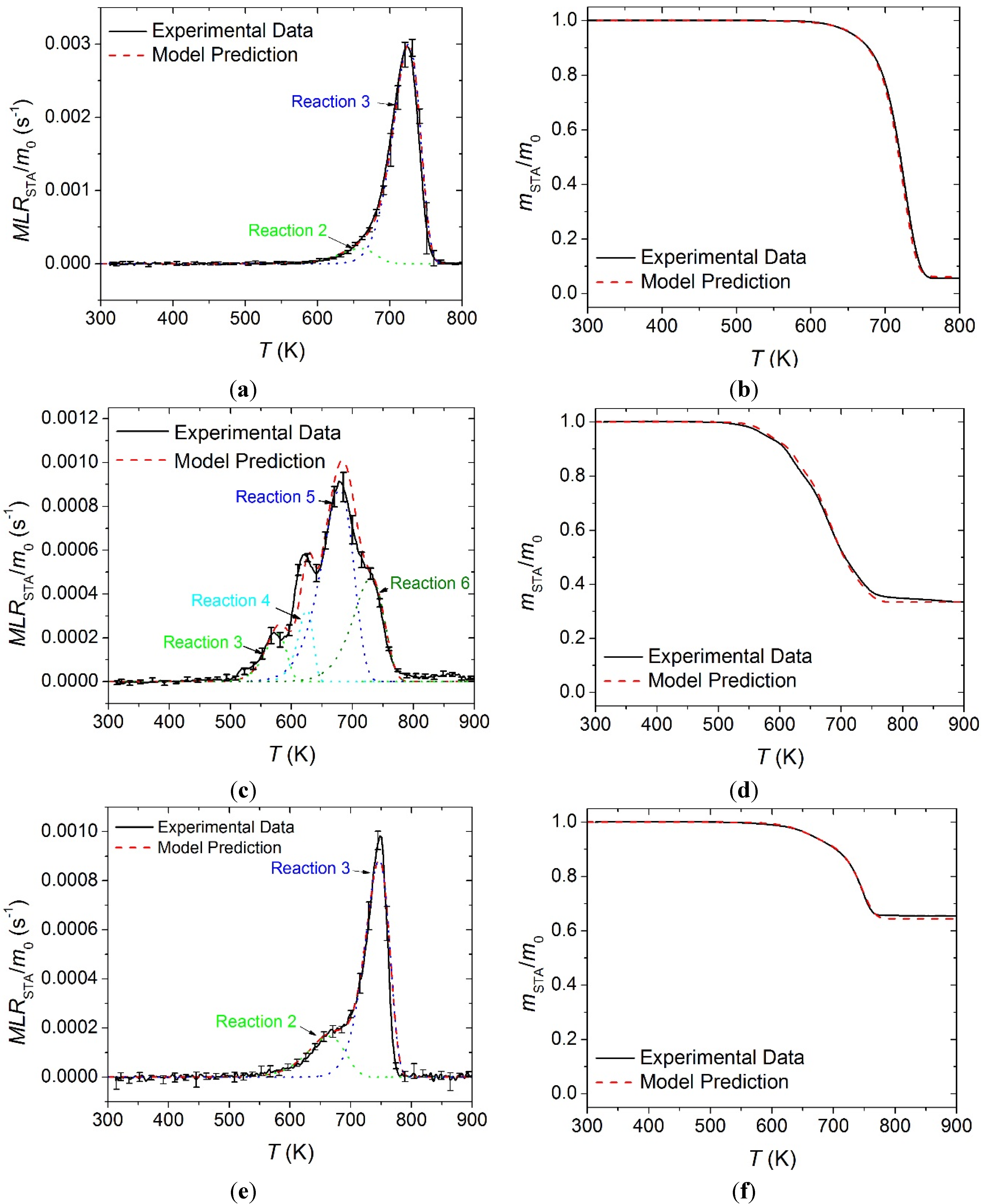
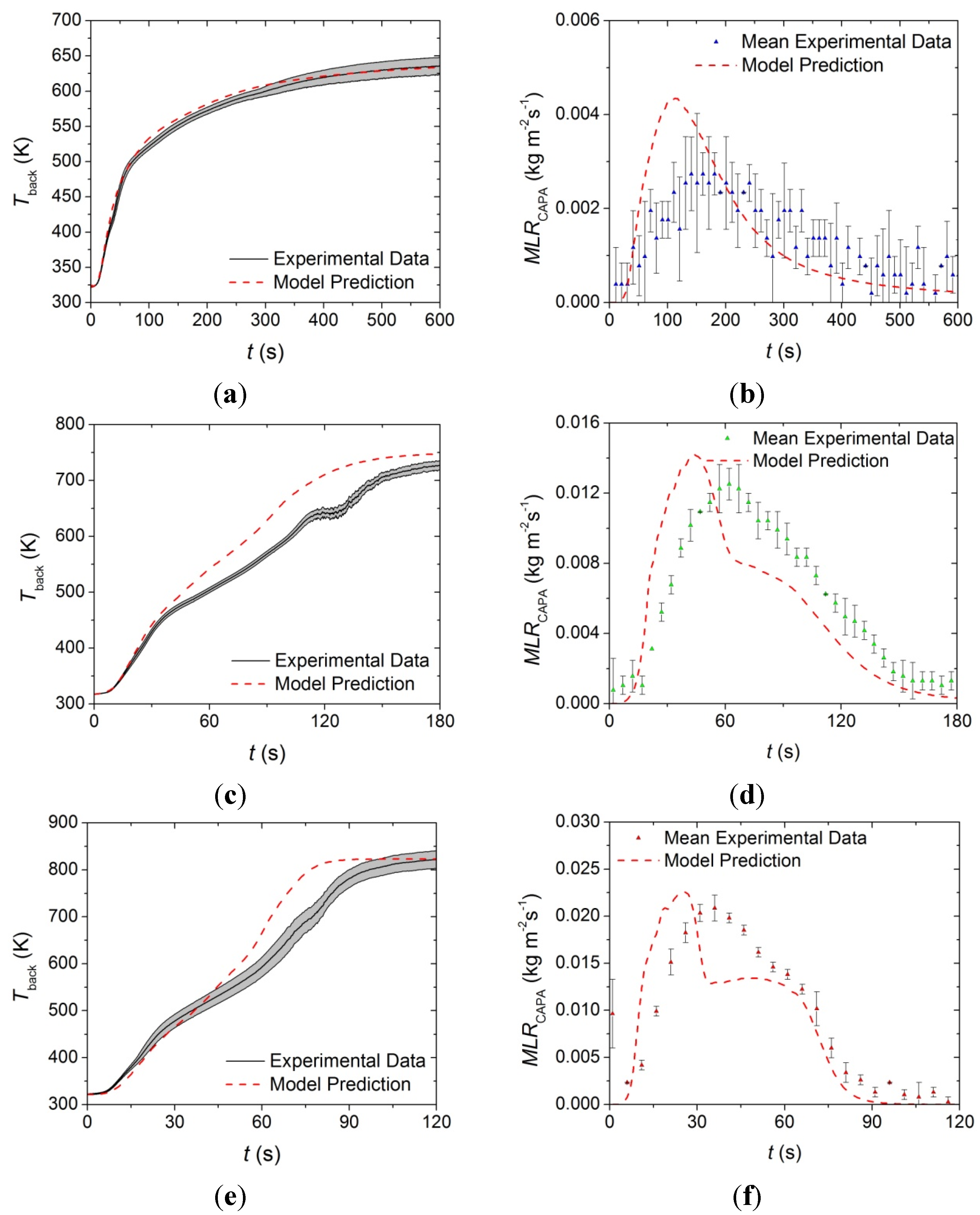
The STA normalized mass and normalized
MLR data for all carpet layers is plotted in
Figure 7 along with the curves predicted by the reaction mechanism shown in
Table 1. In
Figure 7,
m0 indicates initial mass of the sample. In general, the reaction mechanisms determined in this work generated curves that agreed well with the experimental
MLR and total mass curves. All error bars displayed in this work correspond to two standard deviations of the mean.
The heat flow rate data collected in STA tests on each layer were analyzed to determine the temperature-dependent heat capacity for all the virgin carpet components. Two linear temperature-dependent relationships were found for the Face Yarnvirgin and Face Yarnmelt components. The heat capacity of all the Middlevirgin components was adequately described with a single temperature-dependent term. It was impossible to assign a heat capacity value to each individual Middlevirgin component, and all were assigned the same value. The base layer sample melted shortly into the tests, and it proved impossible to determine the heat capacity of the Basevirgin component from the collected heat flow rate data. The heat capacity of the Basemelt component was determined with a linear temperature-dependence and it was assumed that the same expression could adequately describe the heat capacity of the Basevirgin component.
Figure 7.
Normalized mass loss rate (MLR) and normalized mass data collected in STA experiments and model predicted curves for: (a) and (b) face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars indicate two standard deviations of the mean experimental data.
Figure 7.
Normalized mass loss rate (MLR) and normalized mass data collected in STA experiments and model predicted curves for: (a) and (b) face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars indicate two standard deviations of the mean experimental data.
The Face Yarn
char and Middle
char components were characterized by low masses and a porous structure that compromised the thermal contact between the sample and the crucible and yielded unreliable heat flow rate measurements. These char components were assigned a single heat capacity that was measured as the mean value for the chars produced by seven common polymers [
21]. The heat capacity of the Base
char component was determined by conducting independent tests on the char produced from thermal degradation of the base layer sample. The heat capacity of the char did not follow a recognizable functional form, so the arithmetic mean of the data over the entire tested temperature range was defined as the heat capacity of the Base
char component.
The heat capacity of the Face Yarnint. component was defined as the mean between the heat capacities of the Face Yarnmelt component evaluated at 560 K and the Face Yarnchar component. The heat capacity of the Middle3,melt and Middle4,melt components was defined as the mean between the heat capacity of the Middlevirgin components evaluated at 500 K and the Middlechar component. The heat capacity of the Baseint component was defined as the mean between the heat capacities of the Basemelt component evaluated at 600 K and the Basechar component. The heat capacity of the reactant was evaluated at a different temperature for each layer based on the temperature at which the intermediate was produced and subsequently reacted.
The heat capacity of the gaseous volatiles produced during thermal degradation of the carpet samples was assumed to be well approximated by hydrocarbons ranging in length from one to eight carbon atoms in the temperature range of 400 K to 500 K. This resulted in the specific heat capacity of all gaseous volatiles defined as 1800 J·kg
−1·K
−1. The expressions determined for the heat capacity of each component are provided in
Table 2.
Table 2.
Heat capacity values for each component in the carpet composite.
Table 2.
Heat capacity values for each component in the carpet composite.
| Component | c [J kg−1 K−1] | Method |
|---|
| Face Yarnvirgin | 8.2T − 1180 | STA |
| Face Yarnmelt | 3.6T + 580 | STA |
| Face Yarnint. | 2150 | Assumed |
| Face Yarnchar, Middlechar | 1700 | [21] |
| Middle1,virgin, Middle2,virgin, Middle3,virgin, Middle4,virgin | 4.2T | STA |
| Middle3,melt, Middle4,melt | 1900 | Assumed |
| Basevirgin | 2.0T + 1000 | Assumed |
| Basemelt | 2.0T + 1000 | STA |
| Baseint. | 1525 | Assumed |
| Basechar | 850 | STA |
| Face Yarnvolatiles, Middlevolatiles, Basevolatiles | 1800 | Assumed |
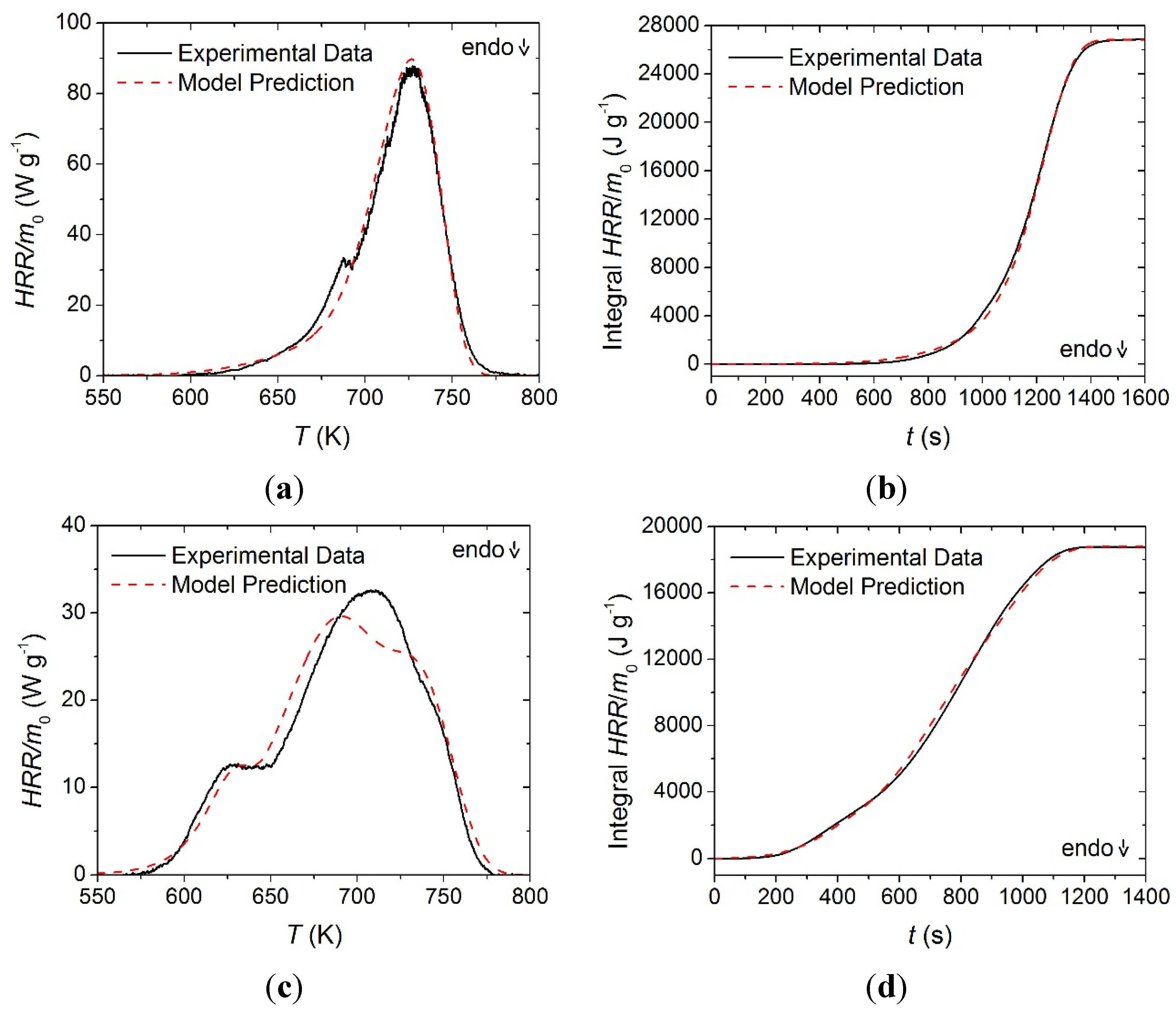
The integral between the experimental heat flow rate curve and the sensible enthalpy baseline was defined as the enthalpy absorbed by the sample undergoing phase transition or thermal degradation. Each heat of reaction was assigned to the reaction that occurred at the temperature range corresponding to the heat flow rate peak. It was difficult to differentiate between the two thermal degradation reactions in the heat flow rate curve for the face yarn and the base layer and the total integral of the peak was divided between the two reactions in approximate proportion to the total mass volatilized in each reaction. The resulting heat flow quantities associated with each reaction are provided in
Table 1. The experimental heat flow rate curves are plotted in
Figure 8 along with the model predictions generated with the parameters summarized in
Table 1 and
Table 2.
Figure 8.
Normalized heat flow rate and integral heat flow rate data collected in STA experiments and model predicted curves for: (a) and (b) face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars indicate two standard deviations of the mean experimental data.
Figure 8.
Normalized heat flow rate and integral heat flow rate data collected in STA experiments and model predicted curves for: (a) and (b) face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars indicate two standard deviations of the mean experimental data.
4.1.2. Heat of Combustion Determination
The MCC data collected on each of the carpet layers at a set heating rate of 0.167 K·s
−1 were analyzed using the degradation kinetics determined from analysis of STA data. The heating rate profile observed in MCC experiments was different than the profile observed in STA experiments, but was adequately described by the form of Equation (11). The mass loss rate was predicted through a simulation adhering to a heating rate profile that approximated the profile observed in MCC experiments. The agreement between the experimentally observed and modeled heating rate profiles is displayed in
Figure 9.
Figure 9.
Experimentally observed and modeled heating rate histories typical of the Microscale Combustion Calorimetry (MCC) experiments conducted in this work. The coefficients for Equation (14) that describe the modeled curve are the following: a = 0.168 K s−1, b = 0.0039 s−1, f = 0.0065 s−1, g = 0.256.
Figure 9.
Experimentally observed and modeled heating rate histories typical of the Microscale Combustion Calorimetry (MCC) experiments conducted in this work. The coefficients for Equation (14) that describe the modeled curve are the following: a = 0.168 K s−1, b = 0.0039 s−1, f = 0.0065 s−1, g = 0.256.
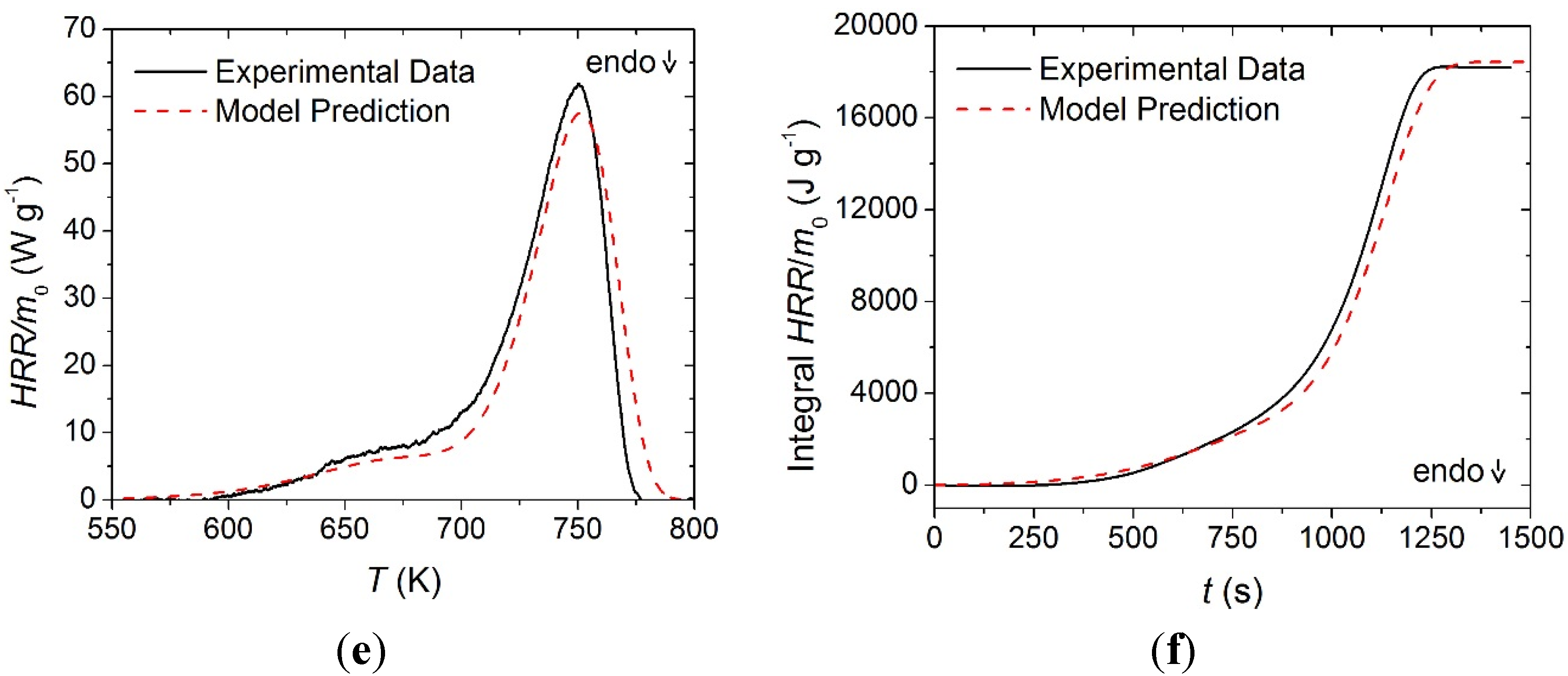
A heat of combustion was assigned to the gaseous components (labeled as volatiles) produced in each of the thermal degradation reactions identified for each material. The mean HRR curve measured for each layer was shifted in temperature to properly align with the MLR curve predicted with the corresponding reaction mechanism. The magnitude of the shift in the data for each layer was based on the principle that heat release required a concurrent mass loss. By applying this shift, the MLR curves agreed well with the rising and falling edges of the HRR curves. The face yarn HRR curve was shifted upward in temperature by 14 K, the middle layer curve was shifted upward in temperature by 15 K, and the base layer was shifted upward in temperature by 5 K.
The offsets between the MCC data and the STA-based kinetics are likely caused by the differences in the heat transport characteristics to and within the crucibles in these two instruments. The current analysis was based on an assumption that the STA provides a more reliable sample temperature control than the MCC. The heats of combustion determined through this analysis are provided in
Table 3 and the experimental and predicted
HRR curves and integral
HRR curves are provided in
Figure 10. The MCC data was highly repeatable for all materials, and error bars were omitted because the magnitude of the scatter was insignificant. Contrary to the convention used to define the heats of reaction, positive values in
Table 3 correspond to exothermic processes.
Table 3.
Effective heat of combustion values for the volatile species released in each reaction. Positive heats of combustion are exothermic.
Table 3.
Effective heat of combustion values for the volatile species released in each reaction. Positive heats of combustion are exothermic.
| Volatile Species | hc (J kg−1) |
|---|
| Face Yarnvolatiles,reaction 2 | 2.4 × 107 |
| Face Yarnvolatiles,reaction 3 | 2.9 × 107 |
| Middlevolatiles,reaction 3 | 0 |
| Middlevolatiles,reaction 4 | 1.6 × 107 |
| Middlevolatiles,reaction 5 | 2.4 × 107 |
| Middlevolatiles,reaction 6 | 5.0 × 107 |
| Basevolatiles,reaction 2 | 3.4 × 107 |
| Basevolatiles,reaction 3 | 5.9 × 107 |
The HRR curve collected on the middle layer was shifted such that the falling edge of the curve corresponded to the falling edge of the MLR curve. After applying this offset in the temperature scale, the first thermal degradation reaction appeared to correspond to a zero magnitude heat release. This led to the conclusion that the volatile species produced in this reaction had no associated heat of combustion. The other values determined for the heats of combustion of volatile species were within a reasonable range for common fuels.
Figure 10.
Normalized heat release rate and integral heat release rate data collected in MCC experiments and model predicted curves for: (a) and (b) the face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars were omitted due to small magnitude scatter.
Figure 10.
Normalized heat release rate and integral heat release rate data collected in MCC experiments and model predicted curves for: (a) and (b) the face yarn layer; (c) and (d) the middle layer; and (e) and (f) the base layer. Error bars were omitted due to small magnitude scatter.
4.1.3. Absorption Coefficient and Emissivity Determination
The reflection loss coefficient,
γ, accounts for reflection of incident radiation at the interface between the material and the gaseous atmosphere. In this work, this coefficient was assumed to be 0.05 for all undegraded carpet layers. This value is the approximate mean of the reflection loss coefficients for common polymers tabulated by Tsilingiris [
26] and supported by the work of Linteris,
et al. [
25] and Försth and Roos [
28].
The data collected in transmitted heat flux measurements on all the sample materials and used to calculate the absorption coefficients are provided in
Table 4. Since the virgin face yarn was observed to melt shortly after the beginning of radiant heating, the absorption coefficient of the melted face yarn was measured instead of the virgin face yarn. The density of the melted face yarn used in the calculation is discussed later in this section.
Table 4.
Measurements used to calculate the absorption coefficient for each virgin and melt component.
Table 4.
Measurements used to calculate the absorption coefficient for each virgin and melt component.
| Layer | | δ (m) | ρ (kg m−3) | κ (m2 kg−1) |
|---|
| Face Yarn Melt | 0.025 | 0.0008 ± 0.0001 | 625 | 7.17 |
| Middle Layer | 0.026 | 0.0013 ± 0.0001 | 582 | 4.69 |
| Middle Layer | 0.020 | 0.0016 ± 0.0001 | 582 | 4.09 |
| Base Layer | 0.010 | 0.0010 ± 0.0001 | 1060 | 4.25 |
| Base Layer | 0.005 | 0.0010 ± 0.0001 | 1060 | 4.90 |
Since the radiant flux transmitted through the melted face yarn was measured, the density used to model the Face Yarnmelt component was also used to calculate the absorption coefficient. The densities of the middle layer and the base layer defined in the individual layer models were used to calculate the absorption coefficient of each of those layers. The mean of the individual measurements of the absorption coefficient for each layer was calculated to define the absorption coefficient in the models. Approximate values were assigned to each component based on the transmitted heat flux measurements (7 m2·kg−1 for the face yarn, 4.4 m2·kg−1 for the middle layer, and 4.6 m2·kg−1 for the base layer). The absorption coefficients of the melt and intermediate components were assigned the same absorption coefficient as the virgin component for all layers.
It was observed during gasification tests on the upper layer that the char components were optically dark and appeared to be graphitic. To make the simulations consistent with this observation, the absorption coefficient of the char components was defined such that all radiation was absorbed at the surface of the sample (100 m2·kg−1). The char formed during thermal degradation of the base layer did not appear to be optically dark. The absorption coefficient for the component was assumed to be equal to the absorption coefficient of all other base layer components.
The relationship between the reflection loss coefficient and emissivity of optically thick materials (must add to unity) led to the definition of the emissivity of all of the virgin and melt components in each of the carpet layers as 0.95. Due to observations of the samples in gasification tests on each of the layers, the char components were assumed to have lower emissivities than the virgin components. It was assumed that the char components had high carbon content, and the emissivity of all chars was expected to be similar to graphite. The emissivity of graphite was measured at elevated temperatures as approximately 0.86 [
29] and was defined as the emissivity of the char components. These definitions are consistent with results of a study by Försth and Roos, who conducted experiments on various colors of PVC and vinyl carpets and found that in some instances, the emissivity tended to decrease as the samples degraded [
28]. The emissivity of the intermediate components was defined as the mean value between the virgin components and the char components (
).
4.1.4. Thermal Conductivity Determination
It was observed that the thickness of the face yarn decreased by a factor of approximately five upon melting. This observation was difficult to confirm in gasification tests because the surface of the sample was completely covered with rapidly regenerating bubbles shortly after melting occurred. However, it was reproduced in a furnace with the temperature set to the melting point of the face yarn (approximately 490 K). A small mass of porous char was produced by the face yarn during degradation, though the thickness of the face yarn layer did not change significantly after melting. The density of the Face Yarnmelt component was defined five times larger than the density of the Face Yarnvirgin component, and the density of the Face Yarnint and Face Yarnchar components were defined proportional to the stoichiometric coefficient for the condensed phase product of each reaction to simulate a constant thickness for the layer after melting.
All Middle
virgin components were defined with the same density because it was impossible to identify and separate each individual initial component. Two of the middle layer components underwent phase changes that did not affect the geometry of the sample, so the Middle
melt components were defined with the same density as the Middle
virgin components. The Base
virgin component went through a phase change without the geometry of the layer changing considerably and the density of the
component was defined equivalent to the virgin component density. The overall thickness of the middle layer and the base layer remained approximately constant throughout the CAPA tests. The densities of the Middle
char, Base
int and Base
char components were defined proportional to the associated stoichiometric coefficients in the reaction mechanism to maintain a constant thickness in the simulations. The definitions for the densities of all components are provided in
Table 5.
Table 5.
Full set of thermophysical properties used in the individual upper layer model and base layer model.
Table 5.
Full set of thermophysical properties used in the individual upper layer model and base layer model.
| Component | ρ (kg m−1) | k (W m−1 K−1) | | κ (m2 κg−1) |
|---|
| Face Yarn |
| Face Yarnvirgin | 125 | 0.05 | 0.95 | 7 |
| Face Yarnmelt | 625 | 0.05 | 0.95 | 7 |
| Face Yarnint. | 575 | 0.025 + 6.5 × 10−10T3 | 0.905 | 7 |
| Face Yarnchar | 34.5 | 11 × 10−10T3 | 0.86 | 100 |
| Middle Layer |
| Middle1,virgin, Middle2,virgin, Middle3,virgin, Middle4,virgin, Middle3,melt, Middle4,melt | 582 | 0.05 | 0.95 | 4.4 |
| Middlechar | 194.4 | 11 × 10−10T3 | 0.86 | 100 |
| Base Layer |
| Basevirgin, Basemelt | 1060 | 0.25 – 2.85 × 10−4T | 0.95 | 4.6 |
| Baseint. | 975.2 | 0.125 – 1.425 × 10−4T + 3.5 × 10−10T3 | 0.905 | 4.6 |
| Basechar | 692.4 | 7 × 10−10T3 | 0.86 | 4.6 |
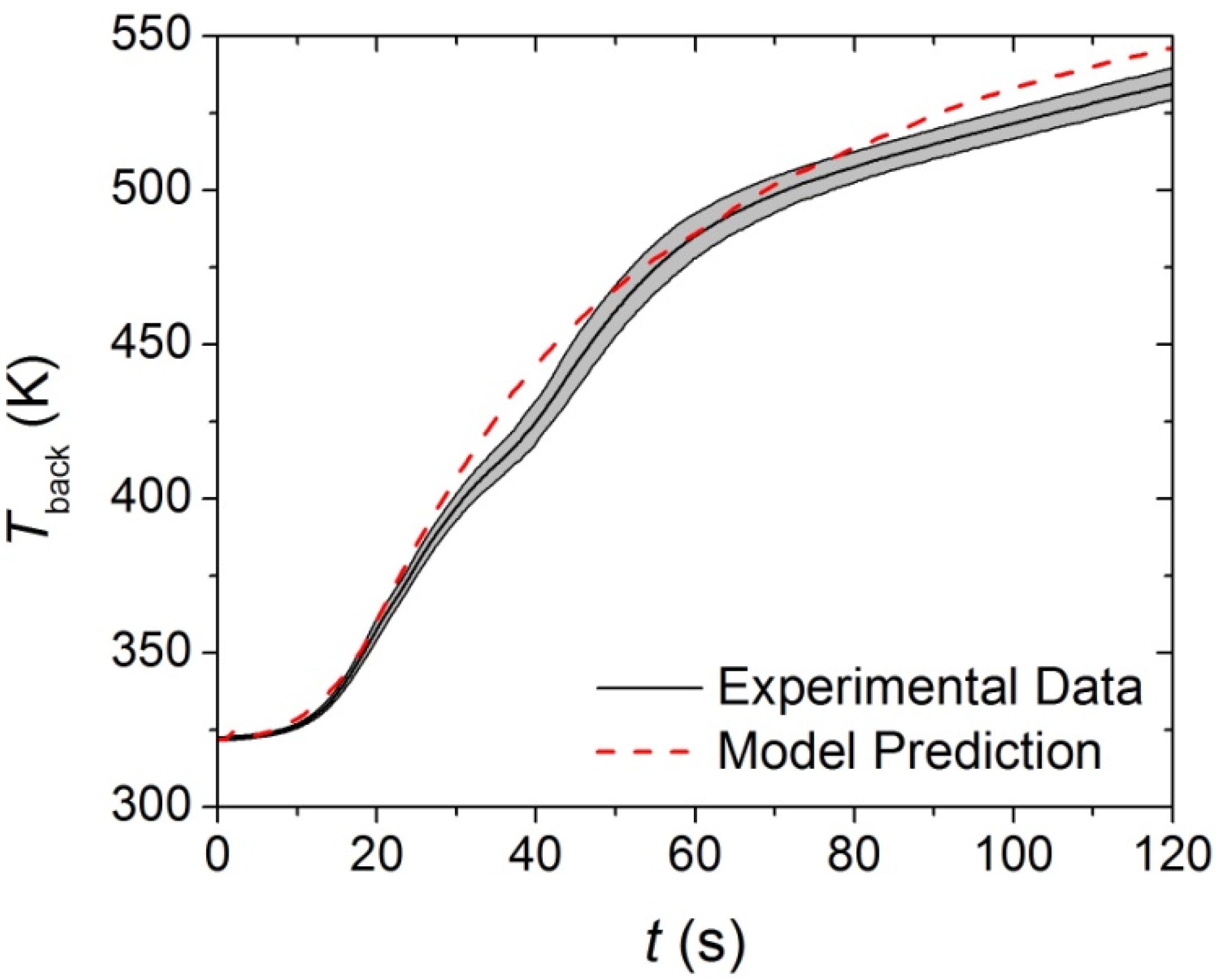
The thermal conductivities of the components in each layer of the carpet composite were the only remaining undefined parameters that affected the pyrolysis model predictions. Inverse analyses were conducted on the
Tback data collected in the CAPA tests using the ThermaKin modeling environment. The initial rise of the
Tback data curve was chosen as the target for the virgin and melt components because these were the only components that affected the
Tback curve early in the tests. The model prediction for the upper layer (face yarn and middle layer) is compared to the experimental data in
Figure 11. The temperature prediction was not sensitive to the thermal transport parameters of the char and intermediate components for the time range that corresponded to the initial rise of
Tback.
It was found that a single, constant value for the thermal conductivity (k = 0.05 W·m−1·K−1) of the Face Yarnvirgin, Middlevirgin, Face Yarnmelt and Middlemelt components was adequate to describe the rising edge of the Tback curve. There was no evidence in the data of a change in the thermal conductivity from the virgin components to the melt components. Though this thermal conductivity value is low for a mixture of solid polymers and is more typical of air at elevated temperatures (650 K), the structure of the carpet upper layer supports a thermal conductivity value lower than the typical range for polymers. The face yarn was made of fibrous filaments woven into a yarn and the majority of the volume of the defined face yarn layer was air (or, in the case of the gasification tests, nitrogen). Furthermore, the face yarn melted shortly after exposure to the cone heater and the melted face yarn was characterized by rapidly regenerating bubbles. The middle layer was structured as a mesh interwoven with face yarn and although the density of the middle layer was larger than the face yarn, gases still made a large contribution to the volume of the layer.
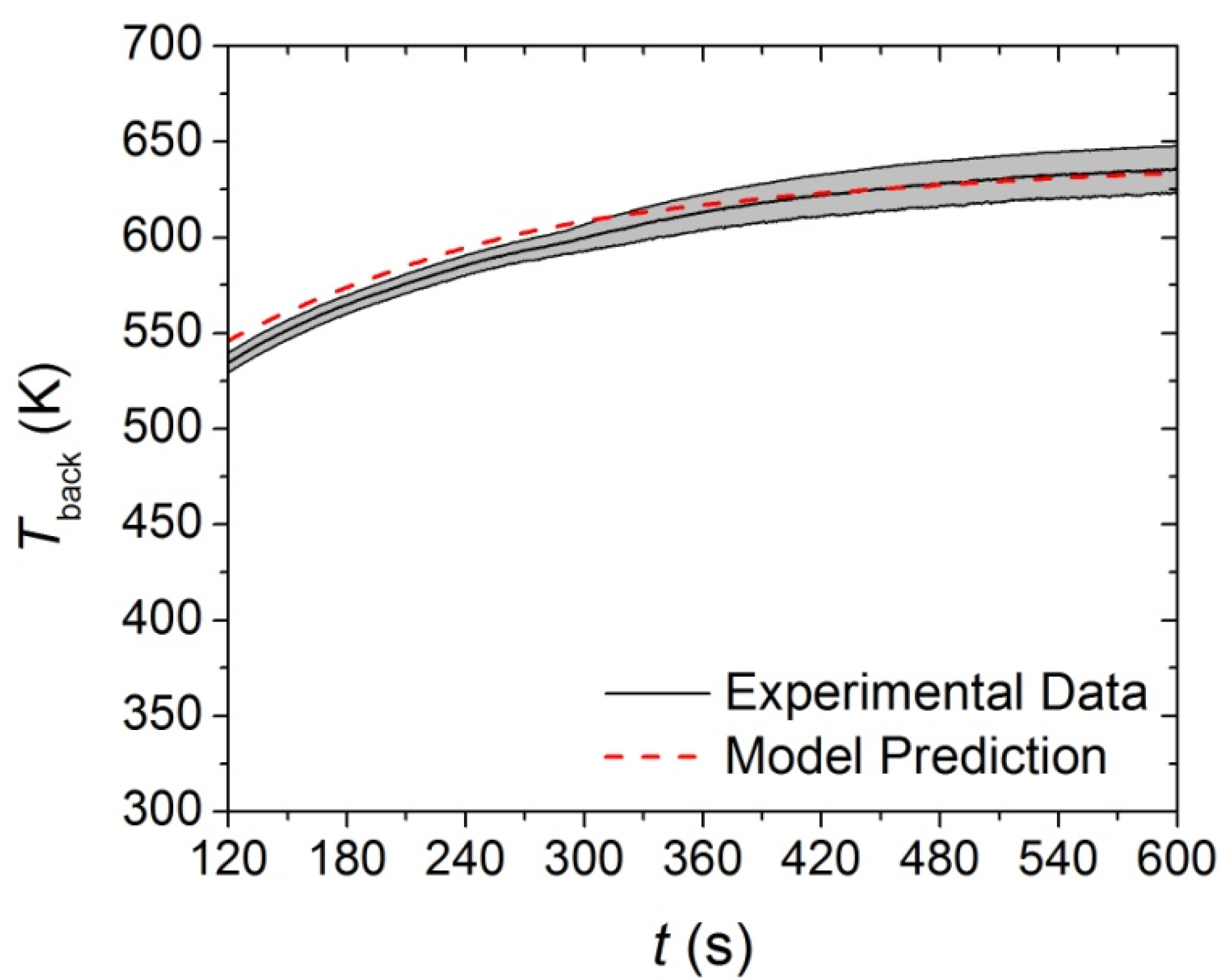
Figure 11.
First 120 s of experimental Tback curve measured in Controlled Atmosphere Pyrolysis Apparatus (CAPA) tests and corresponding model predicted curve for the upper layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
Figure 11.
First 120 s of experimental Tback curve measured in Controlled Atmosphere Pyrolysis Apparatus (CAPA) tests and corresponding model predicted curve for the upper layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
Although the thermal conductivity determined for the upper layer is the only value that provides an adequate agreement between the predicted curve and the experimental data, it is probable that some physical phenomena that occurred during testing are not represented in the model. As mentioned in
Section 3.2.4, the edges of the upper layer sample tended to curl inward during CAPA tests, and the method of securing the sample to the holder was only partially effective in reducing the deformation. It is possible that the decrease in exposed area may have caused the samples to deviate from one-dimensional behavior. The back surface of the upper layer samples was textured which may have compromised the thermal contact between the sample and the aluminum foil, resulting in inaccurate
Tback profiles.
An inverse analysis was conducted on the
Tback data collected on the upper layer subjected to a heat flux of 30 kW·m
−2 to determine the thermal conductivities of the char components. The target data for the inverse analysis was chosen as the slowly rising portion of
Tback that was observed after 120 s in the gasification tests. Due to the high porosity of the chars produced in each layer, radiation was assumed to be the dominant mode of heat transfer through the char. The radiation diffusion approximation [
30] was invoked to describe the thermal conductivity of all the char components. An attempt was made to describe all the char components with a single thermal conductivity expression of the form
βT3 and it was determined that a single value of
β adequately described the
Tback profile in the final 480 s of the curve. The thermal conductivities of all the intermediate components were defined as the mean of the thermal conductivities of the corresponding virgin component and char component. For the face yarn intermediate, this produced an expression with a constant term and a
T3 term. The agreement between the
Tback predictions and the experimental data collected on the upper layer at a heat flux of 30 kW·m
−2 are shown in
Figure 12. The full set of parameters that were determined for the upper layer of the carpet composite to describe thermal transport to and within the solid sample are provided in
Table 5.
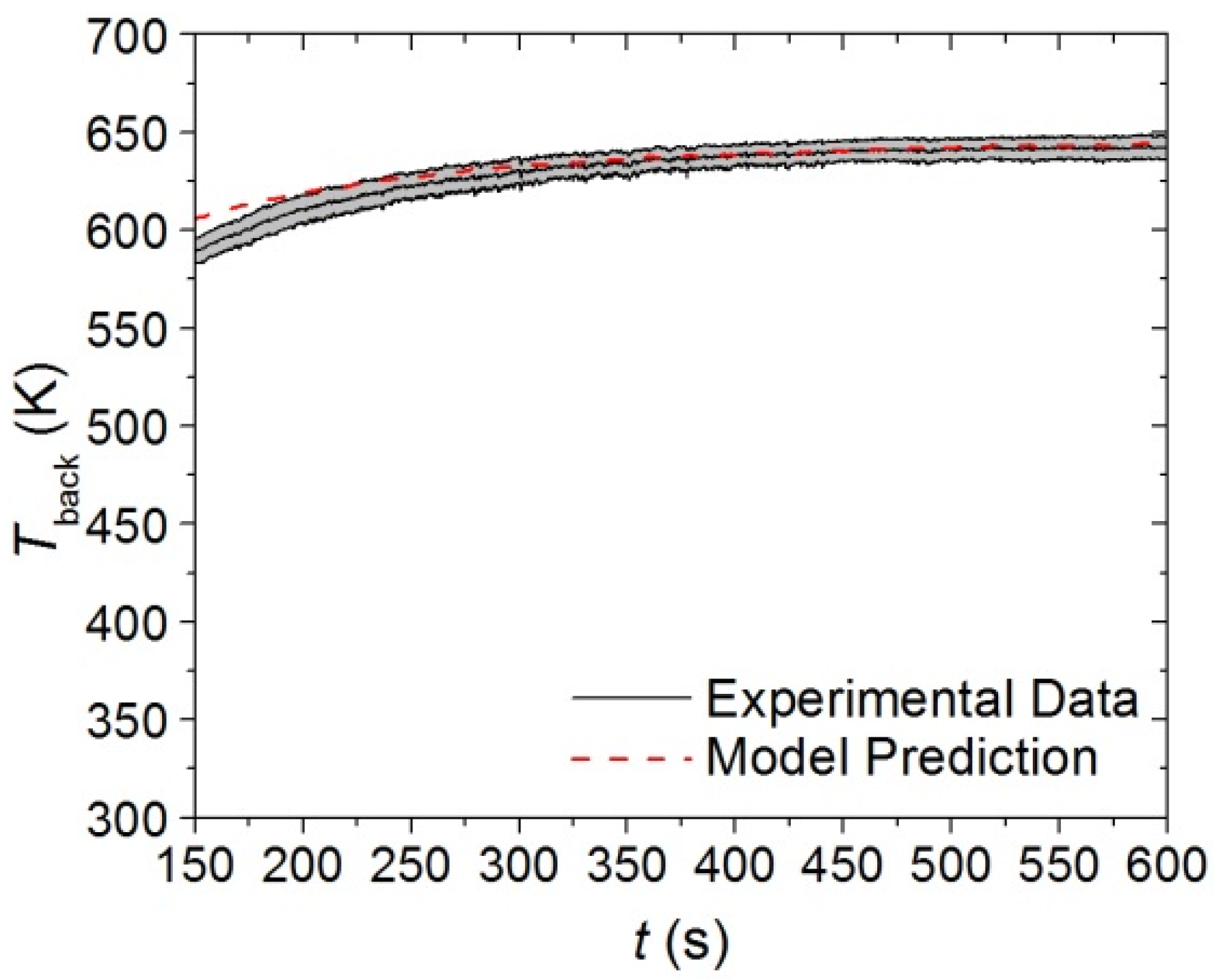
Figure 12.
Final 480 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the upper layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
Figure 12.
Final 480 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the upper layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
An inverse analysis to determine the thermal conductivity of the virgin and melt components was conducted on the data collected in CAPA tests on the base layer at a heat flux of 30 kW·m
−2. The target data was identified as the slope of the initial increase in the
Tback. Inadequate agreement between the model prediction and the experimental data was produced with a single, constant value for the thermal conductivity of the virgin and melt components. A linearly decreasing thermal conductivity for the
and
components was able to adequately predict both the fast and slow rising portions of the initial 150 s of the
Tback curve. The agreement between the experimental curve and the model prediction are provided in
Figure 13.
Figure 13.
First 150 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the base layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
Figure 13.
First 150 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the base layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
The data collected in the CAPA tests on the base layer were analyzed to determine the thermal conductivity of the
component. The resulting experimental and predicted curves for the
Tback of the base layer are provided in
Figure 14. A single, constant value of
β in the
βT3 expression adequately described the
Tback profile in the final 450 s of the curve. The thermal conductivity of the Base
int component was defined as the mean between the Base
virgin and Base
char components, which resulted in a form with a constant, linear, and
T3 term. The full set of parameters that define the base layer thermophysical properties are provided in
Table 5.
Figure 14.
Final 450 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the base layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.
Figure 14.
Final 450 s of experimental Tback curve measured in CAPA tests and corresponding model predicted curve for the base layer exposed to a radiant flux of 30 kW·m−2. The shaded region corresponds to two standard deviations of the mean experimental data.