Sustained Administration of Hormones Exploiting Nanoconfined Diffusion through Nanochannel Membranes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Levothyroxine
| Properties | Osteocalcin | Levothyroxine | Testosterone |
|---|---|---|---|
| Mass (Da) | 5929 | 777 | 288 |
| Net Charge (pH 7.4) | −6 | −0.5 | 0 |
| Radius (Å) | 10 | 4.3 | 4.1 |
| LogD (PH 7.4) | ~−6 | 1.76 | 3.16 |
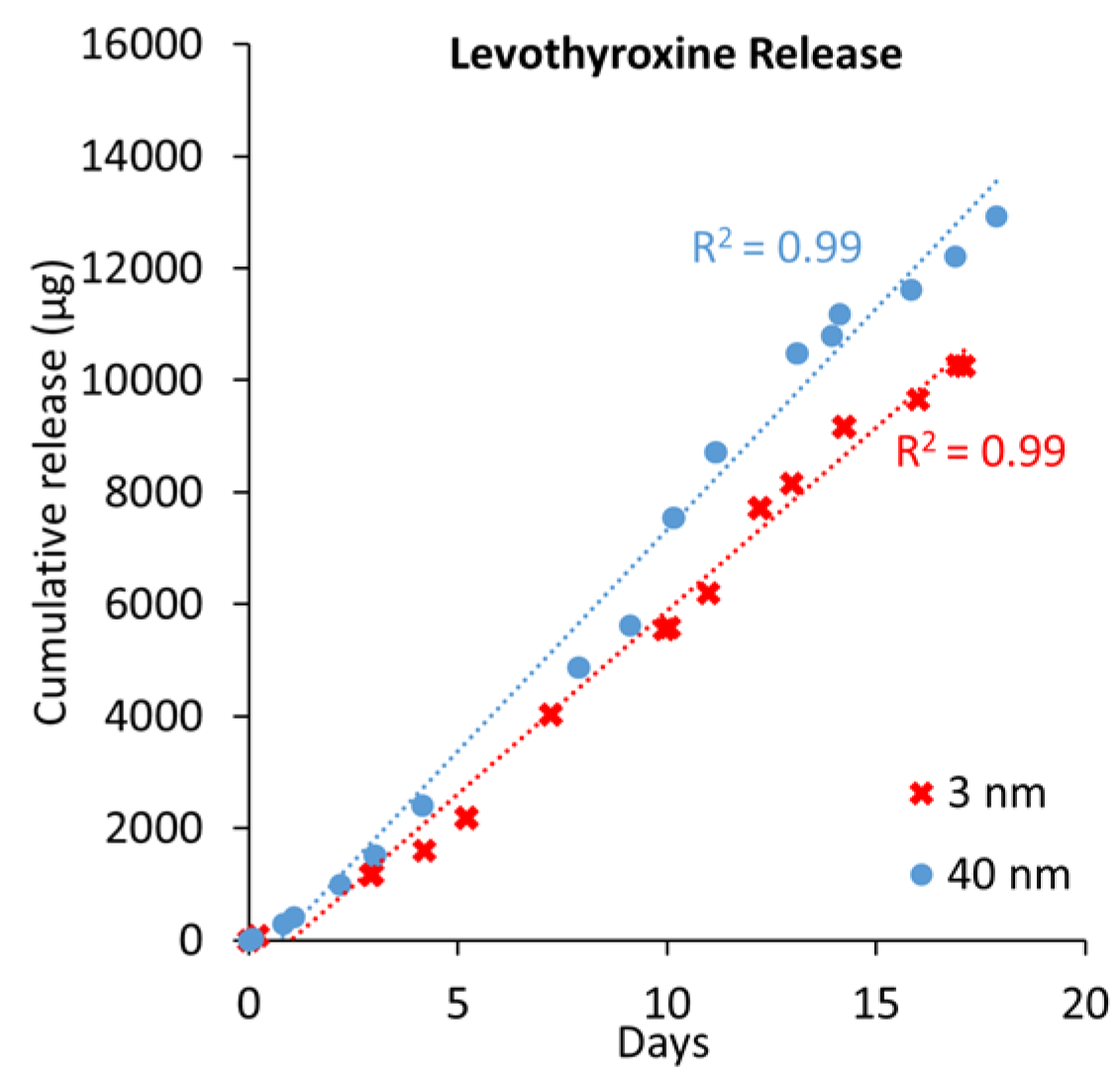
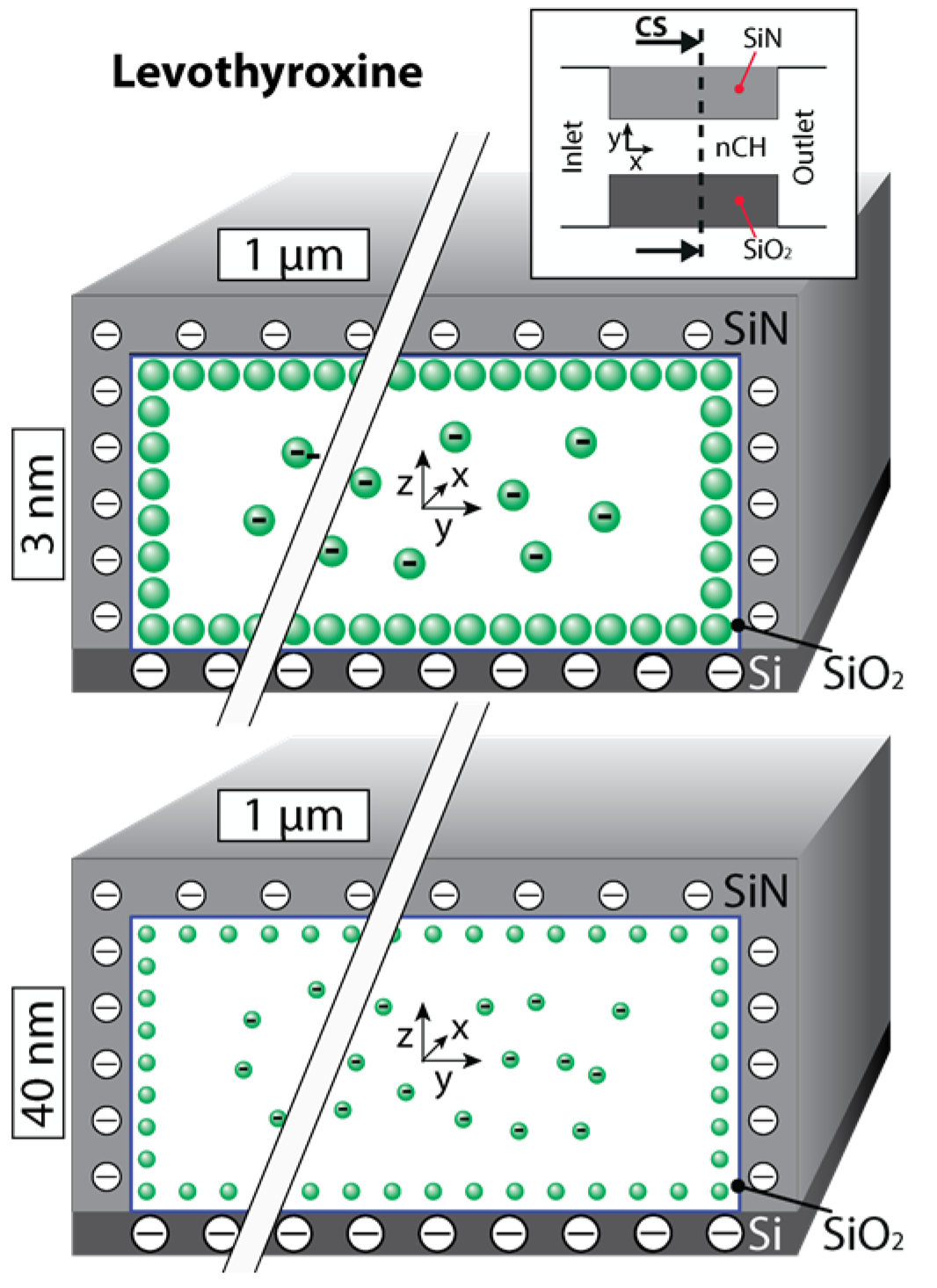
2.2. Osteocalcin
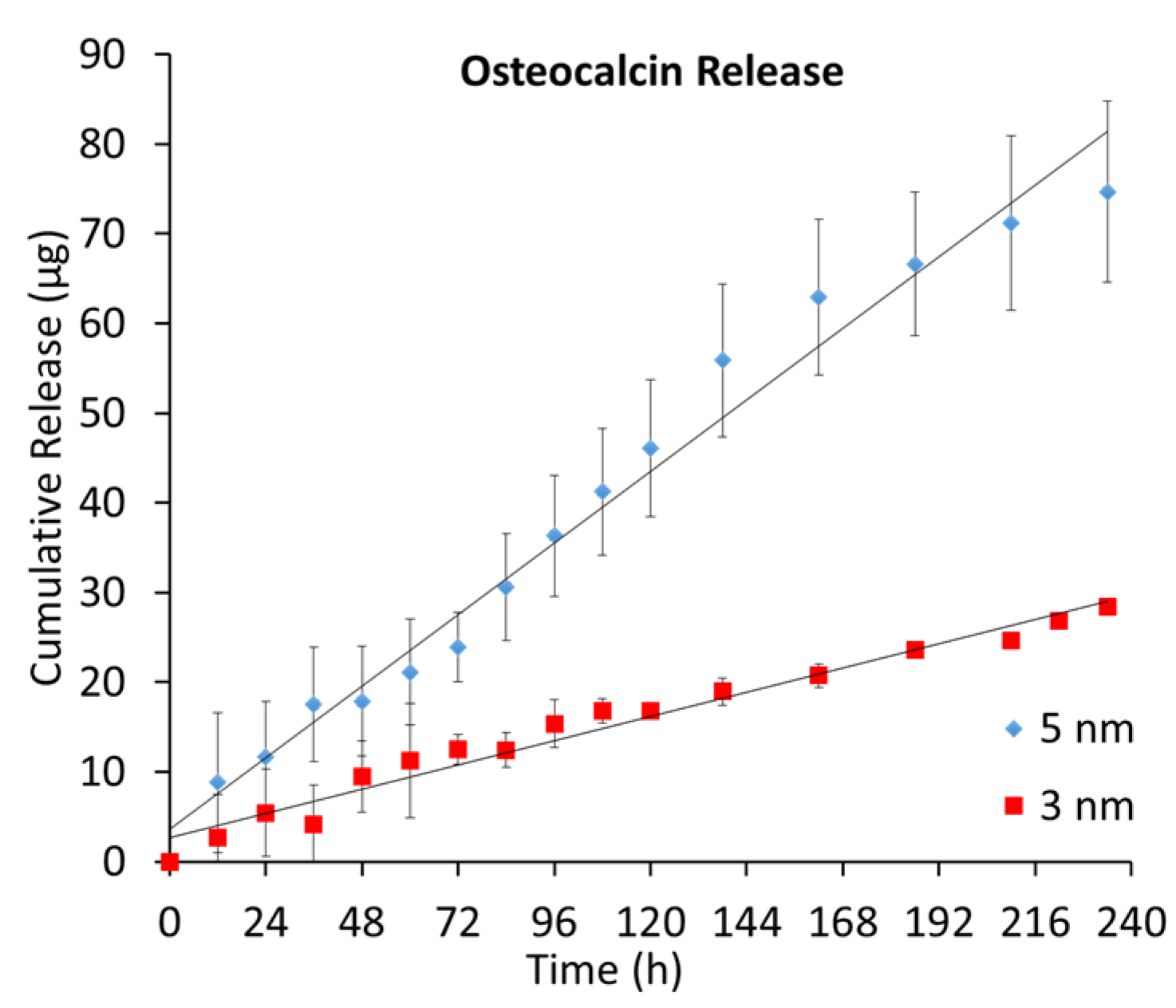
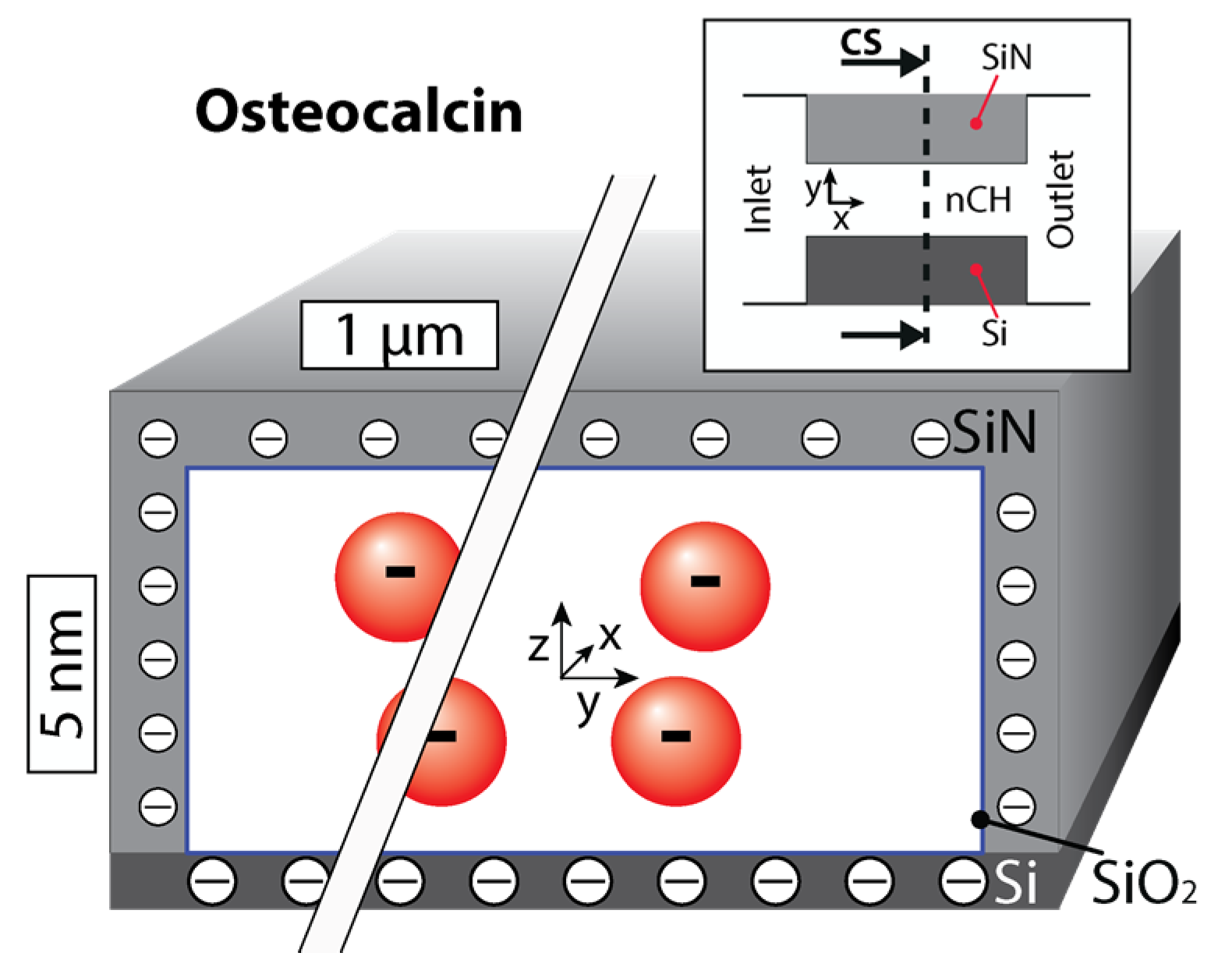
2.3. Testosterone

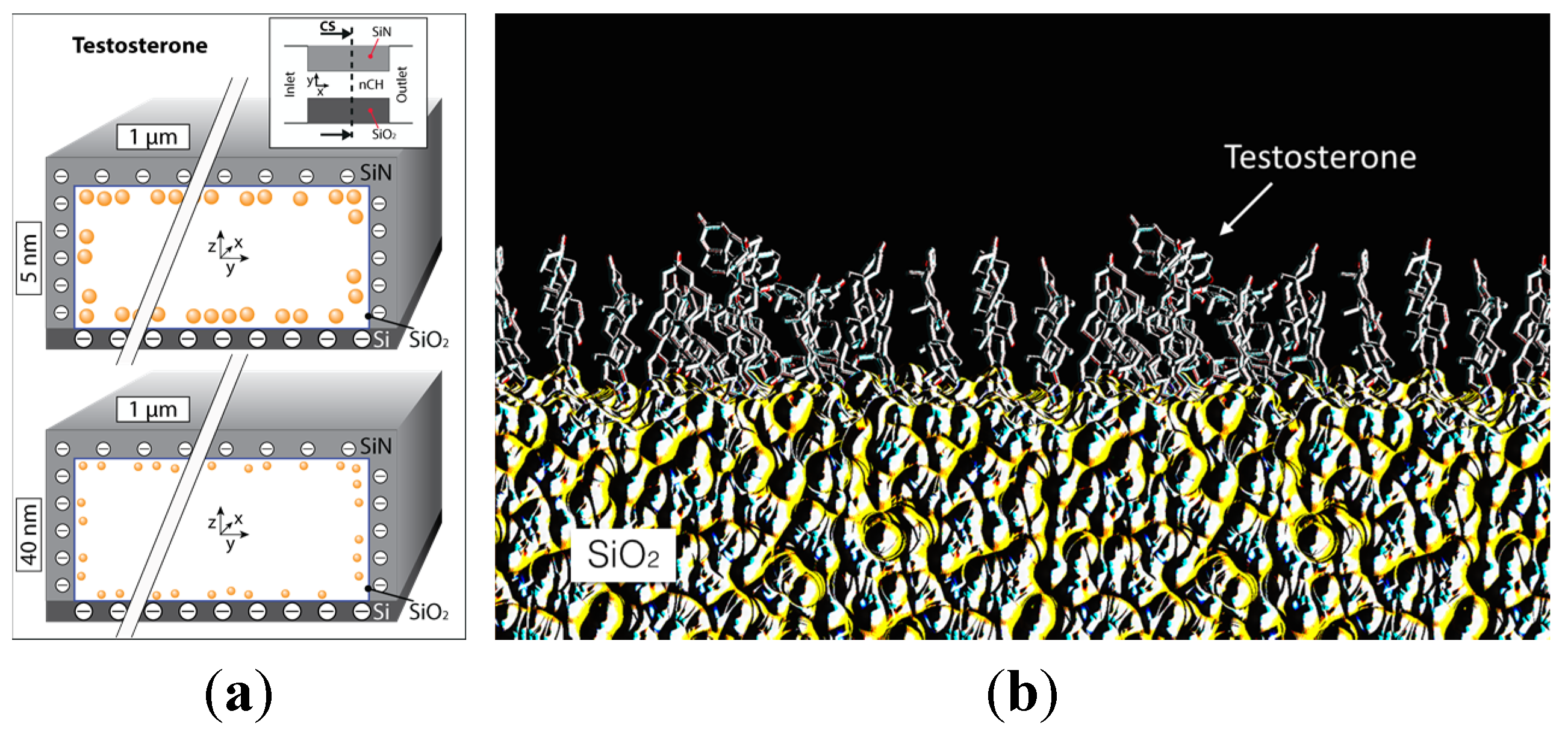
3. Materials and Methods
3.1. Nanochannel Membrane Fabrication

3.2. Levothyroxine Release
3.3. Osteocalcin Release
3.4. Testosterone Release
3.5. Computational Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Keating, N.L.; Cleary, P.D.; Rossi, A.S.; Zaslavsky, A.M.; Ayanian, J.Z. Use of hormone replacement therapy by postmenopausal women in the united states. Ann. Intern. Med. 1999, 130, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D.; Humphrey, L.L.; Nygren, P.; Teutsch, S.M.; Allan, J.D. Postmenopausal hormone replacement therapy: Scientific review. JAMA 2002, 288, 872–881. [Google Scholar] [CrossRef] [PubMed]
- Snyder, P.J.; Peachey, H.; Berlin, J.A.; Rader, D.; Usher, D.; Loh, L.; Hannoush, P.; Dlewati, A.; Holmes, J.H.; Santanna, J.; et al. Effect of transdermal testosterone treatment on serum lipid and apolipoprotein levels in men more than 65 years of age. Am. J. Med. 2001, 111, 255–260. [Google Scholar] [CrossRef]
- Agersø, H.; Møller-Pedersen, J.; Cappi, S.; Thomann, P.; Jesussek, B.; Senderovitz, T. Pharmacokinetics and pharmacodynamics of a new formulation of recombinant human growth hormone administered by zomajet 2 vision, a new needle-free device, compared to subcutaneous administration using a conventional syringe. J. Clin. Pharmacol. 2002, 42, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Cázares-Delgadillo, J.; Ganem-Rondero, A.; Kalia, Y.N. Human growth hormone: New delivery systems, alternative routes of administration, and their pharmacological relevance. Eur. J. Pharm. Biopharm. 2011, 78, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Basaria, S.; Wahlstrom, J.T.; Dobs, A.S. Anabolic-androgenic steroid therapy in the treatment of chronic diseases. J. Clin. Endocrinol. Metab. 2001, 86, 5108–5117. [Google Scholar] [CrossRef] [PubMed]
- Kornstein, S.G.; Toups, M.; Rush, A.J.; Wisniewski, S.R.; Thase, M.E.; Luther, J.; Warden, D.; Fava, M.; Trivedi, M.H. Do menopausal status and use of hormone therapy affect antidepressant treatment response? Findings from the sequenced treatment alternatives to relieve depression (STAR* D) study. J. Women Health 2013, 22, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Mosca, L.; Collins, P.; Herrington, D.M.; Mendelsohn, M.E.; Pasternak, R.C.; Robertson, R.M.; Schenck-Gustafsson, K.; Smith, S.C., Jr.; Taubert, K.A.; Wenger, N.K.; et al. Hormone replacement therapy and cardiovascular disease: A statement for healthcare professionals from the american heart association. Circulation 2001, 104, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.H.; Lee, M.J.; Kim, S.J.; Oh, H.J.; Kim, H.R.; Han, J.H.; Koo, H.M.; Doh, F.M.; Park, J.T.; Han, S.H. Preservation of renal function by thyroid hormone replacement therapy in chronic kidney disease patients with subclinical hypothyroidism. J. Clin. Endocrinol. Metab. 2012, 97, 2732–2740. [Google Scholar] [CrossRef] [PubMed]
- Turgeon, J.L.; McDonnell, D.P.; Martin, K.A.; Wise, P.M. Hormone therapy: Physiological complexity belies therapeutic simplicity. Science 2004, 304, 1269–1273. [Google Scholar] [CrossRef] [PubMed]
- Ferrati, S.; Nicolov, E.; Zabre, E.; Geninatti, T.; Shirkey, B.A.; Hudson, L.; Hosali, S.; Crawley, M.; Khera, M.; Palapattu, G.; et al. The nanochannel delivery system for constant testosterone replacement therapy. J. Sex. Med. 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Nicolov, E.; Ferrati, S.; Goodall, R.; Hudson, L.; Hosali, S.; Crowley, M.; Palapattu, G.; Khera, M.; Grattoni, A. Mp43-20 nanotechnology-based implant for long term testosterone replacement. J. Urol. 2014, 191, e485–e486. [Google Scholar] [CrossRef]
- Fidotti, E. A history of growth hormone injection devices. J. Pediatr. Endocrinol. Metab. 2001, 14, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Azarbayjani, A.F.; Venugopal, J.R.; Ramakrishna, S.; Lim, F.C.; Chan, Y.W.; Chan, S.Y. Smart polymeric nanofibers for topical delivery of levothyroxine. J. Pharm. Pharm. Sci. 2010, 13, 400–410. [Google Scholar] [PubMed]
- Astaneh, R.; Erfan, M.; Moghimi, H.; Mobedi, H. Changes in morphology of in situ forming PLGA implant prepared by different polymer molecular weight and its effect on release behavior. J. Pharm. Sci. 2009, 98, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Angelova, A.; Angelov, B.; Drechsler, M.; Garamus, V.M.; Lesieur, S. Protein entrapment in pegylated lipid nanoparticles. Int. J. Pharm. 2013, 454, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Angelov, B.; Angelova, A.; Filippov, S.K.; Drechsler, M.; Šteěpaánek, P.; Lesieur, S. Multicompartment lipid cubic nanoparticles with high protein upload: Millisecond dynamics of formation. ACS Nano 2014, 8, 5216–5226. [Google Scholar] [CrossRef] [PubMed]
- Schliecker, G.; Schmidt, C.; Fuchs, S.; Ehinger, A.; Sandow, J.; Kissel, T. In vitro and in vivo correlation of buserelin release from biodegradable implants using statistical moment analysis. J. Control. Release 2004, 94, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Sprintz, M.; Tasciotti, E.; Allegri, M.; Grattoni, A.; Driver, L.C.; Ferrari, M. Nanomedicine: Ushering in a new era of pain management. Eur. J. Pain Suppl. 2011, 5, 317–322. [Google Scholar] [CrossRef]
- Grattoni, A.; Fine, D.; Ziemys, A.; Gill, J.; Zabre, E.; Goodall, R.; Ferrari, M. Nanochannel systems for personalized therapy and laboratory diagnostics. Curr. Pharm. Biotechnol. 2010, 11, 343–365. [Google Scholar] [CrossRef] [PubMed]
- Ferrati, S.; Nicolov, E.; Bansal, S.; Hosali, S.; Landis, M.; Grattoni, A. Docetaxel/2-hydroxypropyl β-cyclodextrin inclusion complex increases docetaxel solubility and release from a nanochannel drug delivery system. Curr. Drug Targets 2015. Epub ahead of print. [Google Scholar] [CrossRef]
- Ferrati, S.; Fine, D.; You, J.; De Rosa, E.; Hudson, L.; Zabre, E.; Hosali, S.; Zhang, L.; Hickman, C.; Sunder Bansal, S.; et al. Leveraging nanochannels for universal, zero-order drug delivery in vivo. J. Control. Release 2013, 172, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Grattoni, A.; Fine, D.; Zabre, E.; Ziemys, A.; Gill, J.; Mackeyev, Y.; Cheney, M.A.; Danila, D.C.; Hosali, S.; Wilson, L.J.; et al. Gated and near-surface diffusion of charged fullerenes in nanochannels. ACS Nano 2011, 5, 9382–9391. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.; Walczak, R.; Boiarski, A.; Cohen, M.; West, T.; Cosentino, C.; Shapiro, J.; Ferrari, M. Tailoring width of microfabricated nanochannels to solute size can be used to control diffusion kinetics. J. Control. Release 2005, 102, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Celia, C.; Ferrati, S.; Bansal, S.; van de Ven, A.L.; Ruozi, B.; Zabre, E.; Hosali, S.; Paolino, D.; Sarpietro, M.G.; Fine, D.; et al. Sustained zero-order release of intact ultra-stable drug-loaded liposomes from an implantable nanochannel delivery system. Adv. Healthc. Mater. 2014, 3, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Pimpinelli, A.; Ferrari, M.; Grattoni, A. Scaling and crossovers in molecular transport in nano-fluidic systems. Appl. Phys. Lett. 2013, 103. [Google Scholar] [CrossRef]
- Ferrati, S.; Nicolov, E.; Bansal, S.; Zabre, E.; Geninatti, T.; Ziemys, A.; Hudson, L.; Ferrari, M.; Goodall, R.; Khera, M.; et al. Delivering enhanced testosterone replacement therapy through nanochannels. Adv. Healthc. Mater. 2015, 4, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Sih, J.; Bansal, S.S.; Filippini, S.; Ferrati, S.; Raghuwansi, K.; Zabre, E.; Nicolov, E.; Fine, D.; Ferrari, M.; Palapattu, G.; et al. Characterization of nanochannel delivery membrane systems for the sustained release of resveratrol and atorvastatin: New perspectives on promoting heart health. Anal. Bioanal. Chem. 2013, 405, 1547–1557. [Google Scholar] [CrossRef] [PubMed]
- Biondi, B.; Wartofsky, L. Combination treatment with T4 and T3: Toward personalized replacement therapy in hypothyroidism? J. Clin. Endocrinol. Metab. 2012, 97, 2256–2271. [Google Scholar] [CrossRef] [PubMed]
- Volpato, N.M.; Silva, R.L.; Brito, A.P.; Goncalves, J.C.; Vaisman, M.; Noël, F. Multiple level C in vitro/in vivo correlation of dissolution profiles of two l-thyroxine tablets with pharmacokinetics data obtained from patients treated for hypothyroidism. Eur. J. Pharm. Sci. 2004, 21, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; Stalcup, A.; Dansereau, R.; Sakr, A. The effect of excipients on the stability of levothyroxine sodium pentahydrate tablets. Int. J. Pharm. 2003, 264, 35–43. [Google Scholar] [CrossRef]
- Ziemys, A.; Grattoni, A.; Fine, D.; Hussain, F.; Ferrari, M. Confinement effects on monosaccharide transport in nanochannels. J. Phys. Chem. B 2010, 114, 11117–11126. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.K.; Sowa, H.; Hinoi, E.; Ferron, M.; Ahn, J.D.; Confavreux, C.; Dacquin, R.; Mee, P.J.; McKee, M.D.; Jung, D.Y.; et al. Endocrine regulation of energy metabolism by the skeleton. Cell 2007, 130, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, C.; Amato, F.; Walczak, R.; Boiarski, A.; Ferrari, M. Dynamic model of biomolecular diffusion through two-dimensional nanochannels. J. Phys. Chem. B 2005, 109, 7358–7364. [Google Scholar] [CrossRef] [PubMed]
- Fine, D.; Grattoni, A.; Zabre, E.; Hussein, F.; Ferrari, M.; Liu, X. A low-voltage electrokinetic nanochannel drug delivery system. Lab Chip 2011, 11, 2526–2534. [Google Scholar] [CrossRef] [PubMed]
- Geninatti, T.; Bruno, G.; Barile, B.; Hood, R.L.; Farina, M.; Schmulen, J.; Canavese, G.; Grattoni, A. Impedance characterization, degradation, and in vitro biocompatibility for platinum electrodes on biomems. Biomed. Microdevices 2015, 17. [Google Scholar] [CrossRef] [PubMed]
- Bruno, G.; Geninatti, T.; Hood, R.L.; Fine, D.; Scorrano, G.; Schmulen, J.; Hosali, S.; Ferrari, M.; Grattoni, A. Leveraging electrokinetics for the active control of dendritic fullerene-1 release across a nanochannel membrane. Nanoscale 2015, 7, 5240–5248. [Google Scholar] [CrossRef] [PubMed]
- Okorn-Schmidt, H.F. Characterization of silicon surface preparation processes for advanced gate dielectrics. IBM J. Res. Dev. 1999, 43, 351–326. [Google Scholar] [CrossRef]
- Binks, B.P.; Fletcher, P.D.I.; Johnson, A.J.; Elliott, R.P. Membrane permeation of testosterone from either solutions, particle dispersions, or particle-stabilized emulsions. Langmuir 2012, 28, 2510–2522. [Google Scholar] [CrossRef] [PubMed]
- Fine, D.; Grattoni, A.; Goodall, R.; Bansal, S.S.; Chiappini, C.; Hosali, S.; van de Ven, A.L.; Srinivasan, S.; Liu, X.; Godin, B.; et al. Silicon micro- and nanofabrication for medicine. Adv. Healthc. Mater. 2013, 2, 632–666. [Google Scholar] [CrossRef] [PubMed]
- Fine, D.; Grattoni, A.; Hosali, S.; Ziemys, A.; De Rosa, E.; Gill, J.; Medema, R.; Hudson, L.; Kojic, M.; Milosevic, M.; et al. A robust nanofluidic membrane with tunable zero-order release for implantable dose specific drug delivery. Lab Chip 2010, 10, 3074–3083. [Google Scholar] [CrossRef] [PubMed]
- Ziemys, A.; Kojic, M.; Milosevic, M.; Kojic, N.; Hussain, F.; Ferrari, M.; Grattoni, A. Hierarchical modeling of diffusive transport through nanochannels by coupling molecular dynamics with finite element method. J. Comput. Phys. 2011, 230, 5722–5731. [Google Scholar] [CrossRef]
- Ziemys, A.; Ferrari, M.; Cavasotto, C.N. Molecular modeling of glucose diffusivity in silica nanochannels. J. Nanosci. Nanotechnol. 2009, 9, 6349–6359. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.C.; Braun, R.; Wang, W.; Gumbart, J.; Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R.D.; Kale, L.; Schulten, K. Scalable molecular dynamics with NAMD. J. Comput. Chem. 2005, 26, 1781–1802. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, W.L.; Chandrasekhar, J.; Madura, J.D.; Impey, R.W.; Klein, M.L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 1983, 79, 926–935. [Google Scholar] [CrossRef]
- Cruz-Chu, E.R.; Aksimentiev, A.; Schulten, K. Water-silica force field for simulating nanodevices. J. Phys. Chem. B 2006, 110, 21497–21508. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geninatti, T.; Hood, R.L.; Bruno, G.; Jain, P.; Nicolov, E.; Ziemys, A.; Grattoni, A. Sustained Administration of Hormones Exploiting Nanoconfined Diffusion through Nanochannel Membranes. Materials 2015, 8, 5276-5288. https://doi.org/10.3390/ma8085241
Geninatti T, Hood RL, Bruno G, Jain P, Nicolov E, Ziemys A, Grattoni A. Sustained Administration of Hormones Exploiting Nanoconfined Diffusion through Nanochannel Membranes. Materials. 2015; 8(8):5276-5288. https://doi.org/10.3390/ma8085241
Chicago/Turabian StyleGeninatti, Thomas, R. Lyle Hood, Giacomo Bruno, Priya Jain, Eugenia Nicolov, Arturas Ziemys, and Alessandro Grattoni. 2015. "Sustained Administration of Hormones Exploiting Nanoconfined Diffusion through Nanochannel Membranes" Materials 8, no. 8: 5276-5288. https://doi.org/10.3390/ma8085241





