Raman Spectroscopy of Two-Dimensional Bi2TexSe3 − x Platelets Produced by Solvothermal Method
Abstract
:1. Introduction
2. Experimental
2.1. Material Synthesis
2.2. Material Characterizations
3. Results and Discussion
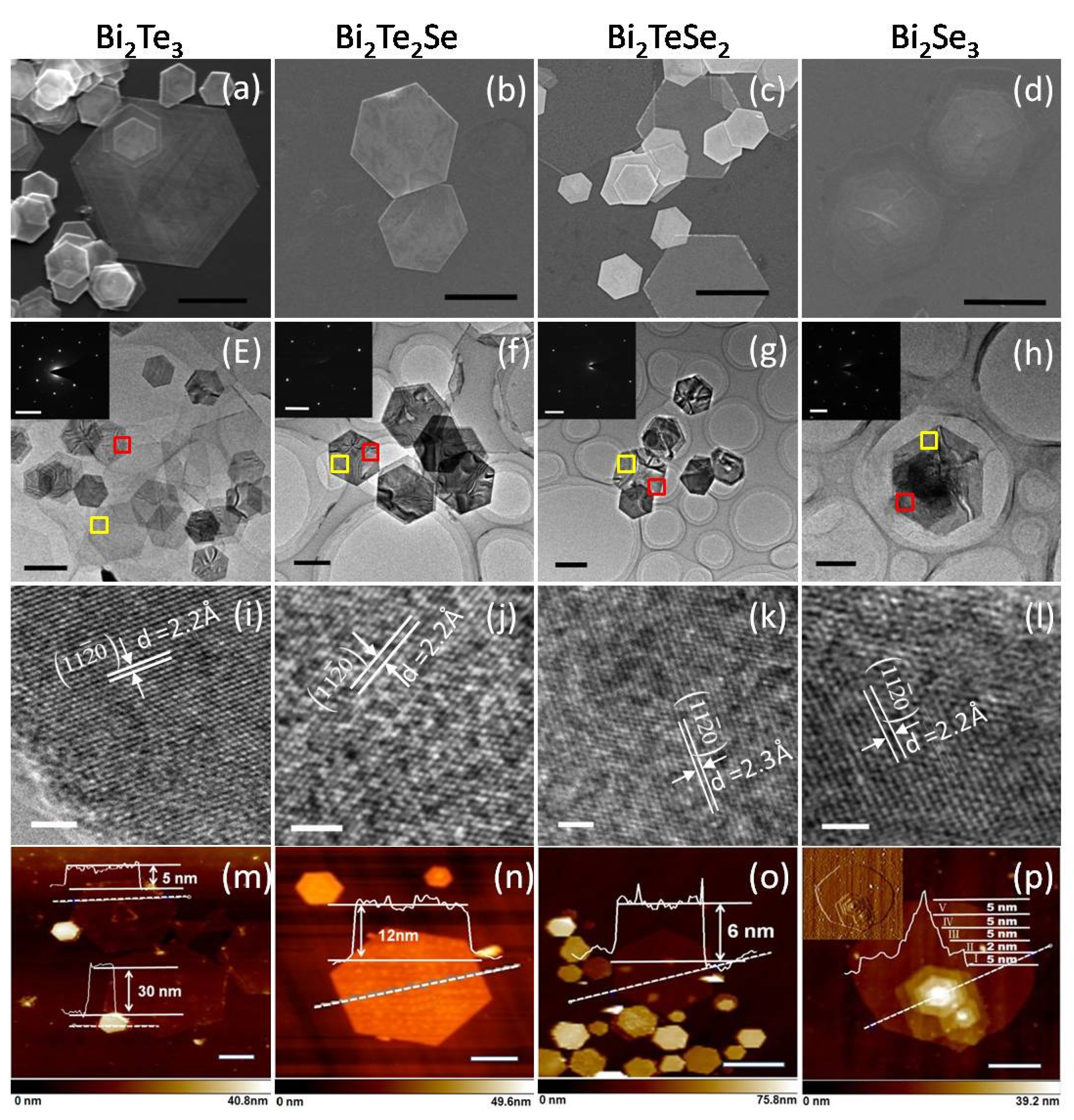
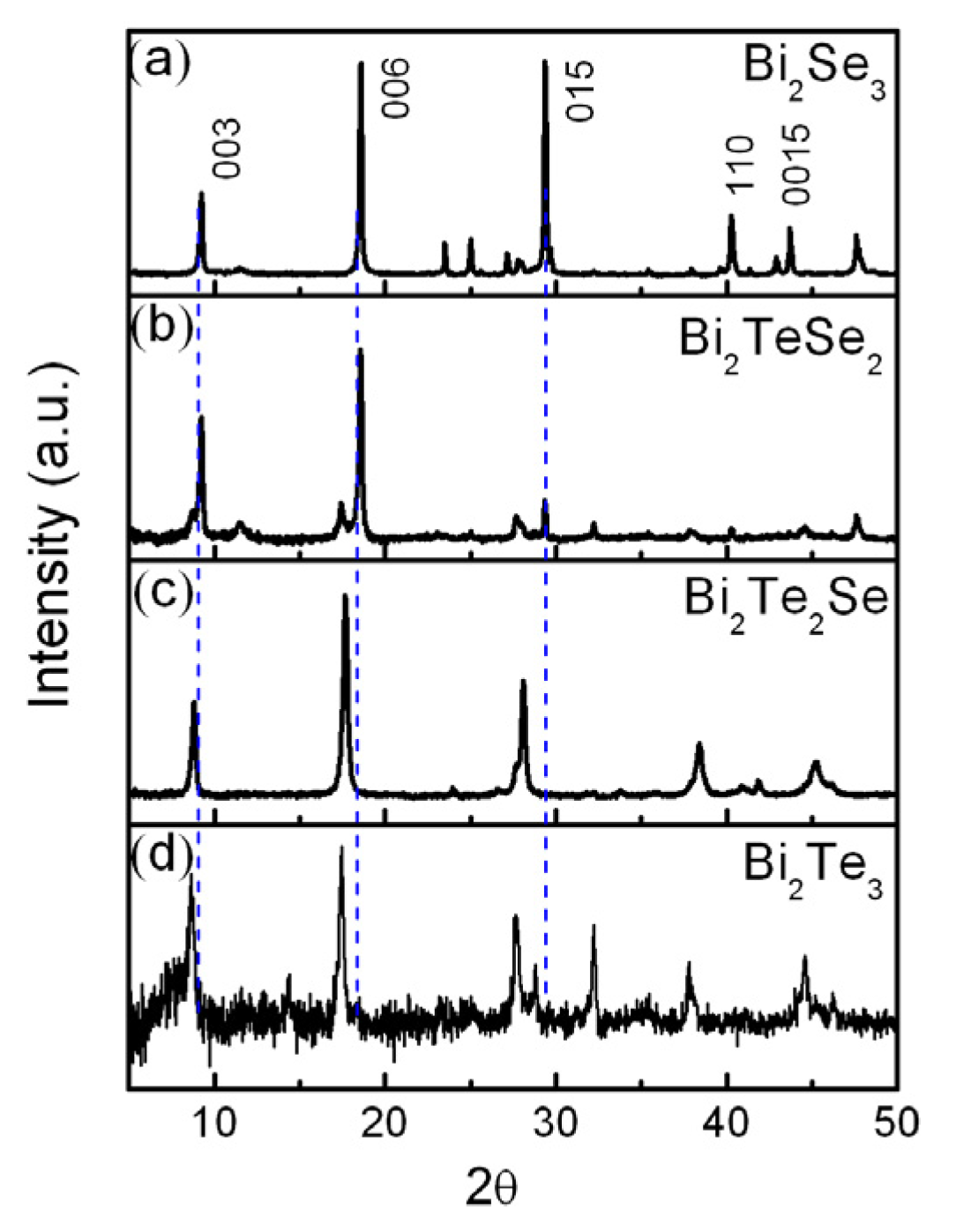
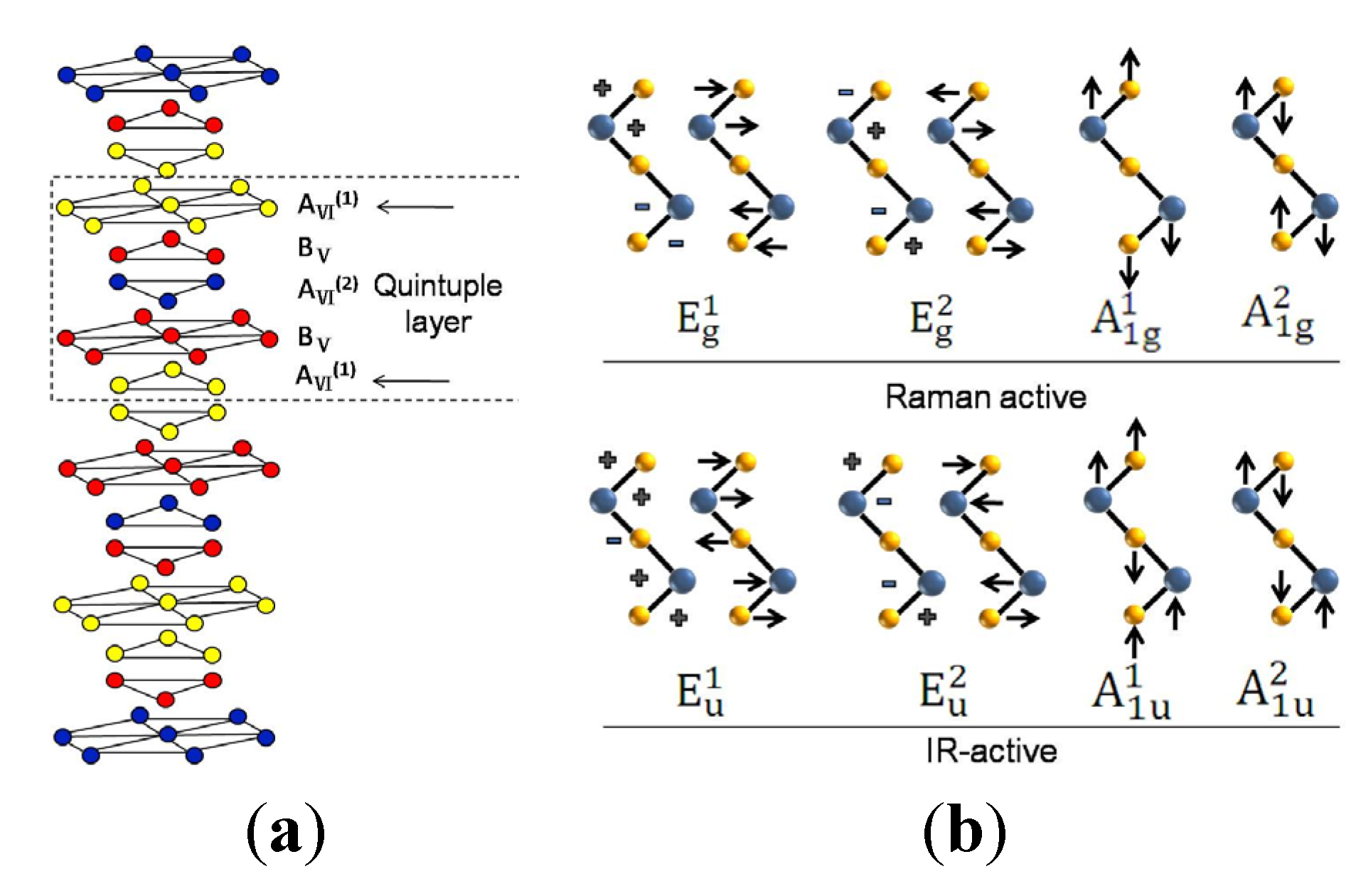
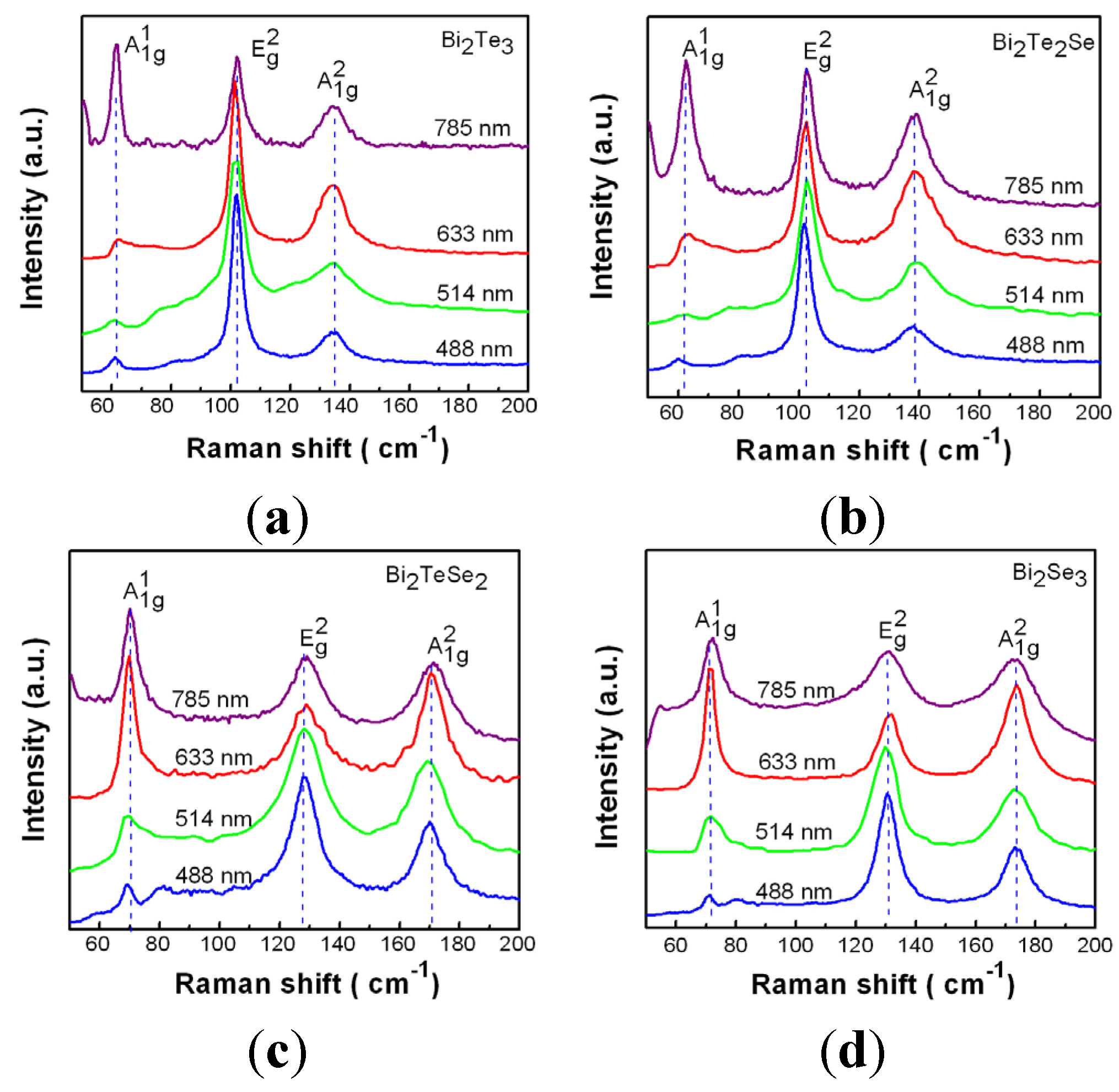
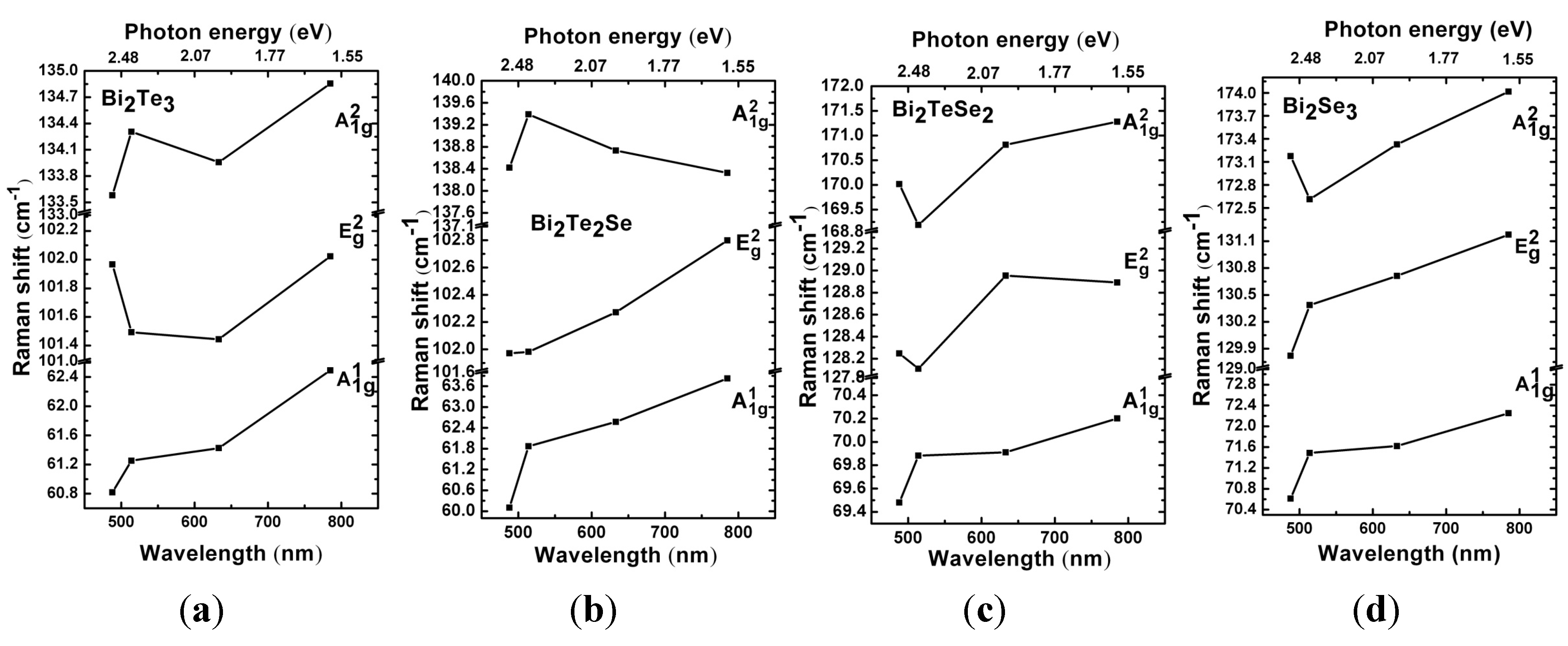
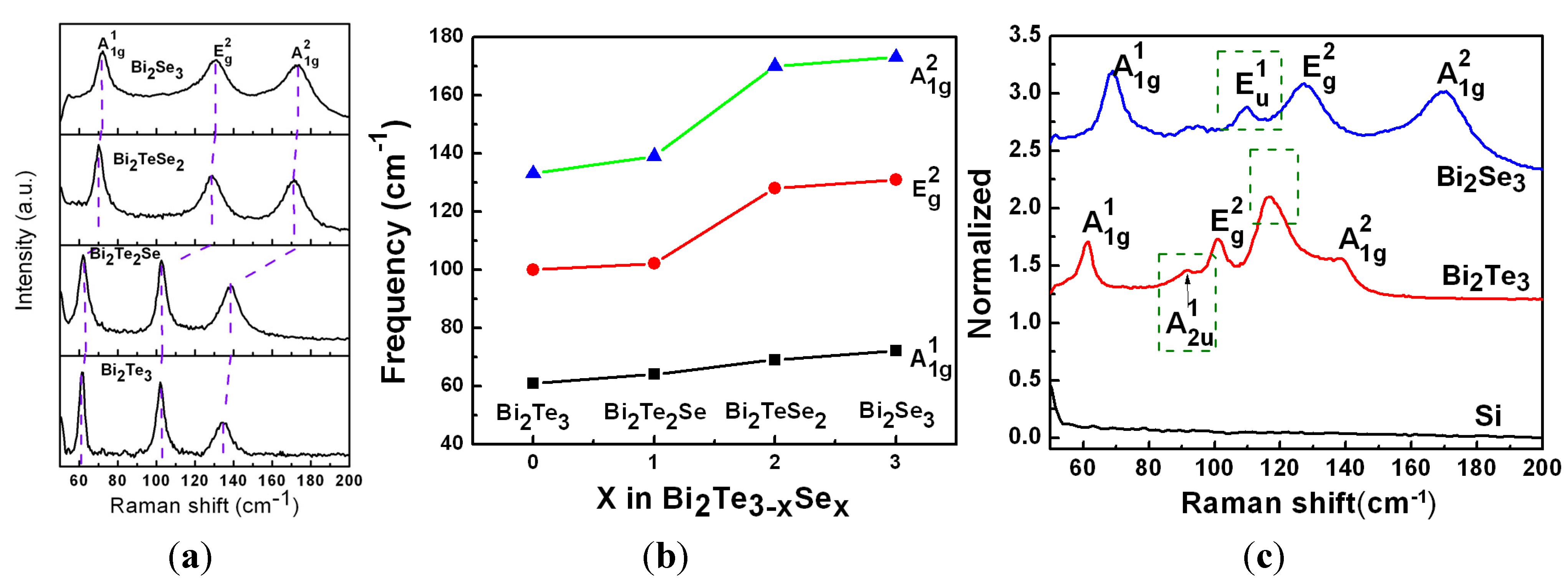
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moore, J.E. The birth of topological insulators. Nature 2010, 464, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Fu, L. Topological crystalline insulators. Phys. Rev. Lett. 2011, 106. [Google Scholar] [CrossRef]
- Butler, S.Z.; Hollen, S.M.; Cao, L.; Cui, Y.; Gupta, J.A.; Gutierrez, H.R.; Heinz, T.F.; Hong, S.S.; Huang, J.; Ismach, A.F.; et al. Progress, challenges, and opportunities in two-dimensional materials beyond graphene. ACS Nano 2013, 7, 2898–2926. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; He, K.; Chang, C.Z.; Song, C.L.; Wang, L.L.; Chen, X.; Jia, J.F.; Fang, Z.; Dai, X.; Shan, W.Y.; et al. Crossover of the three-dimensional topological insulator Bi2Se3 to the two-dimensional limit. Nature Phys. 2010, 6, 584–588. [Google Scholar] [CrossRef]
- Chen, Y.; Analytis, J.; Chu, J.H.; Liu, Z.; Mo, S.K.; Qi, X.L.; Zhang, H.; Lu, D.; Dai, X.; Fang, Z.; et al. Experimental realization of a three-dimensional topological insulator, Bi2Te3. Science 2009, 325, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, C.X.; Qi, X.L.; Dai, X.; Fang, Z.; Zhang, S.C. Topological insulators in Bi2Se3, Bi2Te3 and Sb2Te3 with a single Dirac cone on the surface. Nature Phys. 2009, 5, 438–442. [Google Scholar] [CrossRef]
- Wang, Z.F.; Su, N.; Liu, F. Prediction of a two-dimensional organic topological insulator. Nano Lett. 2013, 13, 2842–2845. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Yuan, J.; Xu, Z.; Chen, C.; Lin, S.; Wang, Y.; Song, J.; Liu, Y.; Khan, Q.; Hoh, H.Y.; Bao, Q.L.; et al. Broadband Photodetector Based on Graphene-Bi2Te3 Heterostructure. ACS Nano 2015, 9, 1886–1894. [Google Scholar] [CrossRef] [PubMed]
- Soni, A.; Yanyuan, Z.; Ligen, Y.; Aik, M.K.K.; Dresselhaus, M.S.; Xiong, Q. Enhanced Thermoelectric Properties of Solution Grown Bi2Te3 – xSex Nanoplatelet Composites. Nano Lett. 2012, 12, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Min, Y.; Moon, G.D.; Kim, B.S.; Lim, B.; Kim, J.S.; Kang, C.Y.; Jeong, U. Quick, controlled synthesis of ultrathin Bi2Se3 nanodiscs and nanosheets. J. Am. Chem. Soc. 2012, 134, 2872–2875. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhu, X.G.; Sun, Y.Y.; Li, Y.Y.; Zhang, T.; Wen, J.; Chen, X.; He, K.; Wang, L.L.; Ma, X.C.; et al. Topological insulator thin films of Bi2Te3 with controlled electronic structure. Adv. Mater. 2011, 23, 2929–2932. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Yu, J.; Wang, H.; Zhang, H. Hydrothermal synthesis of single-crystalline antimony telluride nanobelts. J. Am. Chem. Soc. 2006, 128, 16490–16491. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Wang, W.; Lu, X.; Li, X. Solvothermal synthesis of V–VI binary and ternary hexagonal platelets: The oriented attachment mechanism. Cryst. Growth Des. 2008, 9, 145–150. [Google Scholar] [CrossRef]
- Xu, Y.; Ren, Z.; Ren, W.; Cao, G.; Deng, K.; Zhong, Y. Hydrothermal synthesis of single-crystalline Bi2Te3 nanoplates. Mater. Lett. 2008, 62, 4273–4276. [Google Scholar] [CrossRef]
- Cui, H.; Liu, H.; Li, X.; Wang, J.; Han, F.; Zhang, X.; Boughton, R.I. Synthesis of Bi2Se3 thermoelectric nanosheets and nanotubes through hydrothermal co-reduction method. J. Solid State Chem. 2004, 177, 4001–4006. [Google Scholar] [CrossRef]
- Mehta, R.J.; Zhang, Y.; Karthik, C.; Singh, B.; Siegel, R.W.; Borca-Tasciuc, T.; Ramanath, G. A new class of doped nanobulk high-figure-of-merit thermoelectrics by scalable bottom-up assembly. Nature mater. 2012, 11, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Peng, Z.; Soni, A.; Zhao, Y.; Xiong, Y.; Peng, B.; Wang, J.; Dresselhaus, M.S.; Xiong, Q. Raman spectroscopy of few-quintuple layer topological insulator Bi2Se3 nanoplatelets. Nano Lett. 2011, 11, 2407–2414. [Google Scholar] [CrossRef] [PubMed]
- Song, J.C.; Xia, F.; Zhao, M.; Zhong, Y.L.; Li, W.; Loh, K.P.; Causo, R.A.; Bao, Q.L. Solvothermal growth of bismuth chalcogenide nanoplatelets by the oriented attachment mechanism: An in situ PXRD syudy. Chem. Mater. 2015, 27, 3471–3482. [Google Scholar] [CrossRef]
- Kong, D.; Dang, W.; Cha, J.J.; Li, H.; Meister, S.; Peng, H.; Liu, Z.; Cui, Y. Few-layer nanoplates of Bi2Se3 and Bi2Te3 with highly tunable chemical potential. Nano Lett. 2010, 10, 2245–2250. [Google Scholar] [CrossRef] [PubMed]
- Richter, W.; Becker, C.R. A Raman and far-infrared investigation of phonons in the rhombohedral V2–VI3 compounds Bi2Te3, Bi2Se3, Sb2Te3 and Bi2(Te1 − xSex)3 (0 < x < 1),(Bi1 − ySby)2Te3 (0 < y < 1). Phys. Status solidi 1977, 84, 619–628. [Google Scholar]
- Kullmann, W.; Eichhorn, G.; Rauh, H.; Geick, R.; Eckold, G.; Steigenberger, U. Lattice dynamics and phonon dispersion in the narrow gap semiconductor Bi2Te3 with sandwich structure. Phys. Status solidi 1990, 162, 125–140. [Google Scholar] [CrossRef]
- Zhao, Y.; Luo, X.; Zhang, J.; Wu, J.; Bai, X.; Wang, M.; Jia, J.; Peng, H.; Liu, Z.; Quek, S.Y.; et al. Interlayer vibrational modes in few-quintuple-layer Bi2Te3 and Bi2Se3 two-dimensional crystals: Raman spectroscopy and first-principles studies. Phys. Rev. B 2014, 90. [Google Scholar] [CrossRef]
- Piscanec, S.; Cantoro, M.; Ferrari, A.; Zapien, J.; Lifshitz, Y.; Lee, S.T.; Hofmann, S.; Robertson, J. Raman spectroscopy of silicon nanowires. Phys. Rev. B 2003, 68. [Google Scholar] [CrossRef]
- Russo, V.; Bailini, A.; Zamboni, M.; Passoni, M.; Conti, C.; Casari, C.; Li Bassi, A.; Bottani, C.E. Raman spectroscopy of Bi-Te thin films. J. Raman Spectrosc. 2008, 39, 205–210. [Google Scholar] [CrossRef]
- Akrap, A.; Tran, M.; Ubaldini, A.; Teyssier, J.; Giannini, E.; Van Der Marel, D.; Lerch, P.; Homes, C.C. Optical properties of Bi2Te2Se at ambient and high pressures. Phys. Rev. B 2012, 86. [Google Scholar] [CrossRef]
- Chis, V.; Sklyadneva, I.Y.; Kokh, K.; Volodin, V.; Tereshchenko, O.; Chulkov, E. Vibrations in binary and ternary topological insulators: First-principles calculations and Raman spectroscopy measurements. Phys. Rev. B 2012, 86. [Google Scholar] [CrossRef]
- Saito, R.; Jorio, A.; Souza Filho, A.; Dresselhaus, G.; Dresselhaus, M.; Pimenta, M. Probing phonon dispersion relations of graphite by double resonance Raman scattering. Phys. Rev. Lett. 2001, 88. [Google Scholar] [CrossRef]
- Irfan, B.; Sahoo, S.; Gaur, A.P.; Ahmadi, M.; Guinel, M.J.F.; Katiyar, R.S.; Chatterjee, R. Temperature dependent Raman scattering studies of three dimensional topological insulators Bi2Se3. J. Appl. Phys. 2014, 115. [Google Scholar] [CrossRef]
- Han, X.; Tao, H.; Gong, L.; Wang, X.; Zhao, X.; Yue, Y. Origin of the frequency shift of Raman scattering in chalcogenide glasses. J. Non-Cryst. Solids 2014, 391, 117–119. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, X.; Nilsson, L.; Wen, J.; Wang, G.; Shan, X.; Zhang, Q.; Zhang, S.; Jia, J.; Xue, Q. In situ Raman spectroscopy of topological insulator Bi2Te3 films with varying thickness. Nano Res. 2013, 6, 688–692. [Google Scholar] [CrossRef]
- Pine, A.S.; Dresselhaus, G. Raman spectra and lattice dynamics of tellurium. Phys. Rev. B 1971, 4, 356. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, J.; Utama, M.I.B.; Peng, B.; De la Mata, M.; Arbiol, J.; Xiong, Q. Exciton-phonon coupling in individual ZnTe nanorods studied by resonant Raman spectroscopy. Phys. Rev. B 2012, 85. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, J.; Zhao, M.; Yu, W.; Lu, Y.; Chen, C.; Xu, M.; Li, S.; Loh, K.P.; Qiaoliang, B. Raman Spectroscopy of Two-Dimensional Bi2TexSe3 − x Platelets Produced by Solvothermal Method. Materials 2015, 8, 5007-5017. https://doi.org/10.3390/ma8085007
Yuan J, Zhao M, Yu W, Lu Y, Chen C, Xu M, Li S, Loh KP, Qiaoliang B. Raman Spectroscopy of Two-Dimensional Bi2TexSe3 − x Platelets Produced by Solvothermal Method. Materials. 2015; 8(8):5007-5017. https://doi.org/10.3390/ma8085007
Chicago/Turabian StyleYuan, Jian, Meng Zhao, Wengzhi Yu, Yao Lu, Caiyun Chen, Meng Xu, Shaojuan Li, Kian Ping Loh, and Bao Qiaoliang. 2015. "Raman Spectroscopy of Two-Dimensional Bi2TexSe3 − x Platelets Produced by Solvothermal Method" Materials 8, no. 8: 5007-5017. https://doi.org/10.3390/ma8085007
APA StyleYuan, J., Zhao, M., Yu, W., Lu, Y., Chen, C., Xu, M., Li, S., Loh, K. P., & Qiaoliang, B. (2015). Raman Spectroscopy of Two-Dimensional Bi2TexSe3 − x Platelets Produced by Solvothermal Method. Materials, 8(8), 5007-5017. https://doi.org/10.3390/ma8085007




