Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fabrication of PLA Scaffolds
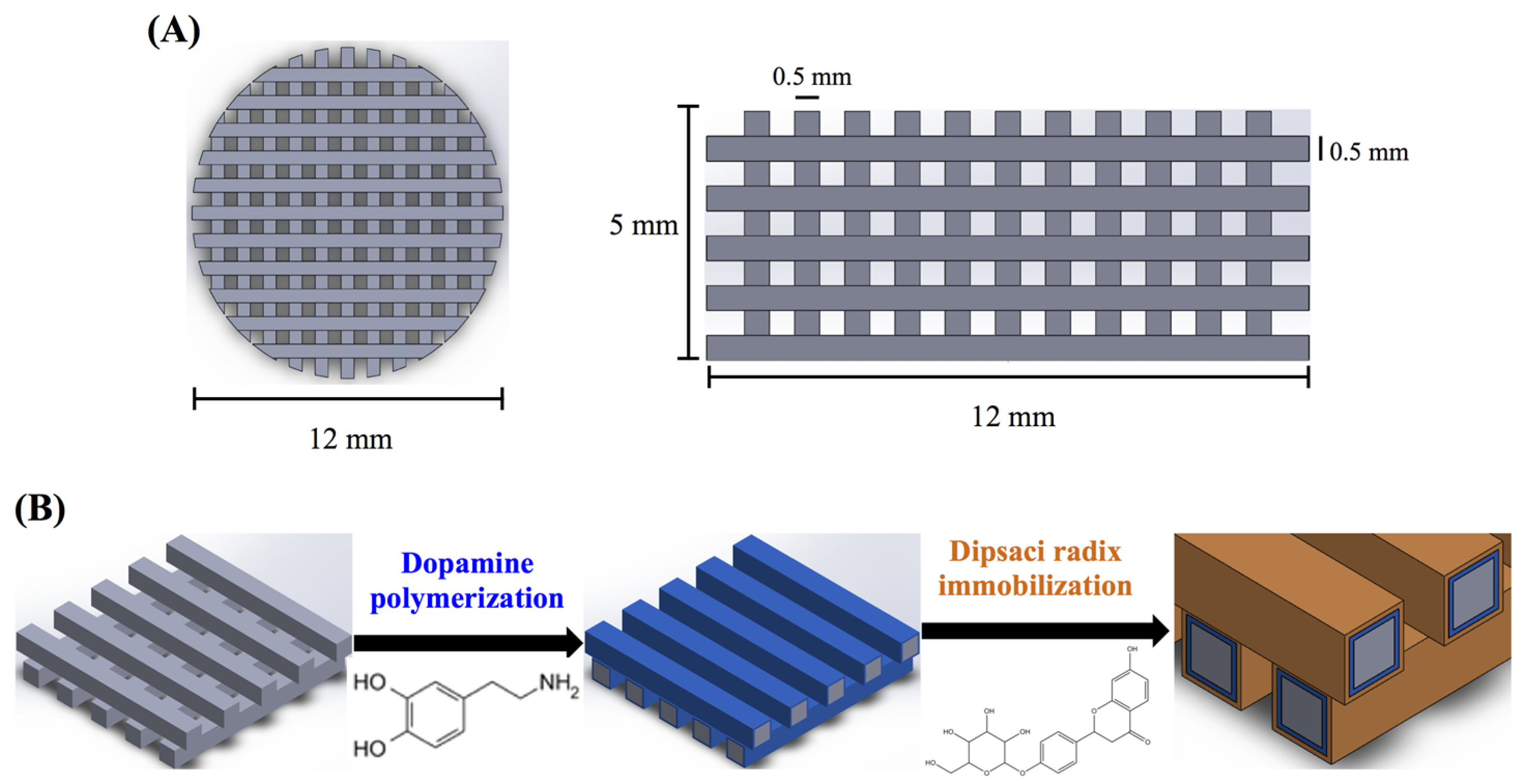
2.2. Preparation of Xu Duan (XD) Powder and Immobilization
2.3. PDA Coating
2.4. In Vitro Release of Xu Duan
2.5. Human Bone-Marrow Mesenchymal Stem Cell Culture
2.6. Cell Adhesion, Proliferation and Cytotoxicity
2.7. Cell Morphology
2.8. Western Blot
2.9. Real-Time PCR
| Genes | Primer Sequences |
|---|---|
| COL | Forward: 5’-AGAACAGCGTGGCCT-3’ |
| Reverse: 5’-TCCGGTGTGACTCGT-3’ | |
| ALP | Forward: 5’-TCAGAAGCTAACACCAACG-3’ |
| Reverse: 5’-TTGTACGTCTTGGAGAGGGC-3’ | |
| BSP | Forward: 5’-TCACCTGTGCCATACCAGTTAA-3’ |
| Reverse: TGAGATGGGTCAGGGTTTAGC-3’ | |
| OC | Forward: 5’-GCAAAGGTGCAGCCTTTGTG-3’ |
| Reverse: GGCTCCCAGCCATTGATACAG-3’ | |
| 18S | Forward: 5’-CGGAACTGAGGCCATGATTAAG-3’ |
| Reverse: 5’-GTATCTGATCGTCTTCGAACCTCC-3’ |
2.10. Alizarin Red S Stain
2.11. Angiogenesis-Related Protein Analysis
2.12. Statistical Analysis
3. Results and Discussion
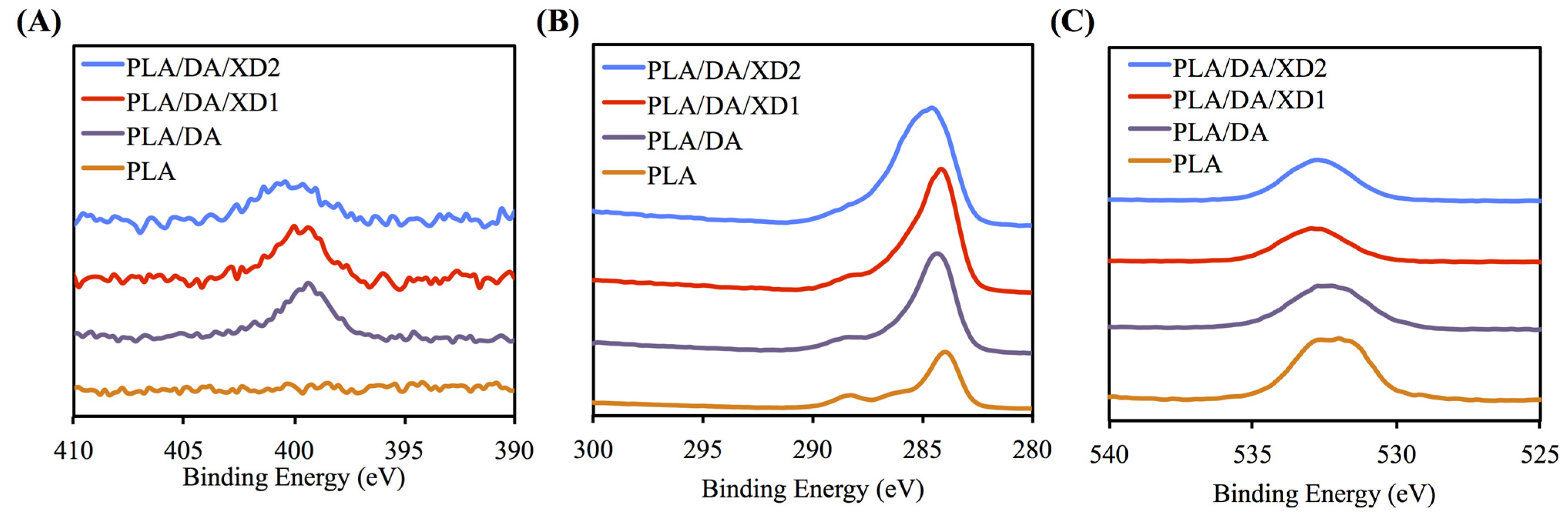
3.1. Characterization of PLA/PDA/XD Scaffolds
| Code | O1s (%) | C1s (%) | N1s (%) |
|---|---|---|---|
| Dopamine | 19.21 | 70.33 | 10.46 |
| XD | 27.69 | 72.31 | N.A. |
| PLA | 33.92 | 66.08 | N.A. |
| PLA/DA | 25.14 | 71.65 | 3.51 |
| PLA/DA/XD1 | 24.33 | 72.79 | 2.88 |
| PLA/DA/XD2 | 27.05 | 71.33 | 1.62 |

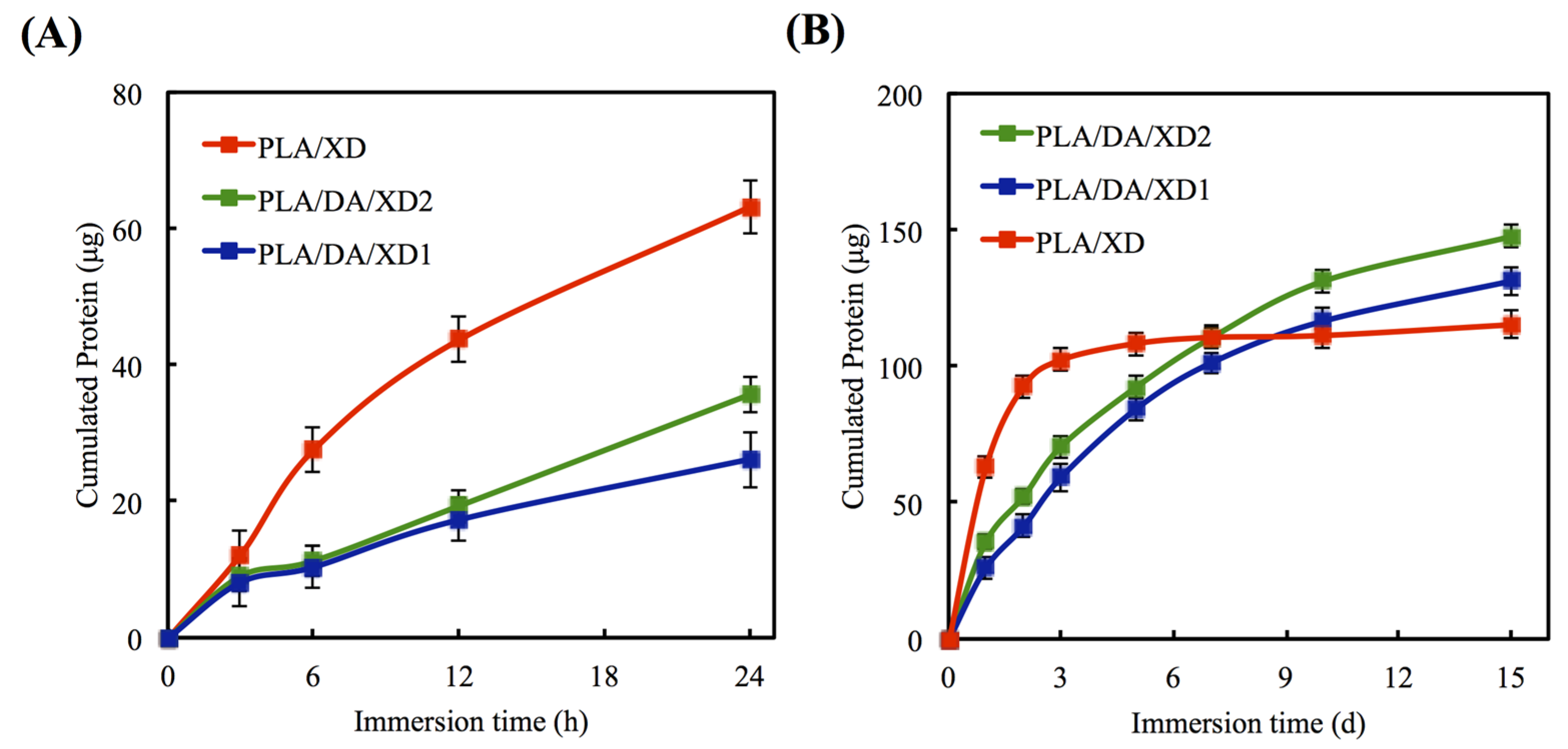
3.2. In Vitro Release of XD
3.3. Cell Adhesion and Morphology
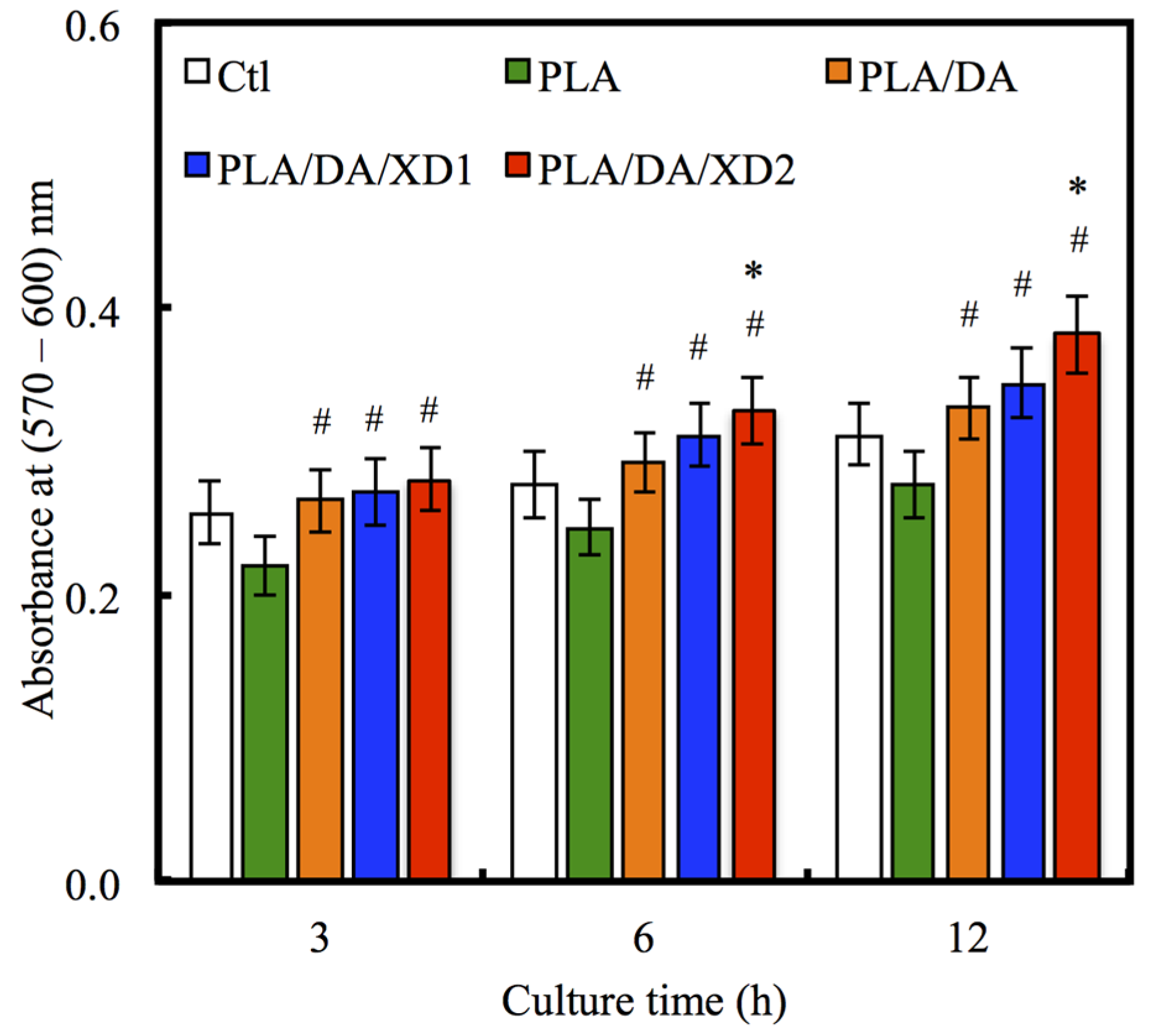

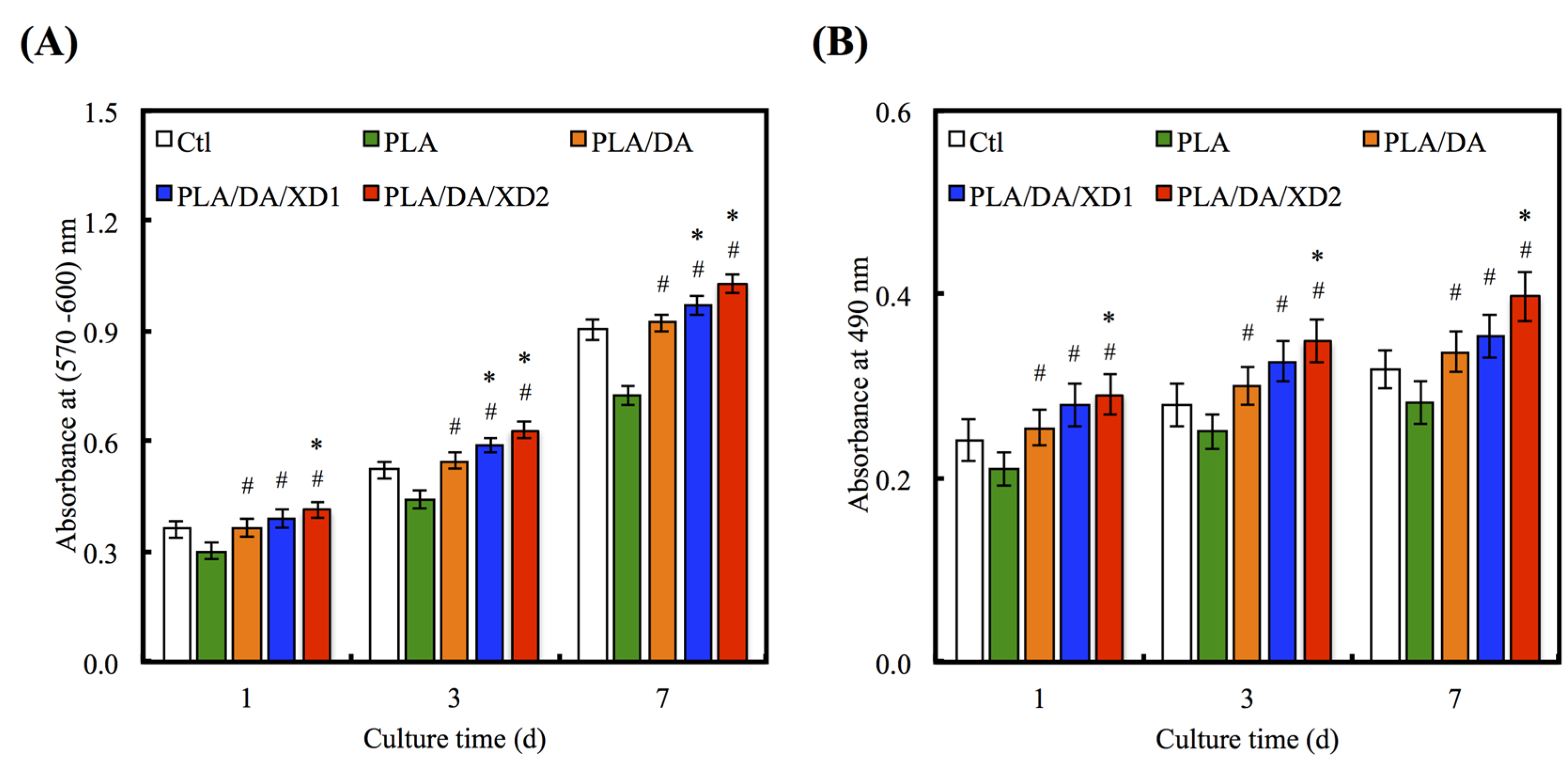
3.4. Cell Proliferation
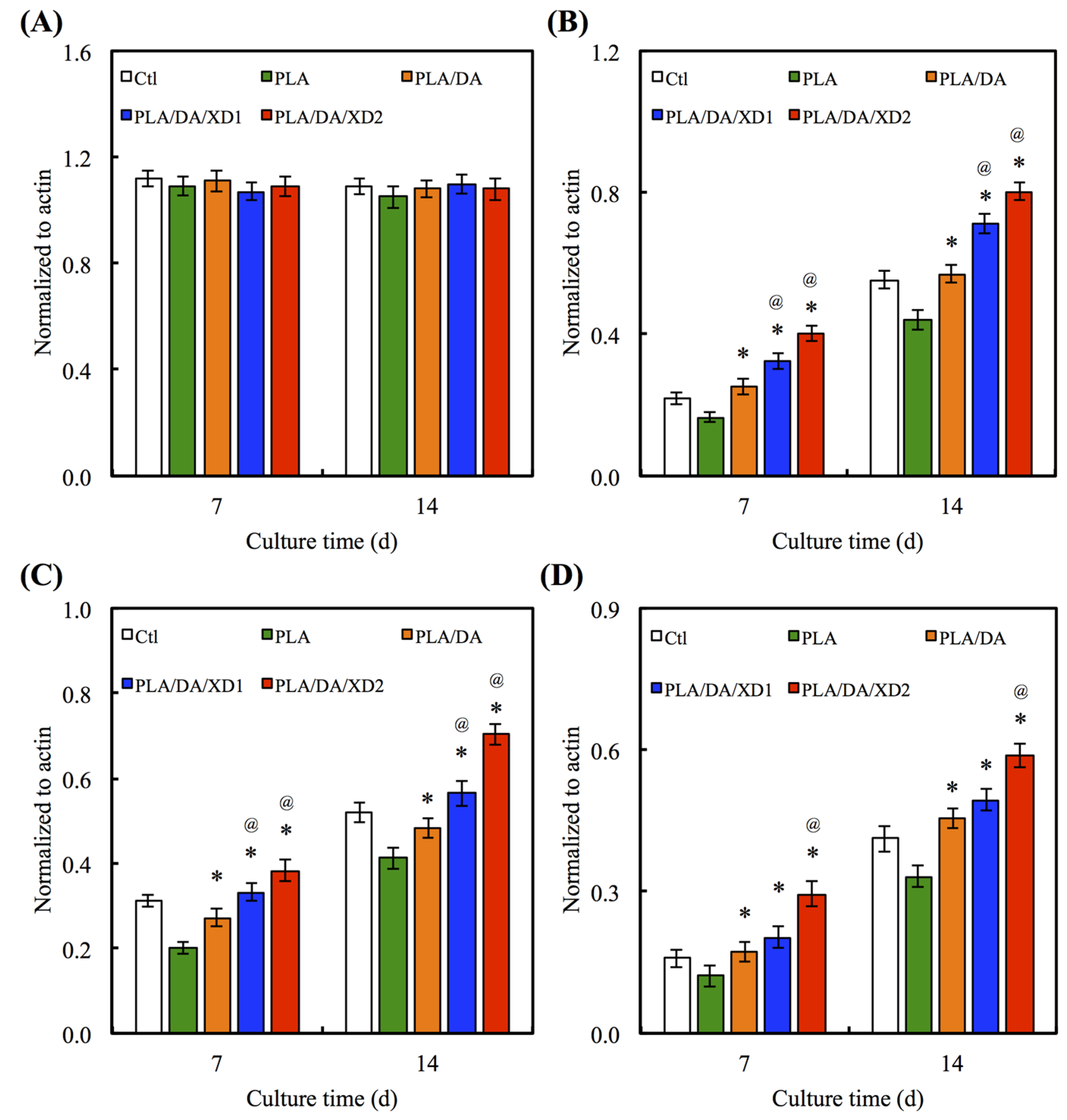
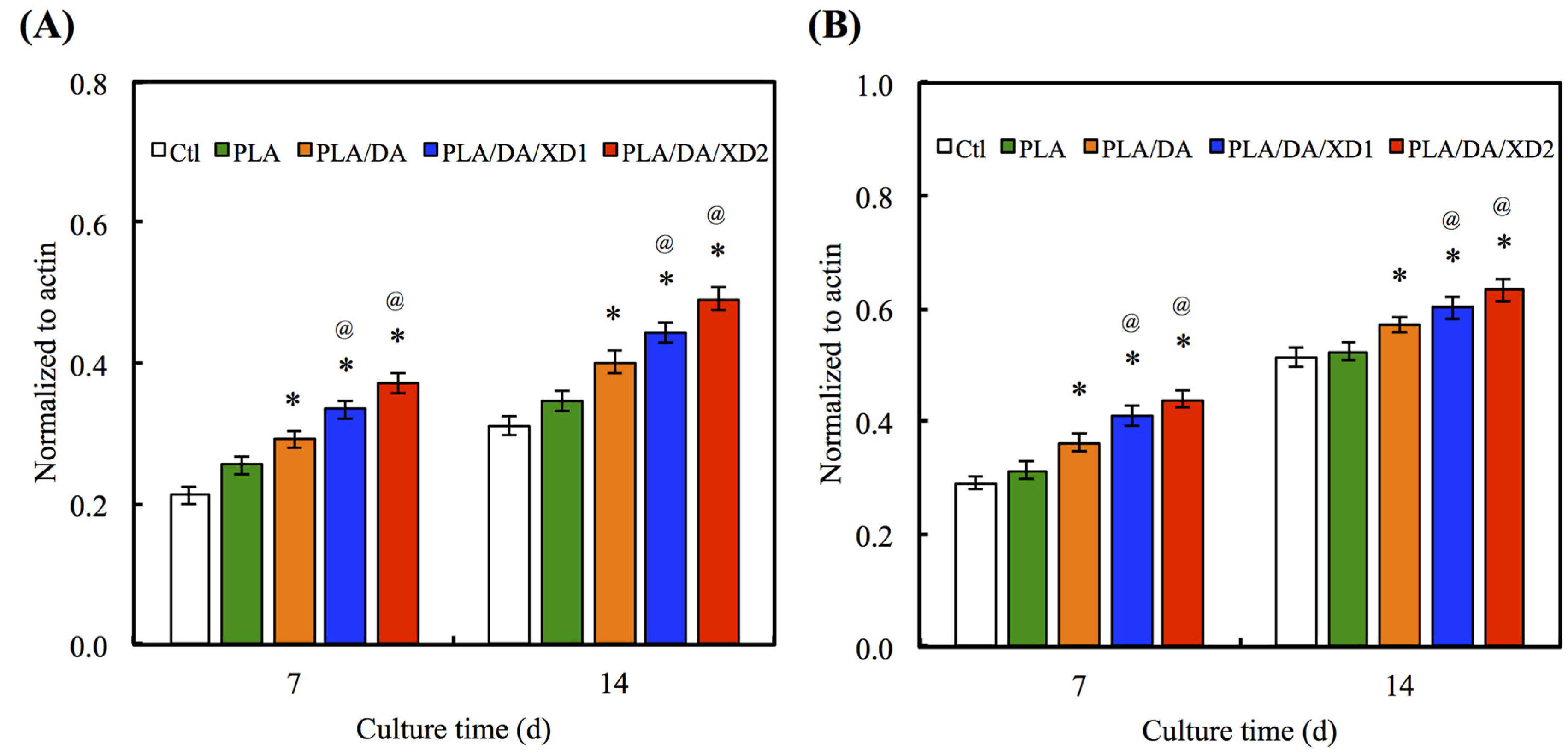
3.5. Osteogenesis Differentiation
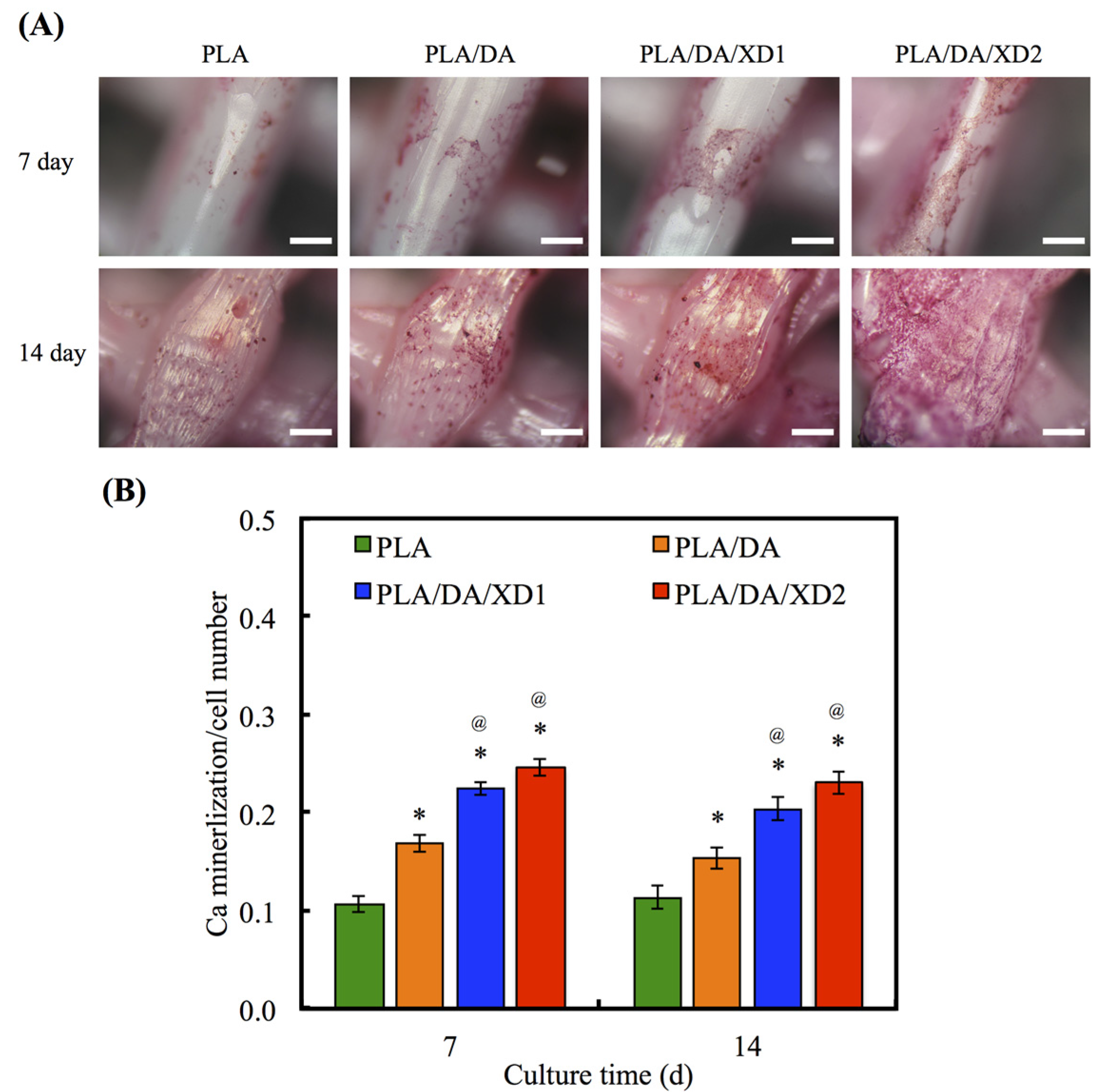
3.6. Calcium Deposition
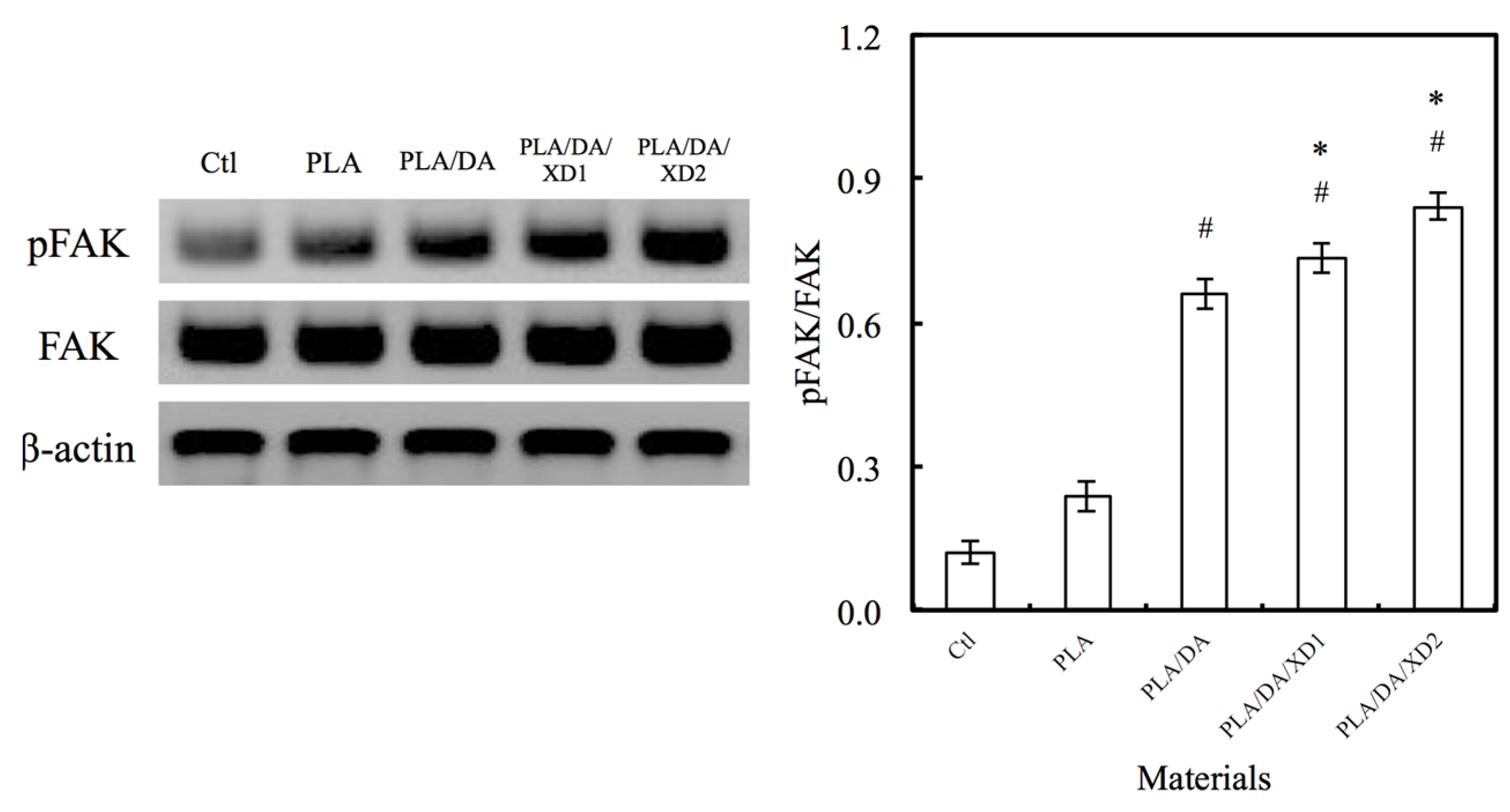
3.7. Angiogenesis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- O’Brien, C.; Holmes, B.; Faucett, S.; Zhang, L.G. 3D printing of nanomaterial scaffolds for complex tissue regeneration. Tissue Eng. Part B Rev. 2014, 21, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Temple, J.P.; Hutton, D.L.; Hung, B.P.; Huri, P.Y.; Cook, C.A.; Kondragunta, R.; Jia, X.; Grayson, W.L. Engineering anatomically shaped vascularized bone grafts with hASCs and 3D-printed PCL scaffolds. J. Biomed. Mater. Res. Part A 2014, 102, 4317–4325. [Google Scholar] [CrossRef] [PubMed]
- Mikos, A.; Herring, S.; Ochareon, P. Engineering complex tissues. Tissue Eng. 2006, 12, 3307–3339. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Fu, S.J.; Lin, Y.C.; Yang, I.K.; Gu, Y. Chitosan-coated electrospun PLA fibers for rapid mineralization of calcium phosphate. Int. J. Biol. Macromol. 2014, 68, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Wu, C.; Chang, J. Dual drug release from electrospun poly(lactic-co-glycolic acid)/mesoporous silica nanoparticles composite mats with distinct release profiles. Acta Biomater. 2012, 8, 1901–1907. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Luo, Y.; Cuniberti, G.; Xiao, Y.; Gelinsky, M. Three-dimensional printing of hierarchical and tough mesoporous bioactive glass scaffolds with a controllable pore architecture, excellent mechanical strength and mineralization ability. Acta Biomater. 2011, 7, 2644–2650. [Google Scholar] [CrossRef] [PubMed]
- Pfister, A.; Landers, R.; Laib, A.; Hübner, U.; Schmelzeisen, R.; Hülhaupt, R. Biofunctional rapid prototyping for tissue-engineering applications: 3D bioplotting versus 3D printing. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 624–638. [Google Scholar] [CrossRef]
- Inzana, J.A.; Olvera, D.; Fuller, S.M.; Kelly, J.P.; Graeve, O.A.; Schwarz, E.M.; Kates, S.L.; Awad, H.A. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials 2014, 35, 4026–4034. [Google Scholar] [CrossRef] [PubMed]
- Fielding, G.A.; Bandyopadhyay, A.; Bose, S. Effects of silica and zinc oxide doping on mechanical and biological properties of 3D printed tricalcium phosphate tissue engineering scaffolds. Dent. Mater. 2012, 28, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Moussa, M.; Carrel, J.P.; Scherrer, S.; Cattani-Lorente, M.; Wiskott, A.; Durual, S. Medium-term function of a 3D printed TCP/HA structure as a new osteoconductive scaffold for vertical bone augmentation: A simulation by BMP-2 activation. Materials 2015, 8, 2174–2190. [Google Scholar] [CrossRef]
- Kai, D.; Liow, S.S.; Loh, X.J. Biodegradable polymers for electrospinning: Towards biomedical applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 45, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.T.; Lin, C.C.; Chen, Y.W.; Yeh, C.H.; Fang, H.Y.; Shie, M.Y. Poly(dopamine) coating of 3D printed poly(lactic acid) scaffolds for bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 56, 165–173. [Google Scholar] [CrossRef]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ai, K.; Lu, L. Polydopamine and its derivative materials: synthesis and promising applications in energy, environmental, and biomedical fields. Chem. Rev. 2014, 114, 5057–5115. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Qi, P.; Wen, F.; Li, X.; Xia, Q.; Maitz, M.F.; Yang, Z.; Shen, R.; Tu, Q.; Huang, N. Mussel-inspired one-step adherent coating rich in amine groups for covalent immobilization of heparin: Hemocompatibility, growth behaviors of vascular cells, and tissue response. ACS Appl. Mater. Interfaces 2014, 6, 14608–14620. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Yang, L.; Lin, M.-F.; Ma, J.; Lu, X.; Lee, P.S. Polydopamine spheres as active templates for convenient synthesis of various nanostructures. Small 2013, 9, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Han, P.; Liu, X.; Xu, M.; Tian, T.; Chang, J.; Xiao, Y. Mussel-inspired bioceramics with self-assembled Ca-P/polydopamine composite nanolayer: Preparation, formation mechanism, improved cellular bioactivity and osteogenic differentiation of bone marrow stromal cells. Acta Biomater. 2014, 10, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Qu, S.; Zheng, X.; Xiong, X.; Fu, R.; Tang, K.; Zhong, Z.; Weng, J. Effect of polydopamine on the biomimetic mineralization of mussel-inspired calcium phosphate cement in vitro. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 44, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Li, L.; Ostrovidov, S.; Shu, Y.; Khademhosseini, A.; Wu, H. Stretchable and micropatterned membrane for osteogenic differentation of stem cells. ACS Appl. Mater. Interfaces 2014, 6, 11915–11923. [Google Scholar] [CrossRef] [PubMed]
- Su, C.C.; Kao, C.T.; Hung, C.J.; Chen, Y.J.; Huang, T.H.; Shie, M.Y. Regulation of physicochemical properties, osteogenesis activity, and fibroblast growth factor-2 release ability of β-tricalcium phosphate for bone cement by calcium silicate. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 37, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Hung, C.J.; Huang, T.H.; Lin, C.C.; Kao, C.T.; Shie, M.Y. Odontogenic differentiation of human dental pulp cells by calcium silicate materials stimulating via FGFR/ERK signaling pathway. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 43, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.C.; Chen, H.M.; Yao, C.H. A novel bone substitute composite composed of tricalcium phosphate, gelatin and drynaria fortunei herbal extract. J. Biomed. Mater. Res. Part A 2008, 84, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.; Lin, Y. Traditional Chinese medicine: An approach to scientific proof and clinical validation. Pharmacol. Ther. 2000, 86, 191–198. [Google Scholar] [CrossRef]
- Wang, J.F.; Zhou, H.; Han, L.Y.; Chen, X.; Chen, Y.Z. Traditional Chinese medicine information database. Clin. Pharmacol. Ther. 2005, 78, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.G.; Zhang, R.; Li, C.; Ma, X.; Liu, L.; Wang, J.P.; Mei, Q.B. The osteoprotective effect of Radix Dipsaci extract in ovariectomized rats. J. Ethnopharmacol. 2009, 123, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.W.K.; Rabie, A.B.M.; Hägg, E.U.O. The effect of crude extract from Radix Dipsaci on bone in mice. Phytother. Res. 2007, 21, 596–598. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.H.; Tsai, H.M.; Chen, Y.S.; Liu, B.S. Fabrication and evaluation of a new composite composed of tricalcium phosphate, gelatin, and Chinese medicine as a bone substitute. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 75, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Shie, M.Y.; Huang, T.H.; Kao, C.T.; Huang, C.H.; Ding, S.J. The effect of a physiologic solution pH on properties of white mineral trioxide aggregate. J. Endod. 2009, 35, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Shie, M.Y.; Ding, S.J.; Chang, H.C. The role of silicon in osteoblast-like cell proliferation and apoptosis. Acta Biomater. 2011, 7, 2604–2614. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Shie, M.Y.; Hung, C.J.; Wu, B.C.; Liu, S.L.; Huang, T.H.; Kao, C.T. Activation of focal adhesion kinase induces extracellular signal- regulated kinase-mediated osteogenesis in tensile force-subjected periodontal ligament fibroblasts but not in osteoblasts. J. Bone Miner. Metab. 2013, 32, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.H.; Ryu, J.; Hong, S.K.; Lee, H.; Park, C.B. General functionalization route for cell adhesion on non-wetting surfaces. Biomaterials 2010, 31, 2535–2541. [Google Scholar] [CrossRef] [PubMed]
- Rim, N.G.; Kim, S.J.; Shin, Y.M.; Jun, I.; Lim, D.W.; Park, J.H.; Shin, H. Mussel-inspired surface modification of poly(L-lactide) electrospun fibers for modulation of osteogenic differentiation of human mesenchymal stem cells. Colloids Surf. B 2012, 91, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; McCloskey, B.D.; Choi, T.H.; Lee, C.; Kim, M.-J.; Freeman, B.D.; Park, H.B. Oxygen concentration control of dopamine-induced high uniformity surface coating chemistry. ACS Appl. Mater. Interfaces 2013, 5, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Cheng, L.; Zhao, J.; Jin, R.; Sun, B.; Shi, Y.; Zhang, L.; Zhang, Y.; Cui, W. bFGF-grafted electrospun fibrous scaffolds via poly(dopamine) for skin wound healing. J. Mater. Chem. B 2014, 2, 3636–3645. [Google Scholar] [CrossRef]
- Kao, C.T.; Huang, T.H.; Chen, Y.J.; Hung, C.J.; Lin, C.C.; Shie, M.Y. Using calcium silicate to regulate the physicochemical and biological properties when using β-tricalcium phosphate as bone cement. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 43, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.F.; Lin, C.C.; Huang, T.H.; Chou, M.Y.; Yang, J.J.; Shie, M.Y. Osteogenesis and angiogenesis properties of dental pulp cell on novel injectable tricalcium phosphate cement by silica doped. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 42, 672–680. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Huang, T.H.; Hung, C.J.; Lai, W.Y.; Kao, C.T.; Shie, M.Y. The synergistic effects of fibroblast growth factor-2 and mineral trioxide aggregate on an osteogenic accelerator in vitro. Int. Endod. J. 2014, 47, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chang, J. Stimulation of proangiogenesis by calcium silicate bioactive ceramic. Acta Biomater. 2013, 9, 5379–5389. [Google Scholar] [CrossRef] [PubMed]
- Chien, C.Y.; Tsai, W.B. Poly(dopamine)-assisted immobilization of Arg-Gly-Asp peptides, hydroxyapatite, and bone morphogenic protein-2 on titanium to improve the osteogenesis of bone marrow stem cells. ACS Appl. Mater. Interfaces 2013, 5, 6975–6983. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.F.; Yao, D.; Yuan, H.; Zhang, S.; Tian, J.; Guo, W. Dipsacus asperoides polysaccharide induces apoptosis in osteosarcoma cells by modulating the PI3K/Akt pathway. Carbohyd. Polym. 2013, 95, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Jung, J.K.; Son, K.H.; Lee, D.H.; Kang, T.M. Inhibitory effects of the root extract of Dipsacus asperoides C.Y. Cheng et al T.M.Ai on collagen-induced arthritis in mice. J. Ethnopharmacol. 2012, 139, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Shie, M.Y.; Ding, S.J. Integrin binding and MAPK signal pathways in primary cell responses to surface chemistry of calcium silicate cements. Biomaterials 2013, 34, 6589–6606. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhang, Y.; Zhou, Y.Z.; Fan, W.; Xiao, Y. A comparative study of mesoporous glass/silk and non-mesoporous glass/silk scaffolds: Physiochemistry and in vivo osteogenesis. Acta Biomater. 2011, 7, 2229–2236. [Google Scholar] [CrossRef] [PubMed]
- Mavrogonatou, E.; Kletsas, D. Differential response of nucleus pulposus intervertebral disc cells to high salt, sorbitol, and urea. J. Cell Phys. 2011, 227, 1179–1187. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.W.; Isenberg, J.S.; Roberts, D.D. Molecular regulation of tumor angiogenesis and perfusion via redox signaling. Chem. Rev. 2009, 109, 3099–3124. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Lin, K.; Jiang, X.; Fang, B.; Xu, Y.; Liu, J.; Zeng, D.; Zhang, M.; Zhang, X.; Chang, J.; Zhang, Z. Effect of nano-structured bioceramic surface on osteogenic differentiation of adipose derived stem cells. Biomaterials 2014, 35, 8514–8527. [Google Scholar] [CrossRef] [PubMed]
- Bi, C.W.C.; Xu, L.; Tian, X.Y.; Liu, J.; Zheng, K.Y.Z.; Lau, C.W.; Lau, D.T.W.; Choi, R.C.Y.; Dong, T.T.X.; Huang, Y.; et al. Fo Shou San, an ancient chinese herbal decoction, protects endothelial function through increasing endothelial nitric oxide synthase activity. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.R.; Black, R.; Kielty, C. PCL-PU composite vascular scaffold production for vascular tissue engineering: Attachment, proliferation and bioactivity of human vascular endothelial cells. Biomaterials 2006, 27, 3608–3616. [Google Scholar] [CrossRef] [PubMed]
- Shome, S.; Dasgupta, P.S.; Basu, S. Dopamine regulates mobilization of mesenchymal stem cells during wound angiogenesis. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Vohra, P.K.; Bhattacharya, R.; Dutta, S.; Sinha, S.; Mukhopadhyay, D. Dopamine regulates phosphorylation of VEGF receptor 2 by engaging Src-homology-2-domain-containing protein tyrosine phosphatase 2. J. Cell Sci. 2009, 122, 3385–3392. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeh, C.-H.; Chen, Y.-W.; Shie, M.-Y.; Fang, H.-Y. Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells. Materials 2015, 8, 4299-4315. https://doi.org/10.3390/ma8074299
Yeh C-H, Chen Y-W, Shie M-Y, Fang H-Y. Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells. Materials. 2015; 8(7):4299-4315. https://doi.org/10.3390/ma8074299
Chicago/Turabian StyleYeh, Chia-Hung, Yi-Wen Chen, Ming-You Shie, and Hsin-Yuan Fang. 2015. "Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells" Materials 8, no. 7: 4299-4315. https://doi.org/10.3390/ma8074299
APA StyleYeh, C.-H., Chen, Y.-W., Shie, M.-Y., & Fang, H.-Y. (2015). Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells. Materials, 8(7), 4299-4315. https://doi.org/10.3390/ma8074299







