Nanoindentation and XPS Studies of Titanium TNZ Alloy after Electrochemical Polishing in a Magnetic Field
Abstract
:1. Introduction
2. Experimental Section
2.1. Sample Preparation and SEM Studies
2.2. Nanoindentation Testing
2.3. XPS Studies
3. Results
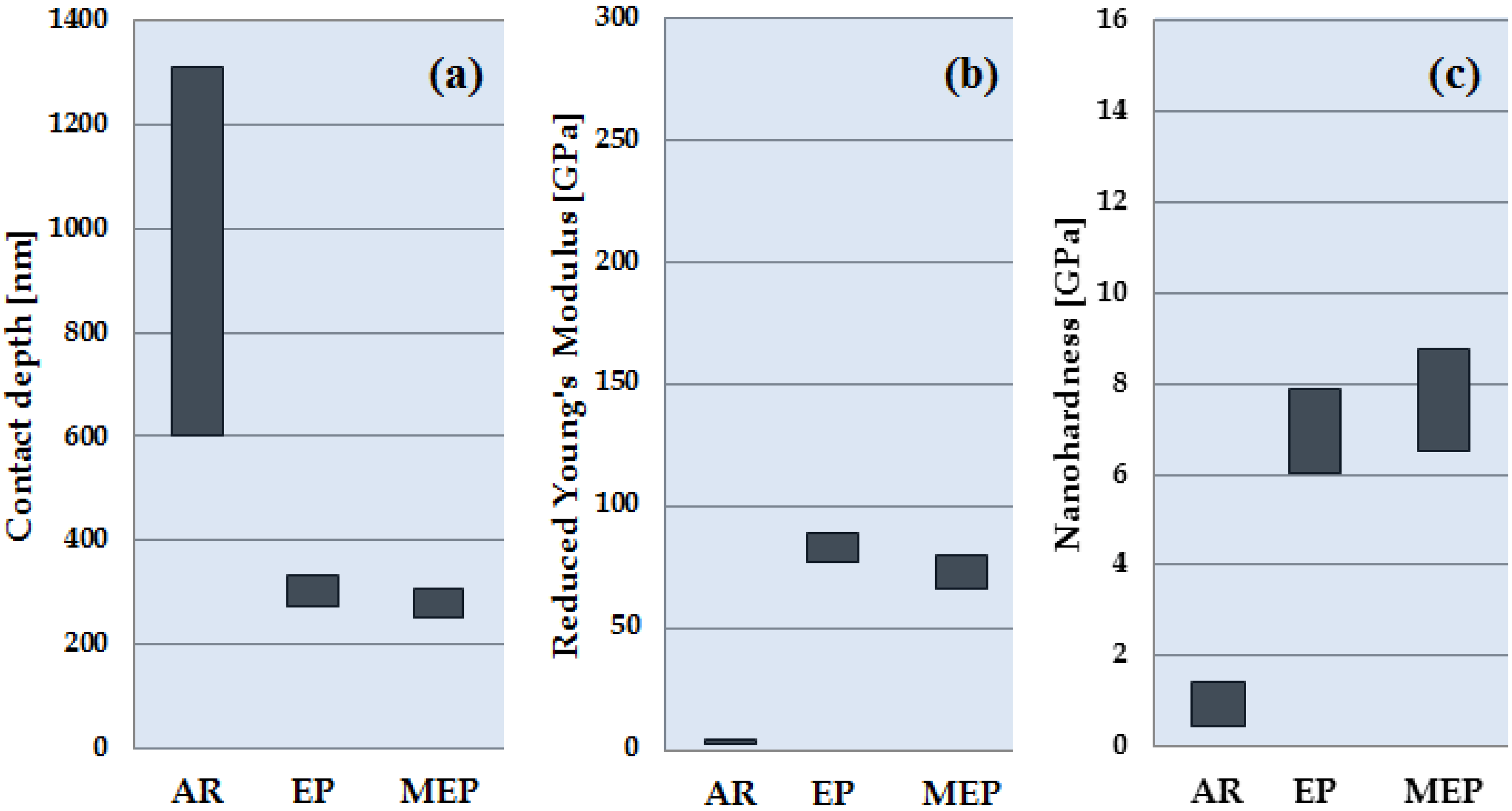
3.1. Nanoindentation Results

| Treatment | Contact Depth (nm) | Reduced Young’s Modulus Er (GPa) | Nanohardness nH (GPa) |
|---|---|---|---|
| AR | 957.09 ± 355.21 | 3.61 ± 0.98 | 0.94 ± 0.48 |
| EP | 303.87 ± 28.79 | 83.08 ± 5.73 | 6.97 ± 0.92 |
| MEP | 280.67 ± 27.53 | 72.95 ± 7.21 | 7.64 ± 1.14 |
3.2. XPS Results
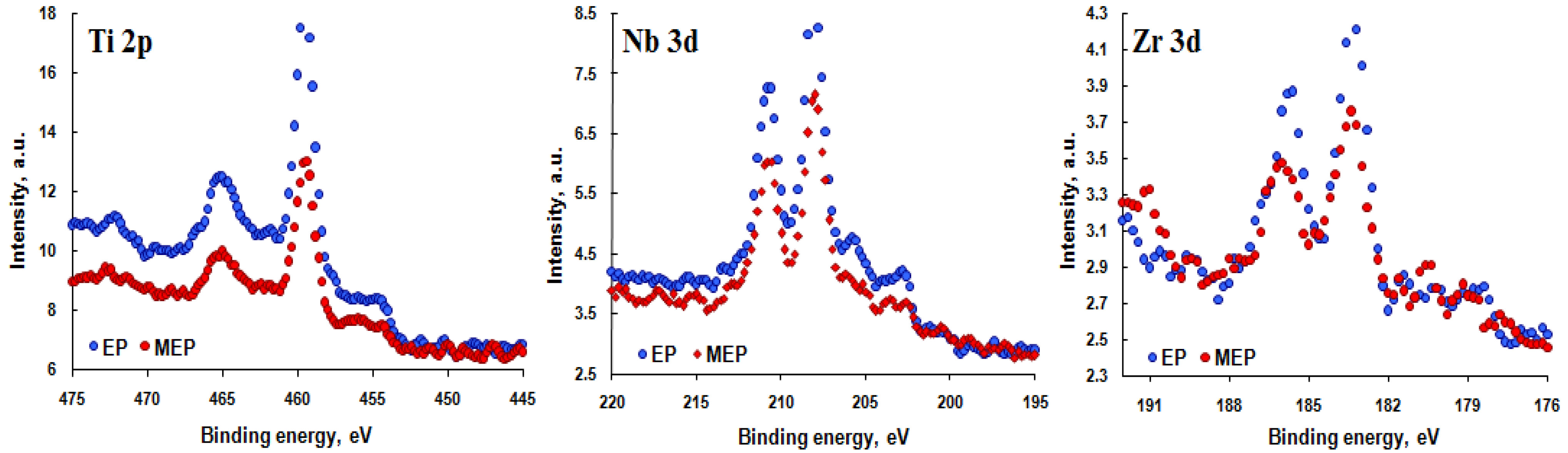

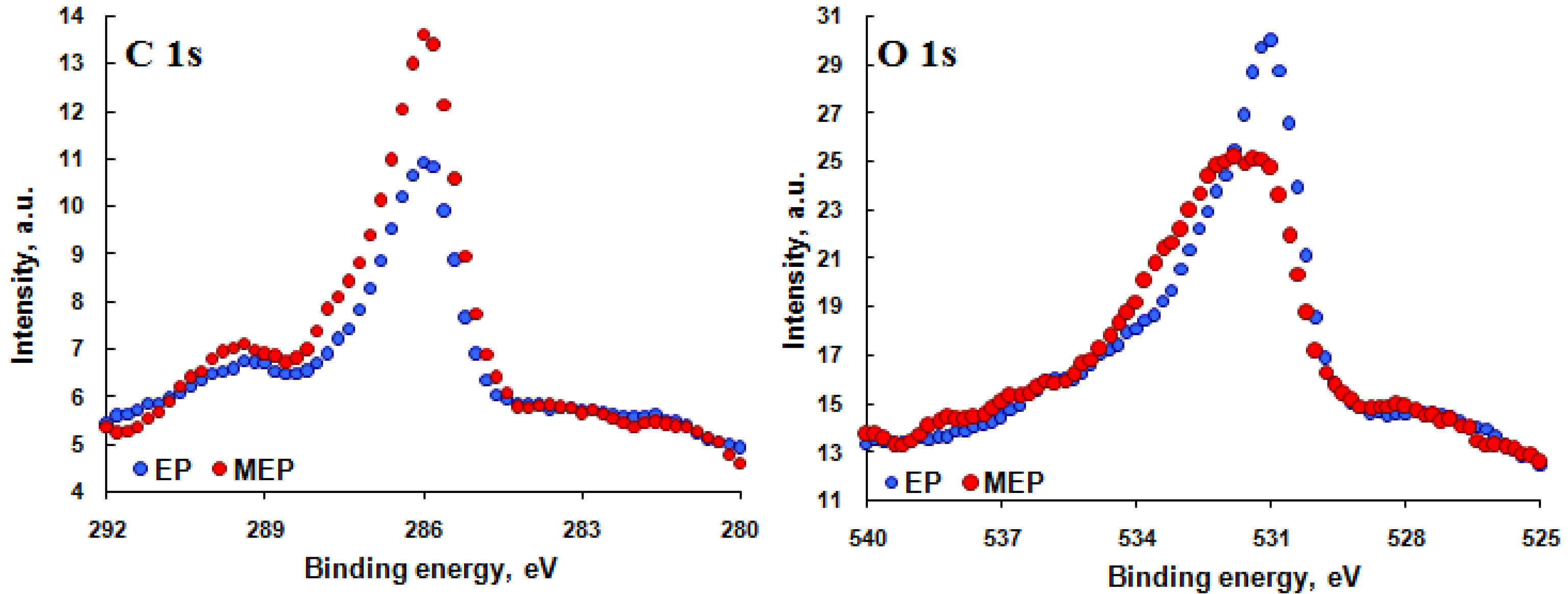
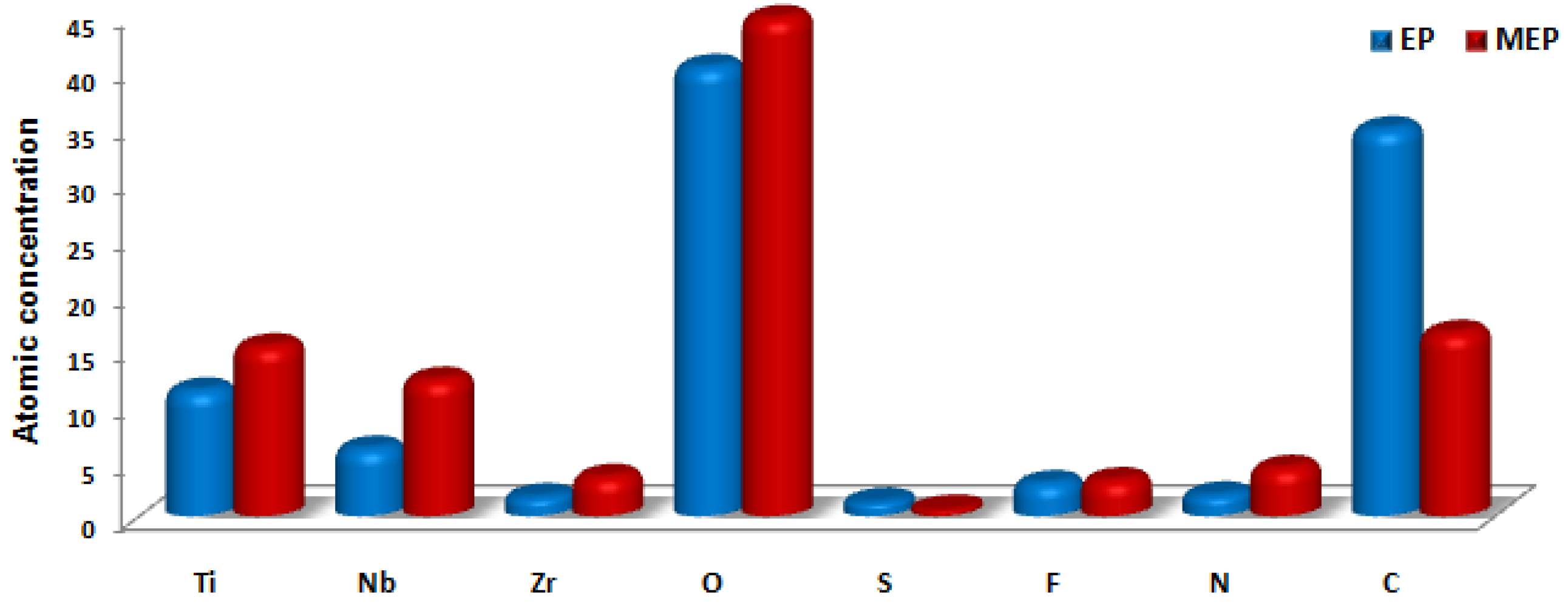
| Element | EP | MEP |
|---|---|---|
| C 1s | 35.1 | 16.4 |
| F 1s | 2.9 | 3.2 |
| N 1s | 2.0 | 4.3 |
| Nb 3d | 6.0 | 12.2 |
| O 1s | 39.9 | 44.6 |
| S 2p | 1.3 | 0.6 |
| Ti 2p | 11.1 | 15.2 |
| Zr 3d | 1.7 | 3.5 |
| Ratio | Matrix | EP | MEP |
|---|---|---|---|
| Ti/(Ti + Nb + Zr) | 0.74 | 0.59 | 0.49 |
| Nb/(Ti + Nb + Zr) | 0.20 | 0.32 | 0.39 |
| Zr/(Ti + Nb + Zr) | 0.06 | 0.09 | 0.12 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pulletikurthi, C.; Munroe, N.; Stewart, D.; Haider, W.; Amruthaluri, S.; Rokicki, R.; Dugrot, M.; Ramaswamy, S. Utility of magneto-electropolished ternary nitinol alloys for blood contacting applications. J. Biomed. Mater. Res. B: Appl. Biomater. 2014. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokosz, K.; Valíček, J.; Rokicki, R. Effect of magnetoelectropolishing on nanohardness and Young’s modulus of titanium biomaterial. Mater. Lett. 2012, 83, 69–72. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Konarski, P.; Rokicki, R.; Valíček, J. SIMS studies of titanium biomaterial hydrogenation after magnetoelectropolishing. Surface Coat. Technol. 2012, 206, 4027–4031. [Google Scholar] [CrossRef]
- Rokicki, R.; Haider, W.; Hryniewicz, T. Influence of sodium hypochlorite treatment of electropolished and magnetoelectropolished nitinol surfaces on adhesion and proliferation of MC3T3 pre-osteoblast cells. J. Mater. Sci.: Mater. Med. 2012, 23, 2127–2139. [Google Scholar] [CrossRef]
- Rokicki, R. The passive oxide film on electropolished titanium. Metal Finish. 1990, 88, 65–66. [Google Scholar]
- Kuhn, A. The electropolishing of titanium and its alloys. Metal Finish. 2004, 102, 80–86. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokicki, R.; Rokosz, K. Corrosion and surface characterization of titanium biomaterial after magnetoelectropolishing. Surface Coat. Technol. 2009, 203, 1508–1515. [Google Scholar] [CrossRef]
- Simka, W.; Kaczmarek, M.; Baron-Wiecheć, A.; Nawrat, G.; Marciniak, J.; Żak, J. Electropolishing and passivation of NiTi shape memory alloy. Electrochim. Acta 2010, 55, 2437–2441. [Google Scholar] [CrossRef]
- Simka, W.; Mosiałek, M.; Nawrat, G.; Nowak, P.; Żak, J.; Szade, J.; Winiarski, A.; Maciej, A.; Szyk-Warszyńska, L. Electrochemical polishing of Ti-13Nb-13Zr Alloy. Surface Coat. Technol. 2012, 213, 239–246. [Google Scholar] [CrossRef]
- Rokicki, R.; Hryniewicz, T.; Rokosz, K. Modifying metallic implants with magnetoelectropolishing. Med. Dev. Diagn. Ind. 2008, 30, 102–111. [Google Scholar]
- Hryniewicz, T.; Rokicki, R.; Rokosz, K. Magnetoelectropolished Titanium Biomaterial; InTech Europe: Rijeka, Croatia, 2011; Chapter 11; pp. 227–248. [Google Scholar]
- Hryniewicz, T.; Rokicki, R.; Rokosz, K. Magnetoelectropolishing for metal surface modification. Trans. Instit. Metal Finish. 2007, 85, 325–332. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokosz, K. On the wear inspection and endurance recovery of nitinol endodontic files. Pomiary Automatyka Kontrola 2009, 55, 247–250. [Google Scholar]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Pluta, Z.; Hryniewicz, T. Quantitative determination of material hardness. J. Quant. Inf. Sci. 2011, 1, 127–134. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokicki, R.; Rokosz, K. Magnetoelectropolishing process improves characteristics of finished metal surfaces. Metal Finish. 2006, 104, 26–33. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokosz, K.; Rokicki, R. Surface investigation of NiTi rotary endodontic instruments after magnetoelectropolishing. In Proceedings of the MRS Proceedings, Biomaterials of XVIII International Materials Research Congress, 9, Biomaterials, Cancun, Mexico, 16–20 August 2009; pp. 21–32.
- Rokosz, K.; Hryniewicz, T.; Valíček, J.; Harničárová, M.; Vyležík, M. Nanoindentation measurements of AISI 316L biomaterial samples after annual immersion in Ringer’s solution followed by electrochemical polishing in a magnetic field. Pomiary Automatyka Kontrola 2012, 58, 460–463. [Google Scholar]
- Hryniewicz, T.; Rokosz, K. Highlights of magnetoelectropolishing. Front. Mater. 2014, 1, 1–7. [Google Scholar] [CrossRef]
- Shuman, D. Computerized image analysis software for measuring indents by AFM. Microscopy-Analysis, P 21, (May 2005). In Nanoindentation; Fischer-Cripps, A.C., Ed.; Springer: New York, NY, USA, 2004. [Google Scholar]
- Oliver, W.C.; Pharr, G.M. Measurement of hardness and elastic modulus by instrumented indentation: Advances in understanding and refinements to methodology. J. Mater. Res. 2004, 19, 3–27. [Google Scholar] [CrossRef]
- Chuang, L.C.; Luo, C.H.; Yang, S. The structure and mechanical properties of thick rutile-TiO2 films using different coating treatments. Appl. Surface Sci. 2011, 258, 297–303. [Google Scholar] [CrossRef]
- Cheng, Y.-T.; Cheng, C.-M. Scaling, dimensional analysis, and indentation measurements. Mater. Sci. Eng. R 2004, 44, 91–149. [Google Scholar] [CrossRef]
- Rokicki, R. Apparatus and method for enhancing electropolishing utilizing magnetic fields. US Patent 7632390, 15 December 2009. [Google Scholar]
- Technical documentation and manual of HYSITRON™. Available online: http://www.hysitron.com (accessed on 14 December 2013).
- Marteleur, M.; Sun, F.; Gloriant, T.; Vermaut, P.; Jacques, P.J.; Prima, F. On the design of new β-metastable titanium alloys with improved work hardening rate thanks to simultaneous TRIP and TWIP effects. Scripta Mater. 2012, 66, 749–752. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokosz, K.; Rokicki, R. Magnetic fields for electropolishing improvement: Materials and systems. Int. Lett. Chem. Phys. Astron. 2014, 4, 98–108. [Google Scholar]
- Crist, B.V. Handbook of Monochromatic XPS Spectra, The Elements and Native Oxides; John Wiley & Sons, Ltd.: Mountain View, CA, USA, 2000. [Google Scholar]
- StatSoft Inc. STATISTICA (data analysis software system), version 10. 2011. Available online: http://www.statsoft.com (accessed on 5 May 2013).
- Wang, S.J.; Ong, C.K. Epitaxial Y-stabilized ZrO2 films on silicon: Dynamic growth process and interface structure. Appl. Phys. Lett. 2002, 80, 2541–2543. [Google Scholar] [CrossRef]
- Reddy, P.A.K.; Reddy, P.V.L.; Sharma, V.M.; Srinivas, B.; Kumari, V.D.; Subrahmanyam, M. Photocatalytic degradation of isoproturon pesticide on C, N and S Doped TiO2. J. Water Resour. Protect. 2010, 2, 235–244. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hryniewicz, T.; Rokosz, K.; Rokicki, R.; Prima, F. Nanoindentation and XPS Studies of Titanium TNZ Alloy after Electrochemical Polishing in a Magnetic Field. Materials 2015, 8, 205-215. https://doi.org/10.3390/ma8010205
Hryniewicz T, Rokosz K, Rokicki R, Prima F. Nanoindentation and XPS Studies of Titanium TNZ Alloy after Electrochemical Polishing in a Magnetic Field. Materials. 2015; 8(1):205-215. https://doi.org/10.3390/ma8010205
Chicago/Turabian StyleHryniewicz, Tadeusz, Krzysztof Rokosz, Ryszard Rokicki, and Frédéric Prima. 2015. "Nanoindentation and XPS Studies of Titanium TNZ Alloy after Electrochemical Polishing in a Magnetic Field" Materials 8, no. 1: 205-215. https://doi.org/10.3390/ma8010205
APA StyleHryniewicz, T., Rokosz, K., Rokicki, R., & Prima, F. (2015). Nanoindentation and XPS Studies of Titanium TNZ Alloy after Electrochemical Polishing in a Magnetic Field. Materials, 8(1), 205-215. https://doi.org/10.3390/ma8010205








