Effect of Organo-Modified Nanoclay on the Thermal and Bulk Structural Properties of Poly(3-hydroxybutyrate)-Epoxidized Natural Rubber Blends: Formation of Multi-Components Biobased Nanohybrids
Abstract
:1. Introduction
2. Results and Discussion
2.1. Surface Analysis and Bulk Structure
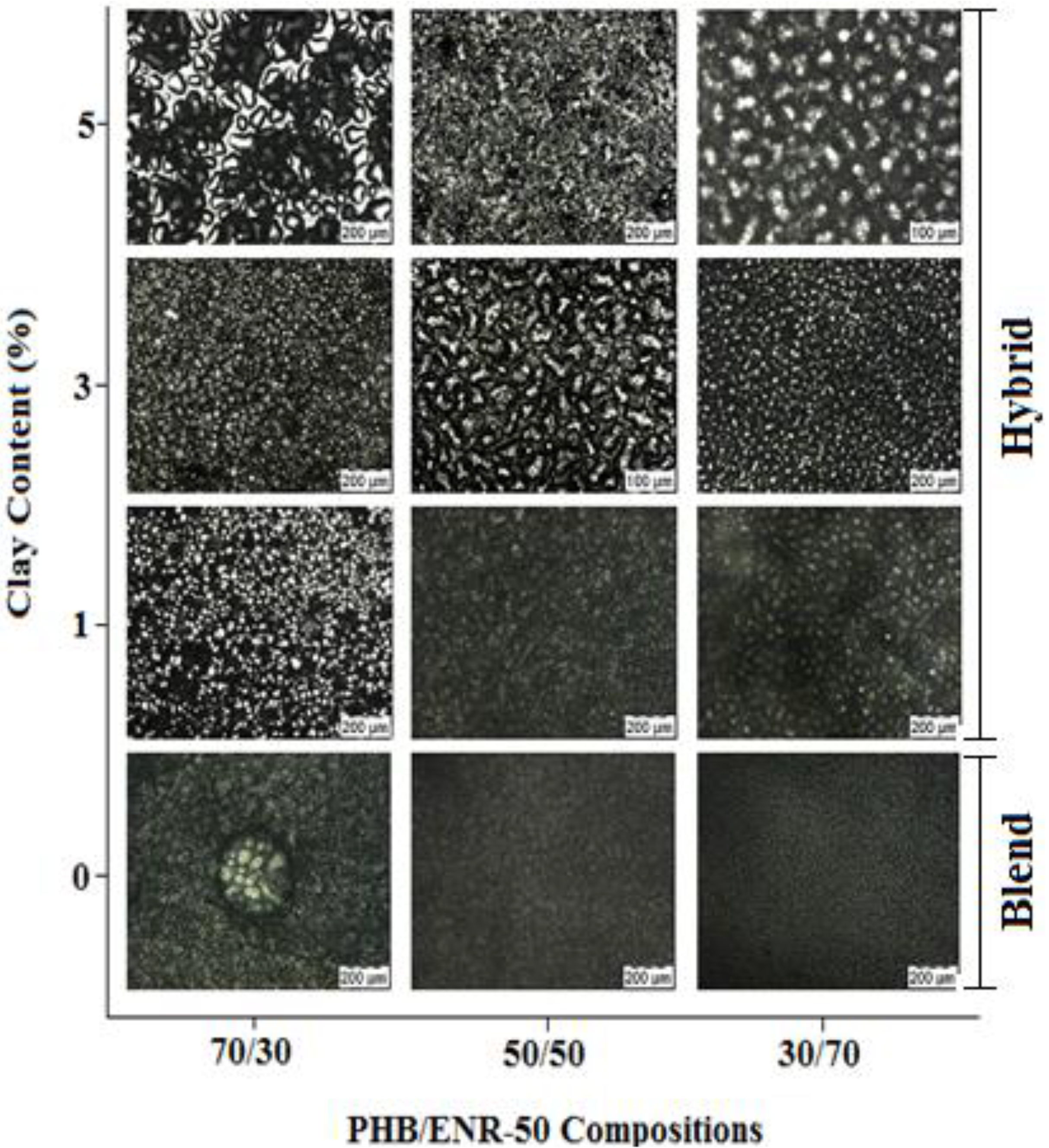

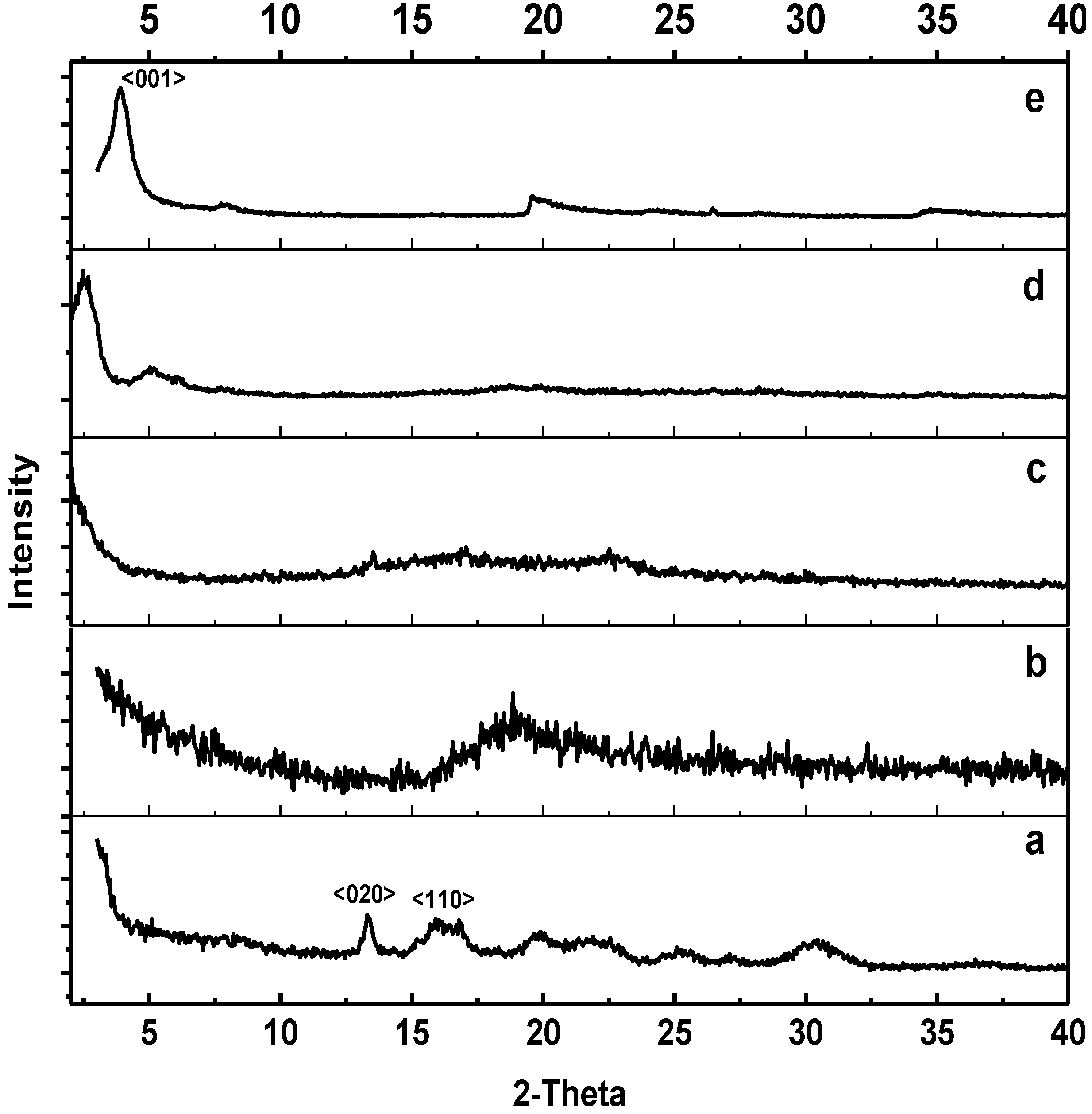
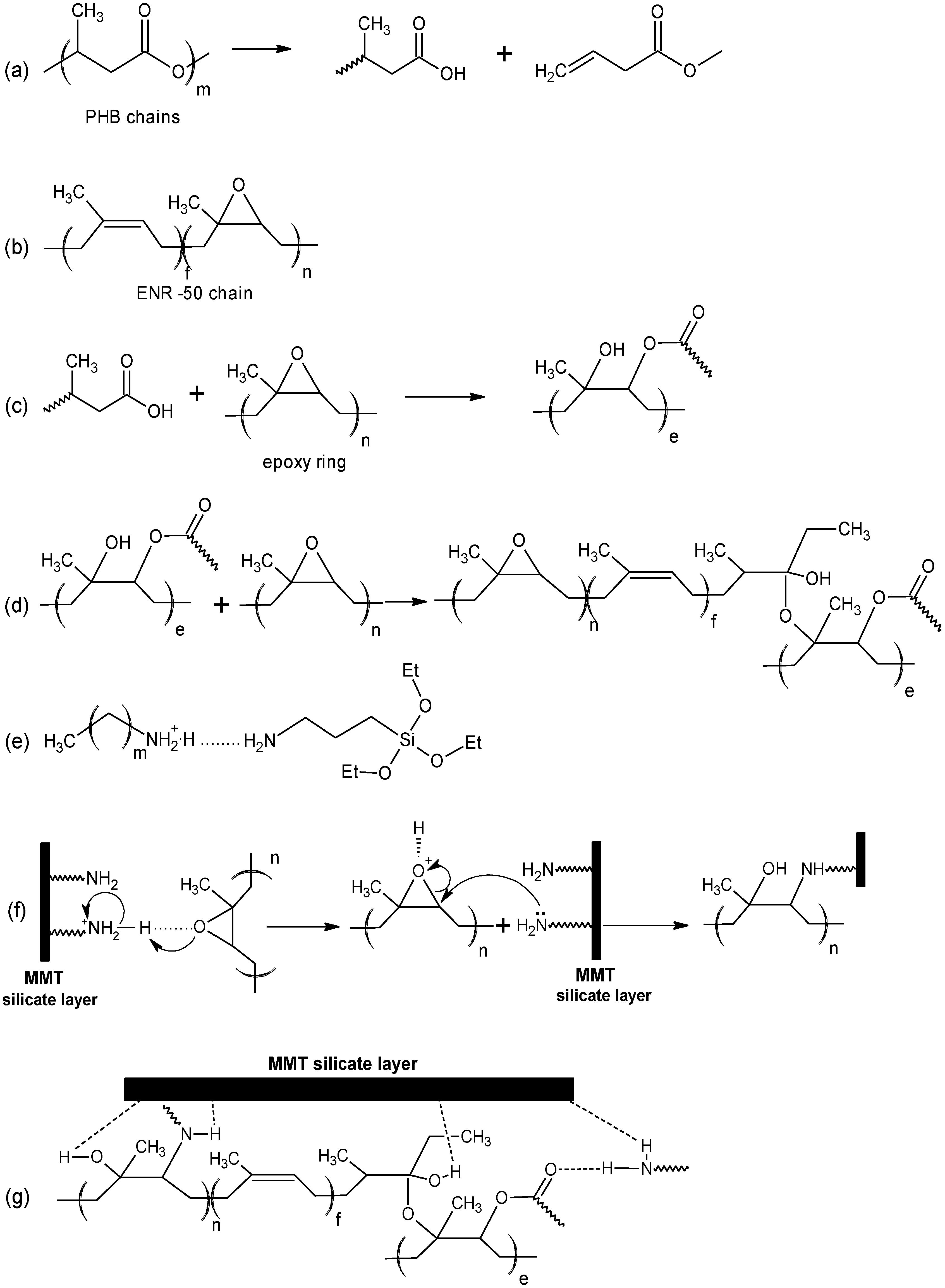
2.2. FTIR Spectroscopy and Proposed Mechanism
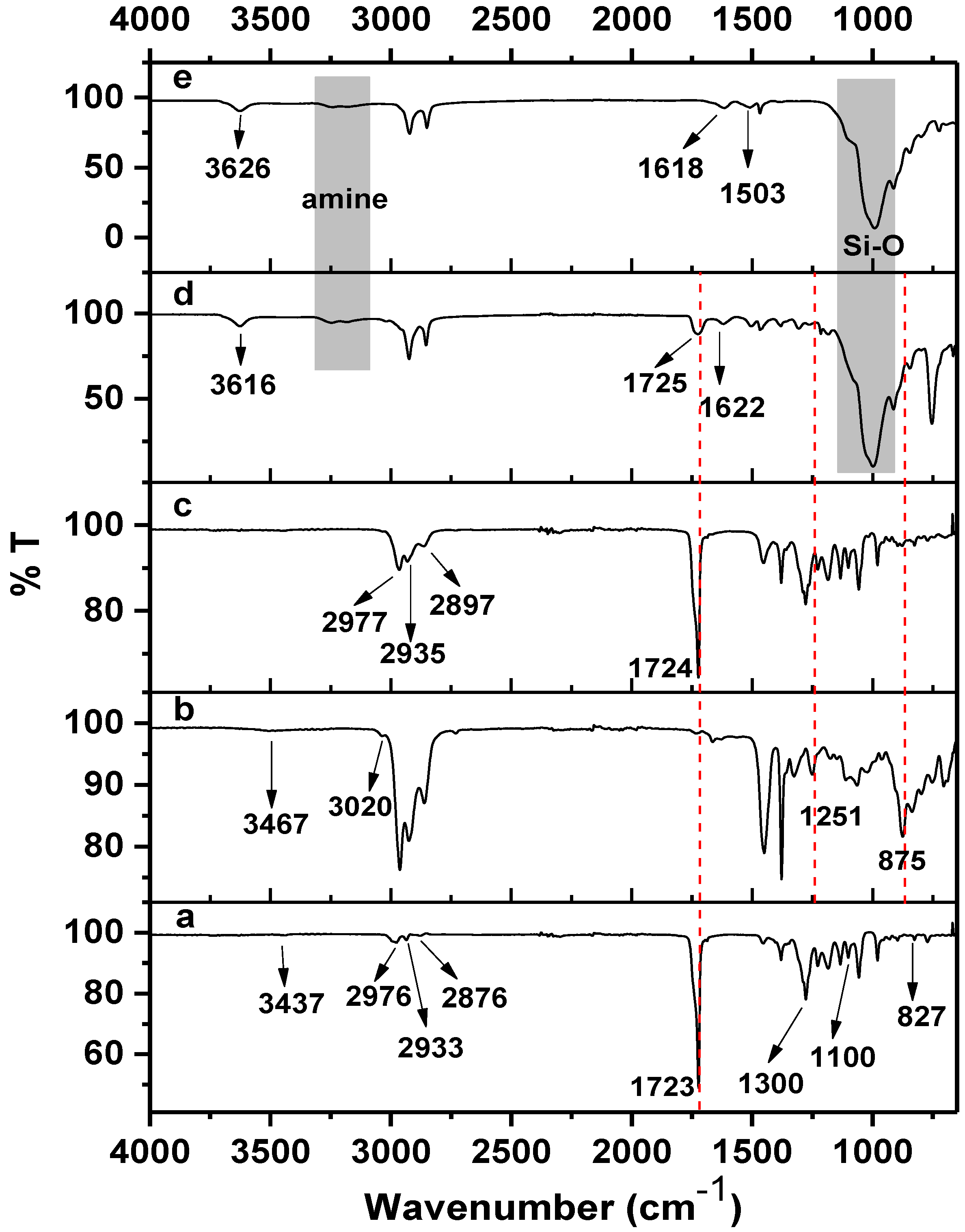
| Functional Group | PHB | ENR–50 | Blend | Nanohybrid | MMT |
|---|---|---|---|---|---|
| (CO)–OH | – | – | 3684,3621 | – | – |
| Si–OH | – | – | – | 3616 | 3626 |
| –OH | 3437 | – | 3464 | 3436 | 3428 |
| Free Amine | – | – | – | 3243,3182 | 3265,3175 |
| –C=CH | – | 3020 | 3019 | – | – |
| Methyl | 2976,1378,827 | 2973,1380 | 2977 | 2977–2895 * | – |
| Methylene | 2933,1453 | 2912 | 2935 | 2928–1466 | 2921,1468 |
| Methine | 2876 | 2853 | 2897 | 2856 | 2851 |
| –(O)C=O | 1723 | – | 1724 | 1725 | – |
| –C–O– | 1300,1100 | – | 1305,1100 | – | – |
| Epoxy | – | 1251,875 | 1218,877 | – | – |
| –Si–O | – | – | – | 1100–915 | 1100–915 |
| –C=C– | – | 759,669 | 1602,760,672 | 1667 | – |
| –NH2+ | – | – | – | 1622 | 1618 |
| –NH2+Amine | – | – | – | 1503 | 1503 |
2.3. Thermal Behavior
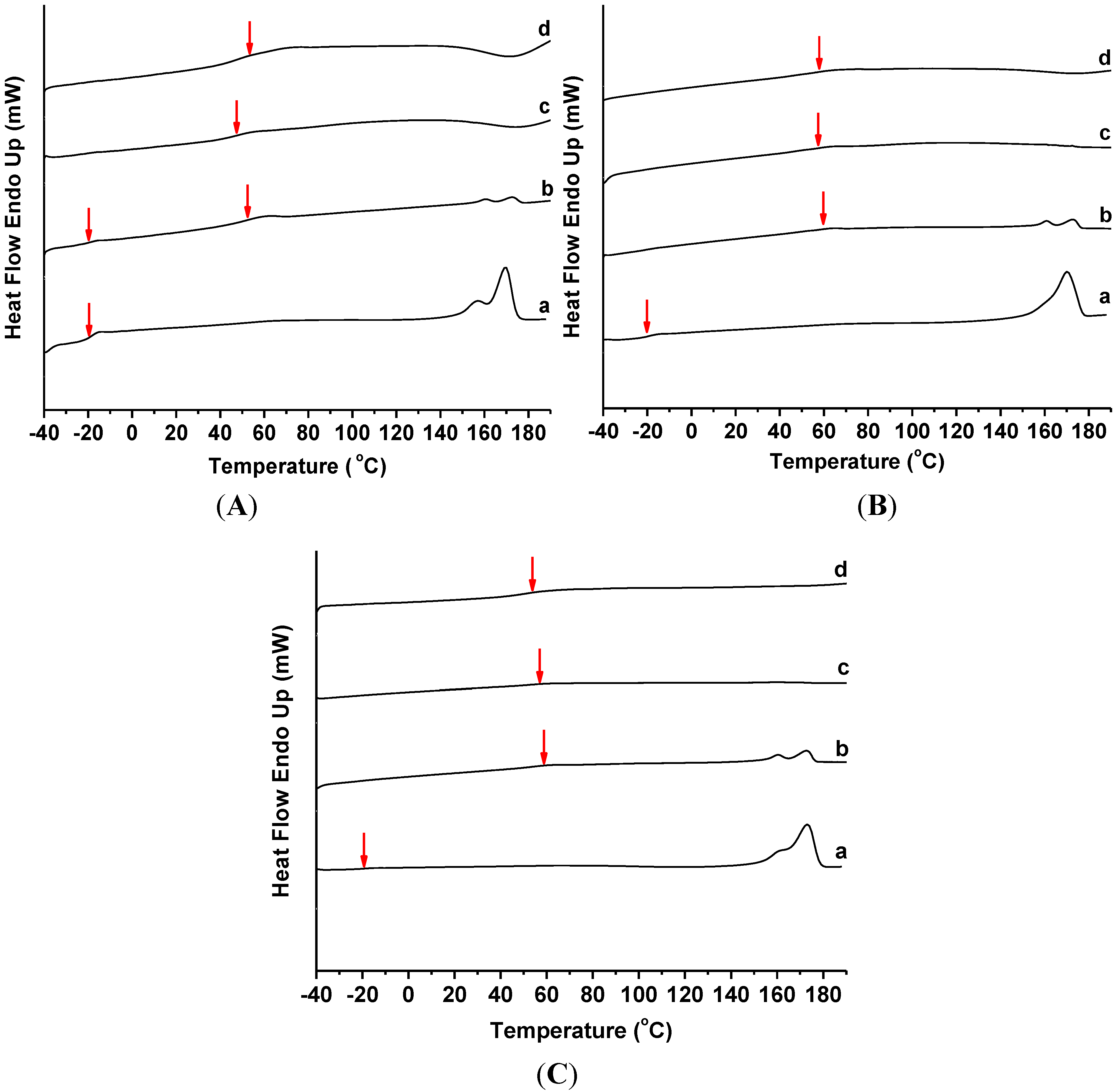
| PHB/ENR-50 composition | Clay content (wt%) | DSC * | TG-DTG ** | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Tg1 (°C) | Tg2 (°C) | Tm (°C) | ∆Hm (J/g) | T10 (°C) | T20 (°C) | T30 (°C) | Tmax1 (°C) | Tmax2 (°C) | ||
| 30/70 | 0 | −18.1 | – | 169.6 | 25.8 | 255.6 | 281.5 | 370.4 | 280.9 | 433.1 |
| 1 | −18.8 | 53.5 | 172.4 | 4.1 | 270.4 | 318.5 | 366.7 | 332.8 | 438.2 | |
| 3 | – | 49.3 | – | – | 296.3 | 366.7 | 403.7 | 310.9 | 432.7 | |
| 5 | – | 60.0 | – | – | 314.8 | 377.8 | 414.8 | 291.0 | 430.7 | |
| 50/50 | 0 | −17.7 | – | 170.2 | 37.5 | 248.2 | 259.3 | 270.4 | 279.1 | 437.8 |
| 1 | – | 60.7 | 172.4 | 9.1 | 266.7 | 281.5 | 307.4 | 313.9 | 431.5 | |
| 3 | – | 57.3 | – | – | 274.1 | 340.7 | 388.9 | 300.0 | 429.5 | |
| 5 | – | 58.6 | – | – | 285.2 | 388.9 | 425.9 | 294.4 | 441.0 | |
| 70/30 | 0 | −19.3 | – | 173.1 | 53.7 | 235.2 | 242.6 | 248.2 | 276.9 | 414.2 |
| 1 | – | 58.0 | 172.8 | 18.3 | 281.5 | 292.6 | 300.0 | 303.6 | 446.7 | |
| 3 | – | 57.4 | 170.0 | 1.9 | 274.1 | 296.3 | 377.8 | 297.9 | 438.7 | |
| 5 | – | 54.3 | – | – | 266.7 | 355.6 | 407.4 | 284.1 | 429.5 | |
2.4. Thermal Decomposition
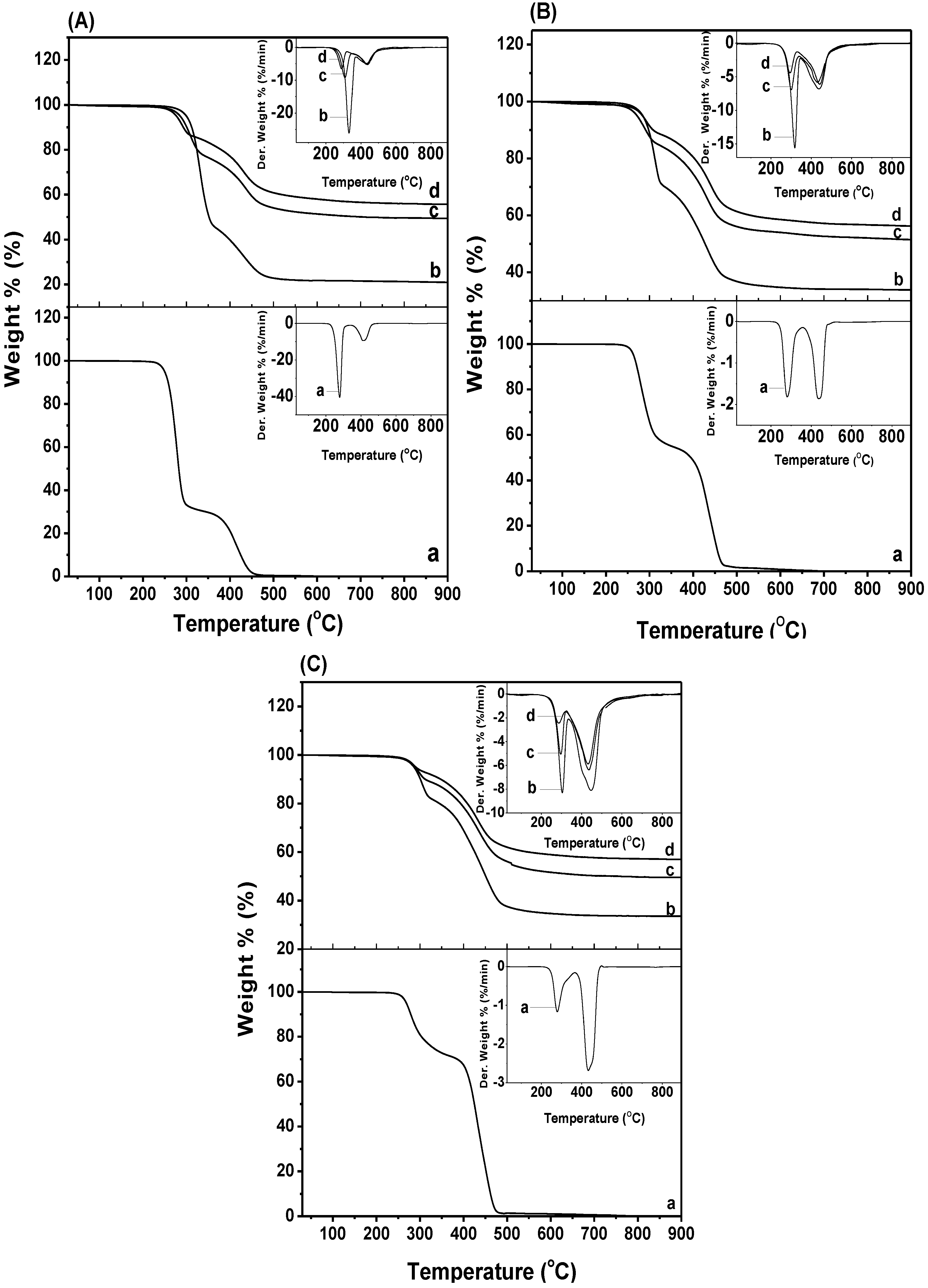
3. Experimental
3.1. Materials
3.2. Preparation of Multi-Component Nanohybrid
3.3. Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Arroyo, M.; Lopezmanchado, M.; Valentin, J.; Carretero, J. Morphology/behavior relationship of nanocomposites based on natural rubber/epoxidized natural rubber blends. Compos. Sci. Technol. 2007, 67, 1330–1339. [Google Scholar] [CrossRef]
- Peng, Z.; Kong, L.X.; Li, S.D.; Chen, Y.; Huang, M.F. Self-assembled natural rubber/silica nanocomposites: Its preparation and characterization. Compos. Sci. Technol. 2007, 67, 3130–3139. [Google Scholar] [CrossRef]
- Botana, A.; Mollo, M.; Eisenberg, P.; Torres Sanchez, R.M. Effect of modified montmorillonite on biodegradable PHB nanocomposites. Appl. Clay Sci. 2010, 47, 263–270. [Google Scholar] [CrossRef]
- Tjong, S. Structural and mechanical properties of polymer nanocomposites. Mater. Sci. Eng. Rep. 2006, 53, 73–197. [Google Scholar] [CrossRef]
- Shaked, Y.; Dodiuk, H.; Kenig, S.; Mccarthy, S. The effect of hyperbranched polymers on processing and thermal stability of biodegradable polyesters. Polym. Eng. Sci. 2009, 49, 559–566. [Google Scholar] [CrossRef]
- Lee, K.Y.; Goettler, L.A. Structure-property relationships in polymer blend nanocomposites. Polym. Eng. Sci. 2004, 44, 1103–1111. [Google Scholar] [CrossRef]
- Yariv, S.; Cross, H. Organo-Clay Complexes and Interactions; DEKKER: New York, NY, USA, 2002. [Google Scholar]
- Reis, K.C.; Pereira, J.; Smith, A.C.; Carvalho, C.W.P.; Wellner, N.; Yakimets, I. Characterization of polyhydroxybutyrate-hydroxyvalerate (PHB-HV)/maize starch blend films. J. Food Eng. 2008, 89, 361–369. [Google Scholar] [CrossRef]
- Gonzalez, A.; Irusta, L.; Fernandezberridi, M.; Iriarte, M.; Iruin, J. Miscibility behavior of amorphous poly(3-hydroxybutyrate) (a-PHB)/styrene-vinyl phenol copolymer (STY-co-VPH) blends applying an association model. Polymer 2004, 45, 1477–1483. [Google Scholar]
- Corradini, E.; Marconcini, J.M.; Agnelli, J.A.M.; Mattoso, L.H.C. Thermoplastic blends of corn gluten meal/starch (CGM/Starch) and corn gluten meal/polyvinyl alcohol and corn gluten meal/poly (hydroxybutyrate-co-hydroxyvalerate) (CGM/PHB-V). Carbohydr. Polym. 2011, 83, 959–965. [Google Scholar] [CrossRef]
- Vogel, C.; Siesler, H.W. Thermal degradation of poly(ɛ-caprolactone), poly(L-lactic acid) and their blends with poly(3-hydroxy-butyrate) studied by TGA/FT-IR spectroscopy. Macromol. Symp. 2008, 265, 183–194. [Google Scholar] [CrossRef]
- Abdelwahab, M.A.; Flynn, A.; Chiou, B.S.; Imam, S.; Orts, W.; Chiellini, E. Thermal, mechanical and morphological characterization of plasticized PLA-PHB blends. Polym. Degrad. Stab. 2012, 97, 1822–1828. [Google Scholar] [CrossRef]
- Chee, M.J.K.; Ismail, J.; Kummerlöwe, C.; Kammer, H.W. Study on miscibility of PEO and PCL in blends with PHB by solution viscometry. Polymer 2002, 43, 1235–1239. [Google Scholar]
- Achilias, D.S.; Panayotidou, E.; Zuburtikudis, I. Thermal degradation kinetics and isoconversional analysis of biodegradable poly(3-hydroxybutyrate)/organomodified montmorillonite nanocomposites. Thermochim. Acta 2011, 514, 58–66. [Google Scholar] [CrossRef]
- Perinovic, S.; Erceg, M.; Kovac, T. Isothermal degradation of poly (3-hydroxybutyrate)/ organically modified montmorillonite nanocomposites. Polym. Compos. 2010, 31, 272–278. [Google Scholar]
- Tharanathan, R. Biodegradable films and composite coatings: Past, present and future. Trends Food Sci. Technol. 2003, 14, 71–78. [Google Scholar] [CrossRef]
- Erceg, M.; Kovacic, T.; Klaric, I. Poly(3-hydroxybutyrate ) nanocomposites: Isothermal degradation and kinetic analysis. Thermochim. Acta 2009, 485, 26–32. [Google Scholar] [CrossRef]
- Mousavioun, P.; George, G.A.; Doherty, W.O.S. Environmental degradation of lignin/poly(hydroxybutyrate) blends. Polym. Degrad. Stab. 2012, 97, 1114–1122. [Google Scholar] [CrossRef]
- Lee, H.K. A Study of Poly(3-hydroxybutyrate)/Epoxidized Natural Rubber Blends: Melt Reaction and Thermal Properties. Ph.D. Thesis, Universiti Sains Malaysia, Penang, Malaysia, 2008. [Google Scholar]
- Lee, H.K.; Ismail, J.; Kammer, H.W.; Bakar, M.A. Melt reaction in blends of poly(3-hydroxybutyrate) (PHB) and epoxidized natural rubber (ENR-50). J. Appl. Polym. Sci. 2004, 95, 113–129. [Google Scholar]
- Salehabadi, A.; Abu Bakar, M. Poly(3-hydroxybutyrate)organo modified montmorillonite nanohybrids; preparation and characterization. Adv. Mater. Res. 2013, 623, 263–270. [Google Scholar]
- Salehabadi, A.; Abu Bakar, M. Epoxidized natural rubber-organomodified montmorillonite nanohybrids; interaction and thermal decomposition. Mater. Sci. Forum 2013, 756, 119–126. [Google Scholar] [CrossRef]
- Holmes, P.A. Applications of PHB—A microbially produced biodegradable thermoplastic. Phys. Technol. 1985, 16, 32–36. [Google Scholar] [CrossRef]
- Zhijiang, C.; Guang, Y. Optical nanocomposites prepared by incorporating bacterial cellulose nanofibrils into poly(3-hydroxybutyrate). Mater. Lett. 2011, 65, 182–184. [Google Scholar] [CrossRef]
- Vu, Y.T.; Mark, J.E.; Pham, L.H.; Engelhardt, M. Clay nanolayer reinforcement of cis-1,4-polyisoprene and epoxidized natural rubber. J. Appl. Polym. Sci. 2001, 82, 1391–1403. [Google Scholar] [CrossRef]
- Ratnam, C.T.; Nasir, M.; Baharin, A.; Zaman, K. Electron beam irradiation of epoxidized natural rubber: FTIR studies. Polym. Int. 2000, 49, 1693–1701. [Google Scholar] [CrossRef]
- Bordes, P.; Hablot, E.; Pollet, E.; Avérous, L. Effect of clay organomodifiers on degradation of polyhydroxyalkanoates. Polym. Degrad. Stab. 2009, 94, 789–796. [Google Scholar] [CrossRef]
- Amico, D.A.D.; Manfredi, L.B.; Cyras, V.P.; de Mar, N.; Justo, J.B.; Plata, M. Relationship between thermal properties, morphology, and crystallinity of nanocomposites based on polyhydroxybutyrate. J. Appl. Polym. Sci. 2011, 123, 200–208. [Google Scholar]
- Leszczy, A.; Njuguna, J.; Pielichowski, K.; Banerjee, J.R. Polymer/montmorillonite nanocomposites with improved thermal properties Part I. Factors influencing thermal stability and mechanisms of thermal stability improvement. Thermochim. Acta 2007, 453, 75–96. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Salehabadi, A.; Bakar, M.A.; Bakar, N.H.H.A. Effect of Organo-Modified Nanoclay on the Thermal and Bulk Structural Properties of Poly(3-hydroxybutyrate)-Epoxidized Natural Rubber Blends: Formation of Multi-Components Biobased Nanohybrids. Materials 2014, 7, 4508-4523. https://doi.org/10.3390/ma7064508
Salehabadi A, Bakar MA, Bakar NHHA. Effect of Organo-Modified Nanoclay on the Thermal and Bulk Structural Properties of Poly(3-hydroxybutyrate)-Epoxidized Natural Rubber Blends: Formation of Multi-Components Biobased Nanohybrids. Materials. 2014; 7(6):4508-4523. https://doi.org/10.3390/ma7064508
Chicago/Turabian StyleSalehabadi, Ali, Mohamad Abu Bakar, and Noor Hana Hanif Abu Bakar. 2014. "Effect of Organo-Modified Nanoclay on the Thermal and Bulk Structural Properties of Poly(3-hydroxybutyrate)-Epoxidized Natural Rubber Blends: Formation of Multi-Components Biobased Nanohybrids" Materials 7, no. 6: 4508-4523. https://doi.org/10.3390/ma7064508
APA StyleSalehabadi, A., Bakar, M. A., & Bakar, N. H. H. A. (2014). Effect of Organo-Modified Nanoclay on the Thermal and Bulk Structural Properties of Poly(3-hydroxybutyrate)-Epoxidized Natural Rubber Blends: Formation of Multi-Components Biobased Nanohybrids. Materials, 7(6), 4508-4523. https://doi.org/10.3390/ma7064508




