Microstructural Characterization of Calcite-Based Powder Materials Prepared by Planetary Ball Milling
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
| Inorganic element (wt %) a | Organic element (wt %) b | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca | Mg | Si | Al | Fe | Sr | Na | P | Ti | C | H | N |
| 35.3 | 0.388 | 0.557 | 0.431 | 0.223 | 0.09 | 1.06 | 0.08 | 0.013 | 11.55 | 0.48 | 0.00 |
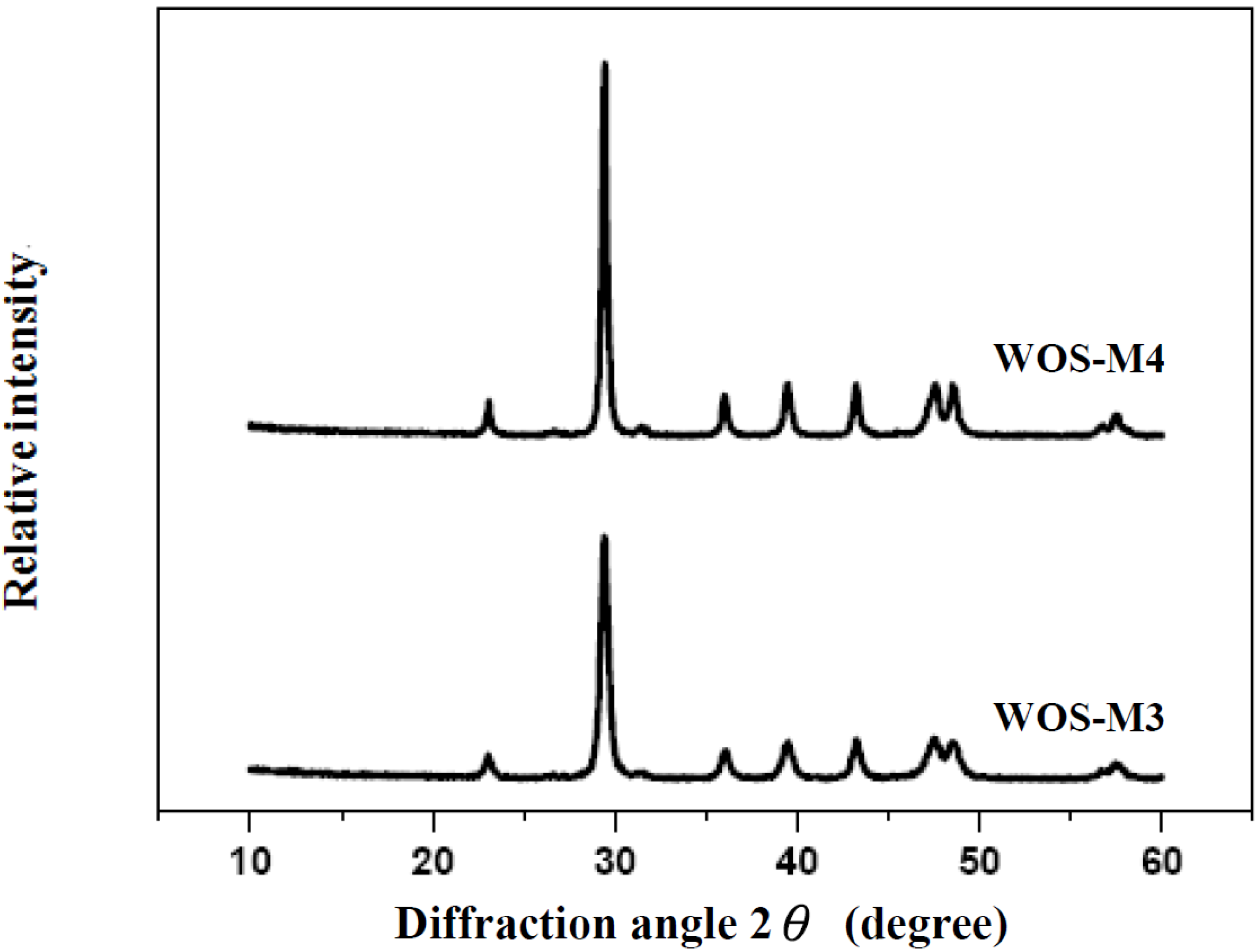
2.2. Experimental Apparatus and Procedures
- Rotational speed: 200, 300, 400, 500, and 600 rpm (denoted as S1, S2, S3, S4, and S5, respectively);
- Grinding time: 5, 10, 30, 60, 120, and 180 min (denoted as T1, T2, T3, T4, T5, and T6, respectively);
- Sample mass: 1.0, 2.5, 5.0, 7.5, and 10.0 g (denoted as M1, M2, M3, M4, and M5, respectively).
2.3. Characterization of Resulting Powder Materials
2.3.1. Pore Property
2.3.2. True Density
2.3.3. Particle Size Distribution
2.3.4. Scanning Electron Microscope (SEM) Observation
3. Results and Discussion
3.1. Pore Property
| Sample ID | BET surface area (m2·g−1) | Total pore volume (cm3·g−1) | True density (g·cm−3) | Porosity (-) f |
|---|---|---|---|---|
| WOS-RW | 4.05 | 0.0244 | 2.596 | 0.06 |
| WOS-S1 a | 5.52 | 0.0276 | 2.622 | 0.07 |
| WOS-S2 a | 7.31 | 0.0532 | 2.611 | 0.12 |
| WOS-S3 a,d | 10.64 e | 0.0660 | 2.606 | 0.15 |
| WOS-S4 a | 9.18 | 0.0579 | 2.584 | 0.13 |
| WOS-S5 a | 8.04 | 0.0454 | 2.575 | 0.11 |
| WOS-T1 b | 4.14 | 0.0359 | 2.620 | 0.09 |
| WOS-T2 b | 7.37 | 0.0429 | 2.621 | 0.10 |
| WOS-T3 b,d | 10.64 e | 0.0660 | 2.606 | 0.15 |
| WOS-T4 b | 9.20 | 0.0682 | 2.590 | 0.15 |
| WOS-T5 b | 6.30 | 0.0382 | 2.599 | 0.09 |
| WOS-T6 b | 5.92 | 0.0322 | 2.582 | 0.08 |
| WOS-M1 c | 5.41 | 0.0484 | 2.508 | 0.11 |
| WOS-M2 c | 7.20 | 0.0536 | 2.577 | 0.12 |
| WOS-M3 c,d | 10.64 e | 0.0660 | 2.606 | 0.15 |
| WOS-M4 c | 7.06 | 0.0474 | 2.636 | 0.11 |
| WOS-M5 c | 6.11 | 0.0368 | 2.641 | 0.09 |
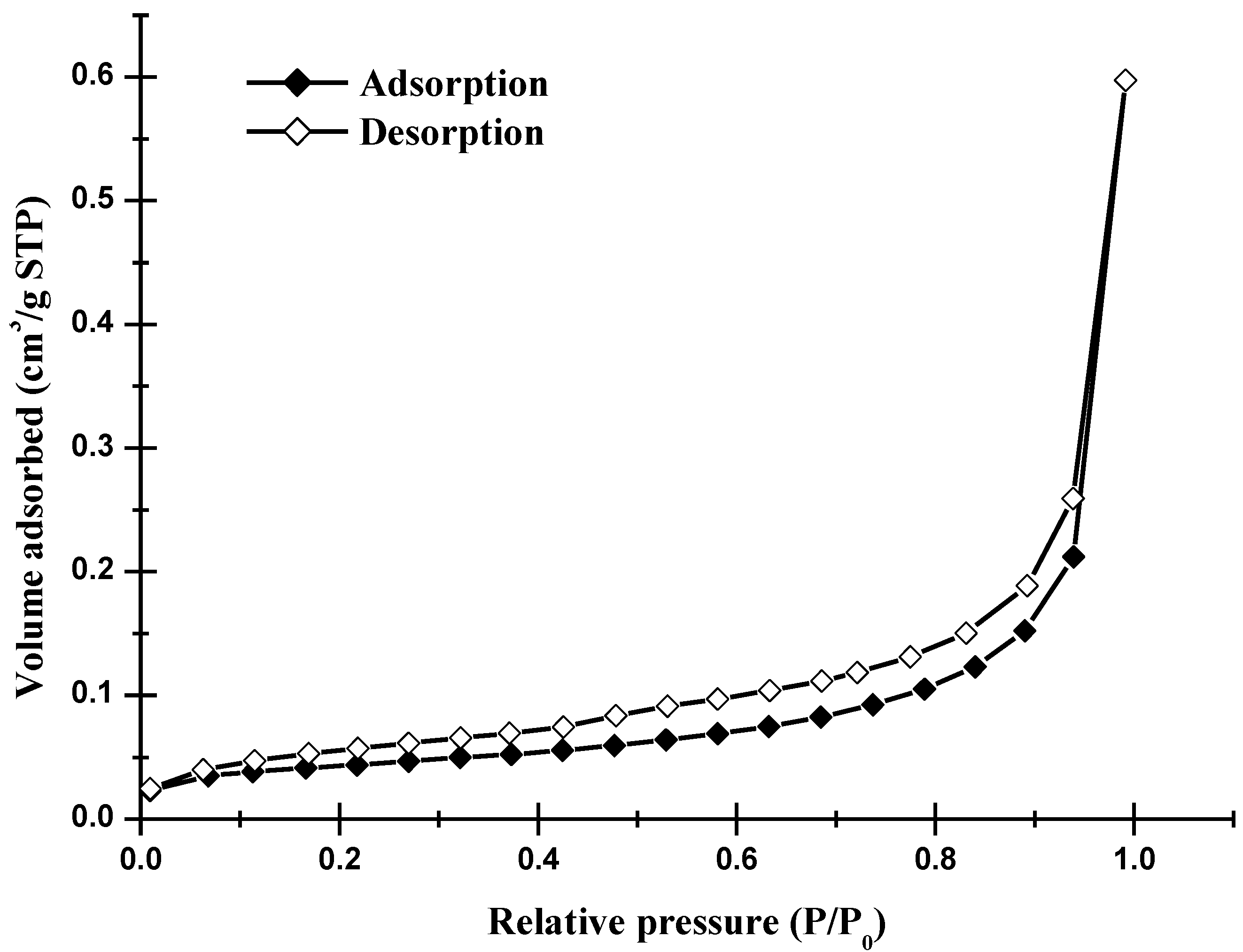
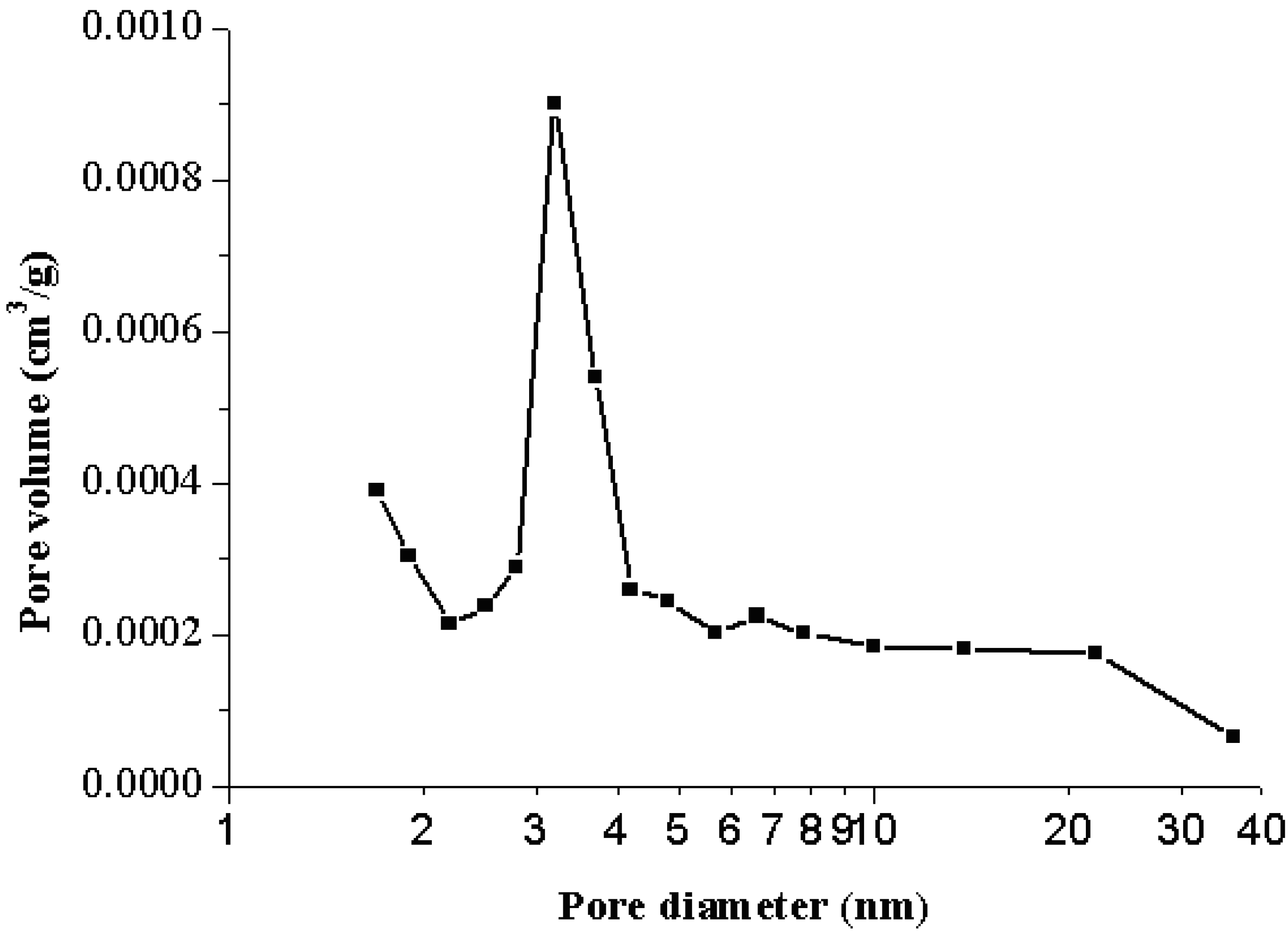
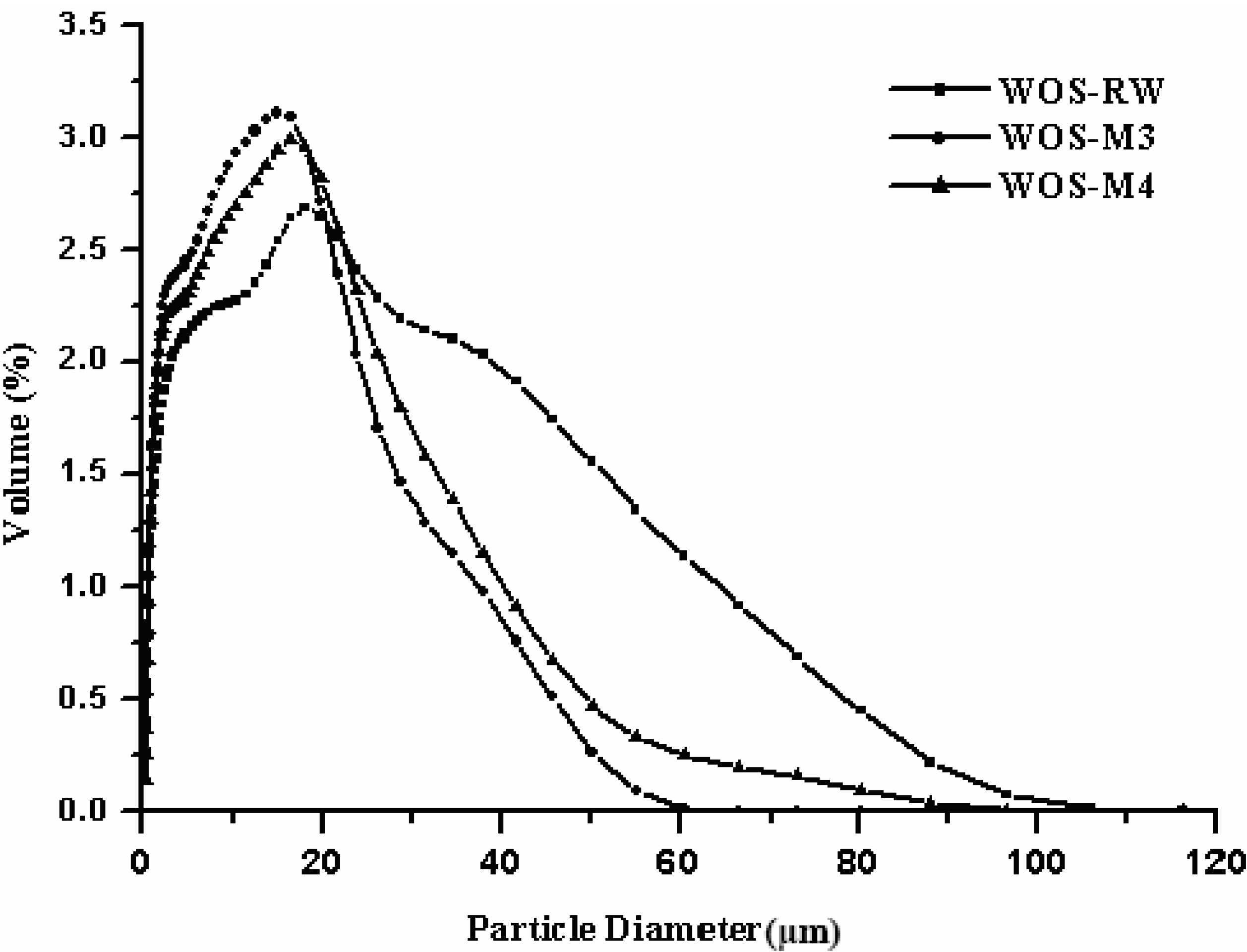
3.2. Particle Size Distribution
3.3. Scanning Electron Microscope (SEM) Observation

4. Conclusions
- Under the rotational speed of 200–600 rpm, grinding time of 5–180 min and sample mass loading of 1–10 g, the grinding treatment significantly changes the surface properties of the calcite-based minerals. The pore properties of the optimal resulting powder are 10.64 m2·g−1, 0.066 cm3·g−1, and 0.15 based on BET surface area, total pore volume and porosity, respectively, as compared to those (i.e., 4.05 m2·g−1, 0.024 cm3·g−1, and 0.06) of the starting material. This finding was also consistent with the particle size measurement (i.e., 9.7 vs. 15.0 μm of mean diameter).
- From the nitrogen adsorption/desorption isotherms, a typical Type II was found in the resulting powders, indicating that the isotherms are characterized by the nonporous materials or materials with macropores or open voids. However, a small hysteresis loop was also seen in the isotherms, suggesting that a small amount of mesopore with wide slit-shaped mouth exists in the fine powder.
- According to the observations in the SEM, the surface roughness can be enhanced as particle size decreases as a result of particle-particle attrition, suggesting that the specific surface areas of resulting powders hence increase with increasing fractural impact during high-energy ball milling.
Acknowledgments
Conflict of Interest
References
- Chen, B.; Peng, X.; Wang, J.G.; Wu, X. Laminated microstructure of bivalva shell and research of biomimetic ceramic/polymer composite. Ceram. Int. 2004, 30, 2011–2014. [Google Scholar] [CrossRef]
- Zolotoyabko, E.; Caspi, E.N.; Fieramosca, J.S.; von Dreele, R.B. Bond lengths differences between the mollusk-made and geological calcium carbonate. Mater. Sci. Eng. A 2009, 524, 77–81. [Google Scholar] [CrossRef]
- Chateigner, D.; Ouhenia, S.; Krauss, C.; Hedegaard, C.; Gil, O.; Morales, M.; Lutterotti, L.; Rousseau, M.; Lopez, E. Voyaging around nacre with the X-ray shuttle: From bio-mineralisation to prosthetics via mollusc phylogeny. Mater. Sci. Eng. A 2010, 528, 37–51. [Google Scholar] [CrossRef]
- Jung, J.H.; Shon, B.H.; Yoo, K.S.; Oh, K.J. Physicochemical characteristics of waste sea shells for acid gas cleaning absorbent. Korean J. Chem. Eng. 2000, 17, 585–592. [Google Scholar] [CrossRef]
- Lee, D.Y.; Jing, S.R.; Lin, Y.F. Using seafood waste as sludge conditioners. Water Sci. Technol. 2001, 44, 301–307. [Google Scholar] [PubMed]
- Tudor, H.E.A.; Gryte, C.C.; Harris, C.C. Seashells: Detoxifying agents for metal-contaminated waters. Water Air Soil Pollut. 2006, 173, 209–242. [Google Scholar] [CrossRef]
- Tsai, W.T.; Chen, H.R.; Kuo, K.C.; Lai, C.Y.; Su, T.C.; Chang, Y.M.; Yang, J.M. The adsorption of methylene blue from aqueous solution using waste aquacultural shell powders. J. Environ. Eng. Manag. 2009, 19, 165–172. [Google Scholar]
- Yoon, H.; Park, S.; Lee, K.; Park, J. Oyster shell as substitute for aggregate in mortar. Waste Manag. Res. 2004, 22, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.I.; Yi, S.T.; Leem, Y.M. Effect of oyster shell substituted for fine aggregate on concrete characteristics: Part I. Fundamental properties. Cem. Concr. Res. 2005, 35, 2175–2182. [Google Scholar] [CrossRef]
- Augspurger, N.R.; Baker, D.H. Phytase improves dietary calcium utilization in chicks, and oyster shell, carbonate, citrate, and citrate-malate forms of calcium are equally bioavailable. Nutr. Res. 2004, 24, 293–301. [Google Scholar] [CrossRef]
- Namasivayam, C.; Sakoda, A.; Suzuki, M. Removal of phosphate by adsorption onto oyster shell powder- kinetic studies. J. Chem. Technol. Biotechnol. 2005, 80, 356–358. [Google Scholar] [CrossRef]
- Park, W.H.; Polprasert, C. Phosphorus adsorption characteristics of oyster shells and alum sludge and their application for nutrient control in constructed wetland system. J. Environ. Sci. Health A 2008, 43, 511–517. [Google Scholar] [CrossRef]
- Choi, Y.M.; Whang, J.H.; Kim, J.M.; Suh, H.J. The effect of oyster shell powder on the extension of the shelf-life of Kimchi. Food Control 2006, 17, 695–699. [Google Scholar] [CrossRef]
- Lee, C.H.; Lee, D.K.; Ali, M.A.; Kim, P.J. Effects of oyster shell on soil chemical and biological properties and cabbage productivity as a liming materials. Waste Manag. 2008, 28, 2702–2708. [Google Scholar] [CrossRef] [PubMed]
- Tongamp, W.; Kano, J.; Zhang, Q.; Saito, F. Simultaneous treatment of PVC and oyster-shell wastes by mechanochemical means. Waste Manag. 2008, 28, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Lowell, S.; Shields, J.E. Powder Surface Area and Porosity, 3rd ed.; Chapman & Hall: London, UK, 1991; pp. 59–65. [Google Scholar]
- Balaz, P.; Dutkova, E. Fine milling in applied mechanochemistry. Miner. Eng. 2009, 22, 681–694. [Google Scholar] [CrossRef]
- Russo, L.; Colangelo, F.; Cioffi, R.; Rea, H.; de Stefano, L. A mechanochemical approach to porous silicon nanoparticles fabrication. Materials 2011, 4, 1023–1033. [Google Scholar] [CrossRef]
- Kano, J.; Saito, F. Correlation of powder characteristics of talc during planetary ball milling with the impact energy of the balls simulated by the particle element method. Powder Technol. 1998, 98, 166–170. [Google Scholar] [CrossRef]
- Choi, H.; Lee, W.; Kim, S. Effect of grinding aids on the kinetics of fine grinding energy consumed of calcite powders by a stirred ball mill. Adv. Powder Technol. 2009, 20, 350–354. [Google Scholar] [CrossRef]
- Tsai, W.T.; Yang, J.M.; Hsu, H.C.; Lin, C.M.; Lin, K.Y.; Chiu, C.H. Development and characterization in eggshell ground by planetary ball milling. Microporous Mesoporous Mater. 2008, 111, 379–386. [Google Scholar] [CrossRef]
- Chen, Z.; Yang, B.; Nan, Z. Modification of calcium carbonate crystals growth by dibenzoic acids. Mater. Chem. Phys. 2012, 132, 601–609. [Google Scholar] [CrossRef]
- Tsai, W.T.; Lai, C.W.; Hsien, K.J. Effect of particle size of activated clay on the adsorption of paraquat from aqueous solution. J. Colloid Interface Sci. 2003, 263, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity, 2nd ed.; Academic Press: London, UK, 1982; pp. 3–12. [Google Scholar]
- Molina-Boisseau, S.; Le Bolay, N. Characterisation of the physicochemical properties of polymers ground in a vibrated bead mill. Powder Technol. 2002, 128, 99–106. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tsai, W.-T. Microstructural Characterization of Calcite-Based Powder Materials Prepared by Planetary Ball Milling. Materials 2013, 6, 3361-3372. https://doi.org/10.3390/ma6083361
Tsai W-T. Microstructural Characterization of Calcite-Based Powder Materials Prepared by Planetary Ball Milling. Materials. 2013; 6(8):3361-3372. https://doi.org/10.3390/ma6083361
Chicago/Turabian StyleTsai, Wen-Tien. 2013. "Microstructural Characterization of Calcite-Based Powder Materials Prepared by Planetary Ball Milling" Materials 6, no. 8: 3361-3372. https://doi.org/10.3390/ma6083361
APA StyleTsai, W.-T. (2013). Microstructural Characterization of Calcite-Based Powder Materials Prepared by Planetary Ball Milling. Materials, 6(8), 3361-3372. https://doi.org/10.3390/ma6083361




