Optical Constants of Crystallized TiO2 Coatings Prepared by Sol-Gel Process
Abstract
:1. Introduction
2. Experimental Section
2.1. TiO2 Sol Synthesis
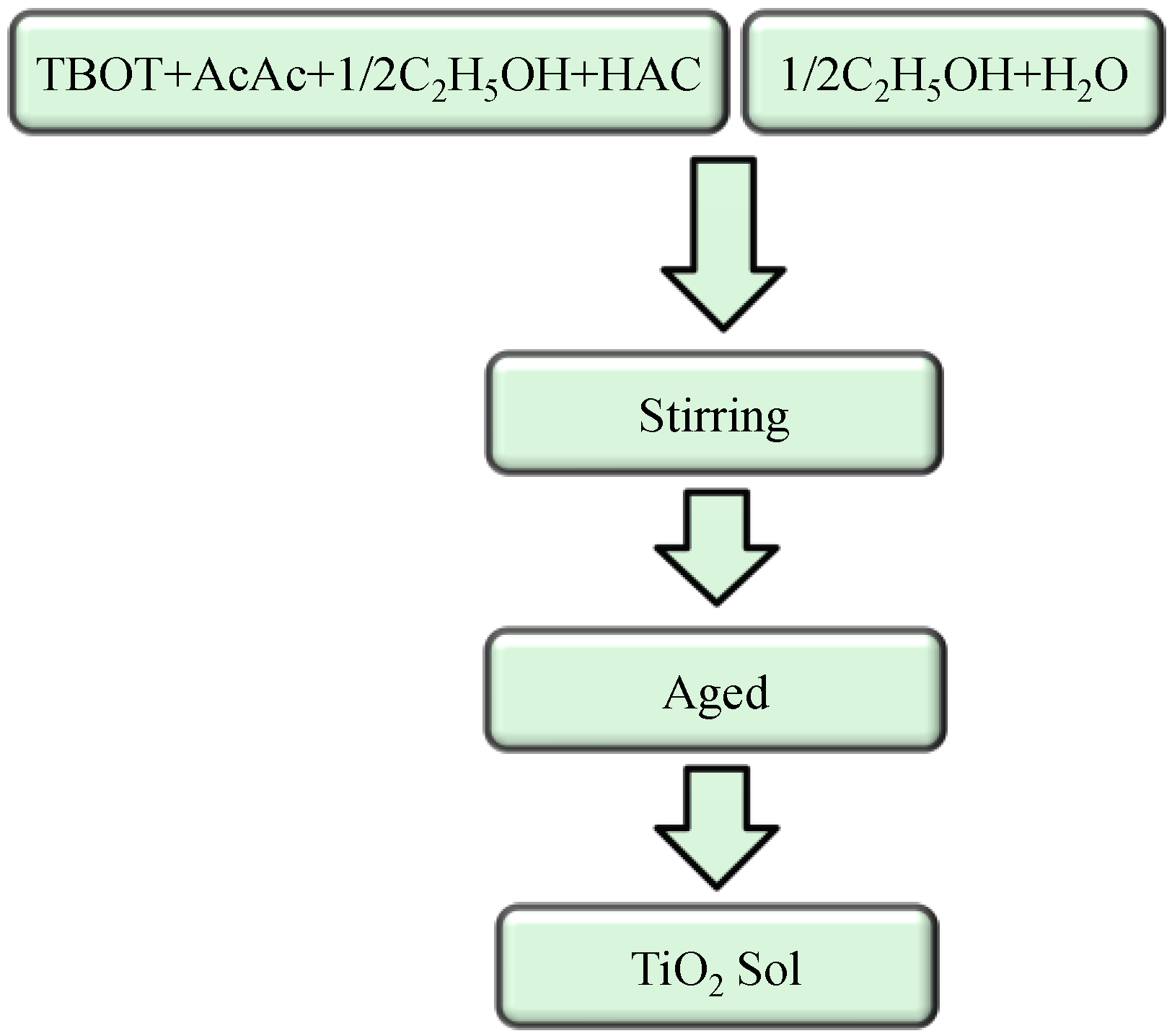
2.2. Coating Preparation
2.3. Characterization
3. Results and Discussion
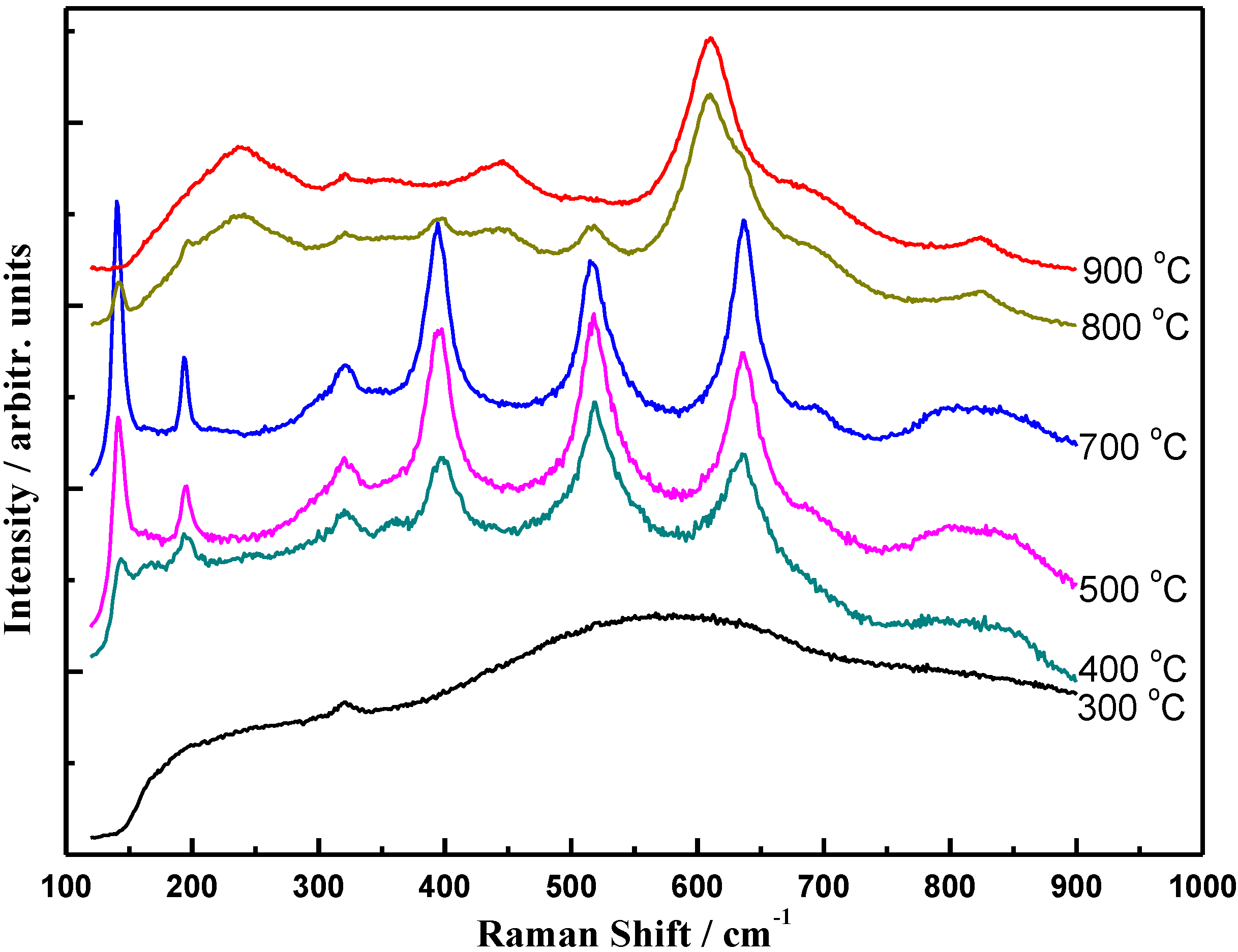
3.1. Structural Properties
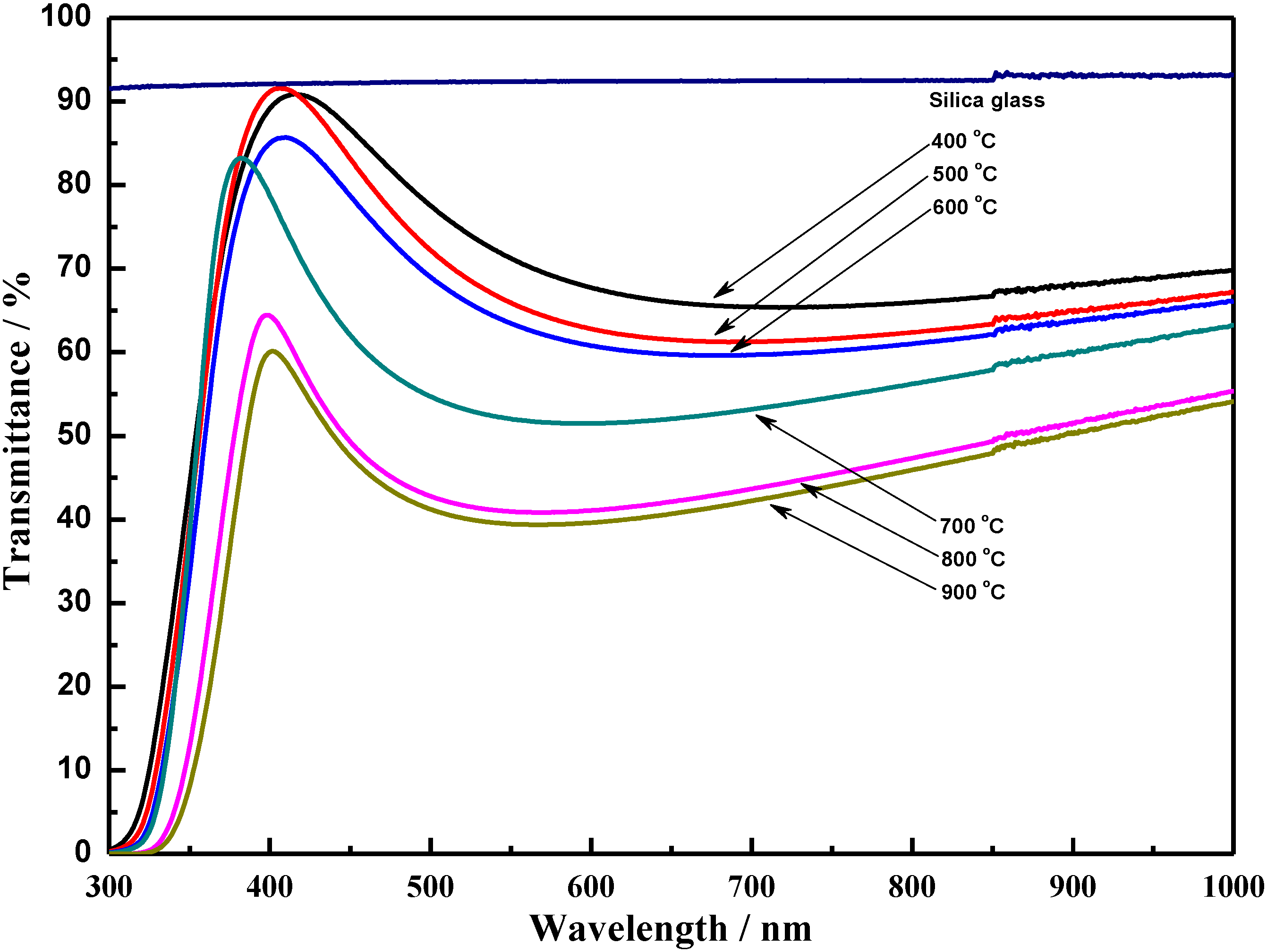
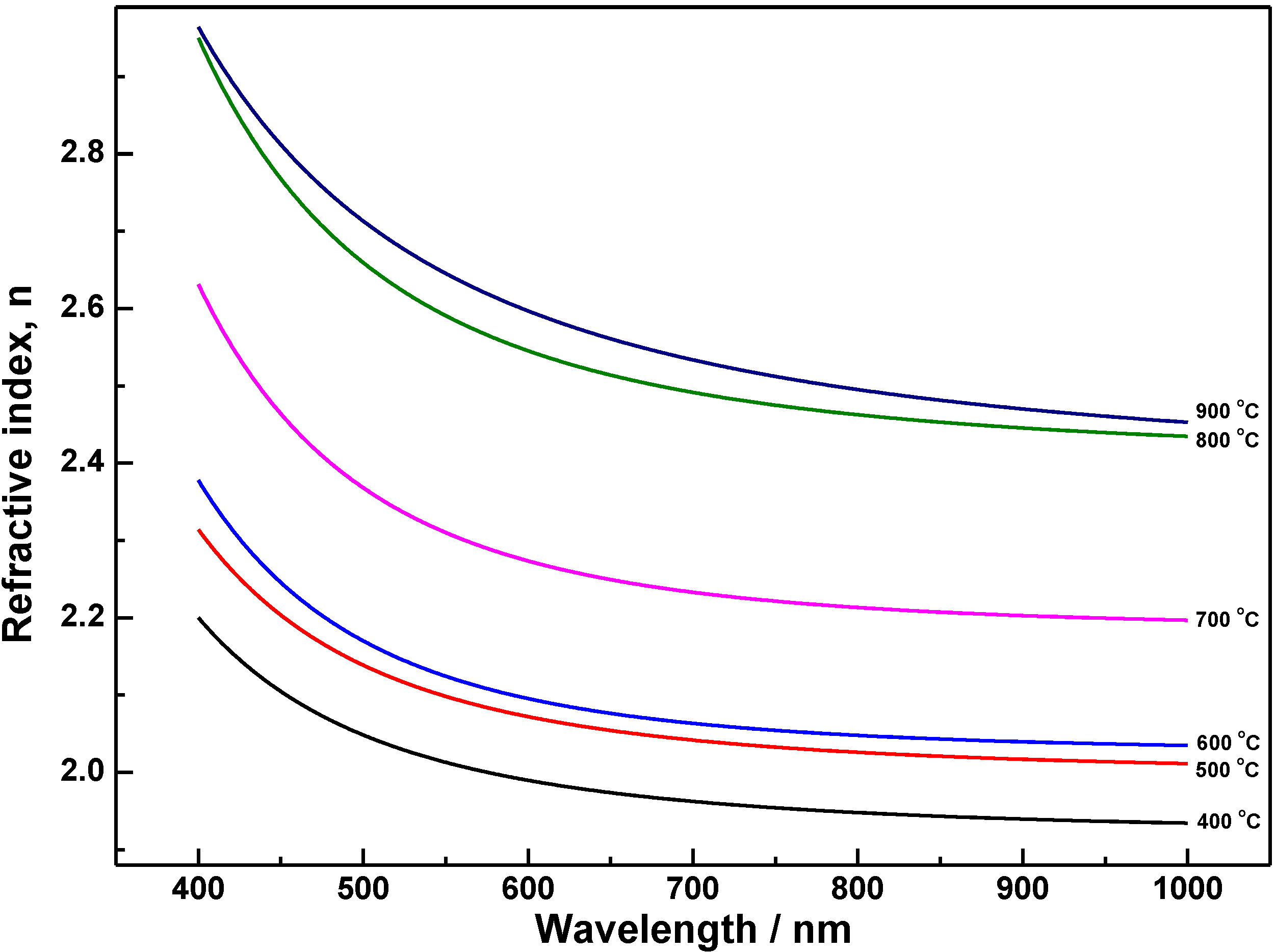
3.2. Optical Properties
| Sample | An | Bn (nm) | Cn (nm) | Ak | Bk (nm) | Ck (nm) | Thickness d (nm) | RMSE |
|---|---|---|---|---|---|---|---|---|
| 400 °C | 1.892 | 0.036 | 5.701 × 10−4 | 3.508 × 10−3 | 1.538 × 10−15 | 2.567 × 10−10 | 96.46 | 0.4452 |
| 500 °C | 1.958 | 0.040 | 1.704 × 10−3 | 5.757 × 10−3 | 2.629 × 10−13 | 7.540 × 10−13 | 89.94 | 0.6306 |
| 600 °C | 1.996 | 0.035 | 3.921 × 10−15 | 7.965 × 10−3 | 2.280 × 10−9 | 1.259 × 10−9 | 86.87 | 0.5689 |
| 700 °C | 2.127 | 0.051 | 1.255 × 10−3 | 8.741 × 10−3 | 2.054 × 10−12 | 4.287 × 10−9 | 70.70 | 0.4936 |
| 800 °C | 2.401 | 0.023 | 0.010 | 2.987 × 10−3 | 4.708 × 10−3 | 4.310 × 10−4 | 61.81 | 0.8760 |
| 900 °C | 2.385 | 0.063 | 4.707 × 10−3 | 0.011 | 5.985 × 10−11 | 1.102 × 10−3 | 61.02 | 0.3872 |
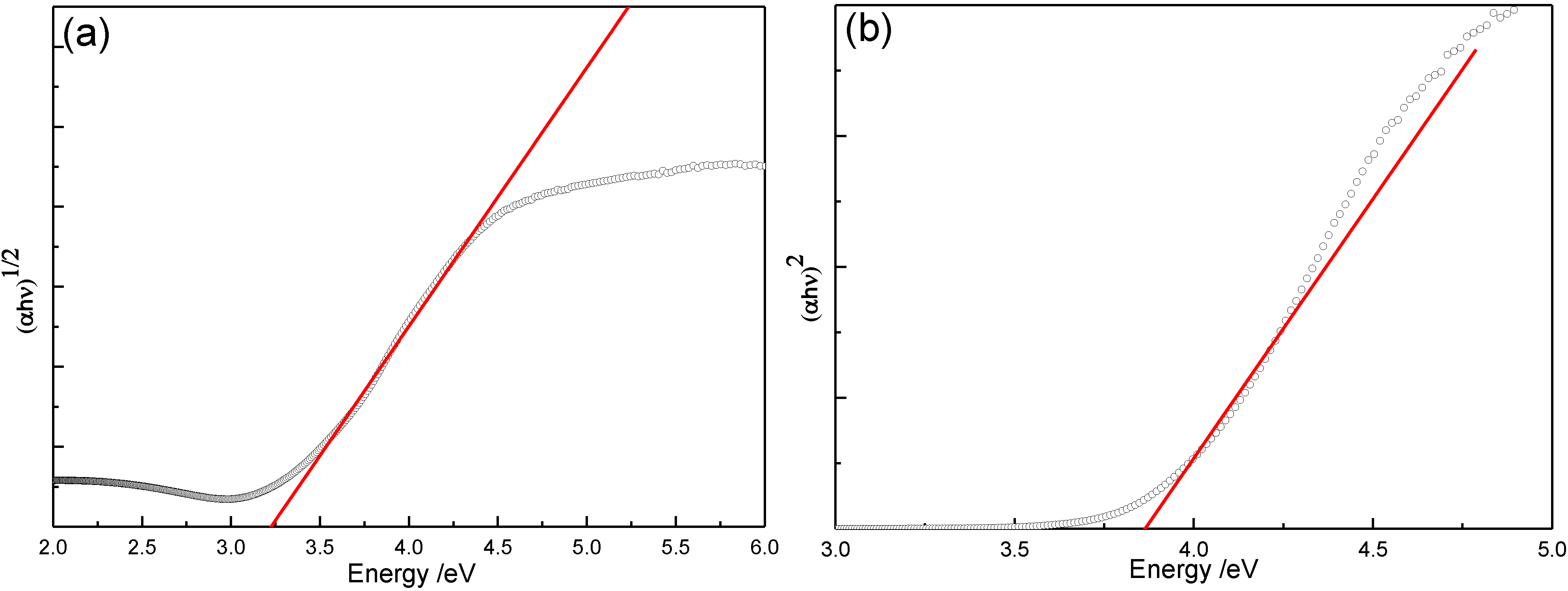
| Sample/°C | 400 | 500 | 600 | 700 | 800 | 900 |
|---|---|---|---|---|---|---|
| Eg-indirect/eV | 3.22 | 3.17 | 3.15 | 3.09 | 2.97 | 2.93 |
| Eg-direct/eV | 3.86 | 3.82 | 3.79 | 3.77 | 3.67 | 3.65 |
3.3. Surface Morphology

3.4. Stability of TiO2 Sol
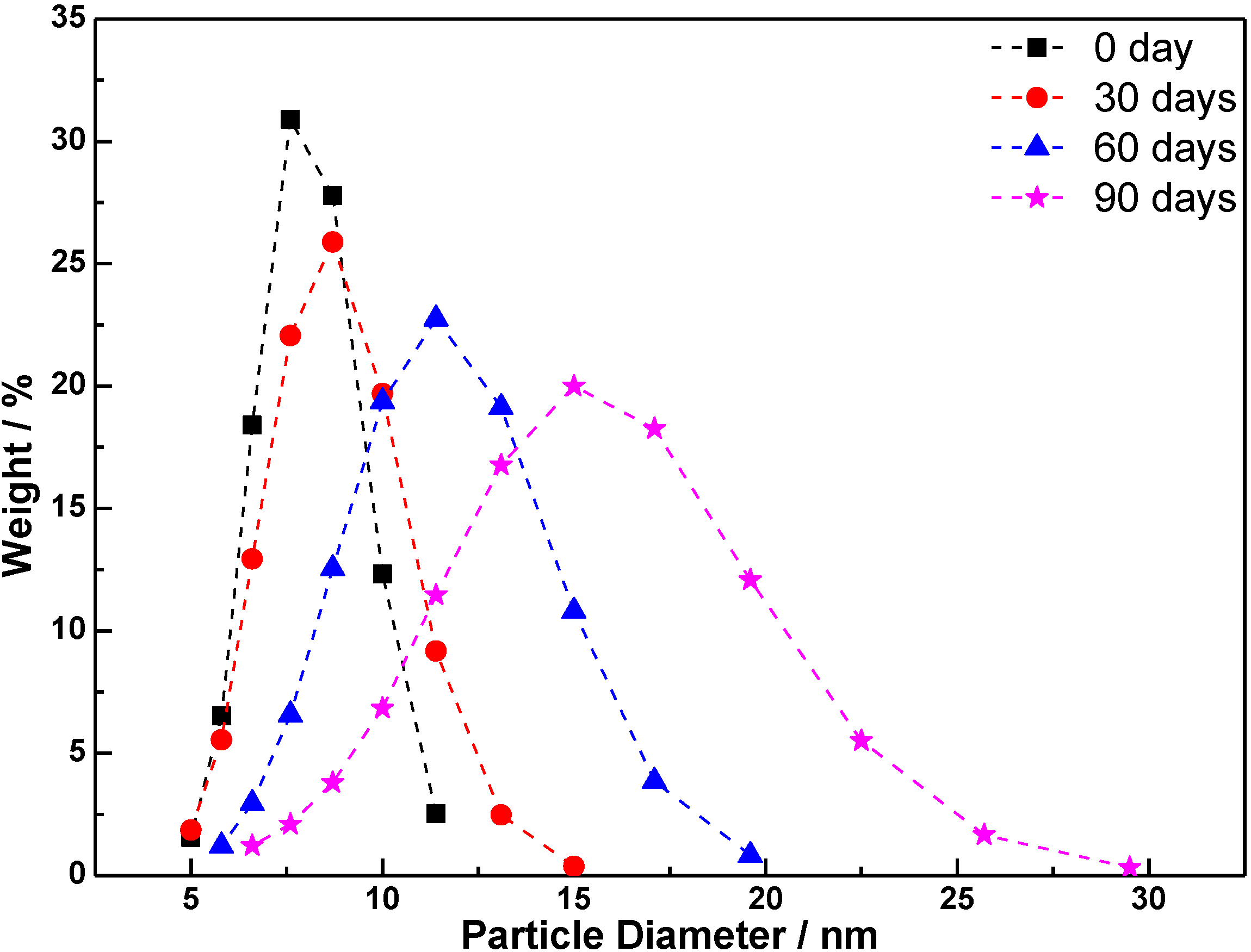
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Bi, Z.-F.; Wang, L.; Liu, X.-H.; Zhang, S.-M.; Dong, M.-M.; Zhao, Q.-Z.; Wu, X.-L.; Wang, K.-M. Optical waveguides in TiO2 formed by He ion implantation. Opt. Express 2012, 20, 6712–6719. [Google Scholar]
- Szeghalmi, A.; Helgert, M.; Brunner, R.; Heyroth, F.; Gösele, U.; Knez, M. Atomic layer deposition of Al2O3 and TiO2 multilayers for applications as bandpass filters and antireflection coatings. Appl. Opt. 2009, 48, 1727–1732. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shen, J. Sol-gel derived durable antireflective coating for solar glass. J. Sol-Gel Sci. Technol. 2010, 53, 322–327. [Google Scholar] [CrossRef]
- Peng, B.; Jungmann, G.; Jäger, C.; Haarer, D.; Schmidt, H.-W.; Thelakkat, M. Systematic investigation of the role of compact TiO2 layer in solid state dye-sensitized TiO2 solar cells. Coordin. Chem. Rev. 2004, 248, 1479–1489. [Google Scholar]
- Sauvage, F.; di Fonzo, F.; Li Bassi, A.; Casari, C.S.; Russo, V.; Divitini, G.; Ducati, C.; Bottani, C.E.; Comte, P.; Graetzel, M. Hierarchical TiO2 photoanode for dye-sensitized solar cells. Nano Lett. 2010, 10, 2562–2567. [Google Scholar] [CrossRef] [PubMed]
- Hamadanian, M.; Sayahi, H.; Zolfaghari, A. Modified multistep electrophoretic deposition of TiO2 nanoparticles to prepare high quality thin films for dye-sensitized solar cell. J. Mater. Sci. 2012, 47, 5845–5851. [Google Scholar] [CrossRef]
- Yamada, Y.; Uyama, H.; Watanabe, S.; Nozoye, H. Deposition at low substrate temperatures of high-quality TiO2 films by radical beam-assisted evaporation. Appl. Opt. 1999, 38, 6638–6641. [Google Scholar] [CrossRef] [PubMed]
- Eiamchai, P.; Chindaudom, P.; Pokaipisit, A.; Limsuwan, P. A spectroscopic ellipsometry study of TiO2 thin films prepared by ion-assisted electron-beam evaporation. Curr. Appl. Phys. 2009, 9, 707–712. [Google Scholar] [CrossRef]
- Albertinetti, N.; Minden, H.T. Granularity in ion-beam-sputtered TiO2 films. Appl. Opt. 1996, 35, 5620–5625. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.; Wang, W.-H.; Hsu, M.-Y.; Wang, L.-C. Characteristics of ion-beam-sputtered high-refractive-index TiO2-SiO2 mixed films. J. Opt. Soc. Am. A 1999, 16, 1477–1483. [Google Scholar] [CrossRef]
- Walczak, M.; Oujja, M.; Marco, J.; Sanz, M.; Castillejo, M. Pulsed laser deposition of TiO2: Diagnostic of the plume and characterization of nanostructured deposits. Appl. Phys. A Mater. 2008, 93, 735–740. [Google Scholar] [CrossRef]
- Gámez, F.; Plaza-Reyes, A.; Hurtado, P.; Guillén, E.; Anta, J.A.; Martínez-Haya, B.; Pérez, S.; Sanz, M.; Castillejo, M.; Izquierdo, J.G.; et al. Nanoparticle TiO2 films prepared by pulsed laser deposition: Laser desorption and cationization of model adsorbates. J. Phys. Chem. C 2010, 114, 17409–17415. [Google Scholar] [CrossRef]
- Dzibrou, D.; Grishin, A.M.; Kawasaki, H.; Suda, Y.; Pankov, V.V. Tailoring optical properties of pulsed laser deposited TiO2 films. J. Phys. Conf. Ser. 2008, 100. [Google Scholar] [CrossRef]
- Ayouchi, R.; Casteleiro, C.; Schwarz, R.; Barrado, J.R.; Martín, F. Optical properties of TiO2 thin films prepared by chemical spray pyrolysis from aqueous solutions. Phys. Status Solidi C 2010, 7, 933–936. [Google Scholar] [CrossRef]
- Battiston, G.A.; Gerbasi, R.; Gregori, A.; Porchia, M.; Cattarin, S.; Rizzi, G.A. PECVD of amorphous TiO2 thin films: Effect of growth temperature and plasma gas composition. Thin Solid Films 2000, 371, 126–131. [Google Scholar] [CrossRef]
- McCurdy, P.R.; Sturgess, L.J.; Kohli, S.; Fisher, E.R. Investigation of the PECVD TiO2–Si(100) interface. Appl. Surf. Sci. 2004, 233, 69–79. [Google Scholar] [CrossRef]
- Phadke, S.; Sorge, J.D.; Hachtmann, S.; Birnie, D.P., III. Broad band optical characterization of sol-gel TiO2 thin film microstructure evolution with temperature. Thin Solid Films 2010, 518, 5467–5470. [Google Scholar] [CrossRef]
- Sankapal, B.R.; Lux-Steiner, M.C.; Ennaoui, A. Synthesis and characterization of anatase-TiO2 thin films. Appl. Surf. Sci. 2005, 239, 165–170. [Google Scholar] [CrossRef]
- Chrysicopoulou, P.; Davazoglou, D.; Trapalis, C.; Kordas, G. Optical properties of very thin (<100 nm) sol-gel TiO2 films. Thin Solid Films 1998, 323, 188–193. [Google Scholar]
- Yusuf, M.M.; Imai, H.; Hirashima, H. Preparation of porous titania film by modified sol-gel method and its application to photocatalyst. J. Sol-Gel Sci. Technol. 2002, 25, 65–74. [Google Scholar] [CrossRef]
- Wen, T.; Gao, J.; Shen, J.; Zhou, Z. Preparation and characterization of TiO2 thin films by the sol-gel process. J. Mater. Sci. 2001, 36, 5923–5926. [Google Scholar] [CrossRef]
- Wang, X.; Shen, J.; Pan, Q. Raman spectroscopy of sol-gel derived titanium oxide thin films. J. Raman Spectrosc. 2011, 42, 1578–1582. [Google Scholar] [CrossRef]
- Yao, J.K.; Huang, H.L.; Ma, J.Y.; Jin, Y.X.; Zhao, Y.A.; Shao, J.D.; He, H.B.; Yi, K.; Fan, Z.X.; Zhang, F.; Wu, Z.Y. High refractive index TiO2 film deposited by electron beam evaporation. Surf. Eng. 2009, 25, 257–260. [Google Scholar] [CrossRef]
- Wang, H.; Hu, Y.; Zhang, L.; Li, C. Self-cleaning films with high transparency based on TiO2 nanoparticles synthesized via flame combustion. Ind. Eng. Chem. Res. 2010, 49, 3654–3662. [Google Scholar] [CrossRef]
- Zhao, L.; Han, M.; Lian, J. Photocatalytic activity of TiO2 films with mixed anatase and rutile structures prepared by pulsed laser deposition. Thin Solid Films 2008, 516, 3394–3398. [Google Scholar] [CrossRef]
- Wang, X.; Shen, J.; Wang, S.; Zhang, Z. Optical constants of sol-gel derived TiO2 films characterized by spectroscopic ellipsometry. Acta Phys. Sin. 2009, 58, 8027–8032. [Google Scholar]
- Ohsaka, T.; Izumi, F.; Fujiki, Y. Raman spectrum of anatase, TiO2. J. Raman Spectrosc. 1978, 7, 321–324. [Google Scholar] [CrossRef]
- Zhang, W.F.; He, Y.L.; Zhang, M.S.; Yin, Z.; Chen, Q. Raman scattering study on anatase TiO2 nanocrystals. J. Phys. D Appl. Phys. 2000, 33, 912–916. [Google Scholar] [CrossRef]
- Jenkins, F.A.; White, H.E. Fundamentals of Optics, 4th ed.; McGraw-Hill, Inc.: New York, NY, USA, 1981. [Google Scholar]
- Wang, X.; Wu, G.; Zhou, B.; Shen, J. Improvement on laser-induced damage threshold of sol-gel ZrO2 coatings by crystal structure tuning. Opt. Express 2012, 20, 24482–24487. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, G.; Zhou, B.; Shen, J. Effect of crystal structure on optical properties of sol-gel derived zirconia thin films. J. Alloys Compd. 2013, 556, 182–187. [Google Scholar] [CrossRef]
- Fabes, B.D.; Birnie, D.P., III; Zelinski, B.J.J. Porosity and composition effects in sol-gel derived interference filters. Thin Solid Films 1995, 254, 175–180. [Google Scholar] [CrossRef]
- Pankove, I. Optical Processes in Semiconductors; Prentice-Hall: Englewood Cliffs, NJ, USA, 1971. [Google Scholar]
- Janitabar-Darzi, S.; Mahjoub, A.R.; Nilchi, A. Investigation of structural, optical and photocatalytic properties of mesoporous TiO2 thin film synthesized by sol-gel templating technique. Physica E 2009, 42, 176–181. [Google Scholar] [CrossRef]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical properties and electronic structure of amorphous germanium. Phys. Status Solidi 1966, 15, 627–637. [Google Scholar] [CrossRef]
- Ghrairi, N.; Bouaicha, M. Structural, morphological, and optical properties of TiO2 thin films synthesized by the electro phoretic deposition technique. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Richards, B.S. Novel Uses of Titanium Dioxide for Silicon Solar Cells. Ph.D. Thesis, University of New South Wales, Sydney, Australia, 2002. [Google Scholar]
- Birnie, D.P., III; Bendzko, N.J. 1H and 13C NMR observation of the reaction of acetic acid with titanium isopropoxide. Mater. Chem. Phys. 1999, 59, 26–35. [Google Scholar] [CrossRef]
- Birnie, D.P., III. Esterification kinetics in titanium isopropoxide-acetic acid solutions. J. Mater. Sci. 2000, 35, 367–374. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.; Wu, G.; Zhou, B.; Shen, J. Optical Constants of Crystallized TiO2 Coatings Prepared by Sol-Gel Process. Materials 2013, 6, 2819-2830. https://doi.org/10.3390/ma6072819
Wang X, Wu G, Zhou B, Shen J. Optical Constants of Crystallized TiO2 Coatings Prepared by Sol-Gel Process. Materials. 2013; 6(7):2819-2830. https://doi.org/10.3390/ma6072819
Chicago/Turabian StyleWang, Xiaodong, Guangming Wu, Bin Zhou, and Jun Shen. 2013. "Optical Constants of Crystallized TiO2 Coatings Prepared by Sol-Gel Process" Materials 6, no. 7: 2819-2830. https://doi.org/10.3390/ma6072819
APA StyleWang, X., Wu, G., Zhou, B., & Shen, J. (2013). Optical Constants of Crystallized TiO2 Coatings Prepared by Sol-Gel Process. Materials, 6(7), 2819-2830. https://doi.org/10.3390/ma6072819






