Thermal Annealing Effect on Optical Properties of Binary TiO2-SiO2 Sol-Gel Coatings
Abstract
:1. Introduction
2. Experimental Section
2.1. Sol Synthesis
2.2. Coating Preparation
2.3. Characterization
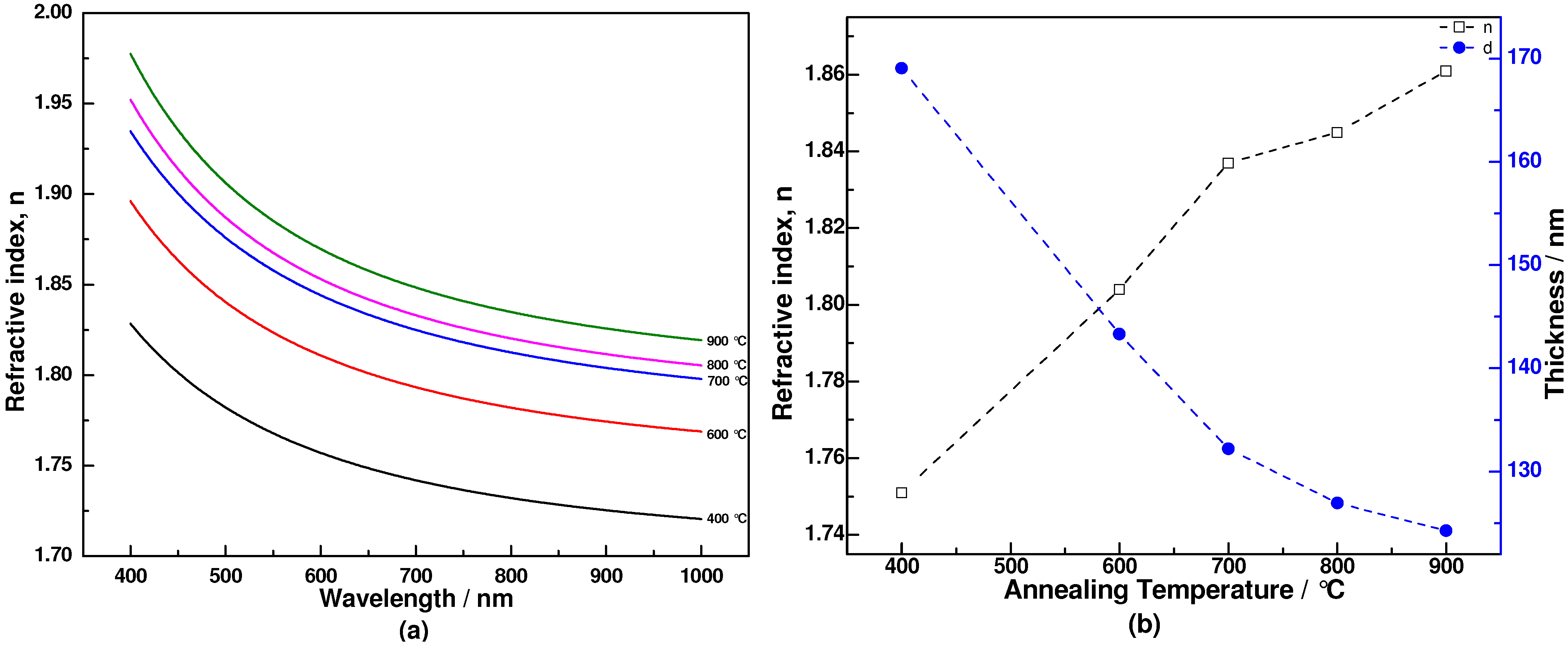
3. Results and Discussion
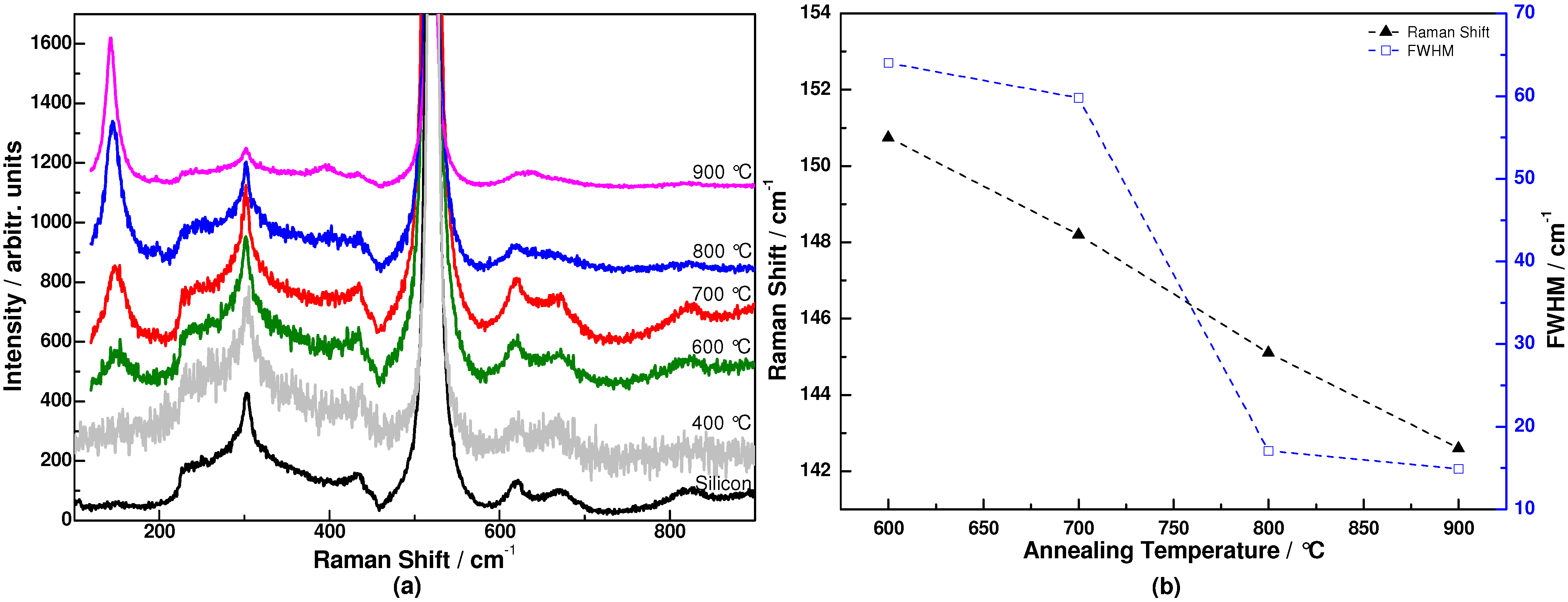
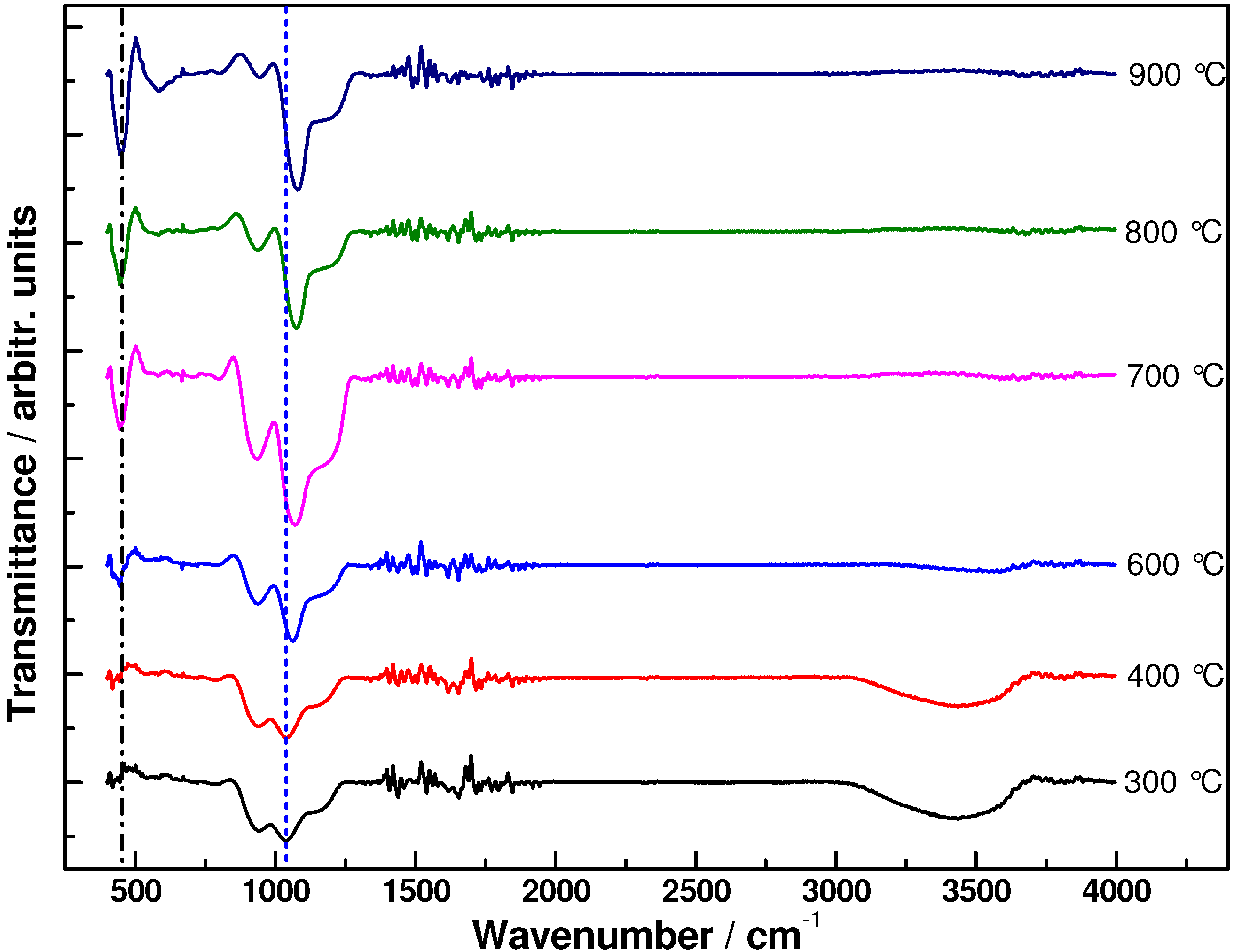
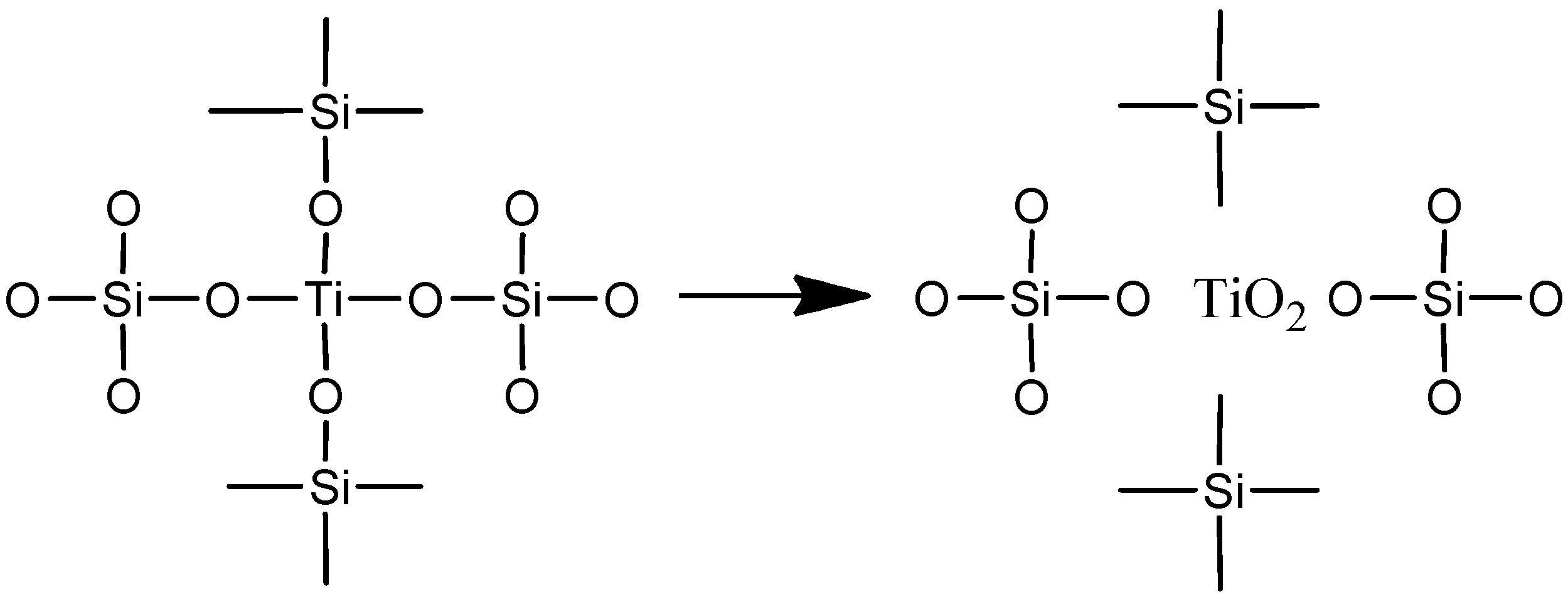
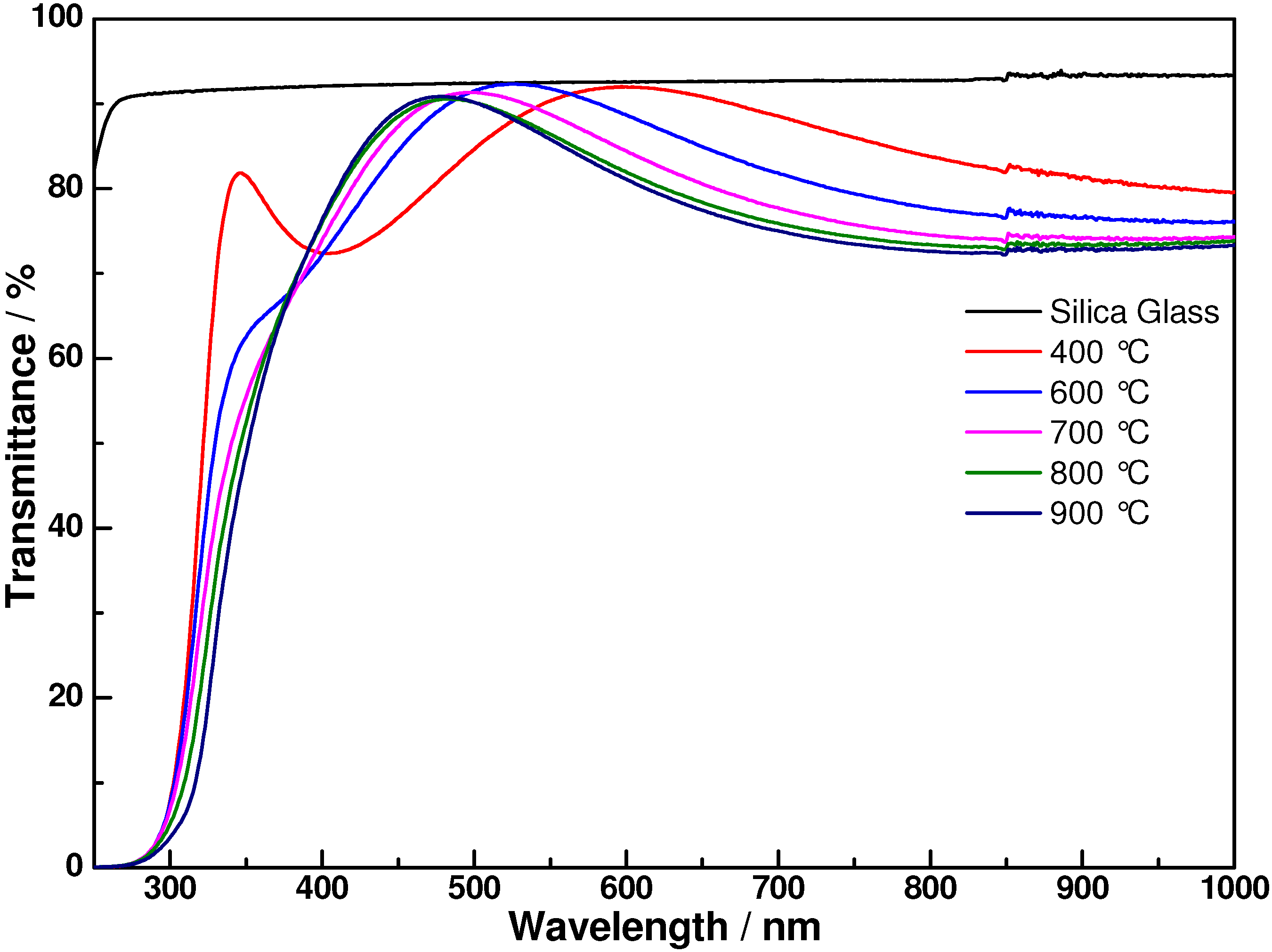
4. Conclusions
Acknowledgments
References
- Pal, M.; Pal, U.; Jiménez, J.M.G.Y.; Pérez-Rodríguez, F. Effects of crystallization and dopant concentration on the emission behavior of TiO2:Eu nanophosphors. Nanoscale Res. Lett. 2012, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shen, J. Sol-gel derived durable antireflective coating for solar glass. J. Sol-Gel Sci. Technol. 2010, 53, 322–327. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, X.; Wang, Z. High-damage-threshold broadband chirped mirror. Chin. Opt. Lett. 2010, 8, 156–158. [Google Scholar] [CrossRef]
- Chen, S.-H.; Wang, C.-H.; Chai, K.-Y.; Chang, T.-H.; Yeh, Y.-W.; Lee, C.-C.; Ku, S.-L.; Huang, C.-C. Polarization beam splitters with autocloned symmetric structure. In Proceeding of Optical Interference Coatings, Tucson, AZ, USA, 6 June 2010.
- Que, W.; Zhou, Y.; Lam, Y.L.; Chan, Y.C.; Cheng, S.D.; Li, H.P.; Liu, J.; Kam, C.H. TiO2/SiO2/ORMOSIL hybrid material planar waveguides prepared at low-temperature by sol-gel processing. Proc. SPIE 1999, 3896, 446–452. [Google Scholar]
- Yang, L.L.; Lai, Y.S.; Chen, J.S.; Tsai, P.H.; Chen, C.L.; Chang, C.J. Compositional tailored sol-gel SiO2-TiO2 thin films: Crystallization, chemical bonding configuration, and optical properties. J. Mater. Res. 2005, 20, 3141–3149. [Google Scholar] [CrossRef]
- Gracia, F.; Yubero, F.; Holgado, J.P.; Espinos, J.P.; Gonzalez-Elipe, A.R.; Girardeau, T. SiO2/TiO2 thin films with variable refractive index prepared by ion beam induced and plasma enhanced chemical vapor deposition. Thin Solid Films 2006, 500, 19–26. [Google Scholar] [CrossRef]
- Sankur, H.; Gunning, W. Crystallization and diffusion in composite TiO2-SiO2 thin films. J. Appl. Phys. 1989, 66, 4747–4751. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Qian, L.Q.; Guo, C.; Jia, X.; Wang, J.W.; Tang, W.H. Natural superhydrophilic TiO2/SiO2 composite thin films deposited by radio frequency magnetron sputtering. J. Alloy. Compd. 2009, 479, 532–535. [Google Scholar] [CrossRef]
- Sarkar, D.K.; Desbiens, E.; Khakani, M.A.E. High-k titanium silicate dielectric thin films grown by pulsed-laser deposition. Appl. Phys. Lett. 2002, 80, 294–296. [Google Scholar] [CrossRef]
- Nilchi, A.; Janitabar-Darzi, S.; Mahjoub, A.R.; Rasouli-Garmarodi, S. New TiO2/SiO2 nanocomposites-phase transformations and photocatalytic studies. Colloids Surf. A 2010, 361, 25–30. [Google Scholar] [CrossRef]
- Louis, B.; Krins, N.; Faustini, M.; Grosso, D. Understanding crystallization of anatase into binary SiO2/TiO2 sol-gel optical thin films: An in situ thermal ellipsometry analysis. J. Phys. Chem. C 2011, 115, 3115–3122. [Google Scholar] [CrossRef]
- Song, C.F.; Lv, M.K.; Yang, P.; Xu, D.; Yuan, D.R. Structure and photoluminescence properties of sol-gel TiO2-SiO2 films. Thin Solid Films 2002, 413, 155–159. [Google Scholar] [CrossRef]
- Hodroj, A.; Chaix-Pluchery, O.; Audier, M.; Gottlieb, U.; Deschanvres, J.L. Thermal annealing of amorphous Ti–Si–O thin films. J. Mater. Res. 2008, 23, 755–759. [Google Scholar] [CrossRef]
- Smitha, V.; Manjumol, K.; Baiju, K.; Ghosh, S.; Perumal, P.; Warrier, K. Sol-gel route to synthesize titania-silica nano precursors for photoactive particulates and coatings. J. Sol-Gel Sci. Technol. 2010, 54, 203–211. [Google Scholar] [CrossRef]
- Machida, M.; Norimoto, K.; Watanabe, T.; Hashimoto, K.; Fujishima, A. The effect of SiO2 addition in super-hydrophilic property of TiO2 photocatalyst. J. Mater. Sci. 1999, 34, 2569–2574. [Google Scholar] [CrossRef]
- Wang, X.; Shen, J.; Pan, Q. Raman spectroscopy of sol-gel derived titanium oxide thin films. J. Raman Spectrosc. 2011, 42, 1578–1582. [Google Scholar] [CrossRef]
- Pärna, R.; Joost, U.; Nõmmiste, E.; Käämbre, T.; Kikas, A.; Kuusik, I.; Kink, I.; Hirsimäki, M.; Kisand, V. Effect of different annealing temperatures and SiO2/Si(100) substrate on the properties of nickel containing titania thin sol-gel films. Phys. Status Solidi A 2012, 209, 953–965. [Google Scholar] [CrossRef]
- Bersani, D.; Lottici, P.P.; Ding, X.-Z. Phonon confinement effects in the raman scattering by TiO2 nanocrystals. Appl. Phys. Lett. 1998, 72, 73–75. [Google Scholar] [CrossRef]
- Darmawan, A.; Smart, S.; Julbe, A.; Diniz da Costa, J.C. Iron oxide silica derived from sol-gel synthesis. Materials 2011, 4, 448–456. [Google Scholar] [CrossRef]
- Ono, H.; Ikarashi, T.; Ando, K.; Kitano, T. Infrared studies of transition layers at SiO2/Si interface. J. Appl. Phys. 1998, 84, 6064–6069. [Google Scholar] [CrossRef]
- Janicki, V.; Sancho-Parramon, J.; Stenzel, O.; Lappschies, M.; Görtz, B.; Rickers, C.; Polenzky, C.; Richter, U. Optical characterization of hybrid antireflective coatings using spectrophotometric and ellipsometric measurements. Appl. Opt. 2007, 46, 6084–6091. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, F.A.; White, H.E. Fundamentals of Optics, 4th ed.; McGraw-Hill, Inc.: New York, NY, USA, 1981. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, X.; Wu, G.; Zhou, B.; Shen, J. Thermal Annealing Effect on Optical Properties of Binary TiO2-SiO2 Sol-Gel Coatings. Materials 2013, 6, 76-84. https://doi.org/10.3390/ma6010076
Wang X, Wu G, Zhou B, Shen J. Thermal Annealing Effect on Optical Properties of Binary TiO2-SiO2 Sol-Gel Coatings. Materials. 2013; 6(1):76-84. https://doi.org/10.3390/ma6010076
Chicago/Turabian StyleWang, Xiaodong, Guangming Wu, Bin Zhou, and Jun Shen. 2013. "Thermal Annealing Effect on Optical Properties of Binary TiO2-SiO2 Sol-Gel Coatings" Materials 6, no. 1: 76-84. https://doi.org/10.3390/ma6010076
APA StyleWang, X., Wu, G., Zhou, B., & Shen, J. (2013). Thermal Annealing Effect on Optical Properties of Binary TiO2-SiO2 Sol-Gel Coatings. Materials, 6(1), 76-84. https://doi.org/10.3390/ma6010076





