2.2. Discrimination of Immature and Mature Fibers and Assessment of Cellulose Maturity
Mature and immature fibers are determined easily from conventional microscopy, but the degree of maturity is difficult to assess. This is because the microscopic procedure is subjective and depends on one’s judgment to assign the fibers into the appropriate class of either immature or mature fibers. On the ATR-FTIR spectral intensity differences between immature and mature fibers, Liu
et al. determined the key wavelengths first and then developed two simple algorithms (
R1 and
R2) for their discrimination [
16]. They observed that the
R1 values increase with
R2 values among a data set consisting of 402 seed bolls (
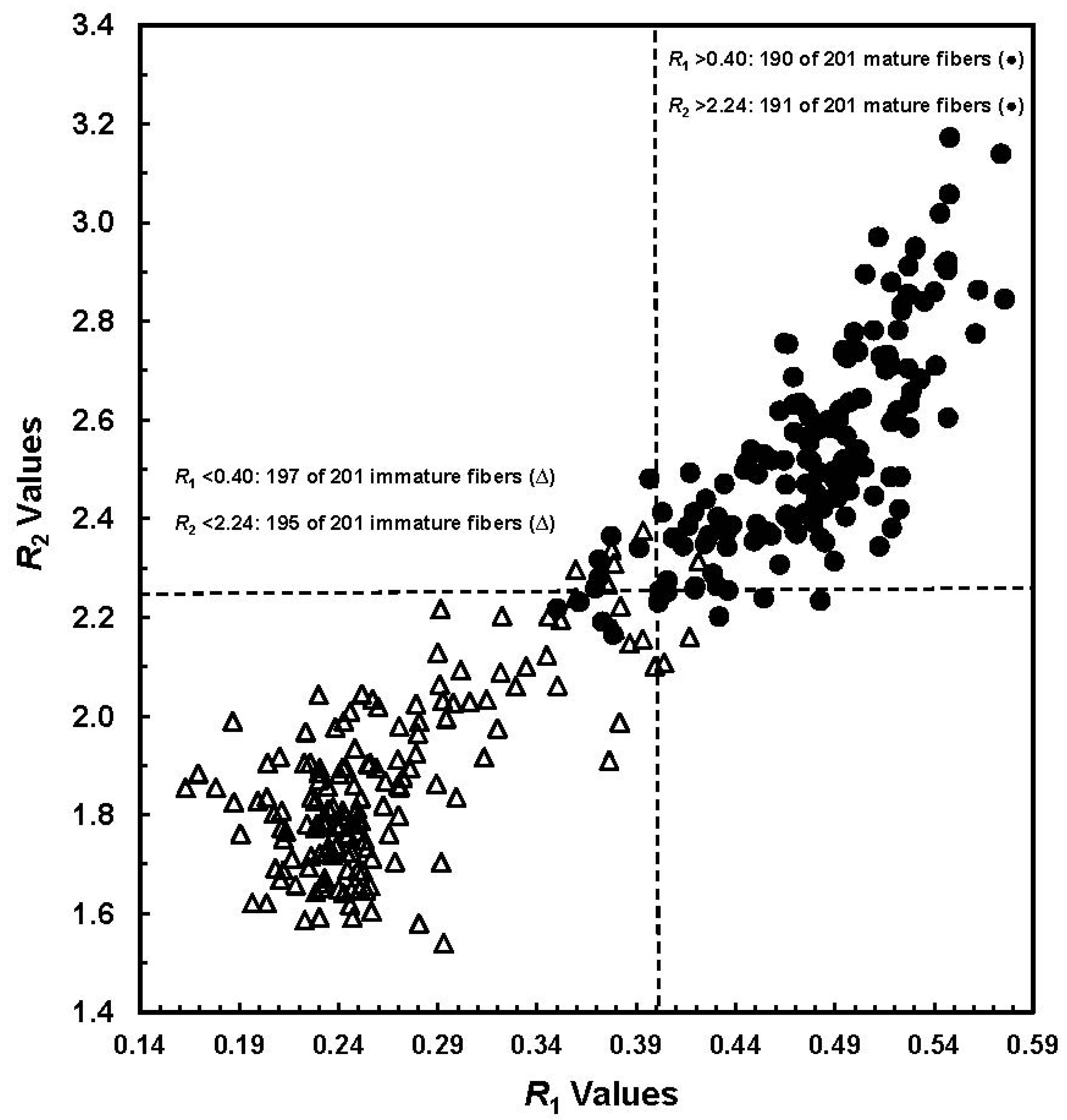
Figure 2). With an
R1 threshold value at 0.40, 197 of 201 (98.0%) immature fibers and 190 of 201 (94.5%) mature samples were correctly classified. By setting an
R2 threshold value at 2.24, only six immature fibers and 10 mature samples were misidentified, yielding an overall 96% accuracy in their correct differentiation.
Figure 2.
Plot of
R1 values
vs. R2 values from a total of 402 manually selected seed cottons (Reprinted with permission from [
16]. Copyright 2011 SAGE).
Figure 2.
Plot of
R1 values
vs. R2 values from a total of 402 manually selected seed cottons (Reprinted with permission from [
16]. Copyright 2011 SAGE).
Further, they proposed a formula to estimate the degree of cotton cellulose maturity (
MIR) by representing the
R1 values. In this concept, the
MIR values of 0.0 and 1.0 were assigned to the most immature and mature fibers, respectively. Thus, immature fibers whose
R1 < 0.40 correspond to a
MIR < 0.58 in the maturity range of 0 to 1.0, and
vice versa. In order to validate the efficiency of accessing the
MIR from direct ATR-FTIR measurement, cotton fibers with various maturity readings as determined from traditional image analysis (IA) and advanced fiber information system (AFIS) were taken. Within small and selected samples, strong correlations between
MIR against referenced IA and AFIS maturity readings were reported.
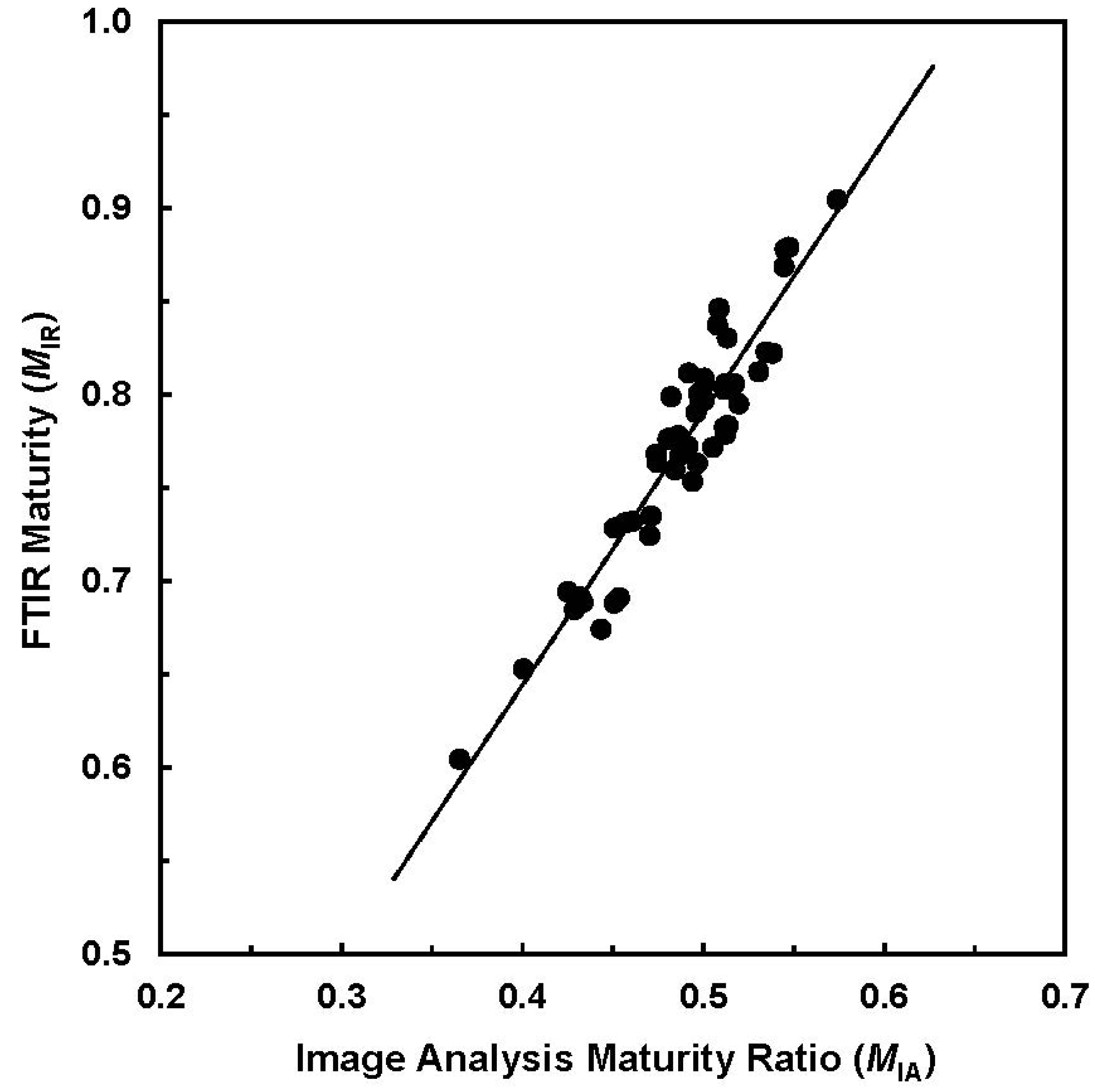
Figure 3 provides a relationship between two maturity measurements (IA against ATR-FTIR) for the selected 50 samples.
Figure 3.
Relationship of
MIA from image analysis (IA) procedure
vs. MIR from attenuated total reflection (ATR) sampling device based Fourier transform infrared (ATR-FTIR) measurement (R
2 = 0.894) (Reprinted with permission from [
16]. Copyright 2011 SAGE).
Figure 3.
Relationship of
MIA from image analysis (IA) procedure
vs. MIR from attenuated total reflection (ATR) sampling device based Fourier transform infrared (ATR-FTIR) measurement (R
2 = 0.894) (Reprinted with permission from [
16]. Copyright 2011 SAGE).
2.3. Determination of Cellulose Crystallinity
Numerous hydroxyl groups in fiber cellulose are involved in complicated intra- and inter-molecular hydrogen bonds, which yield high-order (crystalline) and low-order (amorphous) regions. Crystallinity index (
CI) has been used to describe the relative amount of crystalline portion in a simple two-phase model (crystalline
vs. amorphous). In practice,
CI of cotton fiber cellulose is determined predominantly by a curve-fitting process that extracts individual crystalline peaks from the X-ray diffraction (XRD) intensity profile. In general, XRD determination of cellulose
CI provides a qualitative or semi-quantitative evaluation of the amounts of either crystalline or amorphous components in a sample [
17]. Hence, appropriate cellulose standards are desired to calibrate or validate the XRD measurement. However, these absolute standards are not easy to be prepared or obtained.
In the preceding ATR-FTIR study of immature and mature cotton fibers [
16], the algorithm
R2 was linked with the relative amount of
Iβ to
Iα crystal form. Similar to the conception of representing the
R1 value, the
R2 values were converted to
CIIR [
18]. If the most immature and mature fibers were assigned the
CIIR value of 0% and 100%, respectively, the corresponding
R2 values should be the smallest and largest ones in
Figure 2. In turn, immature fibers with
R2 < 2.24 might have
CIIR < 42%, and mature fibers with
R2 > 2.24 have
CIIR of >42%.
Along with the idea identical to simple algorithms for ATR-FTIR spectral analysis, Liu
et al. developed a four-band ratio (
R3) that related the XRD intensity with the crystalline information [
18]. Relationship between the
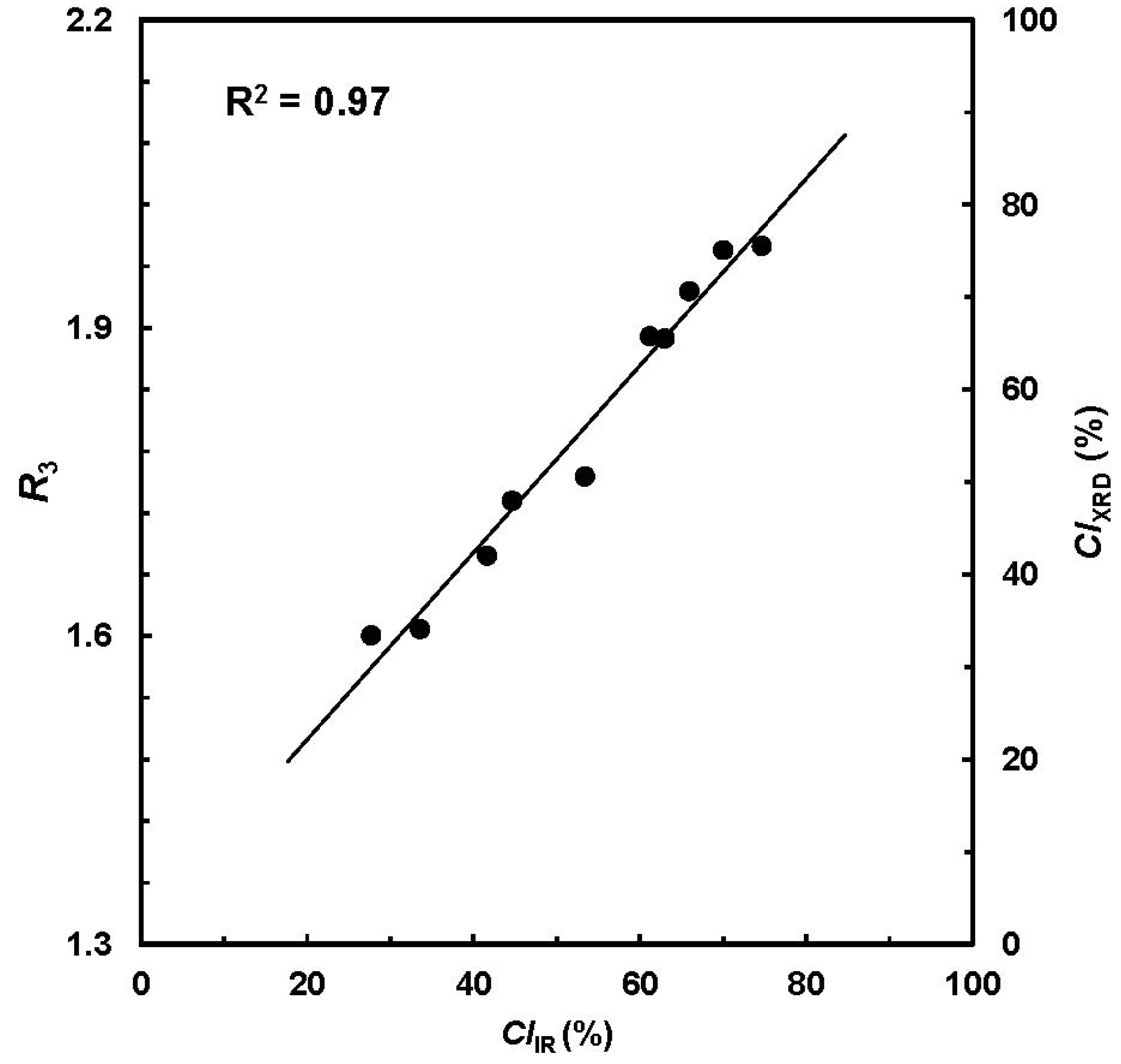
CIIR from ATR-FTIR spectra and
R3 from XRD pattern showed a high correlation of R
2 > 0.90 (
Figure 4). Next, the
R3 values were converted into respective
CIXRD readings in the range of 0% to 100%, which is depicted as a secondary vertical axis in
Figure 4.
Figure 4.
Plot of
CIIR from ATR-FTIR procedure
vs. CIXRD from X-ray diffraction (XRD) measurement. (Reprinted with permission from [
18]. Copyright 2012 Ingentaconnect)
Figure 4.
Plot of
CIIR from ATR-FTIR procedure
vs. CIXRD from X-ray diffraction (XRD) measurement. (Reprinted with permission from [
18]. Copyright 2012 Ingentaconnect)
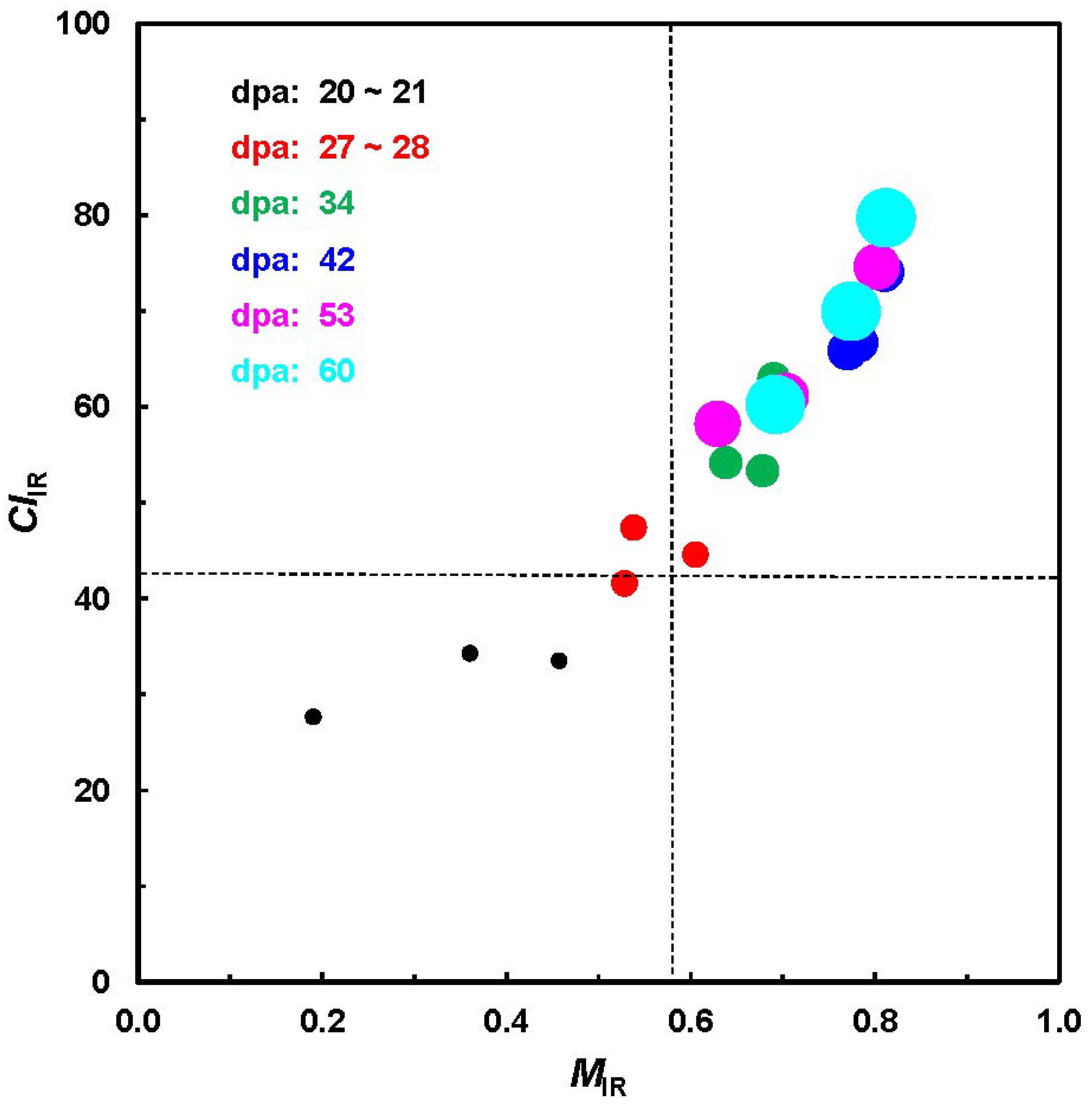
As one implementation,
Figure 5 shows the relationship between
MIR and
CIIR from a total of 18 cotton bolls, representing three varieties and six growth periods from the unique ATR-FTIR measurement. In the line of expectation, both
MIR and
CIIR increase from shorter to longer dpa for all varieties. The pattern in
Figure 5 is very similar to that in a previous investigation (
Figure 2) and also reveals a relatively clear separation among the fibers with differing dpa.
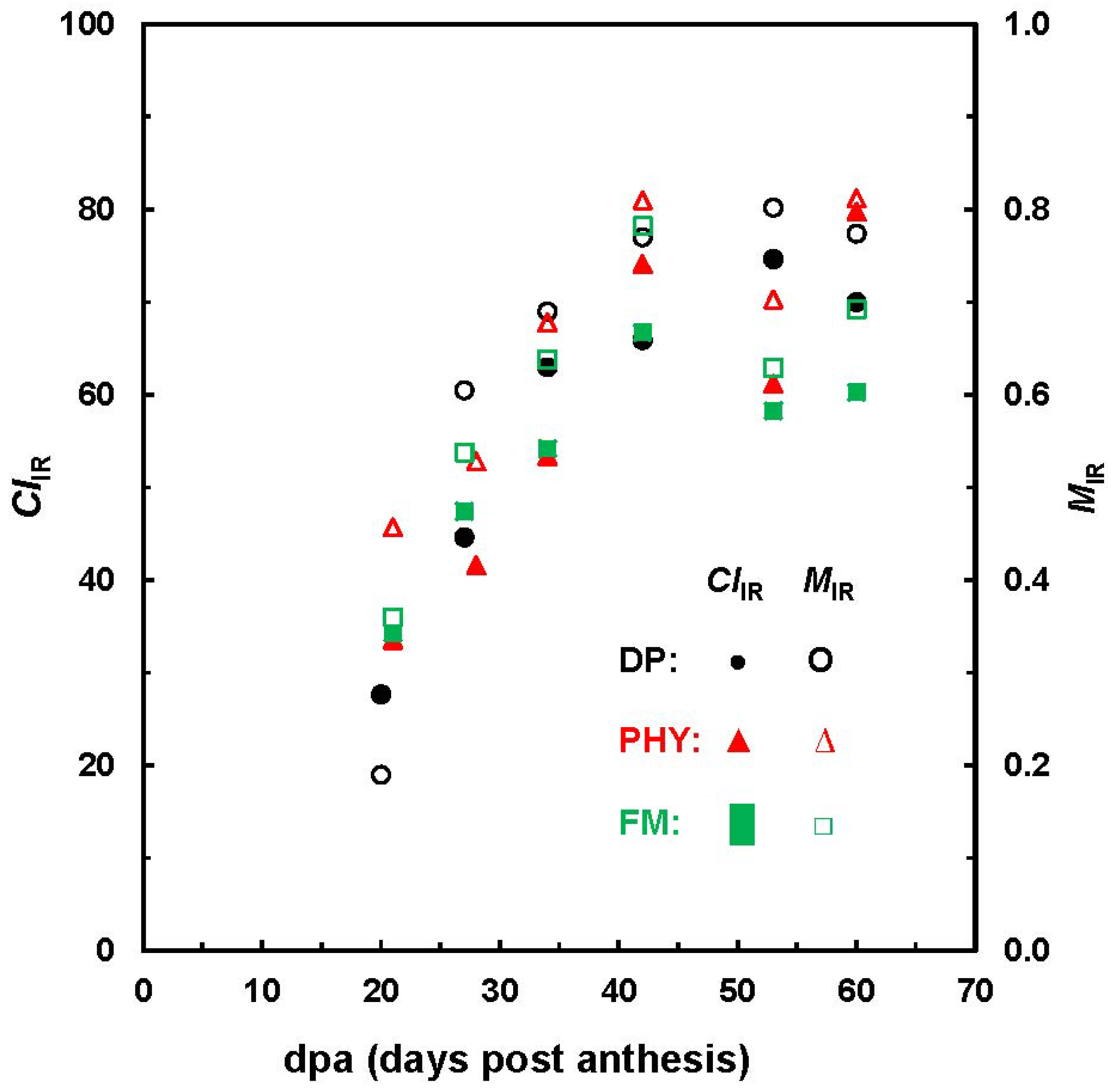
Figure 6 highlights the evolution of
MIR and
CIIR with developmental stages. In general, this observation is well consistent with those from XRD analysis on Maxxa and SJ-2 Acala varieties [
14]. Hsieh
et al. reported a steady increase of crystallinity from 38% to 57% between 24 and 60 dpa for Maxxa fibers and a maximum degree of crystallinity (55%) at 34 dpa for Acala fibers. Given the limited number of cotton bolls in this study, it is inappropriate to draw any solid comparisons about the three varieties examined, because fiber growth depends on a number of factors, such as genotype, grown locations and crop managements.
Figure 5.
Relationship of MIR against CIIR values from ATR-FTIR procedure.
Figure 5.
Relationship of MIR against CIIR values from ATR-FTIR procedure.
Figure 6.
Relationship between MIR and CIIR values against fiber dpa.
Figure 6.
Relationship between MIR and CIIR values against fiber dpa.
Careful examination of results from different strategies by independent investigators suggests that, in general, immature fibers could have the characteristics of less than 21–28 dpa,
MIR < 0.58 (in the maturity scale of 0 to 1.0) and
CIIR < 42% (in the crystallinity scale of 0 to 100%) [
11,
12,
14,
16,
18]. To interpret the practical samples, a margin of error of 5% to 10% or more should be considered.
2.4. Prediction of Cotton Stelometer Fiber Strength
Strength of cotton fibers is one of several important end-use attributes. To measure cotton strength at the level of either single fiber or flat bundle fibers, a number of testing devices have been in practice. A traditional laboratory testing method known as Stelometer is one of them, which is a different and also complementary tool to the automatic high volume instrument (HVI™) system [
19]. With the capability of micro sampling, ATR-FTIR spectra were collected on force-induced broken specimens in fiber bundles (
Figure 7) and then were correlated with Stelometer strength (or tenacity) values via partial least squares (PLS) regression [
19].
Figure 7.
Scheme of broken points to flat bundle fibers from Stelometer strength measurement and ATR-FTIR micro sampling spots (Reprinted with permission from [
19]. Copyright 2012 American Society of Agricultural & Biological Engineers).
Figure 7.
Scheme of broken points to flat bundle fibers from Stelometer strength measurement and ATR-FTIR micro sampling spots (Reprinted with permission from [
19]. Copyright 2012 American Society of Agricultural & Biological Engineers).
By comparing the statistics of optimal results in calibration and validation sets from individual spectral region, Liu
et al. reported the optimal PLS model performance from the 1800 to 800 cm
−1 region [
19]. However, the resultant spectral model indicated some difficulty in quantitative determination of cotton Stelometer strength for quality control purposes. They attributed this limitation to an apparent lack of uniformity of strength distribution in native fibers and different sampling spot size between spectral (~2 mm in diameter) and Stelometer (~10 mm width × 15 mm length) measurements. To remove the outlier samples that had large differences (or errors) between measured and spectral predicted strength, they applied a 90% confidence interval rule subjectively. As expected, the recalibrated model in the 1800 to 800 cm
−1 revealed a remarkable improvement in model performance. The result suggested the potential of the ATR-FTIR technique in the quantitative determination of fiber strength. Undoubtedly, more studies are necessary to unravel the strength quality trait and to elucidate the strength mechanism.
Hsieh
et al. probed the relationship between single fiber strengths and crystalline structures of greenhouse-grown Maxxa and Acala cotton fibers at varying growth stages [
14]. They found that single fiber breaking tenacities of Acala fibers do not vary at and beyond 21 dpa, while those of Maxxa cottons appear to be positively related to the secondary cell wall thickening process. They emphasized that, besides the crystallinity and crystallite sizes, other structural parameters, such as fibril orientation and residual stress, may also play key roles in affecting the single fiber strength of cotton fibers.
2.5. Two-Dimensional Correlation Characterization of Fiber Cellulose Development
Two-dimensional correlation (2D-Corr) spectroscopy, a universal and modern technique of vibrational spectral analysis, was originally developed as 2D IR correlation spectroscopy by Noda [
20]. Years later, he introduced a more applicable and simple mathematical formalism to perform the generalized 2D-Corr analysis [
21], which has been considerably applied to a variety of optical spectroscopic techniques under different types of external perturbations and waveforms.
Major advantages of 2D-Corr analysis include the enhancement of spectral resolution by spreading peaks over the second dimension, the band assignments through the correlation analysis and probing the complex sequence of events arising from the changes in a system. To obtain 2D-Corr spectra and also to interpret them reasonably, a limited number of spectra were arranged in an increasing or decreasing variable. On the other hand, one of specific challenges might be how to implement the 2D-Corr analysis in large and diverse spectral sets that are common in model development and include multivariate variations in chemical and physical properties. Being a method to explore the variations within a diverse data set, PCA was attempted to classify the spectra of samples [
22].
Despite the effort of extracting specific information from relatively sharp IR bands of cotton fibers in the 1800–600 cm
−1 fingerprint region, their spectral features have not been well understood, mainly due to the complexity of IR bands from a mixture of constituents in cotton fibers, slight spectral difference between fiber cellulose and noncellulosic polysaccharides (e.g., sugars), as well as between the amorphous and crystalline celluloses in the 1500–1200 cm
−1 region [
23]. In order to facilitate the interpretation of cotton cellulose IR spectra, Liu
et al. explored 2D-Corr spectroscopy to analyze the ATR bands in the 1800–650 cm
−1 IR region for spectral differences and characteristic band assignments between immature and mature fibers [
24]. Examples of 2D-Corr spectra of immature and mature fibers are given in
Figure 9 and
Figure 10, respectively, while their source spectra (or conventional one-dimensional spectra) are shown in
Figure 8 for comparison.
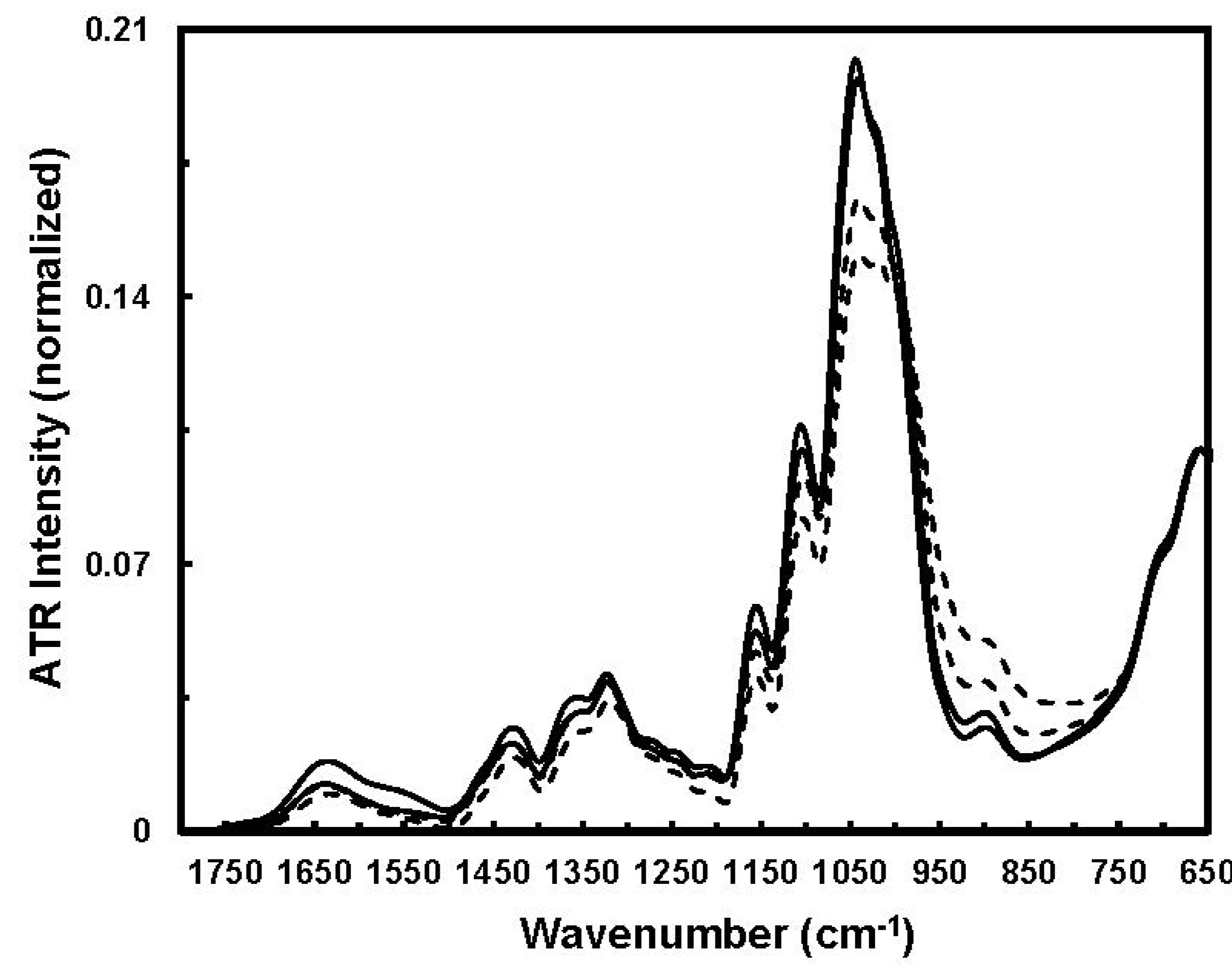
Figure 8 indicates apparent spectral intensity variations in the 1800–650 cm
−1 region between immature and mature fibers. With respect to cotton growth, cellulose concentrating induces fiber density augmentation and cellulose chain rearrangement through the formation of effective inter- and intra-molecular hydrogen bonding and also inter-chain van der Waals interactions. Clearly, the cellulose chains occur in amorphous and crystalline regions. Hence, a spectral intensity decrease in the 1050–1000 cm
−1 region might be associated with the functional groups in amorphous regions, and spectral intensity increase in the 1000–950 cm
−1 region could arise from crystalline regions.
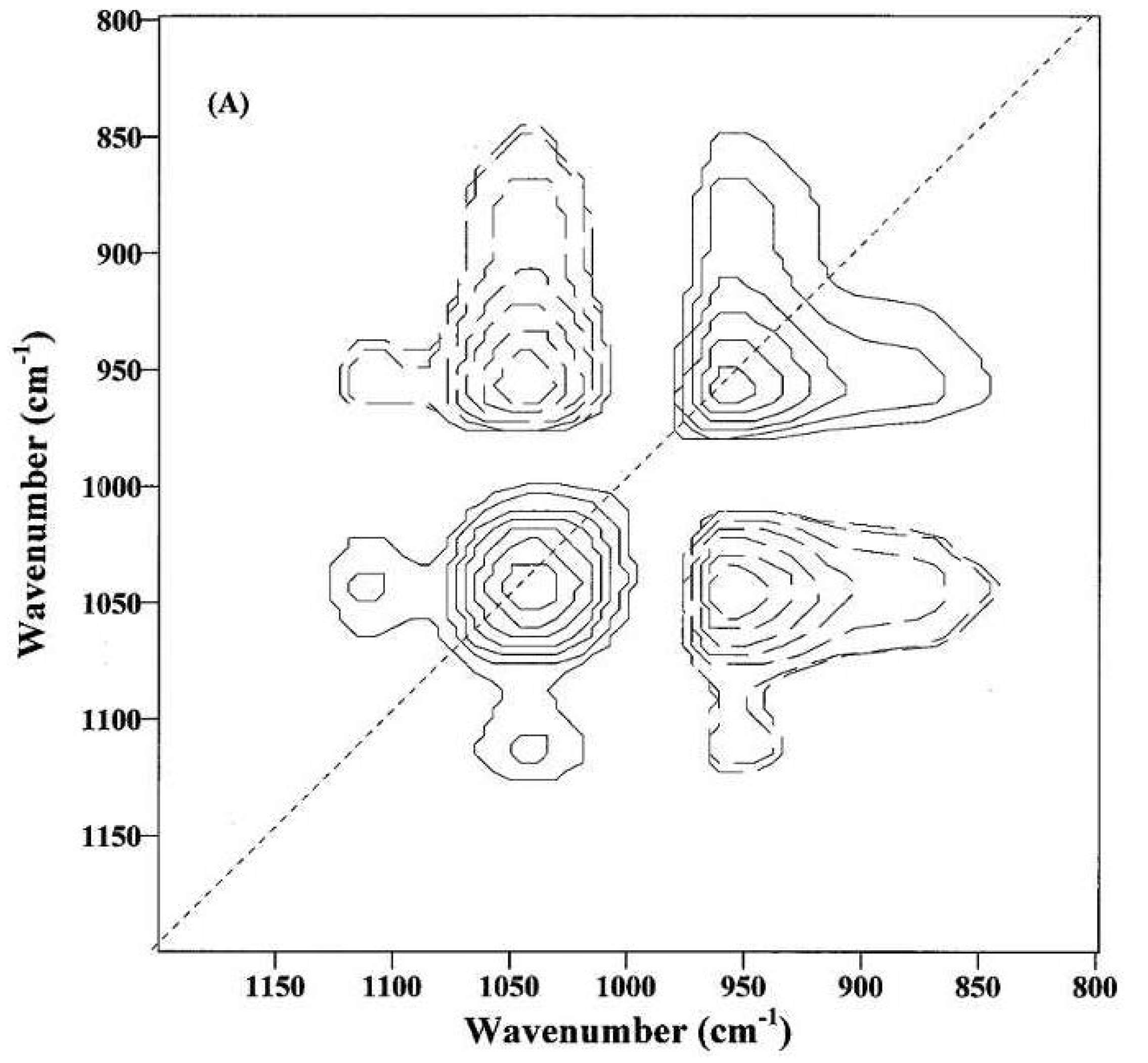
Figure 9 was obtained from immature fibers and might provide information on the compositional and structural changes during the initial fiber development. The appearance of at least two dominant autopeaks (1048 and 968 cm
−1) at the diagonal position indicates that spectral intensities of these two bands vary greatly with the growth of cotton fibers. The positive cross-peaks (solid lines) at the off-diagonal position are observed between the 1048 cm
−1 band and the two bands at 1152 and 1110 cm
−1, while the negative cross-peaks (dotted lines) are found between the 1048 cm
−1 band and the 968 cm
−1 band. Hence, the spectral intensity at 968 cm
−1 increases, while those at 1152, 1110 and 1048 cm
−1 decrease, as the cotton fibers grow.
Figure 8.
Typical ATR-FTIR spectra of immature (solid line) and mature (dashed line) fibers (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
Figure 8.
Typical ATR-FTIR spectra of immature (solid line) and mature (dashed line) fibers (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
Figure 9.
Two-dimensional correlation (2D-Corr) spectrum of immature fibers in the 1200–800 cm
−1 IR region (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
Figure 9.
Two-dimensional correlation (2D-Corr) spectrum of immature fibers in the 1200–800 cm
−1 IR region (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
With the biosynthesis of cellulose from small and non-cellulosic components (e.g., sugars), fiber cellulose is deposited, and subsequent concentrating might produce a relative increase of crystalline regions and simultaneous decrease of amorphous regions through more effective hydrogen bonding. Apparently, IR absorptions of cellulose chains in amorphous regions differ from those in crystalline regions. To this point, ATR intensity variations observed in
Figure 9 are mostly associated with the variations in fiber noncellulosic and cellulosic compositions, which undoubtedly could be presented in more or less the order of amorphous and crystalline regions. Consequently, an increase of spectral intensity at 968 cm
−1 could be related to the C–O stretching mode of primary alcohols (–C
6H
2–O
6H) in crystalline regions of fiber celluloses. On the other hand, the reduction of spectral intensity at 1048 cm
−1 might arise from at least one of two sources, either the C–O stretching mode of primary alcohols (–C
6H
2–O
6H) in amorphous regions of fiber celluloses or the C–O vibrational mode of such noncellulosic components as sugars.
An apparent distinction between
Figure 10, a 2D-Corr spectrum from mature fibers, and
Figure 9, is the appearance of at least two new autopeaks at 1042 and 956 cm
−1 and also several cross-peaks associated with them. Both autopeaks shift to lower frequencies compared to those at 1048 and 968 cm
−1 in
Figure 9, likely indicating an increase in the cellulose amount that produces more extensive molecular interactions and crystalline portions during this later stage. In the same manner, the reduction of the 1042 cm
−1 band intensity is positively correlated with the intensity decreases at 1152 and 1110 cm
−1 and also negatively correlated with the increase in the band intensity at 956 cm
−1. This suggests that the 956 cm
−1 band is associated with the C–O stretching mode of primary alcohols (–C
6H
2–O
6H) in crystalline celluloses, and the 1042 cm
−1 band might come from the C–O stretching mode of primary alcohols (–C
6H
2–O
6H) in amorphous celluloses.
Figure 10.
2D-Corr spectrum of mature fibers in the 1200–800 cm
−1 infrared (IR) region (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
Figure 10.
2D-Corr spectrum of mature fibers in the 1200–800 cm
−1 infrared (IR) region (Reprinted with permission from [
24]. Copyright 2012 Ingentaconnect).
Two cross-peaks at 1152 and 1110 cm
−1 in
Figure 10 appear at similar positions to those in
Figure 9, indicating that these two bands are greatly affected by the amount of cellulose instead of the hydrogen bonding effect that usually causes the shifting in absorption bands. Thus, these two bands could be from amorphous regions, and possibly, the 1152 cm
−1 band is mainly due to the antisymmetric C–O–C stretching mode of the glycosidic linkage, while the 1110 cm
−1 band can be attributed to the C–O stretching mode of secondary alcohols (–C
2–O
2H).
In addition, negative cross-peaks between the 1048 cm
−1 band in
Figure 9 or the 1042 cm
−1 band in
Figure 10, with at least one band at 895 cm
−1 from the 950 to 850 cm
−1 region, suggest the intensity increase of the 895 cm
−1 band with fiber development. This is anticipated, since intensity increase of the 895 cm
−1 band could be indicative of the increase in glycosidic linkage in fiber cellulose.