Deposition of Metal-Organic Frameworks by Liquid-Phase Epitaxy: The Influence of Substrate Functional Group Density on Film Orientation
Abstract
:1. Introduction
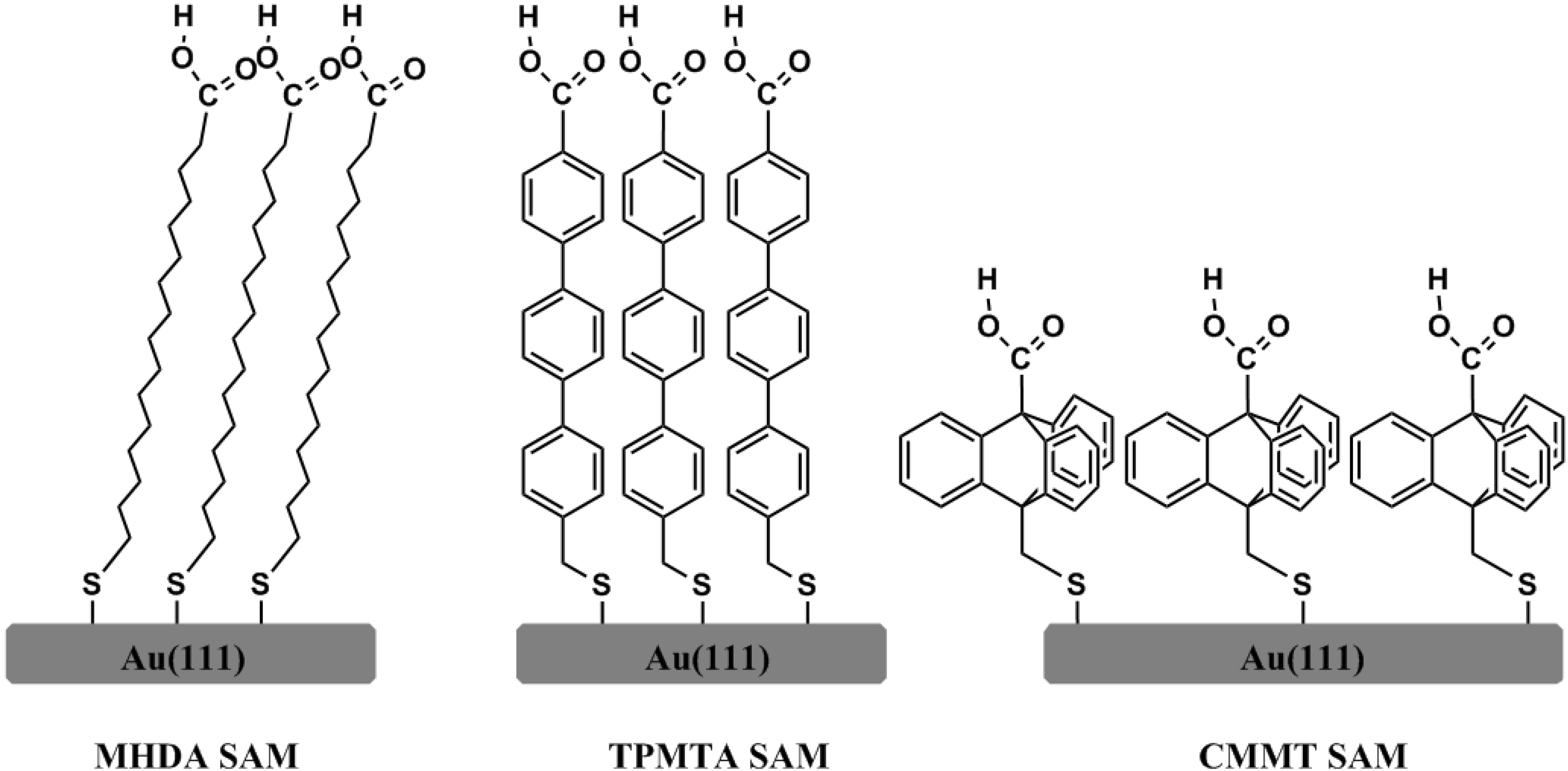
2. Experimental Section
2.1. SAMs Preparation
2.2. MOF Thin Films Preparation
2.3. X-ray Diffraction (XRD)
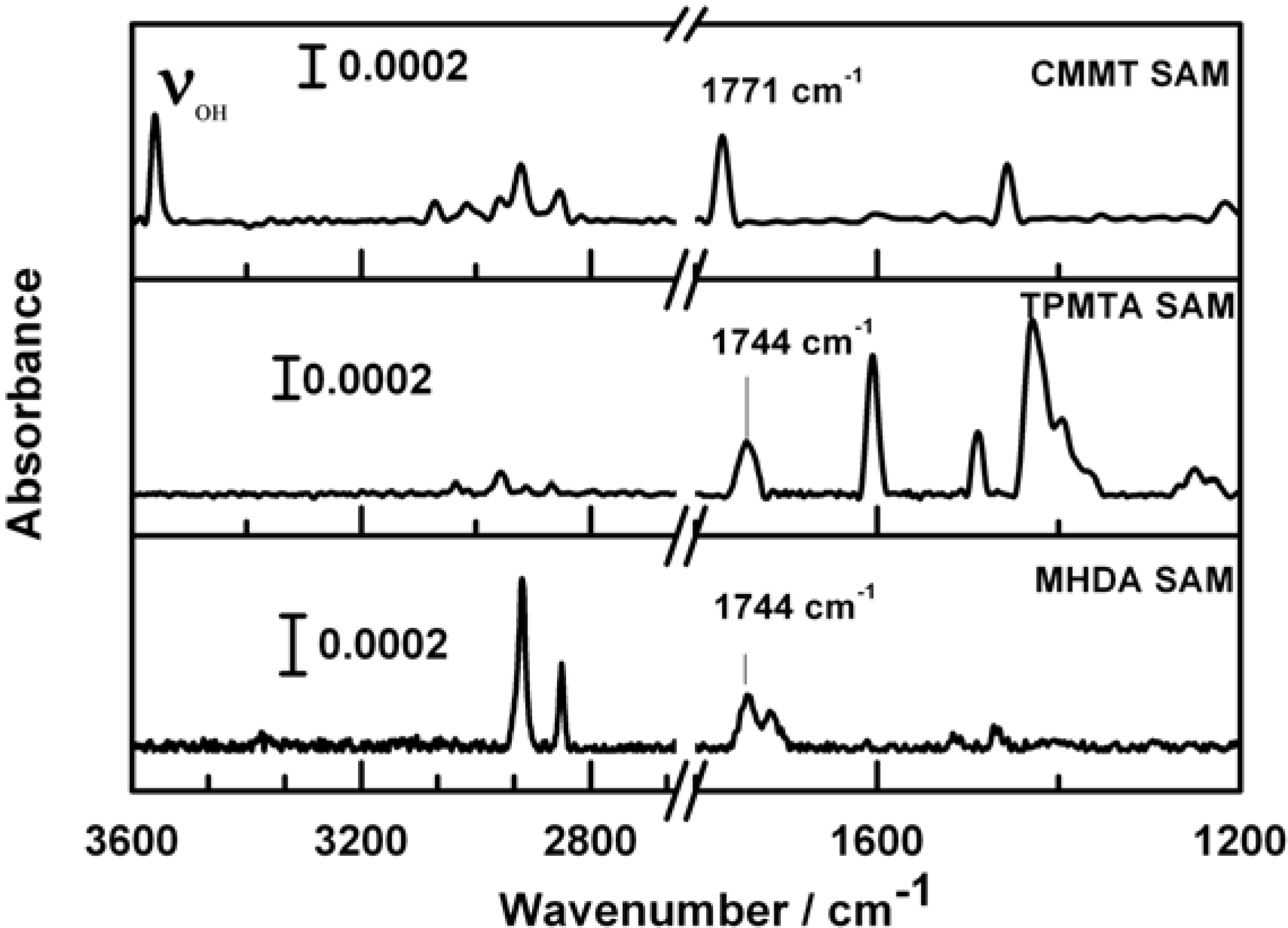
2.4. Infrared (IR) Spectroscopy
3. Results and Discussion
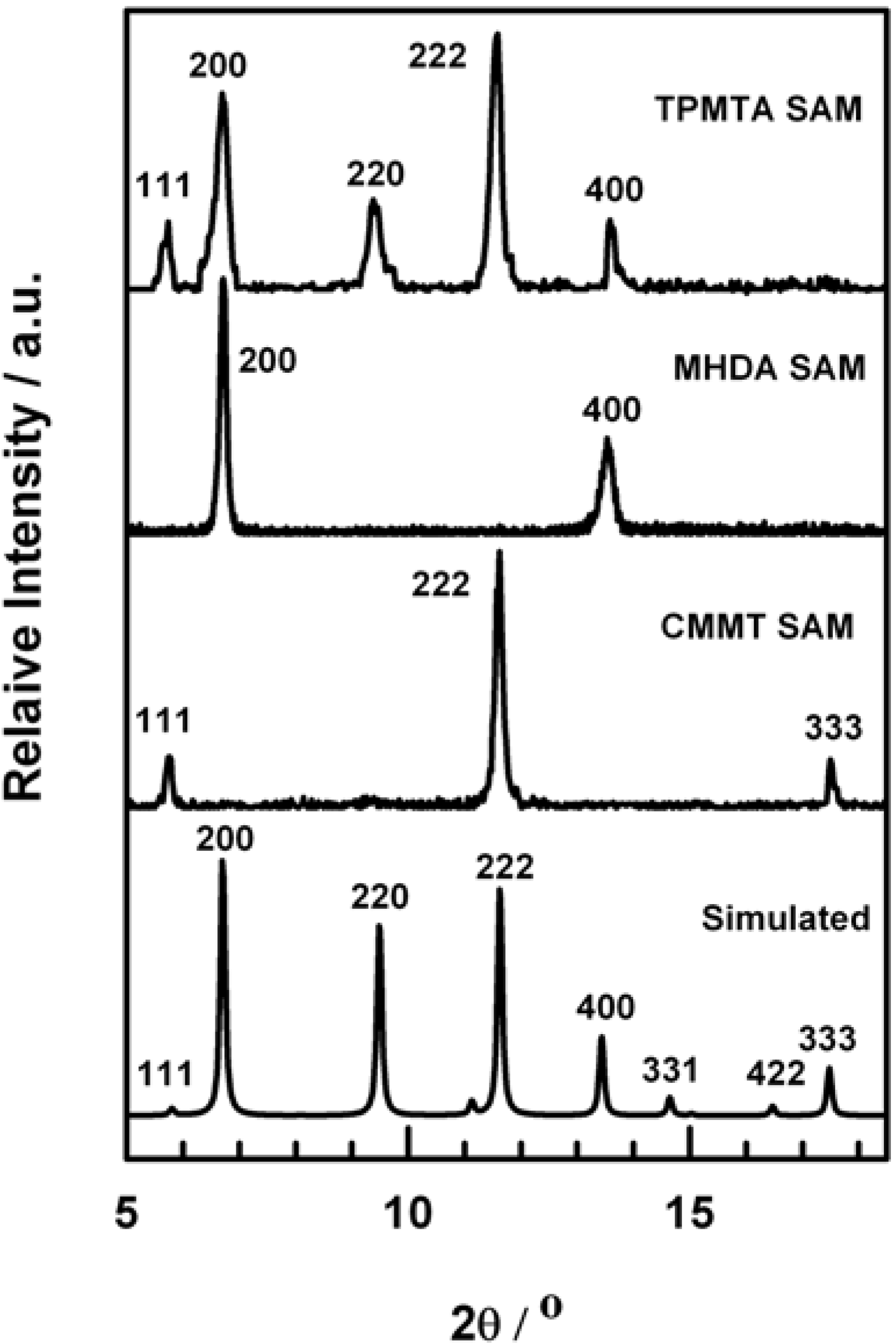
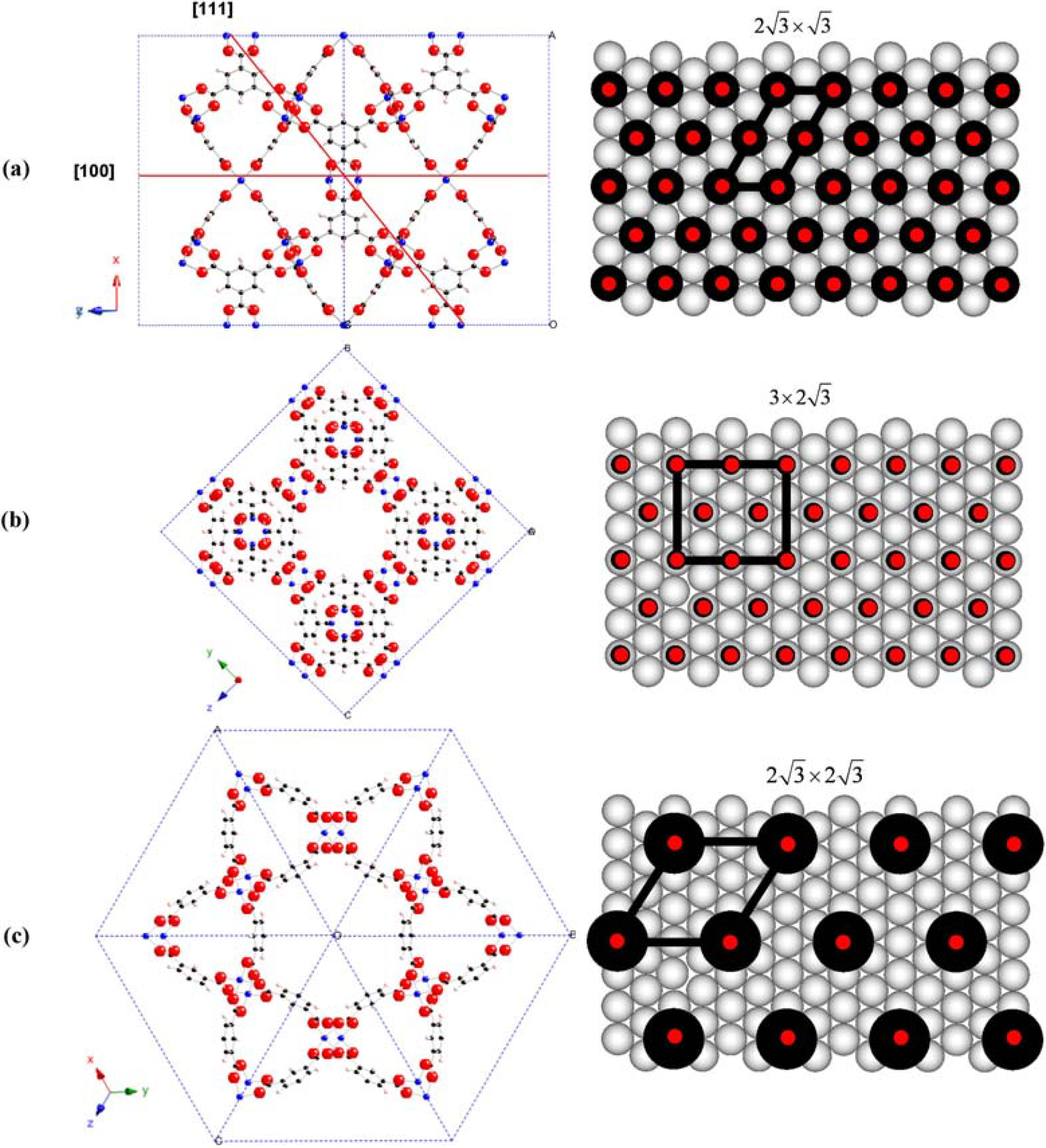
| SAMs | Unit cell | Area of unit cell (Å2) | Number of molecules per unit cell | Density (COOH groups per nm2) |
|---|---|---|---|---|
| MHDA | 86.6 | 4 | 4.6 | |
| TPMTA | 43.3 | 2 | 4.6 | |
| CMMT | 86.2 | 1 | 1.2 |
4. Conclusions
Acknowledgments
References
- Ferey, G. Microporous solids: From organically templated inorganic skeletons to hybrid frameworks ... ecumenism in chemistry. Chem. Mater. 2001, 13, 3084–3098. [Google Scholar] [CrossRef]
- Hoskins, B.F.; Robson, R. Design and construction of a new class of scaffolding-like materials comprising infinite polymeric frameworks of 3-D-linked molecular rods-a reappraisal of the Zn(Cn)2 and Cd(Cn)2 structures and the synthesis and structure of the diamond-related frameworks [N(Ch3)4][Cuiznii(Cn)4] and Cui[4,4′,4″,4‴-Tetracyanotetraphenylmethane]Bf4.XC6H5No2. J. Am. Chem. Soc. 1990, 112, 1546–1554. [Google Scholar] [CrossRef]
- Kitagawa, S.; Kitaura, R.; Noro, S. Functional porous coordination polymers. Angew. Chem. Int. Ed. 2004, 43, 2334–2375. [Google Scholar] [CrossRef]
- Rowsell, J.L.C.; Yaghi, O.M. Metal-organic frameworks: A new class of porous materials. Microporous Mesoporous Mater. 2004, 73, 3–14. [Google Scholar] [CrossRef]
- Maji, T.K.; Uemura, K.; Chang, H.C.; Matsuda, R.; Kitagawa, S. Expanding and shrinking porous modulation based on pillared-layer coordination polymers showing selective guest adsorption. Angew. Chem. Int. Ed. 2004, 43, 3269–3272. [Google Scholar] [CrossRef]
- Snurr, R.Q.; Hupp, J.T.; Nguyen, S.T. Prospects for nanoporous metal-organic materials in advanced separations processes. AIChE J. 2004, 50, 1090–1095. [Google Scholar] [CrossRef]
- Isaeva, V.I.; Kustov, L.M. The application of metal-organic frameworks in catalysis (Review). Petrol. Chem. 2010, 50, 167–180. [Google Scholar] [CrossRef]
- Lee, J.; Farha, O.K.; Roberts, J.; Scheidt, K.A.; Nguyen, S.T.; Hupp, J.T. Metal-organic framework materials as catalysts. Chem. Soc. Rev. 2009, 38, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- Ranocchiari, M.; van Bokhoven, J.A. Catalysis by metal-organic frameworks: Fundamentals and opportunities. Phys. Chem. Chem. Phys. 2011, 13, 6388–6396. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.S.; Whang, D.; Lee, H.; Jun, S.I.; Oh, J.; Jeon, Y.J.; Kim, K. A homochiral metal-organic porous material for enantioselective separation and catalysis. Nature 2000, 404, 982–986. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.Y.; Liu, C.J. Cu-3(BTC)(2): CO oxidation over MOF based catalysts. Chem. Commun. 2011, 47, 2167–2169. [Google Scholar] [CrossRef]
- Serre, C.; Horcajada, P.; Vallet-Regi, M.; Sebban, M.; Taulelle, F.; Ferey, G. Metal-organic frameworks as efficient materials for drug delivery. Angew. Chem. Int. Ed. 2006, 45, 5974–5978. [Google Scholar] [CrossRef]
- Allendorf, M.D.; Houk, R.J.T.; Andruszkiewicz, L.; Talin, A.A.; Pikarsky, J.; Choudhury, A.; Gall, K.A.; Hesketh, P.J. Stress-induced chemical detection using flexible metal-organic frameworks. J. Am. Chem. Soc. 2008, 130, 14404–14405. [Google Scholar] [CrossRef] [PubMed]
- Hermes, S.; Schroder, F.; Amirjalayer, S.; Schmid, R.; Fischer, R.A. Loading of porous metal-organic open frameworks with organometallic CVD precursors: Inclusion compounds of the type [LnM](a)@MOF-5. J. Mater. Chem. 2006, 16, 2464–2472. [Google Scholar] [CrossRef]
- Hermes, S.; Schroter, M.K.; Schmid, R.; Khodeir, L.; Muhler, M.; Tissler, A.; Fischer, R.W.; Fischer, R.A. Metal@MOF: Loading of highly porous coordination polymers host lattices by metal organic chemical vapor deposition. Angew. Chem. Int. Ed. 2005, 44, 6237–6241. [Google Scholar] [CrossRef]
- Meilikhov, M.; Yusenko, K.; Esken, D.; Turner, S.; van Tendeloo, G.; Fischer, R.A. Metals@MOFs-loading MOFs with metal nanoparticles for hybrid functions. Eur. J. Inorg. Chem. 2010, 2010, 3701–3714. [Google Scholar] [CrossRef]
- Cheetham, A.K.; Jain, P.; Ramachandran, V.; Clark, R.J.; Zhou, H.D.; Toby, B.H.; Dalal, N.S.; Kroto, H.W. Multiferroic behavior associated with an order-disorder hydrogen bonding transition in Metal-Organic Frameworks (MOFs) with the perovskite ABX(3) architecture. J. Am. Chem. Soc. 2009, 131, 13625–13627. [Google Scholar] [CrossRef] [PubMed]
- Drillon, M.; Rogez, G.; Viart, N. Multiferroic materials: The attractive approach of Metal-Organic Frameworks (MOFs). Angew. Chem. Int. Ed. 2010, 49, 1921–1923. [Google Scholar] [CrossRef]
- Ramesh, R. Materials Science emerging routes to multiferroics. Nature 2009, 461, 1218–1219. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.J.; Yue, Y.F.; Qian, G.D.; Chen, B.L. Luminescent functional metal-organic frameworks. Chem. Rev. 2012, 112, 1126–1162. [Google Scholar] [CrossRef] [PubMed]
- Ferey, G.; Millange, F.; Morcrette, M.; Serre, C.; Doublet, M.L.; Greneche, J.M.; Tarascon, J.M. Mixed-valence Li/Fe-based metal-organic frameworks with both reversible redox and sorption properties. Angew. Chem. Int. Ed. 2007, 46, 3259–3263. [Google Scholar] [CrossRef]
- Fischer, R.A.; Zacher, D.; Schmid, R.; Wöll, C. Surface chemistry of metal-organic frameworks at the liquid-solid interface. Angew. Chem. Int. Ed. 2011, 50, 176–199. [Google Scholar] [CrossRef]
- Shekhah, O.; Liu, J.; Fischer, R.A.; Wöll, C. MOF thin films: Existing and future applications. Chem. Soc. Rev. 2011, 40, 1081–1106. [Google Scholar] [CrossRef] [PubMed]
- Zacher, D.; Shekhah, O.; Wöll, C.; Fischer, R.A. Thin films of metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1418–1429. [Google Scholar] [CrossRef] [PubMed]
- Dragasser, A.; Shekhah, O.; Zybaylo, O.; Shen, C.; Buck, M.; Wöll, C.; Schlettwein, D. Redox mediation enabled by immobilised centres in the pores of a metal-organic framework grown by liquid phase epitaxy. Chem. Commun. 2012, 48, 663–665. [Google Scholar] [CrossRef]
- Kreno, L.E.; Leong, K.; Farha, O.K.; Allendorf, M.; van Duyne, R.P.; Hupp, J.T. Metal-organic framework materials as chemical sensors. Chem. Rev. 2012, 112, 1105–1124. [Google Scholar] [CrossRef] [PubMed]
- Allendorf, M.D.; Schwartzberg, A.; Stavila, V.; Talin, A.A. A roadmap to implementing metal-organic frameworks in electronic devices: Challenges and critical directions. Chem. Eur. J. 2011, 17, 11372–11388. [Google Scholar] [CrossRef] [PubMed]
- Hanke, M.; Arslan, H.K.; Bauer, S.; Zybaylo, O.; Christophis, C.; Gliemann, H.; Rosenhahn, A.; Wöll, C. The biocompatibility of metal-organic framework coatings: An investigation on the stability of SURMOFs with regard to water and selected cell culture media. Langmuir 2012, 28, 6877–6884. [Google Scholar] [CrossRef] [PubMed]
- Biemmi, E.; Scherb, C.; Bein, T. Oriented growth of the metal organic framework Cu-3(BTC)(2)(H2O)(3)center dot xH(2)O tunable with functionalized self-assembled monolayers. J. Am. Chem. Soc. 2007, 129, 8054–8055. [Google Scholar] [CrossRef] [PubMed]
- Zacher, D.; Liu, J.N.; Huber, K.; Fischer, R.A. Nanocrystals of [Cu-3(btc)(2)] (HKUST-1): A combined time-resolved light scattering and scanning electron microscopy study. Chem. Commun. 2009, 9, 1031–1033. [Google Scholar] [CrossRef]
- Shekhah, O.; Wang, H.; Kowarik, S.; Schreiber, F.; Paulus, M.; Tolan, M.; Sternemann, C.; Evers, F.; Zacher, D.; Fischer, R.A.; et al. Step-by-step route for the synthesis of metal-organic frameworks. J. Am. Chem. Soc. 2007, 129, 15118–15119. [Google Scholar] [CrossRef] [PubMed]
- Shekhah, O.; Wang, H.; Paradinas, M.; Ocal, C.; Schupbach, B.; Terfort, A.; Zacher, D.; Fischer, R.A.; Wöll, C. Controlling interpenetration in metal-organic frameworks by liquid-phase epitaxy. Nat. Mater. 2009, 8, 481–484. [Google Scholar] [CrossRef] [PubMed]
- Shekhah, O.; Wang, H.; Strunskus, T.; Cyganik, P.; Zacher, D.; Fischer, R.; Wöll, C. Layer-by-layer growth of oriented metal organic polymers on a functionalized organic surface. Langmuir 2007, 23, 7440–7442. [Google Scholar] [CrossRef] [PubMed]
- Shekhah, O.; Wang, H.; Zacher, D.; Fischer, R.A.; Wöll, C. Growth mechanism of metal-organic frameworks: Insights into the nucleation by employing a step-by-step route. Angew. Chem. Int. Ed. 2009, 48, 5038–5041. [Google Scholar] [CrossRef]
- Kind, M.; Wöll, C. Organic surfaces exposed by self-assembled organothiol monolayers: Preparation, characterization, and application. Prog. Surf. Sci. 2009, 84, 230–278. [Google Scholar] [CrossRef]
- Stavila, V.; Volponi, J.; Katzenmeyer, A.M.; Dixon, M.C.; Allendorf, M.D. Kinetics and mechanism of metal-organic framework thin film growth: Systematic investigation of HKUST-1 deposition on QCM electrodes. Chem. Sci. 2012, 3, 1531–1540. [Google Scholar] [CrossRef]
- Arnold, R.; Azzam, W.; Terfort, A.; Wöll, C. Preparation, modification, and crystallinity of aliphatic and aromatic carboxylic acid terminated self-assembled monolayers. Langmuir 2002, 18, 3980–3992. [Google Scholar] [CrossRef]
- Himmel, H.J.; Terfort, A.; Wöll, C. Fabrication of a carboxyl-terminated organic surface with self-assembly of functionalized terphenylthiols: The importance of hydrogen bond formation. J. Am. Chem. Soc. 1998, 120, 12069–12074. [Google Scholar] [CrossRef]
- Liu, J.; Ribas-Arino, J.; Käfer, D.; Winkler, S.; Schüpbach, B.; Zhang, W.; Bashir, A.; Shekhah, O.; Kind, M.; Terfort, A.; et al.; Karlsruhe Institute of Technology, Eggenstein-Leopoldshafen, Germany Unpublished work. 2012.
- Zacher, D.; Yusenko, K.; Betard, A.; Henke, S.; Molon, M.; Ladnorg, T.; Shekhah, O.; Schupbach, B.; Arcos, T.D.; Krasnopolski, M.; Meilikhov, M.; Winter, J.; Terfort, A.; Wöll, C.; Fischer, R.A. Liquid-phase epitaxy of multicomponent layer-based porous coordination polymer thin films of [M(L)(P)0.5] type: Importance of deposition sequence on the oriented growth. Chem. Eur. J. 2011, 17, 1448–1455. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Glenz, A.; Muhler, M.; Wöll, C. A new dual-purpose ultrahigh vacuum infrared spectroscopy apparatus optimized for grazing-incidence reflection as well as for transmission geometries. Rev. Sci. Instrum. 2009, 80, 113108. [Google Scholar] [CrossRef] [PubMed]
- Arslan, H.K.; Shekhah, O.; Wohlgemuth, J.; Franzreb, M.; Fischer, R.A.; Woll, C. High-throughput fabrication of uniform and homogenous MOF coatings. Adv. Funct. Mater. 2011, 21, 4228–4231. [Google Scholar] [CrossRef]
- Munuera, C.; Shekhah, O.; Wang, H.; Woll, C.; Ocal, C. The controlled growth of oriented metal-organic frameworks on functionalized surfaces as followed by scanning force microscopy. Phys. Chem. Chem. Phys. 2008, 10, 7257–7261. [Google Scholar] [CrossRef] [PubMed]
- Azzam, W.; Bashir, A.; Terfort, A.; Strunskus, T.; Wöll, C. Combined STM and FTIR characterization of terphenylalkanethiol monolayers on Au(111): Effect of alkyl chain length and deposition temperature. Langmuir 2006, 22, 3647–3655. [Google Scholar] [CrossRef] [PubMed]
- Poirier, G.E.; Tarlov, M.J. The C(4 × 2) superlattice of N-Alkanethiol monolayers self-assembled an Au(111). Langmuir 1994, 10, 2853–2856. [Google Scholar] [CrossRef]
- Liu, J.X.; Schupbach, B.; Bashir, A.; Shekhah, O.; Nefedov, A.; Kind, M.; Terfort, A.; Wöll, C. Structural characterization of self-assembled monolayers of pyridine-terminated thiolates on gold. Phys. Chem. Chem. Phys. 2010, 12, 4459–4472. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, R.G.; Dubois, L.H.; Allara, D.L. Fundamental-studies of microscopic wetting on organic-surfaces. 1. Formation and structural characterization of a self-consistent series of polyfunctional organic monolayers. J. Am. Chem. Soc. 1990, 112, 558–569. [Google Scholar] [CrossRef]
- Gliemann, H.; Wöll, C. Epitaxially grown metal-organic frameworks. Mater. Today 2012, 15, 110–116. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, J.; Shekhah, O.; Stammer, X.; Arslan, H.K.; Liu, B.; Schüpbach, B.; Terfort, A.; Wöll, C. Deposition of Metal-Organic Frameworks by Liquid-Phase Epitaxy: The Influence of Substrate Functional Group Density on Film Orientation. Materials 2012, 5, 1581-1592. https://doi.org/10.3390/ma5091581
Liu J, Shekhah O, Stammer X, Arslan HK, Liu B, Schüpbach B, Terfort A, Wöll C. Deposition of Metal-Organic Frameworks by Liquid-Phase Epitaxy: The Influence of Substrate Functional Group Density on Film Orientation. Materials. 2012; 5(9):1581-1592. https://doi.org/10.3390/ma5091581
Chicago/Turabian StyleLiu, Jinxuan, Osama Shekhah, Xia Stammer, Hasan K. Arslan, Bo Liu, Björn Schüpbach, Andreas Terfort, and Christof Wöll. 2012. "Deposition of Metal-Organic Frameworks by Liquid-Phase Epitaxy: The Influence of Substrate Functional Group Density on Film Orientation" Materials 5, no. 9: 1581-1592. https://doi.org/10.3390/ma5091581
APA StyleLiu, J., Shekhah, O., Stammer, X., Arslan, H. K., Liu, B., Schüpbach, B., Terfort, A., & Wöll, C. (2012). Deposition of Metal-Organic Frameworks by Liquid-Phase Epitaxy: The Influence of Substrate Functional Group Density on Film Orientation. Materials, 5(9), 1581-1592. https://doi.org/10.3390/ma5091581






