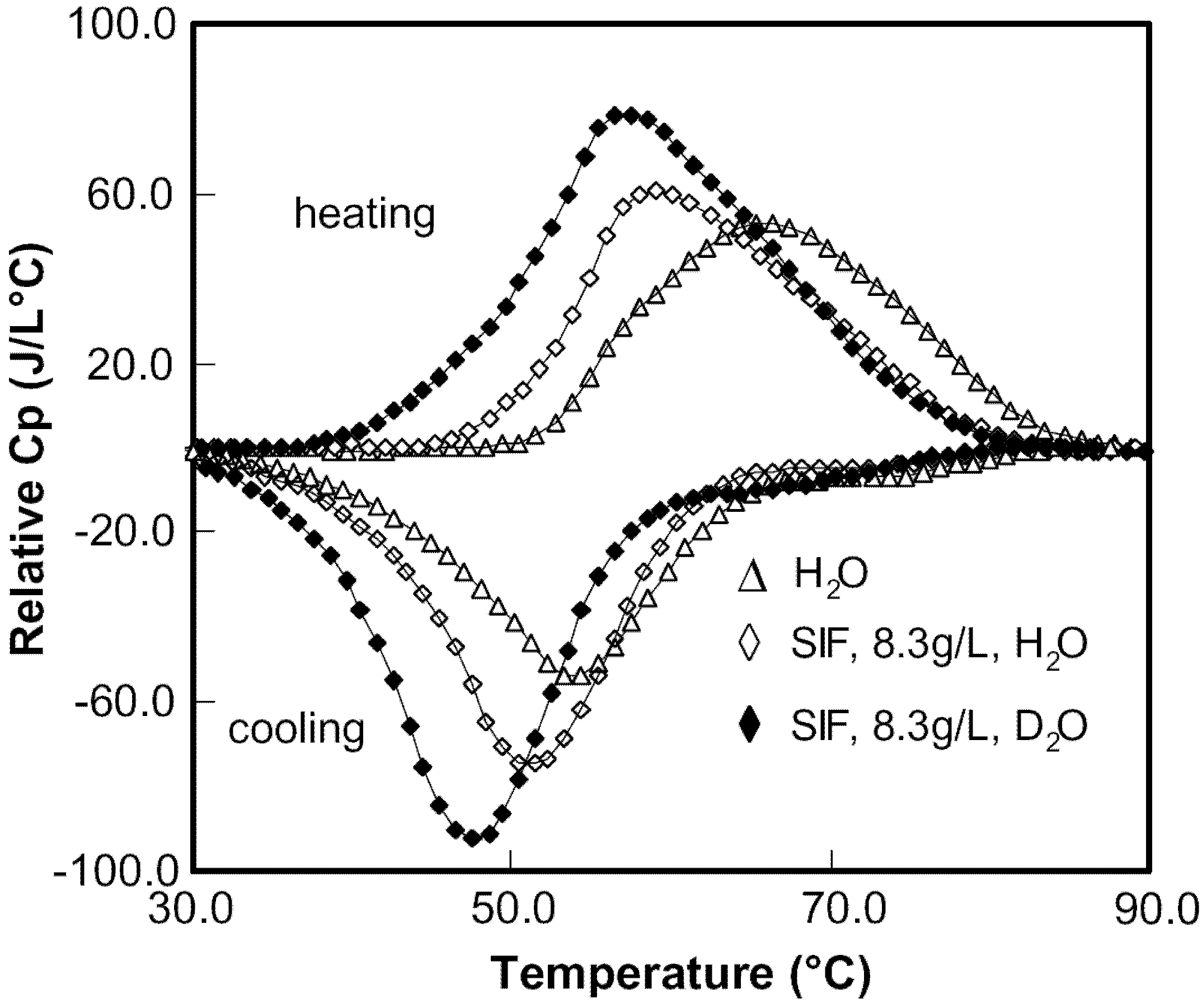
3.2. Investigations Using DSC
Although the effects of salt on some sol-gel systems, including MC, have been previously investigated [
16,
27,
28], few have reported about the effects of salt on the gelation of HPMC, even though HPMC has more applications in pharmaceutics and bio-fields.
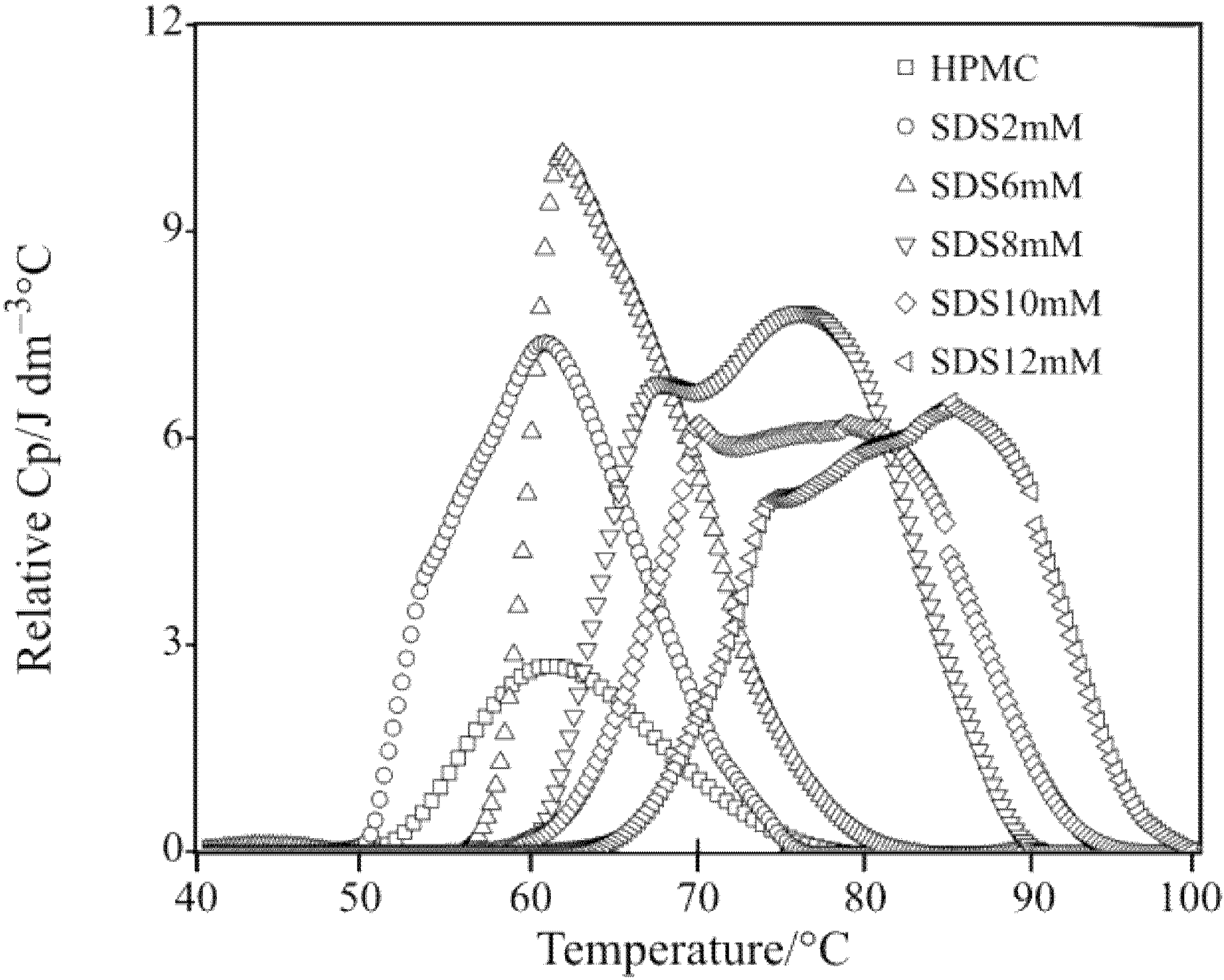
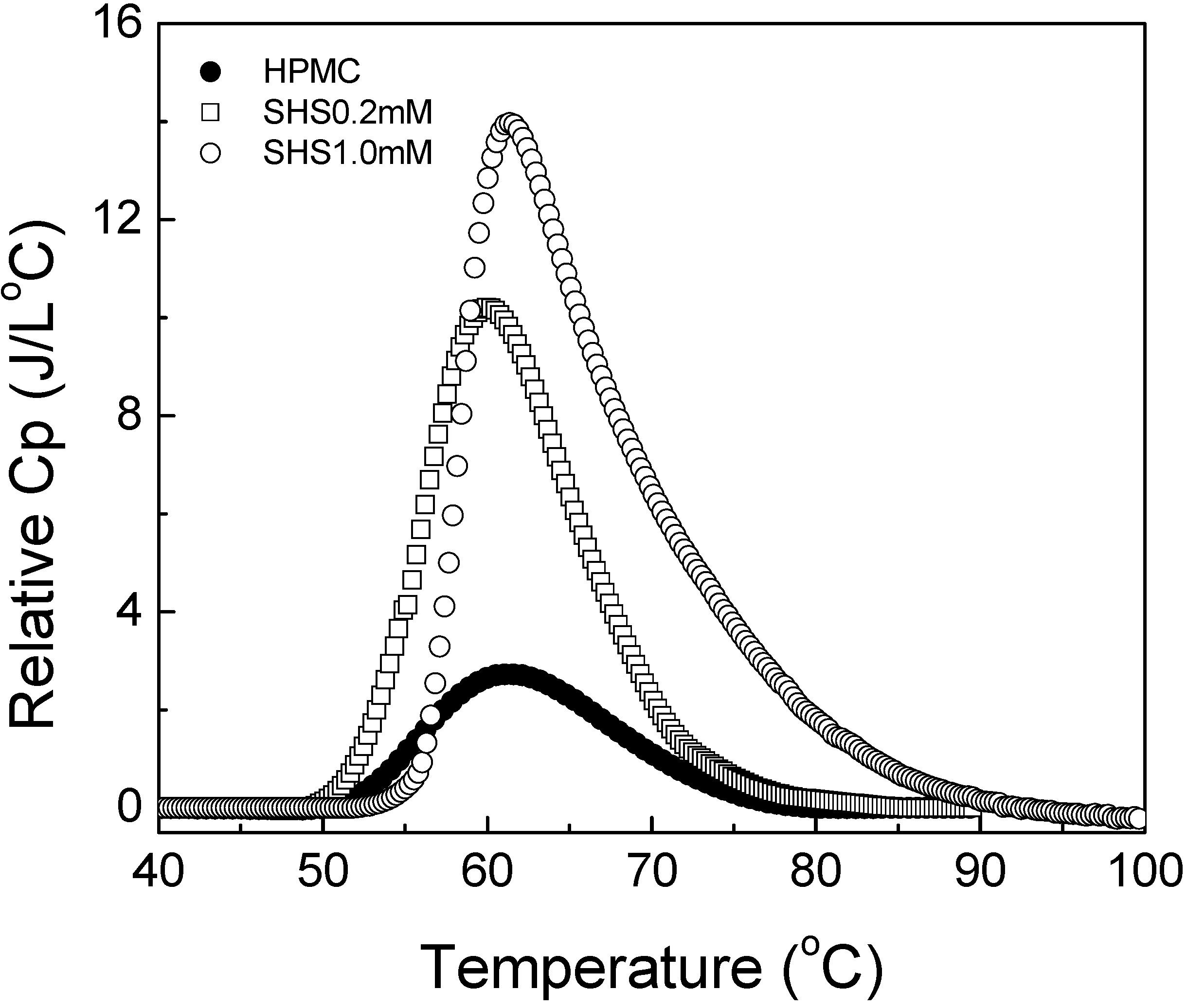
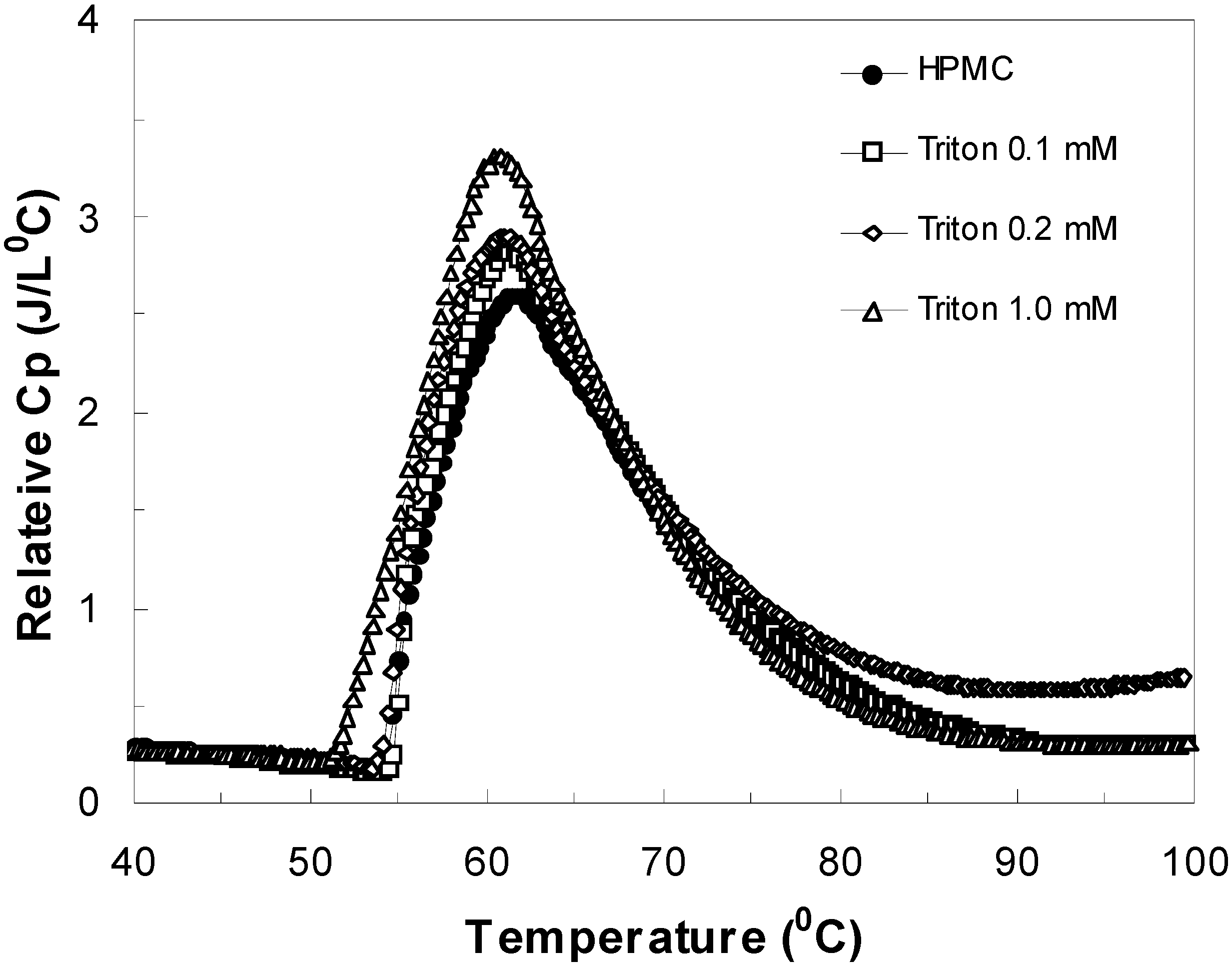
Figure 1.
Differential scanning calorimetry (DSC) scans showing Cp plots for HPMC solutions in H2O and D2O with various salts.
Figure 1.
Differential scanning calorimetry (DSC) scans showing Cp plots for HPMC solutions in H2O and D2O with various salts.
Mitchell
et al. [
29] showed that various salts that lower the cloud point of HPMC gels follow the order of the Hofmeister series. Since turbidity measurement method has many limitations; microcalorimetry, which is a more powerful technique, is used to study the salting mechanism for HPMC gels. As seen in
Figure 1, addition of NaCl shifted the DSC plot leftward whereas the presence of NaI in the HPMC solution caused the DSC curve to shift towards the right. Thus, NaCl led to the decrease in gelation temperature indicating salting-out phenomenon [
15] whereas NaI was the cause for the salting-in phenomenon that led to the increase in the gelation temperature. The plots in
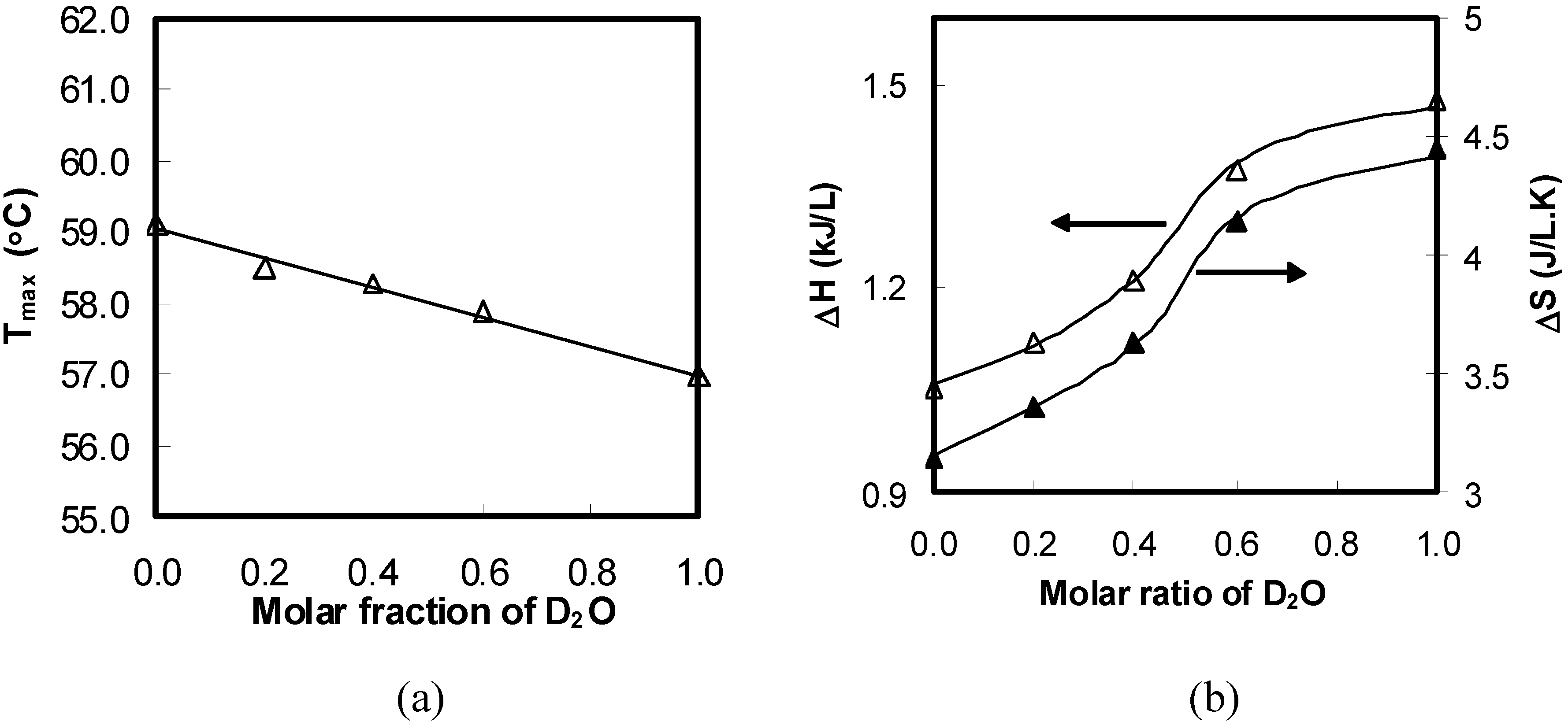
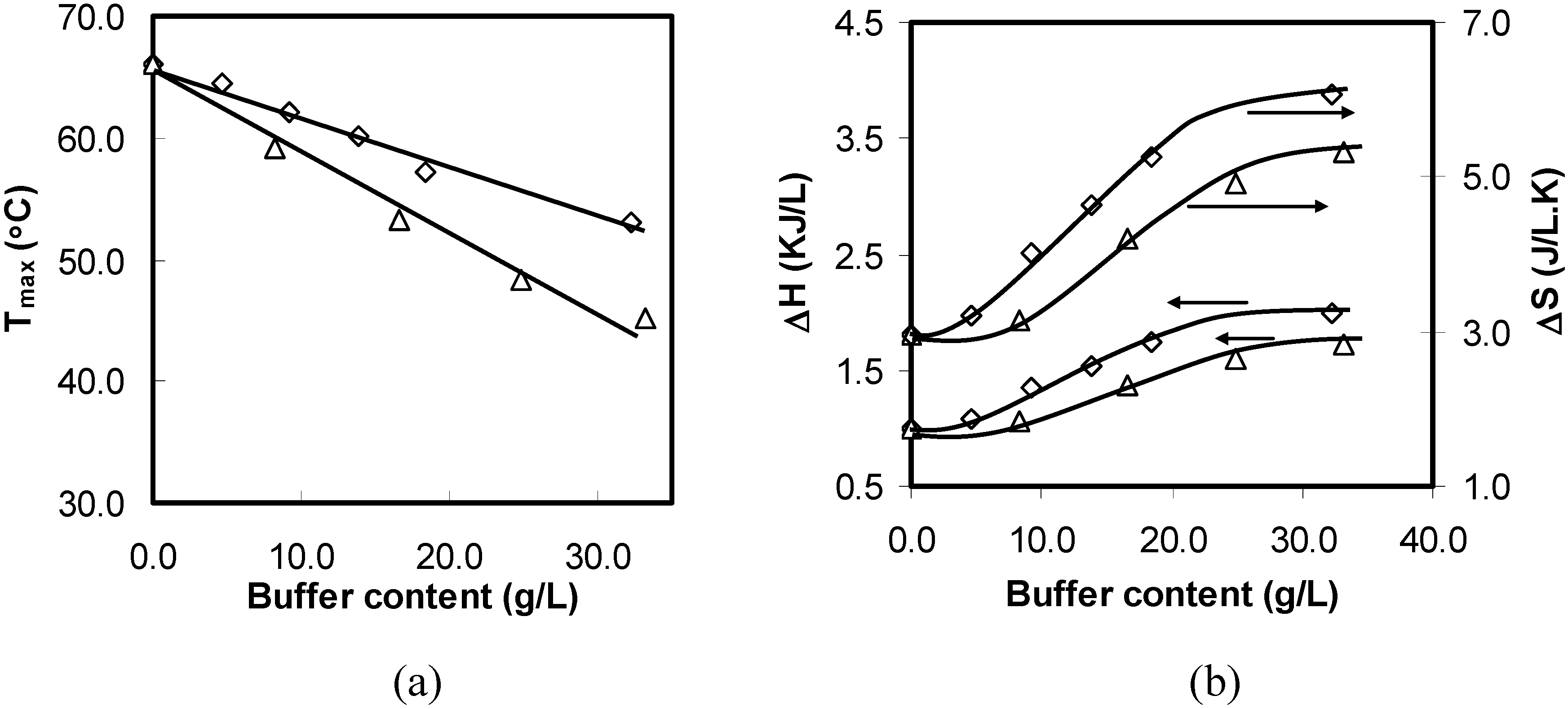
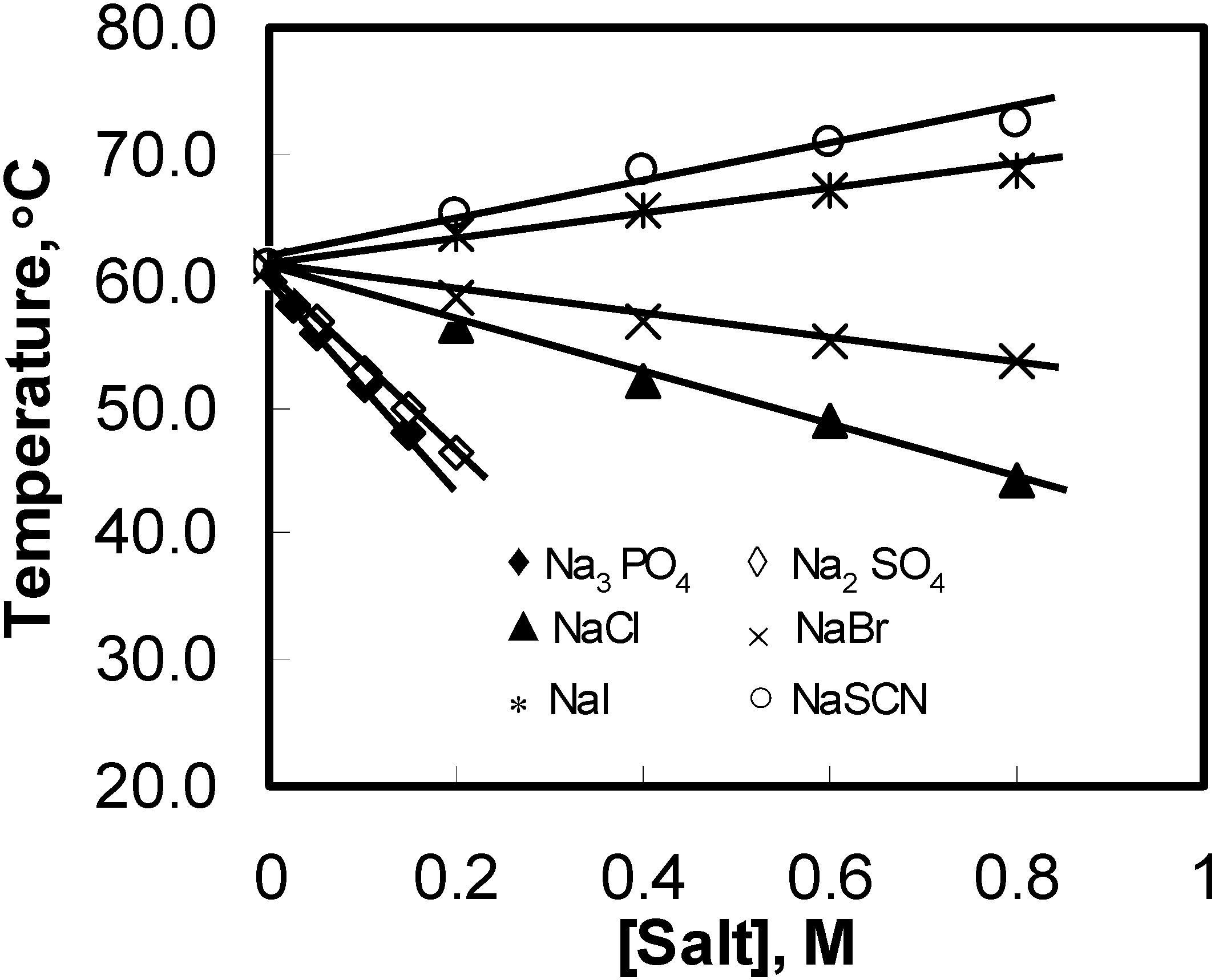
Figure 2 show a linear decrease in the peak temperature (
) with the concentration of salt in the solutions. This indicates that all ionic effects have a linear dependence on the salt concentration.
Figure 2.
Peak temperature (T) variations for aqueous solutions of 1 wt% HPMC as a function of the added salts concentration during the heating process.
Figure 2.
Peak temperature (T) variations for aqueous solutions of 1 wt% HPMC as a function of the added salts concentration during the heating process.
The magnitude of
lowered with the addition of kosmotropic anions and increased in the presence of chaotropes. Moreover, the ability of ions to change the values of
depended strongly on the anions and was characterized with the slope (
) of the fitted straight line (see
Figure 2). The more negative the
value was, the stronger the salting-out effect observed. PO
43− had the strongest salting-out effect amongst all of investigated anions, and
decreased sharply with a negative
of −86.0 °C/M. The slopes for the other three kosmotropic anions (SO
42–, Cl
–, and Br
–) were −72.6, −20.6, and −9.2 °C/M, respectively. The negative slope for the kosmotropic anions indicated that they promoted the sol–gel transition of the HPMC solutions. In contrast to kosmotropic anions, the slopes of chaotropic anions such as I− and SCN− were positive, and they were 9.4 and 14.3 °C/M, respectively, which indicated that they retarded the thermal gelation of the HPMC solutions. The results showed that the effects of anions on
of the HPMC solutions followed the sequence of the Hofmeister series.
As most studies have pointed out and demonstrated that the ions exert their salting-out/salting-in effects via their water structuring capability and not via the direct interactions between the ions and the polymer chains [
30,
31,
32]; a similar mechanism is used to explain the Hofmeister series in this study.
The effects of ions on water structure are attributed to the competitions between ion-water interactions and water-water interactions [
21]. The former interactions are dominated by charge density, and the latter interactions are dominated by hydrogen bonding. The ability of halide to lower
decreases with its size (
Table 2, regression coefficient = 1.0) [
33].
Table 2.
Linear correlation between
of the fitted curves and the entropy of hydration (ΔS
hydr), viscosity B coefficient of the anions, the anion radius. Note: ΔS
hydr, viscosity B coefficient, and anion radius are from reference [
30].
Table 2.
Linear correlation between
of the fitted curves and the entropy of hydration (ΔShydr), viscosity B coefficient of the anions, the anion radius. Note: ΔShydr, viscosity B coefficient, and anion radius are from reference [30].
| Ions | (°C/mol) | ΔShydr (J/K mol) | Viscosity B coefficient (L/mol) | Radius (Å) |
| PO43− | −86.0 | −421 | 0.495 | 2.38 |
| SO42− | −72.6 | −200 | 0.206 | 2.30 |
| Cl− | −20.6 | −75 | −0.005 | 1.81 |
| Br− | −9.2 | −59 | −0.033 | 1.96 |
| I− | 9.4 | −36 | −0.073 | 2.20 |
| SCN− | 14.3 | – | −0.103 | 2.13 |
Because the chloride ion has the smallest radius and largest charge density among the halides investigated in this study, it has the strongest ability to compete for water molecules and form hydrogen bonds with water molecules, which minimizes or weakens water-polymer hydrogen bonding. On the other hand, the low charge density of ions such as I– provides the weakest competition for water molecules, which thus leads to salting-in effects and increased the solubility of the polymer.
Hribar
et al. [
22] argued that the effect of salt on the degree of water structuring is determined mainly by the entropies of ion salvation and viscosity changes. Anions with more negative entropy values are considered more effective in arranging water molecules in an orderly manner. A plot of the
values versus the corresponding ΔS
hydr of the various salts anions yielded a straight line with a regression coefficient of 0.84, indicating that these two parameters were not well correlated linearly. On the other hand, viscosity B coefficient was used to quantify the degree of water structuring because it was related to the viscosity of an aqueous salt solution. Anions with a large viscosity B coefficient value exhibited strong water structure making capabilities. The regression coefficient for the
verses viscosity B coefficient of the anions plots was found to be 0.91.
Even though the linear correlation between slopes
(for
) and ΔS
hydr and viscosity B coefficient was not well-defined, most anions followed a sequence. These seem to demonstrate that different mechanisms were involved in thermogelation of HPMC in presence of salts [
34].
An alternative explanation proposed by Hribar
et al. [
22] is that water structure is determined by the balance between electrostatics and hydrogen bonding. Multivalent anions and the smallest anions such as F
− and Cl
− cause a strong electrostatic orientation of water molecules with respect to the anion, which makes water structure more ordered.
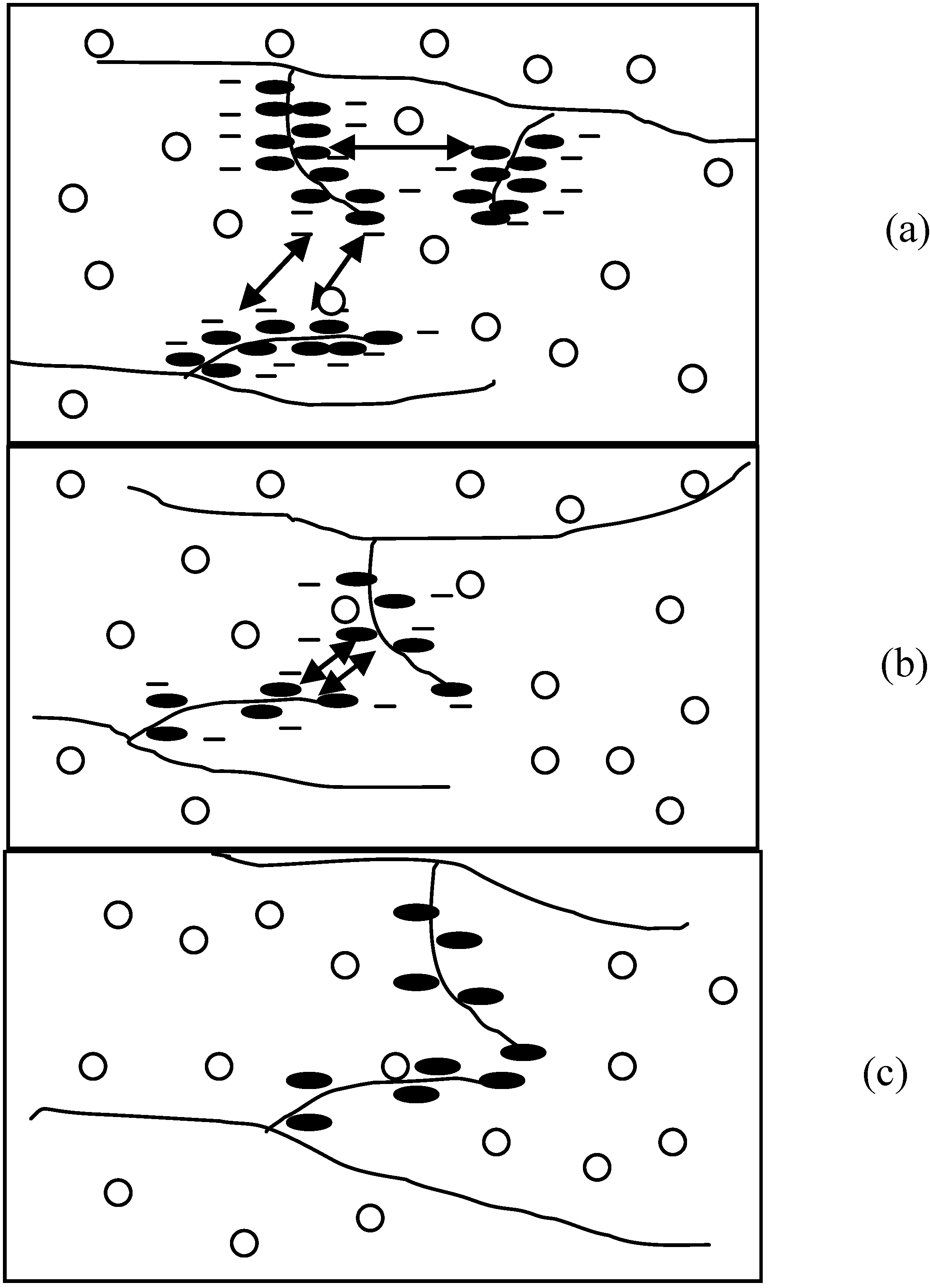
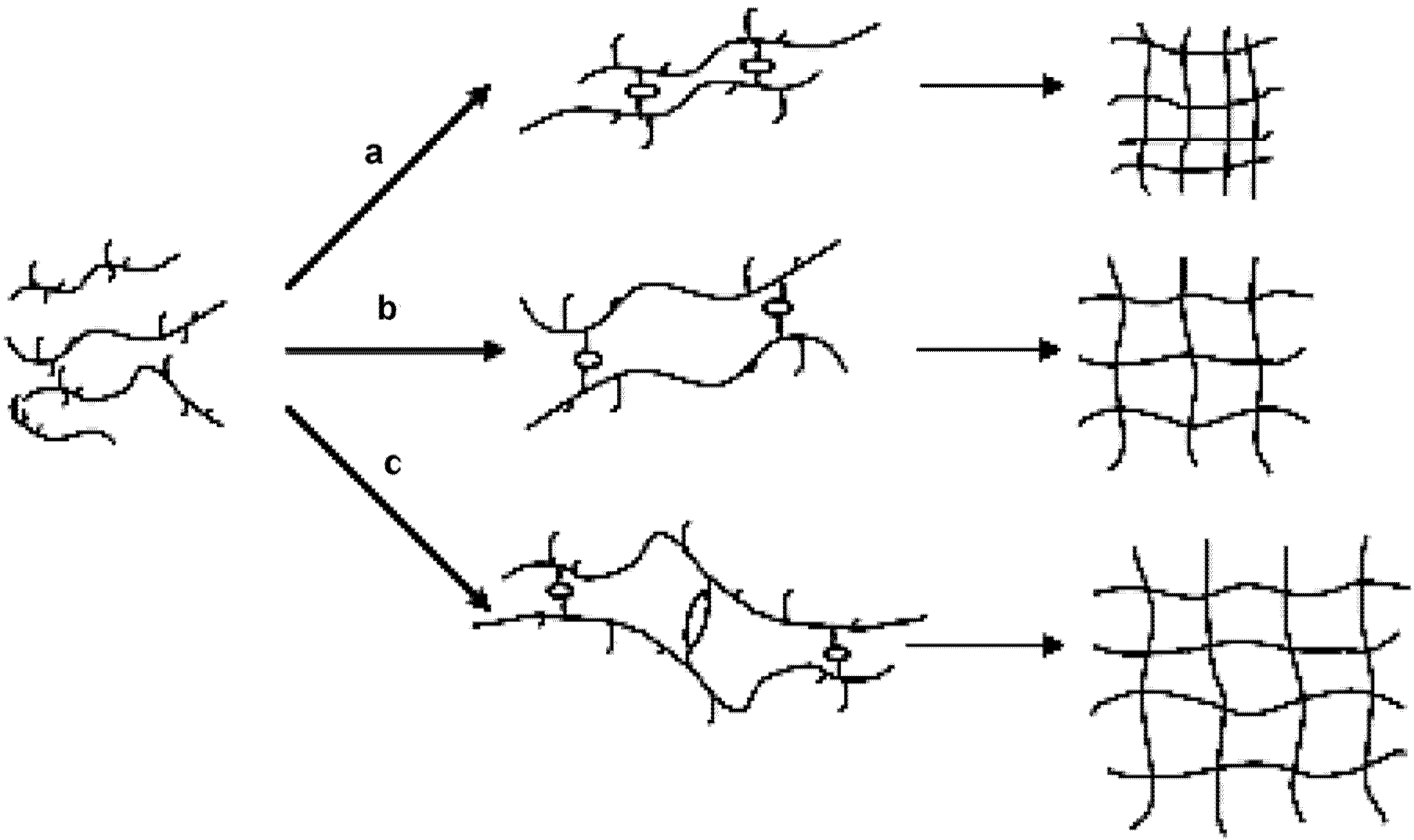
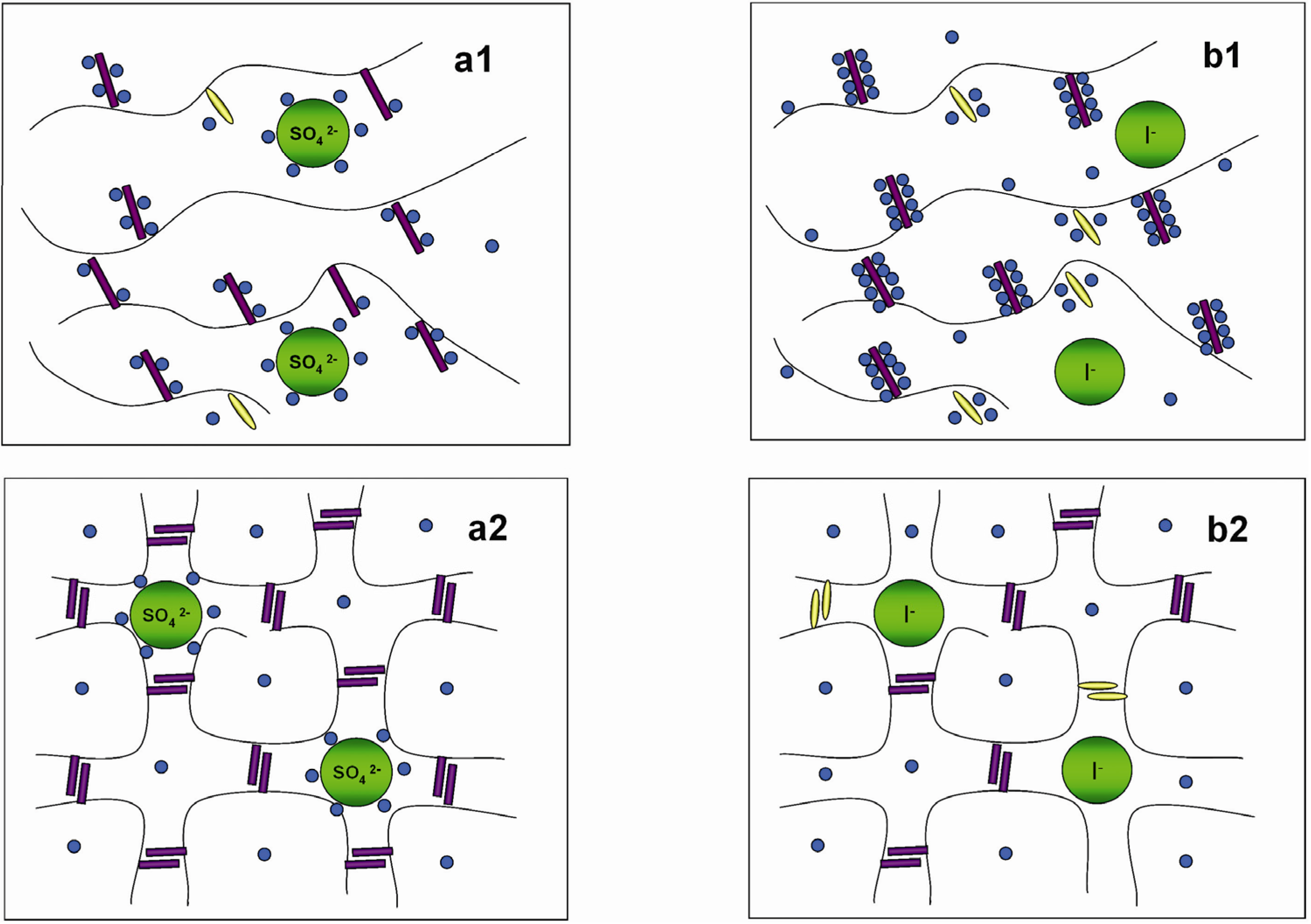
The figure below illustrated the physical structures of HPMC hydrogels in respectively 0.2 M Na
2SO
4 and 0.2 M NaI solutions. The figure legends are as follows: HPMC main chain (
![Materials 04 01861 i001]()
); water molecules (
![Materials 04 01861 i002]()
); hydrophobic substitution (
![Materials 04 01861 i003]()
); hydroxyl group (
![Materials 04 01861 i004]()
); ions (
![Materials 04 01861 i005]()
); hydrophobic interaction (
![Materials 04 01861 i006]()
); interchain hydrogen bonding (
![Materials 04 01861 i007]()
).
Figure 3.
Physical structures of HPMC hydrogels in (a) 0.2 M Na2SO4 in H2O; (b) 0.2 M NaI in H2O (a1, b1 at lower temperatures and a2, b2 at higher temperatures).
Figure 3.
Physical structures of HPMC hydrogels in (a) 0.2 M Na2SO4 in H2O; (b) 0.2 M NaI in H2O (a1, b1 at lower temperatures and a2, b2 at higher temperatures).
As a result of more ordered water structure, a less number of water molecules are freely available to solvate the polymer chains. This facilitates and accelerates the hydrophobic association of methyl substitutions causing reduction in
of the HPMC solutions (refer to
Figure 3(a1,a2)). In contrast, large monovalent anions such as I
− and SCN
− have low charge densities, and water structures around them are less organized. Having no restraint on their movement, these water molecules tend to increase hydrogen bonding between them and the polymer, which results in an increase in
of the HPMC solutions as shown schematically in
Figure 3(b1,b2).
On the basis of these analyses, it is clear that both mechanisms could be applied to interpret the results in this study. The trivalent anion (PO
43−) was more effective in the salting-out effects than the divalent anion (SO
42−), followed by the monovalent anions such as Cl
−. However, the trends did not relate well to the radius of the ions (refer
Table 2). This is a strong indication that the valency was a dominating factor in comparison to the anionic radius.
Multivalent anions such as PO
43− and SO
42− had a strong salting-out effect as compared to monovalent anions. It was interesting that the pattern of thermograms of HPMC in the presence of SO
42− was much broader than that of monovalent anions (
Figure 1). The corresponding
is illustrated in
Figure 4.
Figure 4.
variations for aqueous solutions of 1 wt% HPMC as a function of the added salts concentration during the heating process.
Figure 4.
variations for aqueous solutions of 1 wt% HPMC as a function of the added salts concentration during the heating process.
for all monovalent anions was almost constant. It rose sharply with increasing concentration of a salt with multivalent anions. The results suggest that the effect of monovalent anions was more cooperative, whereas that of multivalent anions was less cooperative. Multivalent anions have strong ability to compete for water molecules in a solution. It is, however, unlikely that all such ions could have competed for water molecules at the water-HPMC interfaces because of their large size and tetrahedral coordination. Additionally, the ring structure of HPMC would have made it difficult for the multivalent anions to approach the water cages. Therefore, it may be construed that the water cages were weakened to a different extent by multivalent anions than they were by the monovalent anions. In other words, the strength of the water cages had a larger polydispersity in the presence of multivalent anions.
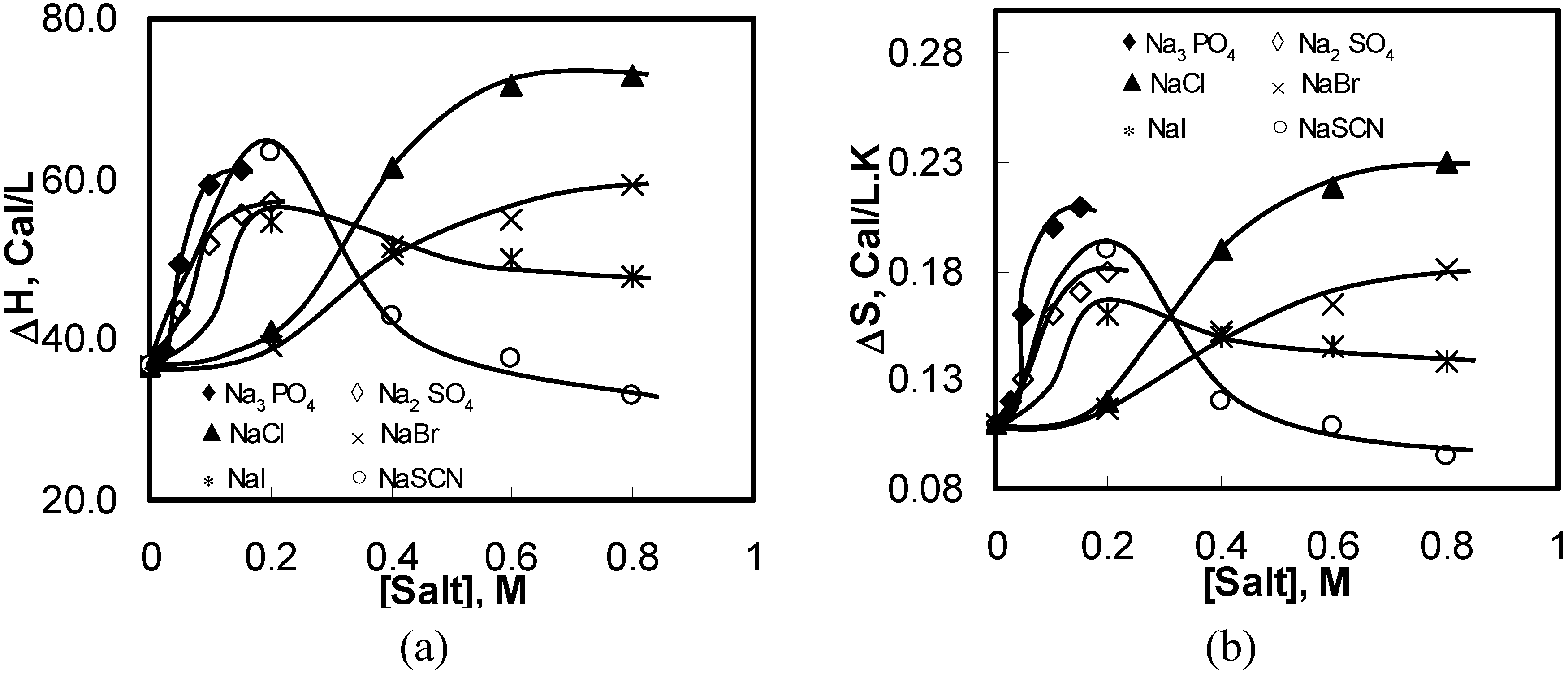
Figure 5.
Thermodynamic properties of 1 wt% HPMC aqueous solutions of as a function of the concentration of the added salts during the heating process: (a) ΔH; (b) ΔS.
Figure 5.
Thermodynamic properties of 1 wt% HPMC aqueous solutions of as a function of the concentration of the added salts during the heating process: (a) ΔH; (b) ΔS.
Figure 5 shows variations in the endothermic ΔH and ΔS values for the aqueous solutions of HPMC with various salts and salt concentrations. The endothermic ΔH and ΔS increased with the increasing salting-out salt concentration. All salting-out salts showed similar trends as NaCl shows. The changes in the ΔH and ΔS values for HPMC in salting-out salt solutions could be explained using the same mechanism as for the NaCl. On the other hand, the endothermic values of ΔH and ΔS for salting-in salts such as NaI and NaSCN showed a different pattern than salting-out salts. Both ΔH and ΔS increased initially with increasing salt concentration until the salt concentration reached 0.2 M. Subsequently, the quantities decreased with further increase in the salt concentration.
As stated earlier, salting-in salts are the demolishers of the oriented structure of water molecules, which enhances the intermolecular hydrogen bonding and allows denser water cages developing around the side chains of the HPMC molecules (see
Figure 3(b1,b2)). As a result, higher amount of energy is required to break these water cages before any hydrophobic association between the HPMC molecules leading to the gel formation. With further increase in the salt concentration, the water cages strengthen further; some are too strong to be broken even at high temperatures. This eventually reduced the energy requirement for further hydrophobic association and gel formation causing a reduction in ΔH and ΔS values.
3.3. Rheological Behavior
Viscoelastic characteristics of HPMC gels were studied using micro-DSC in terms of G′ and G″. The changes in G′ of HPMC upon heating as the effects of different salts additives are illustrated in
Figure 6. Typical samples of 0.2 M Na
2SO
4, 0.8 M NaCl, and 0.8 M NaI are chosen for comparison.
Figure 6.
G′ as a function of temperature for the aqueous solution of 1 wt% HPMC with various salt additives measured during heating process (frequency = 1 rad/s, heating rate = 1 °C/min).
Figure 6.
G′ as a function of temperature for the aqueous solution of 1 wt% HPMC with various salt additives measured during heating process (frequency = 1 rad/s, heating rate = 1 °C/min).
The general pattern exhibited in G′ values for the samples in the presence of Na2SO4 and NaI were found to be similar to that of NaCl. The curves shifted towards lower temperature in the presence of the salting-out salt NaCl. This tendency became more pronounced in the presence of Na2SO4, a multivalent salting-out salt. In contrast to salting-out salts, the G′ curve for NaI shifted towards higher temperature, indicating salting-in effect. The final values of G′ were salt-dependent.
Table 3.
G′ and G″ for HPMC aqueous solutions with various salts and salt concentrations measured at 70 °C, 1 rad/s frequency and 5 wt% strain.
Table 3.
G′ and G″ for HPMC aqueous solutions with various salts and salt concentrations measured at 70 °C, 1 rad/s frequency and 5 wt% strain.
| Aqueous solution | 0.8M NaI | 0.2M NaCl | 0.4M NaCl | 0.6M NaCl | 0.8M NaCl | 0.2M Na2SO4 |
| G′ (Pa) | 16.33 | 11.25 | 19.24 | 31.48 | 40.88 | 44.60 | 65.38 |
| G″ (Pa) | 2.91 | 2.82 | 4.80 | 3.08 | 3.57 | 11.60 | 18.74 |
As seen in
Table 3, G′ increased with the addition of salting-out salts, whereas it decreased in the presence of salting-in salts. This means that the gel was strengthened in the presence of salting-out salts and weakened when salting-in salts were added in. A similar trend was reported by Cho
et al. [
35] in their study of the effects of salts on the viscosity of polyorganophosphazenes. As discussed earlier, the thermally induced gelation of HPMC solutions mainly involves hydrophobic association, which leads to a three-dimensional network. Therefore, gel strength is governed basically by the hydrophobic associations.
In the presence of salting-out salts, the number of physical junctions formed by the hydrophobic association and the strength of association went up, resulting in the increased gel strength [
35]. In contrast, NaI showed a salting-in effect and enhanced the overall solubility of the HPMC chains in water, thereby causing a decrease in the gel strength. G′ values increased with the increasing NaCl concentration. Moreover, the trend correlated well with that of the ΔH. This demonstrates further that the gel strength is affected by the hydrophobic associations.

 ); water molecules (
); water molecules (  ); hydrophobic substitution (
); hydrophobic substitution (  ); hydroxyl group (
); hydroxyl group (  ); ions (
); ions (  ); hydrophobic interaction (
); hydrophobic interaction (  ); interchain hydrogen bonding (
); interchain hydrogen bonding (  ).
).


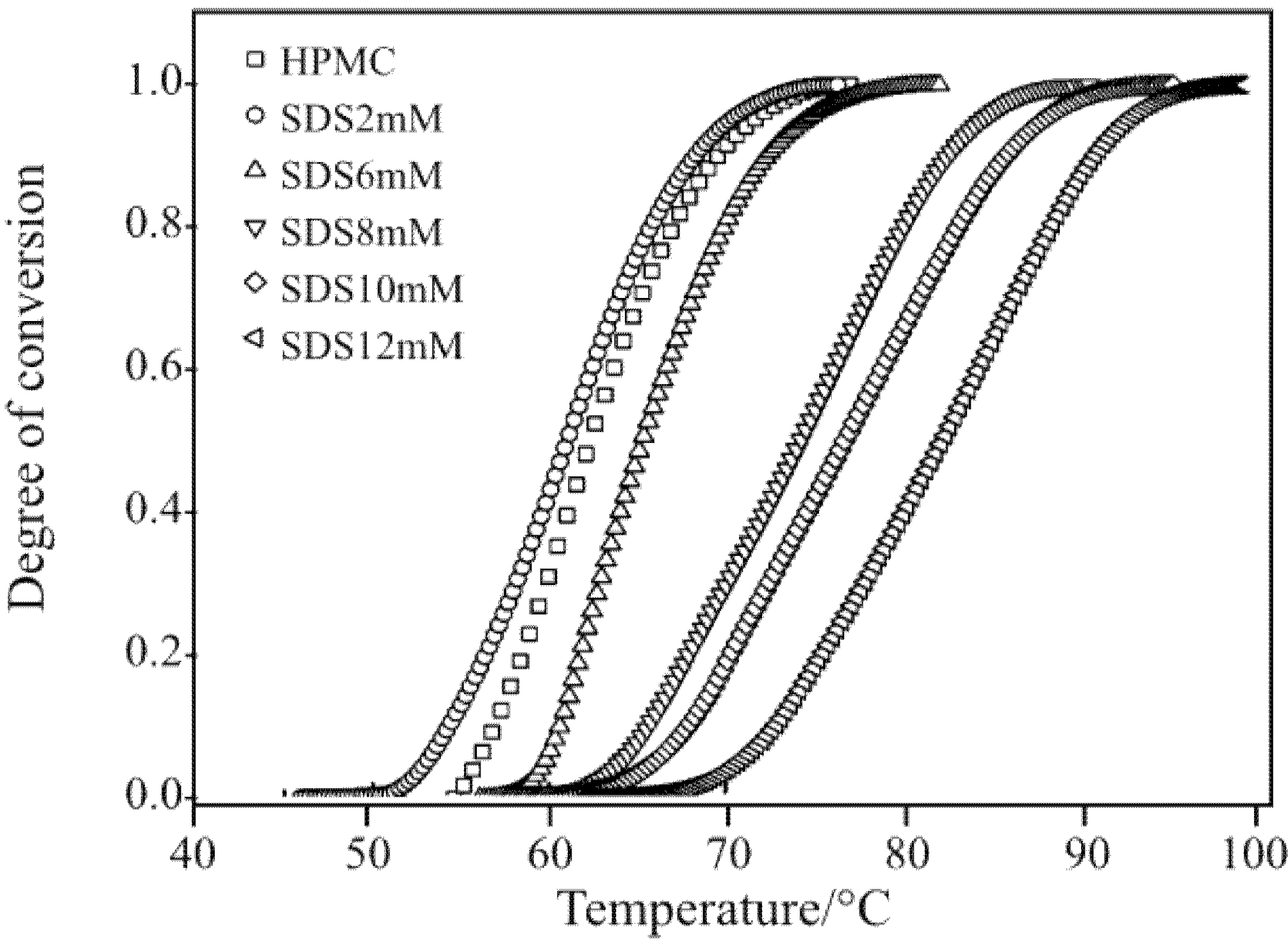
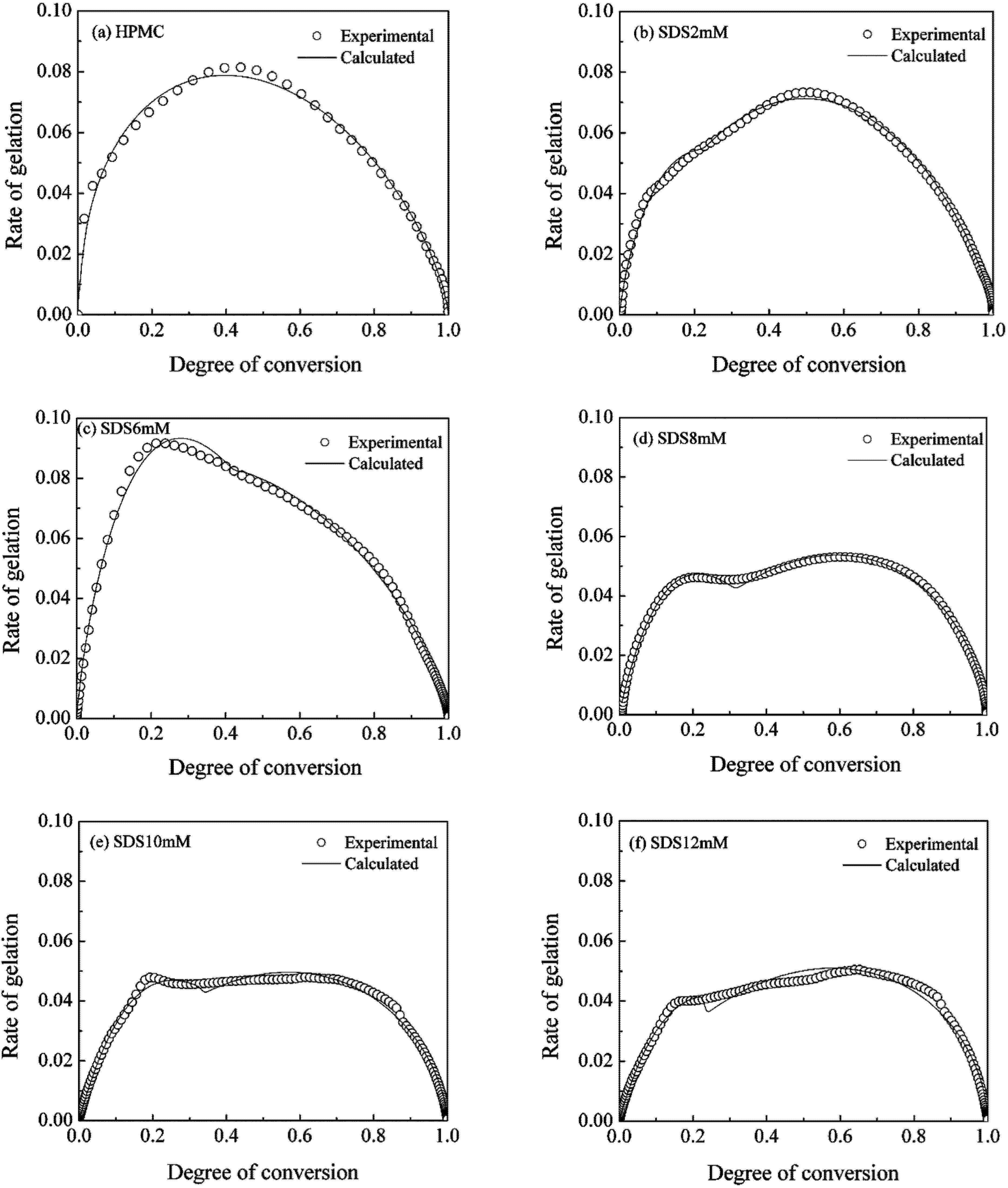

 representing the polar head of surfactant molecule and
representing the polar head of surfactant molecule and  representing the electro-static repulsory, Figure 14 depicts the molecular interaction between HPMC and the surfactants.
representing the electro-static repulsory, Figure 14 depicts the molecular interaction between HPMC and the surfactants.