2.1. Bi2NiMnO6 High-Pressure Synthesized Bulk
Bi
2NiMnO
6 cannot be synthesized under an ambient-pressure condition. However, we found that the compound is obtained by means of high-pressure synthesis. Although small amounts of impurities were included, nearly single phase sample (>97%) was obtained by the synthesis at 6 GPa and 800 °C [
19].
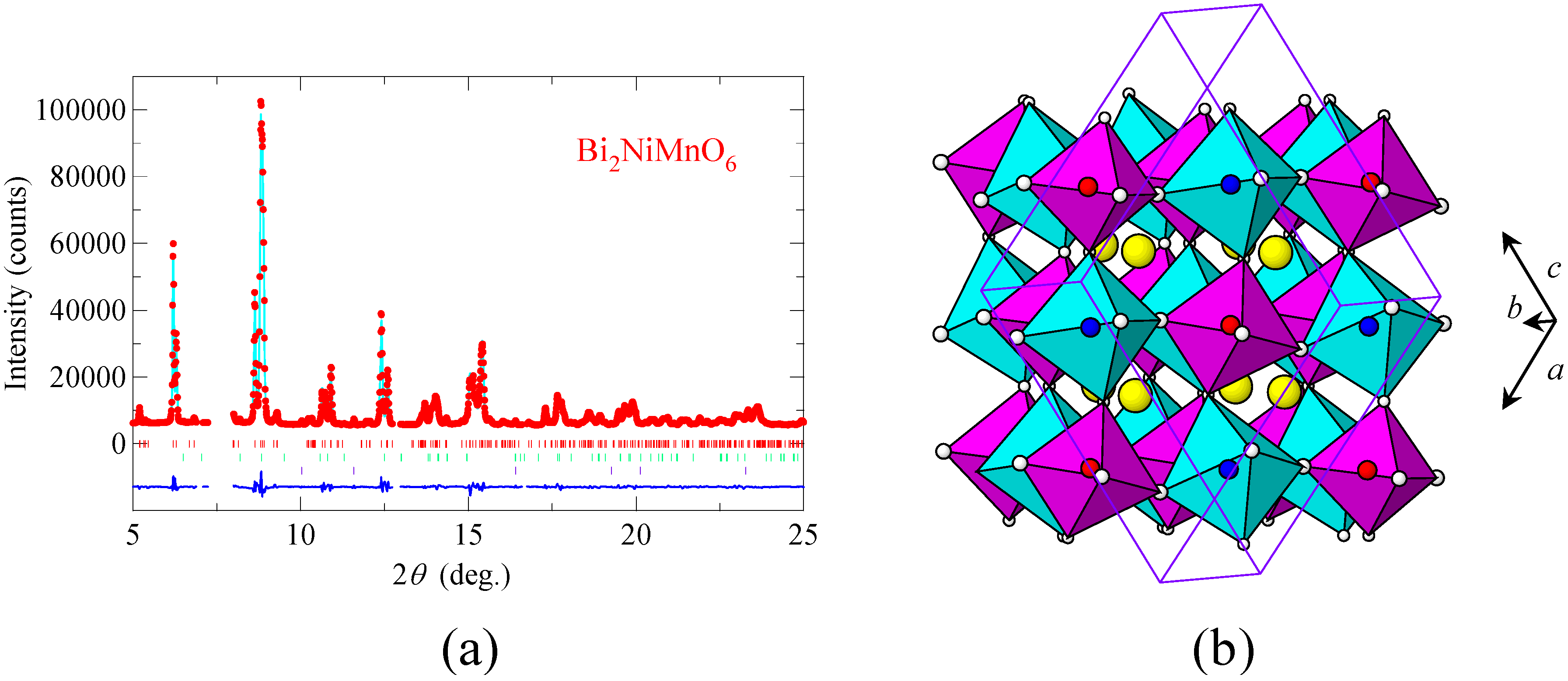
Figure 2(a) shows the synchrotron X-ray powder diffraction pattern taken at room temperature and the result of the structure refinement with the Rietveld method. The diffraction pattern shows that the material is crystallized with the Bi
2NiMnO
6 double-perovskite structure, with the monoclinic (space group
C2) unit cell of
a = 9.4646(4) Å,
b = 5.4230(2) Å,
c = 9.5431(4) Å, and
β = 107.823(2)°. At the initial stage of the refinement, Ni
2+ and Mn
4+ were randomly located at the three crystallographic sites (M(1), M(2), and M(3), with multiplicities of 2, 4, and 2, respectively) in the monoclinic structure. It was found that the M-O bond lengths were considerably shorter for the M(2) site than M(1) and M(3), so the small Mn
4+ ion was assigned to the M(2) site, and large Ni
2+ ions were assigned to M(1) and M(3) at the final stage. The refined structural parameters are summarized in
Table 1. The results of the structural analysis revealed that Bi
3+ is located at the A site and that Ni
2+ and Mn
4+ ions are ordered in a rock-salt configuration at the B site as shown in
Figure 2(b). Bond valence sums [
23] calculated from the refined structural parameters were 2.14 and 2.17 for the Ni sites, and 3.62 for the Mn site, which also support the structure model. The NiO
6 and MnO
6 octahedra of Bi
2NiMnO
6 are rather isotropic, reflecting the absence of Jahn-Teller distortions, which is also consistent with the Ni
2+ (
t2g6eg2) and Mn
4+ (
t2g3) oxidation states of the transition-metal ions in the compound.
Figure 2.
(a) Synchrotron X-ray powder diffraction pattern of Bi2NiMnO6 and the Rietveld refinement profile. The observed (red dot), calculated (light-blue line), and difference (bottom blue line) patterns are shown. Bragg reflections are indicated by tick marks. Diffractions from impurities (0.65 wt% Bi2(CO3)O2 and 1.92 wt% NiO) are also included in the refinement; (b) Crystal structure of Bi2NiMnO6. The blue octahedra correspond to NiO6 and the red octahedra correspond to MnO6. Bi ions are shown in yellow spheres. The monoclinic unit cell is superimposed.
Figure 2.
(a) Synchrotron X-ray powder diffraction pattern of Bi2NiMnO6 and the Rietveld refinement profile. The observed (red dot), calculated (light-blue line), and difference (bottom blue line) patterns are shown. Bragg reflections are indicated by tick marks. Diffractions from impurities (0.65 wt% Bi2(CO3)O2 and 1.92 wt% NiO) are also included in the refinement; (b) Crystal structure of Bi2NiMnO6. The blue octahedra correspond to NiO6 and the red octahedra correspond to MnO6. Bi ions are shown in yellow spheres. The monoclinic unit cell is superimposed.
Table 1.
Refined structure parameters of Bi2NiMnO6 at room temperature. Space group C2, a = 9.4646(4) Å, b = 5.4230(2) Å, c = 9.5431(4) Å, and β = 107.823(2) °. Rwp = 4.79% and RI = 0.64%. The same thermal parameter values (B) were given for Bi(1) and Bi(2), and also for Ni and Mn, respectively. The B value of O was fixed during the refinement.
Table 1.
Refined structure parameters of Bi2NiMnO6 at room temperature. Space group C2, a = 9.4646(4) Å, b = 5.4230(2) Å, c = 9.5431(4) Å, and β = 107.823(2) °. Rwp = 4.79% and RI = 0.64%. The same thermal parameter values (B) were given for Bi(1) and Bi(2), and also for Ni and Mn, respectively. The B value of O was fixed during the refinement.
| atom | site | x | y | z | B (Å2) |
|---|
| Bi(1) | 4c | 0.133(1) | −0.023(12) | 0.378(1) | 0.672(5) |
| Bi(2) | 4c | 0.369(1) | 0.035(12) | 0.123(1) | 0.672 (=Bi(1)) |
| Ni(1) | 2a | 0 | 0 | 0 | 0.40(7) |
| Ni(2) | 2b | 0.5 | 0.015(2) | 0.5 | 0.40 (=Ni(1)) |
| Mn | 4c | 0.243(3) | 0.013(13) | 0.749(3) | 0.40 (=Ni(1)) |
| O(1) | 4c | 0.111(5) | −0.061(15) | 0.849(6) | 0.8 |
| O(2) | 4c | 0.420(4) | 0.042(14) | 0.680(5) | 0.8 |
| O(3) | 4c | 0.146(9) | 0.276(18) | 0.636(9) | 0.8 |
| O(4) | 4c | 0.333(4) | 0.242(14) | 0.413(5) | 0.8 |
| O(5) | 4c | 0.377(5) | 0.204(12) | 0.899(5) | 0.8 |
| O(6) | 4c | 0.162(8) | 0.216(17) | 0.126(9) | 0.8 |
The noncentrosymmetric
C2 space group of the compound allows a spontaneous polarization along the
b axis, and a calculation based on an ionic point charge model with the obtained structural parameters gives a polarization of 20 μC/cm
2. The Bi
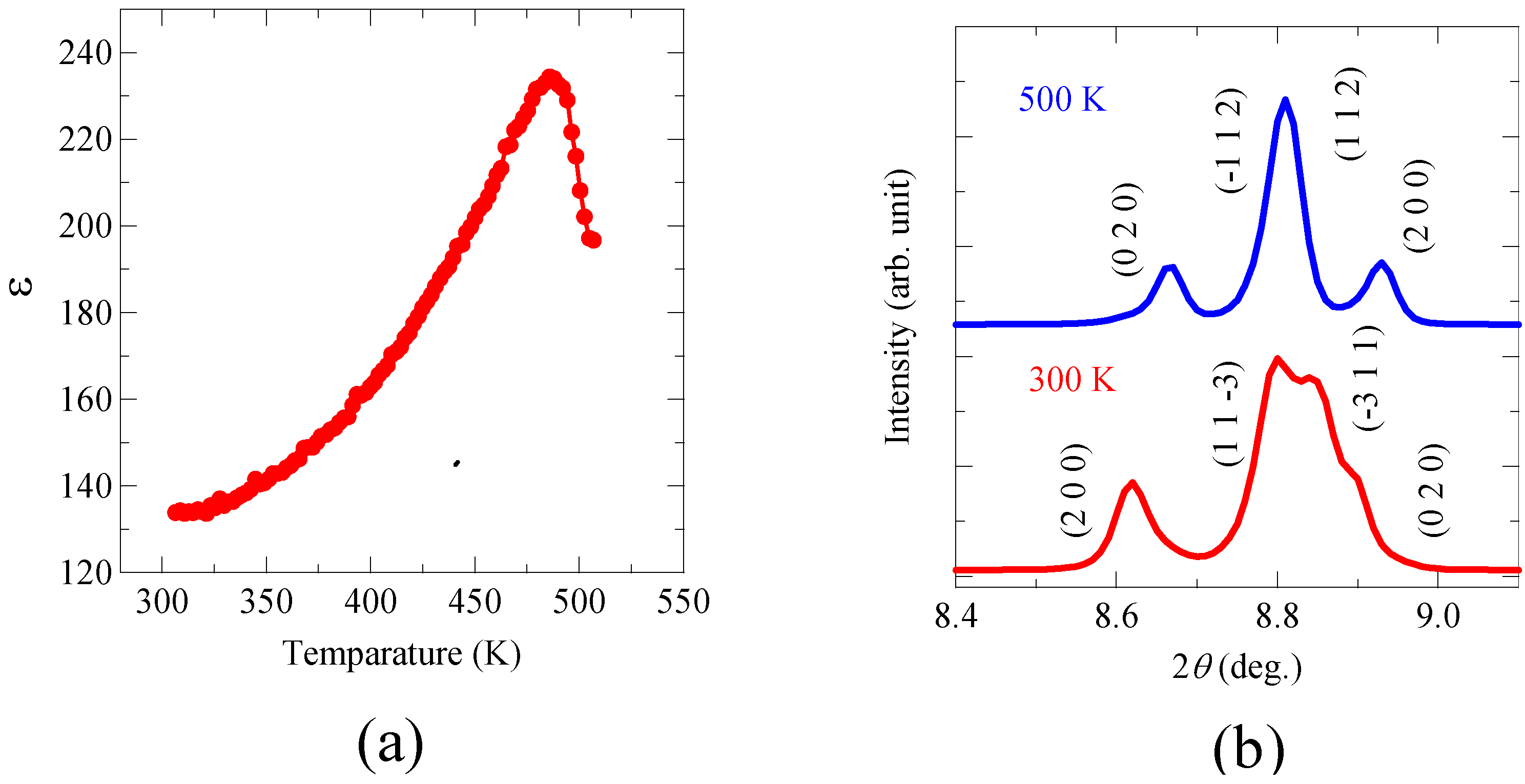
3+ ion at the A site in the perovskite structure should give rise to significant anisotropic structural distortion in the material. The temperature dependence of the dielectric constant, ε, shown in
Figure 3(a) also exhibits a typical ferroelectric behavior with a ferroelectric Curie temperature of about 485 K. Correspondingly, the crystal structure changes from the room-temperature noncentrosymmetric phase to a high-temperature centrosymmetric phase with a monoclinic structure (space group,
P2
1/
n; a = 5.4041(2) Å,
b = 5.5669(1) Å,
c = 7.7338(2) Å, and
β = 90.184(2) °) above the Curie temperature as shown in
Figure 3(b). Thus, the structural and dielectric measurement results confirm that Bi
2NiMnO
6 is ferroelectric.
Figure 3.
(a) Temperature dependence of dielectric constant of Bi2NiMnO6 measured at 10 kHz. A peak at 485 K indicates the ferroelectric Curie temperature; (b) Powder X-ray diffraction patterns taken at 300 and 500 K. The indices of the diffraction pattern at 300 K are for the noncentrosymmetric C2 structure, while those at 500 K for the centrosymmetric monoclinic P21/n structure. The results clearly show the ferroelectric transition at 485 K of Bi2NiMnO6.
Figure 3.
(a) Temperature dependence of dielectric constant of Bi2NiMnO6 measured at 10 kHz. A peak at 485 K indicates the ferroelectric Curie temperature; (b) Powder X-ray diffraction patterns taken at 300 and 500 K. The indices of the diffraction pattern at 300 K are for the noncentrosymmetric C2 structure, while those at 500 K for the centrosymmetric monoclinic P21/n structure. The results clearly show the ferroelectric transition at 485 K of Bi2NiMnO6.
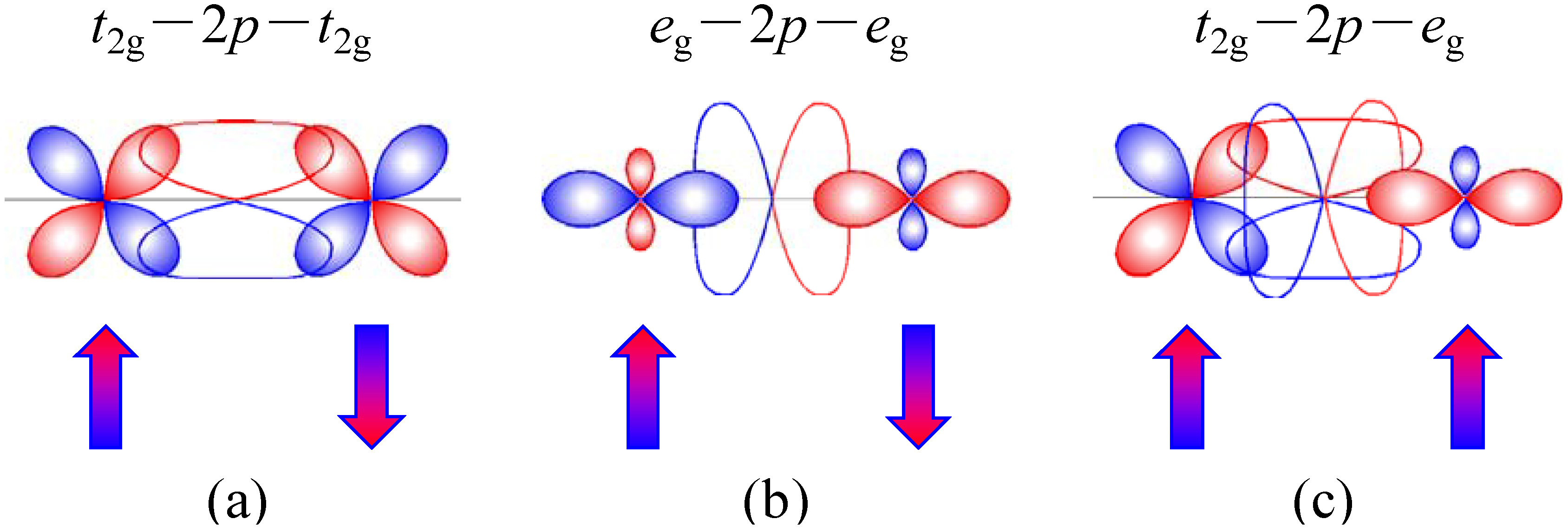
The rock-salt configuration of the Ni
2+ and Mn
4+ ions in the double-perovskite structure gives the magnetic exchange path of Ni
2+-O-Mn
4+. Since a Ni
2+ ion has the
t2g6eg2 electron configuration with
eg electrons while a Mn
4+ ion has no
eg electron (
t2g3), a ferromagnetic interaction is expected between the adjacent spins according to the Kanamori-Goodenough rule.
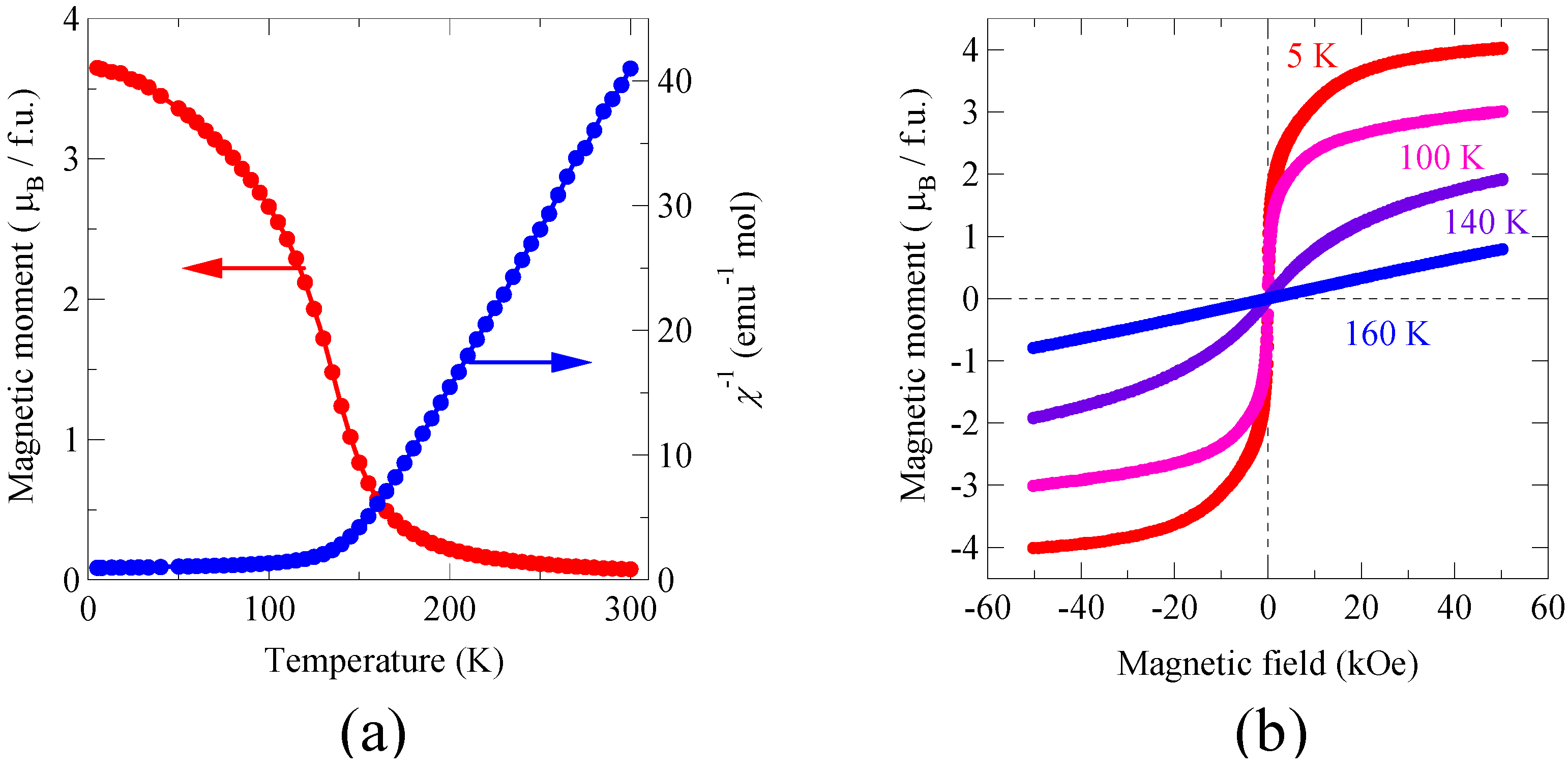
Figure 4(a) shows the temperature dependence of the magnetic moment and inverse magnetic susceptibility measured under an external field of 10 kOe. Magnetization curves measured at temperatures from 5 to 160 K are also shown in
Figure 4(b). The material shows a typical ferromagnetic behavior below 140 K. The magnetic susceptibility above the transition temperature obeys the Curie-Weiss law and the Weiss constant is about 140 K, which also suggests the ferromagnetic interaction between the Ni
2+ and Mn
4+ spins. The observed saturated magnetization at 5 K is 4.1 μ
B/f.u., which is close to 5 μ
B/f.u. expected from the ferromagnetic ordering of Ni
2+ (
S = 1) and Mn
4+ (
S = 3/2). This is in sharp contrast with the BiFeO
3 compound, in which magnetic Fe
3+ spins are aligned antiferromagnetically through the superexchange interaction. The slight deviation from the expected saturated moment is probably due to small amount of antisite disorder of the Ni
2+ and Mn
4+ ions. The resulting Ni-O-Ni and Mn-O-Mn magnetic paths produce antiferromagnetic interactions and thus reduce the saturated magnetization. It should be noted that a sample obtained by quenching from 800 °C after the high-pressure synthesis resulted in the random mixing of Ni and Mn, and that a substantial decrease of the saturated magnetic moment was observed. The magnetic measurement results clearly show that Bi
2NiMnO
6 is ferromagnetic. Therefore, we can conclude that the material is a multiferroic, and both ferroelectric and ferromagnetic properties are observed below 140 K.
In Bi2NiMnO6 the ferroelectric transition temperature is 485 K and the ferromagnetic transition temperature is 140 K, and the two ferroic orderings occur independently. Even though, both ferroelectric and ferromagnetic orderings, i.e., the multiferroic property, appear below 140 K.
Figure 4.
(a) Temperature dependence of the magnetization and inverse magnetic susceptibility of Bi2NiMnO6 measured under an external field of 10 kOe. The increase in the magnetic moment below 140 K suggests the ferromagnetic transition at that temperature; (b) Magnetization curves measured from 5 to 160 K. The ferromagnetic hysteresis was observed for the measurements at 5, 100, and 140 K.
Figure 4.
(a) Temperature dependence of the magnetization and inverse magnetic susceptibility of Bi2NiMnO6 measured under an external field of 10 kOe. The increase in the magnetic moment below 140 K suggests the ferromagnetic transition at that temperature; (b) Magnetization curves measured from 5 to 160 K. The ferromagnetic hysteresis was observed for the measurements at 5, 100, and 140 K.
2.2. Bi2NiMnO6 Epitaxially Grown Thin Film
As described above, Bi
2NiMnO
6 is a meta-stable phase synthesized under high-pressure conditions. The compound can also be obtained in a thin-film form by using epitaxial strain from the substrate lattice. A Bi
2NiMnO
6 thin film was thus grown epitaxially on a SrTiO
3 substrate by a pulsed laser deposition method [
20].
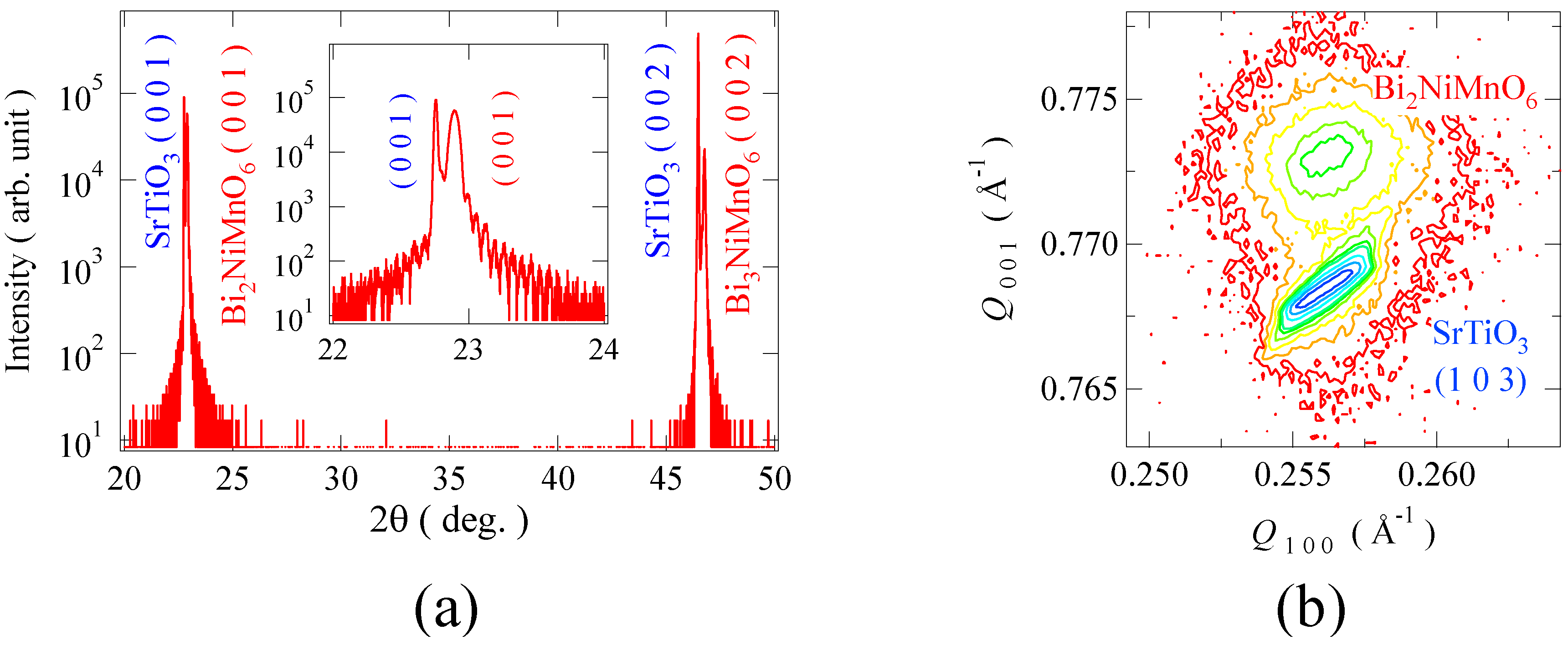
Figure 5(a) shows the
θ-2
θ X-ray diffraction pattern of the Bi
2NiMnO
6 thin film grown on the SrTiO
3 (0 0 1) substrate. In the diffraction pattern, only (0 0
l) reflections of the perovskite structure are observed, and the out-of-plane lattice parameter,
c, is ascertained to be 3.87 Å from the data.
Figure 5(b) shows a logarithmic intensity map in reciprocal lattice space around the (1 0 3) reflection for the Bi
2NiMnO
6 thin film. We can see that the in-plane lattice parameter of the film matches that of the substrate perovskite structure (
a = 3.91 Å) while the out-of-plane lattice parameter (3.87 Å) is smaller than the substrate cell. The results of the X-ray diffraction measurements indicate that the strained pseudo-tetragonal Bi
2NiMnO
6 thin film epitaxially grows on the SrTiO
3 substrate. In addition, the reflection high energy diffraction (RHEED) pattern strongly suggests a double-perovskite structure. Electron beam incidence from the [1 1 0] direction gives weak streaks of the 2-fold superstructure, whereas the [1 0 0] incidence shows no such a superstructure reflection. These results imply that the synthesized Bi
2NiMnO
6 thin film has a √2
ap×√2
ap superstructure, which originates from a rock-salt-type ordering of the B site ions in the double-perovskite structure.
Figure 5.
(a) X-ray diffraction patterns of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) Logarithmic intensity reciprocal space map around (1 0 3) reflection of the thin film. In addition to the SrTiO3 substrate peaks, Bragg reflections of the perovskite Bi2NiMnO6 thin film are clearly seen and the results also confirm the epitaxial growth of the film on the substrate lattice.
Figure 5.
(a) X-ray diffraction patterns of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) Logarithmic intensity reciprocal space map around (1 0 3) reflection of the thin film. In addition to the SrTiO3 substrate peaks, Bragg reflections of the perovskite Bi2NiMnO6 thin film are clearly seen and the results also confirm the epitaxial growth of the film on the substrate lattice.
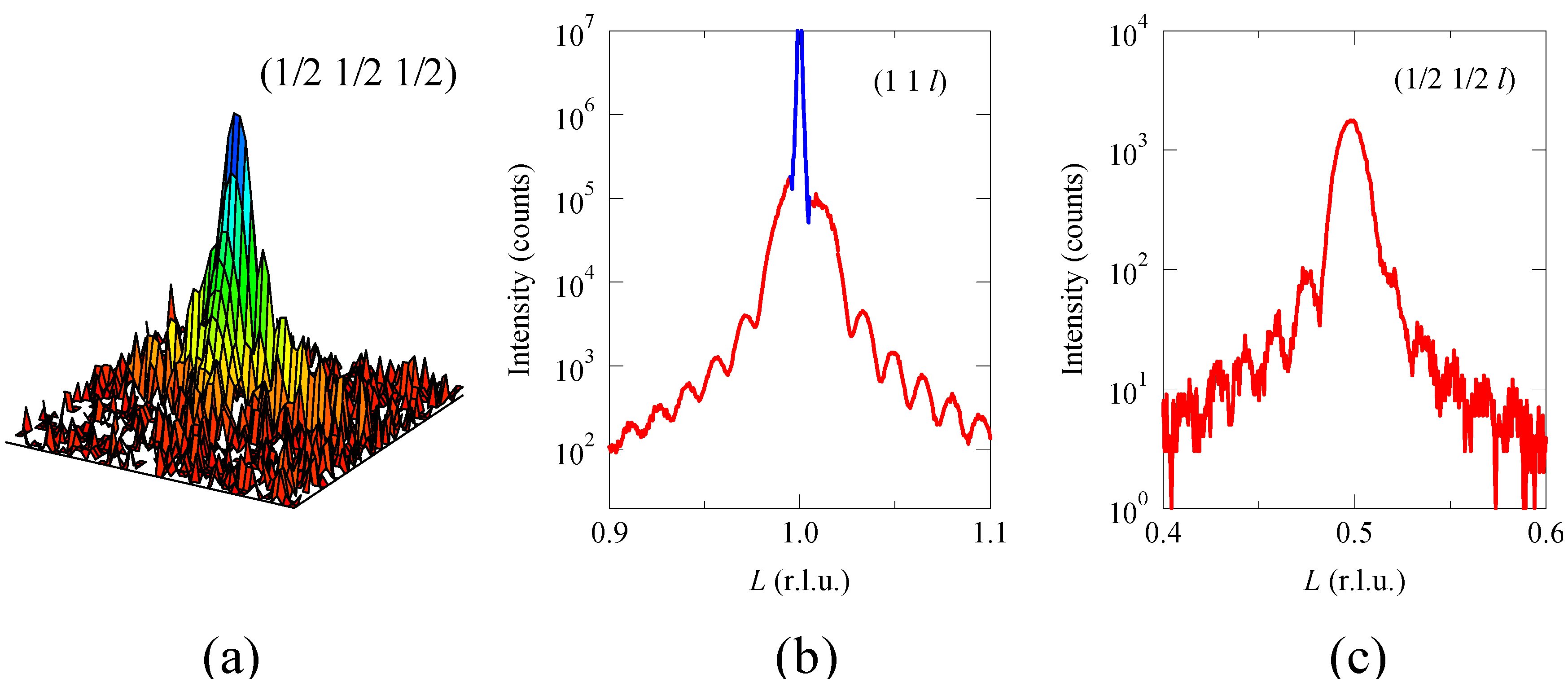
A clear evidence of the rock-salt configuration of the Ni
2+ and Mn
4+ ions in the perovskite structure is also seen in the (1/2 1/2 1/2) superstructure reflection in a reciprocal lattice space intensity map obtained by synchrotron X-ray diffraction as shown in
Figure 6. Here, the reciprocal-space coordinates correspond to the cubic SrTiO
3 substrate. The observed superstructure reflection is consistent with that from a 2
a × 2
a × 2
c ordered structure. It is noted that the observed intensity of the superstructure reflection is about 1×10
3 cps, which is only 1% of the (1 1 1) fundamental reflection intensity. And the weak intensity of the (1/2 1/2 1/2) superstructure reflection is well reproduced from a simple ordered structure model where Ni
2+ and Mn
4+ ions are ordered in the rock-salt configuration in the perovskite structure. The calculated intensities of the (1/2 1/2 1/2) and (1 1 1) reflections for the rock-salt arrangement of the Ni and Mn ions are 94 and 33,055, respectively, for the maximum peak intensity of 100,000 for the (1 0 1) reflection. Note that the calculated (1/2 1/2 1/2) diffraction intensity for the random arrangement of the Ni and Mn ions at the B site in the perovskite structure is zero. Also note that the Laue’s-intensity oscillation is observed on the superstructure diffraction peak with the same interval as that observed on the (1 1 1) reflection, confirming that both (1 1 1) and (1/2 1/2 1/2) diffraction peaks originate from the structure of the grown Bi
2NiMnO
6 film.
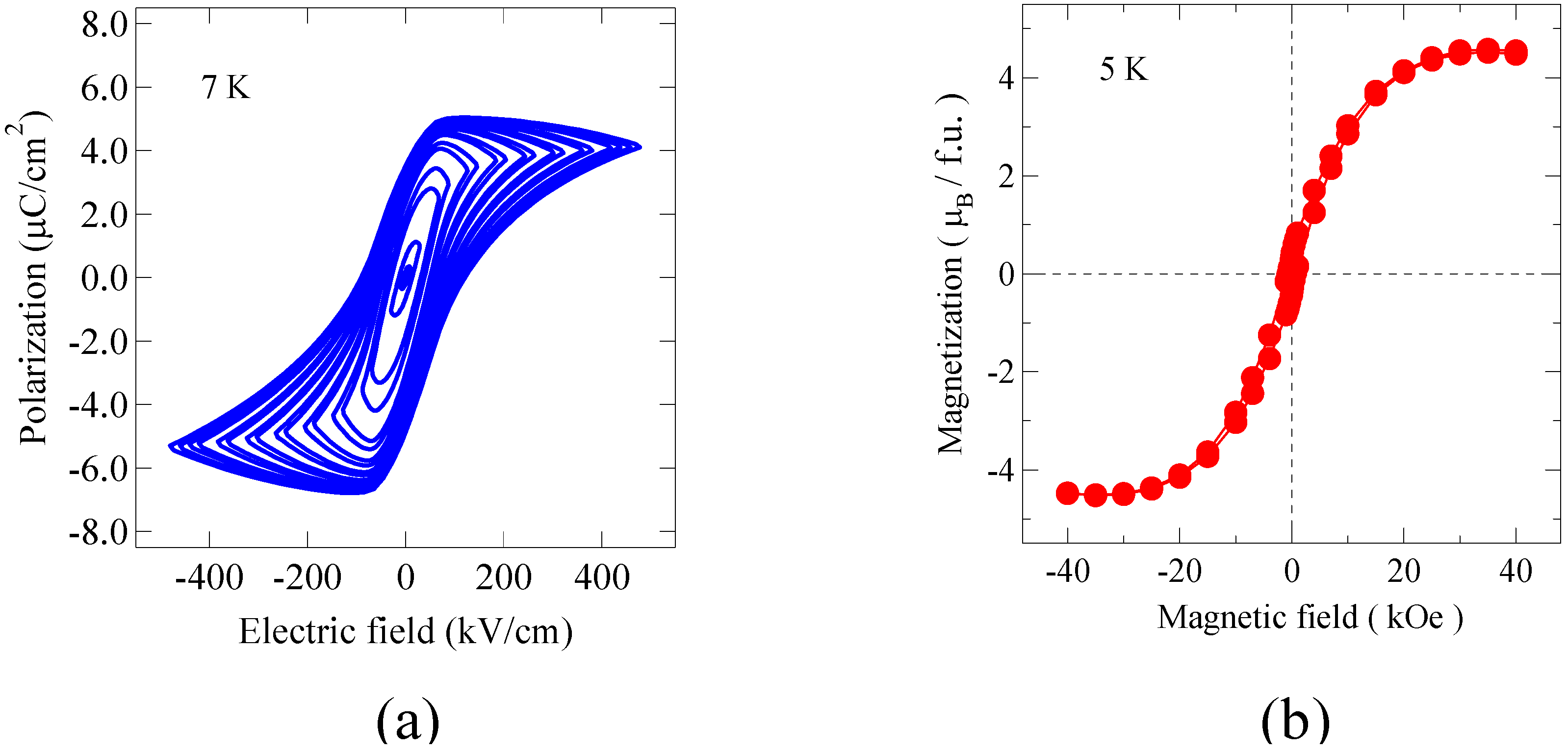
The ferroelectric behavior of the Bi
2NiMnO
6 thin film is confirmed by the polarization hysteresis measured at 7 K as shown in
Figure 7(a). Although the observed slightly distorted hysteresis loop includes an effect of leakage current, we can see the saturated polarization of about 5 μC/cm
2 above 80 kV/cm. The observed polarization is quite small compared to the polarization of about 20 μC/cm
2 calculated from the refined bulk crystal structure. If the essential crystal structure of the thin film is similar to that of bulk with the
C2 noncentrosymmetric space group, the polarization vector should be along the [1 1 1] direction of the simple perovskite structure, and thus the observed ferroelectric polarization of the film should be a projection along the out-of-plane direction. This may explain the difference between the bulk and thin film in the polarization value.
Figure 7(b) shows a magnetization behavior of the Bi
2NiMnO
6 film at 5 K. The observed saturated magnetization is 4.2 μ
B/f.u., which is close to the 5 μ
B/f.u. that would be expected from the ferromagnetic ordering of Ni
2+ (
S = 1) and Mn
4+ (
S = 3/2) spins. The magnetization is also consistent with the observed moment of the bulk sample synthesized under high pressure. The important point is that our Bi
2NiMnO
6 thin film shows both ferromagnetic and ferroelectric properties, that is the multiferroic property, at low temperatures.
Figure 6.
(a) Synchrotron X-ray logarithmic intensity reciprocal space map around (1/2 1/2 1/2) reflection of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) (1 1 1) and (c) (1/2 1/2 1/2) intensity profiles along the L direction of the thin film. A sharp diffraction peak (blue) in (b) originates from the SrTiO3 substrate. A weak intensity of the (1/2 1/2 1/2) superstructure reflection, which is only 1% of the (1 1 1) fundamental reflection, can be observed in the synchrotron X-ray diffraction, confirming the rock-salt type ordering of the Ni2+ and Mn4+ ions in the Bi2NiMnO6 double perovskite structure.
Figure 6.
(a) Synchrotron X-ray logarithmic intensity reciprocal space map around (1/2 1/2 1/2) reflection of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) (1 1 1) and (c) (1/2 1/2 1/2) intensity profiles along the L direction of the thin film. A sharp diffraction peak (blue) in (b) originates from the SrTiO3 substrate. A weak intensity of the (1/2 1/2 1/2) superstructure reflection, which is only 1% of the (1 1 1) fundamental reflection, can be observed in the synchrotron X-ray diffraction, confirming the rock-salt type ordering of the Ni2+ and Mn4+ ions in the Bi2NiMnO6 double perovskite structure.
Then, we looked at the coupling between the ferroelectric and ferromagnetic properties of our Bi
2NiMnO
6 film by measuring the dielectric property around the ferromagnetic transition temperature.
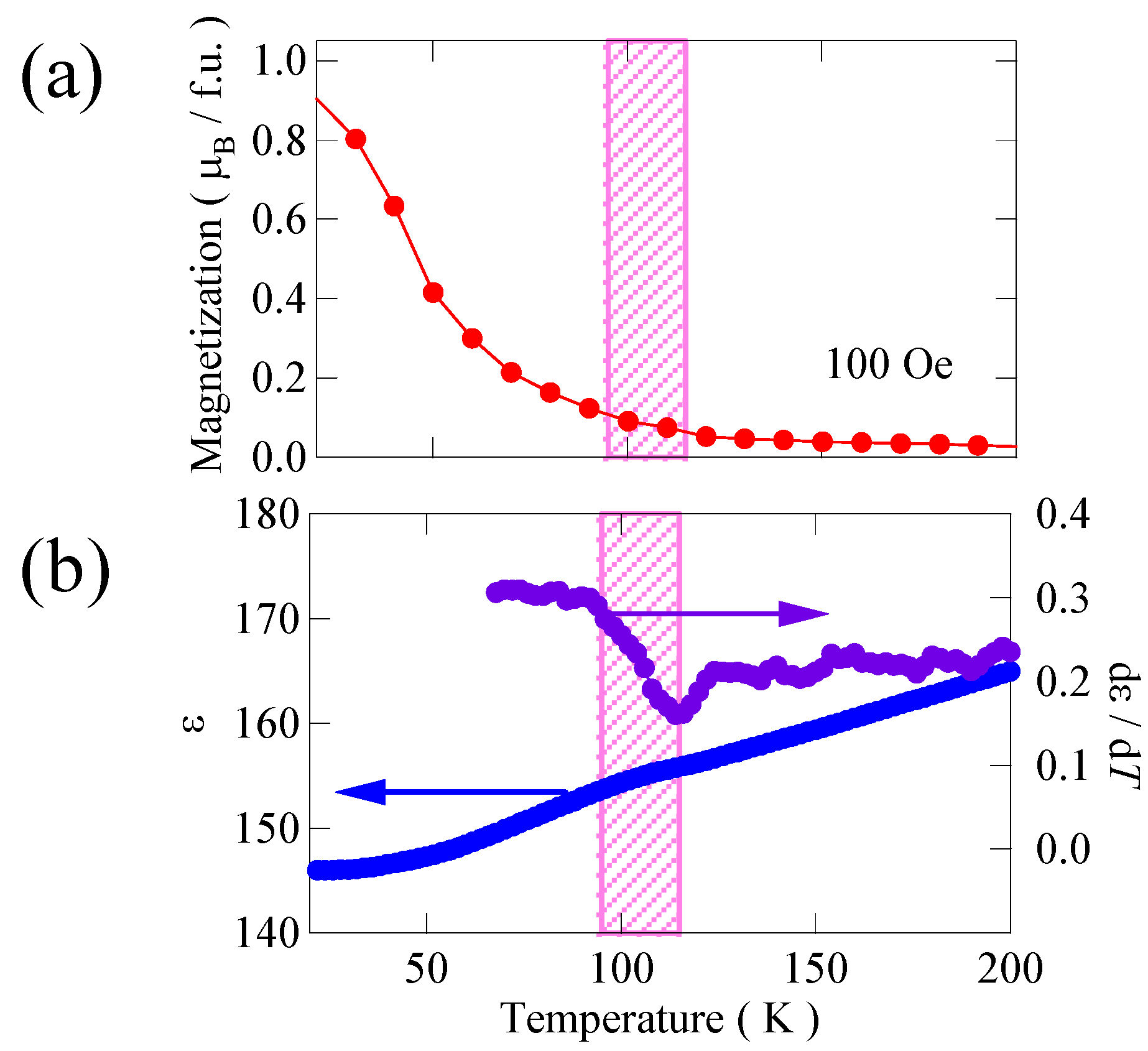
Figure 8(a) shows the temperature dependence of the magnetization of the Bi
2NiMnO
6 thin film measured under a 100 Oe magnetic field applied to the in-plane direction. The magnetization increases below about 100 K, suggesting that the ferromagnetic transition of the present Bi
2NiMnO
6 film is around 100 K. Although this transition temperature is lower than 140 K observed with the bulk, structural strain of the epitaxially grown film may change the ferromagnetic transition temperature. As shown in
Figure 8(b), the observed dielectric constant shows a very small anomaly near the ferromagnetic transition temperature. Although the change is very small, the anomaly near the ferromagnetic transition temperature appears to show some interplay between the ferromagnetic and ferroelectric interactions. Thus, the coupling between ferromagnetic and ferroelectric interactions seems to exist in Bi
2NiMnO
6, but it is quite small.
Figure 7.
(a) Ferroelectric hysteresis curves of the Bi2NiMnO6 thin film measured at 7 K of the film. Clear polarization hysteresis loops confirm the ferroelectricity of the film; (b) Magnetization curve measured at 5 K of the thin film. A typical ferromagnetic behavior was seen.
Figure 7.
(a) Ferroelectric hysteresis curves of the Bi2NiMnO6 thin film measured at 7 K of the film. Clear polarization hysteresis loops confirm the ferroelectricity of the film; (b) Magnetization curve measured at 5 K of the thin film. A typical ferromagnetic behavior was seen.
Figure 8.
(a) Temperature dependence of magnetization of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) Temperature dependence of dielectric constant and the temperature derivative of the film. An anomaly in ε near the ferromagnetic transition temperature (near 100 K indicated in pink) suggests some interplay between the ferromagnetic and ferroelectric interactions.
Figure 8.
(a) Temperature dependence of magnetization of Bi2NiMnO6 thin film grown on SrTiO3 substrate; (b) Temperature dependence of dielectric constant and the temperature derivative of the film. An anomaly in ε near the ferromagnetic transition temperature (near 100 K indicated in pink) suggests some interplay between the ferromagnetic and ferroelectric interactions.
2.3. Bi2FeCrO6 Artificial Superlattice Thin Film
Another possible multiferroic compound is a double perovskite, Bi
2FeCrO
6, because electron configurations of Fe
3+ and Cr
3+ are
t2g3eg2 and
t2g3, respectively. However, a bulk sample synthesized under a high-pressure condition did not show large magnetization from the ferromagnetism [
24]. The observed saturated magnetization of a thin film sample was also more than an order of magnitudes smaller than that expected from the ideal value of the ferromagnetic ordering of Fe
3+ and Cr
3+ spins [
25]. These should result from disorder between Fe
3+ and Cr
3+ ions at the B site in the perovskite structure because they are isovalent and have similar ionic radii. We thus made a BiFeO
3/BiCrO
3 artificial superlattice with a 1/1 stacking period on a perovskite-structure SrTiO
3 (1 1 1) substrate [
22]. While BiFeO
3 and BiCrO
3 [
26] are antiferromagnets, these 1/1 superlattice along [1 1 1] direction contain Fe-O-Cr bonds producing the rock-salt configuration in the perovskite structure (
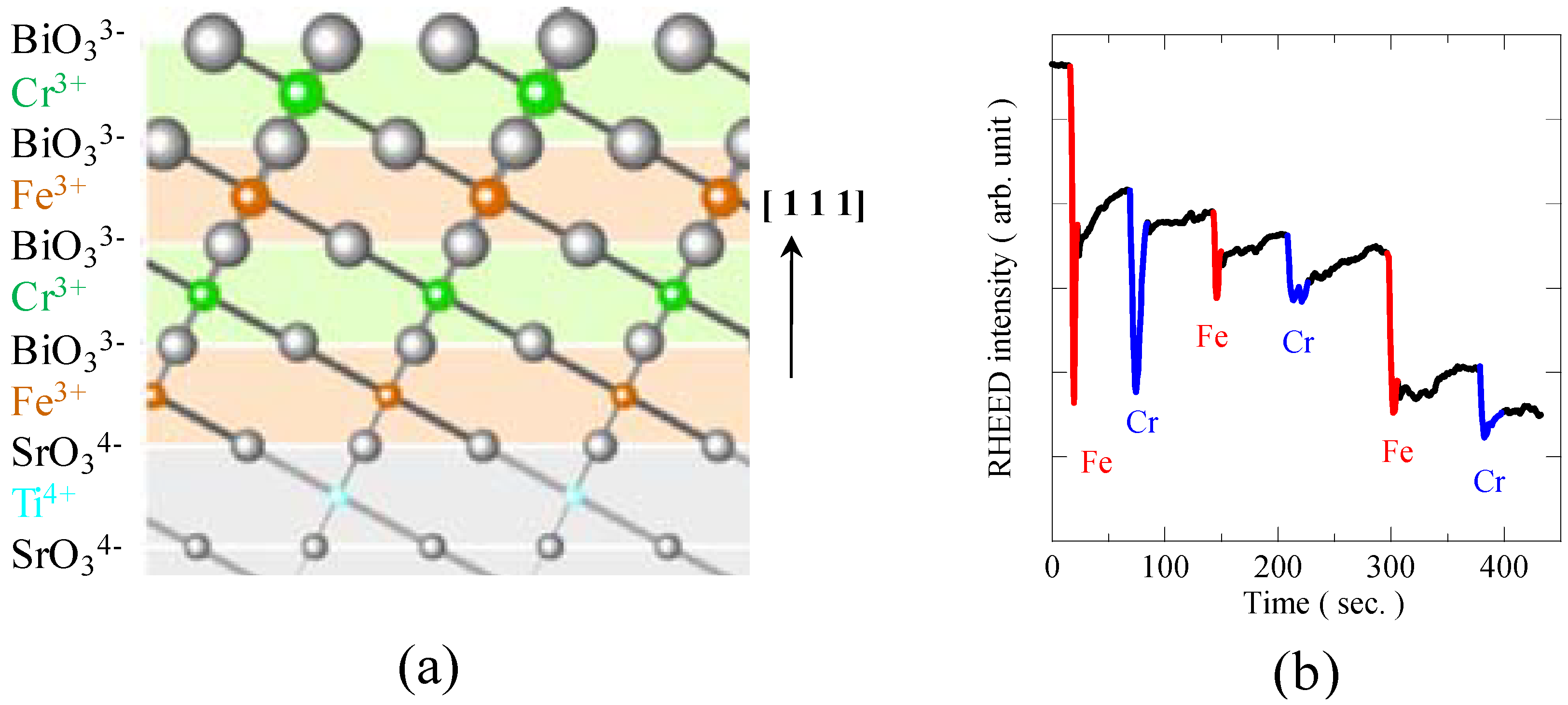
Figure 9(a)).
The 1/1 artificial superlattice of BiFeO
3/BiCrO
3 was grown epitaxially on a SrTiO
3 substrate by a pulsed laser deposition method. Two-dimensional layer-by-layer growth was confirmed by monitoring the RHEED intensity oscillations
in situ from the start of the deposition to the end.
Figure 9(b) shows the change in RHEED intensity during the deposition of a 6-monolayer (ML) superlattice. So far, clear oscillations during the depositions for the growth of only 2 ML ([BiFeO
3/BiCrO
3]
1), 4 ML ([BiFeO
3/BiCrO
3]
2), and 6 ML ([BiFeO
3/BiCrO
3]
3) were observed. Although for the sample identification it is difficult to obtain the X-ray diffraction patterns from such very thin film samples, the results of magnetic and ferroelectric property measurements appear to show multiferroic property of the prepared artificial superlattice.
Figure 9.
(a) Schematic figure of BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate. Monolayers of BiFeO3 and BiCrO3 are shown in orange and green, respectively. One-by-one stacking of the BiFeO3 and BiCrO3 monolayers along [1 1 1] direction produces the rock-salt configuration of Bi3+ and Cr3+ ions in the perovskite structure, so the Fe3+-O-Cr3+ magnetic path is expected to lead to the ferromagnetism; (b) RHEED intensity oscillation during the deposition of the 6-monolayer artificial superlattice thin film, confirming the two-dimensional layer-by-layer growth.
Figure 9.
(a) Schematic figure of BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate. Monolayers of BiFeO3 and BiCrO3 are shown in orange and green, respectively. One-by-one stacking of the BiFeO3 and BiCrO3 monolayers along [1 1 1] direction produces the rock-salt configuration of Bi3+ and Cr3+ ions in the perovskite structure, so the Fe3+-O-Cr3+ magnetic path is expected to lead to the ferromagnetism; (b) RHEED intensity oscillation during the deposition of the 6-monolayer artificial superlattice thin film, confirming the two-dimensional layer-by-layer growth.
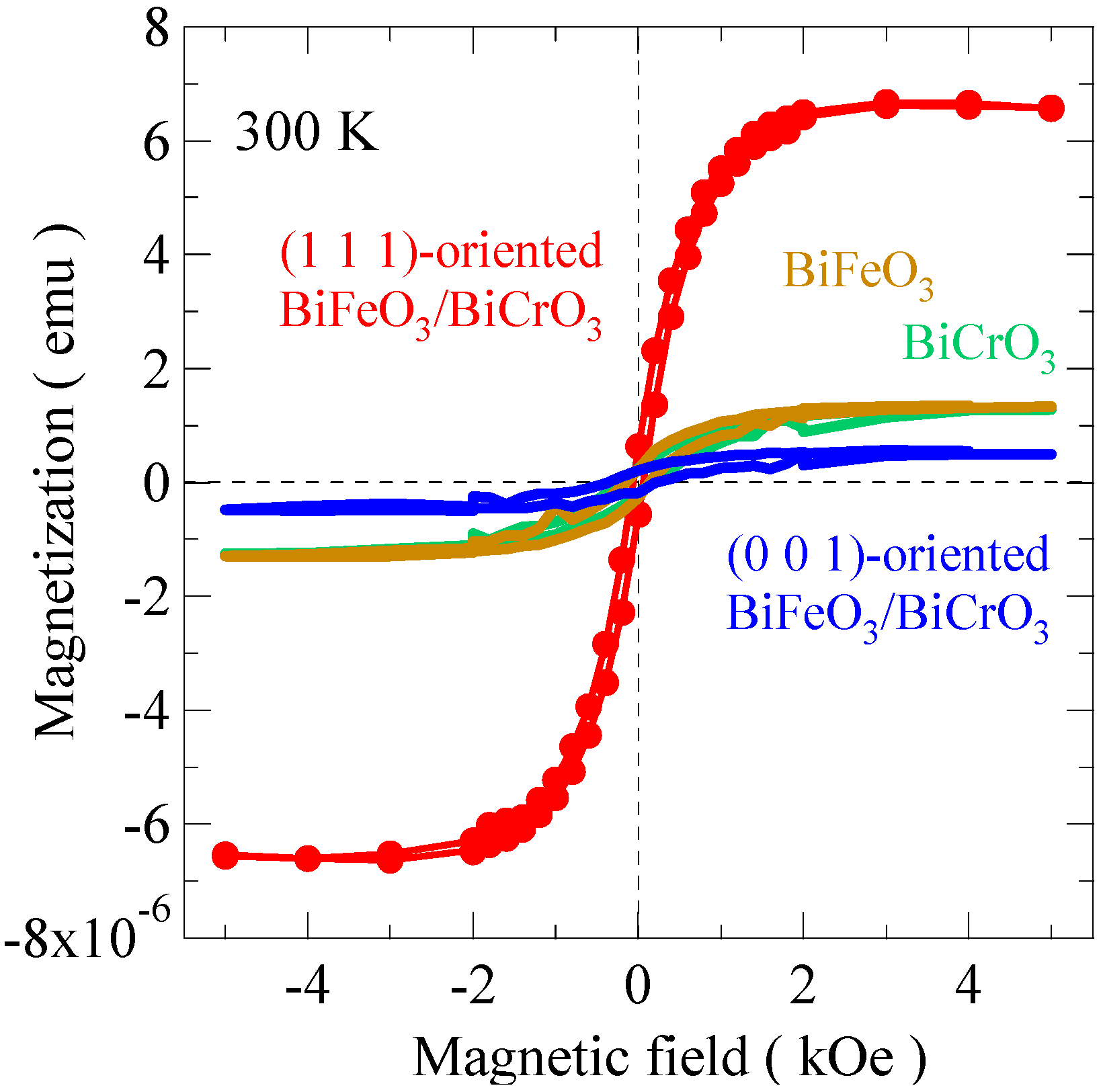
Figure 10 shows the magnetization curves of the 4-ML ([BiFeO
3/BiCrO
3]
2) artificial superlattice film measured at 300 K. Those of 4-ML BiFeO
3 and 4-ML BiCrO
3 grown on SrTiO
3 (1 1 1), and that of a 4-ML ([BiFeO
3/BiCrO
3]
2) artificial superlattice film grown on a SrTiO
3 (0 0 1) substrate are also shown in the figure. The magnetization in the BiFeO
3/BiCrO
3 superlattices evidently depends on the orientations and the results are consistent with the Kanamori-Goodenough rule. The magnetization signals of the (0 0 1) superlattice are quite small and compatible with the extrinsic magnetization from the substrate. In the (0 0 1) superlattice the magnetic interaction between the monolayers is antiferromagnetic, so the stacking does not lead to ferromagnetism. The magnetic response from each single material film with the (1 1 1) orientation was also small. Therefore, the magnetization results strongly suggest that the BiFeO
3/BiCrO
3 artificial superlattice with the 1/1 stacking period along the [1 1 1] direction produced the ferromagnetic moment.
Figure 10.
Magnetization curves measured at 300 K of 4-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate (red). Those of 4-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (0 0 1) substrate (blue), and 4-monolayer BiFeO3 (brown) and 4-monolayer BiCrO3 (green) thin films grown on SrTiO3 (1 1 1) substrates are also shown. Only the (1 1 1)-oriented BiFeO3/BiCrO3 artificial superlattice shows the large magnetization, confirming the ferromagnetic property of the film.
Figure 10.
Magnetization curves measured at 300 K of 4-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate (red). Those of 4-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (0 0 1) substrate (blue), and 4-monolayer BiFeO3 (brown) and 4-monolayer BiCrO3 (green) thin films grown on SrTiO3 (1 1 1) substrates are also shown. Only the (1 1 1)-oriented BiFeO3/BiCrO3 artificial superlattice shows the large magnetization, confirming the ferromagnetic property of the film.
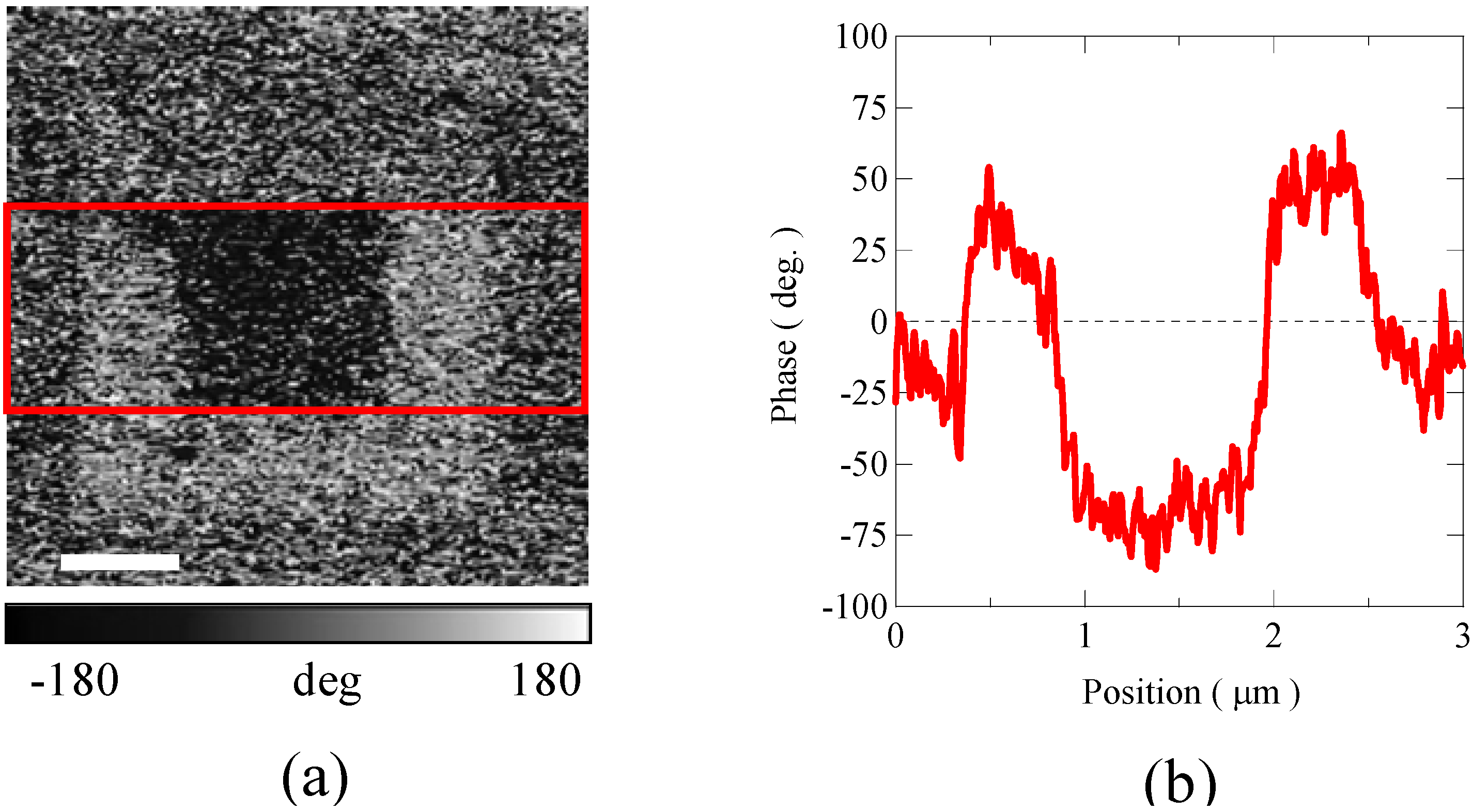
Figure 11.
(a) Non-linear dielectric constant microscope phase image taken at room temperature of 6-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate. The white scale bar indicates 600 nm. The image was taken after applying 10V in the outer square area (2 × 2 μm2) and then −10 V in the center square area (1 × 1 μm2). (b) Phase shift of the image obtained by averaging the data in the rectangle area surrounded by red lines in (a). The result shows that the ferroelectric polarization domains in the artificial superlattice BiFeO3/BiCrO3 thin film can be switched by the applied voltage.
Figure 11.
(a) Non-linear dielectric constant microscope phase image taken at room temperature of 6-monolayer BiFeO3/BiCrO3 artificial superlattice grown on SrTiO3 (1 1 1) substrate. The white scale bar indicates 600 nm. The image was taken after applying 10V in the outer square area (2 × 2 μm2) and then −10 V in the center square area (1 × 1 μm2). (b) Phase shift of the image obtained by averaging the data in the rectangle area surrounded by red lines in (a). The result shows that the ferroelectric polarization domains in the artificial superlattice BiFeO3/BiCrO3 thin film can be switched by the applied voltage.
The ferroelectric properties of the (1 1 1) orientated BiFeO
3/BiCrO
3 artificial superlattices were investigated by polarization domain switching using a scanning non-linear dielectric constant microscope. The measurements were performed at room temperature and an AC voltage of 0.5–2.0 V
p-p was applied between the back side of the substrate and the probe tip during the scans. Positively and negatively poled voltage patterns were observed in the scans of a 6-ML superlattice as shown in
Figure 11. The result demonstrates that polarization reversal is indeed possible proving that the film is ferroelectric and that the ferroelectric polarization domains can be switched by the applied voltage. All the experimental results on ferromagnetic and ferroelectric properties suggest that the grown Bi
2FeCrO
6 artificial superlattice shows multiferroic property at room temperature.