Overall Performance Enhancement of Epoxy Resins Loaded with Non-Covalently Modified Carbon Nanotubes and Graphene Nanosheets
Abstract
1. Introduction
- Preserved structure and conductivity: Non-covalent functionalization (e.g., π–π stacking with surfactants, polymers, or aromatic molecules) adsorbs onto CNT/graphene surfaces without breaking C–C bonds, thereby maintaining their stiffness, strength, and electrical conductivity [2].
- Better dispersion and reduced agglomeration: Epoxy resins are polar and viscous, while pristine CNTs/graphene tend to bundle via van der Waals forces. Non-covalent modifiers improve compatibility with the epoxy matrix, leading to finer, more uniform dispersion and fewer stress-concentrating agglomerates [1,2,3].
- Improved interfacial adhesion: The modifier layer can interact with the epoxy network (via hydrogen bonding, polar interactions, or mechanical interlocking), creating a more effective load-transfer interface while still leveraging the high modulus of carbon nanotubes and graphene [1].
- Lower percolation thresholds: Non-covalently modified carbon nanotubes or graphene nanosheets efficiently reduce the electrical percolation threshold (EPT), often below 0.1–0.5 wt%, by improving filler dispersion, preventing agglomeration, and preserving high aspect ratios.
2. Materials and Methods
3. Results and Discussion
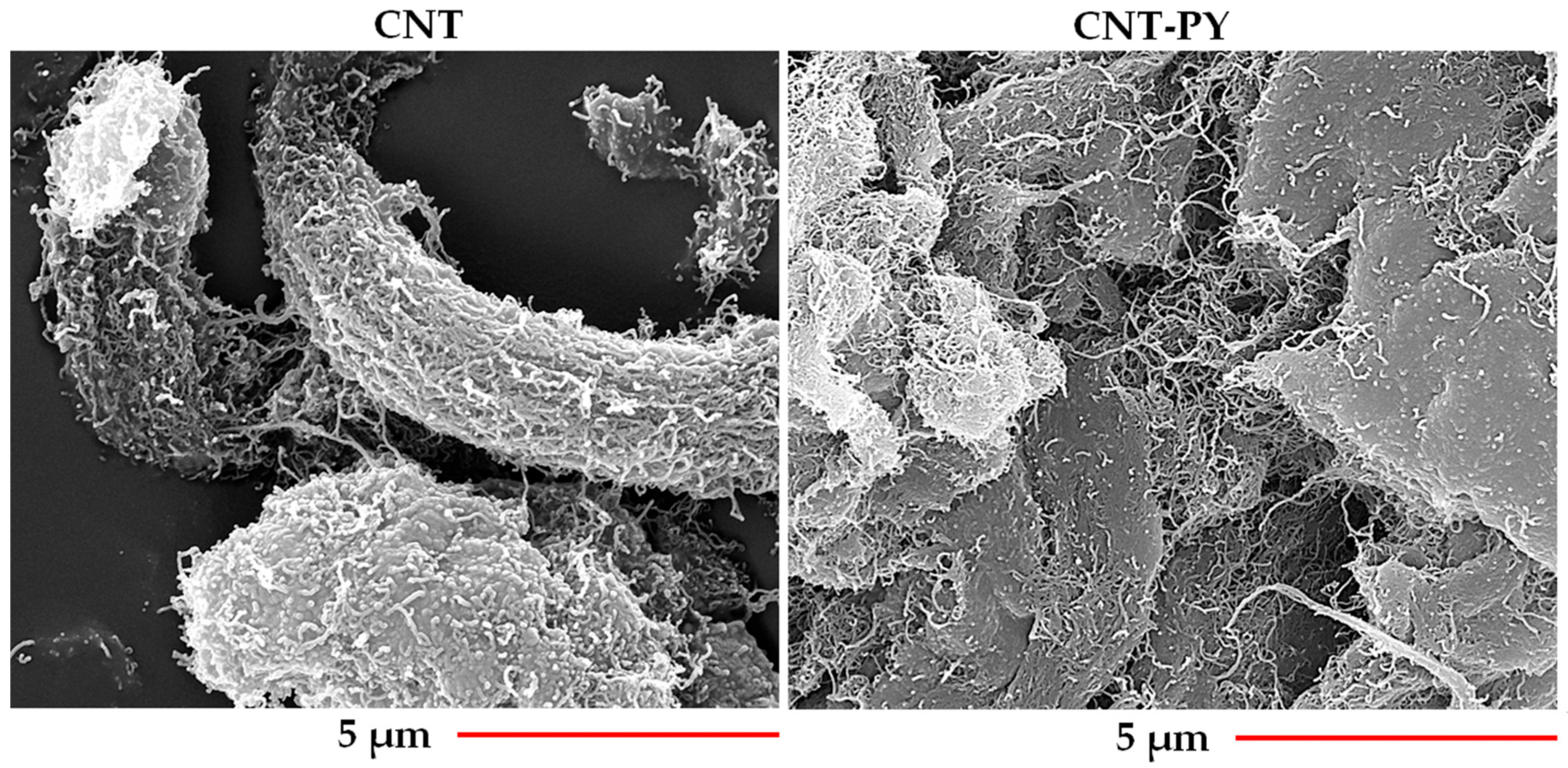
3.1. Morphology of Unfunctionalized and Functionalized Carbon Nanofillers
3.2. DSC Investigation and TGA Analysis
3.3. Rheological Properties
3.4. Self-Healing Performance of the Epoxy Composites
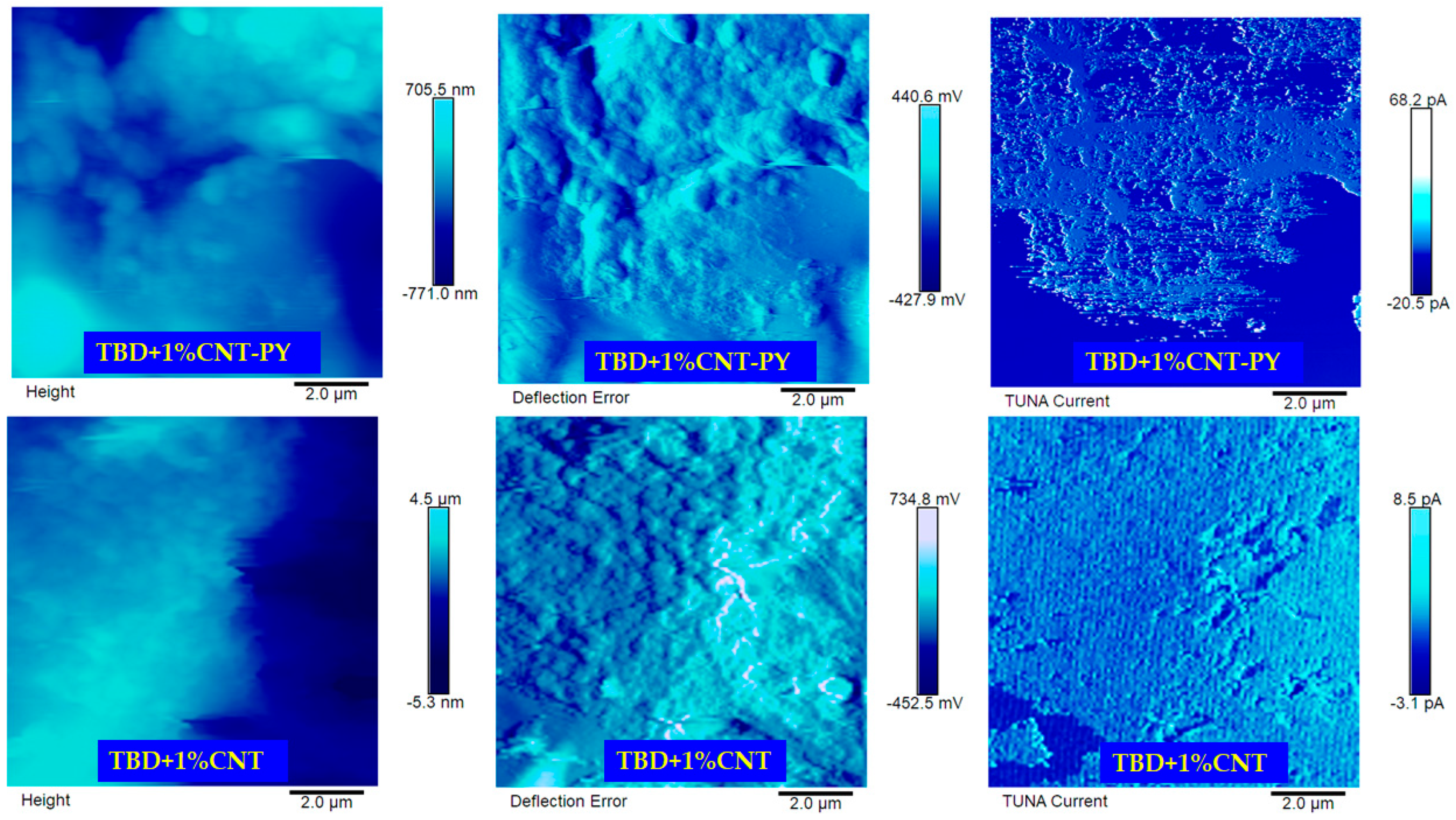
3.5. Morphology and Nanoscale Electrical Property Mapping by Means of TUNA of the Epoxy Nanocomposites
4. Conclusions
- The non-covalent modification does not disrupt the layered structure of G and allows for better separation of CNT-PY compared to untreated CNT.
- Thermal analysis shows that this non-covalent modification significantly increases the thermal stability of unfunctionalized G by about 70 °C and enhances the oxidative thermostability of the composites. Pristine CNT remains stable up to about 520 °C, while modified CNT-PY experiences about 10 wt% weight loss at this temperature due to pyrene degradation.
- The degree of cure (DC) values of the functionalized G-PY and CNT-PY nanocomposites cured under isothermal heating are about 90%, meeting aviation industry standards. All samples cured at 200 °C show cross-link onset temperatures above 180 °C, while uncured samples begin cross-linking at around 125 °C.
- A significant decrease in viscosity occurs with functionalized graphene (G-PY) and functionalized carbon nanotubes (CNT), thus impacting industrial preparation processes.
- Non-covalently modified carbon nanotubes (CNT-PY) and graphene (G-PY) enhance the self-healing properties of epoxy resin, achieving efficiencies of 30% and 15%, respectively. Their superior dispersion and interfacial compatibility facilitate better healing compared to unmodified versions.
- The nanocomposite with functionalized carbon nanotubes (CNT-PY) showed the highest efficiency compared to the one with graphene (G-PY). This is due to better network formation and interactions during healing, as CNTs improve conductivity, crack bridging, and localized heating. Graphene’s rigidity limits polymer movement, reducing healing efficiency.
- The TUNA analysis shows that the TBD + 1%G-PY sample exhibits significant electric currents ranging from −4.1 pA to 12.7 pA, which confirms the high conductivity of 0.1 S/m. The electric currents measured for the sample TBD + 1%CNT-PY range from −20. 5 pA to 68. 2 pA and are well correlated with the high conductivity of 0. 391 S/m. The good electrical performance at the nanoscale is due to effective non-covalent functionalization with PY.
- These results show that non-covalently modified G-PY and CNT-PY can be effectively used to create functional nanocomposites for structural applications while preserving the electronic structure and properties of the original materials.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spinelli, G.; Guarini, R.; Batakliev, T.; Guadagno, L.; Raimondo, M. Testing, Experimental Design, and Numerical Analysis of Nanomechanical Properties in Epoxy Hybrid Systems Reinforced with Carbon Nanotubes and Graphene Nanoparticles. Polymers 2024, 16, 3420. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Zhang, H.; Tzounis, L.; Santagiuliana, G.; Bilotti, E.; Papageorgiou, D.G. Multifunctional epoxy nanocomposites reinforced by two-dimensional materials: A review. Carbon 2021, 185, 57–81. [Google Scholar] [CrossRef]
- Tee, Z.Y.; Yeap, S.P.; Hassan, C.S.; Kiew, P.L. Nano and non-nano fillers in enhancing mechanical properties of epoxy resins: A brief review. Polym.-Plast. Technol. Mater. 2022, 61, 709–725. [Google Scholar] [CrossRef]
- Nobile, M.R.; Raimondo, M.; Naddeo, C.; Guadagno, L. Rheological and Morphological Properties of Non-Covalently Functionalized Graphene-Based Structural Epoxy Resins with Intrinsic Electrical Conductivity and Thermal Stability. Nanomaterials 2020, 10, 1310. [Google Scholar] [CrossRef]
- Ponnamma, D.; Sung, S.H.; Hong, J.; Ahn, K.H.; Varughese, K.T.; Thomas, S. Influence of non-covalent functionalization of carbon nanotubes on the rheological behavior of natural rubber latex nanocomposites. Eur. Polym. J. 2014, 53, 147–159. [Google Scholar] [CrossRef]
- Sharma, S.K.; Miladinović, S.; Sharma, L.K.; Gajević, S.; Sharma, Y.; Sharma, M.; Čukić, S.; Stojanović, B. Graphene/CNT Nanocomposites: Processing, Properties, and Applications. Nanomaterials 2026, 16, 100. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, L.; Raimondo, M.; Vertuccio, L.; Naddeo, C.; Barra, G.; Longo, P.; Lamberti, P.; Spinelli, G.; Nobile, M. Morphological, rheological and electrical properties of composites filled with carbon nanotubes functionalized with 1-pyrenebutyric acid. Compos. Part B Eng. 2018, 147, 12–21. [Google Scholar] [CrossRef]
- Chen, C.; Li, X.; Wen, Y.; Liu, J.; Li, X.; Zeng, H.; Xue, Z.; Zhou, X.; Xie, X. Noncovalent engineering of carbon nanotube surface by imidazolium ionic liquids: A promising strategy for enhancing thermal conductivity of epoxy composites. Compos. Part A Appl. Sci. Manuf. 2019, 125, 105517. [Google Scholar] [CrossRef]
- Teng, C.C.; Ma, C.C.M.; Lu, C.H.; Yang, S.Y.; Lee, S.H.; Hsiao, M.C.; Yen, M.Y.; Chiou, K.C.; Lee, T. Thermal conductivity and structure of non-covalent functionalized graphene/epoxy composites. Carbon 2011, 49, 5107–5116. [Google Scholar] [CrossRef]
- Silva, S.; Barbosa, J.M.; Sousa, J.D.; Paiva, M.C.; Teixeira, P.F. High-Performance PEEK/MWCNT Nanocomposites: Combining Enhanced Electrical Conductivity and Nanotube Dispersion. Polymers 2024, 16, 583. [Google Scholar] [CrossRef]
- Ma, P.C.; Siddiqui, N.A.; Marom, G.; Kim, J.K. Dispersion and functionalization of carbon nanotubes for polymer-based nanocomposites: A review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 1345–1367. [Google Scholar] [CrossRef]
- Wernik, J.M.; Meguid, S.A. Recent Developments in Multifunctional Nanocomposites Using Carbon Nanotubes. Appl. Mech. Rev. 2010, 63, 050801. [Google Scholar] [CrossRef]
- Novikov, I.V.; Krasnikov, D.V.; Vorobei, A.M.; Zuev, Y.I.; Butt, H.A.; Fedorov, F.S.; Gusev, S.A.; Safonov, A.A.; Shulga, E.V.; Konev, S.D.; et al. Multifunctional Elastic Nanocomposites with Extremely Low Concentrations of Single-Walled Carbon Nanotubes. ACS Appl. Mater. Interfaces 2022, 14, 18866–18876. [Google Scholar] [CrossRef]
- Kusumahastuti, D.K.A.; Winarni, I.; Mardawati, E.; Iswanto, A.H.; Lubis, M.A.R. Synthesis, characterization, applications, and environmental impacts of nanofluid thermosetting adhesives for sustainable bio-composites: A review. Case Stud. Chem. Environ. Eng. 2025, 11, 101161. [Google Scholar] [CrossRef]
- Sun, Y.; Li, C.; Zhang, F.; Xue, J.; Zheng, Q. Surface and Interfacial Engineering for Multifunctional Nanocarbon Materials. ACS Nano 2025, 19, 1944–1980. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, L.; Sorrentino, A.; Longo, R.; Raimondo, M. Multifunctional Properties of Polyhedral Oligomeric Silsesquioxanes (POSS)-Based Epoxy Nanocomposites. Polymers 2023, 15, 2297. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, L.; Naddeo, C.; Sorrentino, A.; Raimondo, M. Thermo-Mechanical Performance of Epoxy Hybrid System Based on Carbon Nanotubes and Graphene Nanoparticles. Nanomaterials 2023, 13, 2427. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, L.; Raimondo, M.; Vertuccio, L.; Mauro, M.; Guerra, G.; Lafdi, K.; De Vivo, B.; Lamberti, P.; Spinelli, G.; Tucci, V. Optimization of graphene-based materials outperforming host epoxy matrices. RSC Adv. 2015, 5, 36969–36978. [Google Scholar] [CrossRef]
- Raimondo, M.; Guadagno, L.; Vertuccio, L.; Naddeo, C.; Barra, G.; Spinelli, G.; Lamberti, P.; Tucci, V.; Lafdi, K. Electrical conductivity of carbon nanofiber reinforced resins: Potentiality of Tunneling Atomic Force Microscopy (TUNA) technique. Compos. Part B Eng. 2018, 143, 148–160. [Google Scholar] [CrossRef]
- Raimondo, M.; Guadagno, L.; Speranza, V.; Bonnaud, L.; Dubois, P.; Lafdi, K. Multifunctional graphene/POSS epoxy resin tailored for aircraft lightning strike protection. Compos. Part B Eng. 2018, 140, 44–56. [Google Scholar] [CrossRef]
- Di Crescenzo, A.; Ettorre, V.; Fontana, A. Non-covalent and reversible functionalization of carbon nanotubes. Beilstein J. Nanotechnol. 2014, 5, 1675–1690. [Google Scholar] [CrossRef]
- Dubey, R.; Dutta, D.; Sarkar, A.; Chattopadhyay, P. Functionalized carbon nanotubes: Synthesis, properties and applications in water purification, drug delivery, and material and biomedical sciences. Nanoscale Adv. 2021, 3, 5722–5744. [Google Scholar] [CrossRef] [PubMed]
- Bhagavathi Kandy, S.; Simon, J.P.; Cheng, W.; Zank, J.; Saito, K.; Bhattacharyya, A.R. Effect of Organic Modification on Multiwalled Carbon Nanotube Dispersions in Highly Concentrated Emulsions. ACS Omega 2019, 4, 6647–6659. [Google Scholar] [CrossRef]
- Tuncel, D. Non-covalent interactions between carbon nanotubes and conjugated polymers. Nanoscale 2011, 3, 3545–3554. [Google Scholar] [CrossRef]
- Nobile, M.R.; Naddeo, C.; Raimondo, M.; Guadagno, L. Effect of functionalized carbon nanofillers on the rheological behavior of structural epoxy resins. AIP Conf. Proc. 2019, 2196, 020027. [Google Scholar] [CrossRef]
- Mohammed, M.; Jawad, A.J.M.; Mohammed, A.M.; Oleiwi, J.K.; Tijjani, A.; Osman, A.F.; Dahham, O.S.; Betar, B.O.; Gopinath, S.C.; Jaafar, M. Challenges and advancement in water absorption of natural fiber-reinforced polymer composites. Polym. Test. 2023, 124, 108083. [Google Scholar] [CrossRef]
- Sharma, S.K.; Gajević, S.; Sharma, L.K.; Sharma, Y.; Sharma, M.; Ivanović, L.; Milojević, S.; Stojanović, B. Self-Healing Polymer Nanocomposites: Mechanisms, Structure-Property Relationships, and Emerging Applications. Polymers 2026, 18, 276. [Google Scholar] [CrossRef]
- Fuseini, M.; Zaghloul, M.M.Y.; Abakar, D.; Yousry Zaghloul, M.M. Review of epoxy nano-filled hybrid nanocomposite coatings for tribological applications. FlatChem 2025, 49, 100768. [Google Scholar] [CrossRef]
- Caglayan, C.; Kim, G.; Yun, G.J. CNT-Reinforced Self-Healable Epoxy Dynamic Networks Based on Disulfide Bond Exchange. ACS Omega 2022, 7, 43480–43491. [Google Scholar] [CrossRef]
- Zhan, J.; Lei, Z.; Zhang, Y. Non-covalent interactions of graphene surface: Mechanisms and applications. Chem 2022, 8, 947–979. [Google Scholar] [CrossRef]
- Li, T.; Meng, Z.; Keten, S. Interfacial mechanics and viscoelastic properties of patchy graphene oxide reinforced nanocomposites. Carbon 2020, 158, 303–313. [Google Scholar] [CrossRef]










Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Raimondo, M.; Guadagno, L. Overall Performance Enhancement of Epoxy Resins Loaded with Non-Covalently Modified Carbon Nanotubes and Graphene Nanosheets. Materials 2026, 19, 1569. https://doi.org/10.3390/ma19081569
Raimondo M, Guadagno L. Overall Performance Enhancement of Epoxy Resins Loaded with Non-Covalently Modified Carbon Nanotubes and Graphene Nanosheets. Materials. 2026; 19(8):1569. https://doi.org/10.3390/ma19081569
Chicago/Turabian StyleRaimondo, Marialuigia, and Liberata Guadagno. 2026. "Overall Performance Enhancement of Epoxy Resins Loaded with Non-Covalently Modified Carbon Nanotubes and Graphene Nanosheets" Materials 19, no. 8: 1569. https://doi.org/10.3390/ma19081569
APA StyleRaimondo, M., & Guadagno, L. (2026). Overall Performance Enhancement of Epoxy Resins Loaded with Non-Covalently Modified Carbon Nanotubes and Graphene Nanosheets. Materials, 19(8), 1569. https://doi.org/10.3390/ma19081569






