Influence of Electrochemical Oxidation in H2SO4 and H3PO4 on the Electrochemical Behavior of Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Experimental Procedure
2.3. Ex Situ Characterization
3. Results and Discussion
3.1. Surface Morphology and Chemical Composition of Ti-6Al-4V ELI Before and After Anodic Oxidation
3.2. Evaluation of Vickers Microhardness of Ti-6Al-4V ELI Alloy Before and After Anodic Oxidation
3.3. Roughness of Ti-6Al-4V ELI Alloy Before and After Anodic Oxidation
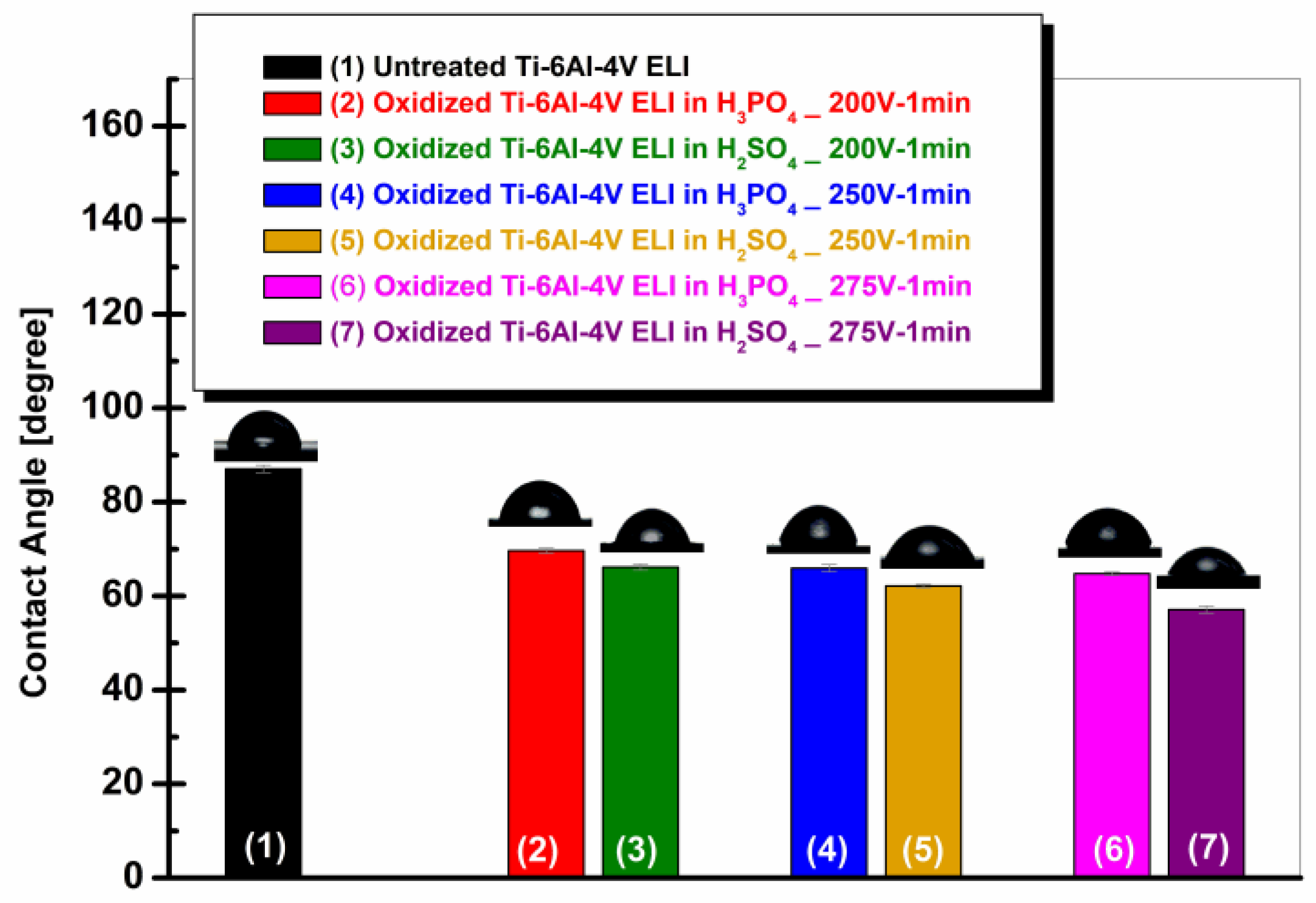
3.4. Wettability of Ti-6Al-4V ELI Alloy Before and After Anodic Oxidation
3.5. Comparative Corrosion Performance of Untreated and Electrochemically Treated Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments
3.5.1. Open Circuit Potential (OCP)
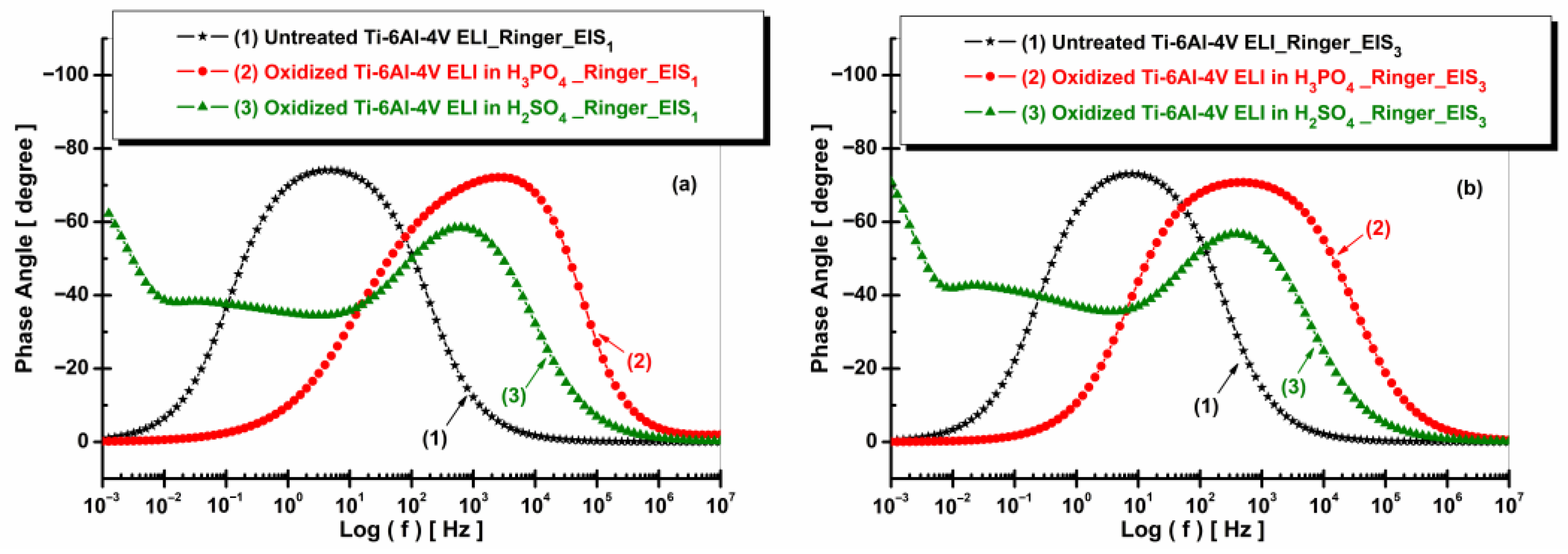
3.5.2. Electrochemical Impedance Spectroscopy (EIS)
| Parameters of the Equivalent Electrical Circuit /Units | Measurement 1 | Untreated Ti-6Al-4V ELI | Oxidized Ti-6Al-4V ELI in 1 M H3PO4 200 V-1 min | Oxidized Ti-6Al-4V ELI in 1 M H2SO4 200 V-1 min |
|---|---|---|---|---|
| Rs [Ω cm2] | EIS1 | 71.45 | 53.66 | 64.23 |
| EIS3 | 94.36 | 56.31 | 68.41 | |
| CPE-P [F/cm2] | EIS1 | 2.948 × 10−5 | 1.040 × 10−6 | 2.532 × 10−6 |
| EIS3 | 1.792 × 10−5 | 7.658 × 10−7 | 4.508 × 10−6 | |
| α | EIS1 | 0.872 | 0.912 | 0.804 |
| EIS3 | 0.873 | 0.822 | 0.784 | |
| Rp [MΩ cm2] | EIS1 | 45.263 | 0.00826 | 2.969 |
| EIS3 | 37.977 | 3.140 | 3.040 | |
| CPE-Tox [F/cm2] | EIS1 | - | 6.382 × 10−8 | - |
| EIS3 | - | 2.994 × 10−9 | - | |
| α | EIS1 | - | 0.969 | - |
| EIS3 | - | 0.995 | - | |
| Rp [MΩ cm2] | EIS1 | - | 65.254 | - |
| EIS3 | - | 56.025 | - | |
| Wo-R [kΩ cm2] | EIS1 | - | - | 186.180 |
| EIS3 | - | - | 203.940 | |
| Wo-Tox [F/cm2] | EIS1 | - | - | 128.6 |
| EIS3 | - | - | 144.3 | |
| Wo-α | EIS1 | - | - | 0.441 |
| EIS3 | - | - | 0.488 |
| Parameters of the Equivalent Electrical Circuit /Units | Measurement 1 | Untreated Ti-6Al-4V ELI | Oxidized Ti-6Al-4V ELI in 1 M H3PO4 200 V-1 min | Oxidized Ti-6Al-4V ELI in 1 M H2SO4 200 V-1 min |
|---|---|---|---|---|
| Rs [Ω cm2] | EIS1 | 68.96 | 68.86 | 67.89 |
| EIS3 | 82.24 | 79.68 | 152.3 | |
| CPE-P [F/cm2] | EIS1 | 4.172 × 10−5 | 2.545 × 10−7 | 3.410 × 10−6 |
| EIS3 | 6.493 × 10−5 | 2.625 × 10−5 | 2.619 × 10−5 | |
| α | EIS1 | 0.890 | 0.966 | 0.915 |
| EIS3 | 0.887 | 0.825 | 0.886 | |
| Rp [MΩ cm2] | EIS1 | 24.575 | 1.218 | 1.740 |
| EIS3 | 36.632 | 0.525 | 3.191 | |
| CPE-Tox [F/cm2] | EIS1 | - | 9.508 × 10−7 | - |
| EIS3 | - | 9.177 × 10−7 | - | |
| α | EIS1 | - | 0.702 | - |
| EIS3 | - | 0.704 | - | |
| Rp [MΩ cm2] | EIS1 | - | 30.276 | - |
| EIS3 | - | 51.230 | - | |
| Wo-R [kΩ cm2] | EIS1 | - | - | 175.880 |
| EIS3 | - | - | 158.610 | |
| Wo-Tox [F/cm2] | EIS1 | - | - | 103.3 |
| EIS3 | - | - | 72.9 | |
| Wo-α | EIS1 | - | - | 0.544 |
| EIS3 | - | - | 0.560 |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Benea, L.; Neaga, V.; Bogatu, N.; Axente, E.R. Monitoring and Evaluation of Corrosion at the Interface of Zirconium Alloy Biomaterials Under Simulated Oxidative Biological Environment. Int. J. Mol. Sci. 2025, 26, 10537. [Google Scholar] [CrossRef] [PubMed]
- Benea, L.; Bogatu, N.; Neaga, V.; Axente, E.R. Electrochemical Oxidation of Ti-Grad 23 Alloy for Biomedical Applications: Influence of TiO2 Formation on Their Morphology, Composition, Wettability, and Chemical Corrosion. Molecules 2026, 31, 251. [Google Scholar] [CrossRef] [PubMed]
- Barjaktarević, D.R.; Cvijović-Alagić, I.L.; Dimić, I.D.; Đokić, V.R.; Rakin, M.P. Anodization of Ti-Based Materials for Biomedical Applications: A Review. Metall. Mater. Eng. 2016, 22, 129–143. [Google Scholar] [CrossRef]
- Marin, E.; Lanzutti, A. Biomedical Applications of Titanium Alloys: A Comprehensive Review. Materials 2024, 17, 114. [Google Scholar] [CrossRef]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Sul, Y.T.; Johansson, C.; Petronis, S.; Krozer, A.; Jeong, Y.; Wennerberg, A.; Albrektsson, T. Characteristics of the surface oxides on turned and electrochemically oxidized pure titanium implants up to dielectric breakdown:: The oxide thickness, micropore configurations, surface roughness, crystal structure and chemical composition. Biomaterials 2002, 23, 491–501. [Google Scholar] [CrossRef]
- Benea, L.; Ravoiu, A.; Neaga, V.; Axente, E.R. Using Applied Electrochemistry to Obtain Nanoporous TiO2 Films on Ti6Al4V Implant Alloys and Their Preclinical In Vitro Characterization in Biological Solutions. Coatings 2023, 13, 614. [Google Scholar] [CrossRef]
- Niinomi, M. Mechanical properties of biomedical titanium alloys. Mater. Sci. Eng. A 1998, 243, 231–236. [Google Scholar] [CrossRef]
- Niinomi, M.; Nakai, M.; Hieda, J. Development of new metallic alloys for biomedical applications. Acta Biomater. 2012, 8, 3888–3903. [Google Scholar] [CrossRef]
- Sidambe, A.T. Biocompatibility of Advanced Manufactured Titanium Implants—A Review. Materials 2014, 7, 8168–8188. [Google Scholar] [CrossRef]
- Revathi, A.; Borrás, A.D.; Muñoz, A.I.; Richard, C.; Manivasagam, G. Degradation mechanisms and future challenges of titanium and its alloys for dental implant applications in oral environment. Mater. Sci. Eng. C 2017, 76, 1354–1368. [Google Scholar] [CrossRef]
- Pan, J.; Thierry, D.; Leygraf, C. Electrochemical impedance spectroscopy study of the passive oxide film on titanium for implant application. Electrochim. Acta 1996, 41, 1143–1153. [Google Scholar] [CrossRef]
- Jáquez-Muñoz, J.M.; Gaona-Tiburcio, C.; Méndez-Ramírez, C.T.; Baltazar-Zamora, M.Á.; Estupinán-López, F.; Bautista-Margulis, R.G.; Cuevas-Rodríguez, J.; Flores-De los Rios, J.P.; Almeraya-Calderón, F. Corrosion of Titanium Alloys Anodized Using Electrochemical Techniques. Metals 2023, 13, 476. [Google Scholar] [CrossRef]
- Jáquez-Muñoz, J.M.; Gaona-Tiburcio, C.; Chacón-Nava, J.; Cabral-Miramontes, J.; Nieves-Mendoza, D.; Maldonado-Bandala, E.; Delgado, A.D.; Flores-De los Rios, J.P.; Bocchetta, P.; Almeraya-Calderón, F. Electrochemical Corrosion of Titanium and Titanium Alloys Anodized in H2SO4 and H3PO4 Solutions. Coatings 2022, 12, 325. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, H.; Ding, X.; Yan, Q.; Wang, L.; Ma, W. Simulation of anodizing current-time curves and morphology evolution of TiO2 nanotubes anodized in electrolytes with different NH4F concentrations. Electrochim. Acta 2015, 176, 1083–1091. [Google Scholar] [CrossRef]
- Laurindo, C.A.H.; Torres, R.D.; Mali, S.A.; Gilbert, J.L.; Soares, P. Incorporation of Ca and P on anodized titanium surface: Effect of high current density. Mater. Sci. Eng. C 2014, 37, 223–231. [Google Scholar] [CrossRef]
- Chamanzadeh, Z.; Noormohammadi, M.; Zahedifar, M. Self-organized and uniform TiO2 nanotube arrays with optimized NH4F concentration in electrolyte by high voltage electrochemical anodization. Mater. Res. Express 2018, 5, 55025. [Google Scholar] [CrossRef]
- Prando, D.; Branna, A.; Diamanti, M.V.; Beretta, S.; Bolzoni, F.; Ormellese, M. Electrochemical anodizing treatment to enhance localized corrosion resistance of pure titanium. J. Appl. Biomater. Funct. Mater. 2017, 15, e19–e24. [Google Scholar] [CrossRef] [PubMed]
- Sul, Y.T.; Johansson, C.B.; Jeong, Y.; Albrektsson, T. The Electrochemical Oxide Growth Behaviour on Titanium in Acid and Alkaline Electrolytes. Med. Eng. Phys. 2001, 23, 329–346. [Google Scholar] [CrossRef]
- Park, H.H.; Park, I.S.; Kim, K.S.; Jeon, W.Y.; Park, B.K.; Kim, H.S.; Bae, T.S.; Lee, M.H. Bioactive and electrochemical characterization of TiO2 nanotubes on titanium via anodic oxidation. Electrochim. Acta 2010, 55, 6109–6114. [Google Scholar] [CrossRef]
- Ribeiro, B.; Offoiach, R.; Rahimi, E.; Salatin, E.; Lekka, M.; Fedrizzi, L. On Growth and Morphology of TiO2 Nanotubes on Ti6Al4V by Anodic Oxidation in Ethylene Glycol Electrolyte: Influence of Microstructure and Anodization Parameters. Materials 2021, 14, 2540. [Google Scholar] [CrossRef]
- Lestari, F.P.; Sari, Y.R.; Rokhmanto, F.; Asmaria, T.; Pramono, A.W. Surface modification of Ti-6Al-4V alloy by anodization technique at low potential to produce oxide layer. J. Electron. Electromed. Eng. Med. Inform. 2020, 2, 93–102. [Google Scholar] [CrossRef]
- Diamanti, M.V.; Del Curto, B.; Pedeferri, M. Anodic oxidation of titanium: From technical aspects to biomedical applications. J. Appl. Biomater. Biomech. 2011, 9, 55–69. [Google Scholar] [CrossRef]
- Teng, F.Y.; Tai, I.C.; Wang, M.W.; Wang, Y.J.; Hung, C.C.; Tseng, C.C. The structures, electrochemical and cell performance of titania films formed on titanium by micro-arc oxidation. J. Taiwan Inst. Chem. Eng. 2014, 45, 1331–1337. [Google Scholar] [CrossRef]
- Ohtsu, N.; Yokoi, K. Surface structure and photocatalytic performance of an anodic oxide layer fabricated on titanium in a nitrate/ethylene glycol electrolyte with different treatment durations. Surf. Coat. Technol. 2016, 294, 109–114. [Google Scholar] [CrossRef]
- Uttiya, S.; Contarino, D.; Prandi, S.; Carnasciali, M.M.; Gemme, G.; Mattera, L.; Rolandi, R.; Canepa, M.; Cavalleri, O. Anodic oxidation of titanium in sulphuric acid and phosphoric acid electrolytes. J. Mater. Sci. Nanotechnol. 2014, 1, S106. [Google Scholar] [CrossRef]
- Wilk, M.; Klimek, L. Oxide layers on titanium obtained by anodizing in orthophosphoric acid. Arch. Mater. Sci. Eng. 2018, 94, 11–17. [Google Scholar] [CrossRef]
- Benea, L.; Celis, J.P. Reactivity of porous titanium oxide film and chitosan layer electrochemically formed on Ti-6Al-4V alloy in biological solution. Surf. Coat. Technol. 2018, 354, 145–152. [Google Scholar] [CrossRef]
- Cui, X.; Kim, H.-M.; Kawashita, M.; Wang, L.; Xiong, T.; Kokubo, T.; Nakamura, T. Preparation of bioactive titania films on titanium metal via anodic oxidation. Dent. Mater. 2009, 25, 80–86. [Google Scholar] [CrossRef]
- Alipal, J.; Lee, T.C.; Koshy, P.; Abdullah, H.Z.; Idris, M.I. Evolution of anodised titanium for implant applications. Heliyon 2021, 7, e07408. [Google Scholar] [CrossRef]
- Yang, B.; Gan, L.; Qu, Y.; Yue, C. Anti-inflammatory properties of bioactive titanium metals. J. Biomed. Mater. Res. A 2010, 94, 700–705. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Biao, M.; Chen, Y.; Xie, M.; Yang, B. Regulating the osteogenic function of rhBMP-2 by different titanium surface properties. J. Biomed. Mater. Res. A 2016, 104, 1882–1893. [Google Scholar] [CrossRef]
- Mori, Y.; Masahashi, N.; Aizawa, T. A Review of Anodized TiNbSn Alloys for Improvement in Layer Quality and Application to Orthopedic Implants. Materials 2022, 15, 5116. [Google Scholar] [CrossRef]
- Regonini, D. Anodised TiO2 Nanotubes: Synthesis, Growth Mechanism and Thermal Stability. Ph.D. Thesis, University of Bath, Bath, UK, 1 September 2008. [Google Scholar]
- Chen, S.; Gou, Y.; Wang, H.; Jian, K.; Wang, J. Preparation and characterization of high-temperature resistant ZrC–ZrB2 nanocomposite ceramics derived from single-source precursor. Mater. Des. 2017, 117, 257–264. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, D.; Gao, M.; Li, D.; Song, Y.; Jin, R.; Ma, W.; Zhu, X. Growth of anodic TiO2 nanotubes in mixed electrolytes and novel method to extend nanotube diameter. Electrochim. Acta 2015, 160, 33–42. [Google Scholar] [CrossRef]
- Katunar, M.R.; Gomez Sanchez, A.; Santos Coquillat, A.; Civantos, A.; Martinez Campos, E.; Ballarre, J.; Vico, T.; Baca, M.; Ramos, V.; Cere, S. In vitro and in vivo characterization of anodised zirconium as a potential material for biomedical applications. Mater. Sci. Eng. C 2017, 75, 957–968. [Google Scholar] [CrossRef]
- Gomez Sanchez, A.; Ballarre, J.; Orellano, J.C.; Duffó, G.; Ceré, S. Surface modification of zirconium by anodisation as material for permanent implants: In vitro and in vivo study. J. Mater. Sci. Mater. Med. 2013, 24, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Kuromoto, N.K.; Simão, R.A.; Soares, G.A. Titanium oxide films produced on commercially pure titanium by anodic oxidation with different voltages. Mater. Charact. 2007, 58, 114–121. [Google Scholar] [CrossRef]
- Shabani, M.; Zamiri, R. Effect of applied voltage and substrate preparation on surface modification of anodically oxidized titanium. J. Ovonic Res. 2014, 10, 43–53. [Google Scholar]
- Jain, S.; Williamson, R.S.; Roach, M.D. Surface characterization, shear strength, and bioactivity of anodized titanium prepared in mixed-acid electrolytes. Surf. Coat. Technol. 2017, 325, 594–603. [Google Scholar] [CrossRef]
- Tanaka, S.-i.; Fukushima, Y.; Nakamura, I.; Tanaki, T.; Jerkiewicz, G. Preparation and Characterization of Microporous Layers on Titanium by Anodization in Sulfuric Acid with and without Hydrogen Charging. ACS Appl. Mater. Interfaces 2013, 5, 3340–3347. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kushwaha, M.K. Anodization of Titanium Alloy (Grade-5) and Influence of Anodic Voltage and Electrolytic Concentration on Morphology and Aesthetic Appearance of an Oxide Layer. J. Emerg. Technol. Innov. Res. 2019, 6, 545–551. [Google Scholar]
- Chu, C.L.; Wang, R.M.; Yin, L.H.; Pu, Y.P.; Lin, P.H.; Dong, Y.S.; Chung, C.Y.; Yeung, K.W.K.; Chu, P.K. Effects of anodic oxidation in H2SO4 electrolyte on the biocompatibility of NiTi shape memory alloy. Mater. Lett. 2008, 62, 3512–3514. [Google Scholar] [CrossRef]
- Benea, L. Metode Avansate de Investigare a Materialelor; Editura Academica: Galati, Romania, 2017; pp. 1–312. [Google Scholar]
- Benea, L.; Simionescu-Bogatu, N. Reactivity and Corrosion Behaviors of Ti6Al4V Alloy Implant Biomaterial under Metabolic Perturbation Conditions in Physiological Solutions. Materials 2021, 14, 7404. [Google Scholar] [CrossRef] [PubMed]













Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Benea, L.; Bogatu, N.; Neaga, V.; Axente, E.R. Influence of Electrochemical Oxidation in H2SO4 and H3PO4 on the Electrochemical Behavior of Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments. Materials 2026, 19, 1530. https://doi.org/10.3390/ma19081530
Benea L, Bogatu N, Neaga V, Axente ER. Influence of Electrochemical Oxidation in H2SO4 and H3PO4 on the Electrochemical Behavior of Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments. Materials. 2026; 19(8):1530. https://doi.org/10.3390/ma19081530
Chicago/Turabian StyleBenea, Lidia, Nicoleta Bogatu, Veaceslav Neaga, and Elena Roxana Axente. 2026. "Influence of Electrochemical Oxidation in H2SO4 and H3PO4 on the Electrochemical Behavior of Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments" Materials 19, no. 8: 1530. https://doi.org/10.3390/ma19081530
APA StyleBenea, L., Bogatu, N., Neaga, V., & Axente, E. R. (2026). Influence of Electrochemical Oxidation in H2SO4 and H3PO4 on the Electrochemical Behavior of Ti-6Al-4V ELI Alloy in Artificial Biological Media Mimicking Physiological and Pathological Environments. Materials, 19(8), 1530. https://doi.org/10.3390/ma19081530







