Tribocorrosion Behavior of Mg Alloys on Sliding Friction in Hank’s Balanced Salt Solution
Abstract
1. Introduction
2. Experimental Procedures
2.1. Material Preparation
2.2. Vickers Hardness Measurement
2.3. Sliding Wear Test
2.4. Corrosion Volume-Loss Measurement
2.5. In Situ Tribo-Electrochemical Measurement
2.6. Microstructure Observation of Wear Track
3. Results
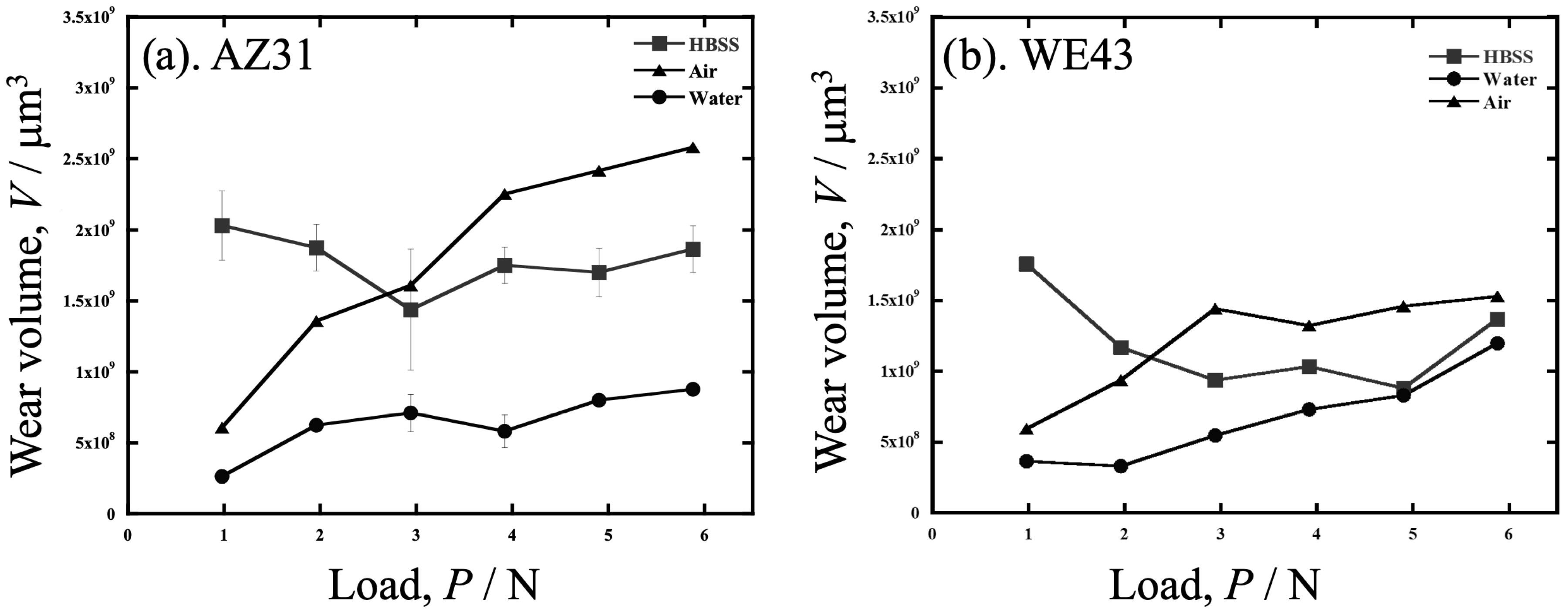
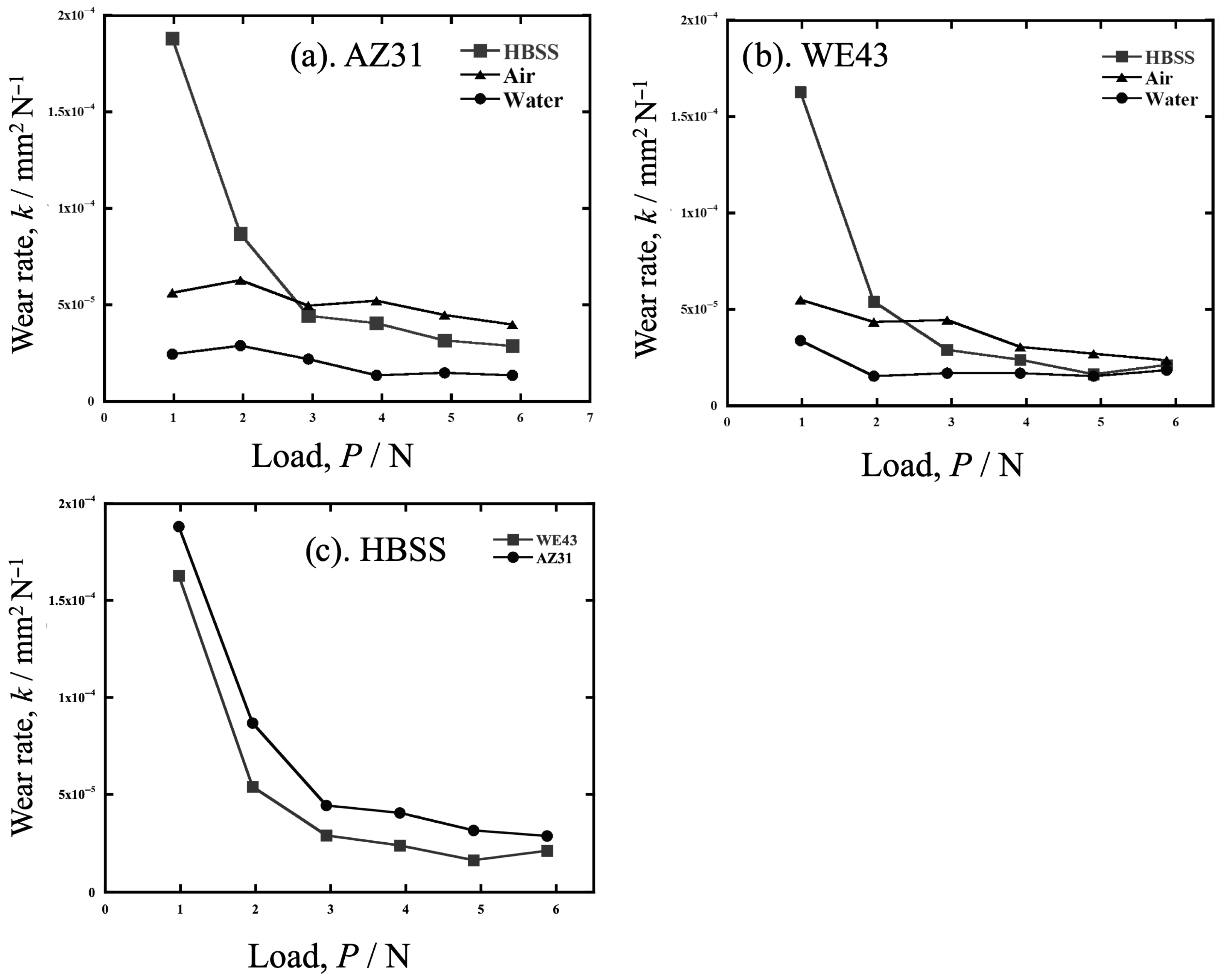
3.1. Wear Volume Loss After Sliding Wear
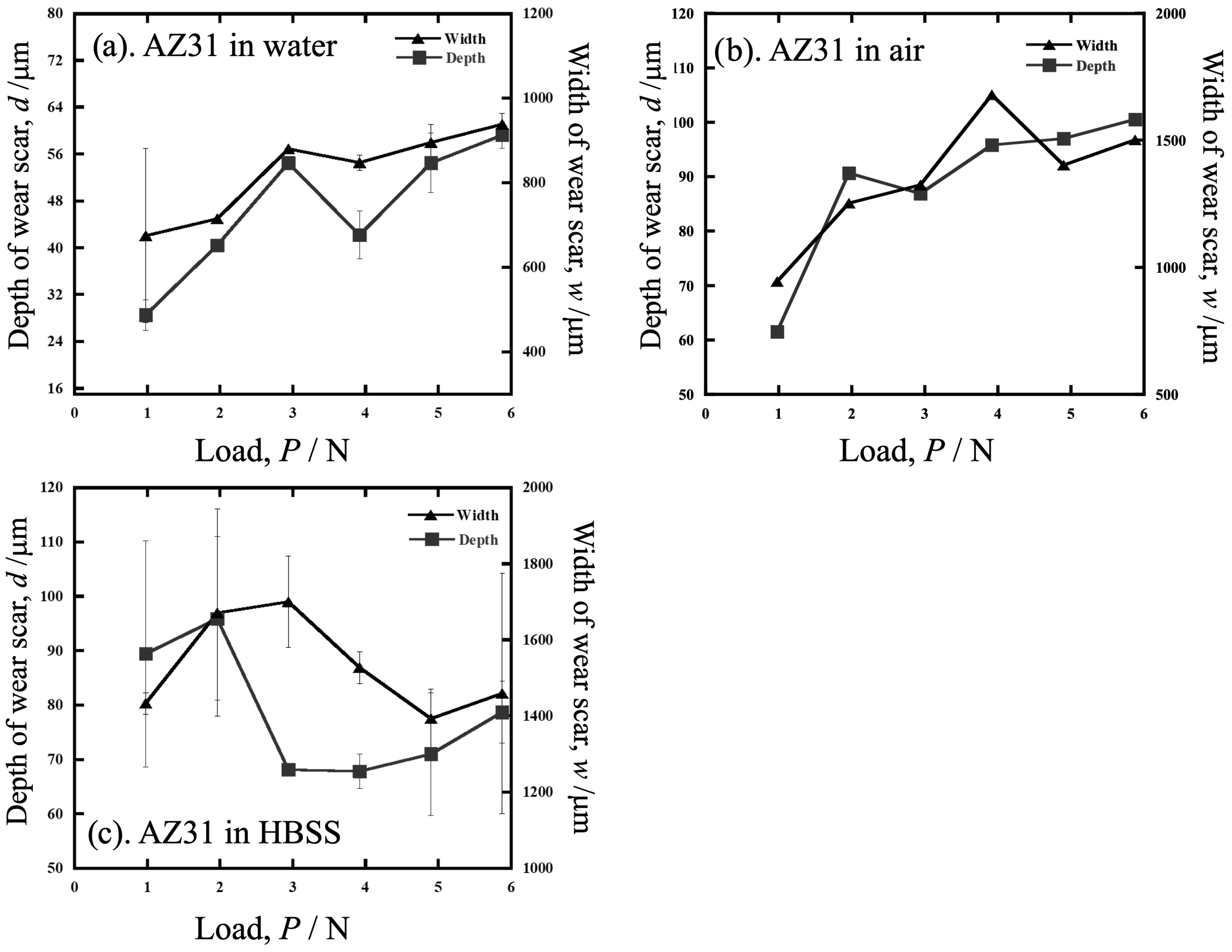
3.2. Worn Surface Observation of AZ31
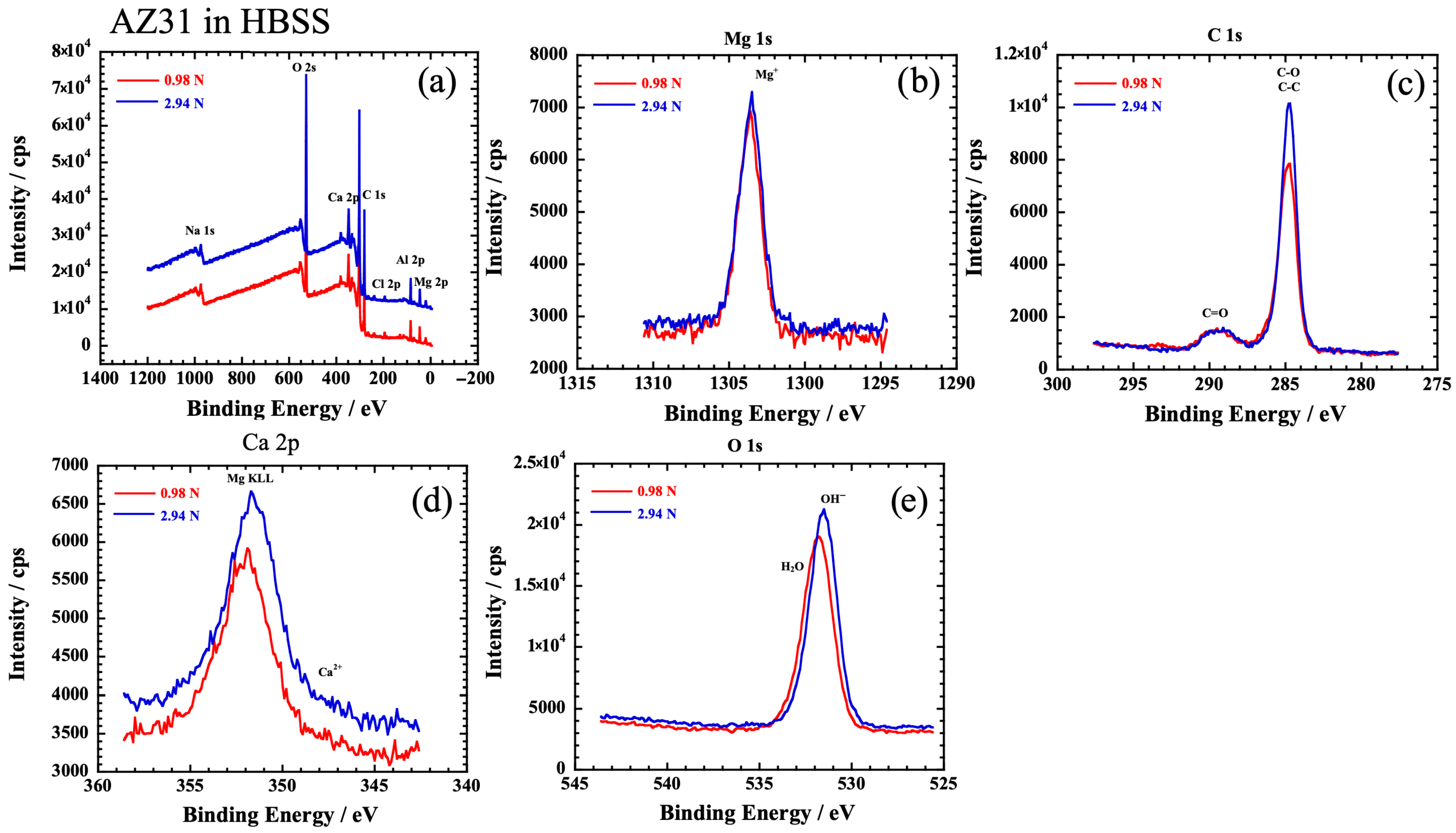
3.3. Corrosion Behavior
4. Discussion
4.1. Wear in Water
4.2. Wear in HBSS
4.2.1. Relationships Between Applied Load and Tribocorrosion
4.2.2. Lubricating Components That Most Affect Corrosive Wear
4.3. Mechanism of Tribocorrosion
5. Summary and Conclusions
- (1)
- Comparing the load dependence of wear in air, deionized water, and HBSS, the wear of both the AZ31 and WE43 alloys was greater at loads above 2 to 3 N in the order of air > HBSS > water. Below 2 to 3 N, the wear of HBSS increased and was the largest. In the comparison between the alloys, the amount of wear in water was similar at each load; however, the increase in the amount of wear with increasing load was more pronounced for AZ31, that is, AZ31 showed a stronger load dependence.
- (2)
- However, when converted into a specific wear rate, the specific wear of AZ31 and WE43 was not significantly different at any load, and the effect of the atmosphere or liquid was also small, except for HBSS.
- (3)
- For AZ31 and WE43, the specific wear rate in water was smaller than that in air and HBSS. This was due to the improved corrosion and wear resistance resulting from the formation of a protective Mg(OH)2 gel. The Cl− ions in HBSS prohibit gel formation.
- (4)
- The wear rate in HBSS increased drastically with a load decrease of less than 3 N. This result indicates that the wear in HBSS is affected by factors other than the load, running distance, and hardness.
- (5)
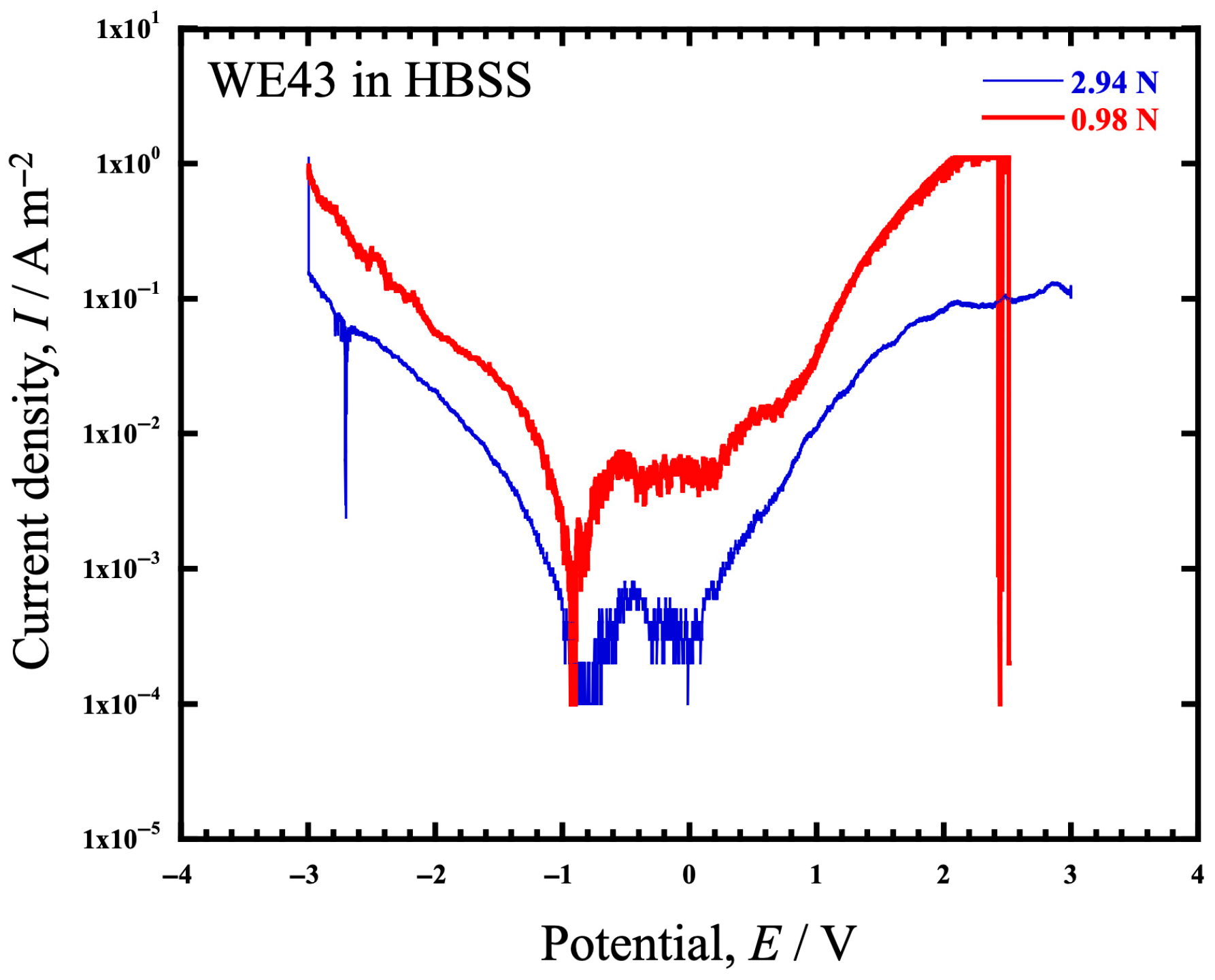
- Electrochemical measurements of WE43 in HBSS showed that the polarization behavior differed between a load of 0.98 N and 2.94 N, with a tendency for corrosion to occur more likely at 0.98 N.
- (6)
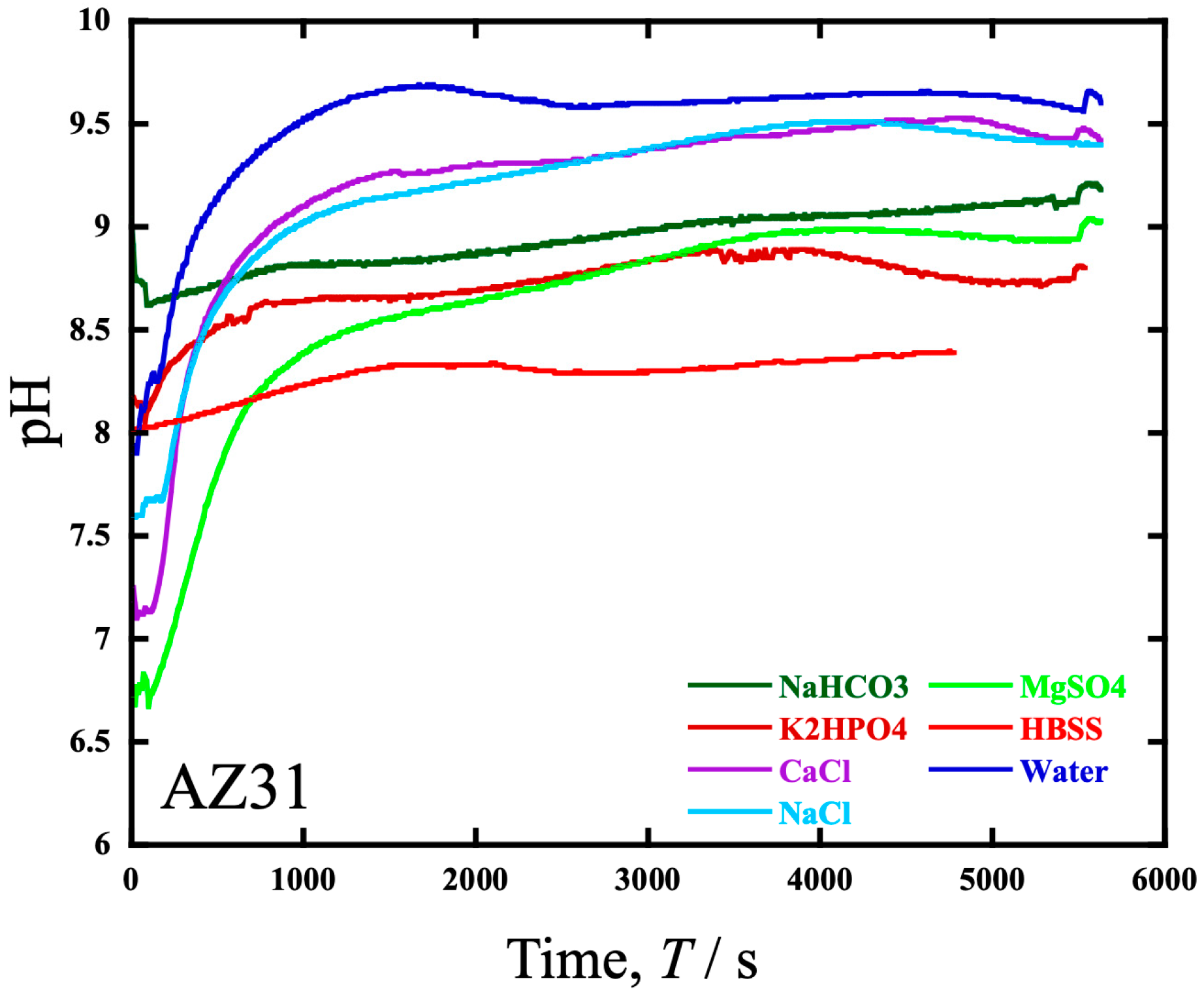
- Similar to the wear in HBSS, the wear in NaHCO3 aq. exhibited a tendency to increase in the low-load range. The amount of wear also increased under Cl− and D-glucose conditions, which significantly influenced the increase in volume loss.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yamamoto, R. Biomedical application of magnesium alloys. Jpn. Inst. Light Met. 2008, 58, 570–576. [Google Scholar] [CrossRef]
- Hiromoto, S. Corrosion evaluation of biodegradable magnesium alloys and improvement of corrosion resistance with calcium phosphate coatings. Jpn. Inst. Light Met. 2014, 64, 203–210. [Google Scholar] [CrossRef]
- Amukarimi, S.; Mozafari, M. Biodegradable magnesium-based biomaterials: An overview of challenges and opportunities. MedComm 2021, 2, 123–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, W.; Liu, J.; Wang, L.; Tang, Y.; Wang, K. A review on magnesium alloys for biomedical applications. Front. Bioeng. Biotechnol. 2022, 10, 953344. [Google Scholar] [CrossRef]
- Sezer, N.; Evis, Z.; Kayhan, S.M.; Tahmasebifar, A.; Koç, M. Review of magnesium-based biomaterials and their applications. J. Magnes. Alloys 2018, 6, 23–43. [Google Scholar] [CrossRef]
- Heublein, B. Biocorrosion of magnesium alloys: A new principle in cardiovascular implant technology. Heart 2003, 89, 651–656. [Google Scholar] [CrossRef]
- Kuwahara, H.; Al-Abdullat, Y.; Mazaki, N.; Tsutsumi, S.; Aizawa, T. Precipitation of Magnesium Apatite on Pure Magnesium Surface during Immersing in Hank’s Solution. Mater. Trans. 2001, 42, 1317–1321. [Google Scholar] [CrossRef]
- Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr. Opin. Solid State Mater. Sci. 2008, 12, 63–72. [Google Scholar] [CrossRef]
- Witte, F. The history of biodegradable magnesium implants: A review. Acta Biomater. 2010, 6, 1680–1692. [Google Scholar] [CrossRef] [PubMed]
- Waksman, R.; Pakala, R.; Baffour, R.; Seabron, R.; Hellinga, D.; Tio, F.O. Short-term effects of biocorrodible iron stents in porcine coronary arteries. J. Interv. Cardiol. 2008, 21, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Meyer-Lindenberg, A.; Wirth, C.; Windhagen, H. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 2005, 26, 3557–3563. [Google Scholar] [CrossRef]
- Yao, R.; Wang, H.; Shan, R.; Liu, L.; Zhao, Y.; Sun, Y.; Yao, X.; Huang, D.; Hang, R. Biodegradable porous Zn-1Mg-3βTCP scaffold for bone defect repair: In vitro and in vivo evaluation. J. Mater. Sci. Technol. 2023, 162, 189–202. [Google Scholar] [CrossRef]
- Sangeetha, K.; Jisha Kumari, A.V.; Venkatesan, J.; Sukumaran, A.; Aisverya, S.; Sudha, P.N. 13-Degradable metallic biomaterials for cardiovascular applications. In Fundamental Biomaterials: Metals; Balakrishnan, P., Sadasivan, S.M., Thomas, S., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 285–298. [Google Scholar]
- Khan, A.R.; Grewal, N.S.; Zhou, C.; Yuan, K.; Zhang, H.-J.; Jun, Z. Recent advances in biodegradable metals for implant applications: Exploring in vivo and in vitro responses. Results Eng. 2023, 20, 101526. [Google Scholar] [CrossRef]
- Hiromoto, S. Biodegradable Materials, Present and Prospects ~Surface Coatings for Corrosion Control of Bioabsorbable Magnesium~. Mater. Jpn. 2012, 51, 319–322. [Google Scholar] [CrossRef][Green Version]
- Wazzan, A.R.; Robinson, L.B. Elastic constants of magnesium-lithium alloys. Phys. Rev. 1967, 155, 586–594. [Google Scholar] [CrossRef]
- Slutsky, L.J.; Garland, C.W. Elastic constants of magnesium from 4.2°K to 300°K. Phys. Rev. 1957, 107, 972–976. [Google Scholar] [CrossRef]
- Hardie, D. The elastic properties of magnesium solid solutions. Acta Metall. 1971, 19, 719–723. [Google Scholar] [CrossRef]
- Shi, H.; Xu, C.; Hu, X.; Gan, W.; Wu, K.; Wang, X. Improving the Young’s modulus of Mg via alloying and compositing—A short review. J. Magnes. Alloys 2022, 10, 2009–2024. [Google Scholar] [CrossRef]
- Poyansky, L. An Insight to Magnesium (Mg). 2021. Available online: https://www.azom.com/properties.aspx?ArticleID=618 (accessed on 1 June 2024).
- Jiang, L.; Xu, F.; Xu, Z.; Chen, Y.; Zhou, X.; Wei, G.; Ge, H. Biodegradation of AZ31 and WE43 Magnesium Alloys in Simulated Body Fluid. Int. J. Electrochem. Sci. 2015, 10, 10422–10432. [Google Scholar] [CrossRef]
- Jayasathyakawin, S.; Ravichandran, M.; Naveenkumar, R.; Radhika, N.; Ismail, S.O.; Mohanavel, V. Recent advances in magnesium alloys for biomedical applications: A review. Mater. Today Commun. 2025, 42, 111239. [Google Scholar] [CrossRef]
- Meng, X.; Liu, A.; Wu, C.; Han, X.; Yang, Q.; Qiu, H.; Li, X.; Cai, M.; Duan, T.; Wang, Z. Research Progress of Magnesium Alloys and Its Alloys in Medical Applications. Int. J. Gen. Med. 2025, 18, 7101–7126. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, T. Surface treatment of titanium in medical applications. J. Jpn. Inst. Light Met. 2005, 55, 553–556. [Google Scholar] [CrossRef]
- Yamada, R.; Hosaka, T.; Yoshihara, S.; MacDonald, B.J. Effect of Equal-Channel Angular Pressing on corrosion behavior of magnesium alloy for bioabsorbable stents. J. Jpn. Inst. Light Met. 2019, 69, 579–586. [Google Scholar] [CrossRef][Green Version]
- Rahman, M.; Chowdhury, M.A.; Mia, M.S.; Ali, M.R.; Rahman, A.; Ali, M.O.; Mahmud, S. Fabrication and characterization of hybrid coating on Mg–Zn–Ca Mg alloy for enhanced corrosion and degradation resistance as medical implant. Ceram. Int. 2022, 48, 23314–23324. [Google Scholar] [CrossRef]
- Ivănescu, M.C.; Munteanu, C.; Cimpoeșu, R.; Vlad, M.D.; Istrate, B.; Lupu, F.C.; Șindilar, E.V.; Vlasa, A.; Stan, C.I.; Ivănescu, M.L.; et al. The Influence of Zn and Ca Addition on the Microstructure, Mechanical Properties, Cytocompatibility, and Electrochemical Behavior of WE43 Alloy Intended for Orthopedic Applications. Medicina 2025, 61, 1271. [Google Scholar] [CrossRef]
- Feyerabend, F.; Fischer, J.; Holtz, J.; Witte, F.; Willumeit, R.; Drücker, H.; Vogt, C.; Hort, N. Evaluation of short-term effects of rare earth and other elements used in magnesium alloys on primary cells and cell lines. Acta Biomater. 2010, 6, 1834–1842. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, A.; Wu, J.; Guo, S.; Sun, Q. Application and Perspectives: Magnesium Materials in Bone Regeneration. ACS Biomater. Sci. Eng. 2024, 10, 3514–3527. [Google Scholar] [CrossRef]
- Dong, J.; Lin, T.; Shao, H.; Wang, H.; Wang, X.; Song, K.; Li, Q. Advances in degradation behavior of biomedical magnesium alloys: A review. J. Alloys Compd. 2022, 908, 164600. [Google Scholar] [CrossRef]
- Zhai, C.; Dai, C.Y.; Lv, X.; Shi, B.; Li, Y.R.; Yang, Y.; Fan, D.; Lee, E.-S.; Sun, Y.; Jiang, H.B. Fluoride Coatings on Magnesium Alloy Implants. Bioinorg. Chem. Appl. 2022, 2022, 7636482. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.; Tan, P.-H.; Zhao, Z.-H.; Cui, L.-Y.; Kannan, M.B.; Li, S.-Q.; Liu, C.-B.; Zou, Y.-H.; Zhang, F.; Chen, Z.-Y.; et al. In vitro degradation and multi-antibacterial mechanisms of β-cyclodextrin@curcumin embodied Mg(OH)2/MAO coating on AZ31 magnesium alloy. J. Mater. Sci. Technol. 2023, 132, 179–192. [Google Scholar] [CrossRef]
- Zhang, Z.-Y.; An, Y.-L.; Wang, X.-S.; Cui, L.-Y.; Li, S.-Q.; Liu, C.-B.; Zou, Y.-H.; Zhang, F.; Zeng, R.-C. In vitro degradation, photo-dynamic and thermal antibacterial activities of Cu-bearing chlorophyllin-induced Ca–P coating on magnesium alloy AZ31. Bioact. Mater. 2022, 18, 284–299. [Google Scholar] [CrossRef]
- Jiang, P.; Hou, R.; Chen, T.; Bai, L.; Li, J.; Zhu, S.; Wang, L.; Willumeit-Römer, R.; Guan, S. Enhanced degradation performance and promoted bone regeneration of novel CaCO3-based hybrid coatings on magnesium alloy as bioresorbable orthopedic implants. Chem. Eng. J. 2023, 467, 143460. [Google Scholar] [CrossRef]
- Luo, J.; Ajami, S.; Yu, H.-M.; Ma, S.; Chen, S.-M.; Zhou, F.; Wang, P.-W.; Yao, X.-D.; Liu, C. Fretting corrosion of screws contribute to the fixation failure of the femoral neck: A case report. Biosurface Biotribol. 2019, 5, 118–123. [Google Scholar] [CrossRef]
- Brown, S.A.; Simpson, J.P. Crevice and fretting corrosion of stainless-steel plates and screws. J. Biomed. Mater. Res. 1981, 15, 867–878. [Google Scholar] [CrossRef]
- Li, W.; Li, N.; Zheng, Y.; Yuan, G. Fretting properties of biodegradable Mg-Nd-Zn-Zr alloy in air and in Hank’s solution. Sci. Rep. 2016, 6, 35803. [Google Scholar] [CrossRef]
- Mathew, M.T.; Srinivasa Pai, P.; Pourzal, R.; Fischer, A.; Wimmer, M.A. Significance of Tribocorrosion in Biomedical Applications: Overview and Current Status. Adv. Tribol. 2009, 2009, 250986. [Google Scholar] [CrossRef]
- Akiyama, E.; Maruyama, N. Fretting Corrosion Fatigue of Biomaterials. Zair.-Kankyo 2015, 63, 378. [Google Scholar] [CrossRef][Green Version]
- Mishina, H.; Hase, A. Effect of the adhesion force on the equation of adhesive wear and the generation process of wear elements in adhesive wear of metals. Wear 2019, 432–433, 202936. [Google Scholar] [CrossRef]
- Kawamoto, M.; Yamamoto, T.; Okabayashi, K. On the Law of Abrasive Wear of Cast Iron (Relationship to the Holm’s Law of Wear). J. Jpn. Inst. Met. 1973, 37, 633–638. [Google Scholar] [CrossRef][Green Version]
- Archard, J.F. Contact and Rubbing of Flat Surfaces. J. Appl. Phys. 1953, 24, 981–988. [Google Scholar] [CrossRef]
- Tunold, R.; Holtan, H.; Berge, M.B.H.; Lasson, A.; Steen-Hansen, R. The corrosion of magnesium in aqueous solution containing chloride ions. Corros. Sci. 1977, 17, 353–365. [Google Scholar] [CrossRef]
- Hiromoto, S. Corrosion Behaviour of Magnesium Alloys for Biomedical Use. Zair.-Kankyo 2014, 63, 371–377. [Google Scholar] [CrossRef][Green Version]
- Hanawa, T. Materials for metallic stents. J. Artif. Organs 2009, 12, 73–79. [Google Scholar] [CrossRef]
- Hara, N.; Kobayashi, Y.; Kagaya, D.; Akao, N. Formation and breakdown of surface films on magnesium and its alloys in aqueous solutions. Corros. Sci. 2007, 49, 166–175. [Google Scholar] [CrossRef]
- Zeng, R.-C.; Li, X.-T.; Li, S.-Q.; Zhang, F.; Han, E.-H. In vitro degradation of pure Mg in response to glucose. Sci. Rep. 2015, 5, 13026. [Google Scholar] [CrossRef]
- Feliu, S.; Samaniego, A.; El-Hadad, A.A.; Llorente, I. The effect of NaHCO3 treatment time on the corrosion resistance of commercial magnesium alloys AZ31 and AZ61 in 0.6M NaCl solution. Corros. Sci. 2013, 67, 204–216. [Google Scholar] [CrossRef]
- Al-Abdullat, Y.; Tsutsumi, S.; Nakajima, N.; Ohta, M.; Kuwahara, H.; Ikeuchi, K. Surface Modification of Magnesium by NaHCO3 and Corrosion Behavior in Hank’s Solution for New Biomaterial Applications. Mater. Trans. 2001, 42, 1777–1780. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Tokuda, M.; Inaba, C.; Iwasaki, G.; Machino, H.; Deguchi, N. Plastic Deformation of AZ31 Magnesium Alloy under Various Temperature Conditions. J. Soc. Mater. Sci. Jpn. 2008, 57, 688–695. [Google Scholar] [CrossRef][Green Version]
- Gu, R.J.; Yang, H.; Zhan, M.; Li, H.; Li, H.W. Research on the springback of thin-walled tube NC bending based on the numerical simulation of the whole process. Comput. Mater. Sci. 2008, 42, 537–549. [Google Scholar] [CrossRef]
- Yang, H.; Huang, L.; Zhan, M. Coupled thermo-mechanical FE simulation of the hot splitting spinning process of magnesium alloy AZ31. Comput. Mater. Sci. 2010, 47, 857–866. [Google Scholar] [CrossRef]
- Suzuki, K.; Chino, Y.; Huang, X.; Mabuchi, M. Elastic and Damping Properties of AZ31 Magnesium Alloy Sheet Processed by High-Temperature Rolling. Mater. Trans. 2011, 52, 2040–2044. [Google Scholar] [CrossRef][Green Version]
- Giraud, S.; Canel, J. Young’s modulus of some SOFCs materials as a function of temperature. J. Eur. Ceram. Soc. 2008, 28, 77–83. [Google Scholar] [CrossRef]
- Fogaing, E.Y.; Lorgouilloux, Y.; Huger, M.; Gault, C.P. Young’s modulus of zirconia at high temperature. J. Mater. Sci. 2006, 41, 7663–7666. [Google Scholar] [CrossRef]









| Al | Zn | Mn | Fe | Si | Cu | Ni | Mg | |
|---|---|---|---|---|---|---|---|---|
| mass% | 2.76 | 0.84 | 0.2708 | 0.0024 | 0.0116 | 0.0014 | 0.0007 | bal. |
| Zn + Ag | Y | Cu | Mn | Fe | Nd | Zr | RE | Mg | |
|---|---|---|---|---|---|---|---|---|---|
| mass% | 0.03 | 4.00 | 0.002 | 0.01 | 0.001 | 2.3 | 0.48 | 1.1 | bal. |
| Test Load (N) | Ecorr (V) | Icorr (A/m2) | Ip (A/m2) | Epit (V) |
|---|---|---|---|---|
| 0.98 | −0.929 | 5.58 × 10−4 | 5.21 × 10−3 | 0.218 |
| 2.94 | −0.853 | 5.90 × 10−5 | 3.44 × 10−4 | 0.083 |
| O | Na | Mg | P | Ca | Y | Nd | C | |
|---|---|---|---|---|---|---|---|---|
| mol% | 25.74 | nd | 57.37 | 1.59 | nd | 4.03 | 2.1 | 9.17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Miura, E.; Shiraishi, C.; Hiromoto, S. Tribocorrosion Behavior of Mg Alloys on Sliding Friction in Hank’s Balanced Salt Solution. Materials 2026, 19, 1513. https://doi.org/10.3390/ma19081513
Miura E, Shiraishi C, Hiromoto S. Tribocorrosion Behavior of Mg Alloys on Sliding Friction in Hank’s Balanced Salt Solution. Materials. 2026; 19(8):1513. https://doi.org/10.3390/ma19081513
Chicago/Turabian StyleMiura, Eri, Chihiro Shiraishi, and Sachiko Hiromoto. 2026. "Tribocorrosion Behavior of Mg Alloys on Sliding Friction in Hank’s Balanced Salt Solution" Materials 19, no. 8: 1513. https://doi.org/10.3390/ma19081513
APA StyleMiura, E., Shiraishi, C., & Hiromoto, S. (2026). Tribocorrosion Behavior of Mg Alloys on Sliding Friction in Hank’s Balanced Salt Solution. Materials, 19(8), 1513. https://doi.org/10.3390/ma19081513







