3.1. Characterization of Raw Materials
This subsection presents the results of the microstructural and phase analyses of quartz sand from the Gorbunovo deposit (West Kazakhstan region), which was used as the primary silica-containing raw material in the control glass composition.
Figure 1 shows the SEM image of the quartz sand surface. The quartz grains exhibit a natural morphology with irregular, angular edges and minor traces of surface wear caused by natural mechanical and chemical erosion processes. The surface appears relatively clean, with no significant foreign inclusions, indicating a low level of contamination and good raw material quality.
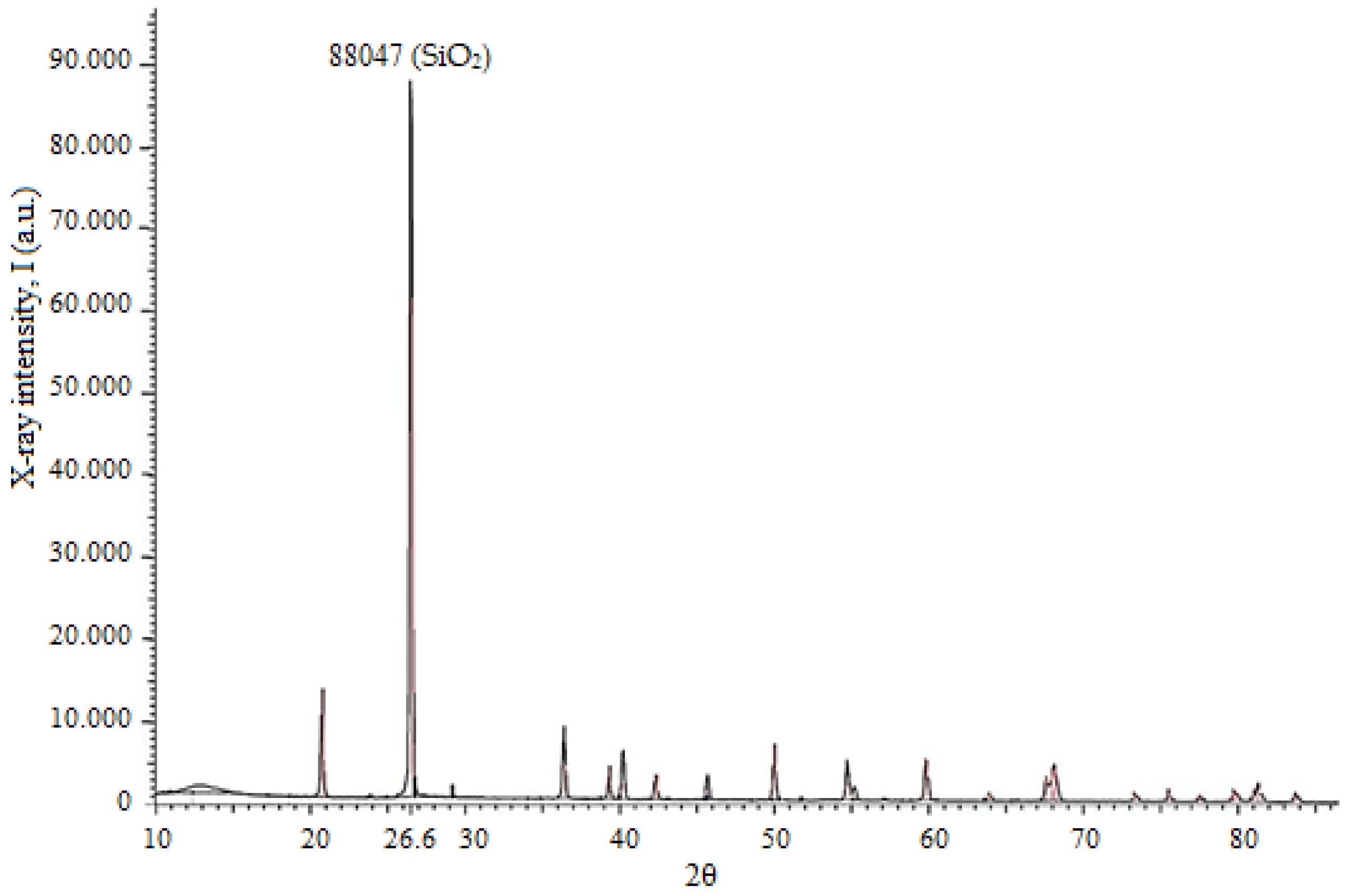
The phase composition of the quartz sand was further investigated using X-ray diffraction (XRD) analysis (
Figure 2).
The XRD pattern (
Figure 2) confirms that the sample is predominantly composed of quartz (SiO
2). The characteristic diffraction peak observed at 2θ ≈ 26.6° corresponds to the (101) crystallographic plane of quartz. The high intensity and sharpness of this peak indicate a high degree of crystallinity and the predominance of quartz in the sample. Minor low-intensity reflections may correspond to trace secondary phases or impurities; however, their significantly lower intensity confirms the high purity and structural homogeneity of the material.
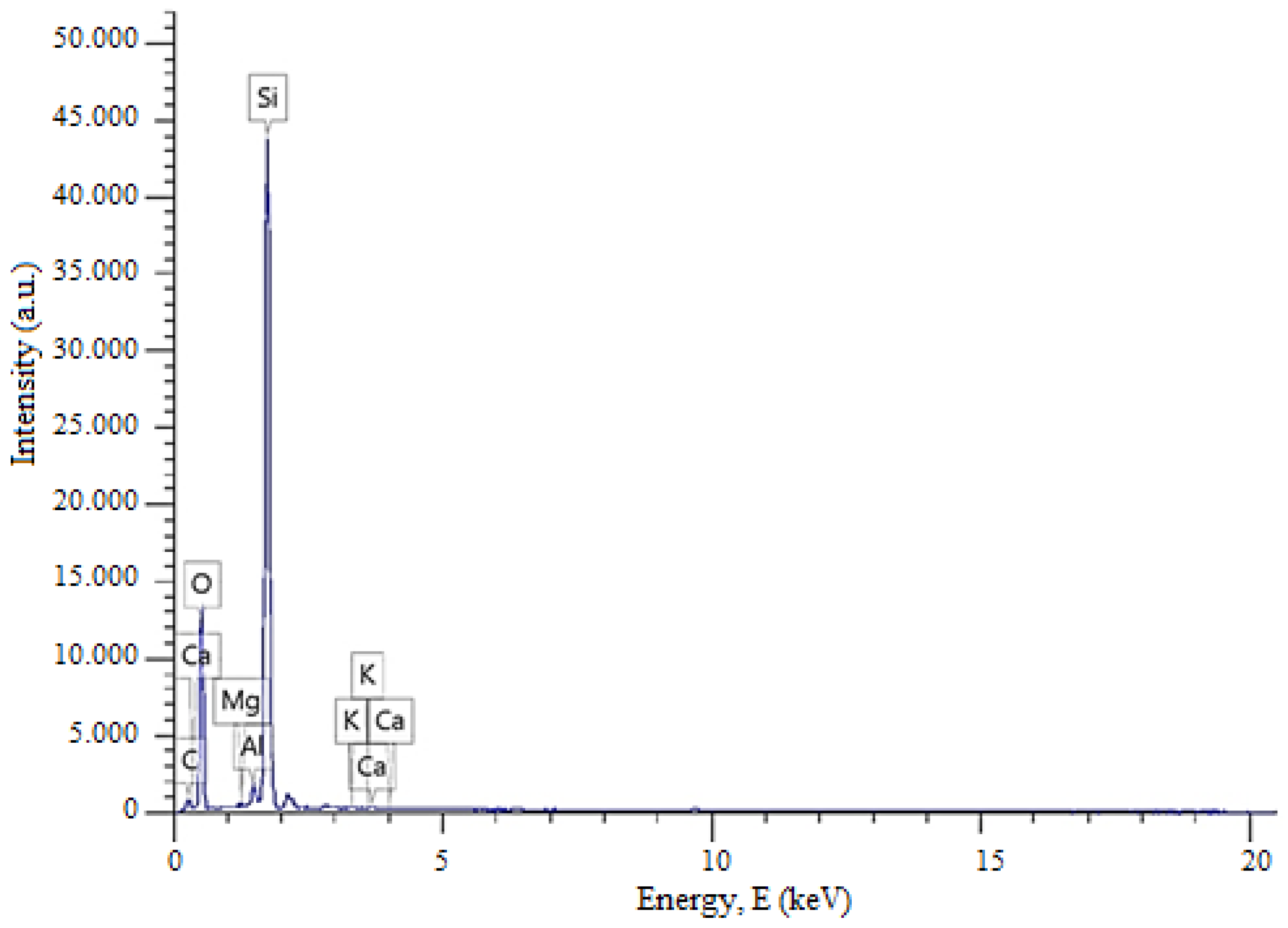
Figure 3 presents the energy-dispersive X-ray spectroscopy (EDS) analysis of quartz sand from the Gorbunovo deposit in the West Kazakhstan region of the Republic of Kazakhstan. The EDS results further confirm the elemental composition and high purity of the sample, consistent with the XRD analysis.
The graph shows the elemental composition of the studied sample, where the peak heights reflect the relative concentrations of the elements, and the peak energies are characteristic of specific chemical elements. In this case, silicon and oxygen exhibit the highest peaks, confirming the quartz nature of the material. The quantitative elemental composition of quartz sand from the Gorbunovo deposit in the West Kazakhstan region of the Republic of Kazakhstan is presented in
Table 2.
The XRD and EDS analyses indicate that the studied quartz sand sample predominantly consists of silicon and oxygen, uniformly distributed throughout the structure. Minor elements such as Mg, Al, K, and Ca are present in trace amounts, likely due to localized inclusions of clay minerals or aluminosilicates. This reflects the high purity and structural homogeneity of the material. The relatively high carbon content detected by EDS (
Table 2) should not be considered part of the intrinsic composition of quartz sand. The FTIR spectrum of the studied sample (
Figure 4), recorded in the range of 4000–400 cm
−1, is characterized by absorption bands corresponding to Si–O–Si vibrations typical of silica-based materials. For improved visualization and more accurate band assignment, the spectral region below 1500 cm
−1 is presented in an enlarged view (
Figure 4).
The most intense absorption band observed at approximately 1080–1100 cm−1 is attributed to the asymmetric stretching vibrations of Si–O–Si bonds, which is characteristic of silica. Additional bands located at around 790–800 cm−1 and 460–470 cm−1 correspond to the symmetric stretching and bending vibrations of Si–O–Si bonds, respectively. These bands are typical of crystalline silicon dioxide (quartz phase), indicating the presence of a crystalline component in the sample.
No strong or well-defined absorption bands related to carbonate groups (typically observed in the 1400–1500 cm−1 region) are present in the spectrum. A weak feature observed near ~1530 cm−1 is more likely associated with minor surface contamination or residual organic matter rather than carbonate minerals.
Therefore, the carbon detected by EDS analysis is unlikely to originate from carbonate phases. Instead, it is attributed to loss on ignition (LOI), including surface contamination, residual organic matter (e.g., humic substances), and possible carbon coating applied during SEM–EDS analysis. Accordingly, carbon is not considered a structural component of the material.
These band assignments are consistent with previously reported FTIR spectra of silica and crystalline quartz [
38].
In contrast, the FTIR spectra of the synthesized borosilicate glasses exhibit additional absorption bands associated with Si–O–Si and B–O vibrations, confirming the formation of a borosilicate network structure. The absence of additional impurity-related bands indicates the formation of a relatively homogeneous and well-structured glass matrix.
The structural and elemental composition of chalk from the Melovye Gorki deposit (West Kazakhstan, Republic of Kazakhstan) was studied using scanning electron microscopy (SEM) and X-ray diffraction (XRD) analyses (
Figure 5 and
Figure 6).
SEM analysis revealed that the chalk has a characteristic fine-grained, granular, and porous surface structure, likely formed by aggregates of individual particles. Variations in grayscale correspond to differences in density: light areas indicate denser or heavier fragments, whereas dark areas indicate lighter or more porous regions.
The elemental composition of the chalk was further determined using energy-dispersive X-ray spectroscopy (EDS) (
Figure 6).
The EDS spectrum shows that the chalk is predominantly composed of calcium, oxygen, and carbon, consistent with calcium carbonate (CaCO
3). The detected carbon (C) signal is attributed to the carbon inherently present in calcite (CaCO
3), which is the principal mineral phase of the chalk. Minor peaks of silicon, magnesium, and aluminum indicate the presence of natural clay-silicate inclusions. The quantitative elemental composition is summarized in
Table 3.
The combined SEM and EDS results confirm that the chalk sample is predominantly composed of calcite (CaCO3) with minor natural impurities such as clay and silicate minerals. The fine-grained, porous structure observed by SEM is typical of natural chalk, and the elemental composition supports its natural origin.
As noted, diatomite represents another source of silicon dioxide. Unlike quartz sand, diatomite exhibits a higher reactivity and a porous structure, making it particularly suitable for glass production. Its predominance of amorphous SiO2, high purity, and finely dispersed structure facilitate melting reactions, reduce glass melting temperatures, and lower energy costs. Additionally, its natural origin and local availability make diatomite an economically viable and environmentally friendly raw material for the glass industry.
The X-ray diffraction (XRD) pattern of diatomite from West Kazakhstan is shown in
Figure 7. The diffractogram exhibits a broad diffuse halo in the range of 2θ ≈ 20–25°, which is characteristic of amorphous silica and confirms that the dominant phase is biogenic opal (SiO
2·nH
2O).
In addition to the amorphous phase, several distinct diffraction peaks are observed, indicating the presence of minor crystalline components. The most intense peak at 2θ ≈ 26.6° is indexed to α-quartz (SiO2), corresponding to the (101) crystallographic plane (ICDD PDF No. 46-1045).
Additional reflections at approximately 2θ ≈ 20.9°, 36.5°, and 50.1° are also attributed to α-quartz, corresponding to the (100), (110), and (112) planes, respectively.
The presence of these reflections confirms that quartz is the main crystalline impurity in the studied diatomite. Minor low-intensity peaks may also be related to trace silicate or clay mineral phases typically associated with natural sedimentary deposits.
The dominance of the amorphous halo over crystalline peaks suggests that the material predominantly consists of biogenic amorphous silica with a low degree of crystallinity. Such structural characteristics are typical for natural diatomite and are highly beneficial for glass production, since amorphous silica demonstrates higher reactivity compared to crystalline quartz. The presence of minor crystalline quartz does not significantly affect the suitability of the material for borosilicate glass synthesis; however, it may slightly increase the melting temperature compared to fully amorphous silica. Overall, the XRD results confirm that West Kazakhstan diatomite is mainly composed of amorphous SiO2 with small amounts of crystalline phases, making it a promising sustainable raw material for borosilicate glass production.
The XRF data are presented as elemental equivalents recalculated from oxide compositions. The results are summarized in
Table 4. Thermal treatment induces notable changes in the elemental composition of diatomite, primarily due to the removal of physically and structurally bound water and the redistribution of oxide phases.
As shown in
Table 4, thermal treatment results in an increase in the mass fraction of silicon from 32.283% to 37.924%, indicating silica enrichment of the material. The iron content also increases significantly, from 26.008% to 35.665%, while potassium, titanium, and calcium show moderate increases. These changes suggest activation of glass-forming and modifying oxides during heating, which is beneficial for melt formation in glass production.
In contrast, a substantial decrease in aluminum content is observed, from 24.400% to 4.364%, along with reductions in copper, arsenic, and manganese. This behavior may be attributed to partial volatilization, phase transformation, or redistribution of these elements during thermal modification. The observed changes in elemental composition confirm the structural transformation of diatomite during heat treatment and indicate an increase in its reactivity and glass-forming potential, supporting its use as an effective alternative silica-containing raw material in glass batch formulations.
3.2. Characterization of Glass Products
The microstructure of the synthesized borosilicate glasses was investigated using scanning electron microscopy (SEM), and the results are presented in
Figure 8.
As shown in
Figure 8a, the glass corresponding to Composition I (control sample) exhibits a relatively dense amorphous structure; however, slight microstructural heterogeneities can be observed. Although the surface appears generally smooth, minor contrast variations suggest incomplete homogenization of the melt. This may be attributed to the limited reactivity of quartz sand, which typically requires higher temperatures and longer melting durations for complete dissolution.
In contrast, the SEM image of Composition II (
Figure 8b), which contains both quartz sand and diatomite, demonstrates a noticeably improved microstructure. The glass appears more uniform, with reduced contrast variations and fewer localized inhomogeneities. The partial replacement of quartz with diatomite enhances silica dissolution due to its amorphous nature, high porosity, and large specific surface area. As a result, more efficient interaction between silica and calcium-containing components is achieved, promoting better melt homogenization and formation of a more uniform silicate network.
A further improvement is observed in Composition III (
Figure 8c), where quartz sand is completely replaced by diatomite. The SEM micrograph reveals a highly homogeneous and dense amorphous structure with a smooth and uniform morphology. No visible crystalline inclusions, sharp boundaries, or phase separations are detected. This indicates that the use of diatomite as the primary silica source significantly enhances melt reactivity and facilitates complete glass network formation under the same processing conditions.
The observed differences in microstructure can be explained by the physicochemical properties of diatomite from West Kazakhstan, which is rich in amorphous silica and characterized by a highly porous structure. These features accelerate silica dissolution and improve mass transfer during melting, resulting in a more uniform distribution of network-forming and modifying oxides.
Overall, the comparative analysis of SEM images clearly demonstrates that increasing the proportion of diatomite (from Composition I to III) leads to progressive improvement in microstructural uniformity. The incorporation of diatomite suppresses microstructural defects, enhances melt homogenization, and promotes the formation of a consistent borosilicate glass network. These results strongly confirm that diatomite from West Kazakhstan is a highly effective and sustainable alternative silica source for borosilicate glass production, offering improved structural quality compared to conventional quartz-based systems.
To further correlate the observed microstructural features with compositional variations, the combined SEM and EDS results reveal a clear structure–property relationship. The progressive incorporation of diatomite leads to improved microstructural homogeneity, as evidenced by the transition from heterogeneous regions in Composition I to a dense and uniform amorphous matrix in Composition III. This structural refinement is directly associated with the increased presence of alkali and alkaline earth elements (Na, K, Mg) identified by EDS, which act as network modifiers. These modifiers reduce melt viscosity and enhance mass transfer during melting, promoting more complete homogenization and formation of a stable silicate network. Consequently, the improved structural uniformity is expected to result in enhanced physicochemical properties, including better thermal stability, chemical durability, and reduced defect density. Thus, the use of diatomite not only alters the composition but also positively influences the structure and performance of the resulting glass materials.
To further verify these compositional effects, energy-dispersive X-ray spectroscopy (EDS) was employed to analyze and compare the elemental composition of borosilicate glass samples synthesized from different raw material compositions. The corresponding spectra are presented in
Figure 9.
As shown in
Figure 9a, the EDS spectrum of Composition I (control sample), prepared using quartz sand from the Gorbunovo deposit and chalk from the Melovye Gorki deposit, is dominated by oxygen (O), silicon (Si), and calcium (Ca). These elements represent the primary constituents of the silicate glass network and the calcium source introduced by chalk. The pronounced Ca peak confirms the contribution of calcium-containing fluxes, while the strong Si and O signals correspond to the silicate framework. A minor carbon (C) signal is also detected, which is most likely related to surface contamination or sample preparation rather than the intrinsic composition of the glass.
The peaks initially attributed to aluminum (Al) were carefully re-evaluated. Their positions do not fully correspond to the characteristic Al Kα energy (~1.49 keV), indicating that the previous assignment may not be accurate. In particular, the peak observed near ~1.7 keV is more consistent with Si, while the feature around ~2.1 keV may arise from peak overlap or background effects. Therefore, the presence of Al in Composition I cannot be conclusively confirmed from the current EDS data and, if present, is likely at trace levels.
In Composition II (
Figure 9b), where diatomite is introduced alongside quartz sand, the elemental composition becomes more diverse. In addition to the major elements (O, Si, Ca), peaks corresponding to sodium (Na), magnesium (Mg), potassium (K), iron (Fe), and titanium (Ti) are observed. These elements originate from the natural mineral composition of diatomite and indicate the incorporation of additional oxides into the glass structure. Their presence suggests partial modification of the glass network due to the introduction of an alternative silica source.
A more pronounced effect is observed in Composition III (
Figure 9c), where diatomite completely replaces quartz sand as the primary silica source. The EDS spectrum shows relatively higher intensities of alkali (Na, K) and alkaline earth (Mg) elements compared to Composition II, confirming their increased incorporation into the glass matrix. At the same time, the dominant Si and O peaks remain, indicating preservation of the silicate network structure. The presence of Fe and Ti, although in minor amounts, further reflects the natural impurity profile of diatomite.
The incorporation of alkali (Na, K) and alkaline earth (Mg) elements plays an important role in glass formation, as these oxides act as network modifiers. Their presence reduces the viscosity and melting temperature of the glass melt, thereby enhancing melt fluidity and homogenization. Furthermore, trace transition metals such as Fe and Ti may influence the optical properties and chemical durability of the resulting glass, even at low concentrations.
Overall, the comparative EDS analysis demonstrates that increasing the proportion of diatomite (from Composition I to III) leads to a progressive enrichment of the glass composition with modifying oxides. This modification contributes to improved melting behavior and processing efficiency. The results confirm that diatomite from West Kazakhstan serves not only as a silica source but also as a multifunctional raw material that enhances the physicochemical properties of borosilicate glass. These compositional changes are expected to influence the structural ordering within the glass, which can be further investigated using XRD analysis.
X-ray diffraction (XRD) patterns of all synthesized glass samples were recorded to evaluate the presence of crystalline phases. The obtained diffraction patterns exhibit broad halos typical of amorphous materials, without sharp peaks corresponding to crystalline phases. Minor features observed in some samples were compared with standard reference patterns from the International Centre for Diffraction Data (ICDD) database; however, no matches with known crystalline phases were identified. This confirms that the glasses are fully amorphous. The results indicate that the selected synthesis conditions (temperature and duration) are sufficient to ensure complete melting of the raw materials and the formation of homogeneous glass without crystallization or phase separation.
The XRD patterns presented in
Figure 10 were analyzed to identify the crystalline phases corresponding to the weak diffraction peaks marked with (*).
For Composition I, the XRD pattern is dominated by a broad diffuse halo centered at 2θ ≈ 30–32°, which is characteristic of a predominantly amorphous borosilicate glass structure. No distinct sharp diffraction peaks are observed, indicating that crystalline phases, if present, exist only in trace amounts. This suggests that silica and other components are largely dissolved within the glass network.
In Composition II, a weak diffraction peak at approximately 2θ ≈ 26.6° is attributed to cristobalite (SiO2) (ICDD PDF No. 00-082-0512). An additional feature near 2θ ≈ 39–40° may correspond to wollastonite (CaSiO3) (ICDD PDF No. 00-043-1460), formed through the interaction between SiO2 and CaO during melting. The low intensity of these peaks indicates that these crystalline phases are present only in minor quantities.
In Composition III, similar weak diffraction peaks are observed within the 2θ ≈ 27–48° range, indicating a slightly increased degree of structural ordering. The peak near 26.6° again corresponds to cristobalite (SiO2) (ICDD PDF No. 00-082-0512), suggesting partial crystallization of amorphous silica during cooling. Peaks around 39–41° may correspond to wollastonite (CaSiO3) (ICDD PDF No. 00-043-1460), and additional weak reflections in the 44–46° range are associated with Fe- and Ti-containing oxides, such as hematite (Fe2O3) or rutile (TiO2), originating from natural impurities in diatomite.
Despite these minor crystalline phases, all samples remain predominantly amorphous, as evidenced by the dominant broad halo. This confirms that the fundamental borosilicate glass network is preserved, and that diatomite from West Kazakhstan can be effectively used as an alternative silica source without compromising the structural integrity of the glass. Overall, increasing the proportion of diatomite slightly promotes the formation of secondary crystalline phases, but the amorphous nature of the glass is retained.
3.3. Thermal, Chemical, and Water Resistance of Glass
The thermal stability, chemical resistance, and water resistance of the synthesized borosilicate glass samples were evaluated to assess the influence of glass batch composition on material properties.
The results of the heat resistance tests are presented in
Figure 11. Glass compositions I and III exhibit the highest thermal stability, withstanding temperature differences up to 160 °C without visible damage. In contrast, composition II shows lower thermal resistance, approximately 120 °C, likely due to variations in the ratio of network-forming to modifying oxides, which increase the coefficient of thermal expansion. The higher thermal stability of samples I and III indicates the formation of a stronger and more thermally stable three-dimensional network.
The chemical resistance of the glass samples was assessed by measuring the amount of alkali (Na
2O) released into water, following GOST 10134-2017. The results are summarized in
Table 5. Samples I and II are classified as hydrolytic class IV, indicating relatively low chemical resistance, while sample III shows higher resistance (class III), as evidenced by lower HCl consumption and minimal Na
2O release.
Water resistance tests at 98 °C confirmed similar trends (
Table 6). Samples I and II belong to water resistance class V, with higher XA values indicating more intense leaching. Sample III exhibited the lowest XA value, corresponding to minimal component leaching and the highest water resistance.
To ensure consistency in the chemical and water resistance tests, all synthesized glass samples were ground and sieved to a uniform particle size of 100–150 µm. This standardization eliminates surface area as a variable, allowing differences in resistance to be attributed primarily to variations in chemical composition and network structure.
The differences observed in the properties of the synthesized glasses are related not only to the use of diatomite as a silica source but also to variations in the overall chemical composition. Since the batch compositions differ in the ratio of network-forming and modifying oxides, the resulting glasses have different oxide contents, which significantly influence thermal, chemical, and water resistance. Therefore, the improved properties of diatomite-containing glasses are due to the combined effect of amorphous silica, higher reactivity, and its influence on the glass network structure, rather than solely the presence of diatomite.
The diatomite used in our study contains a significant amount of iron oxide, imparting a greenish tint to the resulting glasses. In contrast, glasses prepared from quartz sand, which contain negligible iron, appear colorless. While this work focuses on thermal and mechanical properties, this note clarifies differences in optical appearance.
The glass transition temperatures (Tg) of the synthesized borosilicate glasses were measured using differential scanning calorimetry (DSC), providing insight into structural variations among compositions. Tg values indicate the temperature at which the glass network softens, reflecting the connectivity and rigidity of the silicate–borate framework. Additionally, the coefficients of thermal expansion (CTE) were determined, showing that diatomite-containing glasses exhibit slightly higher CTE values due to minor fluxing oxides and the influence of amorphous silica on the network. Complementary FTIR spectra confirm the formation of fully connected silicate–borate networks, correlating structural features with thermal behavior.
All synthesized glasses were visually inspected and analyzed using X-ray diffraction (XRD) to verify the formation of a fully amorphous structure. No crystalline phases or phase separation were detected, and all glasses appeared homogeneous macroscopically. As noted, diatomite-containing glasses exhibited a slight greenish tint, while quartz-based glasses were colorless and transparent. No significant opacity or inhomogeneity was observed, confirming that the applied processing parameters allowed the formation of uniform borosilicate glasses.