Optimization Design of Metakaolin-Based Geopolymer Solidification for Potassium Copper Hexacyanoferrate After Cs+ Adsorption Using Response Surface Methodology
Highlights
- A quadratic regression model based on RSM optimizes the geopolymer mix for Cs immobilization with a high predictive accuracy (R2 > 0.99).
- The H2O/Na2O ratio is the most critical factor affecting compressive strength and leaching resistance, followed by Na2O/Al2O3 and SiO2/Al2O3.
- The optimal mix (Na2O/Al2O3 = 0.84, SiO2/Al2O3 = 2.8, and H2O/Na2O = 10.23) yields MPa a strength of 23.41 MPa and ultra-low 42-day Cs leaching.
- A quantitative mix design methodology for the efficient solidification of Cs-laden adsorbents using metakaolin-based geopolymer is provided.
- Microstructural analyses (SEM/XRD/FT-IR) confirm effective Cs encapsulation, supporting the geopolymer’s stability in nuclear waste treatment.
- The model offers a scalable strategy for immobilizing other radionuclides (e.g., Sr2+ and Co2+) in multi-nuclide radioactive wastewater.
Abstract
1. Introduction
2. Models and Methodology
2.1. Response Surface Methodology
2.2. Materials
2.3. Experimental Process
2.4. Analytical Methods
3. Results and Discussion
3.1. Effects of Single Factors on Cesium Leaching
3.1.1. Effect of Na2O/Al2O3 on Cesium Leaching
3.1.2. Effect of SiO2/Al2O3 on Cesium Leaching
3.1.3. Effect of H2O/Na2O on Cesium Leaching
3.2. Response Surface Analysis
3.2.1. Analysis of Response Surface Models
3.2.2. Response Surface Plots
3.3. Optimization
Macro Performance
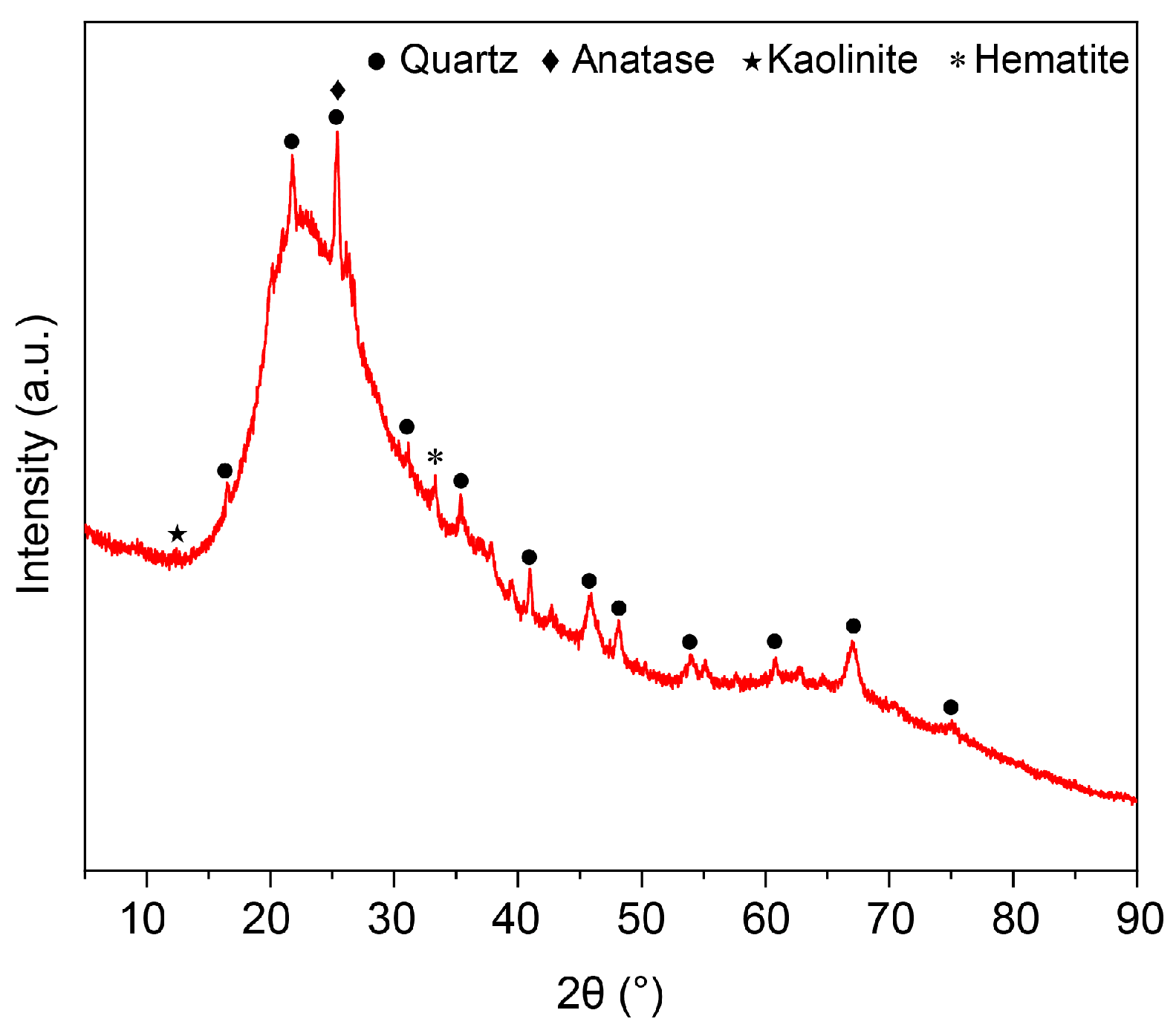
3.4. Microstructural Properties
4. Conclusions
4.1. Summary and Findings
4.2. Outlook and Future Work
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GP | Geopolymer |
| RSM | Response Surface Methodology |
| PB | Prussian Blue |
| GPMs | Geopolymer Microspheres |
| CV | Coefficient of Variation |
| CLF | Cumulative Leaching Fraction |
| MK | Metakaolin |
| XRD | X-ray Diffraction |
| SEM | Scanning Electronic Microscopy |
| EDS | Energy Dispersive X-ray Spectroscopy |
| FTIR | Fourier Transform Infrared Spectroscopy |
| ICP-MS | Inductively Coupled Plasma Mass Spectrometry |
References
- Ma, H.; Shen, M.; Tong, Y.; Wang, X. Radioactive wastewater treatment technologies: A review. Molecules 2023, 28, 1935. [Google Scholar] [CrossRef] [PubMed]
- Menyah, K.; Wolde-Rufael, Y. CO2 emissions, nuclear energy, renewable energy and economic growth in the US. Energy Policy 2010, 38, 2911–2915. [Google Scholar] [CrossRef]
- Casacuberta, N.; Masqué, P.; Garcia-Orellana, J.; Garcia-Tenorio, R.; Buesseler, K.O. 90Sr and 89Sr in seawater off Japan as a consequence of the Fukushima Dai-ichi nuclear accident. Biogeosciences 2013, 10, 3649–3659. [Google Scholar] [CrossRef]
- Kenyon, J.A.; Buesseler, K.O.; Casacuberta, N.; Castrillejo, M.; Otosaka, S.; Masqué, P.; Drysdale, J.A.; Pike, S.M.; Sanial, V. Distribution and evolution of Fukushima Dai-ichi derived 137Cs, 90Sr, and 129I in surface seawater off the coast of Japan. Environ. Sci. Technol. 2020, 54, 15066–15075. [Google Scholar] [CrossRef] [PubMed]
- Shao, L.; Wei, H.; Lei, H.; Yi, M.; Cui, X.; Wei, Y.; Wang, K. In situ immobilization properties and mechanism of geopolymer microspheres after adsorbing Sr2+ and Cs+ (Sr/Cs@GPMs). Ceram. Int. 2023, 49, 10807–10821. [Google Scholar] [CrossRef]
- Yaqub, M.; Mee-Ngern, L.; Lee, W. Cesium adsorption from an aqueous medium for environmental remediation: A comprehensive analysis of adsorbents, sources, factors, models, challenges, and opportunities. Sci. Total Environ. 2024, 950, 175368. [Google Scholar] [CrossRef]
- Kim, H.; Seon, J.; Yoon, S.; Bae, S.; Choung, S.; Hwang, Y. Roll-to-roll production of a cellulose filter with immobilized Prussian blue for 137Cs adsorption. J. Environ. Chem. Eng. 2020, 8, 104273. [Google Scholar] [CrossRef]
- Lee, I.; Park, C.W.; Yoon, S.S.; Yang, H.-M. Facile synthesis of copper ferrocyanide-embedded magnetic hydrogel beads for the enhanced removal of cesium from water. Chemosphere 2019, 224, 776–785. [Google Scholar] [CrossRef]
- Avramenko, V.; Bratskaya, S.; Zheleznov, V.; Sheveleva, I.; Voitenko, O.; Sergienko, V. Colloid stable sorbents for cesium removal: Preparation and application of latex particles functionalized with transition metals ferrocyanides. J. Hazard. Mater. 2011, 186, 1343–1350. [Google Scholar] [CrossRef]
- Li, J.; Xu, D.; Wang, X.; Liu, K.; Mao, Y.; Wang, M.; Bai, Y.; Wang, W. Encapsulation of cesium with a solid waste-derived sulfoaluminate matrix: A circular economy approach of treating nuclear wastes with solid wastes. J. Hazard. Mater. 2021, 416, 126156. [Google Scholar] [CrossRef]
- Pilania, R.K.; Dube, C.L. Matrices for radioactive waste immobilization: A review. Front. Mater. 2023, 10, 1236470. [Google Scholar] [CrossRef]
- Fahim Huseien, G.; Mirza, J.; Ismail, M.; Ghoshal, S.K.; Abdulameer Hussein, A. Geopolymer mortars as sustainable repair material: A comprehensive review. Renew. Sustain. Energy Rev. 2017, 80, 54–74. [Google Scholar] [CrossRef]
- Liew, Y.-M.; Heah, C.-Y.; Mohd Mustafa, A.B.; Kamarudin, H. Structure and properties of clay-based geopolymer cements: A review. Prog. Mater. Sci. 2016, 83, 595–629. [Google Scholar] [CrossRef]
- Jain, S.; Banthia, N.; Troczynski, T. Leaching of immobilized cesium from NaOH-activated fly ash-based geopolymers. Cem. Concr. Compos. 2022, 133, 104679. [Google Scholar] [CrossRef]
- He, J.; Zhang, J.; Yu, Y.; Zhang, G. The strength and microstructure of two geopolymers derived from metakaolin and red mud-fly ash admixture: A comparative study. Constr. Build. Mater. 2012, 30, 80–91. [Google Scholar] [CrossRef]
- Sarazin, J.; Davy, C.A.; Bourbigot, S.; Tricot, G.; Hosdez, J.; Lambertin, D.; Fontaine, G. Flame resistance of geopolymer foam coatings for the fire protection of steel. Compos. Part B 2021, 222, 109045. [Google Scholar] [CrossRef]
- Zhuang, X.Y.; Chen, L.; Komarneni, S.; Zhou, C.H.; Tong, D.S.; Yang, H.M.; Yu, W.H.; Wang, H. Fly ash-based geopolymer: Clean production, properties and applications. J. Clean. Prod. 2016, 125, 253–267. [Google Scholar] [CrossRef]
- Duan, P.; Yan, C.; Zhou, W. Influence of partial replacement of fly ash by metakaolin on mechanical properties and microstructure of fly ash geopolymer paste exposed to sulfate attack. Ceram. Int. 2016, 42, 3504–3517. [Google Scholar] [CrossRef]
- Ji, Z.; Pei, Y. Bibliographic and visualized analysis of geopolymer research and its application in heavy metal immobilization: A review. J. Environ. Manag. 2019, 231, 256–267. [Google Scholar] [CrossRef]
- Dang, J.; Wang, J.; Wang, X.; Liu, Y.; Liu, H. Efficient removal and stable immobilization of Cs+ by silica-based ammonium phosphomolybdate and geopolymer. Chem. Eng. J. 2025, 503, 158354. [Google Scholar] [CrossRef]
- Shao, W.; Zha, W.; Zhou, X.; Xu, T. Experimental study based on Box–Behnken design and response surface methodology for optimization proportioning of activated lithium slag composite cement-based cementitious materials. Materials 2024, 17, 2651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yue, Y. Influence of waste glass powder usage on the properties of alkali-activated slag mortars based on response surface methodology. Constr. Build. Mater. 2018, 181, 527–534. [Google Scholar] [CrossRef]
- Kockal, N.U.; Ozturan, T. Optimization of properties of fly ash aggregates for high-strength lightweight concrete production. Mater. Des. 2011, 32, 3586–3593. [Google Scholar] [CrossRef]
- Li, D.; Pan, Y.; Liu, C.; Chen, P.; Wu, Y.; Liu, J.; Zheng, Z.; Ma, G. Optimal design of glazed hollow bead thermal insulation mortar containing fly ash and slag based on response surface methodology. Rev. Adv. Mater. Sci. 2023, 62, 20220313. [Google Scholar] [CrossRef]
- Tian, Q.; Nakama, S.; Sasaki, K. Immobilization of cesium in fly ash-silica fume based geopolymers with different Si/Al molar ratios. Sci. Total Environ. 2019, 687, 1127–1137. [Google Scholar] [CrossRef]
- He, P.; Wang, M.; Fu, S.; Jia, D.; Yan, S.; Yuan, J.; Xu, J.; Wang, P.; Zhou, Y. Effects of Si/Al ratio on the structure and properties of metakaolin based geopolymer. Ceram. Int. 2016, 42, 14416–14422. [Google Scholar] [CrossRef]
- Li, K.; Lu, D.; Li, M.; Chen, X.; Xu, Z. Effect of water content on microstructure and reaction process of metakaolin-based geopolymers. J. Chin. Ceram. Soc. 2016, 44, 226–231. [Google Scholar] [CrossRef]
- Box, G.E.P.; Behnken, D.W. Some new three level designs for the study of quantitative variables. Technometrics 1960, 2, 455–475. [Google Scholar] [CrossRef]
- Behera, S.K.; Meena, H.; Chakraborty, S.; Meikap, B.C. Application of response surface methodology (RSM) for optimization of leaching parameters for ash reduction from low-grade coal. Int. J. Min. Sci. Technol. 2018, 28, 621–629. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Y.; Lin, N.; Zhang, Z. Significantly enhanced alkaline stability and cyanide suppression of Prussian blue analogues using montmorillonite for high-performance cesium removal. Sep. Purif. Technol. 2023, 325, 124662. [Google Scholar] [CrossRef]
- GB/T 17671-2021; Test Method of Cement Mortar Strength (ISO Method). State Administration for Market Regulation: Beijing, China; Standardization Administration of the People’s Republic of China: Beijing, China, 2021.
- GB/T 7023-2011; Standard Test Method for Leachability of Low and Intermediate Level Solidified Radioactive Waste Forms. China Institute for Radiation Protection: Taiyuan, China, 2021.
- GB 14569.1-2011; Performance Requirements for Low and Intermediate Level Radioactive Waste Form—Cemented Waste Form. Nuclear and Radiation Safety Center: Beijing, China; China Institute for Radiation Protection: Taiyuan, China, 2011.
- Wang, Y.; Liu, X.; Zhang, W.; Li, Z.; Zhang, Y.; Li, Y.; Ren, Y. Effects of Si/Al ratio on the efflorescence and properties of fly ash based geopolymer. J. Clean. Prod. 2020, 244, 118852. [Google Scholar] [CrossRef]
- Wang, G.; Fan, Z.; Lu, D. Effect of Si and AI raw-material on the preparation and performance of geopolymer. Bull. Chin. Ceram. Soc. 2009, 28, 239–244. [Google Scholar]
- Pouhet, R.; Cyr, M.; Bucher, R. Influence of the initial water content in flash calcined metakaolin-based geopolymer. Constr. Build. Mater. 2019, 201, 421–429. [Google Scholar] [CrossRef]
- Zhu, R.; Wang, S.; Chen, Y.; Xiang, D.; Zhang, L.; Liu, J.; Ye, J. Ultrasound enhanced in-situ chemical oxidation for leaching Ag from zinc leaching residue and response surface optimization. Chem. Eng. J. 2024, 489, 151243. [Google Scholar] [CrossRef]
- Nnanwube, I.A.; Onukwuli, O.D.; Omotioma, M.; Ezekannagha, C.B.; Igwilo, C.N.; Esonye, C.; Anadebe, V.C.; Ume, C.S. Optimization of alumina leaching from microcline using particle swarm optimization and response surface methodology. Sci. Rep. 2025, 15, 9111. [Google Scholar] [CrossRef]
- Abikak, Y.; Bakhshyan, A.; Dyussenova, S.; Gladyshev, S.; Kassymzhanova, A. Optimization of hydrochemical leaching process of kaolinite fraction of bauxite with response surface methodology. Processes 2024, 12, 1440. [Google Scholar] [CrossRef]
- Houssaine Moutiy, E.; Tran, L.-H.; Mueller, K.K.; Coudert, L.; Blais, J.-F. Optimized indium solubilization from LCD panels using H2SO4 leaching. Waste Manag. 2020, 114, 53–61. [Google Scholar] [CrossRef]
- Zhang, L.; Zhai, J. Application of response surface methodology to optimize alkali-activated slag mortar with limestone powder and glass powder. Struct. Concr. 2021, 22, E430–E441. [Google Scholar] [CrossRef]
- Juengsuwattananon, K.; Winnefeld, F.; Chindaprasirt, P. Correlation between initial SiO2/Al2O3, Na2O/Al2O3, Na2O/SiO2 and H2O/Na2O ratios on phase and microstructure of reaction products of metakaolin-rice husk ash geopolymer. Constr. Build. Mater. 2019, 226, 239–244. [Google Scholar] [CrossRef]
- Duxson, P.; Provis, J.L.; Lukey, G.C.; Mallicoat, S.W.; Kriven, W.M.; van Deventer, J.S.J. Understanding the relationship between geopolymer composition, microstructure and mechanical properties. Colloids Surf. A 2005, 269, 47–58. [Google Scholar] [CrossRef]
- Riahi, S.; Nemati, A.; Khodabandeh, A.R.; Baghshahi, S. The effect of mixing molar ratios and sand particles on microstructure and mechanical properties of metakaolin-based geopolymers. Mater. Chem. Phys. 2020, 240, 122223. [Google Scholar] [CrossRef]
- Panias, D.; Giannopoulou, I.P.; Perraki, T. Effect of synthesis parameters on the mechanical properties of fly ash-based geopolymers. Colloids Surf. A 2007, 301, 246–254. [Google Scholar] [CrossRef]
- Wang, H.; Wu, H.; Xing, Z.; Wang, R.; Dai, S. The effect of various Si/Al, Na/Al molar ratios and free water on micromorphology and macro-strength of metakaolin-based geopolymer. Materials 2021, 14, 3845. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Wang, Z.; Lv, M. Multiobjective optimization of precision forging process parameters based on response surface method. Adv. Mater. Sci. Eng. 2015, 2015, 893730. [Google Scholar] [CrossRef][Green Version]
- Hamisi, H.; Jande, Y.A.C.; Hilonga, A. Highly reactive metakaolin: A multi-parameter optimization by response surface methodology. Eng. Res. Express 2023, 5, 045064. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Z.; Wang, Y.; Feng, J. Preparation and properties of alkali activated metakaolin-based geopolymer. Materials 2016, 9, 767. [Google Scholar] [CrossRef]
- Hou, L.; Li, J.; Lu, Z.-y. Effect of Na/Al on formation, structures and properties of metakaolin based Na-geopolymer. Constr. Build. Mater. 2019, 226, 250–258. [Google Scholar] [CrossRef]
- Görhan, G.; Aslaner, R.; Şinik, O. The effect of curing on the properties of metakaolin and fly ash-based geopolymer paste. Compos. Part B 2016, 97, 329–335. [Google Scholar] [CrossRef]
- Provis, J.L.; Lukey, G.C.; Van Deventer, J.S.J. Do Geopolymers Actually Contain Nanocrystalline Zeolites? A Reexamination of Existing Results. Chem. Mater. 2025, 17, 3075–3085. [Google Scholar] [CrossRef]
- Chen, Z.; He, P.; Li, W.; Zhao, S.; Jia, D.; Colombo, P.; Zhou, Y. Cold reaction sintering of geopolymers for enhanced Cs+ immobilization: Synthesis, characterization, and leaching behavior. J. Eur. Ceram. Soc. 2025, 45, 329–335. [Google Scholar] [CrossRef]
- Rees, C.A.; Provis, J.L.; Lukey, G.C.; van Deventer, J.S.J. In situ ATR-FTIR study of the early stages of fly ash geopolymer gel formation. Langmuir 2007, 23, 9076–9082. [Google Scholar] [CrossRef]
- Wang, S.; Chen, X.; Li, W.; Zhang, S.S. Setting behavior, mechanical properties and microstructure of seawater-mixed metakaolin-based geopolymers. Constr. Build. Mater. 2025, 493, 143200. [Google Scholar] [CrossRef]
- Sitarz, M.; Handke, M.; Mozgawa, W. Identification of silicooxygen rings in SiO2 based on IR spectra. Spectrochim. Acta Part A 2000, 56, 1819–1823. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xu, Y.; Wang, J.; Zhang, W.; Ye, J.; Wang, R. The immobilizing performance and mechanism of geopolymer and its derivative materials for high-level radionuclides Cs and Sr: A review. J. Aust. Ceram. Soc. 2024, 60, 1131–1151. [Google Scholar] [CrossRef]







| Levels | Coded Factors | Actual Factors | ||
|---|---|---|---|---|
| Na2O/Al2O3(A) | SiO2/Al2O3(B) | H2O/Na2O(C) | ||
| Low | −1 | 0.6 | 2.8 | 9 |
| 0 | 0 | 0.8 | 3.6 | 10 |
| High | 1 | 1 | 4.4 | 11 |
| Composition | SiO2 | Al2O3 | Ti2O | Fe2O3 | ZrO2 | Na2O | CaO | K2O | P2O5 | MgO |
|---|---|---|---|---|---|---|---|---|---|---|
| MK | 45.07 | 52.06 | 1.25 | 0.42 | 0.03 | 0.24 | 0.08 | 0.20 | 0.25 | 0.40 |
| Silica fume | 97.38 | 0.23 | 0.04 | 0.43 | 1.26 | - | 0.08 | 0.04 | 0.39 | 0.15 |
| Mixture Code | Type | Na2O/Al2O3 | SiO2/Al2O3 | H2O/Na2O | 28-Day Compressive Strength (MPa) | 42-Day Cumulative Fraction Leached (10−5 cm) | |
|---|---|---|---|---|---|---|---|
| 25 °C | 40 °C | ||||||
| S1 | A1B1C2 | 0.6 | 2.8 | 10 | 16.61 | 68.82 | 175.04 |
| S2 | A3B1C2 | 1 | 2.8 | 10 | 24.10 | 124.42 | 258.54 |
| S3 | A1B3C2 | 0.6 | 4.4 | 10 | 11.61 | 76.52 | 153.04 |
| S4 | A3B3C2 | 1 | 4.4 | 10 | 14.93 | 129.13 | 301.94 |
| S5 | A1B2C1 | 0.6 | 3.6 | 9 | 10.20 | 128.25 | 252.79 |
| S6 | A3B2C1 | 1 | 3.6 | 9 | 13.21 | 147.67 | 284.04 |
| S7 | A1B2C3 | 0.6 | 3.6 | 11 | 20.62 | 175.27 | 429.08 |
| S8 | A3B2C3 | 1 | 3.6 | 11 | 27.83 | 262.19 | 508.59 |
| S9 | A2B1C1 | 0.8 | 2.8 | 9 | 12.90 | 67.58 | 160.01 |
| S10 | A2B3C1 | 0.8 | 4.4 | 9 | 7.76 | 73.29 | 167.73 |
| S11 | A2B1C3 | 0.8 | 2.8 | 11 | 27.68 | 125.13 | 259.94 |
| S12 | A2B3C3 | 0.8 | 4.4 | 11 | 21.07 | 183.82 | 377.64 |
| S13 | A2B2C2 | 0.8 | 3.6 | 10 | 16.83 | 140.37 | 257.09 |
| S14 | A2B2C2 | 0.8 | 3.6 | 10 | 17.51 | 134.42 | 258.54 |
| S15 | A2B2C2 | 0.8 | 3.6 | 10 | 18.30 | 137.56 | 250.28 |
| Response | y1 | y2 | y3 |
|---|---|---|---|
| Model | |||
| Standard deviation | 0.67 | 8.66 | 14.79 |
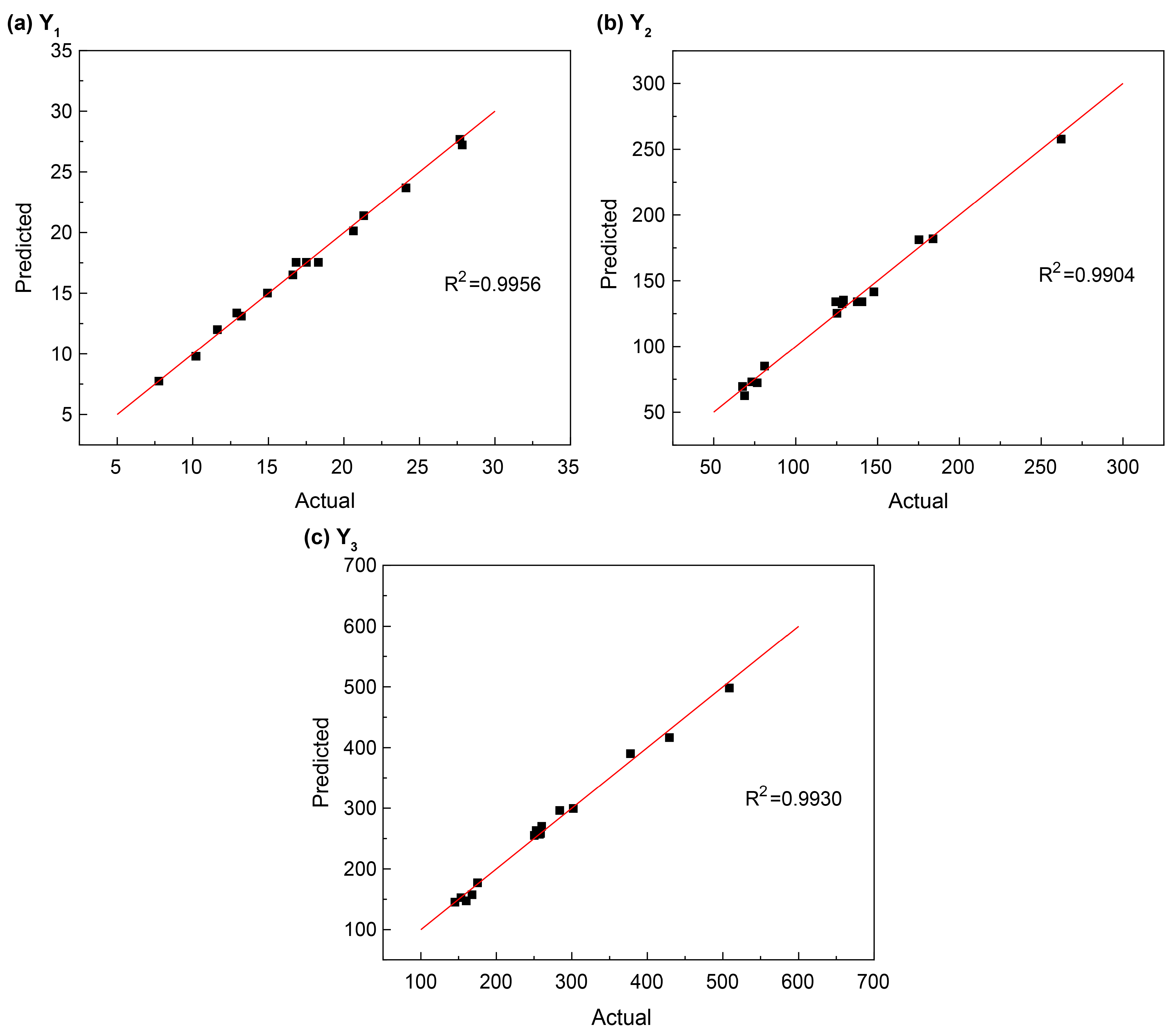
| R2 value | 0.9956 | 0.9904 | 0.9930 |
| F value | 126.22 | 57.22 | 79.02 |
| p-value (Prob > F) | <0.0001 | 0.0002 | <0.0001 |
| Lack of fit (Prob > F) | 0.6309 | 0.5199 | 0.0530 |
| p-value (Prob > F) | |||
| x1-Na2O/Al2O3 | <0.0001 | 0.0009 | 0.0027 |
| x2-SiO2/Al2O3 | 0.0001 | 0.0045 | 0.0016 |
| x3-H2O/Na2O | <0.0001 | <0.0001 | <0.0001 |
| x1x2 | 0.0262 | 0.0670 | 0.0018 |
| x1x3 | 0.0462 | 0.0115 | 0.1637 |
| x2x3 | 0.2101 | 0.0281 | 0.0137 |
| x12 | 0.8262 | 0.0706 | 0.0079 |
| x22 | 0.0661 | <0.0001 | <0.0001 |
| x32 | 0.2037 | 0.0007 | 0.0001 |
| Response | Optimized Mixture (FSM) | |||
|---|---|---|---|---|
| Experimental Value | Predicted Value | Error (%) | ||
| 28-Day Compressive strength (MPa) | 23.41 | 22.36 | 4.70% | |
| 42-Day Cumulative Fraction Leached (10−5 cm) | 25 °C | 79.06 | 74.68 | 5.87% |
| 40 °C | 159.23 | 145.06 | 9.77% | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liao, Y.; Yu, X.; Yuan, X.; Wang, J.; Yan, Y.; Ouyang, G. Optimization Design of Metakaolin-Based Geopolymer Solidification for Potassium Copper Hexacyanoferrate After Cs+ Adsorption Using Response Surface Methodology. Materials 2026, 19, 1469. https://doi.org/10.3390/ma19071469
Liao Y, Yu X, Yuan X, Wang J, Yan Y, Ouyang G. Optimization Design of Metakaolin-Based Geopolymer Solidification for Potassium Copper Hexacyanoferrate After Cs+ Adsorption Using Response Surface Methodology. Materials. 2026; 19(7):1469. https://doi.org/10.3390/ma19071469
Chicago/Turabian StyleLiao, Yuqing, Xingyu Yu, Xinyi Yuan, Jingsong Wang, Yao Yan, and Gaoshang Ouyang. 2026. "Optimization Design of Metakaolin-Based Geopolymer Solidification for Potassium Copper Hexacyanoferrate After Cs+ Adsorption Using Response Surface Methodology" Materials 19, no. 7: 1469. https://doi.org/10.3390/ma19071469
APA StyleLiao, Y., Yu, X., Yuan, X., Wang, J., Yan, Y., & Ouyang, G. (2026). Optimization Design of Metakaolin-Based Geopolymer Solidification for Potassium Copper Hexacyanoferrate After Cs+ Adsorption Using Response Surface Methodology. Materials, 19(7), 1469. https://doi.org/10.3390/ma19071469





