Biopolymer-Based Electrospun Nanofibers for Wound Healing, Regeneration, and Therapeutics
Abstract
1. Introduction
2. Fundamentals of Electrospinning
3. Tissue Engineering Applications of Biopolymer Nanofibers
3.1. Neural Tissue Engineering
3.2. Musculoskeletal Tissue Engineering
3.3. Vascular Tissue Engineering
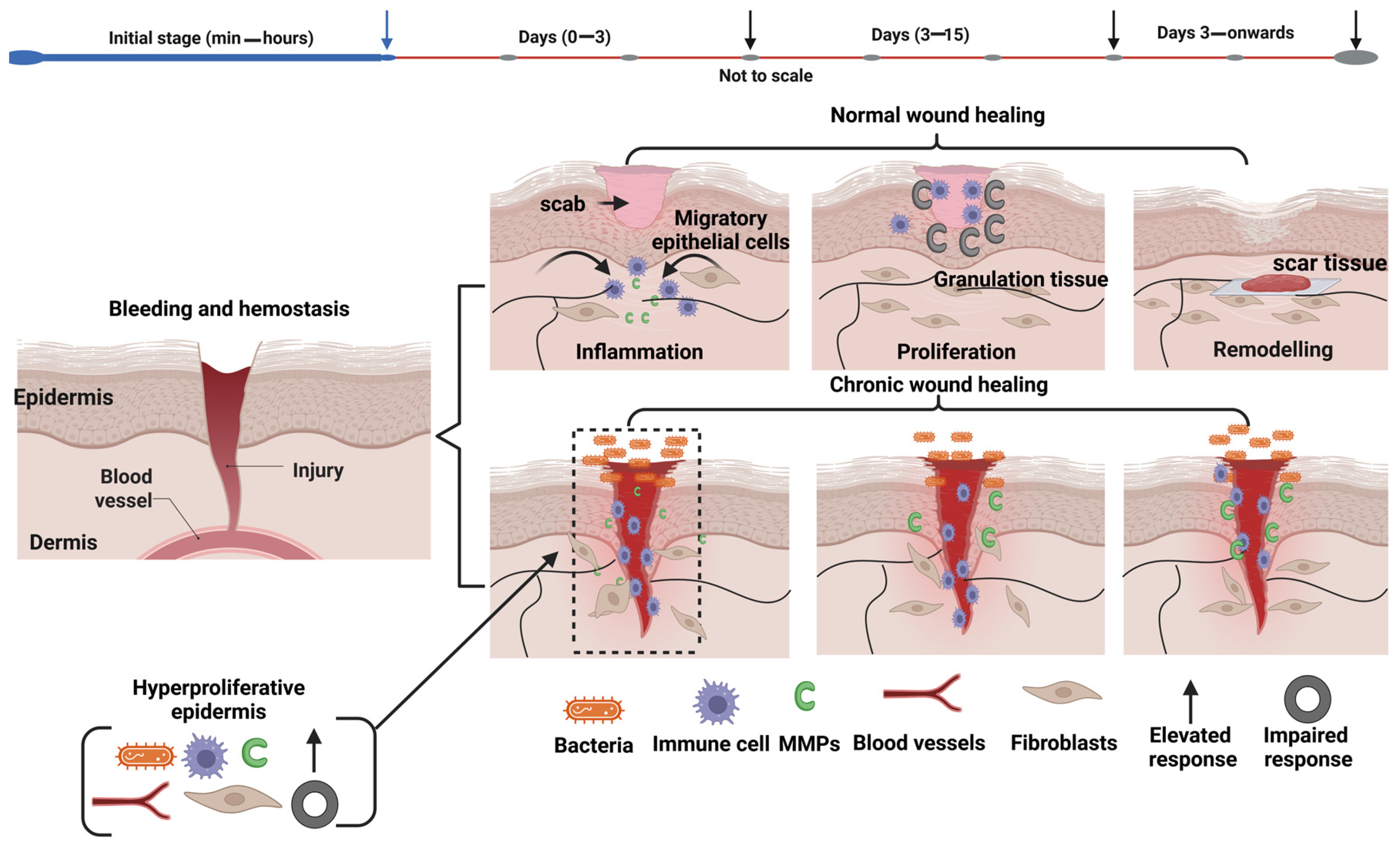
4. Electrospun Biopolymer Nanofibers in Wound Healing Applications
5. Smart and Multifunctional Biomedical Platforms
5.1. Stimuli-Responsive Smart Drug Delivery Systems
5.2. Electrospun Nanofiber-Based Biosensors
5.3. Wearable Nanofiber Systems for Real-Time Monitoring and Personalized Healthcare
5.4. Theranostic Nanofiber Platforms
6. Strategic Integration of Electrospinning with 3D Printing
7. Challenges and Future Perspectives
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BDNF | Brain-Derived Neurotrophic Factor |
| BGM | Blood glucose monitoring |
| CNS | Central nervous system |
| CQD | Carbon quantum dot |
| ECG | Electrocardiography |
| ECM | Extracellular matrix |
| FDM | Fused deposition modeling |
| GCE | Glassy carbon electrodes |
| GNR | Gold nanorods |
| HA | Hyaluronic acid |
| hTSPCs | human tendon progenitor cells |
| LbL | Layer by Layer |
| NGF | Nerve Growth Factor |
| NIR | Near-Infrared |
| NO | Nitric oxide |
| NT | Neurotrophin |
| PAN | Polyacrylonitrile |
| PANi | Polyaniline |
| PCL | Poly(ε-caprolactone) |
| PEO | Poly(ethylene oxide) |
| PLA | Polylactic acid |
| PLGA | Poly(lactic-co-glycolic) acid |
| PLLA | Poly(L-lactic acid |
| PNIPAM | Poly(N-iso-propylacrylamide) |
| PNS | Peripheral nervous system |
| PPY | Polypyrrole |
| PVA | Polyvinyl alcohol |
| RGD | Arginine-Glycine-Aspartic Acid |
| ROS | Reactive oxygen species |
| SF | Silk Fibroin |
References
- Williams, D.F. On the nature of biomaterials. Biomaterials 2009, 30, 5897–5909. [Google Scholar] [CrossRef] [PubMed]
- Zarrintaj, P.; Seidi, F.; Youssefi Azarfam, M.; Khodadadi Yazdi, M.; Erfani, A.; Barani, M.; Chauhan, N.P.S.; Rabiee, N.; Kuang, T.; Kucinska-Lipka, J.; et al. Biopolymer-based composites for tissue engineering applications: A basis for future opportunities. Compos. B Eng. 2023, 258, 110701. [Google Scholar] [CrossRef]
- Abdulsalam, L.; Abubakar, S.; Permatasari, I.; Lawal, A.A.; Uddin, S.; Ullah, S.; Ahmad, I. Advanced Biocompatible and Biodegradable Polymers: A Review of Functionalization, Smart Systems, and Sustainable Applications. Polymers 2025, 17, 2901. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Sharma, M.; Saraf, A.; Bonde, C.; Bonde, S.; Tiwari, K.; Lodha, G.; Singh, S.; Mishra, V. Electrospun nanofibers as multifunctional nanobiosystems for drug delivery and regenerative medicine. In Intelligent Nanobiosystems in Medicine and Healthcare, Volume 2: Applications of Intelligent Nanobiosystems; Academic Press: Cambridge, MA, USA, 2025; pp. 375–416. [Google Scholar] [CrossRef]
- Schiffman, J.D.; Schauer, C.L. A Review: Electrospinning of Biopolymer Nanofibers and their Applications. Polym. Rev. 2008, 48, 317–352. [Google Scholar] [CrossRef]
- Fromager, B.; Marhuenda, E.; Louis, B.; Bakalara, N.; Cambedouzou, J.; Cornu, D. Recent Advances in Electrospun Fibers for Biological Applications. Macromol 2023, 3, 569–613. [Google Scholar] [CrossRef]
- Al-Enizi, A.M.; Zagho, M.M.; Elzatahry, A.A. Polymer-Based Electrospun Nanofibers for Biomedical Applications. Nanomaterials 2018, 8, 259. [Google Scholar] [CrossRef]
- Iacob, A.T.; Drăgan, M.; Ionescu, O.M.; Profire, L.; Ficai, A.; Andronescu, E.; Confederat, L.G.; Lupașcu, D. An Overview of Biopolymeric Electrospun Nanofibers Based on Polysaccharides for Wound Healing Management. Pharmaceutics 2020, 12, 983. [Google Scholar] [CrossRef]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15. [Google Scholar] [CrossRef]
- Oliveira, C.; Sousa, D.; Teixeira, J.A.; Ferreira-Santos, P.; Botelho, C.M. Polymeric biomaterials for wound healing. Front. Bioeng. Biotechnol. 2023, 11, 1136077. [Google Scholar] [CrossRef]
- Downer, M.; Berry, C.E.; Parker, J.B.; Kameni, L.; Griffin, M. Current Biomaterials for Wound Healing. Bioengineering 2023, 10, 1378. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Manna, S.; Roy, S.; Nandi, S.K.; Basak, P. Polymeric biomaterials-based tissue engineering for wound healing: A systemic review. Burn. Trauma 2023, 11, tkac058. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, D.S.; Keerthika, K.; Vijayaragavan, V. Chitosan-Based Biomaterial in Wound Healing: A Review. Cureus 2024, 16, e55193. [Google Scholar] [CrossRef] [PubMed]
- Deo, S.K.; Moschou, E.A.; Peteu, S.F.; Bachas, L.G.; Daunert, S.; Eisenhardt, P.E.; Madou, M.J. ROS-responsive drug delivery systems. Bioeng. Transl. Med. 2016, 1, 239. [Google Scholar] [CrossRef]
- Dumitriu, R.; Vasile, C.; Mitchell, G.; Oprea, A.M. Stimuli-responsive drug delivery system. In Monomers, Oligomers, Polymers, Composites and Nanocomposites Research; Nova Science Publishers, Inc.: New York, NY, USA, 2009; pp. 401–410. [Google Scholar] [CrossRef]
- Halicka, K.; Cabaj, J. Electrospun Nanofibers for Sensing and Biosensing Applications—A Review. Int. J. Mol. Sci. 2021, 22, 6357. [Google Scholar] [CrossRef]
- Wu, L.; Song, Y.; Xing, S.; Li, Y.; Xu, H.; Yang, Q.; Li, Y. Advances in electrospun nanofibrous membrane sensors for ion detection. RSC Adv. 2022, 12, 34866–34891. [Google Scholar] [CrossRef]
- Chen, Z.; Guan, M.; Bian, Y.; Yin, X. Multifunctional Electrospun Nanofibers for Biosensing and Biomedical Engineering Applications. Biosensors 2024, 14, 13. [Google Scholar] [CrossRef]
- Hao, P.; Qiu, S.; Liu, S.; Chen, X. Emerging roles of electrospun nanofibers for real-time motion monitoring. Adv. Compos. Hybrid Mater. 2025, 8, 116. [Google Scholar] [CrossRef]
- Khan, B.; Khalid, R.T.; Amara, U.; Imdad, N.; Masrur, M.H.; Awais, M.; Laghari, A.Q.; Khan, B.; Elgendi, M.; Khoo, B.L.; et al. Electrospun nanofibers for wearable cardiovascular health monitoring. J. Sci. Adv. Mater. Devices 2025, 10, 101030. [Google Scholar] [CrossRef]
- Zhao, P.; Xu, J.; Han, M. Patient-centric care: Unveiling the potential of wearable electronics in clinical practice. Wearable Electron. 2024, 1, 119–136. [Google Scholar] [CrossRef]
- Shajari, S.; Kuruvinashetti, K.; Komeili, A.; Sundararaj, U. The Emergence of AI-Based Wearable Sensors for Digital Health Technology: A Review. Sensors 2023, 23, 9498. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, L.; Sobhana, S.; Yasothamani, V.; Gowsalya, K.; Vivek, R. Multifunctional theranostic nanomedicines for cancer treatment: Recent progress and challenges. Biomed. Eng. Adv. 2023, 5, 100082. [Google Scholar] [CrossRef]
- Yasir, M.; Mishra, R.; Tripathi, A.S.; Maurya, R.K.; Shahi, A.; Zaki, M.E.A.; Al Hussain, S.A.; Masand, V.H. Theranostics: A multifaceted approach utilizing nano-biomaterials. Discov. Nano 2024, 19, 35. [Google Scholar] [CrossRef] [PubMed]
- Joorabloo, A.; Liu, T. Smart theranostics for wound monitoring and therapy. Adv. Colloid Interface Sci. 2024, 330, 103207. [Google Scholar] [CrossRef]
- Alfatama, M.; Choukaife, H.; Doolaanea, A.A. 3D Printing and Electrospinning. In Electrospraying and Electrospinning in Drug Delivery; CRC Press: Boca Raton, FL, USA, 2025; pp. 306–336. [Google Scholar] [CrossRef]
- Ejiohuo, O. A perspective on the synergistic use of 3D printing and electrospinning to improve nanomaterials for biomedical applications. Nano Trends 2023, 4, 100025. [Google Scholar] [CrossRef]
- da Silva Gusmão, R.; Maciel, E.M.; Frattini, W.C.; de Medeiros, E.S.; Fonseca, F.A.H.; Bianco, H.T. Advances and challenges in nanofiber production: Techniques and applications in nanotechnology. Polímeros Ciênc. E Tecnol. 2025, 35, e20250039. [Google Scholar] [CrossRef]
- Kumar, P.; Jain, A.; Sharma, M.; Garg, A.; Ganguly, S.C.; Patel, A.; Khan, Y.; Debnath, B.; Ashique, S.; Hussain, M.S. Nanofibres: A promising approach in drug delivery systems. Int. J. Polym. Mater. Polym. Biomater. 2026, 75, 500–534. [Google Scholar] [CrossRef]
- Das, S.K.; Chakraborty, S.; Naskar, S.; Rajabalaya, R. Techniques and methods used for the fabrication of bionanocomposites. In Bionanocomposites in Tissue Engineering and Regenerative Medicine; Woodhead Publishing: Cambridge, UK, 2021; pp. 17–43. [Google Scholar] [CrossRef]
- Vaseashta, A.; Bolgen, N. (Eds.) Electrospun Nanofibers; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Ahmadi Bonakdar, M.; Rodrigue, D. Electrospinning: Processes, Structures, and Materials. Macromol 2024, 4, 58–103. [Google Scholar] [CrossRef]
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrostat. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Abdulhussain, R.; Adebisi, A.; Conway, B.R.; Asare-Addo, K. Electrospun nanofibers: Exploring process parameters, polymer selection, and recent applications in pharmaceuticals and drug delivery. J. Drug Deliv. Sci. Technol. 2023, 90, 105156. [Google Scholar] [CrossRef]
- Wang, Q.; Ji, P.; Bu, T.; Mao, Y.; He, H.; Ge, N. Recent Progress in the Application of Electrospinning Technology in the Biomedical Field. J. Funct. Biomater. 2025, 16, 266. [Google Scholar] [CrossRef] [PubMed]
- Ameer, J.M.; Anil Kumar, P.R.; Kasoju, N. Strategies to Tune Electrospun Scaffold Porosity for Effective Cell Response in Tissue Engineering. J. Funct. Biomater. 2019, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.; Sant, S.; Nichol, J.W.; Khabiry, M.; Traversa, E.; Khademhosseini, A. Controlling the porosity of fibrous scaffolds by modulating the fiber diameter and packing density. J. Biomed. Mater. Res. Part A 2021, 96, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Toftdal, M.S.; Le Friec, A.; Dong, M.; Han, X.; Chen, M. 3D Electrospun Synthetic Extracellular Matrix for Tissue Regeneration. Small Sci. 2021, 1, 2100003. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Gao, Z.; Mao, X.; Cheng, J.; Huang, L.; Tang, J. Advances in wound dressing based on electrospinning nanofibers. J. Appl. Polym. Sci. 2024, 141, e54746. [Google Scholar] [CrossRef]
- Al-Abduljabbar, A.; Farooq, I. Electrospun Polymer Nanofibers: Processing, Properties, and Applications. Polymers 2022, 15, 65. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.; Lou, L.; Meng, Z. A Review of Chitosan-Based Electrospun Nanofibers for Food Packaging: From Fabrication to Function and Modeling Insights. Nanomaterials 2025, 15, 1274. [Google Scholar] [CrossRef]
- Keshvardoostchokami, M.; Majidi, S.S.; Huo, P.; Ramachandran, R.; Chen, M.; Liu, B. Electrospun Nanofibers of Natural and Synthetic Polymers as Artificial Extracellular Matrix for Tissue Engineering. Nanomaterials 2020, 11, 21. [Google Scholar] [CrossRef]
- Gu, X.; Song, X.; Shao, C.; Zeng, P.; Lu, X.; Shen, X.; Yang, Q. Electrospinning of poly(butylene-carbonate):Effect of Solvents on the Properties of the Nanofibers Film. Int. J. Electrochem. Sci. 2014, 9, 8045–8056. [Google Scholar] [CrossRef]
- Rajeev, M.; Helms, C.C. A Study of the Relationship between Polymer Solution Entanglement and Electrospun PCL Fiber Mechanics. Polymers 2023, 15, 4555. [Google Scholar] [CrossRef]
- Dulnik, J.; Sajkiewicz, P. Crosslinking of Gelatin in Bicomponent Electrospun Fibers. Materials 2021, 14, 3391. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Z.; Venugopal, J.; Huang, Z.M.; Lim, C.T.; Ramakrishna, S. Crosslinking of the electrospun gelatin nanofibers. Polymer 2006, 47, 2911–2917. [Google Scholar] [CrossRef]
- Campiglio, C.E.; Ponzini, S.; De Stefano, P.; Ortoleva, G.; Vignati, L.; Draghi, L. Cross-Linking Optimization for Electrospun Gelatin: Challenge of Preserving Fiber Topography. Polymers 2020, 12, 2472. [Google Scholar] [CrossRef] [PubMed]
- Campiglio, C.E.; Negrini, N.C.; Farè, S.; Draghi, L. Cross-Linking Strategies for Electrospun Gelatin Scaffolds. Materials 2019, 12, 2476. [Google Scholar] [CrossRef]
- Luo, X.; Guo, Z.; He, P.; Chen, T.; Li, L.; Ding, S.; Li, H. Study on structure, mechanical property and cell cytocompatibility of electrospun collagen nanofibers crosslinked by common agents. Int. J. Biol. Macromol. 2018, 113, 476–486. [Google Scholar] [CrossRef]
- Chinnappan, B.A.; Krishnaswamy, M.; Xu, H.; Hoque, M.E. Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters. Polymers 2022, 14, 3719. [Google Scholar] [CrossRef]
- Wang, J.; Windbergs, M. Controlled dual drug release by coaxial electrospun fibers—Impact of the core fluid on drug encapsulation and release. Int. J. Pharm. 2019, 556, 363–371. [Google Scholar] [CrossRef]
- Bullon, J.; Marquez, R.; Zambrano, F.; Ghosh, S.; Yadav, A.; Gurave, P.M.; Srivastava, R.K. Unique Fiber Morphologies from Emulsion Electrospinning—A Case Study of Poly(ε-caprolactone) and Its Applications. Colloids Interfaces 2023, 7, 19. [Google Scholar] [CrossRef]
- Bongiovanni Abel, S.; Montini Ballarin, F.; Abraham, G.A. Combination of electrospinning with other techniques for the fabrication of 3D polymeric and composite nanofibrous scaffolds with improved cellular interactions. Nanotechnology 2020, 31, 172002. [Google Scholar] [CrossRef]
- Omiyale, B.O.; Ogbeyemi, A.; Rasheed, A.A.; Adamolekun, T.M.; Zhang, W.C. Influence of electrospinning parameters on the development of high-quality electrospun nanofibers: A brief critical assessment. Next Nanotechnol. 2025, 8, 100295. [Google Scholar] [CrossRef]
- Ji, D.; Lin, Y.; Guo, X.; Ramasubramanian, B.; Wang, R.; Radacsi, N.; Jose, R.; Qin, X.; Ramakrishna, S. Electrospinning of nanofibres. Nat. Rev. Methods Primers 2024, 4, 1. [Google Scholar] [CrossRef]
- Bobrin, V.A.; Chen, S.P.; Grandes Reyes, C.F.; Sun, B.; Ng, C.K.; Kim, Y.; Purcell, D.; Jia, Z.; Gu, W.; Armstrong, J.W.; et al. Water-Borne Nanocoating for Rapid Inactivation of SARS-CoV-2 and Other Viruses. ACS Nano 2021, 15, 14915–14927. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Wang, J.; Ding, L.; Hu, Y.; Li, W.; Yuan, Z.; Guo, Q.; Zhu, C.; Yu, L.; Wang, H.; et al. Tissue Engineering and Regenerative Medicine: Achievements, Future, and Sustainability in Asia. Front. Bioeng. Biotechnol. 2020, 8, 83. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N. Mechanical and biological properties of scaffold materials. In Functional 3D Tissue Engineering Scaffolds: Materials, Technologies, and Applications; Woodhead Publishing: Cambridge, UK, 2018; pp. 1–21. [Google Scholar] [CrossRef]
- Zhong, J.; Zhang, H.; Yan, J.; Gong, X. Effect of nanofiber orientation of electrospun nanofibrous scaffolds on cell growth and elastin expression of muscle cells. Colloids Surf. B Biointerfaces 2015, 136, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Badmus, M.; Liu, J.; Wang, N.; Radacsi, N.; Zhao, Y. Hierarchically electrospun nanofibers and their applications: A review. Nano Mater. Sci. 2021, 3, 213–232. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, N.; Zhu, Z.; Liu, J.; Qi, X.; Liu, Z.; Zhu, Y.; Wang, X.; Long, Y.; Xiang, H. Electrospun 3D nanofibrous materials and their applications in orthopaedics. Adv. Compos. Hybrid Mater. 2024, 8, 62. [Google Scholar] [CrossRef]
- Echeverria Molina, M.I.; Malollari, K.G.; Komvopoulos, K. Design Challenges in Polymeric Scaffolds for Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 9, 617141. [Google Scholar] [CrossRef]
- Ye, K.; Kuang, H.; You, Z.; Morsi, Y.; Mo, X. Electrospun Nanofibers for Tissue Engineering with Drug Loading and Release. Pharmaceutics 2019, 11, 182. [Google Scholar] [CrossRef]
- Evans, P.J.; Midha, R.; Mackinnon, S.E. The peripheral nerve allograft: A comprehensive review of regeneration and neuroimmunology. Prog. Neurobiol. 1994, 43, 187–233. [Google Scholar] [CrossRef]
- Cao, H.; Liu, T.; Chew, S.Y. The application of nanofibrous scaffolds in neural tissue engineering. Adv. Drug Deliv. Rev. 2009, 61, 1055–1064. [Google Scholar] [CrossRef]
- Yan, B.; Zhang, Y.; Li, Z.; Zhou, P.; Mao, Y. Electrospun nanofibrous membrane for biomedical application. SN Appl. Sci. 2022, 4, 172. [Google Scholar] [CrossRef]
- Liang, Y.; Goh, J.C.H. Polypyrrole-Incorporated Conducting Constructs for Tissue Engineering Applications: A Review. Bioelectricity 2020, 2, 101–119. [Google Scholar] [CrossRef]
- Garrudo, F.F.F.; Mikael, P.E.; Rodrigues, C.A.V.; Udangawa, R.W.; Paradiso, P.; Chapman, C.A.; Hoffman, P.; Colaço, R.; Cabral, J.M.S.; Morgado, J.; et al. Polyaniline-polycaprolactone fibers for neural applications: Electroconductivity enhanced by pseudo-doping. Mater. Sci. Eng. C 2021, 120, 111680. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Ma, P.X. Conducting Polymers for Tissue Engineering. Biomacromolecules 2018, 19, 1764–1782. [Google Scholar] [CrossRef] [PubMed]
- Doblado, L.R.; Martínez-Ramos, C.; Pradas, M.M. Biomaterials for Neural Tissue Engineering. Front. Nanotechnol. 2021, 3, 643507. [Google Scholar] [CrossRef]
- Zhao, Y.; Liang, Y.; Ding, S.; Zhang, K.; Mao, H.Q.; Yang, Y. Application of conductive PPy/SF composite scaffold and electrical stimulation for neural tissue engineering. Biomaterials 2020, 255, 120164. [Google Scholar] [CrossRef]
- Markus, A.; Patel, T.D.; Snider, W.D. Neurotrophic factors and axonal growth. Curr. Opin. Neurobiol. 2002, 12, 523–531. [Google Scholar] [CrossRef]
- Liu, K.; Yan, S.; Liu, Y.; Liu, J.; Li, R.; Zhao, L.; Liu, B. Conductive and alignment-optimized porous fiber conduits with electrical stimulation for peripheral nerve regeneration. Mater. Today Bio 2024, 26, 101064. [Google Scholar] [CrossRef]
- Puranik, N.; Tiwari, S.; Kumari, M.; Yadav, S.K.; Dhakal, T.; Song, M. Advanced Bioactive Polymers and Materials for Nerve Repair: Strategies and Mechanistic Insights. J. Funct. Biomater. 2025, 16, 255. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A Comparative Review of Natural and Synthetic Biopolymer Composite Scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- Hammam, I.A.; Winters, R.; Hong, Z. Advancements in the application of biomaterials in neural tissue engineering: A review. Biomed. Eng. Adv. 2024, 8, 100132. [Google Scholar] [CrossRef]
- Bramson, M.T.K.; Van Houten, S.K.; Corr, D.T. Mechanobiology in Tendon, Ligament, and Skeletal Muscle Tissue Engineering. J. Biomech. Eng. 2021, 143, 070801. [Google Scholar] [CrossRef] [PubMed]
- Citro, V.; Clerici, M.; Boccaccini, A.R.; Della Porta, G.; Maffulli, N.; Forsyth, N.R. Tendon tissue engineering: An overview of biologics to promote tendon healing and repair. J. Tissue Eng. 2023, 14, 20417314231196275. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Wendorff, J.H.; Greiner, A. Use of electrospinning technique for biomedical applications. Polymer 2008, 49, 5603–5621. [Google Scholar] [CrossRef]
- Jiang, T.; Carbone, E.J.; Lo, K.W.H.; Laurencin, C.T. Electrospinning of polymer nanofibers for tissue regeneration. Prog. Polym. Sci. 2015, 46, 1–24. [Google Scholar] [CrossRef]
- Feng, Y.; Shi, Y.; Tian, Y.; Yang, Y.; Wang, J.; Guo, H.; Banitaba, S.N.; Khademolqorani, S.; Li, J. The Collagen-Based Scaffolds for Bone Regeneration: A Journey through Electrospun Composites Integrated with Organic and Inorganic Additives. Processes 2023, 11, 2105. [Google Scholar] [CrossRef]
- Wu, J.; Wang, S.; Zheng, Z.; Li, J. Fabrication of Biologically Inspired Electrospun Collagen/Silk fibroin/bioactive glass composited nanofibrous scaffold to accelerate the treatment efficiency of bone repair. Regen. Ther. 2022, 21, 122. [Google Scholar] [CrossRef]
- Khajavi, R.; Abbasipour, M.; Bahador, A. Electrospun biodegradable nanofibers scaffolds for bone tissue engineering. J. Appl. Polym. Sci. 2016, 133, 42883. [Google Scholar] [CrossRef]
- Ranganathan, S.; Balagangadharan, K.; Selvamurugan, N. Chitosan and gelatin-based electrospun fibers for bone tissue engineering. Int. J. Biol. Macromol. 2019, 133, 354–364. [Google Scholar] [CrossRef]
- Frohbergh, M.E.; Katsman, A.; Botta, G.P.; Lazarovici, P.; Schauer, C.L.; Wegst, U.G.K.; Lelkes, P.I. Electrospun Hydroxyapatite-Containing Chitosan Nanofibers Crosslinked with Genipin for Bone Tissue Engineering. Biomaterials 2012, 33, 9167. [Google Scholar] [CrossRef]
- Li, W.J.; Mauck, R.L.; Cooper, J.A.; Yuan, X.; Tuan, R.S. Engineering controllable anisotropy in electrospun biodegradable nanofibrous scaffolds for musculoskeletal tissue engineering. J. Biomech. 2007, 40, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Thomopoulos, S.; Xia, Y. Electrospun Nanofibers for Regenerative Medicine. Adv. Healthc. Mater. 2011, 1, 10. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Chen, X.; Chen, J.L.; Shen, W.L.; Hieu Nguyen, T.M.; Gao, L.; Ouyang, H.W. The regulation of tendon stem cell differentiation by the alignment of nanofibers. Biomaterials 2010, 31, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.H.; Lee, H.J.; Koh, W.G. Study of myoblast differentiation using multi-dimensional scaffolds consisting of nano and micropatterns. Biomater. Res. 2017, 21, 1. [Google Scholar] [CrossRef]
- Dolgin, J.; Hanumantharao, S.N.; Farias, S.; Simon, C.G.; Rao, S. Mechanical Properties and Morphological Alterations in Fiber-Based Scaffolds Affecting Tissue Engineering Outcomes. Fibers 2023, 11, 39. [Google Scholar] [CrossRef]
- Halder, S.; Tan, G.Z. Musculoskeletal Tissue Regeneration Using Fibrous Biomaterials. In Tissue Repair and Regeneration: Elucidating Cellular and Molecular Mechanisms with Therapeutic Implications; Springer Nature: Cham, Switzerland, 2025; pp. 307–331. [Google Scholar] [CrossRef]
- Ielo, I.; Calabrese, G.; De Luca, G.; Conoci, S. Recent Advances in Hydroxyapatite-Based Biocomposites for Bone Tissue Regeneration in Orthopedics. Int. J. Mol. Sci. 2022, 23, 9721. [Google Scholar] [CrossRef]
- Fan, J.; Abedi-Dorcheh, K.; Vaziri, A.S.; Kazemi-Aghdam, F.; Rafieyan, S.; Sohrabinejad, M.; Ghorbani, M.; Adib, F.R.; Ghasemi, Z.; Klavins, K.; et al. A Review of Recent Advances in Natural Polymer-Based Scaffolds for Musculoskeletal Tissue Engineering. Polymers 2022, 14, 2097. [Google Scholar] [CrossRef]
- Elshabrawy, H.A.; Moustafa, H.A.; Yacoub, M.H.; El-Sherbiny, I.M. Vascular tissue regeneration. In Electrospun Biomaterials for Tissue Engineering and Drug Delivery Applications; Woodhead Publishing: Cambridge, UK, 2025; pp. 665–706. [Google Scholar] [CrossRef]
- Fahad, M.A.; Al Lee, H.Y.; Park, S.; Choi, M.; Shanto, P.C.; Park, M.; Bae, S.H.; Lee, B.T. Small-diameter vascular graft composing of core-shell structured micro-nanofibers loaded with heparin and VEGF for endothelialization and prevention of neointimal hyperplasia. Biomaterials 2024, 306, 122507. [Google Scholar] [CrossRef]
- Huang, Q.; Hu, K.; Xu, C.; Chen, Y.; Guo, L.; Liu, Y.; Bian, L.; Wen, C.; Wang, W.; Xu, W.; et al. A bio-inspired screwed small-diameter vascular graft for endotheliazation and arterial regeneration. Bioact. Mater. 2025, 53, 253. [Google Scholar] [CrossRef]
- Hasan, A.; Memic, A.; Annabi, N.; Hossain, M.; Paul, A.; Dokmeci, M.R.; Dehghani, F.; Khademhosseini, A. Electrospun Scaffolds for Tissue Engineering of Vascular Grafts. Acta Biomater. 2013, 10, 11–25. [Google Scholar] [CrossRef]
- Yau, J.W.; Teoh, H.; Verma, S. Endothelial cell control of thrombosis. BMC Cardiovasc. Disord. 2015, 15, 130. [Google Scholar] [CrossRef] [PubMed]
- Whited, B.M.; Rylander, M.N. The influence of electrospun scaffold topography on endothelial cell morphology, alignment, and adhesion in response to fluid flow. Biotechnol. Bioeng. 2013, 111, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Rickel, A.P.; Deng, X.; Engebretson, D.; Hong, Z. Electrospun nanofiber scaffold for vascular tissue engineering. Mater. Sci. Eng. C 2021, 129, 112373. [Google Scholar] [CrossRef] [PubMed]
- Horan, R.L.; Antle, K.; Collette, A.L.; Wang, Y.; Huang, J.; Moreau, J.E.; Volloch, V.; Kaplan, D.L.; Altman, G.H. In vitro degradation of silk fibroin. Biomaterials 2005, 26, 3385–3393. [Google Scholar] [CrossRef]
- Soffer, L.; Wang, X.; Zhang, X.; Kluge, J.; Dorfmann, L.; Kaplan, D.L.; Leisk, G. Silk-Based Electrospun Tubular Scaffolds for Tissue-Engineered Vascular Grafts. J. Biomater. Sci. Polym. Ed. 2008, 19, 653. [Google Scholar] [CrossRef]
- Liu, H.; Li, X.; Zhou, G.; Fan, H.; Fan, Y. Electrospun sulfated silk fibroin nanofibrous scaffolds for vascular tissue engineering. Biomaterials 2011, 32, 3784–3793. [Google Scholar] [CrossRef]
- Xing, Y.; Gu, Y.; Guo, L.; Guo, J.; Xu, Z.; Xiao, Y.; Fang, Z.; Wang, C.; Feng, Z.G.; Wang, Z. Gelatin coating promotes in situ endothelialization of electrospun polycaprolactone vascular grafts. J. Biomater. Sci. Polym. Ed. 2021, 32, 1161–1181. [Google Scholar] [CrossRef]
- Mirmusavi, M.H.; Gonçalves, V.S.S.; Wischke, C. Gelatin-based porous scaffolds: Design concepts, production, and applications in precision regenerative medicine. Mater. Today Bio 2025, 36, 102710. [Google Scholar] [CrossRef]
- Ma, Z.; He, W.; Yong, T.; Ramakrishna, S. Grafting of gelatin on electrospun poly(caprolactone) nanofibers to improve endothelial cell spreading and proliferation and to control cell Orientation. Tissue Eng. 2005, 11, 1149–1158. [Google Scholar] [CrossRef]
- Fallon, M.E.; Le, H.H.; Bates, N.M.; Yao, Y.; Yim, E.K.F.; Hinds, M.T.; Anderson, D.E.J. Hemocompatibility of micropatterned biomaterial surfaces is dependent on topographical feature size. Front. Physiol. 2022, 13, 983187. [Google Scholar] [CrossRef]
- Camasão, D.B.; Mantovani, D. The mechanical characterization of blood vessels and their substitutes in the continuous quest for physiological-relevant performances. A critical review. Mater. Today Bio 2021, 10, 100106. [Google Scholar] [CrossRef]
- Jeong, Y.; Yao, Y.; Yim, E.K.F. Current understanding of intimal hyperplasia and effect of compliance in synthetic small diameter vascular grafts. Biomater. Sci. 2020, 8, 4383. [Google Scholar] [CrossRef] [PubMed]
- Schultz, G.S.; Chin, G.A.; Moldawer, L.; Diegelmann, R.F. Principles of Wound Healing. In Diabetic Foot Problems; Springer: Cham, Switzerland, 2011; pp. 395–402. [Google Scholar] [CrossRef]
- Kolimi, P.; Narala, S.; Nyavanandi, D.; Youssef, A.A.A.; Dudhipala, N. Innovative Treatment Strategies to Accelerate Wound Healing: Trajectory and Recent Advancements. Cells 2022, 11, 2439. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.; Jadhav, S.S. The application of mesenchymal stem cells to treat thermal and radiation burns. Adv. Drug Deliv. Rev. 2018, 123, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Rieger, K.A.; Birch, N.P.; Schiffman, J.D. Designing electrospun nanofiber mats to promote wound healing—A review. J. Mater. Chem. B 2013, 1, 4531–4541. [Google Scholar] [CrossRef]
- Fookes, F.; Bongiovanni Abel, S.; Martucci, J.F.; Estenoz, D.; Abraham, G.A.; Busatto, C.A. Expanded Nanofibrous Polymeric Mats Incorporating Tetracycline-Loaded Silica Mesoporous Nanoparticles for Antimicrobial Applications. Pharmaceutics 2025, 17, 1335. [Google Scholar] [CrossRef]
- Ekram, B. Review: Functionalization of biopolymer-based electrospun nanofibers for wound healing. J. Mater. Sci. 2025, 60, 8308–8342. [Google Scholar] [CrossRef]
- Ho, T.T.P.; Tran, H.A.; Doan, V.K.; Maitz, J.; Li, Z.; Wise, S.G.; Lim, K.S.; Rnjak-Kovacina, J. Natural Polymer-Based Materials for Wound Healing Applications. Adv. Nanobiomed Res. 2024, 4, 2300131. [Google Scholar] [CrossRef]
- Bhattarai, N.; Edmondson, D.; Veiseh, O.; Matsen, F.A.; Zhang, M. Electrospun chitosan-based nanofibers and their cellular compatibility. Biomaterials 2005, 26, 6176–6184. [Google Scholar] [CrossRef]
- Platon, V.M.; Andreica, B.I.; Anisiei, A.; Rosca, I.; Sandu, I.A.; Mititelu Tartau, L.; Marin, L. Antioxidant-sealed chitosan nanofibers loaded with erythromycin: Tissue regeneration biomaterials with immune-modulating effects in a rat model. Carbohydr. Polym. 2025, 368, 124247. [Google Scholar] [CrossRef]
- Witkowska, K.; Paczkowska-Walendowska, M.; Nagalska, M.; Miklaszewski, A.; Karpiński, T.M.; Plech, T.; Espinar, F.J.O.; Cielecka-Piontek, J. Chitosan/PEO Nanofibers as a Delivery Platform for Sustained Release of Centella asiatica Extract. Int. J. Mol. Sci. 2025, 26, 12134. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Hossain, S.; Mohanty, S.; Gupta, M.K.; Verma, D. Fast acting hemostatic agent based on self-assembled hybrid nanofibers from chitosan and casein. Int. J. Biol. Macromol. 2021, 185, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef]
- Sionkowska, A.; Wisniewski, M.; Skopinska, J.; Kennedy, C.J.; Wess, T.J. Molecular interactions in collagen and chitosan blends. Biomaterials 2004, 25, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Kusnadi, K.; Herdiana, Y.; Rochima, E.; Putra, O.N.; Gazzali, A.M.; Muchtaridi, M. Collagen-Based Nanoparticles as Drug Delivery System in Wound Healing Applications. Int. J. Nanomed. 2024, 19, 11321–11341. [Google Scholar] [CrossRef]
- Arun, A.; Malrautu, P.; Laha, A.; Ramakrishna, S. Gelatin Nanofibers in Drug Delivery Systems and Tissue Engineering. Eng. Sci. 2021, 16, 71–81. [Google Scholar] [CrossRef]
- Ndlovu, S.P.; Ngece, K.; Alven, S.; Aderibigbe, B.A. Gelatin-Based Hybrid Scaffolds: Promising Wound Dressings. Polymers 2021, 13, 2959. [Google Scholar] [CrossRef]
- Chong, E.J.; Phan, T.T.; Lim, I.J.; Zhang, Y.Z.; Bay, B.H.; Ramakrishna, S.; Lim, C.T. Evaluation of electrospun PCL/gelatin nanofibrous scaffold for wound healing and layered dermal reconstitution. Acta Biomater. 2007, 3, 321–330. [Google Scholar] [CrossRef]
- Mohite, P.; Puri, A.; Munde, S.; Dave, R.; Khan, S.; Patil, R.; Singh, A.K.; Tipduangta, P.; Singh, S.; Chittasupho, C. Potential of Chitosan/Gelatin-Based Nanofibers in Delivering Drugs for the Management of Varied Complications: A Review. Polymers 2025, 17, 435. [Google Scholar] [CrossRef]
- Gautam, S.; Chou, C.F.; Dinda, A.K.; Potdar, P.D.; Mishra, N.C. Fabrication and characterization of PCL/gelatin/chitosan ternary nanofibrous composite scaffold for tissue engineering applications. J. Mater. Sci. 2014, 49, 1076–1089. [Google Scholar] [CrossRef]
- Campodoni, E.; Montanari, M.; Dozio, S.M.; Heggset, E.B.; Panseri, S.; Montesi, M.; Tampieri, A.; Syverud, K.; Sandri, M. Blending gelatin and cellulose nanofibrils: Biocomposites with tunable degradability and mechanical behavior. Nanomaterials 2020, 10, 1219. [Google Scholar] [CrossRef] [PubMed]
- Pastrana-Alta, R.Y.; Huarote-Garcia, E.; Egusquiza-Huamani, M.A.; Baena-Moncada, A.M. Antimicrobial activity of chitosan, alginate, pectin, and cellulose-based biopolymer composites with silver, copper oxide, and zinc oxide nanoparticles. RSC Adv. 2025, 15, 35807. [Google Scholar] [CrossRef] [PubMed]
- Datta, D.; Bandi, S.P.; Colaco, V.; Dhas, N.; Saha, S.S.; Hussain, S.Z.; Singh, S. Cellulose-Based Nanofibers Infused with Biotherapeutics for Enhanced Wound-Healing Applications. ACS Polym. Au 2025, 5, 80–104. [Google Scholar] [CrossRef] [PubMed]
- Nuutila, K.; Eriksson, E. Moist Wound Healing with Commonly Available Dressings. Adv. Wound Care 2021, 10, 685. [Google Scholar] [CrossRef]
- Kasoju, N.; Bora, U. Silk fibroin in tissue engineering. Adv. Healthc. Mater. 2012, 1, 393–412. [Google Scholar] [CrossRef]
- Yeo, I.S.; Oh, J.E.; Jeong, L.; Lee, T.S.; Lee, S.J.; Park, W.H.; Min, B.M. Collagen-Based Biomimetic Nanofibrous Scaffolds: Preparation and Characterization of Collagen/Silk Fibroin Bicomponent Nanofibrous Structures. Biomacromolecules 2008, 9, 1106–1116. [Google Scholar] [CrossRef]
- Jeong, S.I.; Krebs, M.D.; Bonino, C.A.; Khan, S.A.; Alsberg, E. Electrospun alginate nanofibers with controlled cell adhesion for tissue engineering. Macromol. Biosci. 2010, 10, 934–943. [Google Scholar] [CrossRef]
- Humaira; Raza Bukhari, S.A.; Shakir, H.A.; Khan, M.; Saeed, S.; Ahmad, I.; Muzammil, K.; Franco, M.; Irfan, M.; Li, K. Hyaluronic acid-based nanofibers: Electrospun synthesis and their medical applications; recent developments and future perspective. Front. Chem. 2022, 10, 1092123. [Google Scholar] [CrossRef]
- Emami, J.; Mostolizadeh, N.; Tabbakhian, M.; Heydari, P.; Kharazi, A.Z.; Minaeiyan, M.; Hasanzadeh, F.; Mirian, M.; Talebi, A. Development and evaluation of gelatin/hyaluronic acid nanofibrous dressing loaded with silver nanoparticles and phenytoin for enhanced wound healing: An in-vitro and in-vivo study. Res. Pharm. Sci. 2025, 20, 610–634. [Google Scholar] [CrossRef]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Adv. Drug Deliv. Rev. 2016, 107, 206–212. [Google Scholar] [CrossRef]
- Zhou, C.; Shi, Q.; Guo, W.; Terrell, L.; Qureshi, A.T.; Hayes, D.J.; Wu, Q. Electrospun bio-nanocomposite scaffolds for bone tissue engineering by cellulose nanocrystals reinforcing maleic anhydride grafted PLA. ACS Appl. Mater. Interfaces 2013, 5, 3847–3854. [Google Scholar] [CrossRef]
- Janmohammadi, M.; Nourbakhsh, M.S. Electrospun polycaprolactone scaffolds for tissue engineering: A review. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 527–539. [Google Scholar] [CrossRef]
- Zeybek, B.; Duman, M. Electrospinning of nanofibrous polycaprolactone (PCL) and collagen-blended polycaprolactone for wound dressing and tissue engineering. Usak Univ. J. Mater. Sci. 2014, 3, 121–134. [Google Scholar] [CrossRef]
- Teixeira, M.A.; Amorim, M.T.P.; Felgueiras, H.P. Poly(Vinyl Alcohol)-Based Nanofibrous Electrospun Scaffolds for Tissue Engineering Applications. Polymers 2019, 12, 7. [Google Scholar] [CrossRef] [PubMed]
- Dev, A.S.; Mohan, N.; Mohan, R.; Boccaccini, A.R. Chitosan-based composite scaffolds for accelerated epidermal-dermal wound healing. Open Explor. 2025, 2, 101336. [Google Scholar] [CrossRef]
- Yilmaz Atay, H. Antibacterial Activity of Chitosan-Based Systems. In Functional Chitosan; Springer: Singapore, 2020; p. 457. [Google Scholar] [CrossRef]
- Hemmingsen, L.M.; Škalko-Basnet, N.; Jøraholmen, M.W. The Expanded Role of Chitosan in Localized Antimicrobial Therapy. Mar. Drugs 2021, 19, 697. [Google Scholar] [CrossRef]
- Bielska, B.; Miłowska, K. Therapeutic Potential of Chitosan-Based and Related Nanocomposite Systems in Wound Management: A Review. Int. J. Mol. Sci. 2025, 26, 11748. [Google Scholar] [CrossRef]
- Mertz, P.M.; Davis, S.C.; Franzen, L.; Uchima, F.D.; Pickett, M.P.; Pierschbacher, M.D.; Polarek, J.W. Effects of an arginine-glycine-aspartic acid peptide-containing artificial matrix on epithelial migration in vitro and experimental second-degree burn wound healing in vivo. J. Burn Care Rehabil. 1996, 17, 199–206. [Google Scholar] [CrossRef]
- de Farias, B.S.; Christ Ribeiro, A.; Jaeschke, D.P.; Ribeiro, E.S.; Gonçalves, J.O.; Vergara, R.F.; Fernandes, S.S.; Dias, D.; Cadaval, T.R.S.; de Almeida Pinto, L.A. Recent Trends in Gelatin Electrospun Nanofibers: Advances in Fabrication, Functionalization, and Applications. Coatings 2025, 15, 1110. [Google Scholar] [CrossRef]
- Kang, J., II; Park, K.M. Advances in gelatin-based hydrogels for wound management. J. Mater. Chem. B 2021, 9, 1503–1520. [Google Scholar] [CrossRef]
- Amirrah, I.N.; Lokanathan, Y.; Zulkiflee, I.; Wee, M.F.M.R.; Motta, A.; Fauzi, M.B. A Comprehensive Review on Collagen Type I Development of Biomaterials for Tissue Engineering: From Biosynthesis to Bioscaffold. Biomedicines 2022, 10, 2307. [Google Scholar] [CrossRef] [PubMed]
- Elango, J.; Hou, C.; Bao, B.; Wang, S.; Maté Sánchez de Val, J.E.; Wenhui, W. The Molecular Interaction of Collagen with Cell Receptors for Biological Function. Polymers 2022, 14, 876. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Wang, N.; Xue, Y.; Ding, T.; Liu, X.; Mo, X.; Sun, J. Electrospun tilapia collagen nanofibers accelerating wound healing via inducing keratinocytes proliferation and differentiation. Colloids Surf. B Biointerfaces 2016, 143, 415–422. [Google Scholar] [CrossRef]
- Mbese, Z.; Alven, S.; Aderibigbe, B.A. Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications. Polymers 2021, 13, 4368. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, S.R.Z.; Dos Reis, E.M.; Apati, G.P.; Meier, M.M.; Nogueira, A.L.; Formolo Garcia, M.C.; Dos Santos Schneider, A.L.; Testa Pezzin, A.P.; Porto, L.M. Biosynthesis and Functionalization of Bacterial Cellulose Membranes with Cerium Nitrate and Silver Nanoparticles. Mater. Res. 2019, 22, e20190054. [Google Scholar] [CrossRef]
- Fahma, F.; Firmanda, A.; Cabral, J.; Pletzer, D.; Fisher, J.; Mahadik, B.; Arnata, I.W.; Sartika, D.; Wulandari, A. Three-Dimensional Printed Cellulose for Wound Dressing Applications. 3D Print. Addit. Manuf. 2023, 10, 1015–1035. [Google Scholar] [CrossRef]
- Teixeira, M.A.; Paiva, M.C.; Amorim, M.T.P.; Felgueiras, H.P. Electrospun Nanocomposites Containing Cellulose and Its Derivatives Modified with Specialized Biomolecules for Enhanced Wound Healing. Nanomaterials 2020, 10, 557. [Google Scholar] [CrossRef]
- Hon, D.N.-S. Chemical Modification of Cellulose. In Chemical Modification of Lignocellulosic Materials; Routledge: Oxfordshire, UK, 2017; pp. 97–127. [Google Scholar] [CrossRef]
- Marinho, E. Cellulose: A comprehensive review of its properties and applications. Sustain. Chem. Environ. 2025, 11, 100283. [Google Scholar] [CrossRef]
- Masoud, A.R.; Velisdeh, Z.J.; Bappy, M.J.P.; Pandey, G.; Saberian, E.; Mills, D.K. Cellulose-Based Nanofibers in Wound Dressing. Biomimetics 2025, 10, 344. [Google Scholar] [CrossRef]
- Martinek, J.; Polomíková, L.; Kudláček, M.; Navrátilová, J.; Mokrejš, P.; Pavlačková, J.; Gál, R. Effect of UV Exposure Time on the Properties of Films Prepared from Biotechnologically Derived Chicken Gelatin. Processes 2025, 13, 91. [Google Scholar] [CrossRef]
- Torres-Giner, S.; Gimeno-Alcañiz, J.V.; Ocio, M.J.; Lagaron, J.M. Comparative Performance of Electrospun Collagen Nanofibers Crosslinked by Means of Different Methods. ACS Appl. Mater. Interfaces 2008, 1, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Rath, G.; Hussain, T.; Chauhan, G.; Garg, T.; Goyal, A.K. Collagen nanofiber containing silver nanoparticles for improved wound-healing applications. J. Drug Target. 2016, 24, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, J.; Padhy, R.; Arun, J.; Murthannagari, V.R.; Ganesh, G. Stimuli-Responsive Drug Delivery Systems: Extensive Overview. Int. J. Appl. Pharm. 2025, 17, 94–106. [Google Scholar] [CrossRef]
- Khrystonko, O.; Rimpelová, S.; Burianová, T.; Švorčík, V.; Lyutakov, O.; Elashnikov, R. Smart multi stimuli-responsive electrospun nanofibers for on-demand drug release. J. Colloid Interface Sci. 2023, 648, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Weng, L.; Xie, J. Smart Electrospun Nanofibers for Controlled Drug Release: Recent Advances and New Perspectives. Curr. Pharm. Des. 2015, 21, 1944–1959. [Google Scholar] [CrossRef]
- Schoeller, J.; Itel, F.; Wuertz-Kozak, K.; Fortunato, G.; Rossi, R.M. pH-Responsive Electrospun Nanofibers and Their Applications. Polym. Rev. 2022, 62, 351–399. [Google Scholar] [CrossRef]
- Wu, J.; Qin, Z.; Jiang, X.; Fang, D.; Lu, Z.; Zheng, L.; Zhao, J. ROS-responsive PPGF nanofiber membrane as a drug delivery system for long-term drug release in attenuation of osteoarthritis. NPJ Regen. Med. 2022, 7, 66. [Google Scholar] [CrossRef]
- Agiba, A.M.; Elsayyad, N.; ElShagea, H.N.; Metwalli, M.A.; Mahmoudsalehi, A.O.; Beigi-Boroujeni, S.; Lozano, O.; Aguirre-Soto, A.; Arreola-Ramirez, J.L.; Segura-Medina, P.; et al. Advances in Light-Responsive Smart Multifunctional Nanofibers: Implications for Targeted Drug Delivery and Cancer Therapy. Pharmaceutics 2024, 16, 1017. [Google Scholar] [CrossRef]
- Singh, B.; Shukla, N.; Kim, J.; Kim, K.; Park, M.H. Stimuli-Responsive Nanofibers Containing Gold Nanorods for On-Demand Drug Delivery Platforms. Pharmaceutics 2021, 13, 1319. [Google Scholar] [CrossRef]
- Alli, Y.A.; Bamisaye, A.; Ige, A.S.; Elabor, R.; Ife-Adediran, O.; Samson, A.O.; Oni, S.O.; Yeboah, A.; Bankole, O.M.; Fapojuwo, D.P.; et al. Coaxial electrospinning as a promising technique for fabricating advanced materials for energy storage applications. Discov. Appl. Sci. 2025, 8, 88. [Google Scholar] [CrossRef]
- Sree, S.; Patra, A.; Arun Prasath, V.; Kambhampati, V.; Xiao, H.-W. Innovations in Electrospinning Techniques for Nanomaterial Synthesis and its applications in the field of Active Food Packaging. J. Future Foods, 2025; in press. [CrossRef]
- Hulanicki, A.; Glab, S.; Ingman, F. Chemical sensors definitions and classification. Pure Appl. Chem. 1991, 63, 1247–1250. [Google Scholar] [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef] [PubMed]
- Abadi, B.; Goshtasbi, N.; Bolourian, S.; Tahsili, J.; Adeli-Sardou, M.; Forootanfar, H. Electrospun hybrid nanofibers: Fabrication, characterization, and biomedical applications. Front. Bioeng. Biotechnol. 2022, 10, 986975. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.O.; Singh, B. Electrochemical Biosensors for Detection of Pesticides and Heavy Metal Toxicants in Water: Recent Trends and Progress. ACS EST Water 2021, 1, 462–478. [Google Scholar] [CrossRef]
- Ali, A.A.; Altemimi, A.B.; Alhelfi, N.; Ibrahim, S.A. Application of Biosensors for Detection of Pathogenic Food Bacteria: A Review. Biosensors 2020, 10, 58. [Google Scholar] [CrossRef]
- Li, P.; Lee, G.H.; Kim, S.Y.; Kwon, S.Y.; Kim, H.R.; Park, S. From Diagnosis to Treatment: Recent Advances in Patient-Friendly Biosensors and Implantable Devices. ACS Nano 2021, 15, 1960–2004. [Google Scholar] [CrossRef]
- Mercante, L.A.; Pavinatto, A.; Pereira, T.S.; Migliorini, F.L.; dos Santos, D.M.; Correa, D.S. Nanofibers interfaces for biosensing: Design and applications. Sens. Actuators Rep. 2021, 3, 100048. [Google Scholar] [CrossRef]
- Mercante, L.A.; Scagion, V.P.; Migliorini, F.L.; Mattoso, L.H.C.; Correa, D.S. Electrospinning-based (bio)sensors for food and agricultural applications: A review. TrAC Trends Anal. Chem. 2017, 91, 91–103. [Google Scholar] [CrossRef]
- Yezer, I.; Demirkol, D.O. Cellulose acetate–chitosan based electrospun nanofibers for bio-functionalized surface design in biosensing. Cellulose 2020, 27, 10183–10197. [Google Scholar] [CrossRef]
- Ratlam, C.; Phanichphant, S.; Sriwichai, S. Development of dopamine biosensor based on polyaniline/carbon quantum dots composite. J. Polym. Res. 2020, 27, 183. [Google Scholar] [CrossRef]
- Fathi, S.; Saber, R.; Adabi, M.; Rasouli, R.; Douraghi, M.; Morshedi, M.; Farid-Majidi, R. Novel competitive voltammetric aptasensor based on electrospun carbon nanofibers-gold nanoparticles modified graphite electrode for Salmonella enterica serovar detection. Biointerface Res. Appl. Chem. 2021, 11, 8702–8715. [Google Scholar] [CrossRef]
- Kilic, N.M.; Gelen, S.S.; Er Zeybekler, S.; Odaci, D. Carbon-Based Nanomaterials Decorated Electrospun Nanofibers in Biosensors: A Review. ACS Omega 2023, 9, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Kanjwal, M.A.; Ghaferi, A. Al: Graphene Incorporated Electrospun Nanofiber for Electrochemical Sensing and Biomedical Applications: A Critical Review. Sensors 2022, 22, 8661. [Google Scholar] [CrossRef]
- Yang, G.; Kampstra, K.L.; Abidian, M.R. High-performance conducting polymer nanofiber biosensors for detection of biomolecules. Adv. Mater. 2014, 26, 4954–4960. [Google Scholar] [CrossRef]
- Ming, D.K.; Sangkaew, S.; Chanh, H.Q.; Nhat, P.T.H.; Yacoub, S.; Georgiou, P.; Holmes, A.H. Continuous physiological monitoring using wearable technology to inform individual management of infectious diseases, public health and outbreak responses. Int. J. Infect. Dis. 2020, 96, 648. [Google Scholar] [CrossRef]
- Dunn, J.; Runge, R.; Snyder, M. Wearables and the Medical Revolution. Per. Med. 2018, 15, 429–448. [Google Scholar] [CrossRef]
- Zhao, Y.S.; Huang, J.; Yang, X.; Wang, W.; Yu, D.G.; He, H.; Liu, P.; Du, K. Electrospun nanofibers and their application as sensors for healthcare. Front. Bioeng. Biotechnol. 2025, 13, 1533367. [Google Scholar] [CrossRef]
- Mpofu, N.S.; Blachowicz, T.; Ehrmann, A.; Ehrmann, G. Wearable Electrospun Nanofibrous Sensors for Health Monitoring. Micro 2024, 4, 798–822. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chiu, C.W. Facile Fabrication of a Stretchable and Flexible Nanofiber Carbon Film-Sensing Electrode by Electrospinning and Its Application in Smart Clothing for ECG and EMG Monitoring. ACS Appl. Electron. Mater. 2021, 3, 676–686. [Google Scholar] [CrossRef]
- Li, J.W.; Huang, B.S.; Chang, C.H.; Chiu, C.W. Advanced electrospun AgNPs/rGO/PEDOT:PSS/TPU nanofiber electrodes: Stretchable, self-healing, and perspiration-resistant wearable devices for enhanced ECG and EMG monitoring. Adv. Compos. Hybrid Mater. 2023, 6, 231. [Google Scholar] [CrossRef]
- Liu, T.; Qu, D.; Guo, L.; Zhou, G.; Zhang, G.; Du, T.; Wu, W. MXene/TPU Composite Film for Humidity Sensing and Human Respiration Monitoring. Adv. Sens. Res. 2024, 3, 2300014. [Google Scholar] [CrossRef]
- Sengupta, D.; Romano, J.; Kottapalli, A.G.P. Electrospun bundled carbon nanofibers for skin-inspired tactile sensing, proprioception, and gesture tracking applications. npj Flex. Electron. 2021, 5, 29. [Google Scholar] [CrossRef]
- Lin, M.F.; Cheng, C.; Yang, C.C.; Hsiao, W.T.; Yang, C.R. A wearable and highly sensitive capacitive pressure sensor integrated with a dual-layer dielectric layer of PDMS microcylinder array and PVDF electrospun fiber. Org. Electron. 2021, 98, 106290. [Google Scholar] [CrossRef]
- Li, X.; Zhang, S.; Li, K.; Yang, Z.; Hu, X.; Zhang, J.; Zhang, D.; Zhang, C.; Liu, Y. Electrospun Micro/Nanofiber-Based Biomechanical Sensors. ACS Appl. Polym. Mater. 2023, 5, 6720–6746. [Google Scholar] [CrossRef]
- Li, X.; Zhuang, Z.; Qi, D.; Zhao, C. High sensitive and fast response humidity sensor based on polymer composite nanofibers for breath monitoring and non-contact sensing. Sens. Actuators B Chem. 2021, 330, 129239. [Google Scholar] [CrossRef]
- Kakria, P.; Tripathi, N.K.; Kitipawang, P. A Real-Time Health Monitoring System for Remote Cardiac Patients Using Smartphone and Wearable Sensors. Int. J. Telemed. Appl. 2015, 2015, 373474. [Google Scholar] [CrossRef]
- Yadav, S.; Yadav, S.; Chaturvedi, V.; Verma, P.; Rajput, A.; Chaubey, R. Telemedicine and Its Role in Innovating the Provision of Healthcare; Springer: Cham, Switzerland, 2022; pp. 1–11. [Google Scholar] [CrossRef]
- Flodgren, G.; Rachas, A.; Farmer, A.J.; Inzitari, M.; Shepperd, S. Interactive telemedicine: Effects on professional practice and health care outcomes. Cochrane Database Syst. Rev. 2015, 12, CD002098. [Google Scholar] [CrossRef]
- Jeyanthi, P.; Gulothungan, G.; Kumar, V.V.; Chopra, H.; Emran, T. Bin: Enhancing postoperative care with telemedicine and remote monitoring for improved recovery and patient safety. Int. J. Surg. 2024, 110, 8205. [Google Scholar] [CrossRef]
- Wang, Q.P.; Chang, W.Y.; Han, M.M.; Hu, Y.X.; Lin, S.S.; Gu, Y.C. Application of telemedicine system for older adults postoperative patients in community: A feasibility study. Front. Public Health 2024, 12, 1291916. [Google Scholar] [CrossRef]
- Creber, A.; Leo, D.G.; Buckley, B.J.R.; Chowdhury, M.; Harrison, S.L.; Isanejad, M.; Lane, D.A. Use of telemonitoring in patient self-management of chronic disease: A qualitative meta-synthesis. BMC Cardiovasc. Disord. 2023, 23, 469. [Google Scholar] [CrossRef] [PubMed]
- Kalash, R.S.; Lakshmanan, V.K.; Cho, C.S.; Park, I.K. Theranostics. In Biomaterials Nanoarchitectonics; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 197–215. [Google Scholar] [CrossRef]
- Al-Thani, A.N.; Jan, A.G.; Abbas, M.; Geetha, M.; Sadasivuni, K.K. Nanoparticles in cancer theragnostic and drug delivery: A comprehensive review. Life Sci. 2024, 352, 122899. [Google Scholar] [CrossRef]
- Kashyap, B.K.; Singh, V.V.; Solanki, M.K.; Kumar, A.; Ruokolainen, J.; Kesari, K.K. Smart Nanomaterials in Cancer Theranostics: Challenges and Opportunities. ACS Omega 2023, 8, 14290–14320. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Sun, C.; Jin, K.; Dong, C. Theranostic Role of Advanced Nanotechnological Tools in Early Brain Metastases in Lung Cancer: An Updated Review. Int. J. Nanomed. 2025, 20, 7215. [Google Scholar] [CrossRef] [PubMed]
- Soares, P.I.P.; Borges, J.P. Recent advances in magnetic electrospun nanofibers for cancer theranostics application. Prog. Nat. Sci. Mater. Int. 2021, 31, 835–844. [Google Scholar] [CrossRef]
- Nikolaou, M.; Avraam, K.; Kolokithas-Ntoukas, A.; Bakandritsos, A.; Lizal, F.; Misik, O.; Maly, M.; Jedelsky, J.; Savva, I.; Balanean, F.; et al. Superparamagnetic electrospun microrods for magnetically guided pulmonary drug delivery with magnetic heating. Mater. Sci. Eng. C 2021, 126, 112117. [Google Scholar] [CrossRef]
- Matos, R.J.R.; Chaparro, C.I.P.; Silva, J.C.; Valente, M.A.; Borges, J.P.; Soares, P.I.P. Electrospun composite cellulose acetate/iron oxide nanoparticles non-woven membranes for magnetic hyperthermia applications. Carbohydr. Polym. 2018, 198, 9–16. [Google Scholar] [CrossRef]
- Abdel Khalek, M.A.; Abdelhameed, A.M.; Abdel Gaber, S.A. The Use of Photoactive Polymeric Nanoparticles and Nanofibers to Generate a Photodynamic-Mediated Antimicrobial Effect, with a Special Emphasis on Chronic Wounds. Pharmaceutics 2024, 16, 229. [Google Scholar] [CrossRef]
- Abdel Khalek, M.A.; Abdel Gaber, S.A.; El-Domany, R.A.; El-Kemary, M.A. Photoactive electrospun cellulose acetate/polyethylene oxide/methylene blue and trilayered cellulose acetate/polyethylene oxide/silk fibroin/ciprofloxacin nanofibers for chronic wound healing. Int. J. Biol. Macromol. 2021, 193, 1752–1766. [Google Scholar] [CrossRef]
- Sun, J.; Fan, Y.; Ye, W.; Tian, L.; Niu, S.; Ming, W.; Zhao, J.; Ren, L. Near-infrared light triggered photodynamic and nitric oxide synergistic antibacterial nanocomposite membrane. Chem. Eng. J. 2021, 417, 128049. [Google Scholar] [CrossRef]
- Ghanavi, J.; Farnia, P.; Velayati, A.A. Nano design of extracellular matrix for tissue engineering. In Nanoarchitectonics in Biomedicine; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 547–583. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, X.; Guo, Q.; Wu, T.; Wang, Y. 3D Bioprinted Scaffolds for Tissue Repair and Regeneration. Front. Mater. 2022, 9, 925321. [Google Scholar] [CrossRef]
- Vaseashta, A.; Demir, D.; Sakım, B.; Aşık, M.; Bölgen, N. Hierarchical Integration of 3D Printing and Electrospinning of Nanofibers for Rapid Prototyping. In Electrospun Nanofibers; Vaseashta, A., Bölgen, N., Eds.; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Chen, W.; Xu, Y.; Liu, Y.; Wang, Z.; Li, Y.; Jiang, G.; Mo, X.; Zhou, G. Three-dimensional printed electrospun fiber-based scaffold for cartilage regeneration. Mater. Des. 2019, 179, 107886. [Google Scholar] [CrossRef]
- Choi, W.S.; Kim, J.H.; Ahn, C.B.; Lee, J.H.; Kim, Y.J.; Son, K.H.; Lee, J.W. Development of a multi-layer skin substitute using human hair keratinic extract-based hybrid 3d printing. Polymers 2021, 13, 2584. [Google Scholar] [CrossRef] [PubMed]
- Khalid Abdullah, K.; Molnár, K. Current Trends and Future Prospects of Integrating Electrospinning With 3D Printing Techniques for Mimicking Bone Extracellular Matrix Scaffolds. J. Polym. Sci. 2025, 63, 1481–1504. [Google Scholar] [CrossRef]
- Sanchaniya, J.V.; Smogor, H.; Gobins, V.; Noël, V.; Lasenko, I.; Rackauskas, S. Layer-by-Layer Integration of Electrospun Nanofibers in FDM 3D Printing for Hierarchical Composite Fabrication. Polymers 2025, 18, 78. [Google Scholar] [CrossRef]
- Pham, Q.P.; Sharma, U.; Mikos, A.G. Electrospinning of polymeric nanofibers for tissue engineering applications: A review. Tissue Eng. 2006, 12, 1197–1211. [Google Scholar] [CrossRef]
- Brown, T.D.; Dalton, P.D.; Hutmacher, D.W. Direct Writing By Way of Melt Electrospinning. Adv. Mater. 2011, 23, 5651–5657. [Google Scholar] [CrossRef]
- Cheng, J.; Jun, Y.; Qin, J.; Lee, S.H. Electrospinning versus microfluidic spinning of functional fibers for biomedical applications. Biomaterials 2017, 114, 121–143. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, L.; Ma, A.; Bai, X.; Zeng, Y.; Liu, D.; Liu, B.; Zhang, W.; Tang, S. Recent advances in coaxial electrospun nanofibers for wound healing. Mater. Today Bio 2024, 29, 101309. [Google Scholar] [CrossRef]
- Bhattarai, R.S.; Bachu, R.D.; Boddu, S.H.S.; Bhaduri, S. Biomedical Applications of Electrospun Nanofibers: Drug and Nanoparticle Delivery. Pharmaceutics 2018, 11, 5. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Karbhari, V.; Shaikh, N.S.; Chillarge, S.; Sirsat, V. Electrospun Nanofibers as Advanced Platforms for Skin Drug Delivery: Formulation Strategies, and Translational Challenges. Int. J. Pharm. Sci. 2026, 4, 1389–1401. [Google Scholar] [CrossRef]
- Venmathi Maran, B.A.; Jeyachandran, S.; Kimura, M. A Review on the Electrospinning of Polymer Nanofibers and Its Biomedical Applications. J. Compos. Sci. 2024, 8, 32. [Google Scholar] [CrossRef]
- Hadisi, Z.; Farokhi, M.; Bakhsheshi-Rad, H.R.; Jahanshahi, M.; Hasanpour, S.; Pagan, E.; Dolatshahi-Pirouz, A.; Zhang, Y.S.; Kundu, S.C.; Akbari, M. Hyaluronic Acid (HA)-Based Silk Fibroin/Zinc Oxide Core-Shell Electrospun Dressing for Burn Wound Management. Macromol. Biosci. 2020, 20, 1900328. [Google Scholar] [CrossRef]
- Maduna, L.; Patnaik, A. Challenges Associated with the Production of Nanofibers. Processes 2024, 12, 2100. [Google Scholar] [CrossRef]
- Yalcinkaya, B.; Buzgo, M. A Guide for Industrial Needleless Electrospinning of Synthetic and Hybrid Nanofibers. Polymers 2025, 17, 3019. [Google Scholar] [CrossRef]
- Beaudoin, É.J.; Kubaski, M.M.; Samara, M.; Zednik, R.J.; Demarquette, N.R. Scaled-Up Multi-Needle Electrospinning Process Using Parallel Plate Auxiliary Electrodes. Nanomaterials 2022, 12, 1356. [Google Scholar] [CrossRef]
- Chen, Y.; Xinsong, L.; Tangying, S. Electrospinning and crosslinking of zein nanofiber mats. J. Appl. Polym. Sci. 2007, 103, 380–385. [Google Scholar] [CrossRef]
- Lhotská, I.; Kholová, A.; Švec, F.; Šatínský, D. Nanofibers prepared from synthetic polymers and biopolymers as advanced extraction materials for sample preparation prior to liquid chromatography. TrAC Trends Anal. Chem. 2024, 180, 117912. [Google Scholar] [CrossRef]
- Delgado, L.M.; Fuller, K.; Zeugolis, D.I. Influence of Cross-Linking Method and Disinfection/Sterilization Treatment on the Structural, Biophysical, Biochemical, and Biological Properties of Collagen-Based Devices. ACS Biomater. Sci. Eng. 2018, 4, 2739–2747. [Google Scholar] [CrossRef]
- Amadori, S.; Torricelli, P.; Rubini, K.; Fini, M.; Panzavolta, S.; Bigi, A. Effect of sterilization and crosslinking on gelatin films. J. Mater. Sci. Mater. Med. 2015, 26, 69. [Google Scholar] [CrossRef]
- Tian, J.; Song, X.; Wang, Y.; Cheng, M.; Lu, S.; Xu, W.; Gao, G.; Sun, L.; Tang, Z.; Wang, M.; et al. Regulatory perspectives of combination products. Bioact. Mater. 2021, 10, 492. [Google Scholar] [CrossRef]
- Reis, M.E.; Bettencourt, A.; Ribeiro, H.M. The regulatory challenges of innovative customized combination products. Front. Med. 2022, 9, 821094. [Google Scholar] [CrossRef]
- Li, W.; Feng, J.; Feng, G.; Feng, S. Wearable technology for athletes: Material innovations, performance monitoring, and emerging paradigms. Microchim. Acta 2026, 193, 76. [Google Scholar] [CrossRef]
- Gulgosteren, E.; Agrali Ermis, S.; Algin Toros, A.; Toros, T.; Serin, E.; Sekeroglu, M.O.; Bahadır Kayisoglu, N. Sweat, tears, and beyond: Advanced wearable sensors for personalized health and athletic performance. Front. Bioeng. Biotechnol. 2025, 13, 1684674. [Google Scholar] [CrossRef]
- Ravizza, A.; De Maria, C.; Di Pietro, L.; Sternini, F.; Audenino, A.L.; Bignardi, C. Comprehensive Review on Current and Future Regulatory Requirements on Wearable Sensors in Preclinical and Clinical Testing. Front. Bioeng. Biotechnol. 2019, 7, 495557. [Google Scholar] [CrossRef]
- Vo, D.K.; Trinh, K.T.L. Advances in Wearable Biosensors for Healthcare: Current Trends, Applications, and Future Perspectives. Biosensors 2024, 14, 560. [Google Scholar] [CrossRef]






| Biopolymer | Origin | Key Advantages | Key Limitations | Common Blending Partners | Ref. |
|---|---|---|---|---|---|
| Chitosan | Natural (chitin) | Antimicrobial, hemostatic, biocompatible, promotes cell proliferation | Difficult to electrospun alone; limited mechanical strength | PVA, PEO, PCL, gelatin | [118,119,120,121] |
| Collagen | Natural (protein) | Excellent ECM biomimicry; promotes cell adhesion/migration via RGD motifs | Weak mechanical properties; requires harsh solvents; rapid degradation | PCL, PLA, chitosan | [122,123,124] |
| Gelatin | Natural (collagen derivative) | Biocompatible; RGD motifs; low immunogenicity; good water retention | Poor mechanical strength; rapid aqueous dissolution; thermal instability | PLA, PCL, PLGA, PVA | [125,126,127,128,129,130] |
| Cellulose | Natural (polysaccharide) | High moisture retention, high structural stability | Limited bioactivity, difficult solubility for electrospinning; requires harsh solvents | PVA, PEO, gelatin, chitosan, PCL | [131,132,133] |
| Silk Fibroin | Natural (protein) | Exceptional mechanical strength/toughness; slow tunable degradation | Requires removal of sericin; limited bioactivity vs. ECM proteins | PEO, PCL, gelatin | [134,135] |
| Alginate | Natural (polysaccharide) | High water absorption (15–20×); promotes moist environment; autolytic debridement | Difficult to electrospun alone; poor mechanical properties | PEO, PVA, chitosan, collagen | [131,136] |
| Hyaluronic Acid | Natural (GAG) | Critical ECM component; exceptional hydration; regulates inflammation and angiogenesis | Difficult to electrospun alone; rapid enzymatic degradation | PVA, PCL, gelatin, PEO | [137,138] |
| PLA | Synthetic (polyester) | FDA-approved; good mechanical properties; controllable degradation (6–24 months) | Hydrophobic; lacks cell recognition sites; acidic degradation products | Gelatin, chitosan, collagen | [139,140] |
| PCL | Synthetic (polyester) | FDA-approved; excellent flexibility; slow degradation (2–4 years); easy to electrospun | Hydrophobic; poor cell adhesion without modification | Chitosan, collagen, gelatin, HA | [141,142] |
| PVA | Synthetic (vinyl polymer) | Water-soluble; non-toxic; excellent co-spinning agent; aqueous processing | Rapid dissolution without crosslinking; limited standalone mechanical properties | Chitosan, alginate, HA, proteins | [143] |
| Nanofiber Composition | Bioactive Agent(s) | Key Functionalities | Key Findings | Ref. |
|---|---|---|---|---|
| PCL/Chitosan | Silver nanoparticles | Antimicrobial, hemostatic | Broad-spectrum activity against S. aureus and P. aeruginosa; ~95% wound closure in 14 days in the rat model | [224] |
| Gelatin/PLA | Curcumin | Anti-inflammatory, antioxidant | Sustained curcumin release over 14 days; reduced TNF-α and IL-6 levels; enhanced fibroblast proliferation | [225] |
| PCL/Collagen (core–shell) | VEGF + bFGF (dual release) | Pro-angiogenic, regenerative | Sequential growth factor release; 3-fold increase in blood vessel density; complete wound closure by day 12 | [226] |
| Silk Fibroin/PEO | EGF | Re-epithelialization, ECM mimicry | Sustained EGF release for 21 days; 2-fold increase in keratinocyte migration rate; thicker neo-epidermis | [227] |
| Chitosan/PVA | ZnO nanoparticles + aloe vera | Antimicrobial, anti-inflammatory, hydrating | Synergistic antibacterial effect; reduced inflammation; ~90% wound closure in 10 days | [224] |
| PCL/Gelatin | Dexamethasone (pH-responsive shell) | Smart anti-inflammatory release | Drug release triggered at alkaline pH (infection); 60% reduction in inflammatory infiltrate | [228] |
| PVA/Alginate | Honey + tetracycline | Antimicrobial, autolytic debridement | Dual-action dressing; effective against biofilm-forming bacteria; enhanced moist wound environment | [166] |
| PLGA/HA | PDGF | Fibroblast recruitment, ECM synthesis | Controlled PDGF release for 28 days; 40% increase in collagen type I deposition; improved scar quality | [229] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Vaseashta, A.; Salel, S.; Bölgen, N. Biopolymer-Based Electrospun Nanofibers for Wound Healing, Regeneration, and Therapeutics. Materials 2026, 19, 1443. https://doi.org/10.3390/ma19071443
Vaseashta A, Salel S, Bölgen N. Biopolymer-Based Electrospun Nanofibers for Wound Healing, Regeneration, and Therapeutics. Materials. 2026; 19(7):1443. https://doi.org/10.3390/ma19071443
Chicago/Turabian StyleVaseashta, Ashok, Sedef Salel, and Nimet Bölgen. 2026. "Biopolymer-Based Electrospun Nanofibers for Wound Healing, Regeneration, and Therapeutics" Materials 19, no. 7: 1443. https://doi.org/10.3390/ma19071443
APA StyleVaseashta, A., Salel, S., & Bölgen, N. (2026). Biopolymer-Based Electrospun Nanofibers for Wound Healing, Regeneration, and Therapeutics. Materials, 19(7), 1443. https://doi.org/10.3390/ma19071443









