Mechanism of UV-C-Induced Oxygen Vacancies Altering the Colour of Dental Zirconia
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization Methods
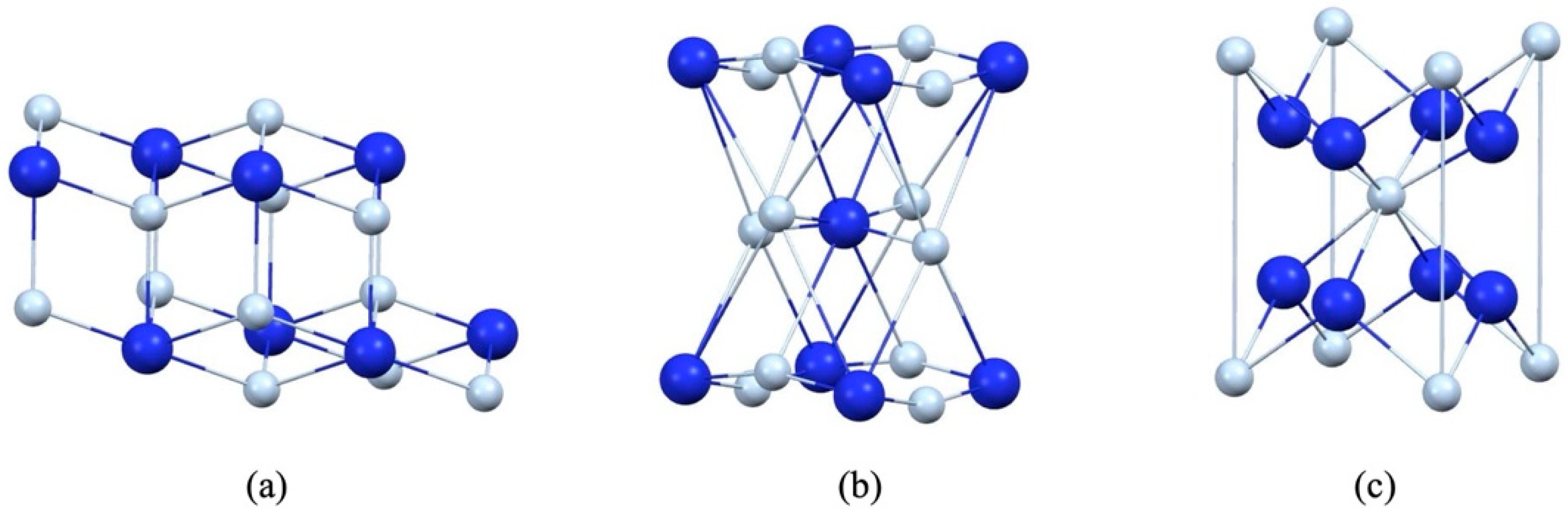
2.3. Computational Calculation
3. Results
3.1. Colour Change Observations
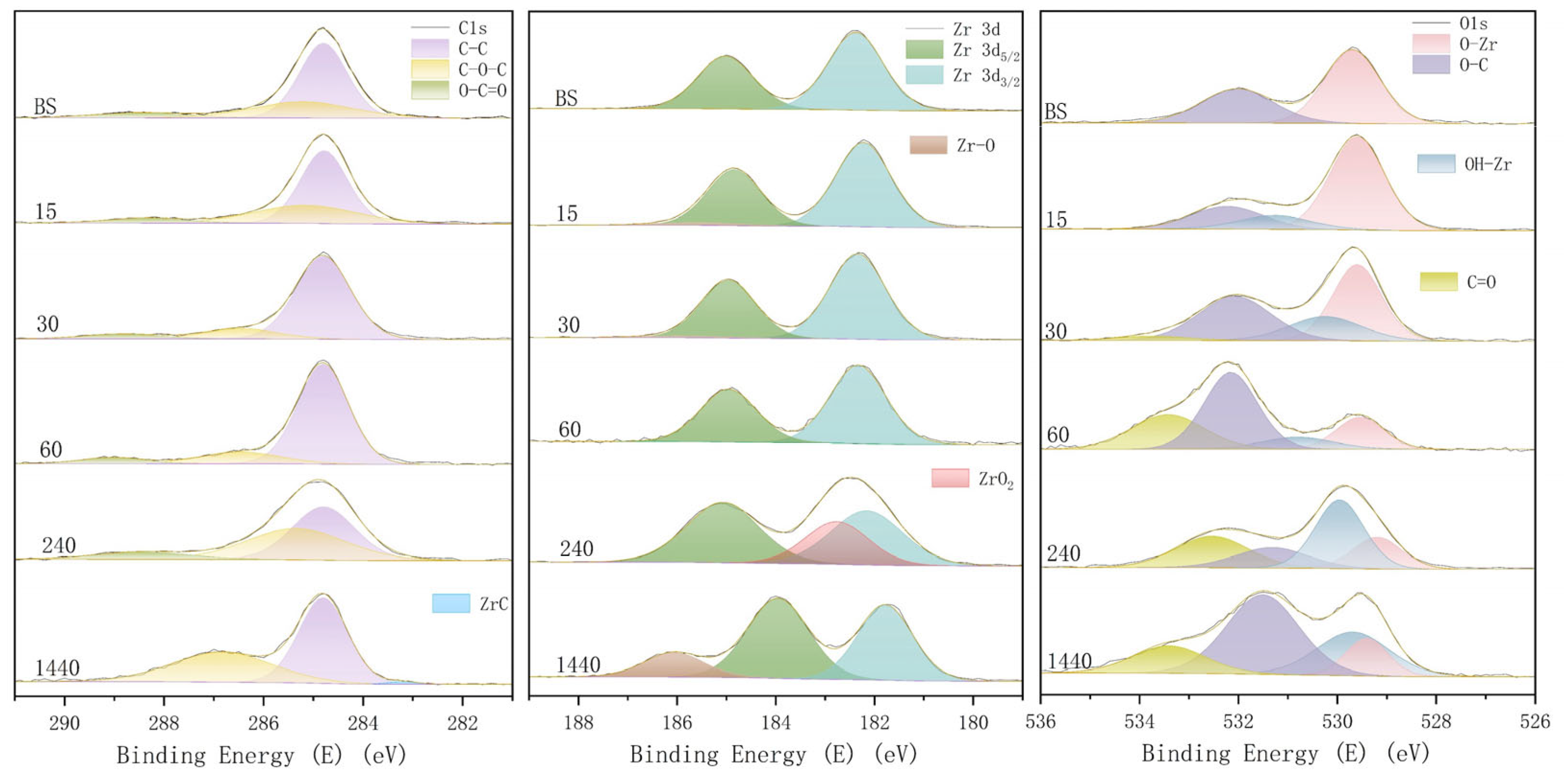
3.2. Surface Composition (XPS)
3.3. Defect Location (EPR)
3.4. Band Gap and DOS Simulation
4. Discussion
4.1. Interpretation of Colour and Feasibility
4.2. Analysis of Surface Chemical Changes
4.3. Confirmation of Paramagnetic Defects
4.4. Proposed Mechanism of UV-C-Induced Colour Change
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pacchioni, G.; Rahman, T.S. Defect engineering of oxide surfaces: Dream or reality? J. Phys. Condens. Matter 2022, 34, 291501. [Google Scholar] [CrossRef]
- Zhang, Y.; Lawn, B.R. Novel Zirconia Materials in Dentistry. J. Dent. Res. 2018, 97, 140–147. [Google Scholar] [CrossRef]
- Bai, X.; Xu, M.; Jin, S.; Pow, E.H.N.; Chen, Y.; Tsoi, J.K.H. Direct colour printing on zirconia using 222 nm UV-C photons. Dent. Mater. 2025, 41, 101–112. [Google Scholar] [CrossRef]
- Kozlovskiy, A.L.; Konuhova, M.; Borgekov, D.B.; Anatoli, I.P. Study of irradiation temperature effect on radiation-induced polymorphic transformation mechanisms in ZrO2 ceramics. Opt. Mater. 2024, 156, 115994. [Google Scholar] [CrossRef]
- Miyazaki, T.; Nakamura, T.; Matsumura, H.; Ban, S.; Kobayashi, T. Current status of zirconia restoration. J. Prosthodont. Res. 2013, 57, 236–261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Agingu, C.; Tsoi, J.K.H.; Yu, H. Effects of Aging on the Color and Translucency of Monolithic Translucent Y-TZP Ceramics: A Systematic Review and Meta-Analysis of In Vitro Studies. BioMed Res. Int. 2021, 2021, 8875023. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, J.I. An unusual mechanism for HOMO-LUMO gap narrowing in a minimal near-IR dye generated by the deprotonation of bis(dicyanomethylene)indan. Chem. Phys. Lett. 2014, 608, 355–359. [Google Scholar] [CrossRef]
- Han, A.; Ding, H.; Tsoi, J.K.H.; Imazato, S.; Matinlinna, J.P.; Chen, Z. Prolonged UV-C Irradiation is a Double-Edged Sword on the Zirconia Surface. ACS Omega 2020, 5, 10. [Google Scholar] [CrossRef]
- Zhang, Y.; Duan, L. First-principles investigations of the crystal and electronic structures, dynamical and mechanical stabilities, Born effective charges and dielectric permittivities of a novel tetragonal zirconia. Phys. B Condens. Matter 2023, 659, 414862. [Google Scholar] [CrossRef]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical Properties and Electronic Structure of Amorphous Germanium. Phys. Status Solidi 1966, 15, 627–637. [Google Scholar] [CrossRef]
- El Mesoudy, A.; Machon, D.; Ruediger, A.; Jaouad, A.; Alibart, F.; Ecoffey, S.; Drouin, D. Band gap narrowing induced by oxygen vacancies in reactively sputtered TiO2 thin films. Thin Solid Films 2023, 769, 139737. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, Z.; Zhang, J.; Shao, C.; Dai, K.; Fan, K.; Liang, C. Interfacial Chemical Bond and Oxygen Vacancy-Enhanced In2O3/CdSe-DETA S-scheme Heterojunction for Photocatalytic CO2 Conversion. Adv. Funct. Mater. 2023, 33, 2214470. [Google Scholar] [CrossRef]
- French, R.H.; Glass, S.J.; Ohuchi, F.S.; Xu, Y.N.; Ching, W.Y. Experimental and theoretical determination of the electronic structure and optical properties of three phases of ZrO2. Phys. Rev. B 1994, 49, 5133–5142. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, R.; Ding, Y.; Zhang, B.; Li, H.; Bai, B.; Li, M.; Cui, Y.; Xiao, J.; Wu, Z.S. Unraveling oxygen vacancy site mechanism of Rh-doped RuO2 catalyst for long-lasting acidic water oxidation. Nat. Commun. 2023, 14, 1412. [Google Scholar] [CrossRef] [PubMed]
- Han, A.; Tsoi, J.K.H.; Matinlinna, J.P.; Zhang, Y.; Chen, Z. Effects of different sterilization methods on surface characteristics and biofilm formation on zirconia in vitro. Dent. Mater. 2018, 34, 272–281. [Google Scholar] [CrossRef]
- Gionco, C.; Paganini, M.C.; Giamello, E.; Burgess, R.; Di Valentin, C.; Pacchioni, G. Paramagnetic defects in polycrystalline zirconia: An EPR and DFT study. Chem. Mater. 2013, 25, 2243–2253. [Google Scholar] [CrossRef]
- Berger, T.; Sterrer, M.; Diwald, O.; Knözinger, E. Charge trapping and photoadsorption of O2 on dehydroxylated TiO2 nanocrystals—An electron paramagnetic resonance study. ChemPhysChem 2005, 6, 2104–2112. [Google Scholar] [CrossRef]
- Zhang, W.; Bian, Z.; Peng, Y.; Tang, H.; Wang, H. Dual-function oxygen vacancy of BiOBr intensifies pollutant adsorption and molecular oxygen activation to remove tetracycline hydrochloride. Chem. Eng. J. 2023, 451, 138731. [Google Scholar] [CrossRef]
- Kurihara, A.; Nakamura, K.; Shishido, S.; Inagaki, R.; Harada, A.; Kanno, T.; Egusa, H. Mechanism underlying ultraviolet-irradiation-induced discoloration of dental zirconia ceramics stabilized with 3 and 5 mol% yttria. Ceram. Int. 2024, 50, 12136–12145. [Google Scholar] [CrossRef]
- Imparato, C.; Fantauzzi, M.; Passiu, C.; Rea, I.; Ricca, C.; Aschauer, U.; Sannino, F.; D’Errico, G.; De Stefano, L.; Rossi, A.; et al. Unraveling the Charge State of Oxygen Vacancies in ZrO2−x on the Basis of Synergistic Computational and Experimental Evidence. J. Phys. Chem. C 2019, 123, 11581–11590. [Google Scholar] [CrossRef]
- Sinhamahapatra, A.; Jeon, J.P.; Kang, J.; Han, B.; Yu, J.S. Oxygen-Deficient Zirconia (ZrO2−x): A New Material for Solar Light Absorption. Sci. Rep. 2016, 6, 27218. [Google Scholar] [CrossRef]
- Li, X.; Wu, X.; Zeng, X.C.; Yang, J. Band-gap engineering via tailored line defects in boron-nitride nanoribbons, sheets, and nanotubes. ACS Nano 2012, 6, 4104–4112. [Google Scholar] [CrossRef]
- Su, X.; Li, W.; Chen, D.; Zhang, S.; Lou, C.; Tian, Q.; Zhao, J.; Zhao, P. Rapid fabrication of oxygen-deficient zirconia by flash sintering treatment. J. Adv. Ceram. 2024, 13, 1881–1890. [Google Scholar] [CrossRef]
- Teeparthi, S.R.; Awin, E.W.; Kumar, R. Dominating role of crystal structure over defect chemistry in black and white zirconia on visible light photocatalytic activity. Sci. Rep. 2018, 8, 5541. [Google Scholar] [CrossRef]
- Forti, M.D.; Gargano, P.H.; Alonso, P.R.; Rubiolo, G.H. Strong pressure dependence of the valence band maximum in tetragonal ZrO2. Phys. Scr. 2024, 99, 025941. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xu, M.; Bai, X.; Yang, S.; Wen, W.; Cho, K.; Lee, Y.-H.; Jin, S.; Tsoi, J.K.H. Mechanism of UV-C-Induced Oxygen Vacancies Altering the Colour of Dental Zirconia. Materials 2026, 19, 1427. https://doi.org/10.3390/ma19071427
Xu M, Bai X, Yang S, Wen W, Cho K, Lee Y-H, Jin S, Tsoi JKH. Mechanism of UV-C-Induced Oxygen Vacancies Altering the Colour of Dental Zirconia. Materials. 2026; 19(7):1427. https://doi.org/10.3390/ma19071427
Chicago/Turabian StyleXu, Mengxiao, Xuedong Bai, Siyu Yang, Weijia Wen, Kiho Cho, Yun-Hong Lee, Shixin Jin, and James Kit Hon Tsoi. 2026. "Mechanism of UV-C-Induced Oxygen Vacancies Altering the Colour of Dental Zirconia" Materials 19, no. 7: 1427. https://doi.org/10.3390/ma19071427
APA StyleXu, M., Bai, X., Yang, S., Wen, W., Cho, K., Lee, Y.-H., Jin, S., & Tsoi, J. K. H. (2026). Mechanism of UV-C-Induced Oxygen Vacancies Altering the Colour of Dental Zirconia. Materials, 19(7), 1427. https://doi.org/10.3390/ma19071427







