Bio-Based and Sustainable Alternatives to Conventional and Synthetic Leather
Abstract
1. Introduction
2. Conventional and Synthetic Leather Markets

3. Synthetic-Leather-Coated Textile
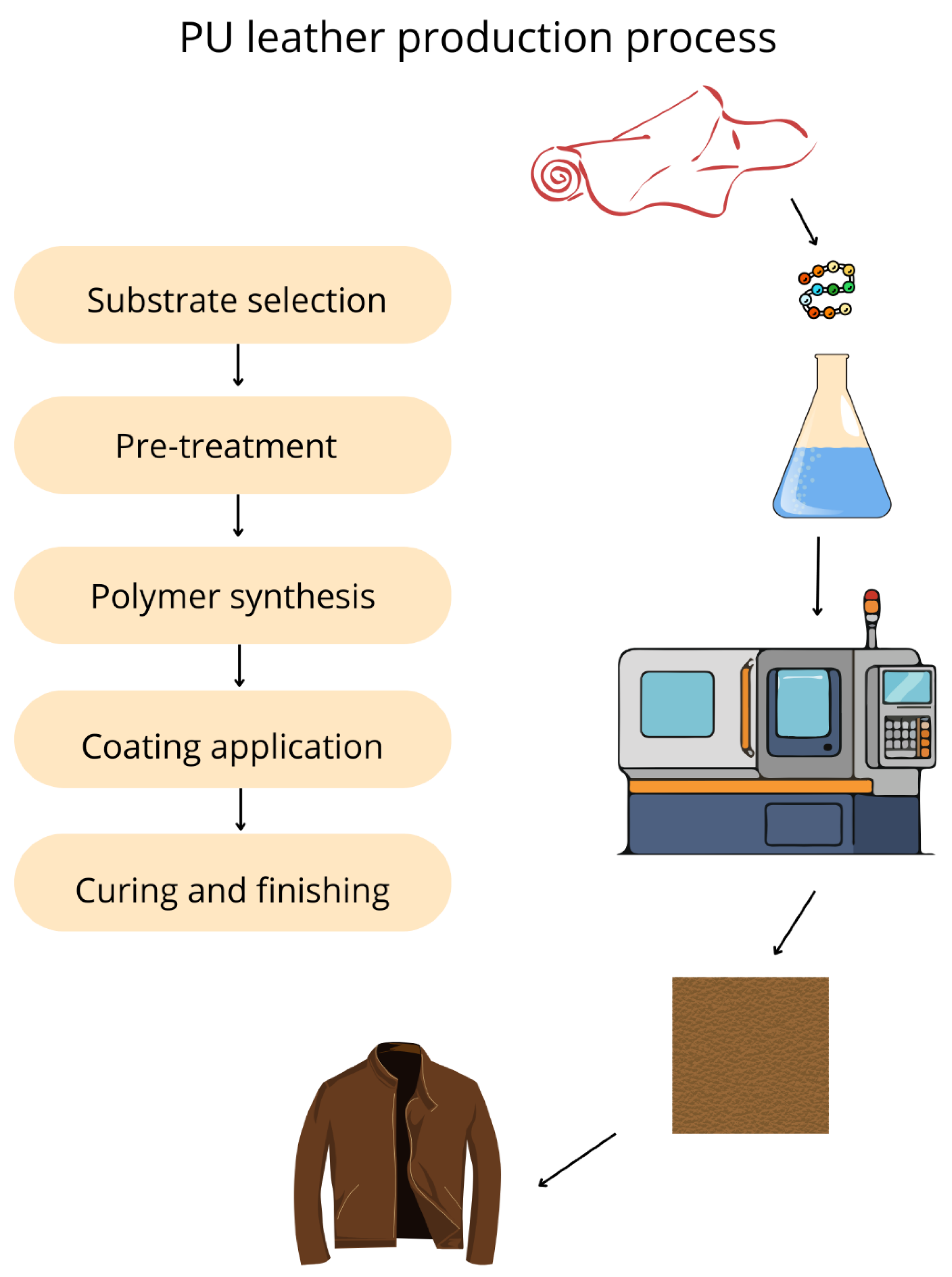
4. Current Trends in Alternative Bio-Leather Materials
4.1. Plant-Derived Leather
| Name | Feedstock | Processing | Mechanical Properties | Sustainability | biodegradability | Main Applications | References |
|---|---|---|---|---|---|---|---|
| Vegea® | Grape pomace (skins, seeds, stalks) | Drying, mechanical fractionation, oil extraction, polymerization of seed oil, grinding to high fiber powder, then spreading and drying | Tensile strength: 7–10 MPa; Tear strength: 20 N; Elongation: <30%; Flex resistance: 50,000 cycles | Carbon footprint: 4–10 kg CO2-eq/m2; Toluene detected in hazardous screenings | High biodegradability | Handbags, footwear, fashion accessories, furniture upholstery, automotive interiors | [14,24,25] |
| Vegatex (AppleSkin®) | Apple pomace | Coagulation process or molded pulp technology; mixing pomace with polymers | Tensile strength: 14 MPa; Tear resistance: 18.4 N/mm; Flex resistance: 50,000 cycles | Hazardous screenings detected butanone oxime and traces of DMFa | Not fully biodegradable (contains polyester/PUR blend) | Shoes, luggage, furnishing, upholstery, apparel | [14,25,26] |
| Desserto® | Cactus Opuntia ficus-indica | Reverse coating process; sun-drying pads, grinding, protein extraction, mixing into liquid bio-resin, then pouring on carrier | Tensile strength: 8–25 MPa; Tear resistance: 37.2 N/mm; Elongation: 30–55%; Flex resistance: 30,000 cycles | Carbon footprint: 1.3–2.0 kg CO2-eq/m2; Hazardous screenings detected butanone oxime, toluene, free isocyanate, folpet, DIBP | Partially biodegradable, varies by backing and coating choice | Handbags, shoes, apparel, furniture, automotive interiors | [14,24,25] |
| Pinatex® | Pineapple leaf fibers | Mechanical extraction of fibers, washing, drying, non-woven mesh formation coated with polymers | Tensile strength: 4.5 MPa; Tear resistance: 31 N/mm; Elongation: 10–20%; Flex resistance: 150,000 cycles | Carbon footprint: 2.7–4.0 kg CO2-eq/m2; Hazardous screenings detected DIBP | High biodegradability, depending on finish | Footwear, jackets, bags, fashion accessories | [14,24,25,26] |
4.2. Bacterial Cellulose Leather

4.3. Mycelium Leather

5. Most Commonly Used Fungal Species for Leather-like Materials
| Fungal Species/Genus | Common Name | Application Focus | References |
|---|---|---|---|
| Ganoderma lucidum | Reishi mushroom | Leather, mats | [33,41,42,43,44,45,46,47,48,49] |
| Pleurotus spp. | Oyster mushrooms | Leather, mats | [38,42,43,44,50,51,52] |
| Fomes fomentarius | Tinder fungus | Leather, mats | [43,50,53] |
| Fomitopsis spp. | Wood decay fungi | Leather, mats | [37,43,50] |
| Trametes versicolor | Turkey tail | Leather, mats | [38,43,45,48,50] |
| Agaricus spp. | Button mushrooms | Leather | [43,50] |
| Phellinus spp. | Wood decay fungi | Leather | [43] |
6. Production Methods of Mycelium Leather Materials
Examples of Mycelium Leather Production Protocols Developed by Different Scientists


| Feature | Mycofabrication of Mycelium-Based Leather from Brown-Rot Fungi (Raman et al., 2022) [48] | Revolutionizing Eco-Friendly Leather Production: A Freeze-Thaw and Liquid Fermentation Approach with Fungal Mycelium (Song et al., 2025) [41] | Fabrication Factors Influencing Mechanical, Moisture- and Water-Related Properties of Mycelium-Based Composites (Appels et al., 2019) [38] | Growing Mycelium Leather: A Paste Substrate Approach with Post-Treatments (Crawford et al., 2024) [42] | Scalable Production of Robust, Moisture-Wicking, and Breathable Superfine Mycelium Fiber/Waterborne Polyurethane Leather-like Textile via Direct Casting and Oven-Drying (Hao et al., 2025) [46] |
|---|---|---|---|---|---|
| Fungal Species | Multiple Polyporales (e.g., Ganoderma lucidum, Fomitella fraxinea) | Ganoderma lucidum | Trametes multicolor, Pleurotus ostreatus | Ganoderma lucidum, Pleurotus djamor | Ganoderma lucidum |
| Substrate | Oak sawdust and rice bran | Liquid culture with glucose and yeast extract | Rapeseed straw, beech sawdust and cotton | Paste substrate (flour, malt extract, xanthan gum, cream of tartar, citric acid) | Poplar sawdust, CaSO4, flour |
| Cultivation Method | Solid-state fermentation in boxes | Liquid fermentation in bioreactor | Solid-state on pressed molds | Paste-based solid-state fermentation in trays | Substrate-based growth in vented polyethylene bags, incubated and peeled, then defibrillated |
| Processing | PEG plasticization, coating, cross linking and hotpressing | Freeze–thaw cycles at −7, −15, −40, or −80 °C, deacetylation, crosslinking, plasticization | No pressing, cold pressing, or heat pressing | plasticization, crosslinking, tanning, hot pressing, dyeing | mycelium blending, casting, oven-drying |
| Chemical Agents Used | PEG (20%), glycerol, ethylene glycol, corn zein, tannic acid | NaOH for deacetylation, genipin and tannic acid (3–5%) for crosslinking, glycerol (20%) for plasticization | None specified for plasticizing; physical pressing only | Glycerol, citric acid, magnesium sulfate, commercial tanner, heat pressing | Mixed with waterborne polyurethane (WPU), oven-dried |
| Mechanical Properties | Tensile Strength: 8.49 ± 0.9 MPa Elastic Modulus: 8.14 MPa ± 0.88 MPa | Tensile Strength: 6.22 MPa Elongation at Break: 18.92% | Tensile Strength: 0.24 ± 0.03 MPa Elastic Modulus: 97 ± 9.0 MPa | Tensile Strength: Max ~1.4 N/cm2 (0.14 MPa) | Tensile Strength: 18.81 MPa Elastic Modulus: 1.81 MPa |
| Density | 1.35–1.46 g/cm3 | Not specified | 0.10–0.39 g/cm3 | Not specified | Not specified |
7. Comparison of Physical Properties of Mycelium Materials with Bovine Leather, Synthetic PU Leather and Commercially Available Bio-Leather Alternatives
| Physical Properties | Thickness [mm] | Tensile Strength [N/mm2] | Tear Resistance [N/mm] | Water Vapor Permeability [mg/(cm2 h)] | Water Vapor Absorption [mg/cm2] | References |
|---|---|---|---|---|---|---|
| Bovine Leather chromium-tanned | 1.27 | 25.39 | 41.11 | 4 | [55] | |
| Bovine leather vegetable-tanned | 2.2 | 32.32 | 67.5 | 1.1 | [55] | |
| Muskin® | 6.22 | 0.2 | 0.5 | 10.4 | 6 | [14] |
| Kombucha | 0.29 | 9.7 | 5.1 | 0.1 | 9.2 | [14] |
| PU coated textile | 1.11 | 10.2 | 17 | 1.1 | 1.4 | [14,56] |
| Desserto® | 1.28 | 9.48 | 47.74 | [55] | ||
| Appleskin® | 1.14 | 14 | 18.8 | 0.4 | 1.7 | [14] |
| Vegea® | 0.95 | 9.4 | 16.6 | 0.6 | 3 | [14] |
| Teak Leaf® | 0.57 | 12.2 | 30.7 | 0.1 | 0.1 | [14] |
| Pinatex® | 1.59 | 5.1 | 68.12 | 6.8 | [55] | |
| SnapPap® | 0.57 | 24.9 | 7.5 | 10.3 | 3.7 | [14] |
| Leatherette | 0.34 | 5.5 | 7.12 | 0.4 | [55] | |
| MyleaTM | 3–9 | 8–11 | 24 | n/a | n/a | [57] |
8. Applications of Mycelium Materials Across Industries
9. Challenges
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PEG | Polyethylene Glycol |
| WPU | Waterborne Polyurethane |
| SMFs | Shredded Mycelium Fibers |
| PET-G | Polyethylene Terephthalate Glycol |
| BOD | Biochemical Oxygen Demand |
| PVC | Polyvinyl Chloride |
| PU | Polyurethane |
| COD | Chemical Oxygen Demand |
| SCOBY | Symbiotic Cultures of Bacteria and Yeast |
| BC | Bacterial Cellulose |
| SEM | Scanning Electron Microscope |
| LCA | Life Cycle Assessment |
| PLA | Polylactic Acid |
References
- Titirici, M.; Baird, S.G.; Sparks, T.D.; Yang, S.M.; Brandt-Talbot, A.; Hosseinaei, O.; Harper, D.P.; Parker, R.M.; Vignolini, S.; Berglund, L.A.; et al. The Sustainable Materials Roadmap. J. Phys. Mater. 2022, 5, 032001. [Google Scholar] [CrossRef]
- Shirvanimoghaddama, K.; Motamedb, B.; Ramakrishnac, S.; Naebe, M. Death by waste: Fashion and textile circular economy case. Sci. Total Environ. 2020, 718, 137317. [Google Scholar] [CrossRef] [PubMed]
- Manshoven, S.; Chistis, M.; Vercalsteren, A.; Arnold, M.; Nicolau, M.; Lafond, E.; Fogh, L.; Coscieme, L. Textiles and the Environment in a Circular Economy; Technical Report; European Environmental Agency: Copenhagen, Denmark, 2019. [Google Scholar]
- EU Strategy for Sustainable and Circular Textiles. Available online: https://environment.ec.europa.eu/strategy/textiles-strategy_en (accessed on 7 July 2025).
- Krishnamoorthy, G.; Sadulla, S.; Sehgal, P.; Mandal, A. Greener approach to leather tanning process: D-Lysine aldehyde as novel tanning agent for chrome-free tanning. J. Clean. Prod. 2013, 42, 277–286. [Google Scholar] [CrossRef]
- Theis, E.; Goetz, A. Chrome Tanning I. The role played by sodium chloride in chrome liquors upon chrome tanning. Ind. Eng. Chem. 1932, 24, 304–307. [Google Scholar] [CrossRef]
- UNIDO-UNEP Report 1991; UNEP: Paris, France, 1991.
- European Commission. JRC-IPTS Report: Dokument Referencyjny Dotyczący Najlepszych Dostępnych Technik (BAT) dla Garbowanie Skór; Dyrektywa 2010/75/UE w sprawie emisji przemysłowych (Zintegrowane zapobieganie zanieczyszczeniom i ich kontrola); European Commission: Seville, Spain, 2013. [Google Scholar]
- Sundar, V.; Rao, J.R.; Muralidharan, C. Cleaner chrome tanning—Emerging options. J. Clean. Prod. 2002, 10, 69–74. [Google Scholar] [CrossRef]
- Ahmed, M.D.; Maraz, K.M. Benefits and problems of chrome tanning in leather processing: Approach a greener technology in leather industry. Mater. Eng. Res. 2021, 3, 156–164. [Google Scholar] [CrossRef]
- Morera, J.; Bacardit, A.; Olle, L.; Bartolı, E.; Borras, M. Minimization of the environmental impact of chrome tanning: A new process with high chrome exhaustion. Chemosphere 2007, 69, 1728–1733. [Google Scholar] [CrossRef]
- Ahmed, D.; Maraz, K.M.; Khan, R.A. Prospects and Challenges of Chrome Tanning: Approach a Greener Technology in Leather Industry. Sci. Rev. 2021, 7, 42–49. [Google Scholar] [CrossRef]
- Jones, M.; Gandia, A.; John, S.; Bismarck, A. Leather-like material biofabrication using fungi. Nat. Sustain. 2021, 4, 9–16. [Google Scholar] [CrossRef]
- Meyer, M.; Dietrich, S.; Schulz, H.; Mondschein, A. Comparison of the Technical Performance of Leather, Artificial Leather, and Trendy Alternatives. Coatings 2021, 11, 226. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, S.; Pan, S.; Zhao, D.; Wei, J.; Zhao, M. Methods for Determination of Plasticizer Migration from Polyvinyl Chloride Synthetic Materials: A Mini Review. J. Leather Sci. Eng. 2022, 4, 8. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Y.; Chen, J.; Chen, R.; Yao, Z.; Huang, J.; Ruan, Y.; Wu, L. A Once-Formed PU/PVC Synthetic Leather and Preparation Method Thereof. Chinese Patent CN104233839, 24 December 2014. [Google Scholar]
- Anam PALF by Ananas Anam: The Premium Textile-Grade Fibre Made from Waste Pineapple Leaves. Available online: https://www.ananas-anam.com/ (accessed on 9 July 2025).
- Wudu, H.; Teshome, Z.; Tesfaye, T.; Solomon, B.; Ebissa, D.T. Animal-Free Leather-Like Material from Pineapple Leaf Fiber Enhanced with Turmeric Dyeing: A Sustainable Alternative to Leather. Wiley J. Eng. 2025, 2025, 9081921. [Google Scholar] [CrossRef]
- Merlino, F.; Tessitore, G. Polymeric Compounds for the Production of Coated Fabrics Including Biomass Derived from Vitis as an Active Ingredient and Filler. Patent No IT201900000073A1, 4 August 2020. [Google Scholar]
- Vegatex: Designed for the Planet Engineered for Change. Available online: https://www.appleskin.com/impact (accessed on 26 September 2025).
- Kefale, G.Y.; Kebede, Z.T.; Birlie, A.A. A Systematic Review on Potential Bio Leather Substitute for Natural Leather. Hindawi J. Eng. 2023, 2023, 1629174. [Google Scholar] [CrossRef]
- VEGEA. Vegea Innovative Bio-Mateials, About Our Company, Innovative Biomaterials for Fashion & Design. Available online: https://www.vegeacompany.com/company/ (accessed on 26 September 2025).
- Kang, J.; Qiu, C. Fruit Extract Leather and Preparation Method Thereof. Patent No TWI829030B, 11 January 2024. [Google Scholar]
- Yizengaw, M.A.; Birlie, A.A.; Tesfaye, T.; Katrikan, R.; Cheklie, E.B.; Girma, Z. Grape Waste as Leather-Like Material Alternative: A Comprehensive Review of Ancient Practices, Current Technologies, and Future Trends. J. Renew. Mater. 2025, 17, 1783. [Google Scholar] [CrossRef]
- Malabadi, R.B.; Kolkar, K.P.; Chalannavar, R.K.; Baijnath, H. Plant-based leather production: An update. World J. Adv. Eng. Technol. Sci. 2025, 14, 31–59. [Google Scholar] [CrossRef]
- Duangsuwan, S.; Junkong, P.; Phinyocheep, P.; Thanawan, S.; Amornsakchai, T. Development of Green Leather Alternative from Natural Rubber and Pineapple Leaf Fiber. Sustainability 2023, 15, 15400. [Google Scholar] [CrossRef]
- Saha, N.; Ngwabebhoh, F.A.; Nguyen, H.T.; Saha, P. Environmentally Friendly and Animal Free Leather: Fabrication and Characterization. In Proceedings of the PPS2019 Europe-Africa Regional Conference of the Polymer Processing Society, Pretoria, South Africa, 18–21 November 2019; AIP Publishing LLC.: Melville, NY, USA, 2019; Volume 2289, p. 020049. [Google Scholar] [CrossRef]
- da Silva Junior, C.J.G.; de Amorim, J.D.P.; de Medeiros, A.D.M.; de Holanda Cavalcanti, A.K.L.; do Nascimento, H.A.; Henrique, M.A.; do Nascimento Maranhão, L.J.C.; Vinhas, G.M.; Souto, K.K.O.; de Santana Costa, A.F.; et al. Design of a Naturally Dyed and Waterproof Biotechnological Leather from Reconstituted Cellulose. J. Funct. Biomater. 2022, 13, 49. [Google Scholar] [CrossRef]
- Amanah, H.; Cahyaningtyas, A.; Renaldi, G.; Fibriana, F.; Mulyani, W.E. Cost-effective production of kombucha bacterial cellulose by evaluating nutrient sources, quality assessment, and dyeing Methods. Environ. Sci. Pollut. Res. 2025, 32, 2713–2725. [Google Scholar] [CrossRef]
- Choi, S.M.; Lee, D.H.; Zo, S.M.; Sood, A.; Han, S.S. Gel-Based Approaches to Vegan Leather: Opportunities and Challenges in Mimicking Leather Properties. Gels 2025, 11, 395. [Google Scholar] [CrossRef]
- Ngwabebhoh, F.A.; Saha, N.; Nguyen, H.T.; Brodnjak, U.V.; Saha, T.; Lengalova, A.; Saha, P. Preparation and Characterization of Nonwoven Fibrous Biocomposites for Footwear Components. Polymers 2020, 12, 3016. [Google Scholar] [CrossRef]
- Nguyen, H.T.; Saha, N.; Ngwabebhoh, F.A.; Zandraa, O.; Saha, T.; Saha, P. Kombucha-derived bacterial cellulose from diverse wastes: A prudent leather alternative. Cellulose 2021, 28, 9335–9353. [Google Scholar] [CrossRef]
- Elsacker, E.; Zhang, M.; Dade-Robertson, M. Fungal Engineered Living Materials: The Viability of Pure Mycelium Materials with Self-Healing Functionalities. Adv. Funct. Mater. 2023, 33, 2301875. [Google Scholar] [CrossRef]
- Madusanka, C.; Udayanga, D.; Nilmini, R.; Rajapaksha, S.; Hewawasam, C.; Manamgoda, D.; Vasco, J. A review of recent advances in fungal mycelium based composites. Discov. Mater. 2024, 4, 13. [Google Scholar] [CrossRef]
- Haneef, M.; Ceseracciu, L.; Canale, C.; Bayer, I.S.; Heredia-Guerrero, J.A. Advanced Materials from Fungal Mycelium: Fabrication and Tuning of Physical Properties. Nat. Sci. Rep. 2017, 7, 41292. [Google Scholar] [CrossRef] [PubMed]
- Elsacker, E.; Vandelook, S.; VanWylick, A.; Ruytinx, J.; De Laet, L.; Peeters, E. A comprehensive framework for the production of mycelium-based lignocellulosic composites. Sci. Total Environ. 2020, 725, 138431. [Google Scholar] [CrossRef]
- Cartabia, M.; Girometta, C.E.; Milanese, C.; Baiguera, R.M.; Buratti, S.; Branciforti, D.S.; Vadivel, D.; Girella, A.; Babbini, S.; Savino, E.; et al. Collection and Characterization of Wood Decay Fungal Strains for Developing Pure Mycelium Mats. J. Fungi 2021, 7, 1008. [Google Scholar] [CrossRef]
- Appels, F.V.W.; Camere, S.; Montalti, M.; Karana, E.; Jansen, K.M.B.; Dijksterhuis, J.; Krijgshelda, P.; Wösten, H.A.B. Fabrication factors influencing mechanical, moisture- and water-related properties of mycelium-based composites. Mater. Des. 2019, 161, 64–71. [Google Scholar] [CrossRef]
- Jones, M.; Bhat, T.; Huynh, T.; Kandare, E.; Yuen, R.; Wang, C.H.; John, S. Waste-derived low-cost mycelium composite construction materials with improved fire safety. Fire Mater. 2018, 42, 816–825. [Google Scholar] [CrossRef]
- Sangosanya, A.-O.; Pistofidou, A. Textile and food waste biodegradation for the production of composite mycelium materials using Pleurotus ostreatus. Res. Dir. Biotechnol. Des. 2024, 2, e24. [Google Scholar] [CrossRef]
- Song, L.; Liu, Y.; Xiao, S.; Yuan, X.; Han, X. Revolutionizing Eco-Friendly Leather Production: A Freeze-Thaw and Liquid Fermentation Approach with Fungal Mycelium. J. Fungi 2025, 11, 326. [Google Scholar] [CrossRef]
- Crawford, A.; Ruthanna Miller, S.; Branco, S.; Fletcher, J.; Stefanov, D. Growing mycelium leather: A paste substrate approach with post-treatments. Res. Dir. Biotechnol. Des. 2024, 2, e6. [Google Scholar] [CrossRef]
- Amobonye, A.; Lalung, J.; Awasthi, M.K.; Pillai, S. Fungal mycelium as leather alternative: A sustainable biogenic material for the fashion industry. Sustain. Mater. Technol. 2023, 38, e00724. [Google Scholar] [CrossRef]
- Elsacker, E.; Vandelook, S.; Peeters, E. Recent technological innovations in mycelium materials as leather substitutes: A patent review. Front. Bioeng. Biotechnol. 2023, 11, 1204861. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, M.R.; Raja, D.; Sri, A.A.; Bilal, S.; Pooja, C.; Sindhuja, K.G. Mycelium-Based Flame Retardant Treatments for Textiles Using Extracts of Turkey Tail, Reishi, and Shiitake. J. Text. Assoc. 2025, 85, 642–648. [Google Scholar] [CrossRef]
- Hao, Y.; Wang, H.; Tian, D.; Zhang, W.; Shi, B. Scalable production of robust, moisture-wicking, and breathable superfine mycelium fiber/waterborne polyurethane leather-like textile via direct casting and oven-drying. Ind. Crops Prod. 2025, 226, 120632. [Google Scholar] [CrossRef]
- Antinori, M.E.; Ceseracciu, L.; Mancini, G.; Heredia-Guerrero, J.A.; Athanassiou, A. Fine-Tuning of Physicochemical Properties and Growth Dynamics of Mycelium-Based Materials. ACS Appl. Bio Mater. 2020, 3, 1044–1051. [Google Scholar] [CrossRef]
- Raman, J.; Kim, D.-S.; Kim, H.-S.; Oh, D.-S.; Shin, H.-J. Mycofabrication of Mycelium-Based Leather from Brown-Rot Fungi. J. Fungi 2022, 8, 317. [Google Scholar] [CrossRef]
- Onorato, C.; Madeu, F.; Tsakalova, M.; Deligkiozi, I.; Karathanasis, A.Z. Navigating the mycelium patent maze: A holistic approach to patent mapping in production technologies. World Pat. Inf. 2024, 76, 102265. [Google Scholar] [CrossRef]
- Kniep, J.; Graupner, N.; Reimer, J.; Müssig, J. Mycelium-based biomimetic composite structures as a sustainable leather alternative. Mater. Today Commun. 2024, 39, 109100. [Google Scholar] [CrossRef]
- Saini, R.; Kaur, G.; Brar, S.K. Textile residue-based mycelium biocomposites from Pleurotus ostreatus. Mycology 2024, 15, 683–689. [Google Scholar] [CrossRef]
- Shankar, M.P.; Hamza, A.; Khalad, A.; Shanthi, G.; Kuppireddy, S.; Kumar, D.S. Engineering mushroom mycelium for a greener built environment: Advancements in mycelium-based biocomposites and bioleather. Food Biosci. 2024, 62, 105577. [Google Scholar] [CrossRef]
- Gandia, A.; Brandhof, J.G.v.D.; Appels, F.V.; Jones, M.P. Flexible Fungal Materials: Shaping the Future. Trends Biotechnol. 2021, 39, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.; Branco, S.; Sheldon, L.; Ebert, L. Experimental Fabrication Methods for Mycelium Leather Production. In Proceedings of the CEES 2023 2nd International Conference on Construction, Energy, Environment & Sustainability, Funchal, Portugal, 27–30 June 2023. [Google Scholar]
- Karana, E.; Blauwhoff, D.; Hultink, E.; Camere, S. When the material grows: A case study on designing (with) mycelium-based materials. Int. J. Des. 2018, 12, 119–136. [Google Scholar]
- Rathinamoorthy, R.; Bharathi, T.S.; Snehaa, M.; Swetha, C. Structural and Chemical Characterization of Mycelium Sheets Developed from Penicillium Camemberti. J. Polym. Environ. 2023, 31, 5152–5165. [Google Scholar] [CrossRef]
- Technical Data Sheet Mylea TM, The Mycelium Leather Mylea Original Reinforced. Available online: www.abc-pf.org/data/report/209.pdf (accessed on 19 July 2025).
- Vandelook, S.; Elsacker, E.; Van Wylick, A.; De Laet, L.; Peeters, E. Current state and future prospects of pure mycelium materials. Fungal Biol. Biotechnol. 2021, 8, 20. [Google Scholar] [CrossRef]
- Sydor, M.; Bonenberg, A.; Doczekalska, B.; Cofta, G. Mycelium-Based Composites in Art, Architecture, and Interior Design: A Review. Polymers 2022, 14, 145. [Google Scholar] [CrossRef]
- Roh, E.K. Mechanical properties and preferences of natural and artificial leathers, and their classification with a focus on leather for bags. J. Eng. Fibers Fabr. 2020, 15, 1558925020968825. [Google Scholar] [CrossRef]
- Mogu. Radical by Nature. Available online: https://mogu.bio/ (accessed on 5 October 2025).
- French, V.; Du, C.; Foster, E.J. Mycelium as a self-growing biobased material for the fabrication of single-layer masks. J. Bioresour. Bioprod. 2023, 8, 399–407. [Google Scholar] [CrossRef]
- Antinori, M.; Contardi, M.; Suarato, G.; Armirotti, A.; Bertorelli, R.; Mancini, G.; Debellis, D. Advanced mycelium materials as potential self-growing biomedical scaffolds. Sci. Rep. 2021, 11, 12630. [Google Scholar] [CrossRef]
- Bustillos, J.; Loganathan, A.; Agrawal, R.; Gonzalez, B.A.; Perez, M.G.; Ramaswamy, S.; Boesl, B.; Agarwal, A. Uncovering the Mechanical, Thermal, and Chemical Characteristics of Biodegradable Mushroom Leather with Intrinsic Antifungal and Antibacterial Properties. ACS Appl. Bio Mater. 2020, 3, 3145–3156. [Google Scholar] [CrossRef]
- Karunarathne, A.; Nabiyeva, G.; Rasmussen, C.J.; Alkhoury, K.; Assem, N.; Bauer, J.; Chester, S.A.; Khalizov, A.F.; Gor, G.Y. Effects of Humidity on Mycelium-Based Leather. ACS Appl. Bio Mater. 2024, 7, 6441–6450. [Google Scholar] [CrossRef] [PubMed]
- Akhter, S.; Jahan, M.S.; Rahman, M.L.; Ruhane, T.A.; Ahmed, M.; Khan, M.A. Revolutionizing Sustainable Fashion: Jute–Mycelium Vegan Leather Reinforced with Polyhydroxyalkanoate Biopolymer Crosslinking from Novel Bacteria. Hindawi Adv. Polym. Technol. 2024, 2024, 1304800. [Google Scholar] [CrossRef]
- Tabandeh, E.; Jędrasik, J.; Czarnecka, M.; Dzierżanowska, J.; Ludwicka, K. SCOBY Cellulose Modified with Apple Powder—Biomaterial with Functional Characteristics. Int. J. Mol. Sci. 2023, 24, 1005. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.; Miller, S.R.; Johnson, M.; Stefanov, D. Mycelium Textile Upholstering: Scaling strategies for bio-leather applications. In 113th ACSA Annual Meeting Proceedings, Repair; ACSA Press: New Orleans, LA, USA, 2025. [Google Scholar] [CrossRef]
- Li, S.; Cao, S.; Wang, X.; Zhang, Y.; Zhang, X.; Lu, W.; Zhu, D. Investigating the mechanism of Zn cross-linking of chitin in a mycelium-based leather substitute and its performance evaluation. Int. J. Biol. Macromol. 2024, 276, 133954. [Google Scholar] [CrossRef]
- Quaratesi, I.; Badea, E.; Călinescu, I.; Sardroudi, N.P.; Zengin, G.; Casas, C.; Bacardit, A. Eco-Friendly Alternatives in Leather Production: Performance of Biodegradable Alginate-Based Retanned Leather Compared to Conventional Leathers and Plant-Based Materials. Appl. Sci. 2024, 14, 10263. [Google Scholar] [CrossRef]
- Kennedy, J. Mycelium Leather: Paving the Way for Green Luxury Amidst Challenges. IDTechEx Research Article. Available online: https://www.idtechex.com/en/research-article/mycelium-leather-paving-the-way-for-green-luxury-amidst-challenges/30640 (accessed on 19 July 2025).
- Bahua, H.; Wijayanti, S.P.; Putra, A.S.; Ariyani, N.R.; Isharyadi, F.; Nuha, N.; Paminto, A.K.; Mulyono, M.; Djarot, I.N.; Widyastuti, N.; et al. Life cycle assessment (LCA) of leather-like materials from mycelium: Indonesian case study. Int. J. Life Cycle Assess. 2024, 29, 1916–1931. [Google Scholar] [CrossRef]
- Williams, E.; Cenian, K.; Golsteijn, L.; Morris, B.; Scullin, M.L. Life cycle assessment of MycoWorks’ Reishi™: The first low-carbon and biodegradable alternative leather. Environ. Sci. Eur. 2022, 34, 120. [Google Scholar] [CrossRef]
- Vas, N.; Munjal, K. Vegan Leather and Environmental Impact: Alternatives, Innovations, and a Case Study on Cactus Leather. Int. J. Environ. Sci. 2025, 11, 1251–1255. [Google Scholar] [CrossRef]
- van den Brandhof, J.G.; Wösten, H.A.B. Risk assessment of fungal materials. Fungal Biol. Biotechnol. 2022, 9, 3. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Oleksińska-Merida, E.; Puchalski, M.; Herczyńska, L. Bio-Based and Sustainable Alternatives to Conventional and Synthetic Leather. Materials 2026, 19, 1198. https://doi.org/10.3390/ma19061198
Oleksińska-Merida E, Puchalski M, Herczyńska L. Bio-Based and Sustainable Alternatives to Conventional and Synthetic Leather. Materials. 2026; 19(6):1198. https://doi.org/10.3390/ma19061198
Chicago/Turabian StyleOleksińska-Merida, Ewa, Michał Puchalski, and Lucyna Herczyńska. 2026. "Bio-Based and Sustainable Alternatives to Conventional and Synthetic Leather" Materials 19, no. 6: 1198. https://doi.org/10.3390/ma19061198
APA StyleOleksińska-Merida, E., Puchalski, M., & Herczyńska, L. (2026). Bio-Based and Sustainable Alternatives to Conventional and Synthetic Leather. Materials, 19(6), 1198. https://doi.org/10.3390/ma19061198







