Culvert Retrofit with Green Filter Media for the Removal of Phosphorus from Stormwater Runoff
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Chemicals
2.3. Preparation of the Green Filter Media
2.4. Experimental Design and Setup
2.5. Sample Analysis
3. Results and Discussion
3.1. Stormwater Characterization
3.2. Optimizing the Composition of Green Filter Media
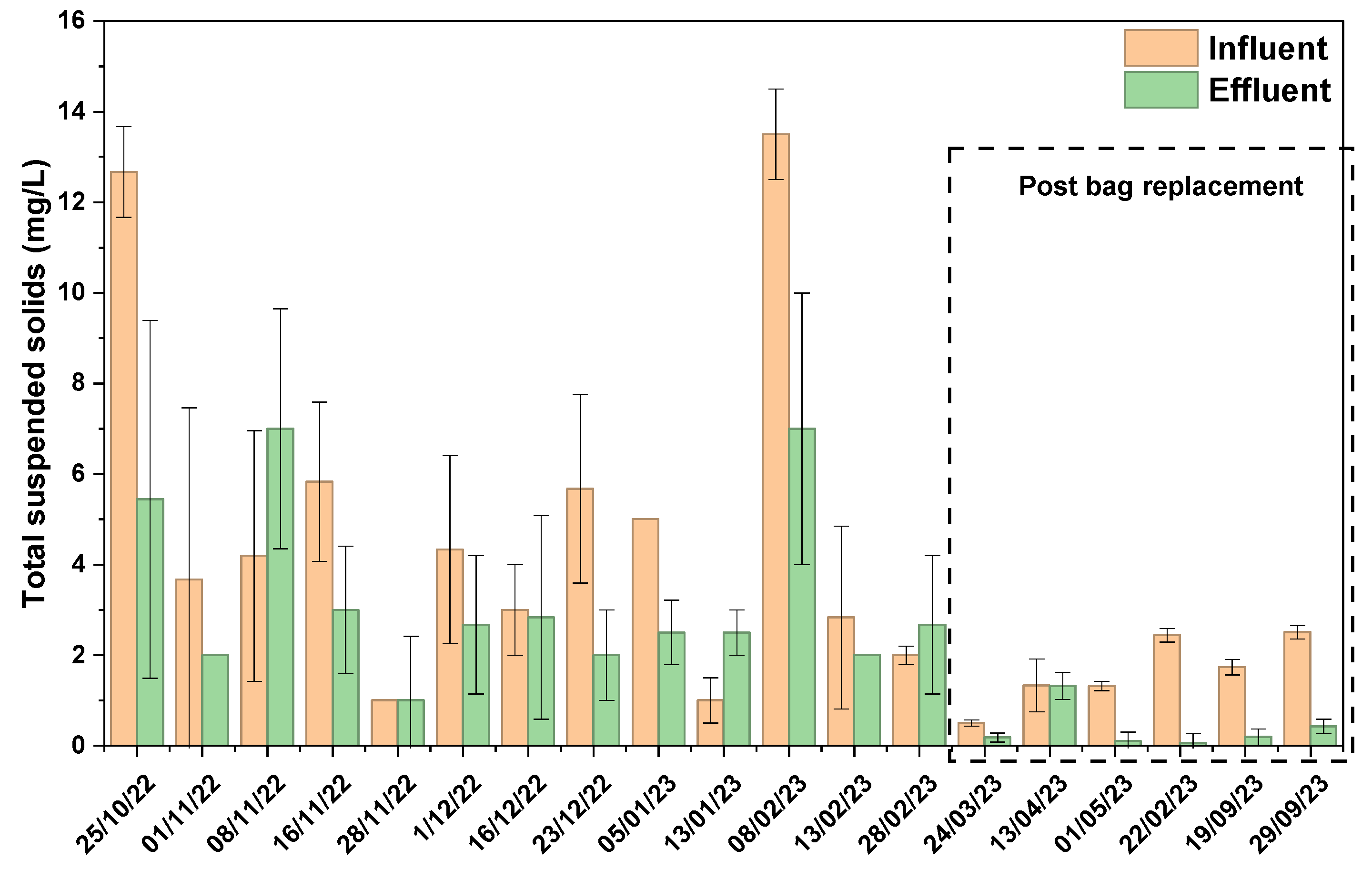
3.3. Green Media Filtration System—Field Application
3.4. Phosphorus Adsorption Analysis on the Spent Filter Media
3.5. Comparative Analysis with Other Field-Scale Studies
3.6. Sustainability Implications
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Al-WTR | aluminum water-treatment residual |
| NJ | New Jersey |
| TCLP | toxicity characteristic leaching procedure |
| USEPA | United States Environmental Protection Agency |
| RCRA | Resource Conservation and Recovery Act |
| TP | total phosphorus |
| TSS | total suspended solids |
| BDL | below detection limit |
| ICP-OES | inductively coupled plasma optical emission spectrometry |
| EPA | Environmental Protection Agency |
| NJDEP | New Jersey Department of Environmental Protection |
| PCB | polychlorinated hydrocarbons |
| PAH | polyaromatic hydrocarbons |
| HAB | harmful algal blooms |
References
- Zeng, F.; Zuo, Z.; Mo, J.; Chen, C.; Yang, X.; Wang, J.; Wang, Y.; Zhao, Z.; Chen, T.; Li, Y.; et al. Runoff Losses in Nitrogen and Phosphorus From Paddy and Maize Cropping Systems: A Field Study in Dongjiang Basin, South China. Front. Plant Sci. 2021, 12, 675121. [Google Scholar] [CrossRef]
- Elser, J.J.; Bracken, M.E.S.; Cleland, E.E.; Gruner, D.S.; Harpole, W.S.; Hillebrand, H.; Ngai, J.T.; Seabloom, E.W.; Shurin, J.B.; Smith, J.E. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 2007, 10, 1135–1142. [Google Scholar] [CrossRef]
- Smith, V.H.; Joye, S.B.; Howarth, R.W. Eutrophication of freshwater and marine ecosystems. Limnol. Oceanogr. 2006, 51, 351–355. [Google Scholar] [CrossRef]
- Short, F.T.; Wyllie-Echeverria, S. Natural and human-induced disturbance of seagrasses. Environ. Conserv. 1996, 23, 17–27. [Google Scholar] [CrossRef]
- Bricker, S.B.; Longstaff, B.; Dennison, W.; Jones, A.; Boicourt, K.; Wicks, C.; Woerner, J. Effects of nutrient enrichment in the nation’s estuaries: A decade of change. Harmful Algae 2008, 8, 21–32. [Google Scholar] [CrossRef]
- Velinsky, D.; Sommerfield, C.; Enache, M.; Charles, D. Nutrient and Ecological Histories in Barnegat Bay. New Jersey; Technical Report; Academy of Natural Sciences: Philadelphia, PA, USA, 2011. [Google Scholar]
- Khatri, N.; Tyagi, S. Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Front. Life Sci. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Munn, M.D.; Frey, J.W.; Tesoriero, A.J.; Black, R.W.; Duff, J.H.; Lee, K.E.; Maret, T.R.; Mebane, C.A.; Waite, I.R.; Zelt, R.B. Understanding the Influence of Nutrients on Stream Ecosystems in Agricultural Landscapes; Circular; US Geological Survey: Reston, VA, USA, 2018; pp. 1–80. [Google Scholar] [CrossRef]
- Na Nagara, V.; Sarkar, D.; Datta, R. Phosphorus and Heavy Metals Removal from Stormwater Runoff Using Granulated Industrial Waste for Retrofitting Catch Basins. Molecules 2022, 27, 7169. [Google Scholar] [CrossRef] [PubMed]
- Göbel, P.; Dierkes, C.; Coldewey, W.G. Storm water runoff concentration matrix for urban areas. J. Contam. Hydrol. 2007, 91, 26–42. [Google Scholar] [CrossRef]
- USEPA. Nonpoint Source: Agriculture; USEPA: Washington, DC, USA, 2022. Available online: https://www.epa.gov/nps/nonpoint-source-agriculture (accessed on 2 August 2025).
- Kennish, M.J.; Fertig, B.M.; Lathrop, R.G. Assessment of Nutrient Loading and Eutrophication in Barnegat Bay-Little Egg Harbor, New Jersey in Support of Nutrient Management Planning; New Jersey Environmental Digital Library: New Brunswick, NJ, USA, 2012. [Google Scholar] [CrossRef]
- Howarth, R.W.; Swaney, D.P.; Butler, T.J.; Marino, R. Rapid Communication: Climatic Control on Eutrophication of the Hudson River Estuary. Ecosystems 2000, 3, 210–215. [Google Scholar] [CrossRef]
- Howarth, R.W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae 2008, 8, 14–20. [Google Scholar] [CrossRef]
- Porter, P.A.; Mitchell, R.B.; Moore, K.J. Reducing hypoxia in the Gulf of Mexico: Reimagining a more resilient agricultural landscape in the Mississippi River Watershed. J. Soil Water Conserv. 2015, 70, 63A–68A. [Google Scholar] [CrossRef]
- Kennish, M. Eutrophication of Mid-Atlantic Coastal Bays. Bull. NJ Acad. Sci. 2009, 54, 1–8. [Google Scholar]
- USEPA. The Effects: Economy; USEPA: Washington, DC, USA, 2023. Available online: https://www.epa.gov/nutrientpollution/effects-economy (accessed on 2 August 2025).
- Wurtsbaugh, W.A.; Paerl, H.W.; Dodds, W.K. Nutrients, eutrophication and harmful algal blooms along the freshwater to marine continuum. WIREs Water 2019, 6, e1373. [Google Scholar] [CrossRef]
- Kennish, M.J.; Bricker, S.B.; Dennison, W.C.; Glibert, P.M.; Livingston, R.J.; Moore, K.A.; Noble, R.T.; Paerl, H.W.; Ramstack, J.M.; Seitzinger, S.; et al. Barnegat Bay–Little Egg Harbor Estuary: Case study of a highly eutrophic coastal bay system. Ecol. Appl. 2007, 17, S3–S16. [Google Scholar] [CrossRef]
- Pang, H.; Ingelido, P.; Hirst, B.; Pflaumer, J.; Witt, A.; Zaman, A.; Aiello, J. Water Quality Condition and Assessment within the Barnegat Bay Watershed between 2011 and 2015. J. Coast. Res. 2017, 78, 7–21. [Google Scholar] [CrossRef]
- Bologna, P.A.X.; Gaynor, J.J.; Meredith, R. Impacts of Invasive Sea Nettles (Chrysaora quinquecirrha) and Ctenophores on Planktonic Community Structure and Bloom Prediction of Sea Nettles Using Molecular Techniques; Final Project Report; New Jersey Department of Environmental Protection (NJDEP): Trenton, NJ, USA, 2015; p. 137. [Google Scholar]
- Lathrop, R.G.; Haag, S.M. Assessment of Seagrass Status in the Barnegat Bay-Little Egg Harbor Estuary System: 2003 and 2009; New Jersey Environmental Digital Library: New Brunswick, NJ, USA, 2011. [Google Scholar] [CrossRef]
- Baker, R.J.; Wieben, C.M.; Lathrop, R.G.; Nicholson, R.S. Concentrations, Loads, and Yields of Total Nitrogen and Total Phosphorus in the Barnegat Bay-Little Egg Harbor Watershed, New Jersey, 1989–2011, at Multiple Spatial Scales; U.S. Geological Survey: Reston, VA, USA, 2014. [Google Scholar]
- Zhao, B.; Zhang, Y.; Dou, X.; Yuan, H.; Yang, M. Granular ferric hydroxide adsorbent for phosphate removal: Demonstration preparation and field study. Water Sci. Technol. 2015, 72, 2179–2186. [Google Scholar] [CrossRef]
- Narkis, N.; Meiri, M. Phosphorus removal by activated alumina. Environ. Pollut. Ser. B Chem. Phys. 1981, 2, 327–343. [Google Scholar] [CrossRef]
- Ament, M.R.; Roy, E.D.; Yuan, Y.; Hurley, S.E. Phosphorus removal, metals dynamics, and hydraulics in stormwater bioretention systems amended with drinking water treatment residuals. J. Sustain. Water Built Environ. 2022, 8, 04022003. [Google Scholar] [CrossRef]
- Vogel, J.R.; Chavez, R.A.; Kandel, S.; Brown, G.O. Field scale demonstration of fly ash amended bioretention cells for stormwater phosphorus removal: A review of 12 years of work. Water 2021, 13, 1632. [Google Scholar] [CrossRef]
- Barca, C.; Troesch, S.; Meyer, D.; Drissen, P.; Andres, Y.; Chazarenc, F. Steel slag filters to upgrade phosphorus removal in constructed wetlands: Two years of field experiments. Environ. Sci. Technol. 2013, 47, 549–556. [Google Scholar] [CrossRef]
- Xia, S.; Liang, S.; Qin, Y.; Chen, W.; Xue, B.; Zhang, B.; Xu, G. Significant improvement of adsorption for phosphate removal by lanthanum-loaded biochar. ACS Omega 2023, 8, 24853–24864. [Google Scholar] [CrossRef]
- USEPA. Method 1311: Toxicity Characteristic Leaching Procedure. EPA SW-846: Test Methods for Evaluating Solid Waste, Physical/Chemical Methods; USEPA: Washington, DC, USA, 1992. [Google Scholar]
- Na Nagara, V.; Sarkar, D.; Barrett, K.; Datta, R. Greening the gray infrastructure: Green adsorbent media for catch basin inserts to remove stormwater pollutants. Environ. Technol. Innov. 2021, 21, 101334. [Google Scholar] [CrossRef]
- USEPA. Method 3050B: Acid Digestion of Sediments, Sludges, and Soils; Revision 2. USEPA: Washington, DC, USA, 1996.
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- Nguyen, M.D.; Adhikari, S.; Mallya, D.S.; Thomas, M.; Surapaneni, A.; Moon, E.M.; Milne, N.A. Reuse of aluminium-based water treatment sludge for phosphorus adsorption: Evaluating the factors affecting and correlation between adsorption and sludge properties. Environ. Technol. Innov. 2022, 27, 102717. [Google Scholar] [CrossRef]
- Lee, L.Y.; Wang, B.; Guo, H.; Hu, J.Y.; Ong, S.L. Aluminum-based water treatment residue reuse for phosphorus removal. Water 2015, 7, 1480–1496. [Google Scholar] [CrossRef]
- Rahmati, R.; Sidhu, V.; Nunez, R.; Datta, R.; Sarkar, D. Correlation of Phosphorus Adsorption with Chemical Properties of Aluminum-Based Drinking Water Treatment Residuals Collected from Various Parts of the United States. Molecules 2022, 27, 7194. [Google Scholar] [CrossRef] [PubMed]
- Babatunde, A.O.; Zhao, Y.Q.; Burke, A.M.; Morris, M.A.; Hanrahan, J.P. Characterization of aluminium-based water treatment residual for potential phosphorus removal in engineered wetlands. Environ. Pollut. 2009, 157, 2830–2836. [Google Scholar] [CrossRef]
- Ait Ichou, A.; Benhiti, R.; Abali, M.; Dabagh, A.; Carja, G.; Soudani, A.; Chiban, M.; Zerbet, M.; Sinan, F. Characterization and sorption study of Zn2 [FeAl]-CO3 layered double hydroxide for Cu (II) and Pb (II) removal. J. Solid State Chem. 2023, 320, 123869. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; Sun, H.; Che, W.; Li, J. A mixed-flow bioretention system amended with water treatment residuals to enhance nitrogen and phosphorus removal performance. Desalination Water Treat. 2021, 236, 144–154. [Google Scholar] [CrossRef]
- O’Neill, S.W.; Davis, A.P. Water treatment residual as a bioretention amendment for phosphorus. II: Long-term column studies. J. Environ. Eng. 2012, 138, 328–336. [Google Scholar] [CrossRef]
- Zhang, Z.; Sarkar, D.; Datta, R.; Deng, Y. Adsorption of perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) by aluminum-based drinking water treatment residuals. J. Hazard. Mater. Lett. 2021, 2, 100034. [Google Scholar] [CrossRef]
- Poor, C.; Kanno, N.; Marumoto, T. Evaluation of a Green Roof Retrofit Using Water Treatment Residuals to Reduce Leaching of Phosphorus. J. Green Build. 2022, 17, 129–139. [Google Scholar] [CrossRef]
- Soleimanifar, H.; Deng, Y.; Barrett, K.; Feng, H.; Li, X.; Sarkar, D. Water treatment residual-coated wood mulch for addressing urban stormwater pollution. Water Environ. Res. 2019, 91, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.T.; Ahammed, M.M. The reuse of water treatment sludge as a coagulant for post-treatment of UASB reactor treating urban wastewater. J. Clean. Prod. 2015, 96, 272–281. [Google Scholar] [CrossRef]





| Parameters | Retention Basin Water Samples | New Jersey Surface Water Quality Standards |
|---|---|---|
| Ammonium-N (mg N/L) | BDL 1 | ~1.30 |
| Nitrate-N (mg N/L) | 0.99 ± 0.12 | 2.00 |
| Total Nitrogen (mg N/L) | 1.80 ± 0.30 | N/A |
| Total Phosphorus (mg P/L) | 0.09 ± 0.04 | 0.05 |
| Total Suspended Solids (mg/L) | 0.36 ± 0.13 | 25.00 |
| Average pH | 7.18 ± 0.14 | 4.50–8.50 |
| Granulated Al-WTR: Granular Carbon | Hydraulic Conductivity (m/s) |
|---|---|
| 1 to 0 | 1.37 × 10−3 |
| 1 to 1 | 1.20 × 10−5 |
| 5 to 1 | 4.03 × 10−5 |
| 0 to 1 | 6.24 × 10−6 |
| TCLP | As | Ba | Cd | Cr | Pb | Hg | Se | Ag | P |
|---|---|---|---|---|---|---|---|---|---|
| Spent Green Filter Media | 8.22 ± 1.78 | BDL 1 | 0.98 ± 0.07 | 12.92 ± 0.15 | BDL | 34.24 ± 20.33 | BDL | BDL | 0.09 ± 0.01 |
| USEPA Limit | 5000 | 100,000 | 1000 | 5000 | 5000 | 200 | 2000 | 5000 | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Bagchi, S.; Zhang, Z.; Olayiwola, O.; Mandala, B.; Datta, R.; Giri, S.; Lathrop, R.; Sarkar, D. Culvert Retrofit with Green Filter Media for the Removal of Phosphorus from Stormwater Runoff. Materials 2026, 19, 1193. https://doi.org/10.3390/ma19061193
Bagchi S, Zhang Z, Olayiwola O, Mandala B, Datta R, Giri S, Lathrop R, Sarkar D. Culvert Retrofit with Green Filter Media for the Removal of Phosphorus from Stormwater Runoff. Materials. 2026; 19(6):1193. https://doi.org/10.3390/ma19061193
Chicago/Turabian StyleBagchi, Somdipta, Zhiming Zhang, Olayinka Olayiwola, Bharadwaj Mandala, Rupali Datta, Subhasis Giri, Richard Lathrop, and Dibyendu Sarkar. 2026. "Culvert Retrofit with Green Filter Media for the Removal of Phosphorus from Stormwater Runoff" Materials 19, no. 6: 1193. https://doi.org/10.3390/ma19061193
APA StyleBagchi, S., Zhang, Z., Olayiwola, O., Mandala, B., Datta, R., Giri, S., Lathrop, R., & Sarkar, D. (2026). Culvert Retrofit with Green Filter Media for the Removal of Phosphorus from Stormwater Runoff. Materials, 19(6), 1193. https://doi.org/10.3390/ma19061193










